Spatiotemporal Epidemiology of Tuberculosis in Thailand from 2011 to 2020
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area and Data Sources
2.2. Rate Standardization
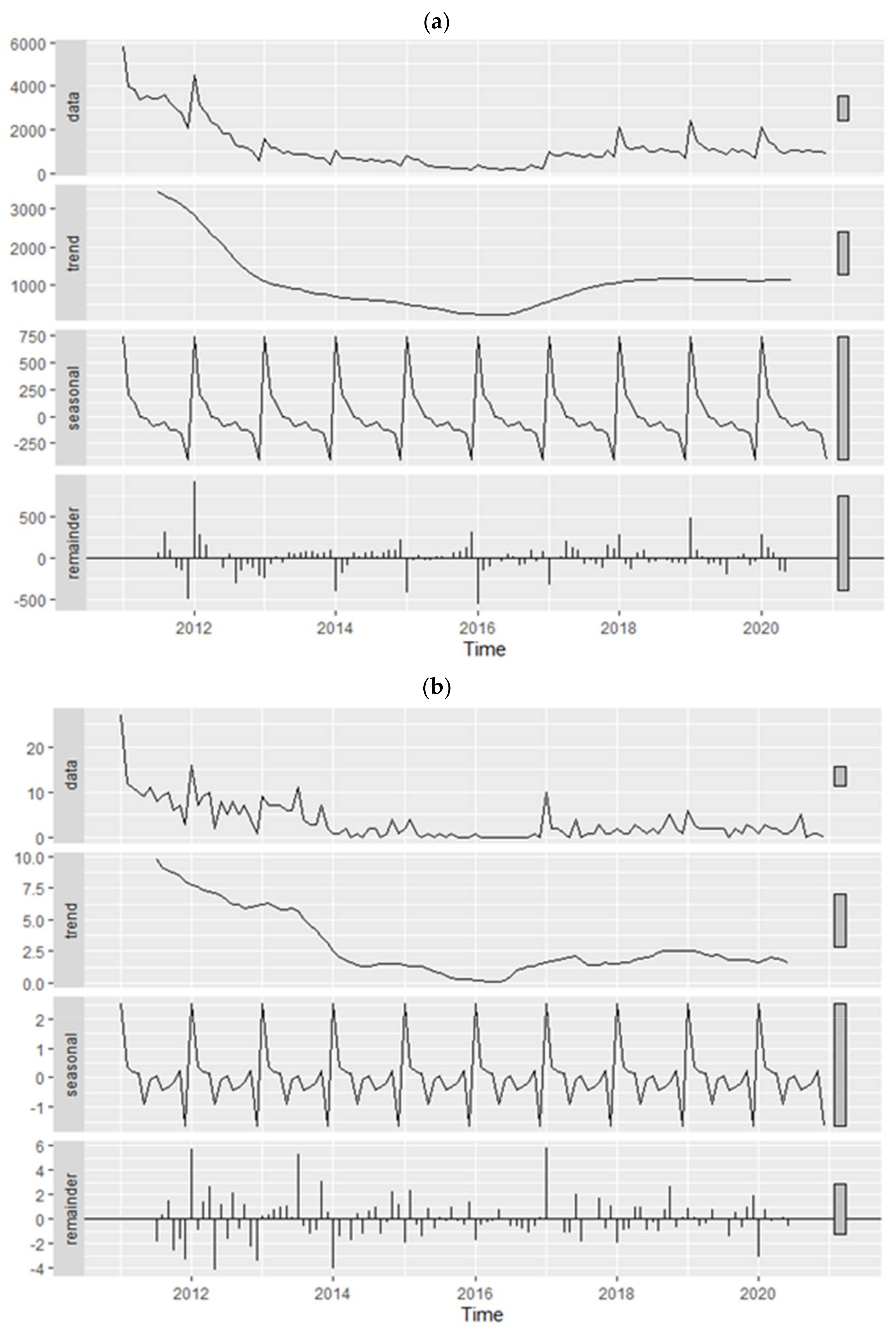
2.3. Descriptive and Temporal Analyses
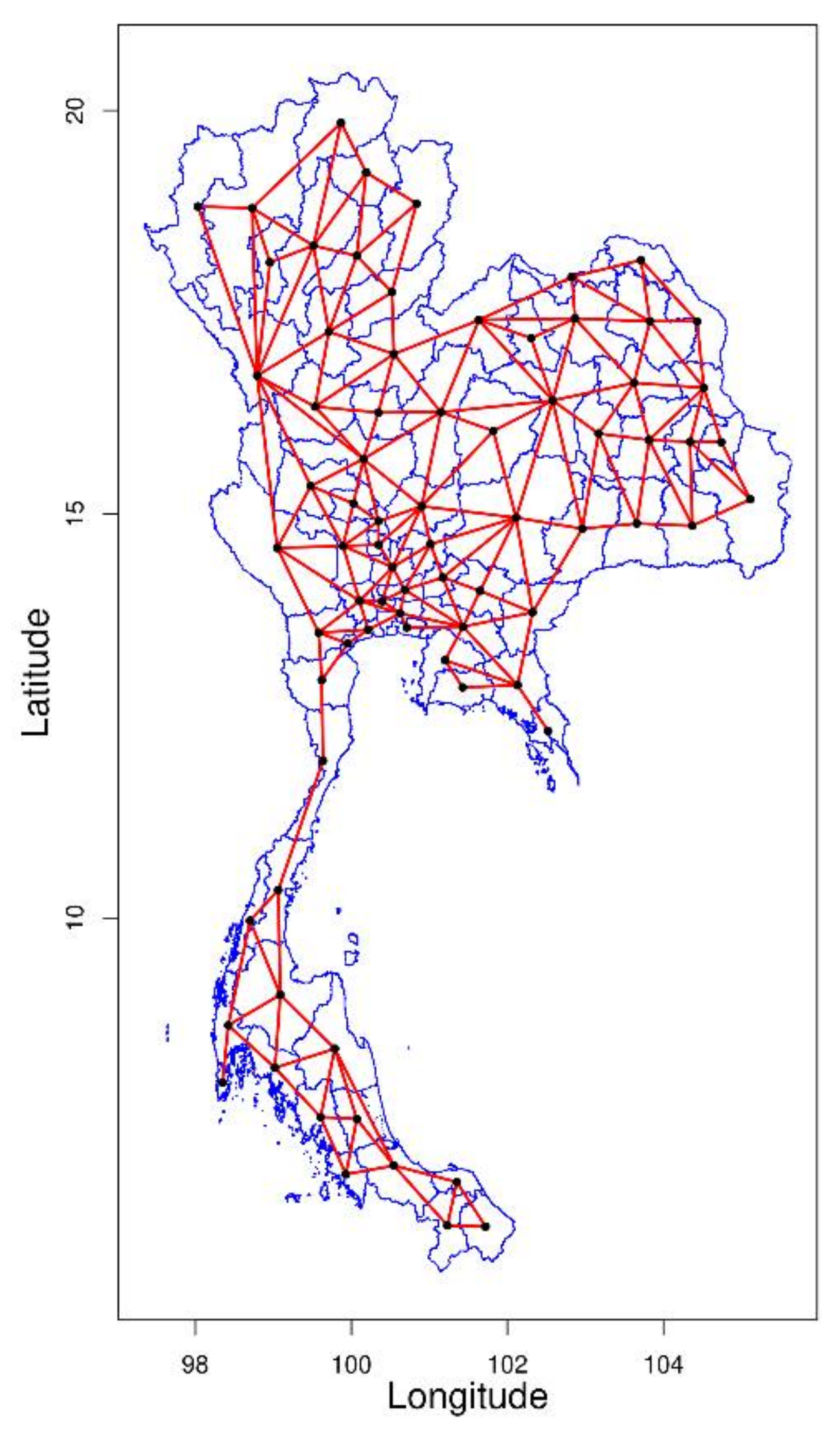
2.4. Spatial Pattern Analysis
2.4.1. Spatial Contiguity Matrices
2.4.2. Global Spatial Detection
2.4.3. Local Spatial Detection
3. Results
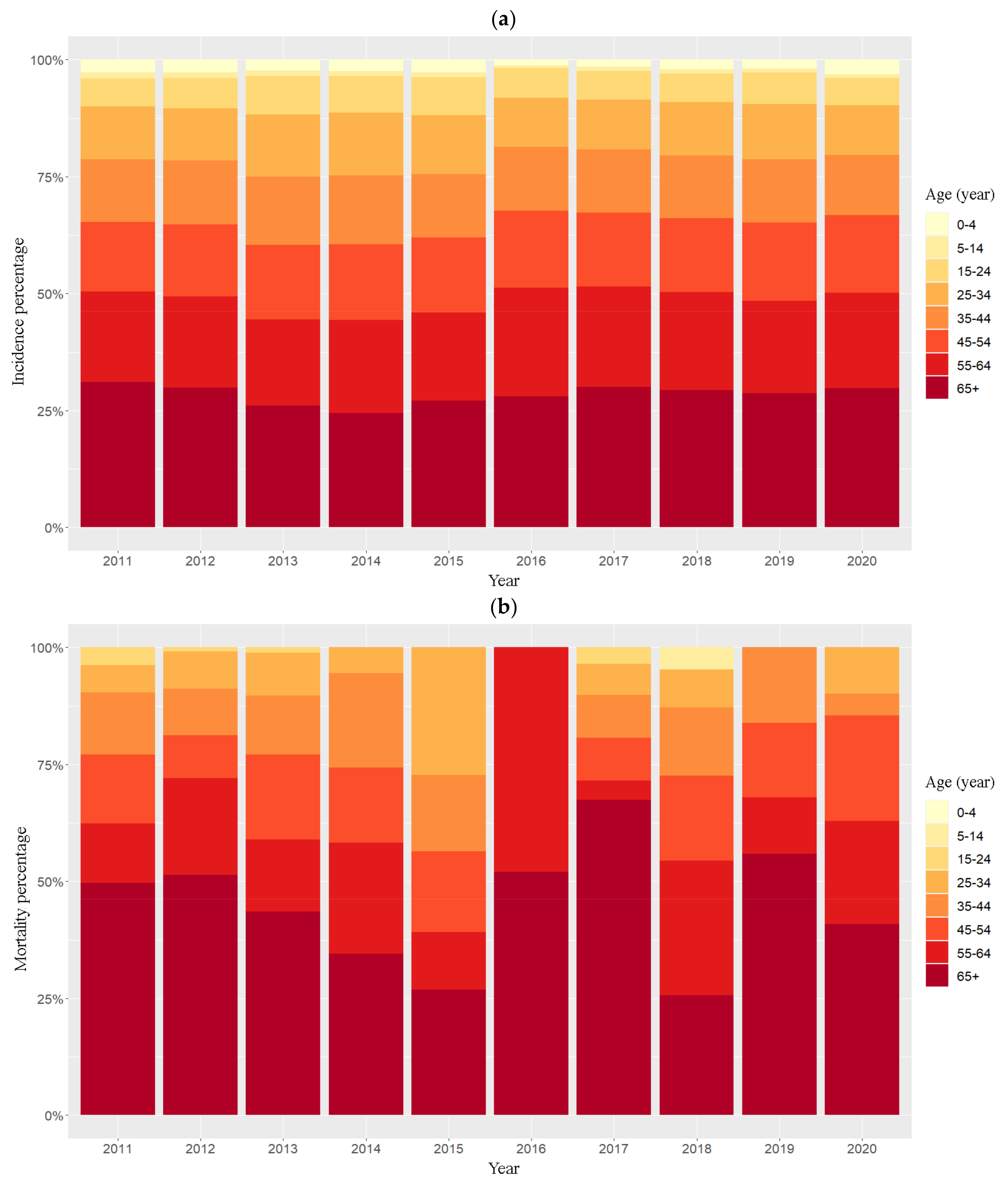
3.1. Descriptive and Temporal Analyses
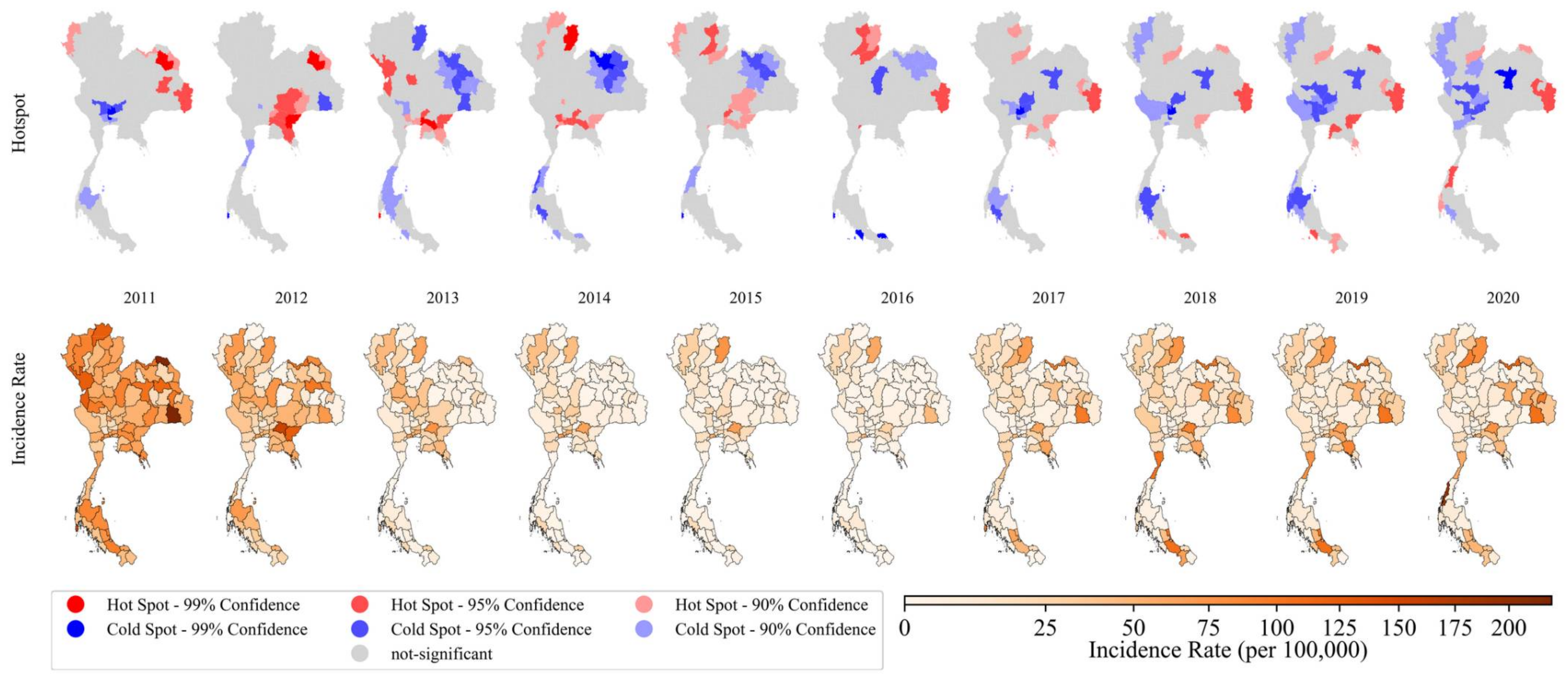
3.2. Spatial Pattern Analysis
3.2.1. Global Spatial Detection
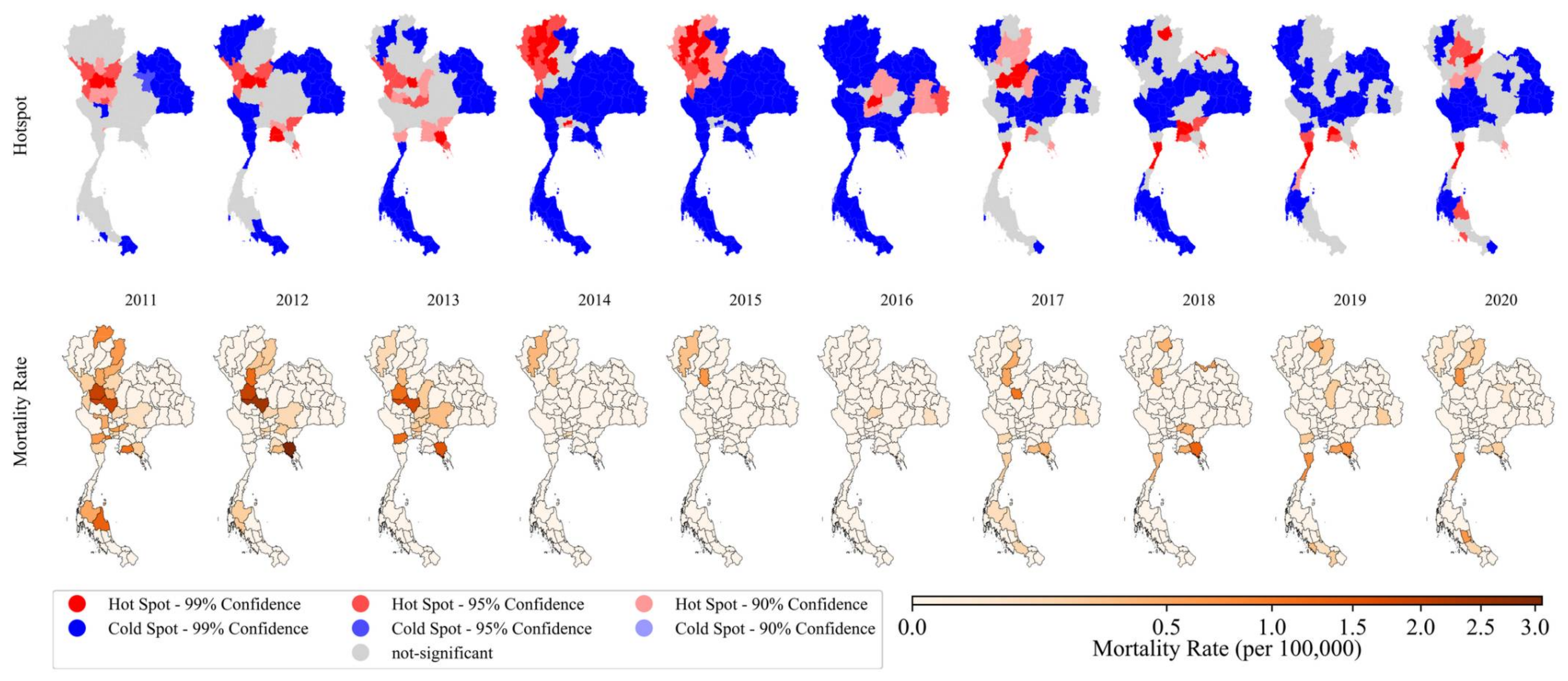
3.2.2. Local Spatial Detection
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chakaya, J.; Khan, M.; Ntoumi, F.; Aklillu, E.; Fatima, R.; Mwaba, P.; Kapata, N.; Mfinanga, S.; Hasnain, S.E.; Katoto, P.D.M.C.; et al. Global Tuberculosis Report 2020—Reflections on the Global TB burden, treatment and prevention efforts. Int. J. Infect. Dis. 2021, 113, S7–S12. [Google Scholar] [CrossRef] [PubMed]
- Holt, E. Tuberculosis on the political agenda. Lancet Infect. Dis. 2018, 18, 30. [Google Scholar] [CrossRef]
- Getnet, F.; Demissie, M.; Assefa, N.; Mengistie, B.; Worku, A. Delay in diagnosis of pulmonary tuberculosis in low-and middle-income settings: Systematic review and meta-analysis. BMC Pulm. Med. 2017, 17, 202. [Google Scholar] [CrossRef] [PubMed]
- Turner, R.D.; Bothamley, G.H. Cough and the transmission of tuberculosis. J. Infect. Dis. 2015, 211, 1367–1372. [Google Scholar] [CrossRef] [PubMed]
