MOET Efficiency in a Spanish Herd of Japanese Black Heifers and Analysis of Environmental and Metabolic Determinants
Abstract
Simple Summary
Abstract
1. Introduction
2. Material and Methods
2.1. Animals and Herd
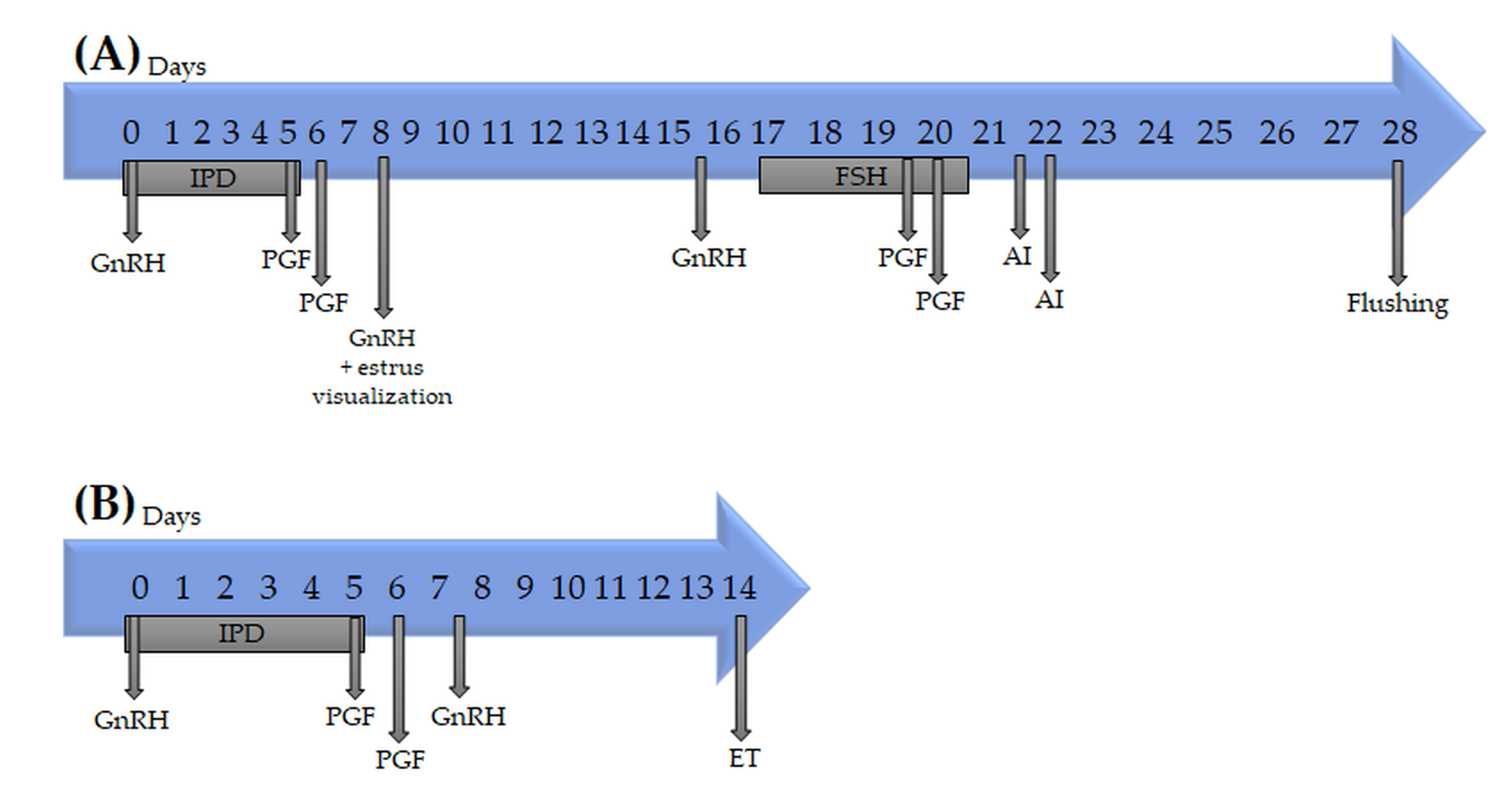
2.2. Donor Synchronization and Superovulation
2.3. Flushing of Embryos and Their Evaluation
2.4. Recipient Synchronization and Embryo Transfer
2.5. Statistical Analysis
3. Results
3.1. Embryo Flushings and Donor Metabolite Analyses
3.2. Embryo Transfers and Recipients’ Pregnancy Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Ethical Statement
References
- Nomura, T.; Honda, T.; Mukai, F. Inbreeding and effective population size of Japanese Black Cattle. J. Anim. Sci. 2001, 79, 366–370. [Google Scholar] [CrossRef] [PubMed]
- Motoyama, M.; Sasaki, K.; Watanabe, A. Wagyu and the factors contributing to its beef quality: A japanese industry overview. Meat Sci. 2016, 120, 10–18. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.B.; Lunt, D.K.; Chung, K.Y.; Choi, C.B.; Tume, R.K.; Zembayashi, M. Adiposity, fatty acid composition, and delta-9 desaturase activity during growth in beef cattle. Anim. Sci. J. 2006, 77, 478–486. [Google Scholar] [CrossRef]
- Nogi, T.; Honda, T.; Mukai, F.; Okagaki, T.; Oyama, K. Heritabilities and genetic correlations of fatty acid compositions in longissimus muscle lipid with carcass traits in Japanese Black Cattle. J. Anim. Sci. 2011, 89, 615–621. [Google Scholar] [CrossRef] [PubMed]
- Gotoh, T.; Albrecht, E.; Teuscher, F.; Kawabata, K.; Sakashita, K.; Iwamoto, H.; Wegner, J. Differences in muscle and fat accretion in Japanese Black and European Cattle. Meat Sci. 2009, 82, 300–308. [Google Scholar] [CrossRef]
- Trenkle, A.; Topel, D.G. Relationship of some endocrine measurements to growth and carcass composition of cattle. J. Anim. Sci. 1978, 46, 1604–1609. [Google Scholar] [CrossRef]
- Matsuzaki, M.; Takizawa, S.; Ogawa, M. Plasma Insulin, metabolite concentrations, and carcass characteristics of Japanese Black, Japanese Brown, and Holstein Steers. J. Anim. Sci. 1997, 75, 3287–3293. [Google Scholar] [CrossRef]
- Chen, G.; Koyama, K.; Yuan, X.; Lee, Y.; Zhou, Y.T.; O’Doherty, R.; Newgard, C.B.; Unger, R.H. Disappearance of body fat in normal rats induced by adenovirus-mediated leptin gene therapy. Proc. Natl. Acad. Sci. USA 1996, 93, 14795–14799. [Google Scholar] [CrossRef]
- Siegrist-Kaiser, C.A.; Pauli, V.; Juge-Aubry, C.E.; Boss, O.; Pernin, A.; Chin, W.W.; Cusin, I.; Rohner-Jeanrenaud, F.; Burger, A.G.; Zapf, J.; et al. Direct effects of leptin on brown and white adipose tissue. J. Clin. Investig. 1997, 100, 2858–2864. [Google Scholar] [CrossRef]
- Minokoshi, Y.; Kim, Y.-B.; Peroni, O.D.; Fryer, L.G.D.; Müller, C.; Carling, D.; Kahn, B.B. Leptin stimulates fatty-acid oxidation by activating amp-activated protein kinase. Nature 2002, 415, 339–343. [Google Scholar] [CrossRef]
