Projecting Future Climate Change-Mediated Impacts in Three Paralytic Shellfish Toxins-Producing Dinoflagellate Species
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Data Collection and Curation
2.2. Predictor Variables
2.3. Modeling
2.4. Post-Analysis
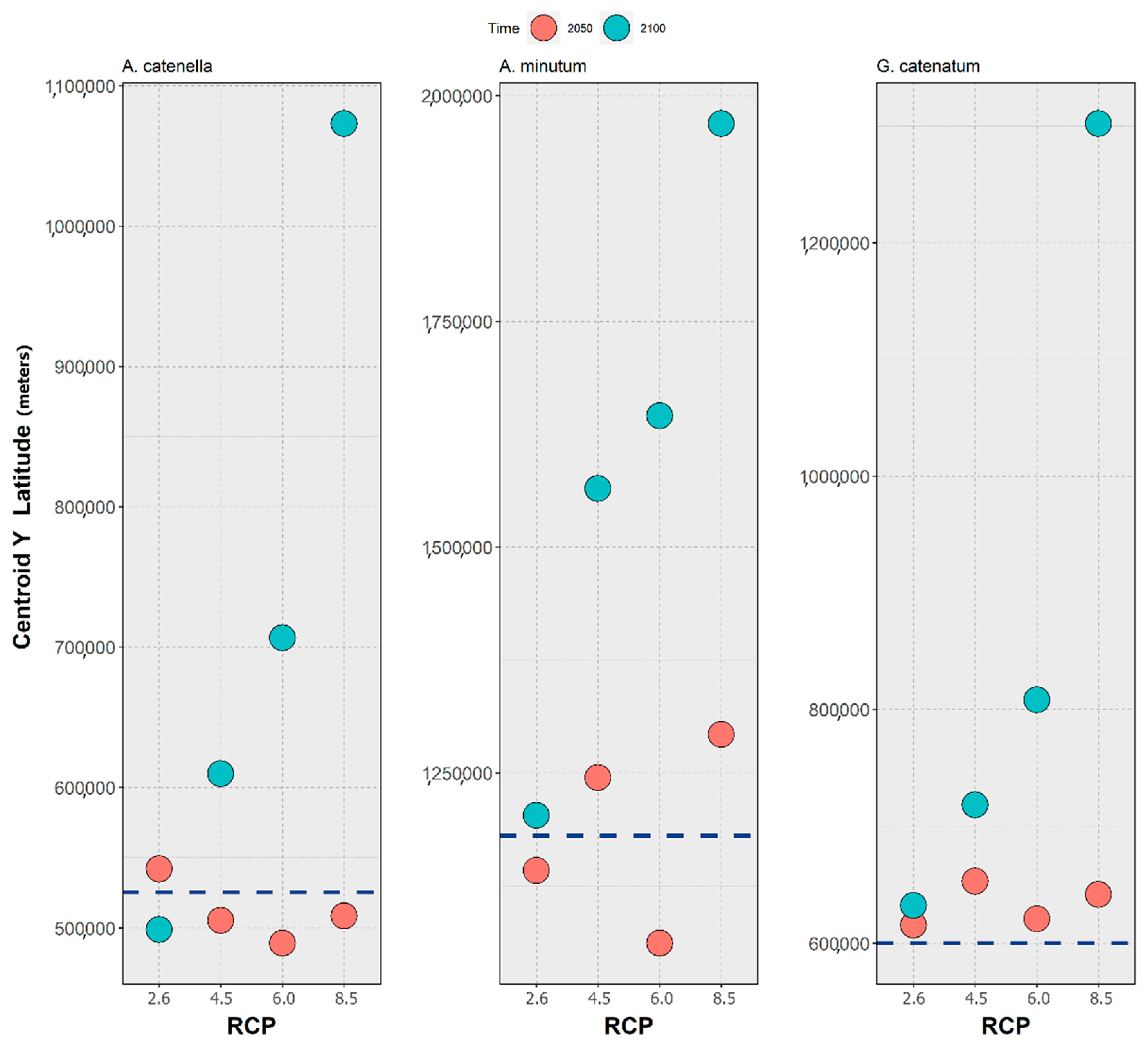
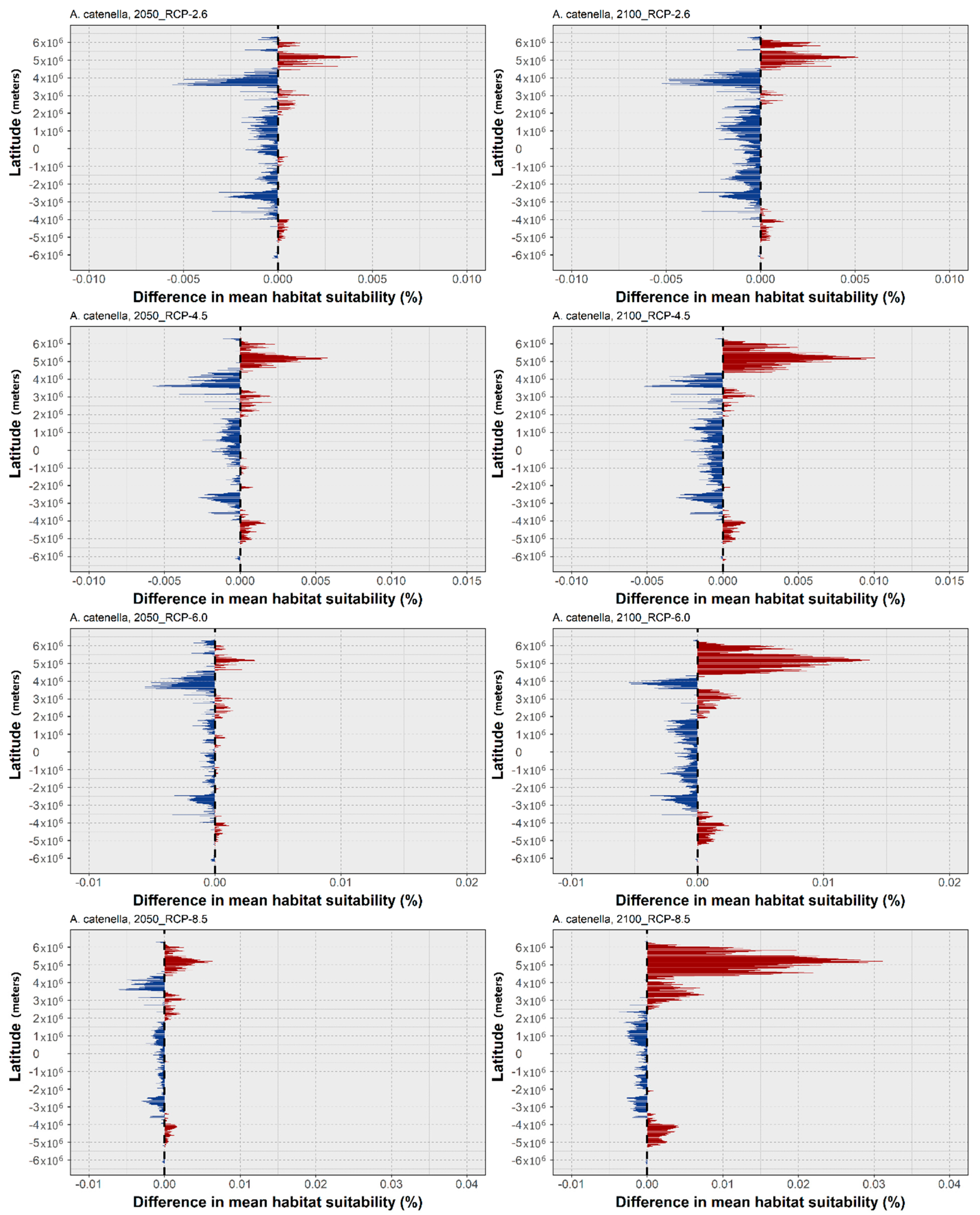
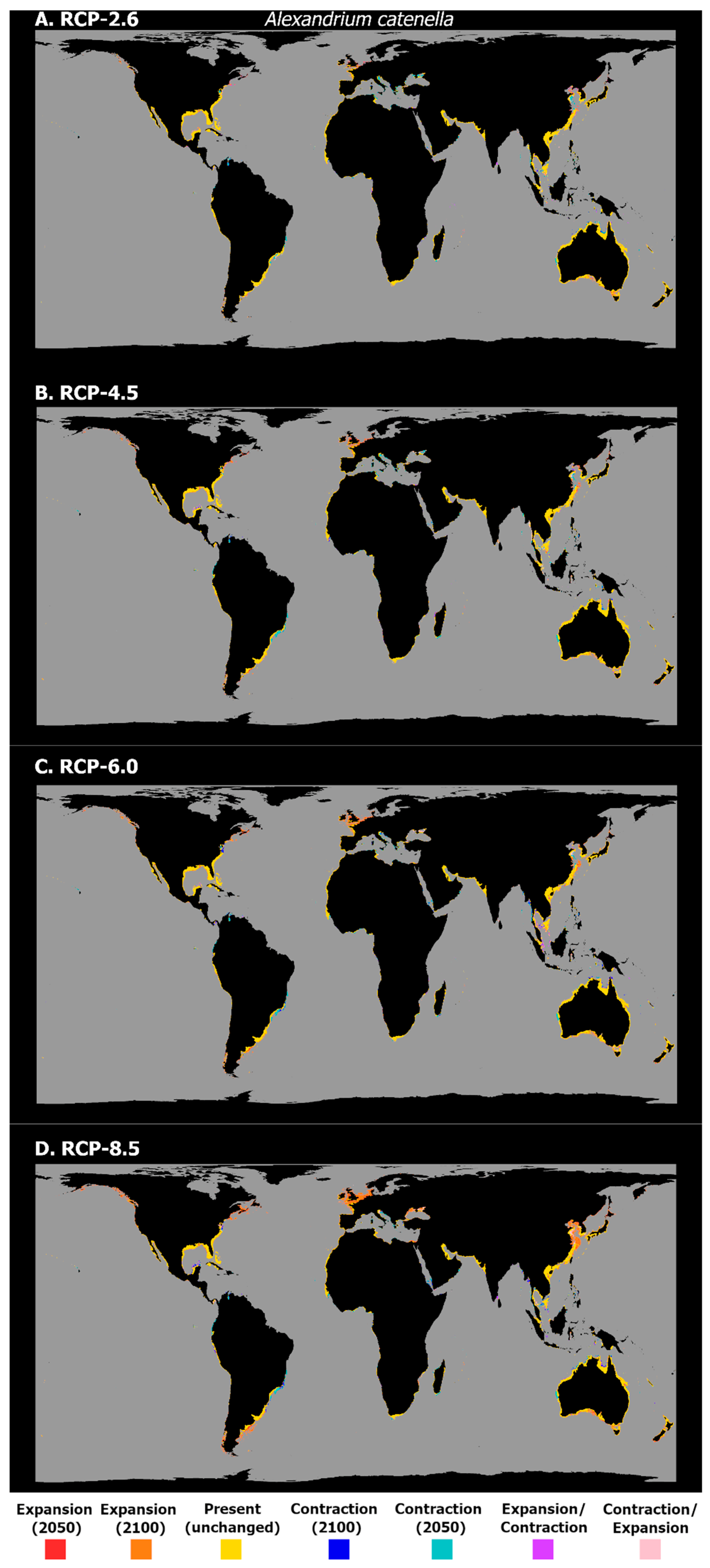
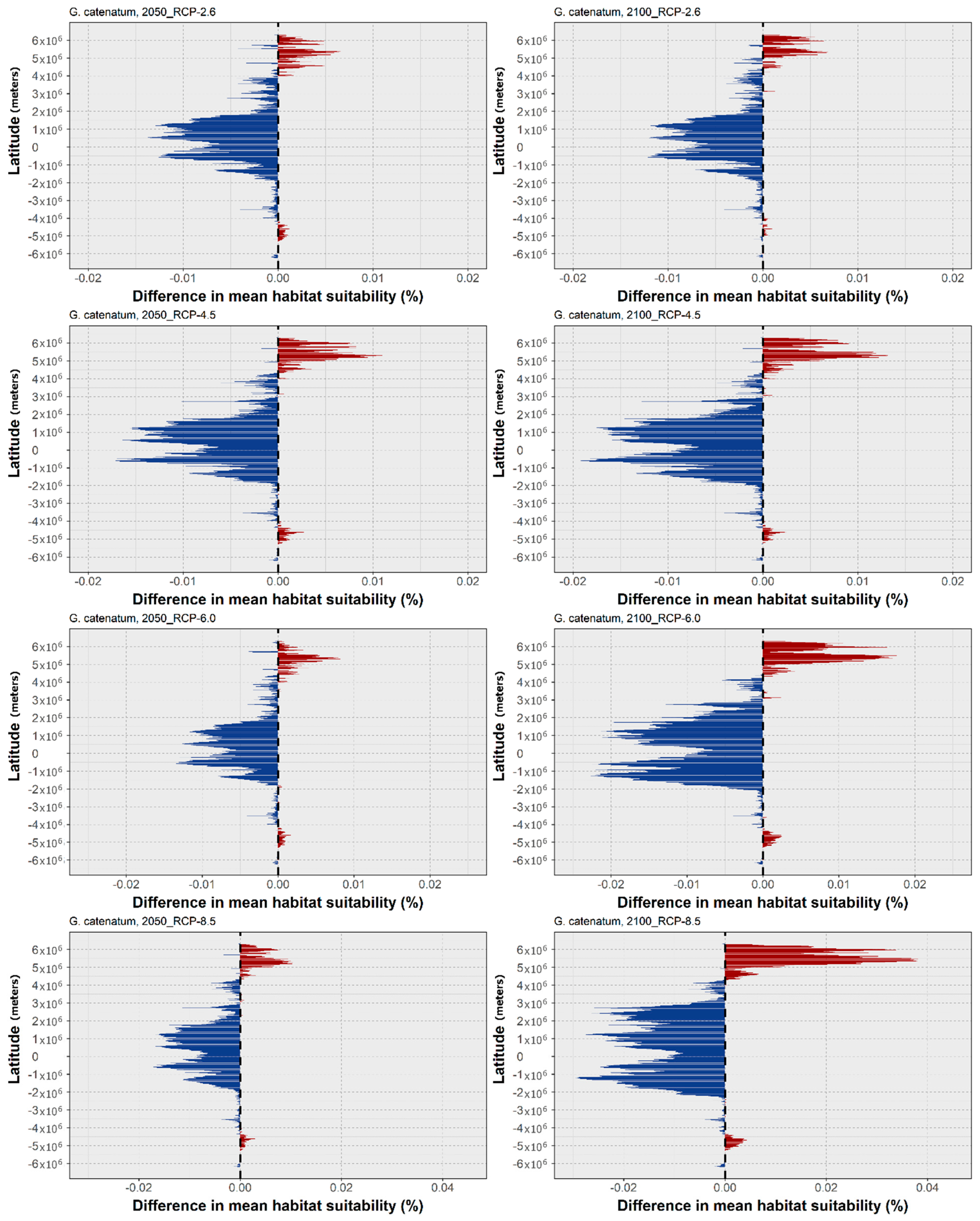
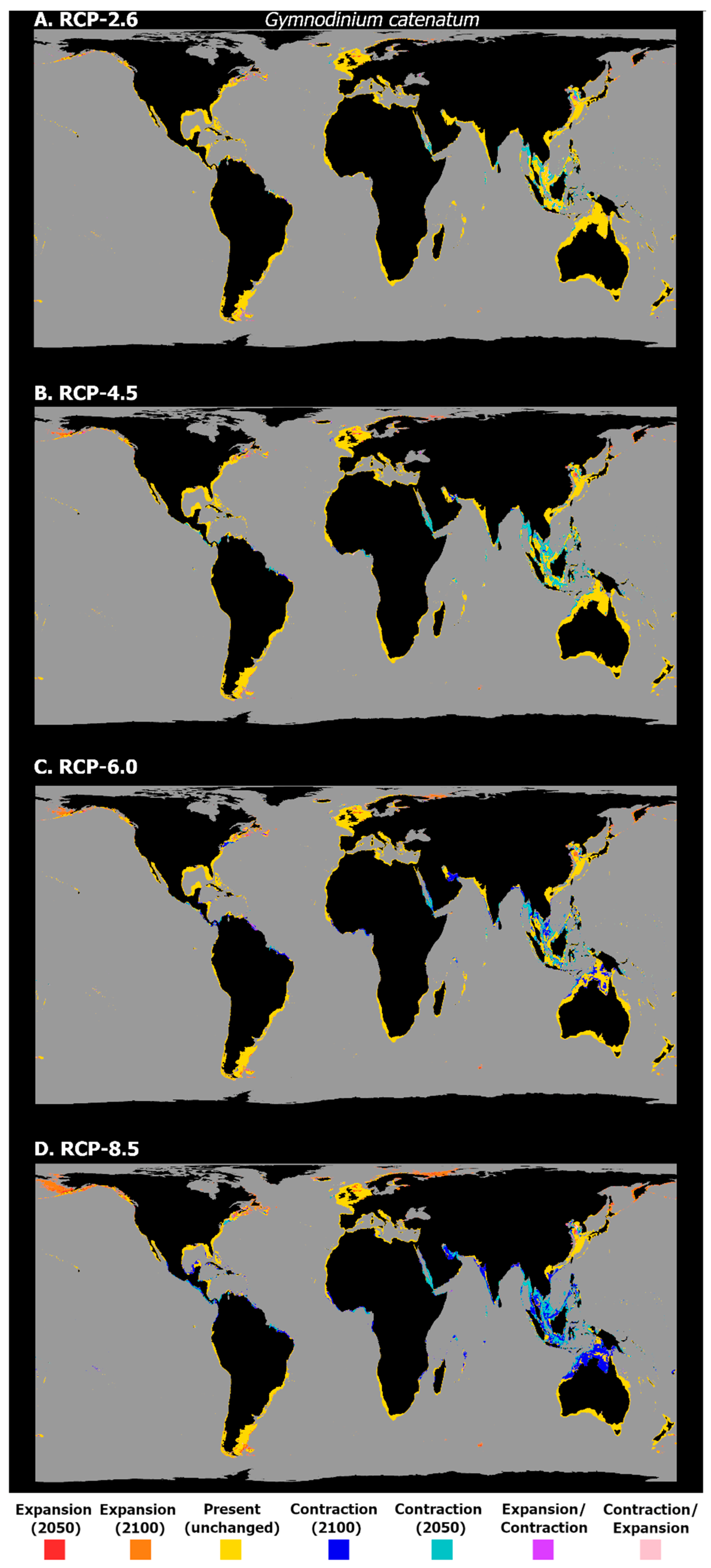
3. Results
4. Discussion
5. Final Considerations
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Glibert, P.M. Eutrophication, Harmful Algae and Biodiversity—Challenging Paradigms in a World of Complex Nutrient Changes. Mar. Pollut. Bull. 2017, 124, 591–606. [Google Scholar] [CrossRef] [PubMed]
- Kudela, R.M.; Raine, R.; Pitcher, G.C.; Gentien, P.; Berdalet, E.; Enevoldsen, H.; Urban, E. Establishment, Goals, and Legacy of the Global Ecology and Oceanography of Harmful Algal Blooms (GEOHAB) Programme. In Global Ecology and Oceanography of Harmful Algal Blooms; Glibert, P.M., Berdalet, E., Burford, M.A., Pitcher, G.C., Zhou, M., Eds.; Ecological Studies; Springer International Publishing: Cham, Switzerland, 2018; pp. 27–49. ISBN 978-3-319-70069-4. [Google Scholar]
- Visciano, P.; Schirone, M.; Berti, M.; Milandri, A.; Tofalo, R.; Suzzi, G. Marine Biotoxins: Occurrence, Toxicity, Regulatory Limits and Reference Methods. Front. Microbiol. 2016, 7, 1051. [Google Scholar] [CrossRef] [PubMed]
- Gao, P.; Wang, P.; Chen, S.; Bi, W.; Lu, S.; He, J.; Wang, X.; Li, K. Effect of Ambient Nitrogen on the Growth of Phytoplankton in the Bohai Sea: Kinetics and Parameters. J. Geophys. Res. Biogeosci. 2020, 125, e2020JG005643. [Google Scholar] [CrossRef]
- Berdalet, E.; Kudela, R.M.; Banas, N.S.; Bresnan, E.; Burford, M.A.; Davidson, K.; Gobler, C.J.; Karlson, B.; Lim, P.T.; Mackenzie, L.; et al. GlobalHAB:: Fostering International Coordination on Harmful Algal Bloom Research in Aquatic Systems. In Global Ecology and Oceanography of Harmful Algal Blooms; Springer: Berlin/Heidelberg, Germany, 2018; pp. 425–447. [Google Scholar] [CrossRef]
- Wang, D.; Zhang, S.; Zhang, H.; Lin, S. Omics Study of Harmful Algal Blooms in China: Current Status, Challenges, and Future Perspectives. Harmful Algae 2021, 107, 102079. [Google Scholar] [CrossRef]
- Zohdi, E.; Abbaspour, M. Harmful Algal Blooms (Red Tide): A Review of Causes, Impacts and Approaches to Monitoring and Prediction. Int. J. Environ. Sci. Technol. 2019, 16, 1789–1806. [Google Scholar] [CrossRef]
- Sha, J.; Xiong, H.; Li, C.; Lu, Z.; Zhang, J.; Zhong, H.; Zhang, W.; Yan, B. Harmful Algal Blooms and Their Eco-Environmental Indication. Chemosphere 2021, 274, 129912. [Google Scholar] [CrossRef]
- Work, T.M.; Moeller, P.D.R.; Beauchesne, K.R.; Dagenais, J.; Breeden, R.; Rameyer, R.; Walsh, W.J.; Abecassis, M.; Kobayashi, D.R.; Conway, C.; et al. Pufferfish Mortality Associated with Novel Polar Marine Toxins in Hawaii. Dis. Aquat. Org. 2017, 123, 87–99. [Google Scholar] [CrossRef]
- Costa, P.R. Impact and Effects of Paralytic Shellfish Poisoning Toxins Derived from Harmful Algal Blooms to Marine Fish. Fish Fish. 2016, 17, 226–248. [Google Scholar] [CrossRef]
- Lu, Y.; Yuan, J.; Lu, X.; Su, C.; Zhang, Y.; Wang, C.; Cao, X.; Li, Q.; Su, J.; Ittekkot, V.; et al. Major Threats of Pollution and Climate Change to Global Coastal Ecosystems and Enhanced Management for Sustainability. Environ. Pollut. 2018, 239, 670–680. [Google Scholar] [CrossRef]
