The Curcumin Supplementation with Piperine Can Influence the Acute Elevation of Exercise-Induced Cytokines: Double-Blind Crossover Study
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
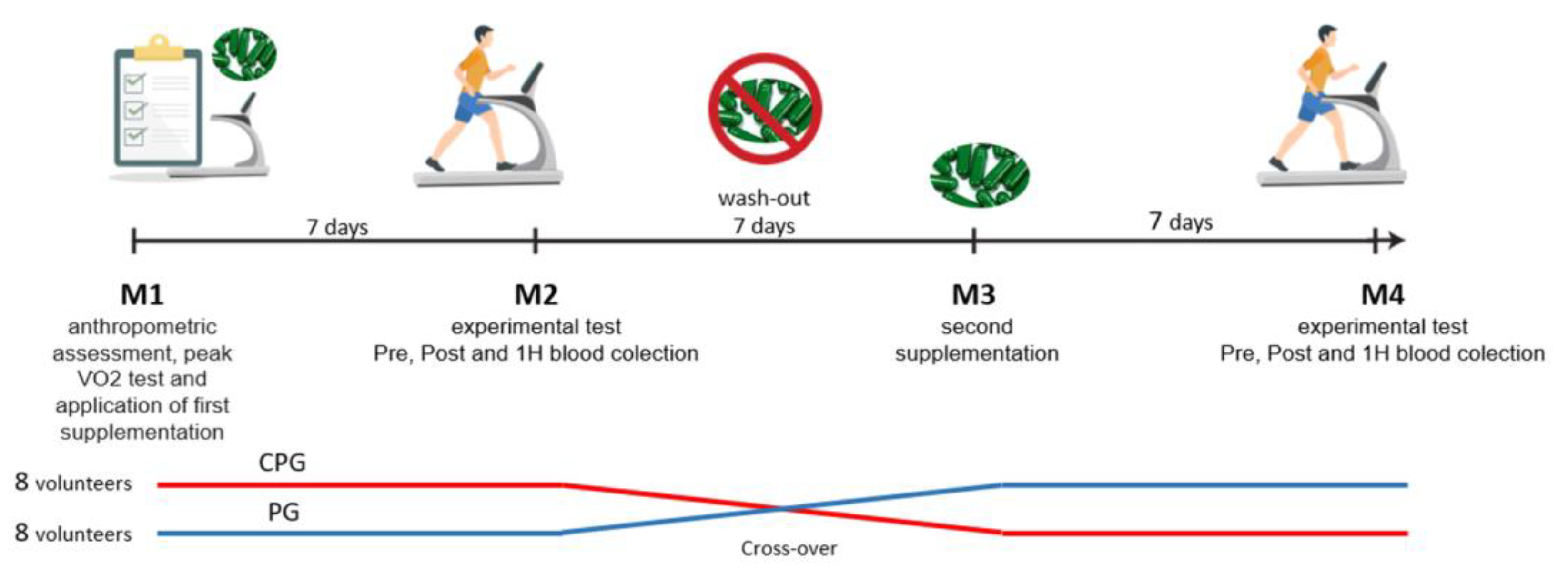
2.1. Study Design and Ethical Approval
2.2. Study Protocol
2.3. Blood Sampling and Laboratory Measurements
2.4. Statistical Analysis
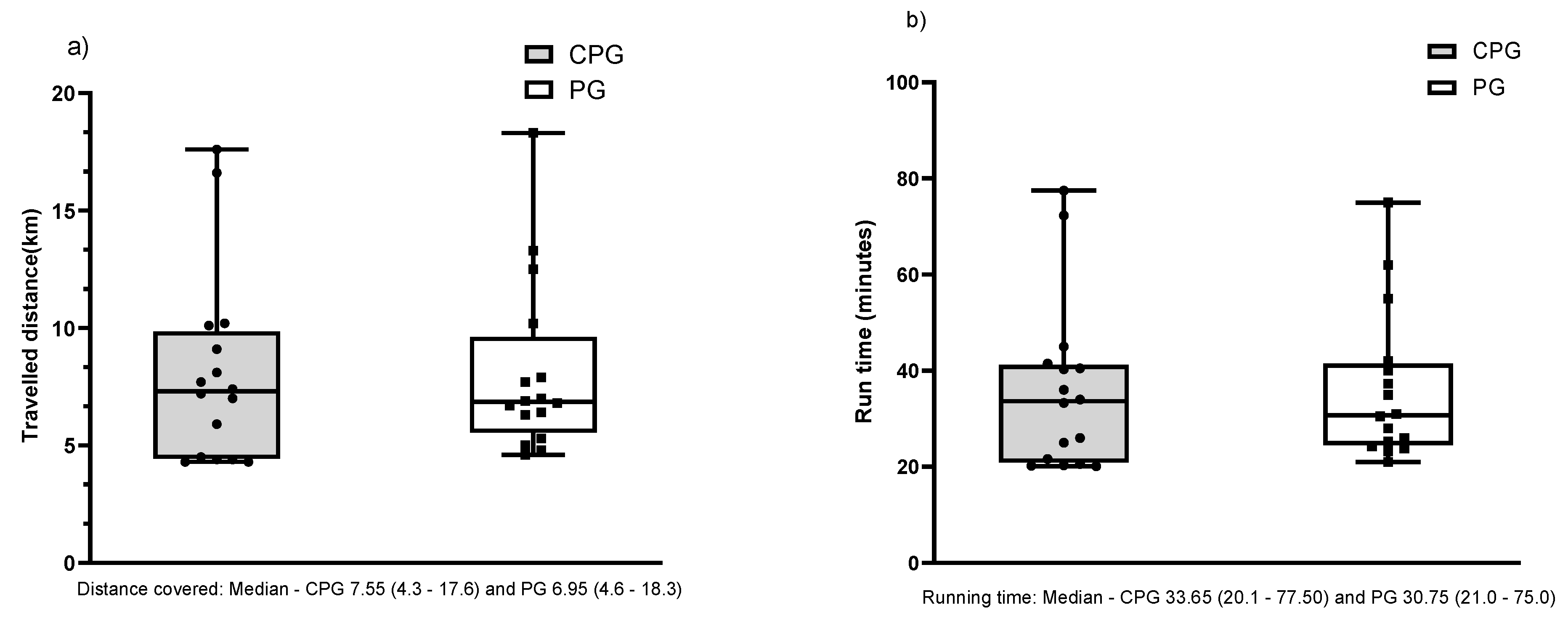
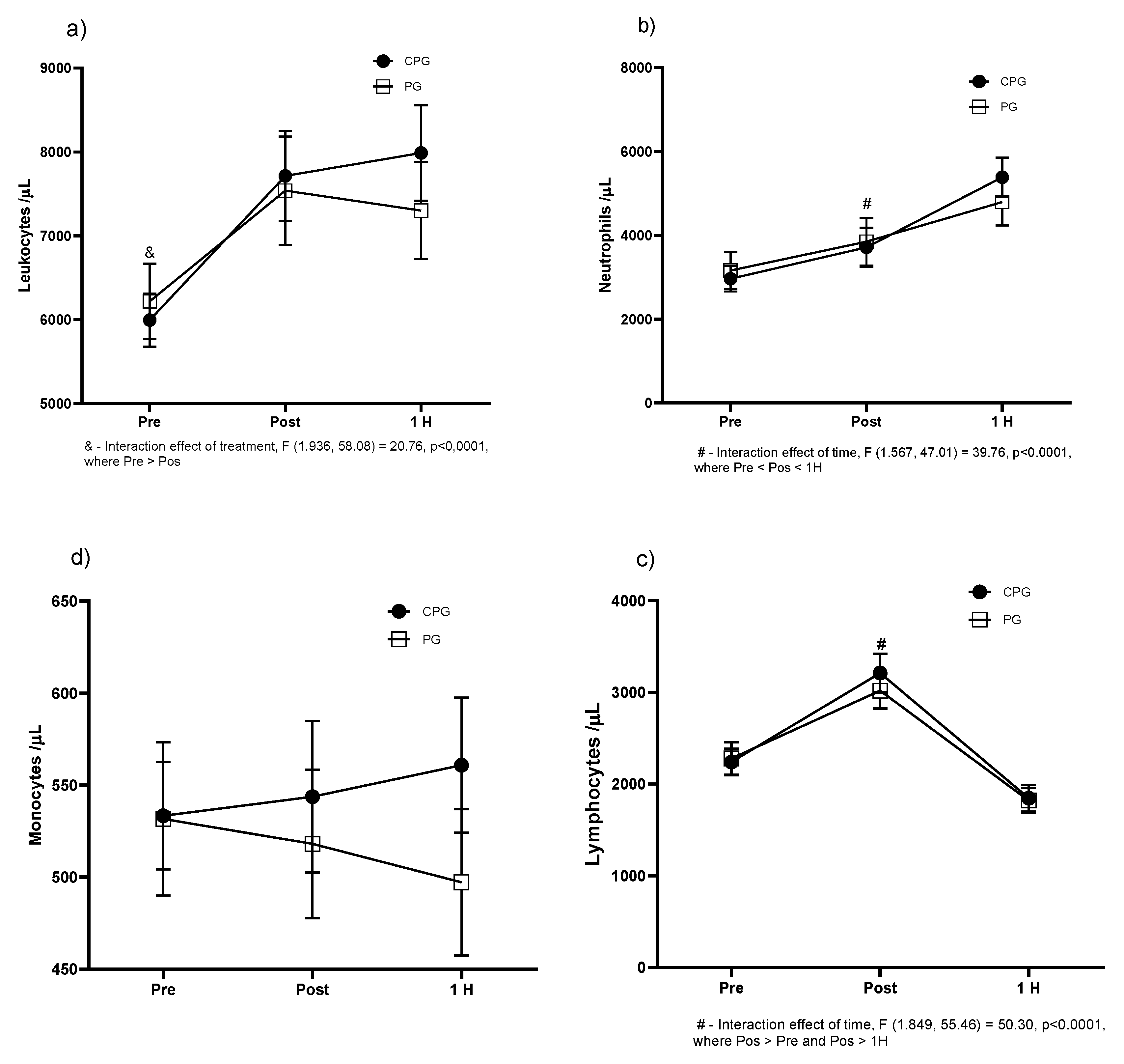
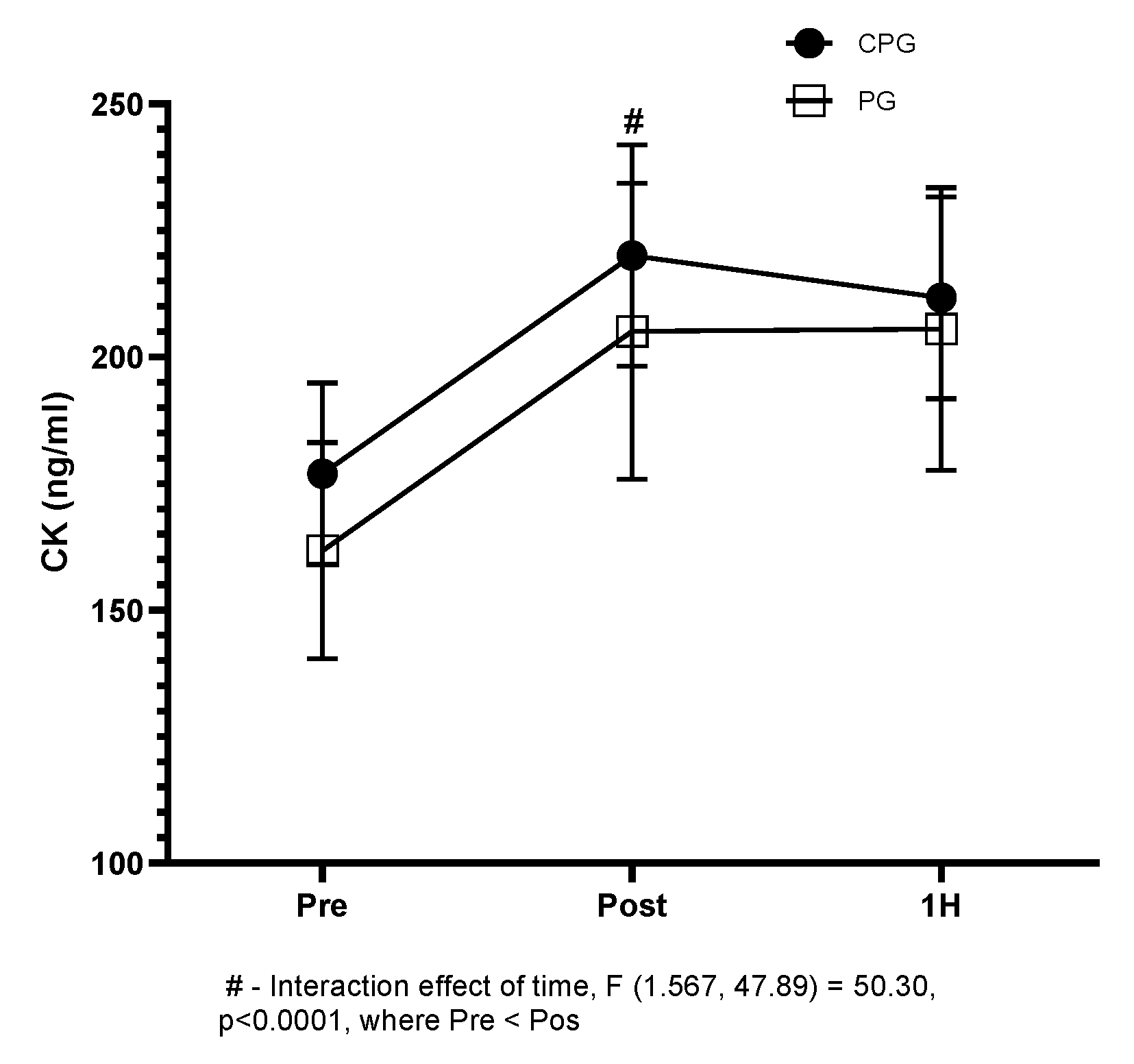
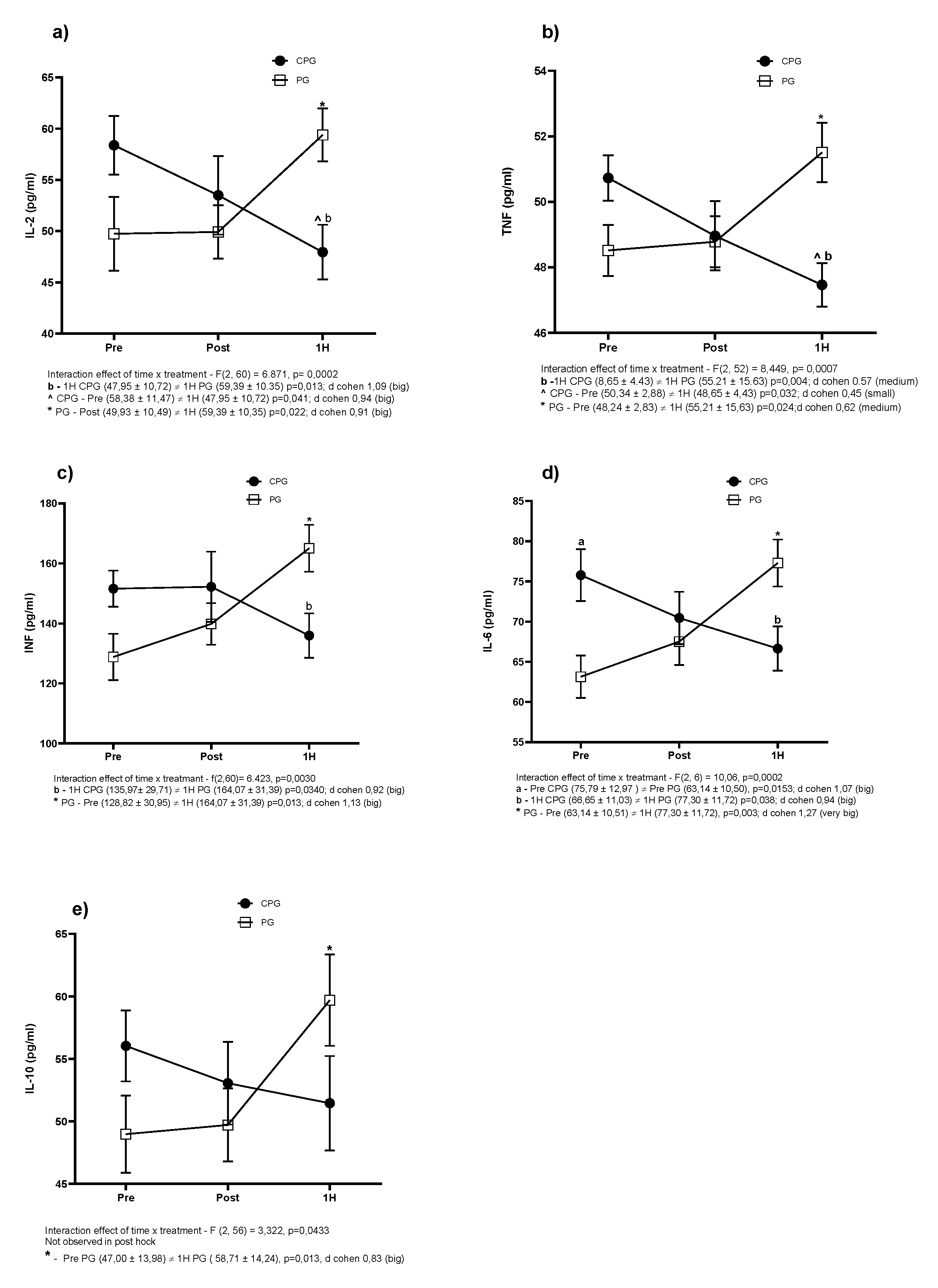
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ramos-Campo, D.J.; Pérez, A.; Ávila-Gandía, V.; Pérez-Piñero, S.; Rubio-Arias, J.P. Impact of Caffeine Intake on 800-m Running Performance and Sleep Quality in Trained Runners. Nutrients 2019, 11, 2040. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kreider, R.B.; Wilborn, C.D.; Taylor, L.; Campbell, B.; Almada, A.L.; Collins, R.; Cooke, M.; Earnest, C.P.; Greenwood, M.; Kalman, D.S.; et al. ISSN exercise & sport nutrition review: Research & recommendations. J. Int. Soc. Sports Nutr. 2010, 7, 7. [Google Scholar] [CrossRef] [Green Version]
- Huang, W.-C.; Chiu, W.-C.; Chuang, H.-L.; Tang, D.-W.; Lee, Z.-M.; Deh-Wei, T.; Chen, F.-A. Effect of Curcumin Supplementation on Physiological Fatigue and Physical Performance in Mice. Nutrients 2015, 7, 905–921. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tanabe, Y.; Chino, K.; Ohnishi, T.; Ozawa, H.; Sagayama, H.; Maeda, S.; Takahashi, H. Effects of oral curcumin ingested before or after eccentric exercise on markers of muscle damage and inflammation. Scand. J. Med. Sci. Sports 2019, 29, 524–534. [Google Scholar] [CrossRef] [PubMed]