- Corva, P.M.; Macedo, G.V.F.; Soria, L.A.; Mazzucco, J.P.; Motter, M.; Villarreal, E.L.; Schor, A.; Mezzadra, C.A.; Melucci, L.M.; Miquel, M.C. Effect of leptin gene polymorphisms on growth, slaughter and meat quality traits of grazing Brangus steers. Genet. Mol. Res. GMR 2009, 8, 105–116. [Google Scholar] [CrossRef] [PubMed]
- Silva, D.B.S.; Crispim, B.A.; Silva, L.E.; Oliveira, J.A.; Siqueira, F.; Seno, L.O.; Grisolia, A.B. Genetic variations in the leptin gene associated with growth and carcass traits in Nellore Cattle. Genet. Mol. Res. GMR 2014, 13, 3002–3012. [Google Scholar] [CrossRef] [PubMed]
- Tian, J.; Zhao, Z.; Zhang, L.; Zhang, Q.; Yu, Z.; Li, J.; Yang, R. Association of the leptin gene E2-169T>C and E3-299T>a mutations with carcass and meat quality traits of the Chinese Simmental-cross steers. Gene 2013, 518, 443–448. [Google Scholar] [CrossRef] [PubMed]
- Kawaguchi, F.; Okura, K.; Oyama, K.; Mannen, H.; Sasazaki, S. Identification of leptin gene polymorphisms associated with carcass traits and fatty acid composition in Japanese Black Cattle. Anim. Sci. J. Nihon Chikusan Gakkaiho 2017, 88, 433–438. [Google Scholar] [CrossRef] [PubMed]
- Buchanan, F.C.; Van Kessel, A.G.; Waldner, C.; Christensen, D.A.; Laarveld, B.; Schmutz, S.M. Hot Topic: An Association between a leptin single nucleotide polymorphism and milk and protein yield. J. Dairy Sci. 2003, 86, 3164–3166. [Google Scholar] [CrossRef]
- Glantz, M.; Månsson, H.L.; Stålhammar, H.; Paulsson, M. Effect of Polymorphisms in the leptin, leptin receptor and Acyl-CoA:Diacylglycerol Acyltransferase 1 (DGAT1) Genes and genetic polymorphism of milk proteins on bovine milk composition. J. Dairy Res. 2012, 79, 110–118. [Google Scholar] [CrossRef] [PubMed]
- Giblin, L.; Butler, S.T.; Kearney, B.M.; Waters, S.M.; Callanan, M.J.; Berry, D.P. Association of bovine leptin polymorphisms with energy output and energy storage traits in progeny tested Holstein-Friesian Dairy Cattle Sires. BMC Genet. 2010, 11, 73. [Google Scholar] [CrossRef]
- Fantuzzi, G.; Faggioni, R. Leptin in the regulation of immunity, inflammation, and hematopoiesis. J. Leukoc. Biol. 2000, 68, 437–446. [Google Scholar]
- Ingvartsen, K.L.; Boisclair, Y.R. Leptin and the regulation of food intake, energy homeostasis and immunity with special focus on periparturient ruminants. Domest. Anim. Endocrinol. 2001, 21, 215–250. [Google Scholar] [CrossRef]
- Yonekura, S.; Oka, A.; Noda, M.; Uozumi, N.; Yonezawa, T.; Katoh, K.; Obara, Y. Relationship between serum leptin concentrations and the marbling scores in Japanese Black cattle. Anim. Sci. J. 2002. [Google Scholar] [CrossRef]
