Reproductive Traits Demonstrate How Well the Mediterranean Stripe-Necked Turtle Mauremys leprosa Can Flourish under Highly Degraded–Polluted Conditions
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Field Procedures and Data Collection
2.3. Statistical Analyses
3. Results
3.1. Body Size of Gravid Females
3.2. Clutch Size
3.3. Egg Sizes in Studied Populations
3.4. Correlation of Clutch Size with Body Size of Gravid Females
3.5. Relationships between Body Size of Gravid Females, Clutch Size, and Egg Size
4. Discussion
4.1. Body Size of Gravid Females
4.2. Clutch Size
4.3. Relationships between Female Body Size, Clutch and Egg Size
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lovich, J.E.; Znari, M.; Baamrane, M.A.A.; Naimi, M.; Mostalih, A. Biphasic Geographic Variation in Sexual Size Dimorphism of Turtle (Mauremys leprosa) Populations along an Environmental Gradient in Morocco. Chelonian Conserv. Biol. 2010, 9, 45–53. [Google Scholar] [CrossRef]
- Tiar-Saadi, M. Bioécologie des Tortues d’eau en Algérie. Ph.D. Thesis, Département de biologie, Université Badji Mokhtar, Annaba, Algeria, 2018. [Google Scholar]
- Attia El Hili, R.; Verneau, O.; Jrijer, J.; Achouri, M. Reassessment of Distribution and Conservation Status of Freshwater Turtles (Testudines) in Tunisia. Salamandra 2020, 56, 362–372. [Google Scholar]
- Toledo, M.A.; de la Concha, A.; Rubio, J.L. Space Use, Distribution and Conservation of the Spanish Pond Turtle (Mauremys leprosa Schewigger, 1812). A Review (Chordata, Geoemydidae). Soc. Esp. Hist. Nat. 2021, 115, 95–110. [Google Scholar]
- Iverson, J.B. A Checklist with Distribution Maps of the Turtles of the World; Erlham College: Richmond, Indiana, 1986; p. 283. [Google Scholar]
- Escoriza, D.; Hassine, J.B. Niche Separation among North-West African Semi-Aquatic Reptiles. Hydrobiologia 2017, 797, 47–56. [Google Scholar] [CrossRef]
- Schleich, H.-H.; Kästle, W.; Kabisch, K. Amphibians and Reptiles of North Africa; Koeltz Scientific Books: Koenigstein Germany, 1996; Volume 63. [Google Scholar]
- Ernst, C.H.; Barbour, R.W.; Altenburg, R.G.M. Turtles of the World; Smithonian Institution Press: Washington, DC, USA, 1989; Volume 272, p. 313. [Google Scholar]