- Sueth-Santiago, V.; Mendes-Silva, G.P.; Decoté-Ricardo, D.; Freire de Lima, M.E. Curcumina, o pó dourado do açafrão-da-terra: Introspecções sobre química e atividades biológicas. Quím. Nova 2015, 38, 538–552. [Google Scholar]
- Gorgani, L.; Mohammadi, M.; Najafpour, G.D.; Nikzad, M. Piperine-The Bioactive Compound of Black Pepper: From Isolation to Medicinal Formulations. Compr. Rev. Food Sci. Food Saf. 2017, 16, 124–140. [Google Scholar] [CrossRef] [PubMed]
- Bielak-Zmijewska, A.; Grabowska, W.; Ciolko, A.; Bojko, A.; Mosieniak, G.; Bijoch, Ł.; Sikora, E. The Role of Curcumin in the Modulation of Ageing. Int. J. Mol. Sci. 2019, 20, 1239. [Google Scholar] [CrossRef] [Green Version]
- Kramer, H.F.; Goodyear, L.J. Exercise, MAPK, and NF-κB signaling in skeletal muscle. J. Appl. Physiol. 2007, 103, 388–395. [Google Scholar] [CrossRef]
- Higgins, M.R.; Izadi, A.; Kaviani, M.; Health, P. Antioxidants and Exercise Performance: With a Focus on Vitamin e and c Supplementation. Int. J. Environ. Res. Public Health 2020, 17, 8452. [Google Scholar] [CrossRef]
- Anand, P.; Thomas, S.G.; Kunnumakkara, A.B.; Sundaram, C.; Harikumar, K.B.; Sung, B.; Tharakan, S.T.; Misra, K.; Priyadarsini, I.K.; Rajasekharan, K.N.; et al. Biological activities of curcumin and its analogues (Congeners) made by man and Mother Nature. Biochem. Pharmacol. 2008, 76, 1590–1611. [Google Scholar] [CrossRef]
