Optimal Laboratory Cultivation Conditions of Limnospira maxima for Large-Scale Production
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Cyanobacteria Strain and Production Medium
2.2. Optical Density and Growth Rate—Biomass
2.3. Biochemical Analysis
2.3.1. Chlorophyll “a” and Total Carotenoids
2.3.2. Phycobiliprotein Measurement
2.3.3. Soluble Proteins Measurement
2.4. Experimental Design and Statistical Analysis
3. Results
3.1. Photosynthetically Active Radiation Measurement
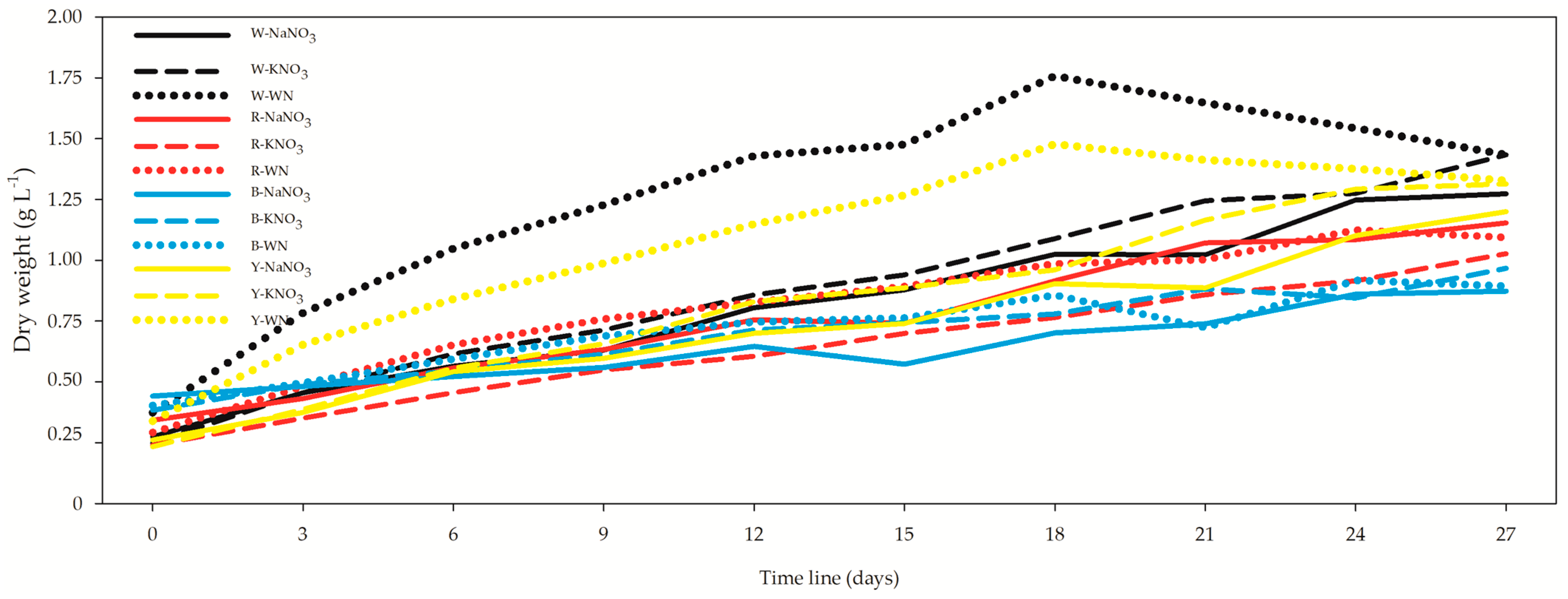
3.2. Optical Density (OD) and Growth Rate (DW)
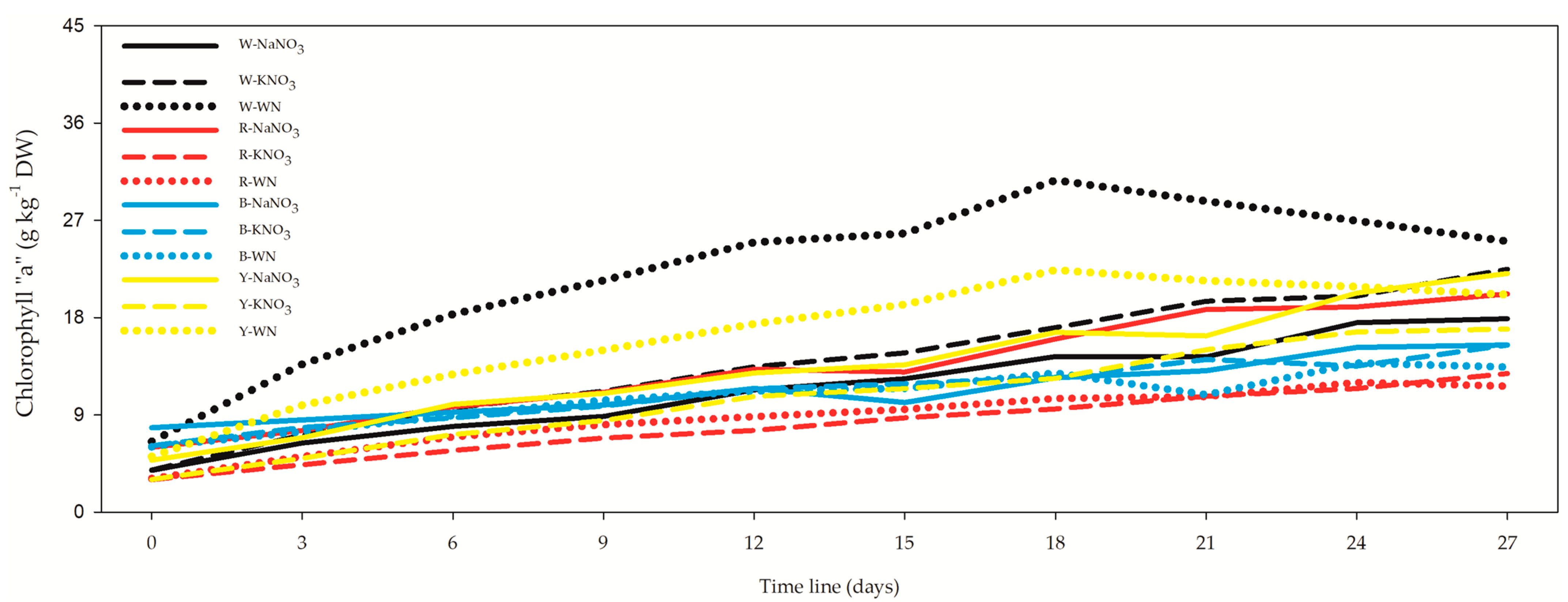
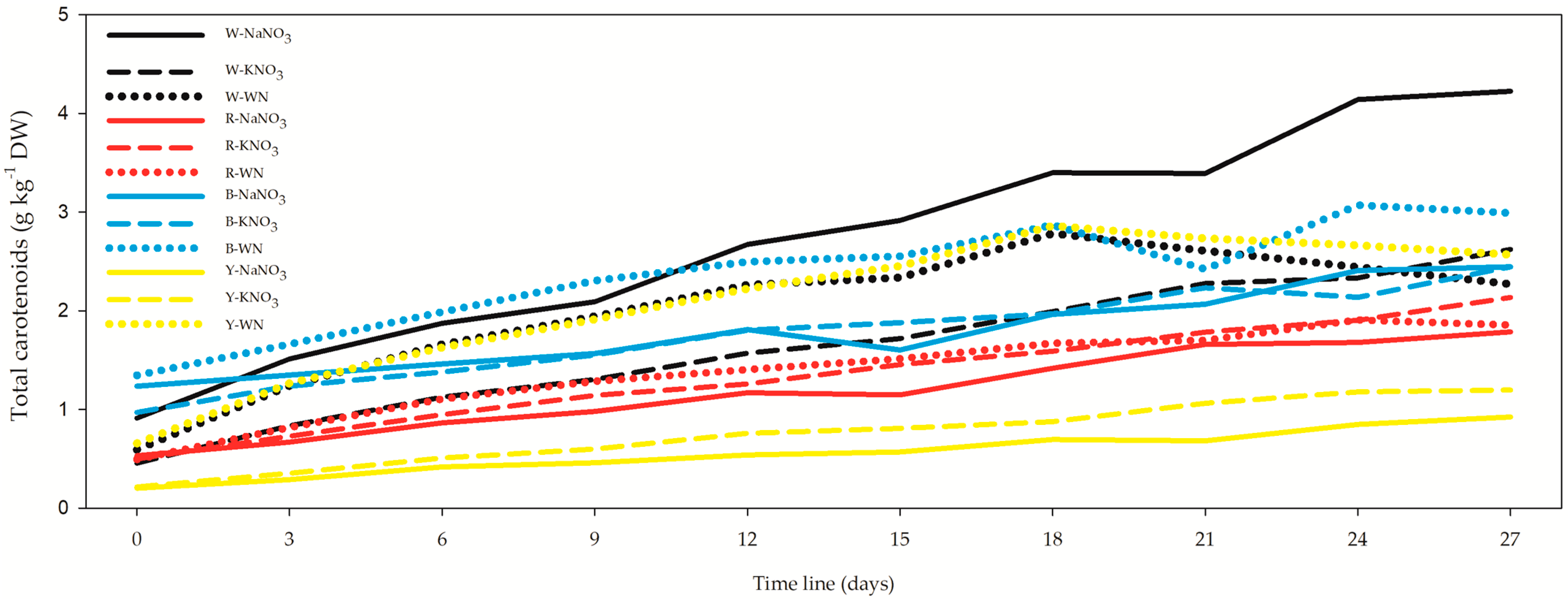
3.3. Chlorophyll “a” (Chla) and Total Carotenoids (Car)
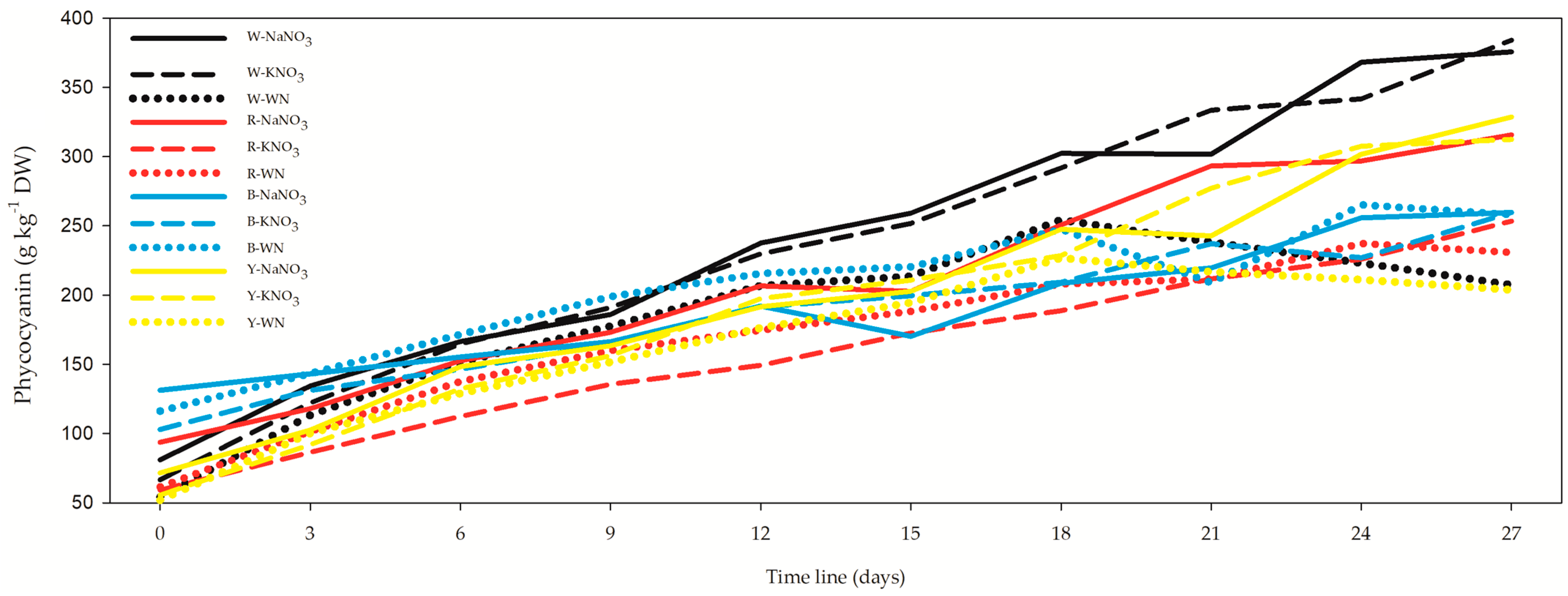
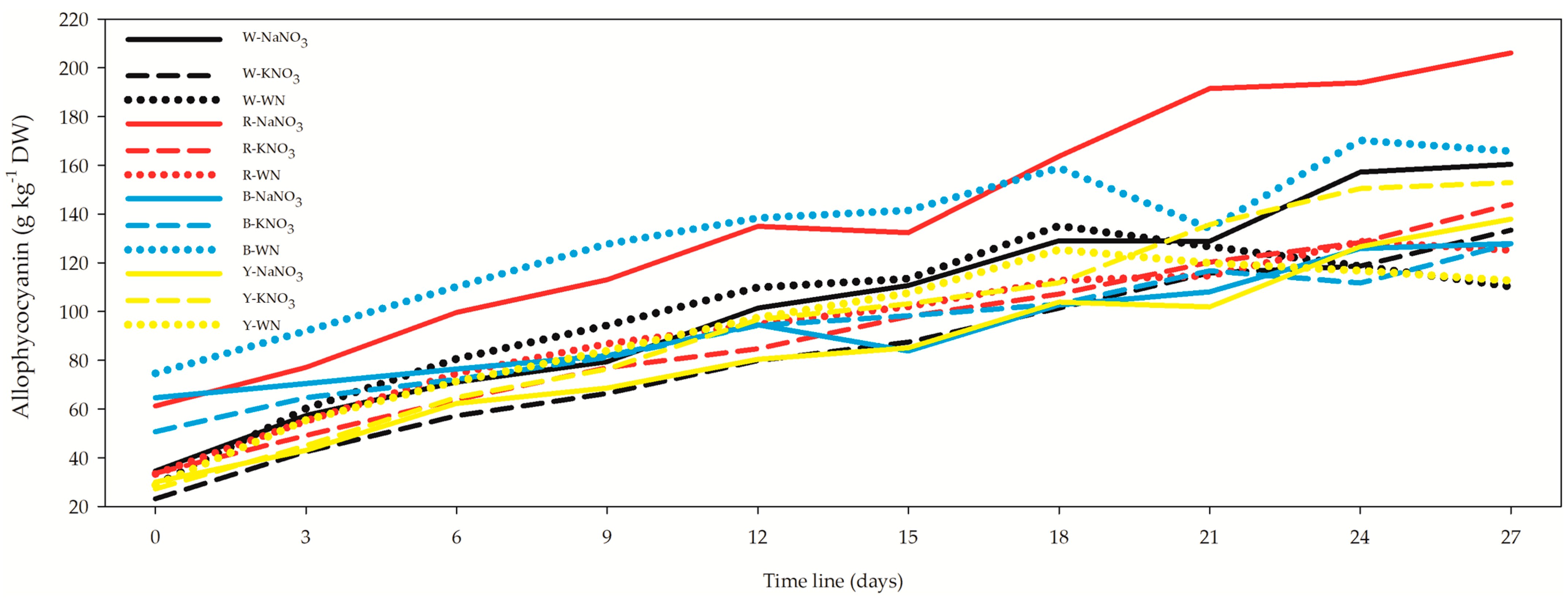
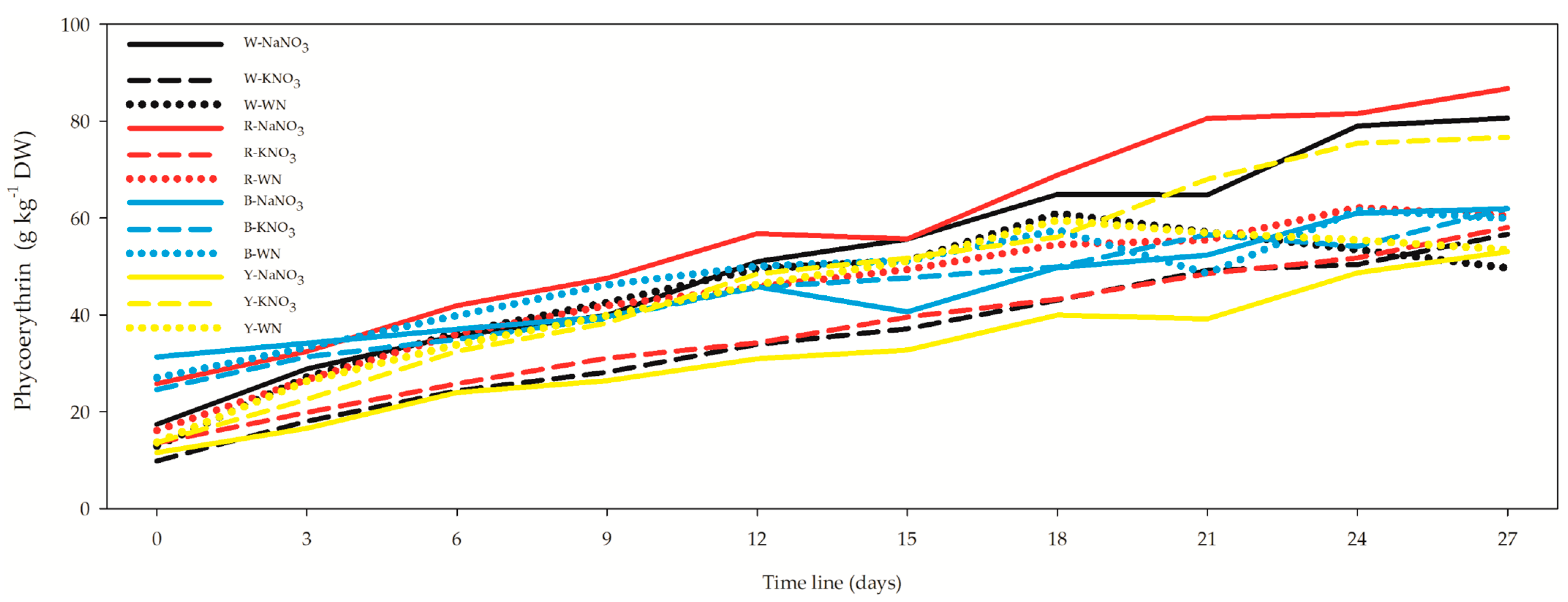
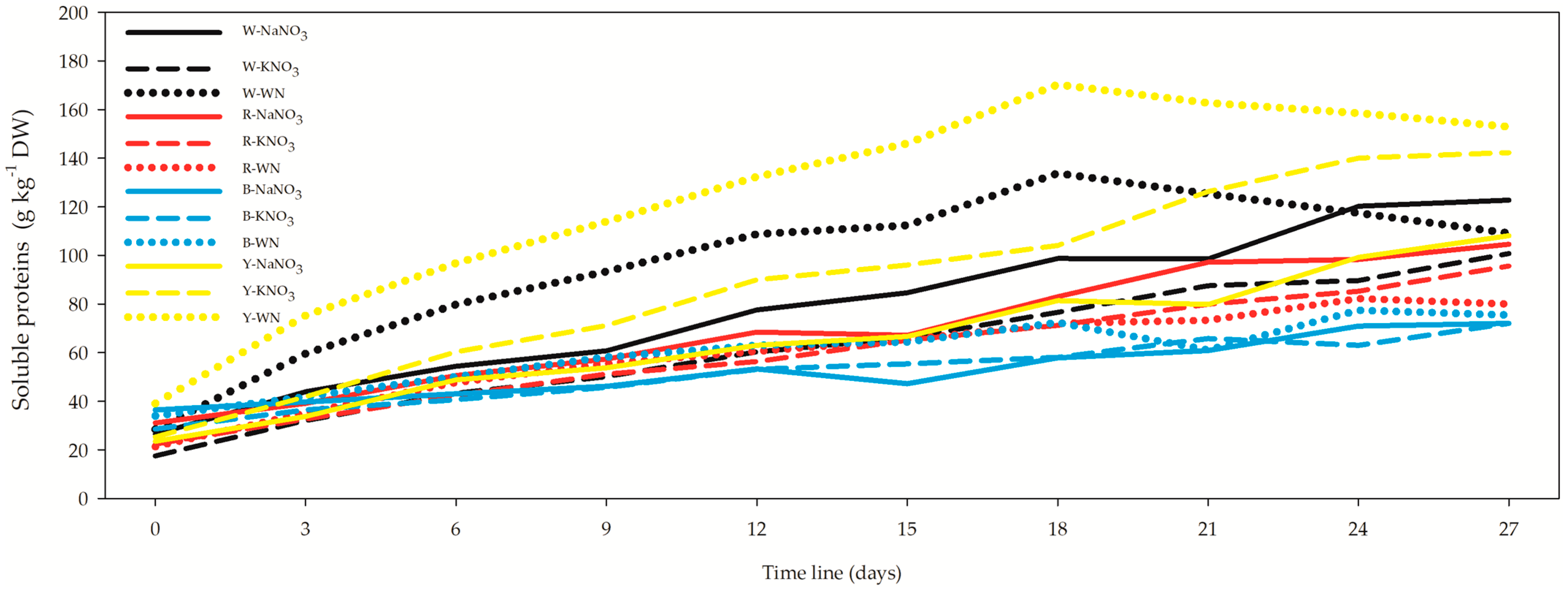
3.4. Phycocyanin, Allophycocyanin, Phycoerythrin, and Soluble Proteins
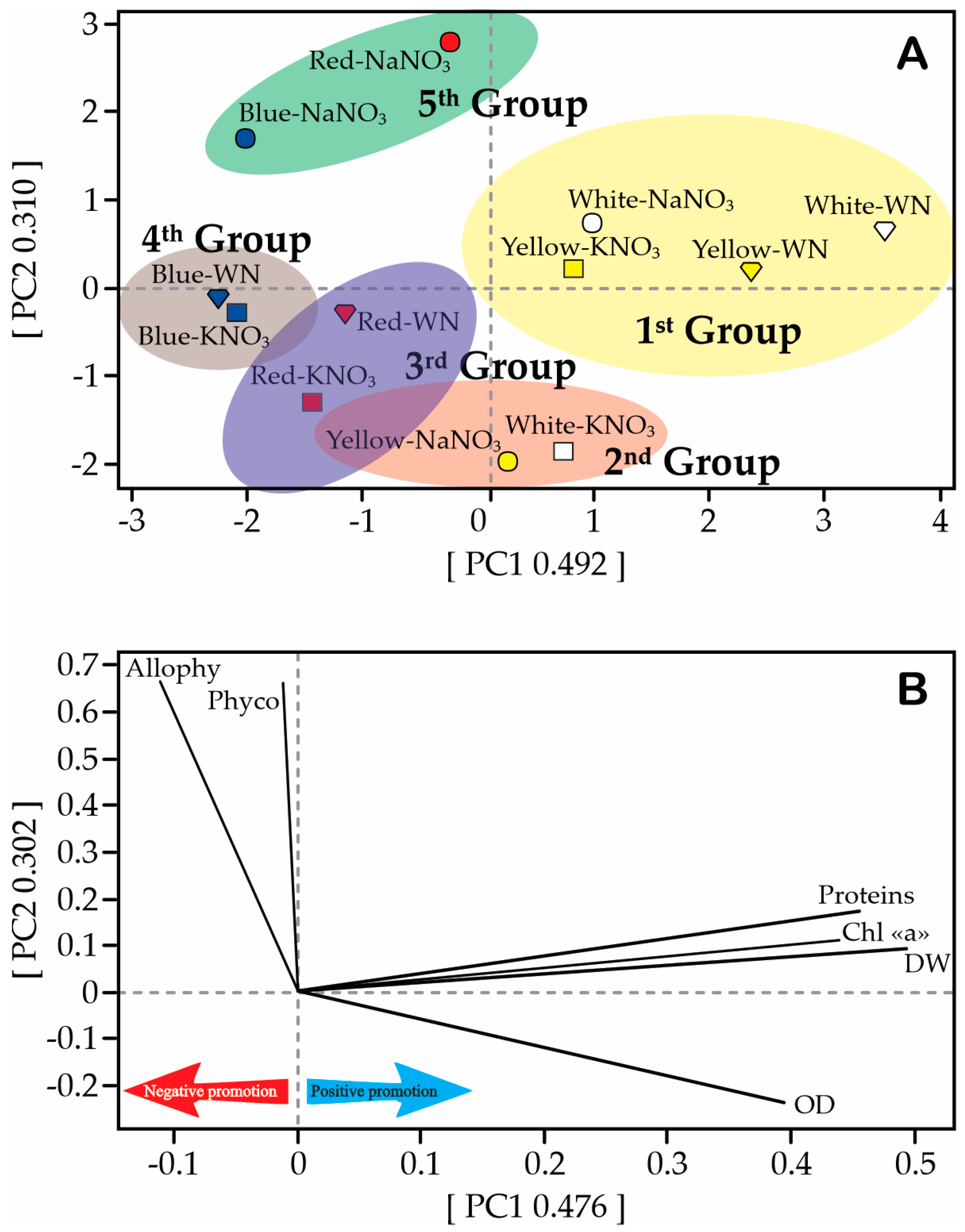
3.5. Principal Component Analysis (PCA)
4. Discussion
| Species | OD | Dry Weight | Chl a | Carotenoids | Phycocyanin | Allophycocyanin | Phycoerythrin | Soluble Proteins | Source | Conditions That Yielded the Highest Values |
|---|---|---|---|---|---|---|---|---|---|---|
| Limnospira maxima | 1.31 | 1.76 g L−1 | 30.70 g kg−1 | 4.22 g kg−1 | 384.11 g k g−1 | 206.12 g kg−1 | 86.68 g kg−1 | 152.86 g kg−1 | This research. | W-KNO3 and Y-KNO3: OD; W-WN: Dry weight and Chla; W-NaNO3: Carotenoids; W-KNO3: phycocyanin; R-NaNO3: allophycocyanin and phycoerythrin; Y-WN: proteins. |
| Arthrospira maxima | - | - | 0.51 g kg−1 | - | 3.20 g kg−1 | 0.19 g kg−1 | 0.97 g kg−1 | - | Park and Dinh [15] | Red and green light: Chl a; blue light: phycobiliproteins. Nitrogen source: NaNO3 |
| Spirulina fussiformis | - | 0.8 g L−1 | 0.0055 g L−1 | - | 2.5 g L−1 | - | - | - | Madhyastha and Vatsala [24] | White light: Dry weight and Chla; green light: phycocyanin; Nitrogen source: NaNO3 |
| Arthrospira maxima | - | 3.8133 g L−1 | 0.065 g L−1 | 0.01 g L−1 | 140 g kg−1 | 30 g kg−1 | - | 735.3 g kg−1 | Milia et al. [28] | Orange light: Dry weight; White and orange lights: Chla; Blue light: proteins, phycocyanin and allophycocyanin. Nitrogen source: NaNO3 |