- Franch, M.; Montori, A.; Sillero, N.; Llorente, G.A. Temporal Analysis of Mauremys leprosa (Testudines, Geoemydidae) Distribution in Northeastern Iberia: Unusual Increase in the Distribution of a Native Species. Hydrobiologia 2015, 757, 129–142. [Google Scholar] [CrossRef]
- Courmont, L.; De Sousa, L. Plan National d’actions en Faveur de l’Emyde lépreuse Mauremys leprosa 2012–2016; Ministère de l’Écologie, du Développement Durable, des Transports et du Logement: Paris, France, 2011; p. 105. [Google Scholar]
- Panzeri, M.; Mori, E.; Mazza, G.; Menchetti, M. Records of Introduced Stripe-Necked Terrapins (Mauremys Species) in Italy. Acta Herpetol. 2014, 9, 227–230. [Google Scholar]
- Pinya, S.; Parpal, L.L.; Sunyer, J.R. Sobre La Presència de Tortugues d’aigua Al· Lòctones d’introducció Recent a l’illa de Mallorca. On the Presence of Recent Introduced Alien Fresh Water Tortoises in the Mallorca Island. Bolletí Soc. Història Nat. Balears 2007, 50, 209–216. [Google Scholar]
- Bakhouche, B.; Tiar, G.; Escoriza, D.; Imed, D. Phenology and Population Structure of the Mediterranean Stripe-Necked Terrapin Mauremys leprosa (Schweigger, 1812) in the Reghaia a Lake (Northern Algeria). Basic Appl. Herpetol. 2019, 33, 43–51. [Google Scholar] [CrossRef]
- El Hassani, M.S.; El Mouden, E.H.; Slimani, T.; Bonnet, X. Morphological and Physiological Assessments Reveal That Freshwater Turtle (Mauremys leprosa) Can Flourish under Extremely Degraded-Polluted Conditions. Chemosphere 2019, 220, 432–441. [Google Scholar] [CrossRef]
- Slimani, T.; El Hassani, M.S.; El Mouden, E.H.; Bonnet, M.; Bustamante, P.; Brischoux, F.; Brault-Favrou, M.; Bonnet, X. Large-Scale Geographic Patterns of Mercury Contamination in Morocco Revealed by Freshwater Turtles. Environ. Sci. Pollut. Res. 2018, 25, 2350–2360. [Google Scholar] [CrossRef]
- Combescot, C.H. Sexualité et Cycle Génital de La Tortue d’eau Algérienne, Emys leprosa Schw. Bull. Société D’Histoire Nat. L’Afrique Nord 1954, 45, 366–377. [Google Scholar]