- Kim, J.H.; Gupta, S.C.; Park, B.; Yadav, V.R.; Aggarwal, B.B. Turmeric (Curcuma longa) inhibits inflam-matory nuclear factor (NF)-κB and NF-κB-regulated gene products and induces death receptors leading to suppressed prolif-eration, induced chemosensitization, and suppressed osteoclastogenesis. Mol. Nutr. Food Res. 2012, 56, 454–465. [Google Scholar] [CrossRef] [Green Version]
- Marquardt, J.U.; Gomez-Quiroz, L.; Camacho, L.O.A.; Pinna, F.; Lee, Y.-H.; Kitade, M.; Domínguez, M.P.; Castven, D.; Breuhahn, K.; Conner, E.A. Curcumin effectively inhibits oncogenic NF-κB signaling and restrains stemness features in liver cancer. J. Hepatol. 2015, 63, 661–669. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ghorbani, Z.; Hekmatdoost, A.; Mirmiran, P. Anti-Hyperglycemic and Insulin Sensitizer Effects of Turmeric and Its Principle Constituent Curcumin. Int. J. Endocrinol. Metab. 2014, 12, e18081. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, Q.; Sun, J.; Mohammadtursun, N.; Wu, J.; Dong, J.; Li, L. Curcumin inhibits cigarette smoke-induced in-flammation via modulating the PPARγ-NF-κB signaling pathway. Food Funct. 2019, 10, 7983–7994. [Google Scholar] [CrossRef] [PubMed]