- Tanimura, T.; Jaramillo, E.; Weil, D.; Raviglione, M.; Lönnroth, K. Financial burden for tuberculosis patients in low-and middle-income countries: A systematic review. Eur. Respir. J. 2014, 43, 1763–1775. [Google Scholar] [CrossRef] [PubMed]
- Dowdy, D.W.; Golub, J.E.; Chaisson, R.E.; Saraceni, V. Heterogeneity in tuberculosis transmission and the role of geographic hotspots in propagating epidemics. Proc. Natl. Acad. Sci. USA 2012, 109, 9557–9562. [Google Scholar] [CrossRef] [PubMed]
- Dangisso, M.H.; Datiko, D.G.; Lindtjørn, B. Spatio-temporal analysis of smear-positive tuberculosis in the Sidama Zone, southern Ethiopia. PLoS ONE 2015, 10, e0126369. [Google Scholar] [CrossRef]
- Xu, B.; Li, J.; Wang, M. Epidemiological and time series analysis on the incidence and death of AIDS and HIV in China. BMC Public Health 2020, 20, 1906. [Google Scholar] [CrossRef]
- Chen, Y.; Leng, K.; Lu, Y.; Wen, L.; Qi, Y.; Gao, W.; Chen, H.; Bai, L.; An, X.; Sun, B.; et al. Epidemiological features and time-series analysis of influenza incidence in urban and rural areas of Shenyang, China, 2010–2018. Epidemiol. Infect. 2020, 148, e29. [Google Scholar] [CrossRef]
- Hassarangsee, S.; Tripathi, N.K.; Souris, M. Spatial Pattern Detection of Tuberculosis: A Case Study of Si Sa Ket Province, Thailand. Int. J. Environ. Res. Public Health 2015, 12, 16005–16018. [Google Scholar] [CrossRef]