- Todd, P.A.; Heery, E.C.; Loke, L.H.L.; Thurstan, R.H.; Kotze, D.J.; Swan, C. Towards an Urban Marine Ecology: Characterizing the Drivers, Patterns and Processes of Marine Ecosystems in Coastal Cities. Oikos 2019, 128, 1215–1242. [Google Scholar] [CrossRef]
- Broadwater, M.H.; Van Dolah, F.M.; Fire, S.E. Vulnerabilities of Marine Mammals to Harmful Algal Blooms. In Harmful Algal Blooms; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2018; pp. 191–222. ISBN 978-1-118-99467-2. [Google Scholar]
- Dickey, R.W.; Plakas, S.M. Ciguatera: A Public Health Perspective. Toxicon 2010, 56, 123–136. [Google Scholar] [CrossRef]
- Masó, M.; Garcés, E. Harmful Microalgae Blooms (HAB); Problematic and Conditions That Induce Them. Mar. Pollut. Bull. 2006, 53, 620–630. [Google Scholar] [CrossRef]
- Anderson, C.R.; Moore, S.K.; Tomlinson, M.C.; Silke, J.; Cusack, C.K. Chapter 17—Living with Harmful Algal Blooms in a Changing World: Strategies for Modeling and Mitigating Their Effects in Coastal Marine Ecosystems. In Coastal and Marine Hazards, Risks, and Disasters; Shroder, J.F., Ellis, J.T., Sherman, D.J., Eds.; Hazards and Disasters Series; Elsevier: Boston, MA, USA, 2015; pp. 495–561. ISBN 978-0-12-396483-0. [Google Scholar]
- Wells, M.L.; Trainer, V.L.; Smayda, T.J.; Karlson, B.S.O.; Trick, C.G.; Kudela, R.M.; Ishikawa, A.; Bernard, S.; Wulff, A.; Anderson, D.M.; et al. Harmful Algal Blooms and Climate Change: Learning from the Past and Present to Forecast the Future. Harmful Algae 2015, 49, 68–93. [Google Scholar] [CrossRef]
- Lopes, V.M.; Costa, P.R.; Rosa, R. Effects of Harmful Algal Bloom Toxins on Marine Organisms. In Ecotoxicology of Marine Organisms; CRC Press: Boca Raton, FL, USA, 2019; pp. 42–88. ISBN 978-1-315-26749-4. [Google Scholar]
- Zingone, A.; Enevoldsen, H.; Zingone, A.; Enevoldsen, H.O. The Diversity of Harmful Algal Blooms: A Challenge for Science and Management. Ocean. Coast. Manag. 2000, 43, 725–748. [Google Scholar] [CrossRef]
- Fensome, R.A.; Taylor, F.J.R.; Norris, G.; Sarjeant, W.A.S.; Wharton, D.I.; Williams, G.L. A Classification of Living and Fossil Dinoflagellates; Sheridon Press: Hanover, PA, USA, 1993; Volume 7. [Google Scholar]
- Smayda, T.J.; Reynolds, C.S. Strategies of Marine Dinoflagellate Survival and Some Rules of Assembly. J. Sea Res. 2003, 49, 95–106. [Google Scholar] [CrossRef]
- Hallegraeff, G.M. A Review of Harmful Algal Blooms and Their Apparent Global Increase. Phycologia 1993, 32, 79–99. [Google Scholar] [CrossRef]
- Anderson, D.M.; Alpermann, T.J.; Cembella, A.D.; Collos, Y.; Masseret, E.; Montresor, M. The Globally Distributed Genus Alexandrium: Multifaceted Roles in Marine Ecosystems and Impacts on Human Health. Harmful Algae 2012, 14, 10–35. [Google Scholar] [CrossRef]
- Young, N.; Sharpe, R.A.; Barciela, R.; Nichols, G.; Davidson, K.; Berdalet, E.; Fleming, L.E. Marine Harmful Algal Blooms and Human Health: A Systematic Scoping Review. Harmful Algae 2020, 98, 101901. [Google Scholar] [CrossRef]
- Balech, E. The Genus Alexandrium Halim (Dinoflagellata); Sherkin Island Marine Station: Cork, Ireland, 1995. [Google Scholar]