| Pseudanabaena mucicola | 0.731 | 0.55 g L−1 | 0.0021 g m−2 | - | 0.4 g L−1 | 0.523 g L−1 | 0.05 g L−1 | - | Khatoon et al. [26] | White light and medium supplemented with NaNO3: OD; White light using wastewater: Dry weight, Chla, allophycocyanin and phycoerythrin; Blue light and medium supplemented with NaNO3: phycocyanin. |
| Limnospira fusiformis | 0.3789 g L−1 | 0.00314 g L−1 | 0.00151 g L−1 | 0.01216 g L−1 | - | - | 0.1507 g L−1 | Pelagatti et al. [32] | Yellow light: Chla, Dry weight, proteins and carotenoids; Blue light: phycocyanin. Nitrogen source: NaNO3. | |
| Arthrospira platensis | 1.706 | 1.415 g L−1 | 25.31 g kg−1 | 6.38 g kg−1 | 167.07 g kg−1 | - | - | - | Lima et al. [42] | Lights composed of 70% red and 30% blue (R70:B30): OD, dry weight, Chla and carotenoids; Light deep red (DR): phycocyanin. Nitrogen source: KNO3. |
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Azadi, H.; Ghazali, S.; Ghorbani, M.; Tan, R.; Witlox, F. Contribution of Small-scale Farmers to Global Food Security: A Meta-analysis. J. Sci. Food Agric. 2023, 103, 2715–2726. [Google Scholar] [CrossRef] [PubMed]
- FAO; FIDA; OMS; PMA; UNICEF. El Estado de la Seguridad Alimentaria y la Nutrición en el Mundo 2022; FAO: Rome, Italy; IFAD: Rome, Italy; WHO: Geneva, Switzerland; WFP: Rome, Italy; UNICEF: New Work, NY, USA, 2022; ISBN 978-92-5-136521-2. [Google Scholar]
- Ravindran, B.; Gupta, S.; Cho, W.-M.; Kim, J.; Lee, S.; Jeong, K.-H.; Lee, D.; Choi, H.-C. Microalgae Potential and Multiple Roles—Current Progress and Future Prospects—An Overview. Sustainability 2016, 8, 1215. [Google Scholar] [CrossRef]
- Yin, C.; Daoust, K.; Young, A.; Tebbs, E.; Harper, D. Tackling Community Undernutrition at Lake Bogoria, Kenya: The Potential of Spirulina (Arthrospira Fusiformis) as a Food Supplement. Afr. J. Food Agric. Nutr. Dev. 2017, 17, 11603–11615. [Google Scholar] [CrossRef]
- Soni, R.A.; Sudhakar, K.; Rana, R.S. Spirulina—From Growth to Nutritional Product: A Review. Trends Food Sci. Technol. 2017, 69, 157–171. [Google Scholar] [CrossRef]