- Kulcsár, M.; Jánosi, S.; Lehtolainen, T.; Kátai, L.; Delavaud, C.; Balogh, O.; Chilliard, Y.; Pyörälä, S.; Rudas, P.; Huszenicza, G. Feeding-Unrelated Factors Influencing the plasma leptin level in ruminants. Domest. Anim. Endocrinol. 2005, 29, 214–226. [Google Scholar] [CrossRef] [PubMed]
- Wu, B.; Zan, L. Enhance beef cattle improvement by embryo biotechnologies. Reprod. Domest. Anim. 2012, 47, 865–871. [Google Scholar] [CrossRef] [PubMed]
- Ferré, L.B.; Kjelland, M.E.; Strøbech, L.B.; Hyttel, P.; Mermillod, P.; Ross, P.J. Review: Recent advances in bovine in vitro embryo production: Reproductive biotechnology history and methods. Anim. Int. J. Anim. Biosci. 2020, 14, 991–1004. [Google Scholar] [CrossRef]
- Numabe, T.; Oikawa, T.; Kikuchi, T.; Horiuchi, T. Production efficiency of Japanese Black calves by transfer of bovine embryos produced in vitro. Theriogenology 2000, 54, 1409–1420. [Google Scholar] [CrossRef]
- Honda, T.; Nomura, T.; Mukai, F. Conservation of genetic diversity in the Japanese Black cattle population by the construction of partially isolated lines. J. Anim. Breed. Genet. Z. Tierz. Zucht. 2005, 122, 188–194. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Sasaki, Y.; Uematsu, M.; Kitahara, G.; Osawa, T. Reproductive performance of Japanese Black Cattle: Association with herd size, season, and parity in commercial cow-calf operations. Theriogenology 2016, 86, 2156–2161. [Google Scholar] [CrossRef]
- Irikura, N.; Uematsu, M.; Kitahara, G.; Osawa, T.; Sasaki, Y. Effects of service number on conception rate in Japanese Black Cattle. Reprod. Domest. Anim. 2018, 53, 34–39. [Google Scholar] [CrossRef]
- Kasimanickam, R.; Kasimanickam, V.; Kappes, A. Timed artificial insemination strategies with or without short-term natural service and pregnancy success in beef heifers. Theriogenology 2021, 166, 97–103. [Google Scholar] [CrossRef]
- White, S.S.; Kasimanickam, R.K.; Kasimanickam, V.R. Fertility after two doses of PGF2α concurrently or at 6-hour interval on the day of CIDR removal in 5-Day CO-Synch progesterone-based synchronization protocols in beef heifers. Theriogenology 2016, 86, 785–790. [Google Scholar] [CrossRef]
- Fernandez-Novo, A.; Santos-Lopez, S.; Pesantez-Pacheco, J.L.; Pérez-Villalobos, N.; Heras-Molina, A.; Gonzalez-Martin, J.V.; Astiz, S. Effects on synchronization and reproductive efficiency of delaying the removal of the intravaginal progesterone device by 24 h in the 5d Co-Synch protocol in heifers. Anim. Open Access J. 2021, 11, 849. [Google Scholar] [CrossRef]
- Konishi, M.; Aoyagi, Y.; Takedomi, T.; Itakura, H.; Itoh, T.; Yazawa, S. Production and transfer of IVF embryos from individual inhibin-immunized cows by ultrasound-guided transvaginal follicular aspiration. J. Vet. Med. Sci. 1996, 58, 893–896. [Google Scholar] [CrossRef] [PubMed]
- Livestock Improvement Association of Japan. Available online: http://liaj.or.jp/giken/gijutsubu/seieki/jyutai.htm (accessed on 18 December 2021).