- Gwitira, I.; Karumazondo, N.; Shekede, M.D.; Sandy, C.; Siziba, N.; Chirenda, J. Spatial patterns of pulmonary tuberculosis (TB) cases in Zimbabwe from 2015 to 2018. PLoS ONE 2021, 16, e0249523. [Google Scholar] [CrossRef]
- Yu, Y.; Wu, B.; Wu, C.; Wang, Q.; Hu, D.; Chen, W. Spatial-temporal analysis of tuberculosis in Chongqing, China 2011–2018. BMC Infect. Dis. 2020, 20, 531. [Google Scholar] [CrossRef]
- Jittimanee, S.; Vorasingha, J.; Mad-asin, W.; Nateniyom, S.; Rienthong, S.; Varma, J.K. Tuberculosis in Thailand: Epidemiology and program performance, 2001–2005. Int. J. Infect. Dis. 2009, 13, 436–442. [Google Scholar] [CrossRef][Green Version]
- Tipayamongkholgul, M.; Podang, J.; Siri, S. Spatial analysis of social determinants for tuberculosis in Thailand. J. Med. Assoc. Thail. Chotmaihet Thangphaet 2013, 96, S116–S121. [Google Scholar]
- Lai, S.; Wangteeraprasert, T.; Sermkaew, T.; Nararak, O.; Binsard, S.; Phanawadee, M.; Ieowongjaroen, I.; Doung-ngern, P.; Tang, X.; Sayavong, C. Evaluation of three main tuberculosis case reporting systems in Satun Province, Thailand, 2011. OSIR J. 2016, 7, 16–23. [Google Scholar]
- Alikhan, M.F.; Swaddiwudhipong, W.; Xu, F.; Nugroho, D.K.; Oo, L.H.; Darnal, J.B.; Din, I.A.B.M.; Aung, Y.N.; Htike, N.W.; Thet, B.S. Evaluation of the National Tuberculosis Database System,“Tuberculosis Case Management (TBCM)”, for its Surveillance Function at Mae Sot Hospital, Thailand. OSIR J. 2021, 14, 20–26. [Google Scholar]
- Rothman, K.J.; Greenland, S.; Lash, T.L. Modern Epidemiology; Wolters Kluwer Health/Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2008; Volume 3. [Google Scholar]