- García-López, D.A.; Olguín, E.J.; González-Portela, R.E.; Sánchez-Galván, G.; De Philippis, R.; Lovitt, R.W.; Llewellyn, C.A.; Fuentes-Grünewald, C.; Parra Saldívar, R. A Novel Two-Phase Bioprocess for the Production of Arthrospira (Spirulina) maxima LJGR1 at Pilot Plant Scale during Different Seasons and for Phycocyanin Induction under Controlled Conditions. Bioresour. Technol. 2020, 298, 122548. [Google Scholar] [CrossRef] [PubMed]
- Dolganyuk, V.; Sukhikh, S.; Kalashnikova, O.; Ivanova, S.; Kashirskikh, E.; Prosekov, A.; Michaud, P.; Babich, O. Food Proteins: Potential Resources. Sustainability 2023, 15, 5863. [Google Scholar] [CrossRef]
- Kulshreshtha, A.; Zacharia, A.J.; Jarouliya, U.; Bhadauriya, P.; Prasad, G.; Bisen, P. Spirulina in Health Care Management. Curr. Pharm. Biotechnol. 2008, 9, 400–405. [Google Scholar] [CrossRef]
- Lafarga, T.; Mayre, E.; Echeverria, G.; Viñas, I.; Villaró, S.; Acién-Fernández, F.G.; Castellari, M.; Aguiló-Aguayo, I. Potential of the Microalgae Nannochloropsis and Tetraselmis for Being Used as Innovative Ingredients in Baked Goods. LWT Food Sci. Technol. 2019, 115, 108439. [Google Scholar] [CrossRef]
- Gal, J.L.; Cole, N.R.; Eggett, D.L.; Johnson, S.M. Growth Comparison of Arthrospira Platensis in Different Vessels: Standard Cylinder vs. Enhanced Surface Area at Low Light. Appl. Phycol. 2023, 4, 1–14. [Google Scholar] [CrossRef]
- Vieira Costa, J.A.; Freitas, B.C.B.; Rosa, G.M.; Moraes, L.; Morais, M.G.; Mitchell, B.G. Operational and Economic Aspects of Spirulina-Based Biorefinery. Bioresour. Technol. 2019, 292, 121946. [Google Scholar] [CrossRef]
- Wang, J.; Cheng, W.; Liu, W.; Wang, H.; Zhang, D.; Qiao, Z.; Jin, G.; Liu, T. Field Study on Attached Cultivation of Arthrospira (Spirulina) with Carbon Dioxide as Carbon Source. Bioresour. Technol. 2019, 283, 270–276. [Google Scholar] [CrossRef] [PubMed]
- Mousavi, M.; Mehrzad, J.; Najafi, M.F.; Zhiani, R.; Shamsian, S.A.A. Nitrate and Ammonia: Two Key Nitrogen Sources for Biomass and Phycocyanin Production by Arthrospira (Spirulina) platensis. J. Appl. Phycol. 2022, 34, 2271–2281. [Google Scholar] [CrossRef]