- Sao, K.; Fujita, T. Elucidation of genes involved in the conception rates of cows decrease. Bull. Oita Prefect. Anim. Ind. Exp. Stn. 2010, 39, 17–19. [Google Scholar]
- Sutmoller, P.; Wrathall, A.E. A Quantitative assessment of the risk of transmission of foot-and-mouth disease, bluetongue and vesicular stomatitis by embryo transfer in cattle. Prev. Vet. Med. 1997, 32, 111–132. [Google Scholar] [CrossRef]
- Thibier, M. Embryo Transfer: A comparative biosecurity advantage in international movements of germplasm. Rev. Sci. Tech. Int. Off. Epizoot. 2011, 30, 177–188. [Google Scholar] [CrossRef]
- Baruselli, P.S.; Ferreira, R.M.; Vieira, L.M.; Souza, A.H.; Bó, G.A.; Rodrigues, C.A. Use of embryo transfer to alleviate infertility caused by heat stress. Theriogenology 2020, 155, 1–11. [Google Scholar] [CrossRef]
- Fernandez-Novo, A.; Pérez-Garnelo, S.S.; Villagrá, A.; Pérez-Villalobos, N.; Astiz, S. The effect of stress on reproduction and reproductive technologies in beef cattle-A review. Anim. Open Access J. 2020, 10, 2096. [Google Scholar] [CrossRef]
- Nishigai, M.; Kamomae, H.; Tanaka, T.; Kaneda, Y. Improvement of pregnancy rate in Japanese Black cows by administration of HCG to recipients of transferred frozen-thawed embryos. Theriogenology 2002, 58, 1597–1606. [Google Scholar] [CrossRef]
- Lukaszewska, J.; Hansel, W. Corpus luteum maintenance during early pregnancy in the cow. J. Reprod. Fertil. 1980, 59, 485–493. [Google Scholar] [CrossRef]
- Setiaji, A.; Oikawa, T. Genetics of heifer reproductive traits in Japanese Black Cattle. Asian-Australas. J. Anim. Sci. 2020, 33, 197–202. [Google Scholar] [CrossRef]
- Tanigawa, S.; Kondo, N.; Ogawa, Y.; Fujiura, T.; Shuqing, H.; Takao, Y.; Fukushima, M.; Watanabe, O.; Kohama, N. The relationship between serum vitamin A level of Japanese black cattle and light reflection on the pupil. In IEEE/SICE International Symposium on System Integration (SII); IEEE: Piscataway, NJ, USA, 2011; pp. 187–191. [Google Scholar] [CrossRef]
- AEMET. Available online: https://www.aemet.es/es/web/datos_abiertos/estadisticas/vigilancia_clima (accessed on 18 December 2021).
- GMAO; NASA. Available online: https://gmao.gsfc.nasa.gov/reanalysis/MERRA-2/ (accessed on 18 December 2021).
- Wheather Spark. Available online: https://es.weatherspark.com/y/42909/Clima-promedio-en-Aliaga-Espa%C3%B1a-durante-todo-el-a%C3%B1o (accessed on 18 December 2021).
- National Research Council. Nutrient Requirements of Beef Cattle: Seventh Revised Edition: Update 2000; The National Academies Press: Washington, DC, USA, 2000. [Google Scholar]
- Stringfellow, D.A.; Givens, M.D.; International Embryo Transfer Society. Manual of the International Embryo Transfer Society: A Procedural Guide and General Information for the Use of Embryo Transfer Technology Emphasizing Sanitary Procedures; International Embryo Transfer Society; Savory, Ill.: Boulder, CO, USA, 2010. [Google Scholar]
- Dochi, O. Direct transfer of frozen-thawed bovine embryos and its application in cattle reproduction management. J. Reprod. Dev. 2019, 65, 389–396. [Google Scholar] [CrossRef]
- Leibo, S.P. A one-step method for direct nonsurgical transfer of frozen-thawed bovine embryos. Theriogenology 1984, 21, 767–790. [Google Scholar] [CrossRef]
- Edmonson, A.J.; Lean, I.J.; Weaver, L.D.; Farver, T.; Webster, G. A body condition scoring chart for Holstein Dairy cows. J. Dairy Sci. 1989, 72, 68–78. [Google Scholar] [CrossRef]
- An, L.; Ling, P.P.; Zhu, X.; Liu, Y.; Zhang, F.; Ma, X.; Xu, B.; Wang, Y.; Du, Z.; Yang, L.; et al. Successful vitrification of in vivo embryos collected from superovulated Japanese Black cattle (Wagyu). Reprod. Domest. Anim. 2016, 51, 255–261. [Google Scholar] [CrossRef] [PubMed]
- American Embryo Transfer Association. Annual Survey of Members. Available online: https://www.aeta.org/survey.asp (accessed on 18 December 2021).