- Naimi, M.; Znari, M.; Lovich, J.E.; Feddadi, Y.; Baamrane, M.A.A. Clutch and Egg Allometry of the Turtle Mauremys leprosa (Chelonia: Geoemydidae) from a Polluted Peri-Urban River in West-Central Morocco. Herpetol. J. 2012, 22, 43–49. [Google Scholar]
- Andreu, A.C.; Villamor, C. Calendario Reproductivo y Tamaño de Puesta En El Galápago Leproso, Mauremys leprosa (Schweigger, 1812) En Doñana, Huelva. Doñana. Acta Vertebr 1989, 16, 167–172. [Google Scholar]
- Da Silva, E. Notes on Clutch Size and Egg Size of Mauremys leprosa from Spain. J. Herpetol. 1995, 29, 484–485. [Google Scholar] [CrossRef]
- Díaz-Paniagua, C.; Andreu, A.C.; Marco, A.; Nuez, M.; Hidalgo-Vila, J.; Perez-Santigosa, N. Data on Nesting, Incubation, and Hatchling Emergence in the Two Native Aquatic Turtle Species (Emys orbicularis and Mauremys leprosa) from Doñana National Park. Basic Appl. Herpetol. 2014, 28, 147–151. [Google Scholar] [CrossRef][Green Version]
- Keller, C. Assessment of Reproductive State in the Turtle Mauremys leprosa: A Comparison between Inguinal Palpation and Radiography. Wildl. Res. 1998, 25, 527–531. [Google Scholar] [CrossRef]
- Maran, J. L’émyde Lépreuse Mauremys leprosa (Schweigger, 1812). Communauté D’Intérêts Pour Tortues En Suisse Bull. 1996, 7, 16–42. [Google Scholar]
- Pérez, M.; Collado, E.; Ramo, C. Crecimiento de Mauremys caspica leprosa (Schweigger, 1812)(Reptilia, Testudines) En La Reserva Biologica de Donana. Doñana Acta Vertebr. 1979, 6, 161–178. [Google Scholar]
- Perez-Quintero, J.C. Primeros Datos Sobre Oologia de Reptiles de La Provincia de Huelva. In Proceedings of the Comunic. IV Congreso Nacional de Herpetologia, Madrid, Spain, 6–9 December 1989. [Google Scholar]
- Segurado, P.; Ayres, C.; Cordero, A. L’Emyde Lépreuse Mauremys leprosa Dans La Péninsule Ibérique. Manouria 2005, 8, 26–27. [Google Scholar]
- Loulida, S.; Naimi, M.; Znari, M. Stress Environnemental et Instabilité Du Développement de l’émyde Lépreuse Mauremys leprosa (Schweigger, 1812) Dans Des Environnements Contrastés En Termes de Pollution Dans Le Bassin Du Tensift, Centre Ouest Du Maroc. J. Int. Sci. Tech. L’Eau L’Environnement 2018, 3, 104–108. [Google Scholar]