- Lawson, A.B. Bayesian Disease Mapping: Hierarchical Modeling in Spatial Epidemiology; Chapman and Hall/CRC: Boca Raton, FL, USA, 2018. [Google Scholar]
- Lawson, A.B.; Rotejanaprasert, C. Childhood brain cancer in Florida: A Bayesian clustering approach. Stat. Public Policy 2014, 1, 99–107. [Google Scholar] [CrossRef]
- Rotejanaprasert, C.; Lawpoolsri, S.; Pan-Ngum, W.; Maude, R.J. Preliminary estimation of temporal and spatiotemporal dynamic measures of COVID-19 transmission in Thailand. PLoS ONE 2020, 15, e0239645. [Google Scholar] [CrossRef]
- Nakaya, T. An Information Statistical Approach to the Modifiable Areal Unit Problem in Incidence Rate Maps. Environ. Plan. A Econ. Space 2000, 32, 91–109. [Google Scholar] [CrossRef]
- Cleveland, R.B.; Cleveland, W.S.; McRae, J.E.; Terpenning, I. STL: A seasonal-trend decomposition. J. Off. Stat. 1990, 6, 3–73. [Google Scholar]
- Dagum, E.B.; Bianconcini, S. Seasonal Adjustment Methods and Real Time Trend-Cycle Estimation; Springer: Berlin/Heidelberg, Germany, 2016. [Google Scholar]
- Nopiah, Z.M.; Baharin, M.N.; Abdullah, S.; Khairir, M.I. Time Series Behaviour of Lower Arm Suspension Fatigue Data Using Classical Decomposition Method. In Proceedings of the 2009 International Conference on Signal Processing Systems, Singapore, 15–17 May 2009; pp. 984–988. [Google Scholar]
- Jordahl, K.; Bossche, J.V.; Fleischmann, M.; McBride, J.; Wasserman, J.; Gerard, J.; Badaracco, A.G.; Snow, A.D.; Tratner, J.; Perry, M.; et al. geopandas/geopandas: v0.8.1 (Version v0.8.1); Zenodo. Available online: https://zenodo.org/record/3946761/export/xd#.Yn365R1BxPY (accessed on 25 March 2022).