- Phillips, P.E.; Jahnke, M.M. Embryo transfer (techniques, donors, and recipients). Vet. Clin. N. Am. Food Anim. Pract. 2016, 32, 365–385. [Google Scholar] [CrossRef] [PubMed]
- Shull, J.W. Managing the problem beef embryo donor. In Proceedings; Society for Theriogenology: Montgomery, AL, USA, 2009; pp. 283–288. [Google Scholar]
- Mikkola, M.; Hasler, J.F.; Taponen, J. Factors affecting embryo production in superovulated Bos Taurus Cattle. Reprod. Fertil. Dev. 2019, 32, 104–124. [Google Scholar] [CrossRef]
- Zoda, A.; Urakawa, M.; Oono, Y.; Ogawa, S.; Satoh, M. Estimation of genetic parameters for superovulatory response traits in Japanese Black Cows. J. Anim. Sci. 2021, 99, skab265. [Google Scholar] [CrossRef]
- Watanabe, U.; Takagi, M.; Yamato, O.; Otoi, T.; Okamoto, K. Retrospective surveillance of metabolic parameters affecting reproductive performance of Japanese Black Breeding cows. J. Vet. Sci. 2014, 15, 283–288. [Google Scholar] [CrossRef]
- Sakatani, M.; Balboula, A.Z.; Yamanaka, K.; Takahashi, M. Effect of summer heat environment on body temperature, estrous cycles and blood antioxidant levels in Japanese Black cow. Anim. Sci. J. Nihon Chikusan Gakkaiho 2012, 83, 394–402. [Google Scholar] [CrossRef]
- Chebel, R.C.; Demétrio, D.G.B.; Metzger, J. Factors affecting success of embryo collection and transfer in large dairy herds. Theriogenology 2008, 69, 98–106. [Google Scholar] [CrossRef]
- Hansen, P.J.; Drost, M.; Rivera, R.M.; Paula-Lopes, F.F.; Al-Katanani, Y.M.; Krininger, C.E.; Chase, C.C. Adverse impact of heat stress on embryo production: Causes and strategies for mitigation. Theriogenology 2001, 55, 91–103. [Google Scholar] [CrossRef]
- Sartori, R.; Sartor-Bergfelt, R.; Mertens, S.A.; Guenther, J.N.; Parrish, J.J.; Wiltbank, M.C. Fertilization and early embryonic development in heifers and lactating cows in summer and lactating and dry cows in winter. J. Dairy Sci. 2002, 85, 2803–2812. [Google Scholar] [CrossRef]
- Roth, Z. Physiology and Endocrinology Symposium: Cellular and molecular mechanisms of heat stress related to bovine ovarian function. J. Anim. Sci. 2015, 93, 2034–2044. [Google Scholar] [CrossRef] [PubMed]
- De Rensis, F.; Lopez-Gatius, F.; García-Ispierto, I.; Morini, G.; Scaramuzzi, R.J. Causes of declining fertility in dairy cows during the warm season. Theriogenology 2017, 91, 145–153. [Google Scholar] [CrossRef] [PubMed]
- Takuma, T.; Sakai, S.; Ezoe, D.; Ichimaru, H.; Jinnouchi, T.; Kaedei, Y.; Nagai, T.; Otoi, T. Effects of season and reproductive phase on the quality, quantity and developmental competence of oocytes aspirated from Japanese Black cows. J. Reprod. Dev. 2010, 56, 55–59. [Google Scholar] [CrossRef] [PubMed]
- Kasimanickam, R.; Kasimanickam, V.; Kastelic, J.P.; Ramsey, K. Metabolic biomarkers, body condition, uterine inflammation and response to superovulation in lactating Holstein Cows. Theriogenology 2020, 146, 71–79. [Google Scholar] [CrossRef]
- Bender, R.W.; Hackbart, K.S.; Dresch, A.R.; Carvalho, P.D.; Vieira, L.M.; Crump, P.M.; Guenther, J.N.; Fricke, P.M.; Shaver, R.D.; Combs, D.K.; et al. Effects of acute feed restriction combined with targeted use of increasing luteinizing hormone content of follicle-stimulating hormone preparations on ovarian superstimulation, fertilization, and embryo quality in lactating dairy cows. J. Dairy Sci. 2014, 97, 764–778. [Google Scholar] [CrossRef] [PubMed]