- Drobnic, F.; Riera, J.; Appendino, G.; Togni, S.; Franceschi, F.; Valle, X.; Pons, A.; Tur, J. Reduction of delayed onset muscle soreness by a novel curcumin delivery system (Meriva®): A randomised, placebo-controlled trial. J. Int. Soc. Sports Nutr. 2014, 11, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Jackson, A.S.; Pollock, M.L. Generalized equations for predicting body density of men. Br. J. Nutr. 1978, 40, 497–504. [Google Scholar] [CrossRef] [Green Version]
- Castagna, C.; Impellizzeri, F.; Cecchini, E.; Rampinini, E.; Alvarez, J.C.; Research, C. Effects of intermittent-endurance fitness on match performance in young male soccer players. J. Strenth Cond. Res. 2009, 23, 1954–1959. [Google Scholar] [CrossRef] [Green Version]
- Garnacho-Castaño, M.V.; Palau-Salvà, G.; Cuenca, E.; Muñoz-González, A.; García-Fernández, P.; del Carmen Lozano-Estevan, M.; Veiga-Herreros, P.; Maté-Muñoz, J.L.; Domínguez, R. Effects of a single dose of beetroot juice on cycling time trial performance at ventilatory thresholds intensity in male triath-letes. J. Int. Soc. Sports Nutr. 2018, 15, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Manoy, P.; Yuktanandana, P.; Tanavalee, A.; Anomasiri, W.; Ngarmukos, S.; Tanpowpong, T.; Honsawek, S. Vitamin D Supplementation Improves Quality of Life and Physical Performance in Osteoarthritis Patients. Nutrients 2017, 9, 799. [Google Scholar] [CrossRef] [Green Version]
- Fielding, R.; Riede, L.; Lugo, J.P.; Bellamine, A. l-Carnitine Supplementation in Recovery after Exercise. Nutrients 2018, 10, 349. [Google Scholar] [CrossRef] [Green Version]