- Rey, S.J.; Anselin, L. PySAL: A Python Library of Spatial Analytical Methods. In Handbook of Applied Spatial Analysis: Software Tools, Methods and Applications; Fischer, M.M., Getis, A., Eds.; Springer: Berlin/Heidelberg, Germany, 2010; pp. 175–193. [Google Scholar] [CrossRef]
- Kulldorff, M. Tests of Spatial Randomness Adjusted for an Inhomogeneity. J. Am. Stat. Assoc. 2006, 101, 1289–1305. [Google Scholar] [CrossRef]
- Songchitruksa, P.; Zeng, X. Getis–Ord Spatial Statistics to Identify Hot Spots by Using Incident Management Data. Transp. Res. Rec. 2010, 2165, 42–51. [Google Scholar] [CrossRef]
- Zhang, K.; Zhang, S. Testing simulated positive spatial autocorrelation by Getis-Ord general G. In Proceedings of the 2015 23rd International Conference on Geoinformatics, Wuhan, China, 19–21 June 2015. [Google Scholar]
- Getis, A.; Ord, J.K. The Analysis of Spatial Association by Use of Distance Statistics. In Perspectives on Spatial Data Analysis; Anselin, L., Rey, S.J., Eds.; Springer: Berlin/Heidelberg, Germany, 2010; pp. 127–145. [Google Scholar] [CrossRef]
- Pfeiffer, D.U.; Robinson, T.P.; Stevenson, M.; Stevens, K.B.; Rogers, D.J.; Clements, A.C. Spatial Analysis in Epidemiology; Oxford University Press: Oxford, UK, 2008. [Google Scholar]
- Ord, J.K.; Getis, A. Local Spatial Autocorrelation Statistics: Distributional Issues and an Application. Geogr. Anal. 1995, 27, 286–306. [Google Scholar] [CrossRef]