- da Fontoura Prates, D.; Radmann, E.M.; Duarte, J.H.; de Morais, M.G.; Costa, J.A.V. Spirulina Cultivated under Different Light Emitting Diodes: Enhanced Cell Growth and Phycocyanin Production. Bioresour. Technol. 2018, 256, 38–43. [Google Scholar] [CrossRef]
- Park, J.; Dinh, T.B. Contrasting Effects of Monochromatic LED Lighting on Growth, Pigments and Photosynthesis in the Commercially Important Cyanobacterium Arthrospira maxima. Bioresour. Technol. 2019, 291, 121846. [Google Scholar] [CrossRef]
- Garcia, A.K.; McShea, H.; Kolaczkowski, B.; Kaçar, B. Reconstructing the Evolutionary History of Nitrogenases: Evidence for Ancestral Molybdenum-cofactor Utilization. Geobiology 2020, 18, 394–411. [Google Scholar] [CrossRef]
- Bachchhav, M.B.; Kulkarni, M.V.; Ingale, A.G. Enhanced Phycocyanin Production from Spirulina platensis Using Light Emitting Diode. J. Inst. Eng. India Ser. E 2017, 98, 41–45. [Google Scholar] [CrossRef]
- Pineda-Rodriguez, Y.Y.; Pompelli, M.F.; Jarma-Orozco, A.; Rodríguez, N.V.; Rodriguez-Paez, L.A. A New and Profitable Protocol to DNA Extraction in Limnospira maxima. Methods Protoc. 2023, 6, 62. [Google Scholar] [CrossRef]
- Rajasekaran, C.; Ajeesh, C.P.M.; Balaji, S.; Shalini, M.; SIVA, R.; Das, R.; Fulzele, D.P.; Kalaivani, T. Effect of Modified Zarrouk’s Medium on Growth of Different Spirulina Strains. Walailak J. Sci. Technol. WJST 2016, 13, 67–75. [Google Scholar]
- Wellburn, A.R. The Spectral Determination of Chlorophylls a and b, as Well as Total Carotenoids, Using Various Solvents with Spectrophotometers of Different Resolution. J. Plant Physiol. 1994, 144, 307–313. [Google Scholar] [CrossRef]
- Lichtenthaler, H.K.; Buschmann, C. Chlorophylls and Carotenoids: Measurement and Characterization by UV-VIS Spectroscopy. Curr. Protoc. Food Anal. Chem. 2001, 1, F4.3.1–F4.3.8. [Google Scholar] [CrossRef]
- Bennett, A.; Bogorad, L. Complementary Chromatic Adaptation in a Filamentous Blue-Green Alga. J. Cell Biol. 1973, 58, 419–435. [Google Scholar] [CrossRef] [PubMed]
- Tandeau, N.; Houmard, J. Adaptation of Cyanobacteria to Environmental Stimuli: New Steps towards Molecular Mechanisms. FEMS Microbiol. Lett. 1993, 104, 119–189. [Google Scholar] [CrossRef]