- Haberkorn, H.; Lambert, C.; Le Goïc, N.; Moal, J.; Suquet, M.; Guéguen, M.; Sunila, I.; Soudant, P. Effects of Alexandrium Minutum Exposure on Nutrition-Related Processes and Reproductive Output in Oysters Crassostrea Gigas. Harmful Algae 2010, 9, 427–439. [Google Scholar] [CrossRef]
- Menezes, M.; Branco, S.; Miotto, M.C.; Alves-de-Souza, C. The Genus Alexandrium (Dinophyceae, Dinophyta) in Brazilian Coastal Waters. Front. Mar. Sci. 2018, 5, 425–447. [Google Scholar] [CrossRef]
- Boyd, P.W.; Doney, S.C. The Impact of Climate Change and Feedback Processes on the Ocean Carbon Cycle. In Ocean Biogeochemistry: The Role of the Ocean Carbon Cycle in Global Change; Fasham, M.J.R., Ed.; Global Change—The IGBP Series (closed); Springer: Berlin/Heidelberg, Germany, 2003; pp. 157–193. ISBN 978-3-642-55844-3. [Google Scholar]
- Condie, S.A.; Oliver, E.C.J.; Hallegraeff, G.M. Environmental Drivers of Unprecedented Alexandrium Catenella Dinoflagellate Blooms off Eastern Tasmania, 2012–2018. Harmful Algae 2019, 87, 101628. [Google Scholar] [CrossRef] [PubMed]
- Guallar, C.; Bacher, C.; Chapelle, A. Global and Local Factors Driving the Phenology of Alexandrium Minutum (Halim) Blooms and Its Toxicity. Harmful Algae 2017, 67, 44–60. [Google Scholar] [CrossRef]
- Santos, M.; Costa, P.R.; Porteiro, F.M.; Moita, M.T. First Report of a Massive Bloom of Alexandrium Minutum (Dinophyceae) in Middle North Atlantic: A Coastal Lagoon in S. Jorge Island, Azores. Toxicon 2014, 90, 265–268. [Google Scholar] [CrossRef]
- da Costa, R.M.; Pereira, L.C.C.; Ferrnández, F. Deterrent Effect of Gymnodinium Catenatum Graham PSP-Toxins on Grazing Performance of Marine Copepods. Harmful Algae 2012, 17, 75–82. [Google Scholar] [CrossRef]
- Negri, A.P. Paralytic Shellfish Toxins in Gymnodinium Catenatum Strains from Six Countries. In Proceedings of the Harmful algal blooms 2000: Proceedings of the Ninth International Conference on Harmful Algal Blooms, Hobart, Australia, 7–11 February 2000. [Google Scholar]
- Hallegraeff, G.M.; Blackburn, S.I.; Doblin, M.A.; Bolch, C.J.S. Global Toxicology, Ecophysiology and Population Relationships of the Chainforming PST Dinoflagellate Gymnodinium Catenatum. Harmful Algae 2012, 14, 130–143. [Google Scholar] [CrossRef]
- Silva, T.; Caeiro, M.F.; Costa, P.R.; Amorim, A. Gymnodinium Catenatum Graham Isolated from the Portuguese Coast: Toxin Content and Genetic Characterization. Harmful Algae 2015, 48, 94–104. [Google Scholar] [CrossRef]
- Fraga, S. Harmful Algal Blooms in Relation to Wind Induced Coastal Upwelling and River Plumes; Instituto Español de Oceanografia: Vigo, Spain, 1995. [Google Scholar]
- Proença, L.; Tamanaha, M.; Souza, N. The Toxic Dinoflagellate Gymnodinium Catenatum Graham in Southern Brazilian Waters: Occurrence, pigments and toxins. Atlântica 2001, 23, 59–65. [Google Scholar]
- Ralston, D.K.; Moore, S.K. Modeling Harmful Algal Blooms in a Changing Climate. Harmful Algae 2020, 91, 101729. [Google Scholar] [CrossRef]
- Ribeiro, S.; Amorim, A.; Andersen, T.J.; Abrantes, F.; Ellegaard, M. Reconstructing the History of an Invasion: The Toxic Phytoplankton Species Gymnodinium Catenatum in the Northeast Atlantic. Biol. Invasions 2012, 14, 969–985. [Google Scholar] [CrossRef]
- Townhill, B.L.; Tinker, J.; Jones, M.; Pitois, S.; Creach, V.; Simpson, S.D.; Dye, S.; Bear, E.; Pinnegar, J.K. Harmful Al-gal Blooms and Climate Change: Exploring Future Distribution Changes. ICES J. Mar. Sci. 2018, 75, 1882–1893. [Google Scholar] [CrossRef]