- Hunter, J.D. Matplotlib: A 2D graphics environment. Comput. Sci. Eng. 2007, 9, 90–95. [Google Scholar] [CrossRef]
- Wingfield, T.; Schumacher, S.G.; Sandhu, G.; Tovar, M.A.; Zevallos, K.; Baldwin, M.R.; Montoya, R.; Ramos, E.S.; Jongkaewwattana, C.; Lewis, J.J. The seasonality of tuberculosis, sunlight, vitamin D, and household crowding. J. Infect. Dis. 2014, 210, 774–783. [Google Scholar] [CrossRef]
- Li, X.-X.; Wang, L.-X.; Zhang, H.; Du, X.; Jiang, S.-W.; Shen, T.; Zhang, Y.-P.; Zeng, G. Seasonal Variations in Notification of Active Tuberculosis Cases in China, 2005–2012. PLoS ONE 2013, 8, e68102. [Google Scholar] [CrossRef]
- Fares, A. Seasonality of tuberculosis. J. Glob. Infect. Dis. 2011, 3, 46–55. [Google Scholar] [CrossRef]
- Yang, X.; Duan, Q.; Wang, J.; Zhang, Z.; Jiang, G. Seasonal Variation of Newly Notified Pulmonary Tuberculosis Cases from 2004 to 2013 in Wuhan, China. PLoS ONE 2014, 9, e108369. [Google Scholar] [CrossRef]
- Nikam, J.; Archer, D.; Nopsert, C. Air Quality in Thailand: Understanding the Regulatory Context; SEI Working Paper; Stockholm Environment Institute: Stockholm, Sweden, 2021. [Google Scholar]
- Tuberculosis, D.O. Thailand Operational Plan to End Tuberculosis 2017–2021; Department of Disease Control, Thailand, Ed.; 2017. Available online: https://www.tbthailand.org/download/Manual/Thailand%20Operational%20Plan%20To%20End%20%20TB_2017_2021.pdf (accessed on 25 March 2022).