- Yaakub, H.; O’Callaghan, D.; Boland, M.P. Effect of type and quantity of concentrates on superovulation and embryo yield in beef heifers. Theriogenology 1999, 51, 1259–1266. [Google Scholar] [CrossRef]
- Otomaru, K.; Shiga, H.; Kanome, J.; Yanagita, K. Blood biochemical values in Japanese Black Breeding cows in Kagoshima Prefecture, Japan. J. Vet. Med. Sci. 2015, 77, 1021–1023. [Google Scholar] [CrossRef]
- Bobe, G.; Young, J.W.; Beitz, D.C. Invited Review: Pathology, etiology, prevention, and treatment of fatty liver in dairy cows. J. Dairy Sci. 2004, 87, 3105–3124. [Google Scholar] [CrossRef]
- LeBlanc, S. Monitoring metabolic health of dairy cattle in the transition period. J. Reprod. Dev. 2010, 56, S29–S35. [Google Scholar] [CrossRef]
- Takahashi, M.; Sawada, K.; Kawate, N.; Inaba, T.; Tamada, H. Improvement of superovulatory response and pregnancy rate after transfer of embryos recovered from Japanese Black cows fed rumen bypass polyunsaturated fatty acids. J. Vet. Med. Sci. 2013, 75, 1485–1490. [Google Scholar] [CrossRef] [PubMed]
- Cerri, R.L.A.; Juchem, S.O.; Chebel, R.C.; Rutigliano, H.M.; Bruno, R.G.S.; Galvão, K.N.; Thatcher, W.W.; Santos, J.E.P. Effect of fat source differing in fatty acid profile on metabolic parameters, fertilization, and embryo quality in high-producing dairy cows. J. Dairy Sci. 2009, 92, 1520–1531. [Google Scholar] [CrossRef] [PubMed]
- Hussein, H.A.; Thurmann, J.-P.; Staufenbiel, R. 24-h variations of blood serum metabolites in high yielding dairy cows and calves. BMC Vet. Res. 2020, 16, 327. [Google Scholar] [CrossRef] [PubMed]
- Katoh, K.; Kouno, S.; Okazaki, A.; Suzuki, K.; Obara, Y. Interaction of GH polymorphism with body weight and endocrine functions in Japanese Black calves. Domest. Anim. Endocrinol. 2008, 34, 25–30. [Google Scholar] [CrossRef] [PubMed]
- Smith, G.D.; Jackson, L.M.; Foster, D.L. Leptin regulation of reproductive function and fertility. Theriogenology 2002, 57, 73–86. [Google Scholar] [CrossRef]
- Batista, A.M.; Gomes, W.A.; Carvalho, C.C.D.; Monteiro, P.L.J.; Silva, F.L.M.; Almeida, F.C.; Soares, P.C.; Carneiro, G.F.; Guerra, M.M.P. Effect of leptin on in vivo goat embryo production. Reprod. Domest. Anim. 2014, 49, 476–480. [Google Scholar] [CrossRef]
- Williams, G.L.; Amstalden, M.; Garcia, M.R.; Stanko, R.L.; Nizielski, S.E.; Morrison, C.D.; Keisler, D.H. Leptin and Its role in the central regulation of reproduction in cattle. Domest. Anim. Endocrinol. 2002, 23, 339–349. [Google Scholar] [CrossRef]
- Zieba, D.A.; Amstalden, M.; Williams, G.L. Regulatory roles of leptin in reproduction and metabolism: A comparative review. Domest. Anim. Endocrinol. 2005, 29, 166–185. [Google Scholar] [CrossRef]
- D’Occhio, M.J.; Baruselli, P.S.; Campanile, G. Influence of nutrition, body condition, and metabolic status on reproduction in female beef cattle: A review. Theriogenology 2019, 125, 277–284. [Google Scholar] [CrossRef]