- Pecl, G.T.; Araújo, M.B.; Bell, J.D.; Blanchard, J.; Bonebrake, T.C.; Chen, I.-C.; Clark, T.D.; Colwell, R.K.; Danielsen, F.; Evengård, B.; et al. Biodiversity Redistribution under Climate Change: Impacts on Ecosystems and Human Well-Being. Science 2017, 355, eaai9214. [Google Scholar] [CrossRef] [PubMed]
- Bindoff, N.; Cheung, W.; Kairo, J.G.; Aristegui, J.; Guinder, V.; Hallberg, R.; Hilmi, N.; Jiao, N.; Karim, M.; Levin, L.; et al. Changing Ocean, Marine Ecosystems, and Dependent Communities (09 SROCC Ch05 FINAL-1). In IPCC Special Report on the Ocean and Cryosphere in a Changing Climate; IPCC: Geneva, Switzerland, 2019; pp. 447–588. [Google Scholar]
- Climate Change 2021: The Physical Science Basis. Contribution of Working Group I to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change; Masson-Delmotte, V., Zhai, P., Pirani, A., Connors, S.L., Péan, C., Berger, S., Caud, N., Chen, Y., Goldfarb, L., Gomis, M.I., et al., Eds.; Cambridge University Press: Cambridge, UK, 2021. [Google Scholar]
- Caldeira, K.; Wickett, M.E. Anthropogenic Carbon and Ocean PH. Nature 2003, 425, 365. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, D.L.; Klein Tank, A.M.G.; Rusticucci, M.; Alexander, L.V.; Brönnimann, S.; Charabi, Y.A.R.; Dentener, F.J.; Dlugokencky, E.J.; Easterling, D.R.; Kaplan, A.; et al. Observations: Atmosphere and Surface. In Climate Change 2013 the Physical Science Basis; Cambridge University Press: Cambridge, UK, 2013; Volume 9781107057999, pp. 159–254. ISBN 978-1-107-05799-9. [Google Scholar]
- Oschlies, A.; Brandt, P.; Stramma, L.; Schmidtko, S. Drivers and Mechanisms of Ocean Deoxygenation. Nat. Geosci. 2018, 11, 467–473. [Google Scholar] [CrossRef]
- Ocean Deoxygenation: Everyone’s Problem. Causes, Impacts, Consequences and Solutions; Laffoley, D., Baxter, J.M., Eds.; IUCN, International Union for Conservation of Nature: Gland, Switzerland, 2019; ISBN 978-2-8317-2013-5. [Google Scholar]
- Harley, C.D.G.; Randall Hughes, A.; Hultgren, K.M.; Miner, B.G.; Sorte, C.J.B.; Thornber, C.S.; Rodriguez, L.F.; Tomanek, L.; Williams, S.L. The Impacts of Climate Change in Coastal Marine Systems. Ecol. Lett. 2006, 9, 228–241. [Google Scholar] [CrossRef]
- Kroeker, K.J.; Kordas, R.L.; Crim, R.; Hendriks, I.E.; Ramajo, L.; Singh, G.S.; Duarte, C.M.; Gattuso, J.-P. Impacts of Ocean Acidification on Marine Organisms: Quantifying Sensitivities and Interaction with Warming. Glob. Chang. Biol. 2013, 19, 1884–1896. [Google Scholar] [CrossRef]
- Sampaio, E.; Santos, C.; Rosa, I.C.; Ferreira, V.; Pörtner, H.-O.; Duarte, C.M.; Levin, L.A.; Rosa, R. Impacts of Hypoxic Events Surpass Those of Future Ocean Warming and Acidification. Nat. Ecol. Evol. 2021, 5, 311–321. [Google Scholar] [CrossRef]
- Shukla, P.R.; Skeg, J.; Buendia, E.C.; Masson-Delmotte, V.; Pörtner, H.-O.; Roberts, D.C.; Zhai, P.; Slade, R.; Connors, S.; van Diemen, S.; et al. Climate Change and Land: An IPCC Special Report on Climate Change, Desertification, Land Degradation, Sustainable Land Management, Food Security, and Greenhouse Gas Fluxes in Terrestrial Ecosystems; IPCC: Geneva, Switzerland, 2019. [Google Scholar]
- IPCC. IPCC, 2014: Climate Change 2014: Synthesis Report. Contribution of Working Groups I, II and III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Core Writing Team, Pachauri, R.K., Meyer, L.A., Eds.; IPCC: Geneva, Switzerland, 2014; p. 151.