- Madhyastha, H.K.; Vatsala, T.M. Pigment Production in Spirulina fussiformis in Different Photophysical Conditions. Biomol. Eng. 2007, 24, 301–305. [Google Scholar] [CrossRef] [PubMed]
- Ortiz-Moreno, M.L.; Cárdenas-Poblador, J.; Agredo, J.; Solarte-Murillo, L.V. Modeling the Effects of Light Wavelength on the Growth of Nostoc ellipsosporum. Univ. Sci. 2020, 25, 113–148. [Google Scholar] [CrossRef]
- Khatoon, H.; Kok Leong, L.; Abdu Rahman, N.; Mian, S.; Begum, H.; Banerjee, S.; Endut, A. Effects of Different Light Source and Media on Growth and Production of Phycobiliprotein from Freshwater Cyanobacteria. Bioresour. Technol. 2018, 249, 652–658. [Google Scholar] [CrossRef]
- Jung, C.H.G.; Waldeck, P.; Sykora, S.; Braune, S.; Petrick, I.; Küpper, J.-H.; Jung, F. Influence of Different Light-Emitting Diode Colors on Growth and Phycobiliprotein Generation of Arthrospira Platensis. Life 2022, 12, 895. [Google Scholar] [CrossRef]
- Milia, M.; Corrias, F.; Addis, P.; Chini Zitelli, G.; Cicchi, B.; Torzillo, G.; Andreotti, V.; Angioni, A. Influence of Different Light Sources on the Biochemical Composition of Arthrospira spp. Grown in Model Systems. Foods 2022, 11, 399. [Google Scholar] [CrossRef]
- Tayebati, H.; Pajoum Shariati, F.; Soltani, N.; Sepasi Tehrani, H. Effect of Various Light Spectra on Amino Acids and Pigment Production of Arthrospira Platensis Using Flat-Plate Photobioreactor. Prep. Biochem. Biotechnol. 2021, 1–12. [Google Scholar] [CrossRef]
- Bahman, M.; Aghanoori, M.; Jalili, H.; Bozorg, A.; Danaee, S.; Bidhendi, M.E.; Amrane, A. Effect of Light Intensity and Wavelength on Nitrogen and Phosphate Removal from Municipal Wastewater by Microalgae under Semi-Batch Cultivation. Environ. Technol. 2022, 43, 1352–1358. [Google Scholar] [CrossRef]
- Mirhosseini, N.; Cano-Europa, E.; Davarnejad, R.; Hallajisani, A.; Franco-Colín, M.; Tavakoli, O.; Blas-Valdivia, V. Cultivations of Arthrospira maxima (Spirulina) Using Ammonium Sulfate and Sodium Nitrate as an Alternative Nitrogen Sources. Iran. J. Fish. Sci. 2021, 20, 475–489. [Google Scholar] [CrossRef]