- Mielgo-Ayuso, J.; Calleja-Gonzalez, J.; Del Coso, J.; Urdampilleta, A.; León-Guereño, P.; Fernández-Lázaro, D. Caffeine Supplementation and Physical Performance, Muscle Damage and Perception of Fatigue in Soccer Players: A Systematic Review. Nutrients 2019, 11, 440. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shin, Y.O.; Lee, J.B. Leukocyte chemotactic cytokine and leukocyte subset responses during ultra-marathon running. Cytokine 2013, 61, 364–369. [Google Scholar] [CrossRef] [PubMed]
- Nieman, D.C. Nutrition, exercise, and immune system function. Clin. Sports Med. 1999, 18, 537–548. [Google Scholar] [CrossRef]
- Tidball, J.G. Inflammatory processes in muscle injury and repair. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2005, 288, R345–R353. [Google Scholar] [CrossRef] [Green Version]
- Assumpção, C.D.O.; Lima, L.; Oliveira, F.B.D.; Greco, C.C.; Denadai, B.S. Exercise-Induced Muscle Damage and Running Economy in Humans. Sci. World J. 2013, 2013, 1–11. [Google Scholar] [CrossRef]
- Qu, C.; Wu, Z.; Xu, M.; Qin, F.; Dong, Y.; Wang, Z.; Zhao, J. Cryotherapy Models and Timing-Sequence Recovery of Exercise-Induced Muscle Damage in Middle-and Long-Distance Runners. J. Athl. Train. 2020, 55, 329–335. [Google Scholar] [CrossRef] [Green Version]
- Millet, G.Y.; Tomazin, K.; Verges, S.; Vincent, C.; Bonnefoy, R.; Boisson, R.-C.; Gergelé, L.; Féasson, L.; Martin, V. Neu-romuscular consequences of an extreme mountain ultra-marathon. PloS ONE 2011, 6, e17059. [Google Scholar] [CrossRef] [Green Version]