- Hallegraeff, G.M.; Anderson, D.M.; Belin, C.; Bottein, M.-Y.D.; Bresnan, E.; Chinain, M.; Enevoldsen, H.; Iwataki, M.; Karlson, B.; McKenzie, C.H.; et al. Perceived Global Increase in Algal Blooms Is Attributable to Intensified Monitoring and Emerging Bloom Impacts. Commun. Earth. Environ. 2021, 2, 117. [Google Scholar] [CrossRef]
- Gobler, C.J.; Doherty, O.M.; Hattenrath-Lehmann, T.K.; Griffith, A.W.; Kang, Y.; Litaker, R.W. Ocean Warming since 1982 Has Expanded the Niche of Toxic Algal Blooms in the North Atlantic and North Pacific Oceans. Proc. Natl. Acad. Sci. USA 2017, 114, 4975–4980. [Google Scholar] [CrossRef]
- McKibben, S.M.; Watkins-Brandt, K.S.; Wood, A.M.; Hunter, M.; Forster, Z.; Hopkins, A.; Du, X.; Eberhart, B.-T.; Peterson, W.T.; White, A.E. Monitoring Oregon Coastal Harmful Algae: Observations and Implications of a Harmful Algal Bloom-Monitoring Project. Harmful Algae 2015, 50, 32–44. [Google Scholar] [CrossRef]
- Silveira, S.B.; Odebrecht, C. Effects of Salinity and Temperature on the Growth, Toxin Production, and Akinete Germination of the Cyanobacterium Nodularia spumigena. Front. Mar. Sci. 2019, 6, 155–160. [Google Scholar] [CrossRef]
- Zhu, J.; Lei, X.; Quan, J.; Yue, X. Algae Growth Distribution and Key Prevention and Control Positions for the Middle Route of the South-to-North Water Diversion Project. Water 2019, 11, 1851. [Google Scholar] [CrossRef]
- Kibler, S.R.; Tester, P.A.; Kunkel, K.E.; Moore, S.K.; Litaker, R.W. Effects of Ocean Warming on Growth and Distribution of Dinoflagellates Associated with Ciguatera Fish Poisoning in the Caribbean. Ecol. Modell. 2015, 316, 194–210. [Google Scholar] [CrossRef]
- Henson, S.A.; Cole, H.S.; Hopkins, J.; Martin, A.P.; Yool, A. Detection of Climate Change-Driven Trends in Phytoplankton Phenology. Glob. Chang. Biol. 2018, 24, e101–e111. [Google Scholar] [CrossRef]
- Moore, S.K.; Mantua, N.J.; Salathé, E.P. Past Trends and Future Scenarios for Environmental Conditions Favoring the Accumulation of Paralytic Shellfish Toxins in Puget Sound Shellfish. Harmful Algae 2011, 10, 521–529. [Google Scholar] [CrossRef]
- Hannah, L.; Midgley, G.F.; Lovejoy, T.; Bond, W.J.; Bush, M.; Lovett, J.C.; Scott, D.; Woodward, F.I. Conservation of Biodiversity in a Changing Climate. Conserv. Biol. 2002, 16, 264–268. [Google Scholar] [CrossRef]
- Zimmermann, N.E.; Edwards Jr, T.C.; Graham, C.H.; Pearman, P.B.; Svenning, J.-C. New Trends in Species Distribution Modelling. Ecography 2010, 33, 985–989. [Google Scholar] [CrossRef]
- Elith, J.; Graham, C.H.; Anderson, R.P.; Dudík, M.; Ferrier, S.; Guisan, A.; Hijmans, R.J.; Huettmann, F.; Leathwick, J.R.; Lehmann, A.; et al. Novel Methods Improve Prediction of Species’ Distributions from Occurrence Data. Ecography 2006, 29, 129–151. [Google Scholar] [CrossRef]
- Araújo, M.; Rahbek, C. How Does Climate Change Affect Biodiversity? Science 2006, 313, 1396–1397. [Google Scholar] [CrossRef]
- Boavida-Portugal, J.; Rosa, R.; Calado, R.; Pinto, M.; Boavida-Portugal, I.; Araújo, M.B.; Guilhaumon, F. Climate Change Impacts on the Distribution of Coastal Lobsters. Mar. Biol. 2018, 165, 186. [Google Scholar] [CrossRef]
- Boavida-Portugal, J.; Guilhaumon, F.; Rosa, R.; Araújo, M.B. Global Patterns of Coastal Cephalopod Diversity Under Climate Change. Front. Mar. Sci. 2022, 8, 740781. [Google Scholar] [CrossRef]
- García-Roselló, E.; Guisande, C.; González-Vilas, L.; González-Dacosta, J.; Heine, J.; Pérez-Costas, E.; Lobo, J.M. A Simple Method to Estimate the Probable Distribution of Species. Ecography 2019, 42, 1613–1622. [Google Scholar] [CrossRef]
- Borges, F.O.; Santos, C.P.; Paula, J.R.; Mateos-Naranjo, E.; Redondo-Gomez, S.; Adams, J.B.; Caçador, I.; Fonseca, V.F.; Reis-Santos, P.; Duarte, B.; et al. Invasion and Extirpation Potential of Native and Invasive Spartina Species Under Climate Change. Front. Mar. Sci. 2021, 8. [Google Scholar] [CrossRef]
- Sinclair, S.; White, M.; Newell, G. How Useful Are Species Distribution Models for Managing Biodiversity under Future Climates? Ecol. Soc. 2010, 15, art8. [Google Scholar] [CrossRef]
- Barton, A.D.; Irwin, A.J.; Finkel, Z.V.; Stock, C.A. Anthropogenic Climate Change Drives Shift and Shuffle in North Atlantic Phytoplankton Communities. Proc. Natl. Acad. Sci. USA 2016, 113, 2964–2969. [Google Scholar] [CrossRef]
- Anderson, C.R.; Berdalet, E.; Kudela, R.M.; Cusack, C.K.; Silke, J.; O’Rourke, E.; Dugan, D.; McCammon, M.; Newton, J.A.; Moore, S.K.; et al. Scaling Up From Regional Case Studies to a Global Harmful Algal Bloom Observing System. Front. Mar. Sci. 2019, 6, 250. [Google Scholar] [CrossRef]
- R Studio Team. R Studio: Integrated Development for R. PBC, Boston, MA. 2022. Available online: http://www.rstudio.com/ (accessed on 26 September 2022).