- Nabenishi, H.; Sugino, F.; Konaka, R.; Yamazaki, A. Conception rate of Holstein and Japanese Black cattle following embryo transfer in southwestern Japan. Anim. Sci. J. Nihon Chikusan Gakkaiho 2018, 89, 1073–1078. [Google Scholar] [CrossRef]
- Rodrigues, M.; Bonotto, A.; Acosta, D.; Boligon, A.A.; Corrêa, M.N.; Brauner, C.C. Effect of oestrous synchrony between embryo donors and recipients, embryo quality and state on the pregnancy rate in beef cattle. Reprod. Domest. Anim. 2018, 53, 152–156. [Google Scholar] [CrossRef] [PubMed]
- Bó, G.A.; Cedeño, A.; Mapletoft, R.J. Strategies to increment in vivo and in vitro embryo production and transfer in cattle. Anim. Reprod. 2019, 16, 411–422. [Google Scholar] [CrossRef] [PubMed]
- Rocha, J.C.; Passalia, F.; Matos, F.D.; Maserati, M.P.; Alves, M.F.; de Almeida, T.G.; Cardoso, B.L.; Basso, A.C.; Nogueira, M.F.G. Methods for assessing the quality of mammalian embryos: How far we are from the gold standard? JBRA Assist. Reprod. 2016, 20, 150–158. [Google Scholar] [CrossRef] [PubMed]
- Hasler, J.F. The Holstein cow in embryo transfer today as compared to 20 years ago. Theriogenology 2006, 65, 4–16. [Google Scholar] [CrossRef] [PubMed]
- Gómez, E.; Muñoz, M.; Gatien, J.; Carrocera, S.; Martín-González, D.; Salvetti, P. Metabolomic identification of pregnancy-specific biomarkers in blood plasma of Bos Taurus beef cattle after transfer of in vitro produced embryos. J. Proteom. 2020, 225, 103883. [Google Scholar] [CrossRef] [PubMed]
- Forde, N.; Simintiras, C.A.; Sturmey, R.G.; Graf, A.; Wolf, E.; Blum, H.; Lonergan, P. Effect of lactation on conceptus-maternal interactions at the initiation of implantation in cattle: I. Effects on the conceptus transcriptome and amino acid composition of the uterine luminal fluid. Biol. Reprod. 2017, 97, 798–809. [Google Scholar] [CrossRef] [PubMed]
- Gomez, E.; Canela, N.; Herrero, P.; Cereto, A.; Gimeno, I.; Carrocera, S.; Martin-Gonzalez, D.; Murillo, A.; Muñoz, M. Metabolites secreted by bovine embryos in vitro predict pregnancies that the recipient plasma metabolome cannot, and vice versa. Metabolites 2021, 11, 162. [Google Scholar] [CrossRef]
| Year | Flushings Order | |||||||
|---|---|---|---|---|---|---|---|---|
| Metabolite | Median (Min–Max) | 2018 | 2019 | 2020 | p-Value | 1 to 4 | >4 | p-Value |
| BHB (mmol/L) | 0.42 (0.06–0.89) | 0.80 (0.29–0.89) | 0.37 (0.06–0.60) | 0.43 (0.28–0.76) | <0.0001 | |||
| NEFA (mmol/L) | 0.24 (0.06–0.77) | |||||||
| TC (mg/L) | 110.0 (64.0–181.0) | 114.0 (101.0–167.0) | 134.0 (67.50–181.0) | 86.50 (64.0–134.0) | <0.0001 | 103.60 (74.63–158.28) | 133, 25 (121, 30–141, 25) | 0.022 |
| HDL (mg/L) | 46.0 (19.0–84.0) | 55.90 (35.0– 65.70) | 54.00 (19.0– 84.0) | 32.50 (26.0–43.0) | <0.0001 | 42.65 (31.50–66.93) | 61.43 (52, 39–67, 36) | 0.002 |
| LDL (mg/L) | 14.0 (3.0–28.0) | |||||||
| GLU (mg/L) | 82.0 (60.0–154.0) | 76.0 (62.0–92.0) | 80.0 (60.0–93.0) | 89.50 (73.0– 154.0) | <0.0001 | 84.0 (73.0–107.50) | 71.69 (68, 25–75, 75) | <0.0001 |
| LAC (mg/L) | 19.60 (4.45–87.0) | 25.67 (11.27–30.48) | 17.0 (4.45–53.80) | 20.0 (7.0–87.0) | 0.047 | |||
| TG (mg/L) | 23.0 (14.0–62.0) | 23.0 (14.0–29.0) | 21.0 (14.0–43.0) | 36.0 (22.0–62.0) | <0.0001 | 26.50 (14.75–45.0) | 24.09 (21, 13–27, 31) | 0.033 |
| UR (mg/dL) | 24.0 (11.0–41.0) | |||||||
| FRU (mg/L) | 275.0 (200.0–508.0) | |||||||