- Delecroix, B.; Abd Elbasset Abaïdia, C.L.; Dawson, B.; Dupont, G. Curcumin and piperine supplementation and recovery following exercise induced muscle damage: A randomized controlled trial. J. Sports Sci. Med. 2017, 16, 147. [Google Scholar]
- Jäger, R.; Purpura, M.; Kerksick, C.M. Eight Weeks of a High Dose of Curcumin Supplementation May Attenuate Performance Decrements Following Muscle-Damaging Exercise. Nutrients 2019, 11, 1692. [Google Scholar] [CrossRef] [Green Version]
- McFarlin, B.K.; Venable, A.S.; Henning, A.L.; Sampson, J.N.B.; Pennel, K.; Vingren, J.L.; Hill, D.W. Reduced inflammatory and muscle damage biomarkers following oral supplementation with bioavailable curcumin. BBA Clin. 2016, 5, 72–78. [Google Scholar] [CrossRef] [Green Version]
- Oh, J.-G.; Hwang, D.-J.; Heo, T.-H. Direct regulation of IL-2 by curcumin. Biochem. Biophys. Res. Commun. 2018, 495, 300–305. [Google Scholar] [CrossRef]
- Pedersen, B.K.; Steensberg, A.; Schjerling, P. Exercise and interleukin-6. Curr. Opin. Hematol. 2001, 8, 137–141. [Google Scholar] [CrossRef] [PubMed]
- Kostrzewa-Nowak, D.; Ciechanowicz, A.; Clark, J.S.; Nowak, R. Damage-Associated Molecular Patterns and Th-Cell-Related Cytokines Released after Progressive Effort. J. Clin. Med. 2020, 9, 876. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ali, B.H.; Al-Salam, S.; Al Suleimani, Y.; Al Kalbani, J.; Al Bahlani, S.; Ashique, M.; Manoj, P.; Al Dhahli, B.; Al Abri, N.; Naser, H.T.; et al. Curcumin Ameliorates Kidney Function and Oxidative Stress in Experimental Chronic Kidney Disease. Basic Clin. Pharmacol. Toxicol. 2018, 122, 65–73. [Google Scholar] [CrossRef] [PubMed]