- Pelagatti, M.; Mori, G.; Falsini, S.; Ballini, R.; Lazzara, L.; Papini, A. Blue and Yellow Light Induce Changes in Biochemical Composition and Ultrastructure of Limnospira fusiformis (Cyanoprokaryota). Microorganisms 2023, 11, 1236. [Google Scholar] [CrossRef]
- De Mooij, T.; De Vries, G.; Latsos, C.; Wijffels, R.H.; Janssen, M. Impact of Light Color on Photobioreactor Productivity. Algal Res. 2016, 15, 32–42. [Google Scholar] [CrossRef]
- Möllers, K.B.; Cannella, D.; Jørgensen, H.; Frigaard, N.-U. Cyanobacterial Biomass as Carbohydrate and Nutrient Feedstock for Bioethanol Production by Yeast Fermentation. Biotechnol. Biofuels 2014, 7, 64. [Google Scholar] [CrossRef]
- Cuellar-Bermudez, S.P.; Struyf, T.; Versluys, M.; Van Den Ende, W.; Wattiez, R.; Muylaert, K. Quantification of Extracellular and Biomass Carbohydrates by Arthrospira under Nitrogen Starvation at Lab-Scale. Algal Res. 2022, 68, 102907. [Google Scholar] [CrossRef]
- Deschoenmaeker, F.; Facchini, R.; Cabrera Pino, J.C.; Bayon-Vicente, G.; Sachdeva, N.; Flammang, P.; Wattiez, R. Nitrogen Depletion in Arthrospira sp. PCC 8005, an Ultrastructural Point of View. J. Struct. Biol. 2016, 196, 385–393. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Yao, C.; Sun, Y.; Chen, W.; Tan, H.; Cao, X.; Xue, S.; Yin, H. Production and Structural Characterization of a New Type of Polysaccharide from Nitrogen-Limited Arthrospira platensis Cultivated in Outdoor Industrial-Scale Open Raceway Ponds. Biotechnol. Biofuels 2019, 12, 131. [Google Scholar] [CrossRef]
- Saxena, R.; Rodríguez-Jasso, R.M.; Chávez-Gonzalez, M.L.; Aguilar, C.N.; Quijano, G.; Ruiz, H.A. Strategy Development for Microalgae Spirulina platensis Biomass Cultivation in a Bubble Photobioreactor to Promote High Carbohydrate Content. Fermentation 2022, 8, 374. [Google Scholar] [CrossRef]
- Sendersky, E.; Kozer, N.; Levi, M.; Moizik, M.; Garini, Y.; Shav-Tal, Y.; Schwarz, R. The Proteolysis Adaptor, NblA, Is Essential for Degradation of the Core Pigment of the Cyanobacterial Light-harvesting Complex. Plant J. 2015, 83, 845–852. [Google Scholar] [CrossRef]