- Martínez-López, E.; Gómez-Ramírez, P.; Espín, S.; Aldeguer, M.P.; García-Fernández, A.J. Influence of a Former Mining Area in the Heavy Metals Concentrations in Blood of Free-Living Mediterranean Pond Turtles (Mauremys leprosa). Bull. Environ. Contam. Toxicol. 2017, 99, 167–172. [Google Scholar] [CrossRef] [PubMed]
- Héritier, L.; Meistertzheim, A.-L.; Verneau, O. Oxidative Stress Biomarkers in the Mediterranean Pond Turtle (Mauremys leprosa) Reveal Contrasted Aquatic Environments in Southern France. Chemosphere 2017, 183, 332–338. [Google Scholar] [CrossRef] [PubMed]
- Gibbons, J.W. Reproductive Dynamics of a Turtle (Pseudemys scripta) Population in a Reservoir Receiving Heated Effluent from a Nuclear Reactor. Can. J. Zool. 1970, 48, 881–885. [Google Scholar] [CrossRef]
- Bergeron, J.M.; Crews, D.; McLachlan, J.A. PCBs as Environmental Estrogens: Turtle Sex Determination as a Biomarker of Environmental Contamination. Environ. Health Perspect. 1994, 102, 780–781. [Google Scholar] [CrossRef]
- Lamb, T.; Bickham, J.W.; Lyne, T.B.; Gibbons, J.W. The Slider Turtle as an Environmental Sentinel: Multiple Tissue Assays Using Flow Cytometric Analysis. Ecotoxicology 1995, 4, 5–13. [Google Scholar]
- Guillette, L.J., Jr.; Crain, D.A. Endocrine-Disrupting Contaminants and Reproductive Abnormalities in Reptiles. Comments Toxicol. 1996, 5, 381–398. [Google Scholar]
- Gibbons, J.W.; Scott, D.E.; Ryan, T.J.; Buhlmann, K.A.; Tuberville, T.D.; Metts, B.S.; Greene, J.L.; Mills, T.; Leiden, Y.; Poppy, S. The Global Decline of Reptiles, Déjà Vu Amphibians: Reptile Species Are Declining on a Global Scale. Six Significant Threats to Reptile Populations Are Habitat Loss and Degradation, Introduced Invasive Species, Environmental Pollution, Disease, Unsustainable Use, and Global Climate Change. BioScience 2000, 50, 653–666. [Google Scholar] [CrossRef]
- Tiar-Saadi, M.; Tiar, G.; Bouslama, Z.; Širokỳ, P. First Data on the Population of the European Pond Turtle Emys orbicularis at Lake Tonga, El Kala National Park, Algeria. Biologia (Bratisl.) 2017, 72, 819–824. [Google Scholar] [CrossRef]
- Ernst, C.H.; Hershey, M.F.; Barbour, R.W. A New Coding System for Hardshelled Turtles. Trans. Ky. Acad. Sci. 1974, 35, 27–28. [Google Scholar]