- Yargic, M.P.; Torgutalp, S.; Akin, S.; Babayeva, N.; Torgutalp, M.; Demirel, H.A.; Demirel, A.H. Acute long-distance trail running increases serum IL-6, IL-15, and Hsp72 levels. Appl. Physiol. Nutr. Metab. 2019, 44, 627–631. [Google Scholar] [CrossRef]
- Pedersen, B.K.; Febbraio, M.A. PMuscle as an endocrine organ: Focus on muscle-derived interleukin-6. Physiol. Rev. 2008, 88, 1379–1406. [Google Scholar] [CrossRef] [Green Version]
- Pedersen, B.K. Muscular interleukin-6 and its role as an energy sensor. Med. Sci. Sports Exerc. 2012, 44, 392–396. [Google Scholar] [CrossRef]
- Janeway, C.A., Jr.; Travers, P.; Walport, M.; Shlomchik, M. Imunobiologia: O sistema imune na saúde e na doença. In Imunobiologia: O Sistema Imune na Saúde e na Doença; Artmed: Porto Alegre, Brazil, 2007; p. 824. [Google Scholar]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Miranda-Castro, S.; Aidar, F.J.; de Moura, S.S.; Marcucci-Barbosa, L.; Lobo, L.F.; de Assis Dias Martins-Júnior, F.; da Silva Filha, R.; Vaz de Castro, P.A.S.; Simões e Silva, A.C.; da Glória de Souza, D.; et al. The Curcumin Supplementation with Piperine Can Influence the Acute Elevation of Exercise-Induced Cytokines: Double-Blind Crossover Study. Biology 2022, 11, 573. https://doi.org/10.3390/biology11040573