- Deschoenmaeker, F.; Facchini, R.; Leroy, B.; Badri, H.; Zhang, C.-C.; Wattiez, R. Proteomic and Cellular Views of Arthrospira Sp. PCC 8005 Adaptation to Nitrogen Depletion. Microbiology 2014, 160, 1224–1236. [Google Scholar] [CrossRef]
- Wegener, K.M.; Singh, A.K.; Jacobs, J.M.; Elvitigala, T.; Welsh, E.A.; Keren, N.; Gritsenko, M.A.; Ghosh, B.K.; Camp, D.G.; Smith, R.D.; et al. Global Proteomics Reveal an Atypical Strategy for Carbon/Nitrogen Assimilation by a Cyanobacterium under Diverse Environmental Perturbations. Mol. Cell. Proteom. 2010, 9, 2678–2689. [Google Scholar] [CrossRef]
- Lima, G.M.; Teixeira, P.C.N.; Teixeira, C.M.L.L.; Filócomo, D.; Lage, C.L.S. Influence of Spectral Light Quality on the Pigment Concentrations and Biomass Productivity of Arthrospira platensis. Algal Res. 2018, 31, 157–166. [Google Scholar] [CrossRef]
- Zittelli, G.; Mugnai, G.; Milia, M.; Cicchi, B.; Silva, A.M.; Angioni, A.; Addis, P.; Torzillo, G. Effects of Blue, Orange and White Lights on Growth, Chlorophyll Fluorescence, and Phycocyanin Production of Arthrospira platensis Cultures. Algal Res. 2022, 61, 102583. [Google Scholar] [CrossRef]
- Lee, S.-H.; Lee, J.E.; Kim, Y.; Lee, S.-Y. The Production of High Purity Phycocyanin by Spirulina platensis Using Light-Emitting Diodes Based Two-Stage Cultivation. Appl. Biochem. Biotechnol. 2016, 178, 382–395. [Google Scholar] [CrossRef] [PubMed]
- Ojit, S.K.; Indrama, T.; Gunapati, O.; Avijeet, S.O.; Subhalaxmi, S.A.; Silvia, C.; Indira, D.W.; Romi, K.; Minerva, S.; Thadoi, D.A.; et al. The Response of Phycobiliproteins to Light Qualities in Anabaena Circinalis. J. Appl. Biol. Biotechnol. 2015, 3, 1–6. [Google Scholar] [CrossRef]
- Joseph, A.; Aikawa, S.; Sasaki, K.; Matsuda, F.; Hasunuma, T.; Kondo, A. Increased Biomass Production and Glycogen Accumulation in apcE Gene Deleted Synechocystis Sp. PCC 6803. AMB Express 2014, 4, 17. [Google Scholar] [CrossRef] [PubMed]
- Thevarajah, B.; Nishshanka, G.K.S.H.; Premaratne, M.; Nimarshana, P.H.V.; Nagarajan, D.; Chang, J.-S.; Ariyadasa, T.U. Large-Scale Production of Spirulina-Based Proteins and c-Phycocyanin: A Biorefinery Approach. Biochem. Eng. J. 2022, 185, 108541. [Google Scholar] [CrossRef]