- Zuffi, M.A.L.; Odetti, F.; Meozzi, P. Body Size and Clutch Size in the European Pond Turtle (Emys orbicularis) from Central Italy. J. Zool. 1999, 247, 139–143. [Google Scholar] [CrossRef]
- Germano, D.J. Ecology of Western Pond Turtles (Actinemys marmorata) at Sewage-Treatment Facilities in the San Joaquin Valley, California. Southwest. Nat. 2010, 55, 89–97. [Google Scholar] [CrossRef]
- Lindeman, P.V. Comparative Life History of Painted Turtles (Chrysemys picta) in Two Habitats in the Inland Pacific Northwest. Copeia 1996, 1, 114–130. [Google Scholar] [CrossRef]
- Polo-Cavia, N.; Engstrom, T.; López, P.; Martín, J. Body Condition Does Not Predict Immunocompetence of Western Pond Turtles in Altered versus Natural Habitats. Anim. Conserv. 2010, 13, 256–264. [Google Scholar] [CrossRef]
- Sidis, I.; Gasith, A. Food Habits of the Caspian Terrapin (Mauremys caspica rivulata) in Unpolluted and Polluted Habitats in Israel. J. Herpetol. 1985, 19, 108–115. [Google Scholar] [CrossRef]
- Souza, F.L.; Abe, A.S. Feeding Ecology, Density and Biomass of the Freshwater Turtle, Phrynops geoffroanus, Inhabiting a Polluted Urban River in South-Eastern Brazil. J. Zool. 2000, 252, 437–446. [Google Scholar] [CrossRef]
- Fritz, U.; Barata, M.; Busack, S.D.; Fritzsch, G.; Castilho, R. Impact of Mountain Chains, Sea Straits and Peripheral Populations on Genetic and Taxonomic Structure of a Freshwater Turtle, Mauremys leprosa (Reptilia, Testudines, Geoemydidae). Zool. Scr. 2006, 35, 97–108. [Google Scholar] [CrossRef]
- Veríssimo, J.; Znari, M.; Stuckas, H.; Fritz, U.; Pereira, P.; Teixeira, J.; Arculeo, M.; Marrone, F.; Sacco, F.; Naimi, M. Pleistocene Diversification in Morocco and Recent Demographic Expansion in the Mediterranean Pond Turtle Mauremys leprosa. Biol. J. Linn. Soc. 2016, 119, 943–959. [Google Scholar] [CrossRef]