Miranda-Castro S, Aidar FJ, de Moura SS, Marcucci-Barbosa L, Lobo LF, de Assis Dias Martins-Júnior F, da Silva Filha R, Vaz de Castro PAS, Simões e Silva AC, da Glória de Souza D, et al. The Curcumin Supplementation with Piperine Can Influence the Acute Elevation of Exercise-Induced Cytokines: Double-Blind Crossover Study. Biology. 2022; 11(4):573. https://doi.org/10.3390/biology11040573
Chicago/Turabian StyleMiranda-Castro, Stéfani, Felipe J. Aidar, Samara Silva de Moura, Lucas Marcucci-Barbosa, Lázaro Fernandes Lobo, Francisco de Assis Dias Martins-Júnior, Roberta da Silva Filha, Pedro Alves Soares Vaz de Castro, Ana Cristina Simões e Silva, Danielle da Glória de Souza, and et al. 2022. "The Curcumin Supplementation with Piperine Can Influence the Acute Elevation of Exercise-Induced Cytokines: Double-Blind Crossover Study" Biology 11, no. 4: 573. https://doi.org/10.3390/biology11040573
APA StyleMiranda-Castro, S., Aidar, F. J., de Moura, S. S., Marcucci-Barbosa, L., Lobo, L. F., de Assis Dias Martins-Júnior, F., da Silva Filha, R., Vaz de Castro, P. A. S., Simões e Silva, A. C., da Glória de Souza, D., da Silva, S. A., de Castro Pinto, K. M., de Paula Costa, G., Silva, A. F., Clemente, F. M., Pereira, W. V. C., & Nunes-Silva, A. (2022). The Curcumin Supplementation with Piperine Can Influence the Acute Elevation of Exercise-Induced Cytokines: Double-Blind Crossover Study. Biology, 11(4), 573. https://doi.org/10.3390/biology11040573