- Laruelle, G.G.; Cai, W.-J.; Hu, X.; Gruber, N.; Mackenzie, F.T.; Regnier, P. Continental Shelves as a Variable but Increasing Global Sink for Atmospheric Carbon Dioxide. Nat. Commun. 2018, 9, 454. [Google Scholar] [CrossRef]
- Sbrocco, E.J.; Barber, P.H. MARSPEC: Ocean Climate Layers for Marine Spatial Ecology. Ecology 2013, 94, 979. [Google Scholar] [CrossRef]
- Peterson, A.; Soberón, J. Species Distribution Modeling and Ecological Niche Modeling: Getting the Concepts Right. Nat. Conserv. 2012, 10, 102–107. [Google Scholar] [CrossRef]
- Schwalm, C.R.; Glendon, S.; Duffy, P.B. RCP8.5 Tracks Cumulative CO2 Emissions. Proc. Natl. Acad. Sci. USA 2020, 117, 19656–19657. [Google Scholar] [CrossRef]
- Tyberghein, L.; Verbruggen, H.; Pauly, K.; Troupin, C.; Mineur, F.; De Clerck, O. Bio-ORACLE: A Global Environmental Dataset for Marine Species Distribution Modelling. Glob. Ecol. Biogeogr. 2012, 21, 272–281. [Google Scholar] [CrossRef]
- Assis, J.; Tyberghein, L.; Bosch, S.; Verbruggen, H.; Serrão, E.A.; De Clerck, O. Bio-ORACLE v2.0: Extending Marine Data Layers for Bioclimatic Modelling. Glob. Ecol. Biogeogr. 2018, 27, 277–284. [Google Scholar] [CrossRef]
- Shipley, B.R.; Bach, R.; Do, Y.; Strathearn, H.; McGuire, J.L.; Dilkina, B. MegaSDM: Integrating Dispersal and Time-Step Analyses into Species Distribution Models. Ecography 2022, 2022. [Google Scholar] [CrossRef]
- Elith, J.; Phillips, S.J.; Hastie, T.; Dudík, M.; Chee, Y.E.; Yates, C.J. A Statistical Explanation of MaxEnt for Ecologists. Divers. Distrib. 2011, 17, 43–57. [Google Scholar] [CrossRef]
- Phillips, S.J.; Dudík, M.; Elith, J.; Graham, C.H.; Lehmann, A.; Leathwick, J.; Ferrier, S. Sample Selection Bias and Presence-Only Distribution Models: Implications for Background and Pseudo-Absence Data. Ecol. Appl. 2009, 19, 181–197. [Google Scholar] [CrossRef]
- Boakes, E.H.; McGowan, P.J.K.; Fuller, R.A.; Chang-qing, D.; Clark, N.E.; O’Connor, K.; Mace, G.M. Distorted Views of Biodiversity: Spatial and Temporal Bias in Species Occurrence Data. PLoS Biol. 2010, 8, e1000385. [Google Scholar] [CrossRef]
- Beck, J.; Ballesteros-Mejia, L.; Nagel, P.; Kitching, I.J. Online Solutions and the ‘Wallacean Shortfall’: What Does GBIF Contribute to Our Knowledge of Species’ Ranges? Divers. Distrib. 2013, 19, 1043–1050. [Google Scholar] [CrossRef]
- Varela, S.; Anderson, R.P.; García-Valdés, R.; Fernández-González, F. Environmental Filters Reduce the Effects of Sampling Bias and Improve Predictions of Ecological Niche Models. Ecography 2014, 37, 1084–1091. [Google Scholar] [CrossRef]
- Barbet-Massin, M.; Jiguet, F.; Albert, C.H.; Thuiller, W. Selecting Pseudo-Absences for Species Distribution Models: How, Where and How Many? Methods Ecol. Evol. 2012, 3, 327–338. [Google Scholar] [CrossRef]
- Lobo, J.M.; Jiménez-Valverde, A.; Hortal, J. The Uncertain Nature of Absences and Their Importance in Species Distribution Modelling. Ecography 2010, 33, 103–114. [Google Scholar] [CrossRef]
- Kramer-Schadt, S.; Niedballa, J.; Pilgrim, J.D.; Schröder, B.; Lindenborn, J.; Reinfelder, V.; Stillfried, M.; Heckmann, I.; Scharf, A.K.; Augeri, D.M.; et al. The Importance of Correcting for Sampling Bias in MaxEnt Species Distribution Models. Divers. Distrib. 2013, 19, 1366–1379. [Google Scholar] [CrossRef]
- Radosavljevic, A.; Anderson, R.P. Making Better Maxent Models of Species Distributions: Complexity, Overfitting and Evaluation. J. Biogeogr. 2014, 41, 629–643. [Google Scholar] [CrossRef]
- Elith, J.; Kearney, M.; Phillips, S. The Art of Modelling Range-Shifting Species. Methods Ecol. Evol. 2010, 1, 330–342. [Google Scholar] [CrossRef]
- Raes, N.; ter Steege, H. A Null-Model for Significance Testing of Presence-Only Species Distribution Models. Ecography 2007, 30, 727–736. [Google Scholar] [CrossRef]
- Araújo, M.B.; New, M. Ensemble Forecasting of Species Distributions. Trends Ecol. Evol. 2007, 22, 42–47. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Berry, P.M.; Dawson, T.P.; Pearson, R.G. Selecting Thresholds of Occurrence in the Prediction of Species Distributions. Ecography 2005, 28, 385–393. [Google Scholar] [CrossRef]
- Early, R.; Sax, D.F. Analysis of Climate Paths Reveals Potential Limitations on Species Range Shifts. Ecol. Lett. 2011, 14, 1125–1133. [Google Scholar] [CrossRef]
- Dai, L.; Yu, R.-C.; Geng, H.-X.; Zhao, Y.; Zhang, Q.-C.; Kong, F.-Z.; Chen, Z.-F.; Zhao, J.-Y.; Zhou, M.-J. Resting Cysts of Alexandrium catenella and A. pacificum (Dinophyceae) in the Bohai and Yellow Seas, China: Abundance, Distribution and Implications for Toxic Algal Blooms. Harmful Algae 2020, 93, 101794. [Google Scholar] [CrossRef]
- Nagai, S.; Chen, H.; Kawakami, Y.; Yamamoto, K.; Sildever, S.; Kanno, N.; Oikawa, H.; Yasuike, M.; Nakamura, Y.; Hongo, Y.; et al. Monitoring of the Toxic Dinoflagellate Alexandrium catenella in Osaka Bay, Japan Using a Massively Parallel Sequencing (MPS)-Based Technique. Harmful Algae 2019, 89, 101660. [Google Scholar] [CrossRef]
- Kim, Y.O.; Choi, J.; Baek, S.H.; Lee, M.; Oh, H.-M. Tracking Alexandrium catenella from Seed-Bed to Bloom on the Southern Coast of Korea. Harmful Algae 2020, 99, 101922. [Google Scholar] [CrossRef]
- Farrell, H.; Brett, S.; Ajani, P.; Murray, S. Distribution of the Genus Alexandrium (Halim) and Paralytic Shellfish Toxins along the Coastline of New South Wales, Australia. Mar. Poll. Bull. 2013, 72, 133–145. [Google Scholar] [CrossRef]
- Dias, J.; Muñoz, J.; Huisman, J.; McDonald, J. Biosecurity Monitoring of Harmful Algal Bloom (HAB) Species in Western Australian Waters: First Confirmed Record of Alexandrium catenella (Dinophyceae). Bioinvasions Rec. 2015, 4, 233–241. [Google Scholar] [CrossRef]
- Harwood, D.T.; Boundy, M.; Selwood, A.I.; van Ginkel, R.; MacKenzie, L.; McNabb, P.S. Refinement and Implementation of the Lawrence Method (AOAC 2005.06) in a Commercial Laboratory: Assay Performance during an Alexandrium catenella Bloom Event. Harmful Algae 2013, 24, 20–31. [Google Scholar] [CrossRef]
- Joyce, L.; Pitcher, G. Cysts of Alexandrium catenella on the West Coast of South Africa: Distribution and Characteristics of Germination. Afr. J. Mar. Sci. 2006, 28, 295–298. [Google Scholar] [CrossRef]