- Rhodin, A.G.J.; Iverson, J.B.; Bour, R.; Fritz, U.; Georges, A.; Shaffer, H.B.; van Dijk, P.P.; Turtle Taxonomy Working Group. Turtles of the World: Annotated Checklist and Atlas of Taxonomy, Synonymy, Distribution, and Conservation Status, 9th ed.; Rhodin, A.G.J., Iverson, J.B., van Dijk, P.P., Stanford, C.B., Goode, E.V., Buhlmann, K.A., Mittermeier, R.A., Eds.; Conservation Biology of Freshwater Turtles and Tortoises: A Compilation Project of the IUCN/SSC Tortoise and Freshwater Turtle Specialist Group. Chelonian Research Monographs; Species Survival Commission: Gland, Switzerland, 2021; Volume 8, pp. 1–472. Available online: https://iucn-tftsg.org/checklist/ (accessed on 19 October 2022).
- Bonnet, X.; Delmas, V.; El-Mouden, H.; Slimani, T.; Sterijovski, B.; Kuchling, G. Is Sexual Body Shape Dimorphism Consistent in Aquatic and Terrestrial Chelonians? Zoology 2010, 113, 213–220. [Google Scholar] [CrossRef]
- Tiar-Saadi, M.; Tiar, G.; Bouslama, Z.; Širokỳ, P. Mechanisms Determining Body Size and Shape Difference in Algerian Spur-Thighed Tortoises (Testudo graeca). Animals 2022, 12, 1330. [Google Scholar] [CrossRef]
- Willemsen, R.E.; Hailey, A. Variation of Adult Body Size of the Tortoise Testudo Hermanni in Greece: Proximate and Ultimate Causes. J. Zool. 1999, 248, 379–396. [Google Scholar] [CrossRef]
- Forsman, A. Variation in Sexual Size Dimorphism and Maximum Body Size among Adder Populations: Effects of Prey Size. J. Anim. Ecol. 1991, 60, 253–267. [Google Scholar] [CrossRef]
- Jorgewich-Cohen, G.; Henrique, R.S.; Dias, P.H.; Sánchez-Villagra, M.R. The Evolution of Reproductive Strategies in Turtles. PeerJ 2022, 10, e13014. [Google Scholar] [CrossRef] [PubMed]
- Bayly, I.A.E. Variation in Sexual Dimorphism in Nonmarine Calanoid Copepods and Its Ecological Significance. Limnol. Oceanogr. 1978, 23, 1224–1228. [Google Scholar] [CrossRef]
- Gasith, A.; Sidis, I. Polluted Water Bodies, the Main Habitat of the Caspian Terrapin (Mauremys caspica rivulata) in Israel. Copeia 1984, 1984, 216–219. [Google Scholar] [CrossRef]
- Díaz-Paniagua, C.; Pérez-Santigosa, N.; Hidalgo-Vila, J.; Florencio, M. Does the Exotic Invader Turtle, Trachemys scripta elegans, Compete for Food with Coexisting Native Turtles? Amphib.-Reptil. 2011, 32, 167–175. [Google Scholar] [CrossRef]