- World Health Organization. Fifth Joint Monitoring Mission of the Bangladesh National Tuberculosis Control Programme; WHO Regional Office for South-East Asia: New Delhi, India, 2011.
- Ben Ayed, H.; Koubaa, M.; Marrakchi, C.; Rekik, K.; Hammami, F.; Smaoui, F. Extrapulmonary tuberculosis: Update on the epidemiology, risk factors and prevention strategies. Int. J. Trop. Dis. 2018, 1. [Google Scholar] [CrossRef]
- Pang, Y.; An, J.; Shu, W.; Huo, F.; Chu, N.; Gao, M.; Qin, S.; Huang, H.; Chen, X.; Xu, S. Epidemiology of extrapulmonary tuberculosis among inpatients, China, 2008–2017. Emerg. Infect. Dis. 2019, 25, 457. [Google Scholar] [CrossRef]
- Dodd, P.J.; Sismanidis, C.; Glaziou, P. Methods for estimating tuberculosis incidence and mortality by age and sex. Int. J. Epidemiol. 2021, 50, 570–577. [Google Scholar] [CrossRef]
- Ncube, R.; Takarinda, K.; Zishiri, C.; Van den Boogaard, W.; Mlilo, N.; Chiteve, C.; Siziba, N.; Trinchán, F.; Sandy, C. Age-stratified tuberculosis treatment outcomes in Zimbabwe: Are we paying attention to the most vulnerable? Public Health Action 2017, 7, 212–217. [Google Scholar] [CrossRef]
- Oshi, D.C.; Oshi, S.N.; Alobu, I.; Ukwaja, K.N. Profile and treatment outcomes of tuberculosis in the elderly in southeastern Nigeria, 2011–2012. PLoS ONE 2014, 9, e111910. [Google Scholar] [CrossRef]
- Gavazzi, G.; Herrmann, F.; Krause, K.-H. Aging and infectious diseases in the developing world. Clin. Infect. Dis. 2004, 39, 83–91. [Google Scholar] [CrossRef]
- World Health, O. Global Tuberculosis Report 2020; World Health Organization: Geneva, Switzerland, 2020.