- Crawford, D.W.; Montero, P.; Daneri, G. Blooms of Alexandrium catenella in Coastal Waters of Chilean Patagonia: Is Subantarctic Surface Water Involved? Front. Mar. Sci. 2021, 8, 612628. [Google Scholar] [CrossRef]
- Persich, G.R.; Kulis, D.M.; Lilly, E.L.; Anderson, D.M.; Garcia, V.M.T. Probable Origin and Toxin Profile of Alexandrium tamarense (Lebour) Balech from Southern Brazil. Harmful Algae 2006, 5, 36–44. [Google Scholar] [CrossRef]
- Penna, A.; Garcés, E.; Vila, M.; Giacobbe, M.G.; Fraga, S.; Lugliè, A.; Bravo, I.; Bertozzini, E.; Vernesi, C. Alexandrium catenella (Dinophyceae), a Toxic Ribotype Expanding in the NW Mediterranean Sea. Mar. Biol. 2005, 148, 13–23. [Google Scholar] [CrossRef]
- Laabir, M.; Jauzein, C.; Genovesi, B.; Masseret, E.; Grzebyk, D.; Cecchi, P.; Vaquer, A.; Perrin, Y.; Collos, Y. Influence of Temperature, Salinity and Irradiance on the Growth and Cell Yield of the Harmful Red Tide Dinoflagellate Alexandrium catenella Colonizing Mediterranean Waters. J. Plankton Res. 2011, 33, 1550–1563. [Google Scholar] [CrossRef]
- Li, Y.; Stumpf, R.P.; McGillicuddy, D.J.; He, R. Dynamics of an Intense Alexandrium Catenella Red Tide in the Gulf of Maine: Satellite Observations and Numerical Modeling. Harmful Algae 2020, 99, 101927. [Google Scholar] [CrossRef]
- Jester, R.J.; Baugh, K.A.; Lefebvre, K.A. Presence of Alexandrium catenella and Paralytic Shellfish Toxins in Finfish, Shellfish and Rock Crabs in Monterey Bay, California, USA. Mar. Biol. 2009, 156, 493–504. [Google Scholar] [CrossRef]
- Tobin, E.D.; Wallace, C.L.; Crumpton, C.; Johnson, G.; Eckert, G.L. Environmental Drivers of Paralytic Shellfish Toxin Producing Alexandrium catenella Blooms in a Fjord System of Northern Southeast Alaska. Harmful Algae 2019, 88, 101659. [Google Scholar] [CrossRef]
- Vandersea, M.W.; Kibler, S.R.; Tester, P.A.; Holderied, K.; Hondolero, D.E.; Powell, K.; Baird, S.; Doroff, A.; Dugan, D.; Litaker, R.W. Environmental Factors Influencing the Distribution and Abundance of Alexandrium catenella in Kachemak Bay and Lower Cook Inlet, Alaska. Harmful Algae 2018, 77, 81–92. [Google Scholar] [CrossRef] [PubMed]
- Anderson, D.M.; Fachon, E.; Pickart, R.S.; Lin, P.; Fischer, A.D.; Richlen, M.L.; Uva, V.; Brosnahan, M.L.; McRaven, L.; Bahr, F.; et al. Evidence for Massive and Recurrent Toxic Blooms of Alexandrium catenella in the Alaskan Arctic. Proc. Natl. Acad. Sci. USA 2021, 118, e2107387118. [Google Scholar] [CrossRef] [PubMed]
- Seto, D.S.; Karp-Boss, L.; Wells, M.L. Effects of Increasing Temperature and Acidification on the Growth and Competitive Success of Alexandrium catenella from the Gulf of Maine. Harmful Algae 2019, 89, 101670. [Google Scholar] [CrossRef] [PubMed]
- Baylón, M.; Sánchez, S.; Bárcena, V.; López, J.; Mamani, E. First record of potentially toxic dinoflagellate, Alexandrium minutum Halim 1960, from Peruvian coastal. Rev. Peru. Biol. 2015, 22, 113–118. [Google Scholar]
- Lewis, A.M.; Coates, L.N.; Turner, A.D.; Percy, L.; Lewis, J. A Review of the Global Distribution of Alexandrium minutum (Dinophyceae) and Comments on Ecology and Associated Paralytic Shellfish Toxin Profiles, with a Focus on Northern Europe. J. Phycol. 2018, 54, 581–598. [Google Scholar] [CrossRef]
- Karlson, B.; Andersen, P.; Arneborg, L.; Cembella, A.; Eikrem, W.; John, U.; West, J.J.; Klemm, K.; Kobos, J.; Lehtinen, S.; et al. Harmful Algal Blooms and Their Effects in Coastal Seas of Northern Europe. Harmful Algae 2021, 102, 101989. [Google Scholar] [CrossRef]
- Bravo, I.; Fraga, S.; Isabel Figueroa, R.; Pazos, Y.; Massanet, A.; Ramilo, I. Bloom Dynamics and Life Cycle Strategies of Two Toxic Dinoflagellates in a Coastal Upwelling System (NW Iberian Peninsula). Deep-Sea Res. II Top. Stud. Oceanogr. 2010, 57, 222–234. [Google Scholar] [CrossRef]
- Li, H.; Fedorov, A.V. Persistent Freshening of the Arctic Ocean and Changes in the North Atlantic Salinity Caused by Arctic Sea Ice Decline. Clim. Dyn. 2021, 57, 2995–3013. [Google Scholar] [CrossRef]
- Band-Schmidt, C.J.; Bustillos-Guzmán, J.J.; López-Cortés, D.J.; Gárate-Lizárraga, I.; Núñez-Vázquez, E.J.; Hernández-Sandoval, F.E. Ecological and Physiological Studies of Gymnodinium catenatum in the Mexican Pacific: A Review. Mar. Drugs 2010, 8, 1935–1961. [Google Scholar] [CrossRef]
- Ferrari, G.M.; Méndez, S.M. Report of Phytoplankton Species Producing Coastal Water Discoloration in Uruguay. Iheringia Ser. Bot. 2000, 54, 3–18. [Google Scholar]
- Rangel, I.; Silva, S. First Records of Gymnodinium catenatum, Gambierdiscus toxicus and Pyrodinium bahamense on Northern Luanda Coast, Angola. Harmful Algae News 2006, 32, 10–11. [Google Scholar]
- Fernando, G. The Toxic Dinoflagellate Gymnodinium catenatum: An Invader in the Mediterranean Sea. Acta Bot. Croat. 2003, 62, 65–72. [Google Scholar]
- Illoul, H.; Maso, M.; Fortuno, J.-M.; Cros, L.; Morales-Blake, A.; Seridji, R. Potentially Harmful Microalgae in Coastal Waters of the Algiers Area (Southern Mediterranea Sea). Cryptogam 2008, 29, 261. [Google Scholar]
- Godhe, A.; Karunasagar, I.; Karunasagar, I. Gymnodinium catenatum on West Coast of India. Harmful Algae News 1996, 15, 1. [Google Scholar]
- Godhe, A.; Karunasagar, I.; Karunasagar, I.; Karlson, B. Dinoflagellate Cysts in Recent Marine Sediments from SW India. Bot. Mar. 2000, 43, 39–48. [Google Scholar] [CrossRef]
- Glibert, P.M.; Landsberg, J.H.; Evans, J.J.; Al-Sarawi, M.A.; Faraj, M.; Al-Jarallah, M.A.; Haywood, A.; Ibrahem, S.; Klesius, P.; Powell, C.; et al. A Fish Kill of Massive Proportion in Kuwait Bay, Arabian Gulf, 2001: The Roles of Bacterial Disease, Harmful Algae, and Eutrophication. Harmful Algae 2002, 1, 215–231. [Google Scholar] [CrossRef]
- Holmes, M.J.; Bolch, C.J.S.; Green, D.H.; Cembella, A.D.; Teo, S.L.M. Singapore Isolates of the Dinoflagellate Gymnodinium catenatum (Dinophyceae) Produce a Unique Profile of Paralytic Shellfish Poisoning Toxins. J. Phycol. 2002, 38, 96–106. [Google Scholar] [CrossRef]
- Bolch, C.J.; Reynolds, M.J. Species Resolution and Global Distribution of Microreticulate Dinoflagellate Cysts. J. Plankton Res. 2002, 24, 565–578. [Google Scholar] [CrossRef][Green Version]
- McMinn, A.; Hallegraeff, G.M.; Thomson, P.; Jenkinson, A.V.; Heijnis, H. Cyst and Radionucleotide Evidence for the Recent Introduction of the Toxic Dinoflagellate Gymnodinium catenatum into Tasmanian Waters. Mar. Ecol. Prog. Ser. 1997, 161, 165–172. [Google Scholar] [CrossRef]