- Bishop, C.A.; Ng, P.; Pettit, K.E.; Kennedy, S.W.; Stegeman, J.J.; Norstrom, R.J.; Brooks, R.J. Environmental Contamination and Developmental Abnormalities in Eggs and Hatchlings of the Common Snapping Turtle (Chelydra serpentina serpentina) from the Great Lakes—St Lawrence River Basin (1989–1991). Environ. Pollut. 1998, 101, 143–156. [Google Scholar] [CrossRef]
- Bishop, C.A.; Brown, G.P.; Brooks, R.J.; Lean, D.R.S.; Carey, J.H. Organochlorine Contaminant Concentrations in Eggs and Their Relationship to Body Size, and Clutch Characteristics of the Female Common Snapping Turtle (Chelydra serpentina serpentina) in Lake Ontario, Canada. Arch. Environ. Contam. Toxicol. 1994, 27, 82–87. [Google Scholar] [CrossRef]
- Bonin, J.; DesGranges, J.-L.; Bishop, C.A.; Rodrigue, J.; Gendron, A.; Elliott, J.E. Comparative Study of Contaminants in the Mudpuppy (Amphibia) and the Common Snapping Turtle (Reptilia), St. Lawrence River, Canada. Arch. Environ. Contam. Toxicol. 1995, 28, 184–194. [Google Scholar] [CrossRef]
- Holliday, D.K.; Roosenburg, W.M.; Elskus, A.A. Spatial Variation in Polycyclic Aromatic Hydrocarbon Concentrations in Eggs of Diamondback Terrapins, Malaclemys terrapin, from the Patuxent River, Maryland. Bull. Environ. Contam. Toxicol. 2008, 80, 119–122. [Google Scholar] [CrossRef][Green Version]
- Berry, J.F.; Shine, R. Sexual Size Dimorphism and Sexual Selection in Turtles (Order Testudines). Oecologia 1980, 44, 185–191. [Google Scholar] [CrossRef]
- Wilbur, H.M.; Morin, P.J. Life History Evolution in Turtles. Biol. Reptil. 1988, 16, 387–439. [Google Scholar]
- Gibbons, J.W.; Greene, J.L.; Patterson, K.K. Variation in Reproductive Characteristics of Aquatic Turtles. Copeia 1982, 1982, 776–784. [Google Scholar] [CrossRef]
- Janzen, F.J.; Warner, D.A. Parent-Offspring Conflict and Selection on Egg Size in Turtles. J. Evol. Biol. 2009, 22, 2222–2230. [Google Scholar] [CrossRef] [PubMed]
- Packard, G.C.; Packard, M.J. The Physiological Ecology of Reptilian Eggs and Embryos. In Biology of the Reptilia; Gans, C., Huey, R.B., Eds.; Alan R. Liss.: New York, NY, USA, 1988; Volume 16, pp. 523–605. [Google Scholar]
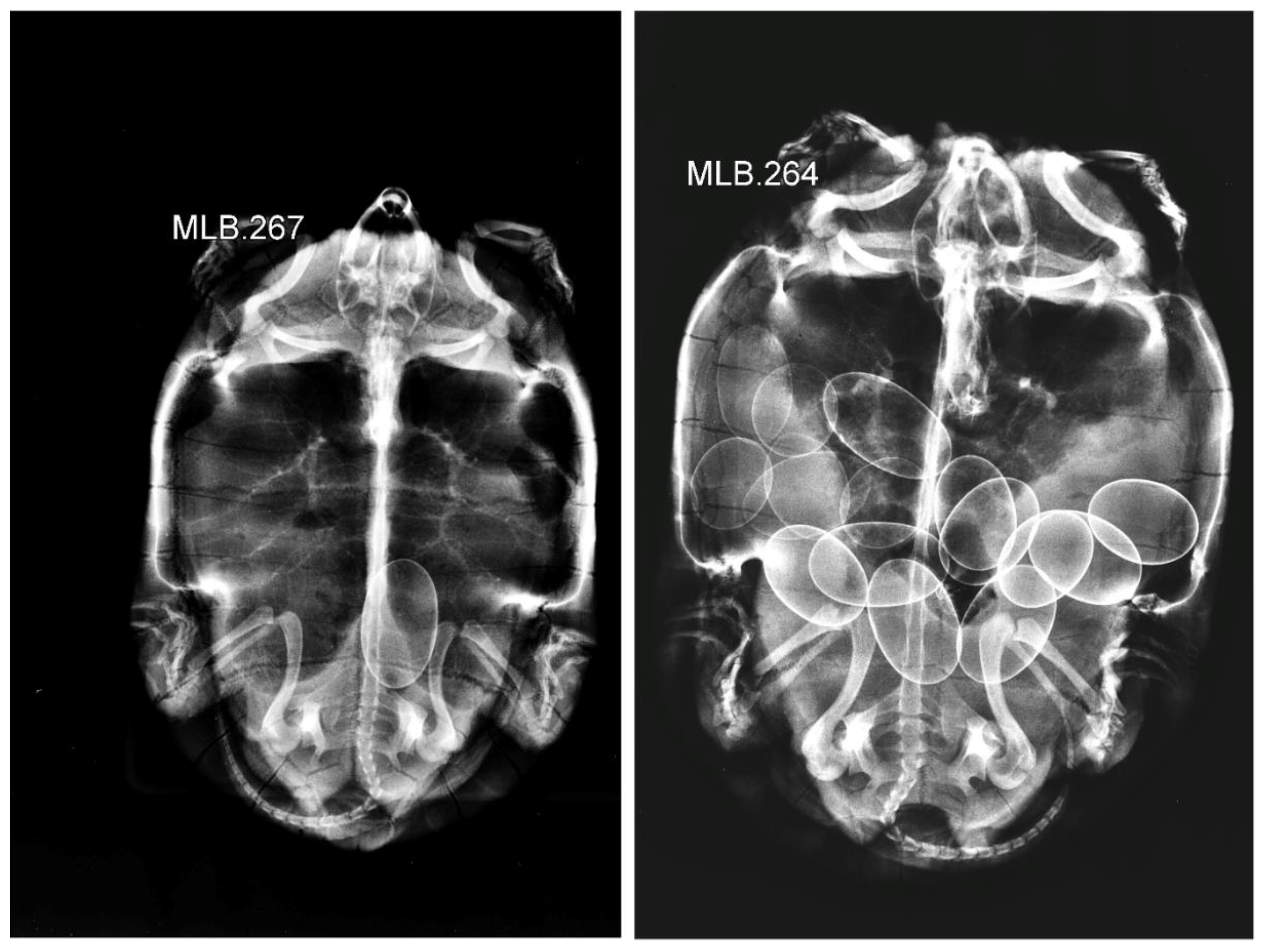
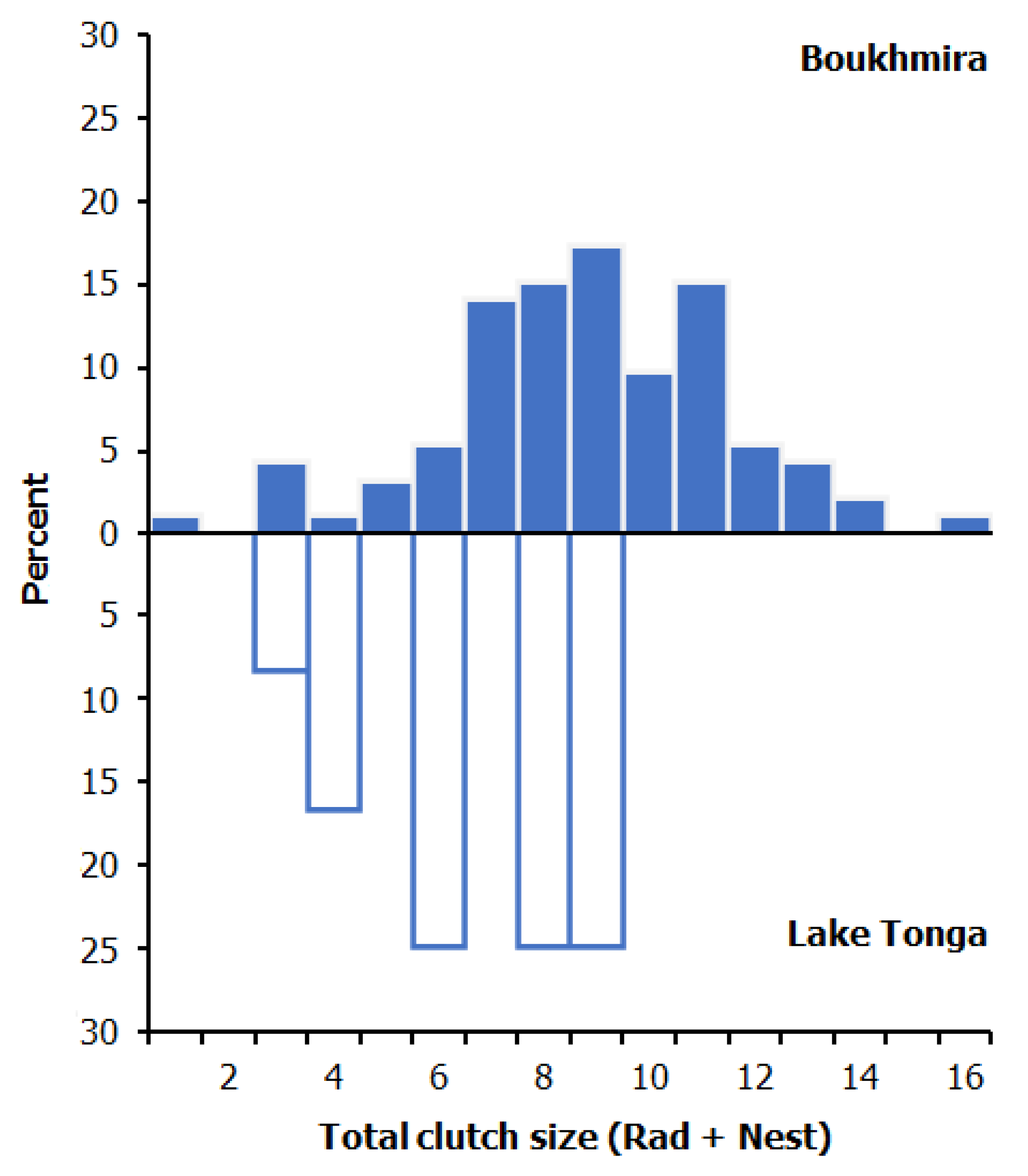