| Light Source | Model | Trademark | Color | Power | PAR (μmol m−2 s−1) | |
|---|---|---|---|---|---|---|
| Theoretical | Measured 1 | |||||
| LED T5 | M22 | Mercury, Kwangchow, China | Write—W | 18 W | 82.8 | 79.14 ± 1.94 |
| LED T5 | M29 | Mercury, Kwangchow, China | Red—R | 18 W | 82.8 | 77.51 ± 2.23 |
| LED T5 | M21 | Mercury, Kwangchow, China | Blue—B | 18 W | 82.8 | 78.10 ± 1.81 |
| LED T5 | M24 | Mercury, Kwangchow, China | Yellow—Y | 18 W | 82.8 | 79.31 ± 1.85 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pineda-Rodríguez, Y.Y.; Herazo-Cárdenas, D.S.; Vallejo-Isaza, A.; Pompelli, M.F.; Jarma-Orozco, A.; Jaraba-Navas, J.d.D.; Cordero-Ocampo, J.D.; González-Berrio, M.; Arrieta, D.V.; Pico-González, A.; et al. Optimal Laboratory Cultivation Conditions of Limnospira maxima for Large-Scale Production. Biology 2023, 12, 1462. https://doi.org/10.3390/biology12121462
Pineda-Rodríguez YY, Herazo-Cárdenas DS, Vallejo-Isaza A, Pompelli MF, Jarma-Orozco A, Jaraba-Navas JdD, Cordero-Ocampo JD, González-Berrio M, Arrieta DV, Pico-González A, et al. Optimal Laboratory Cultivation Conditions of Limnospira maxima for Large-Scale Production. Biology. 2023; 12(12):1462. https://doi.org/10.3390/biology12121462
Chicago/Turabian StylePineda-Rodríguez, Yirlis Yadeth, Diana Sofia Herazo-Cárdenas, Adriana Vallejo-Isaza, Marcelo F. Pompelli, Alfredo Jarma-Orozco, Juan de Dios Jaraba-Navas, Jhony David Cordero-Ocampo, Marianella González-Berrio, Daniela Vegliante Arrieta, Ana Pico-González, and et al. 2023. "Optimal Laboratory Cultivation Conditions of Limnospira maxima for Large-Scale Production" Biology 12, no. 12: 1462. https://doi.org/10.3390/biology12121462
APA StylePineda-Rodríguez, Y. Y., Herazo-Cárdenas, D. S., Vallejo-Isaza, A., Pompelli, M. F., Jarma-Orozco, A., Jaraba-Navas, J. d. D., Cordero-Ocampo, J. D., González-Berrio, M., Arrieta, D. V., Pico-González, A., Ariza-González, A., Aviña-Padilla, K., & Rodríguez-Páez, L. A. (2023). Optimal Laboratory Cultivation Conditions of Limnospira maxima for Large-Scale Production. Biology, 12(12), 1462. https://doi.org/10.3390/biology12121462