| LEP (ng/mL) | 0.19 (0.01–2.28) | 0.11 (0.06–0.27) | 0.84 (0.06–1.48) | 0.08 (0.01–0.39) | <0.0001 | 0.29 (0.08–2.28) | 0.74 (0, 54–1, 09) | 0.016 |
| CR (%; n/N) | p-Value | OR | 95% CI | ||
|---|---|---|---|---|---|
| Recipients’ Parity | Heifers | 44.3% (148/337) | |||
| Primiparous | 58.3% (67/115) | ||||
| Multiparous | 58.8% (118/235) | ||||
| ET number | 1 | 47.0% (235/499) | |||
| >1 | 52.1% (98/188) | ||||
| Heat Stress at ET | Yes | 44.3% (47/106) | 0.042 | 2.06 | 1.026–4.153 |
| No | 49.2% (286/581) |
| Metabolite | Recipient’s Outcome | Concentration Median (Min–Max) | p-Value |
|---|---|---|---|
| BHB (mmol/L) | Pregnant | 0.38 (0.10–0.58) | 0.32 |
| Non-pregnant | 0.39 (0.21–47.0) | ||
| NEFA (mmol/L) | Pregnant | 0.19 (0.02–0.97) | 0.89 |
| Non-pregnant | 0.18 (0.02–0.86) | ||
| TC (mg/L) | Pregnant | 129.0 (53.0–336.0) | 0.025 |
| Non-pregnant | 138.0 (68.0–353.0) | ||
| HDL (mg/L) | Pregnant | 51.0 (25.0–87.50) | 0.33 |
| Non-pregnant | 53.0 (26.0–88.0) | ||
| LDL (mg/L) | Pregnant | 17.0 (4.0–43.0) | 0.031 |
| Non-pregnant | 20.0 (2.50–40.0) | ||
| GLU (mg/L) | Pregnant | 78.0 (41.0–106.0) | 0.005 |
| Non-pregnant | 83.0 (47.50–127.0) | ||
| LAC (mg/L) | Pregnant | 14.0 (3.50–53.0) | 0.93 |
| Non-pregnant | 14.0 (3.0–73.0) | ||
| TG (mg/L) | Pregnant | 23.25 (10.0–118.0) | 0.113 |
| Non-pregnant | 21.0 (7.50–83.0) | ||
| UR (mg/dL) | Pregnant | 19.0 (7.0–49.0) | 0.024 |
| Non-pregnant | 22.0 (2.0–49.0) | ||
| FRU (mg/L) | Pregnant | 309.5 (197.0–398.0) | 0.158 |
| Non-pregnant | 311.0 (246.0–415.0) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vázquez-Mosquera, J.M.; Fernández-Novo, A.; Bonet-Bo, M.; Pérez-Villalobos, N.; Pesántez-Pacheco, J.L.; Pérez-Solana, M.L.; de Mercado, E.; Gardón, J.C.; Villagrá, A.; Sebastián, F.; et al. MOET Efficiency in a Spanish Herd of Japanese Black Heifers and Analysis of Environmental and Metabolic Determinants. Biology 2022, 11, 225. https://doi.org/10.3390/biology11020225
Vázquez-Mosquera JM, Fernández-Novo A, Bonet-Bo M, Pérez-Villalobos N, Pesántez-Pacheco JL, Pérez-Solana ML, de Mercado E, Gardón JC, Villagrá A, Sebastián F, et al. MOET Efficiency in a Spanish Herd of Japanese Black Heifers and Analysis of Environmental and Metabolic Determinants. Biology. 2022; 11(2):225. https://doi.org/10.3390/biology11020225
Chicago/Turabian StyleVázquez-Mosquera, Juan M., Aitor Fernández-Novo, Martin Bonet-Bo, Natividad Pérez-Villalobos, Jose L. Pesántez-Pacheco, Maria Luz Pérez-Solana, Eduardo de Mercado, Juan Carlos Gardón, Arantxa Villagrá, Francisco Sebastián, and et al. 2022. "MOET Efficiency in a Spanish Herd of Japanese Black Heifers and Analysis of Environmental and Metabolic Determinants" Biology 11, no. 2: 225. https://doi.org/10.3390/biology11020225
APA StyleVázquez-Mosquera, J. M., Fernández-Novo, A., Bonet-Bo, M., Pérez-Villalobos, N., Pesántez-Pacheco, J. L., Pérez-Solana, M. L., de Mercado, E., Gardón, J. C., Villagrá, A., Sebastián, F., Pérez-Garnelo, S. S., Martínez, D., & Astiz, S. (2022). MOET Efficiency in a Spanish Herd of Japanese Black Heifers and Analysis of Environmental and Metabolic Determinants. Biology, 11(2), 225. https://doi.org/10.3390/biology11020225