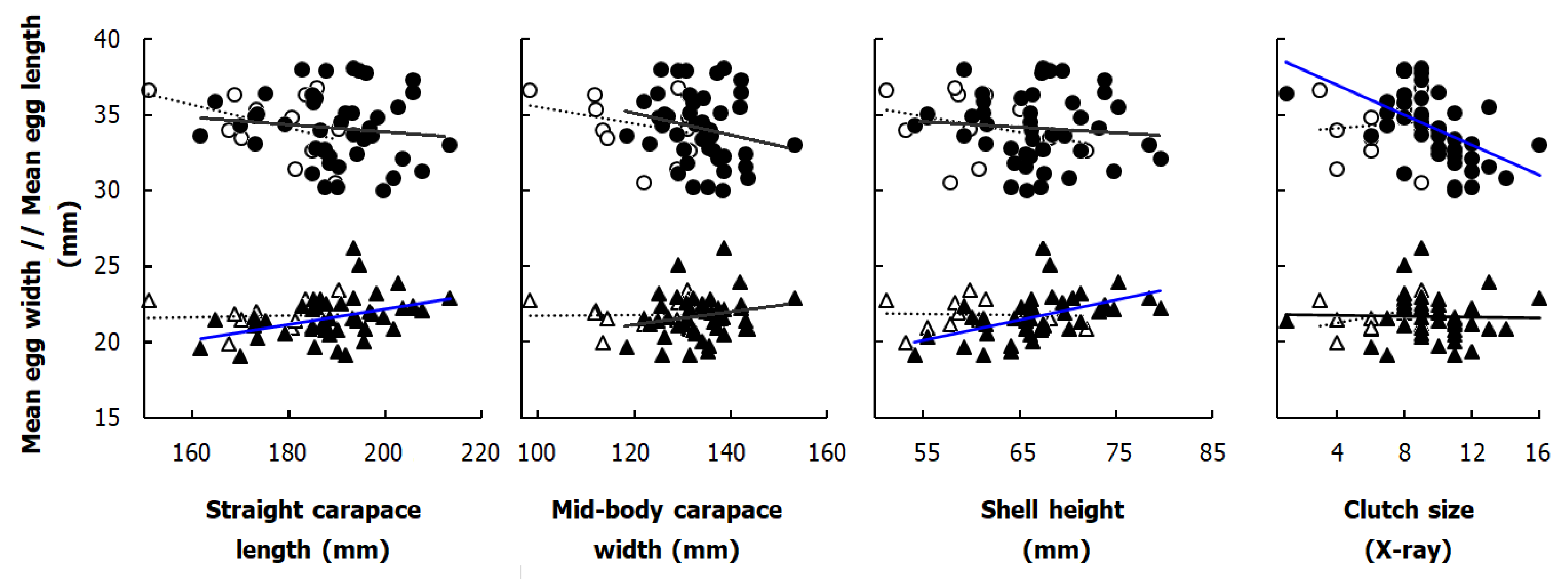
| Scheme. | Boukhmira | Lake Tonga | |||||
|---|---|---|---|---|---|---|---|
| Var. (mm) | Mean ± SD (n = 43) | Smallest Gravid Size (mm) | Largest Gravid Size (mm) | Mean ± SD (n = 12) | Smallest Gravid Size (mm) | Largest Gravid Size (mm) | ANOVA Test |
| SCL | 189.47 ± 11.41 | 161.70 | 213.40 | 177.20 ± 11.45 | 150.90 | 190.30 | F(1, 53) = 10.85, p < 0.01 |
| MCW | 133.74 ± 6.89 | 118.40 | 153.30 | 121.26 ± 10.97 | 98.30 | 131.40 | F(1, 53) = 23.36, p < 0.001 |
| SH | 66.80 ± 5.54 | 54.10 | 79.60 | 60.15 ± 5.97 | 51.20 | 71.90 | F(1, 53) = 13.05, p < 0.001 |
| Site | Boukhmira | Lake Tonga | ||||
|---|---|---|---|---|---|---|
| Var. | Mean ± SD | Range | n | Mean ± SD | Range | n |
| N-CS | 8.00 ± 2.68 | 3–14 | 49 | - | - | - |
| RAD-CS | 9.70 ± 2.45 | 1–16 | 43 | 6.67 ± 2.15 | 3–9 | 12 |
| Total-CS | 8.79 ± 2.70 | 1–16 | 92 | 6.67 ± 2.15 | 3–9 | 12 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gahmous, S.A.; Tiar, G.; Tiar-Saadi, M.; Bouslama, Z.; Široký, P. Reproductive Traits Demonstrate How Well the Mediterranean Stripe-Necked Turtle Mauremys leprosa Can Flourish under Highly Degraded–Polluted Conditions. Biology 2022, 11, 1562. https://doi.org/10.3390/biology11111562
Gahmous SA, Tiar G, Tiar-Saadi M, Bouslama Z, Široký P. Reproductive Traits Demonstrate How Well the Mediterranean Stripe-Necked Turtle Mauremys leprosa Can Flourish under Highly Degraded–Polluted Conditions. Biology. 2022; 11(11):1562. https://doi.org/10.3390/biology11111562
Chicago/Turabian StyleGahmous, Sami Aymen, Ghoulem Tiar, Manel Tiar-Saadi, Zihad Bouslama, and Pavel Široký. 2022. "Reproductive Traits Demonstrate How Well the Mediterranean Stripe-Necked Turtle Mauremys leprosa Can Flourish under Highly Degraded–Polluted Conditions" Biology 11, no. 11: 1562. https://doi.org/10.3390/biology11111562
APA StyleGahmous, S. A., Tiar, G., Tiar-Saadi, M., Bouslama, Z., & Široký, P. (2022). Reproductive Traits Demonstrate How Well the Mediterranean Stripe-Necked Turtle Mauremys leprosa Can Flourish under Highly Degraded–Polluted Conditions. Biology, 11(11), 1562. https://doi.org/10.3390/biology11111562