- Tschirhart, N.; Nosten, F.; Foster, A.M. Access to free or low-cost tuberculosis treatment for migrants and refugees along the Thailand-Myanmar border in Tak province, Thailand. Int. J. Equity Health 2016, 15, 100. [Google Scholar] [CrossRef]
- Chuengsatiansup, K.; Limsawart, W. Tuberculosis in the borderlands: Migrants, microbes and more-than-human borders. Palgrave Commun. 2019, 5, 31. [Google Scholar] [CrossRef]
- Tschirhart, N.; Sein, T.; Nosten, F.; Foster, A.M. Migrant and refugee patient perspectives on travel and tuberculosis along the Thailand-Myanmar border: A qualitative study. PLoS ONE 2016, 11, e0160222. [Google Scholar] [CrossRef]
- Wongkongdech, R.; Srisaenpang, S.; Tungsawat, S. Pulmonary TB among myanmar migrants in samut sakhon province, thailand: A problem or not for the TB control program? Southeast Asian J. Trop. Med. Public Health 2015, 46, 296. [Google Scholar]
- Woldeyohannes, S.; Abera, S. Worldwide spatial and temporal distribution of tuberculosis (TB). J. AIDS Clin. Res. 2015, 6, 2. [Google Scholar] [CrossRef]
- Kulldorff, M. A spatial scan statistic. Commun. Stat.-Theory Methods 1997, 26, 1481–1496. [Google Scholar] [CrossRef]
| Year | Pulmonary | Meningitis | Other Organs | Overall | ||||
|---|---|---|---|---|---|---|---|---|
| Incidence | Mortality | Incidence | Mortality | Incidence | Mortality | Incidence | Mortality | |
| 2011 | 29,825 | 103 | 829 | 6 | 11,384 | 14 | 42,038 | 123 |
| 2012 | 16,403 | 75 | 442 | 3 | 7050 | 4 | 23,895 | 82 |
| 2013 | 7630 | 63 | 233 | 6 | 3225 | 3 | 11,088 | 72 |
| 2014 | 5148 | 15 | 154 | 0 | 2194 | 0 | 7496 | 15 |
| 2015 | 2958 | 10 | 98 | 0 | 1572 | 0 | 4628 | 10 |
| 2016 | 1815 | 1 | 47 | 0 | 1152 | 1 | 3014 | 2 |
| 2017 | 6993 | 24 | 150 | 1 | 3123 | 1 | 10,266 | 26 |
| 2018 | 8954 | 22 | 217 | 1 | 4551 | 1 | 13,722 | 24 |
| 2019 | 8839 | 25 | 224 | 1 | 4823 | 1 | 13,886 | 27 |
| 2020 | 9222 | 19 | 231 | 0 | 4511 | 0 | 13,964 | 19 |
| Years | Incidence Rate | Mortality Rate | ||||
|---|---|---|---|---|---|---|
| p-Value | Interpretation | p-Value | Interpretation | |||
| 2011 | 0.0772 | 0.4087 | Scattered | 0.2137 | 0.01 | Clustered |
| 2012 | 0.0913 | 0.0538 | Clustered | 0.4168 | 0.0416 | Clustered |
| 2013 | 0.1096 | 0.0692 | Clustered | 0.3245 | 0.0907 | Clustered |
| 2014 | 0.1193 | 0.1777 | Scattered | 0.6526 | 0.2942 | Scattered |
| 2015 | 0.1246 | 0.4187 | Scattered | 1.2458 | 0.0364 | Clustered |
| 2016 | 0.1331 | 0.4124 | Scattered | 1.2255 | 0.0234 | Clustered |
| 2017 | 0.0875 | 0.2304 | Scattered | 0.322 | 0.3345 | Scattered |
| 2018 | 0.0923 | 0.4539 | Scattered | 0.2734 | 0.2839 | Scattered |
| 2019 | 0.1011 | 0.301 | Scattered | 0.2486 | 0.2691 | Scattered |
| 2020 | 0.0981 | 0.4566 | Scattered | 0.2652 | 0.1931 | Scattered |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chinpong, K.; Thavornwattana, K.; Armatrmontree, P.; Chienwichai, P.; Lawpoolsri, S.; Silachamroon, U.; Maude, R.J.; Rotejanaprasert, C. Spatiotemporal Epidemiology of Tuberculosis in Thailand from 2011 to 2020. Biology 2022, 11, 755. https://doi.org/10.3390/biology11050755
Chinpong K, Thavornwattana K, Armatrmontree P, Chienwichai P, Lawpoolsri S, Silachamroon U, Maude RJ, Rotejanaprasert C. Spatiotemporal Epidemiology of Tuberculosis in Thailand from 2011 to 2020. Biology. 2022; 11(5):755. https://doi.org/10.3390/biology11050755
Chicago/Turabian StyleChinpong, Kawin, Kaewklao Thavornwattana, Peerawich Armatrmontree, Peerut Chienwichai, Saranath Lawpoolsri, Udomsak Silachamroon, Richard J. Maude, and Chawarat Rotejanaprasert. 2022. "Spatiotemporal Epidemiology of Tuberculosis in Thailand from 2011 to 2020" Biology 11, no. 5: 755. https://doi.org/10.3390/biology11050755
APA StyleChinpong, K., Thavornwattana, K., Armatrmontree, P., Chienwichai, P., Lawpoolsri, S., Silachamroon, U., Maude, R. J., & Rotejanaprasert, C. (2022). Spatiotemporal Epidemiology of Tuberculosis in Thailand from 2011 to 2020. Biology, 11(5), 755. https://doi.org/10.3390/biology11050755