- Mackenzie, L. The risk to New Zealand shellfish aquaculture from paralytic shellfish poisoning (PSP) toxins. N. Z. J. Mar. Freshwater Res. 2014, 48, 430–465. [Google Scholar] [CrossRef]
- Fukuyo, Y.; Kodama, M.; Ogata, T.; Ishimaru, T.; Matsuoka, K.; Okaichi, T.; Maala, A.M.; Ordones, J.A. Occurence of Gymnodinium catenatum in Manila Bay, the Philippines. Toxic Phytoplankton Blooms in the Sea; Elsevier Science Publishers BV: New York, NY, USA, 1993. [Google Scholar]
- Lirdwitayaprasit, T.; Panuksubaksul, D.; Takata, Y.; Sato, S.; Kodama, M.; Fukuyo, Y. Occurrence of Gymnodinium catenatum in the Gulf of Thailand. Mar. Res. Indones. 2008, 33, 87–89. [Google Scholar] [CrossRef]
- Liu, M.; Gu, H.; Krock, B.; Luo, Z.; Zhang, Y. Toxic Dinoflagellate Blooms of Gymnodinium catenatum and Their Cysts in Taiwan Strait and Their Relationship to Global Populations. Harmful Algae 2020, 97, 101868. [Google Scholar] [CrossRef] [PubMed]
- Lin, Z.-R.; Geng, H.-X.; Zhang, Q.-C.; Chen, Z.-F.; Dai, L.; Yu, R.-C. Toxin Production of Dinoflagellate Gymnodinium catenatum Isolated from the East China Sea. Harmful Algae 2022, 113, 102188. [Google Scholar] [CrossRef] [PubMed]
- Gu, H.; Zeng, N.; Xie, Z.; Wang, D.; Wang, W.; Yang, W. Morphology, Phylogeny, and Toxicity of Atama Complex (Dinophyceae) from the Chukchi Sea. Polar. Biol. 2013, 36, 427–436. [Google Scholar] [CrossRef]
- Gyoon, K.H.; Kazumi, M.; Geun, L.S.; Ho, A.K. The Occurrence of a Dinoflagellate Gymnodinium catenatum From Chinhae Bay, Korea. Korean J. Fish. Aquat. Sci. 1996, 29, 837–842. [Google Scholar]
- Matsuoka, K.; Fukuyo, Y. Geographical Distribution of the Toxic Dinoflagellate Gymnodinium catenatum Graham in Japanese Coastal Waters. Bot. Mar. 1994, 37, 495–504. [Google Scholar] [CrossRef]
- Yamamoto, T.; Oh, S.J.; Kataoka, Y. Growth and Uptake Kinetics for Nitrate, Ammonium and Phosphate by the Toxic Dinoflagellate Gymnodinium catenatum Isolated from Hiroshima Bay, Japan. Fish. Sci. 2004, 70, 108–115. [Google Scholar] [CrossRef]
- Orlova, T.Y.; Morozova, T.V.; Gribble, K.E.; Kulis, D.M.; Anderson, D.M. Dinoflagellate Cysts in Recent Marine Sediments from the East Coast of Russia. Bot. Mar. 2004, 47, 184–201. [Google Scholar] [CrossRef]
- Chivers, W.J.; Walne, A.W.; Hays, G.C. Mismatch between Marine Plankton Range Movements and the Velocity of Climate Change. Nat. Commun. 2017, 8, 14434. [Google Scholar] [CrossRef]
- Thomas, M.K.; Kremer, C.T.; Klausmeier, C.A.; Litchman, E. A Global Pattern of Thermal Adaptation in Marine Phytoplankton. Science 2012, 338, 1085–1088. [Google Scholar] [CrossRef]
- Glibert, P.M.; Icarus Allen, J.; Artioli, Y.; Beusen, A.; Bouwman, L.; Harle, J.; Holmes, R.; Holt, J. Vulnerability of Coastal Ecosystems to Changes in Harmful Algal Bloom Distribution in Response to Climate Change: Projections Based on Model Analysis. Glob. Change Biol. 2014, 20, 3845–3858. [Google Scholar] [CrossRef]
- Continuous Plankton Recorder Survey Team. Continuous Plankton Records: Plankton Atlas of the North Atlantic Ocean (1958–1999). II. Biogeographical Charts. Mar. Ecol. Prog. Ser. 2004, 11–75. [Google Scholar]
- Baldrich, Á.M.; Molinet, C.; Reguera, B.; Espinoza-González, O.; Pizarro, G.; Rodríguez-Villegas, C.; Opazo, D.; Mejías, P.; Díaz, P.A. Interannual Variability in Mesoscale Distribution of Dinophysis Acuminata and D. Acuta in Northwestern Patagonian Fjords. Harmful Algae 2022, 115, 102228. [Google Scholar] [CrossRef]
- Oziel, L.; Baudena, A.; Ardyna, M.; Massicotte, P.; Randelhoff, A.; Sallée, J.-B.; Ingvaldsen, R.B.; Devred, E.; Babin, M. Faster Atlantic Currents Drive Poleward Expansion of Temperate Phytoplankton in the Arctic Ocean. Nat. Commun. 2020, 11, 1705. [Google Scholar] [CrossRef]
- Araújo, M.B.; Pearson, R.G.; Rahbek, C. Equilibrium of Species’ Distributions with Climate. Ecography 2005, 28, 693–695. [Google Scholar] [CrossRef]
- Casabianca, S.; Penna, A.; Pecchioli, E.; Jordi, A.; Basterretxea, G.; Vernesi, C. Population Genetic Structure and Connectivity of the Harmful Dinoflagellate Alexandrium minutum in the Mediterranean Sea. Proc. Royal Soc. B 2012, 279, 129–138. [Google Scholar] [CrossRef]
- Beck, J.; Böller, M.; Erhardt, A.; Schwanghart, W. Spatial Bias in the GBIF Database and Its Effect on Modeling Species’ Geographic Distributions. Ecol. Inform. 2014, 19, 10–15. [Google Scholar] [CrossRef]
- Lürling, M. Grazing Resistance in Phytoplankton. Hydrobiologia 2021, 848, 237–249. [Google Scholar] [CrossRef]
- Trainor, A.M.; Schmitz, O.J.; Ivan, J.S.; Shenk, T.M. Enhancing Species Distribution Modeling by Characterizing Predator-Prey Interactions. Ecol. Appl. 2014, 24, 204–216. [Google Scholar] [CrossRef]
- Stockwell, D.R.B.; Peterson, A.T. Controlling Bias in Biodiversity Data. In Predicting Species Occurrences: Issues of Scale and Accuracy; Island Press: Washington, DC, USA, 2002; pp. 537–546. [Google Scholar]


| Species | Pre-Curation | Post-Curation |
|---|---|---|
| Alexandrium catenella | 113 | 108 |
| Alexandrium minutum | 205 | 181 |
| Gymnodinium catenatum | 244 | 116 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Borges, F.O.; Lopes, V.M.; Amorim, A.; Santos, C.F.; Costa, P.R.; Rosa, R. Projecting Future Climate Change-Mediated Impacts in Three Paralytic Shellfish Toxins-Producing Dinoflagellate Species. Biology 2022, 11, 1424. https://doi.org/10.3390/biology11101424
Borges FO, Lopes VM, Amorim A, Santos CF, Costa PR, Rosa R. Projecting Future Climate Change-Mediated Impacts in Three Paralytic Shellfish Toxins-Producing Dinoflagellate Species. Biology. 2022; 11(10):1424. https://doi.org/10.3390/biology11101424
Chicago/Turabian StyleBorges, Francisco O., Vanessa M. Lopes, Ana Amorim, Catarina F. Santos, Pedro Reis Costa, and Rui Rosa. 2022. "Projecting Future Climate Change-Mediated Impacts in Three Paralytic Shellfish Toxins-Producing Dinoflagellate Species" Biology 11, no. 10: 1424. https://doi.org/10.3390/biology11101424
APA StyleBorges, F. O., Lopes, V. M., Amorim, A., Santos, C. F., Costa, P. R., & Rosa, R. (2022). Projecting Future Climate Change-Mediated Impacts in Three Paralytic Shellfish Toxins-Producing Dinoflagellate Species. Biology, 11(10), 1424. https://doi.org/10.3390/biology11101424








