Comparative Analysis of the Complete Mitochondrial Genomes of Five Species of Ricaniidae (Hemiptera: Fulgoromorpha) and Phylogenetic Implications
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Preparation and DNA Extraction
2.2. Sequencing, Assembly, Annotation, and Analysis
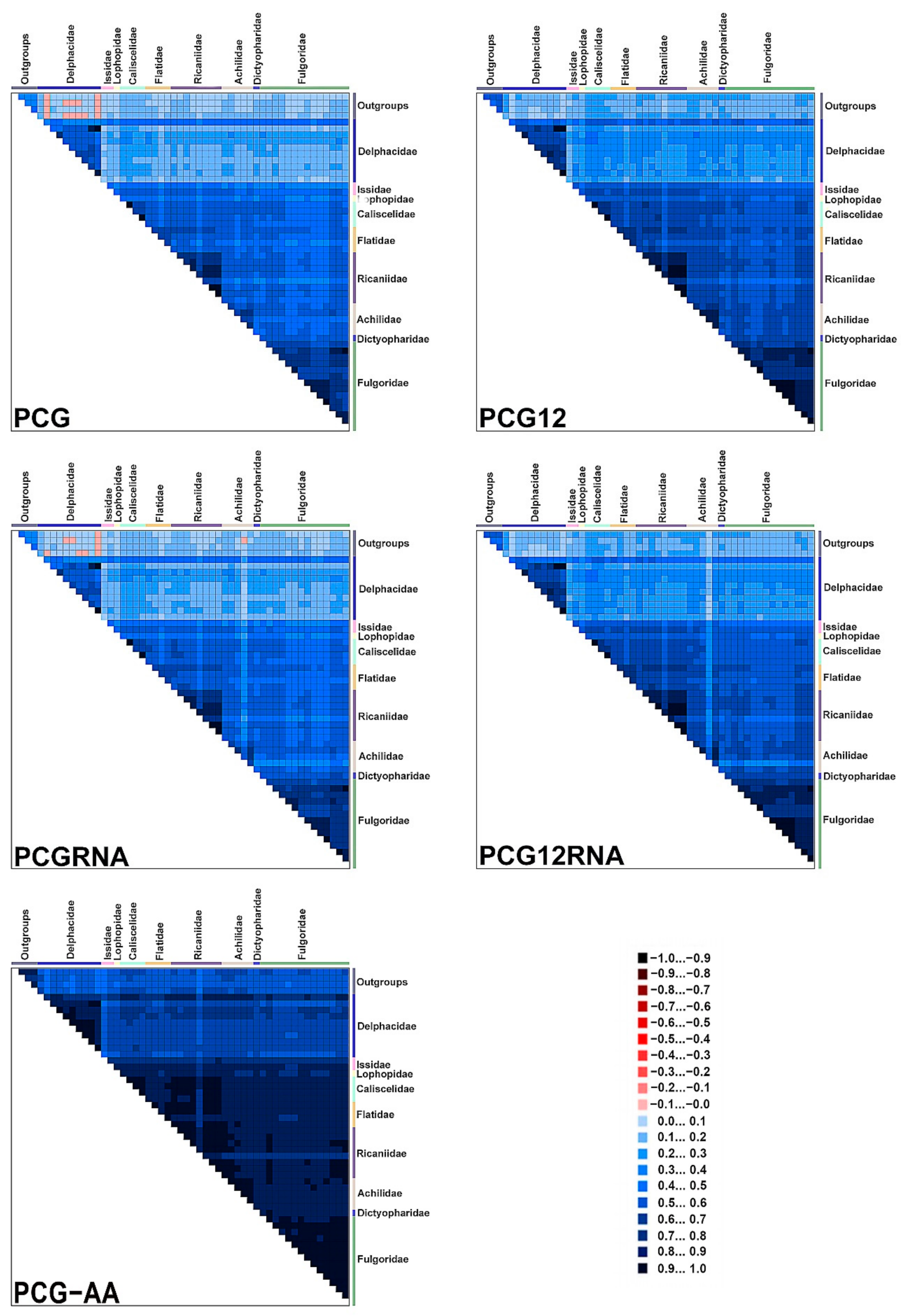
2.3. Mitogenome Sequence Alignment and Analyses of Sequence Heterogeneity
2.4. Phylogenetic Analyses
3. Results
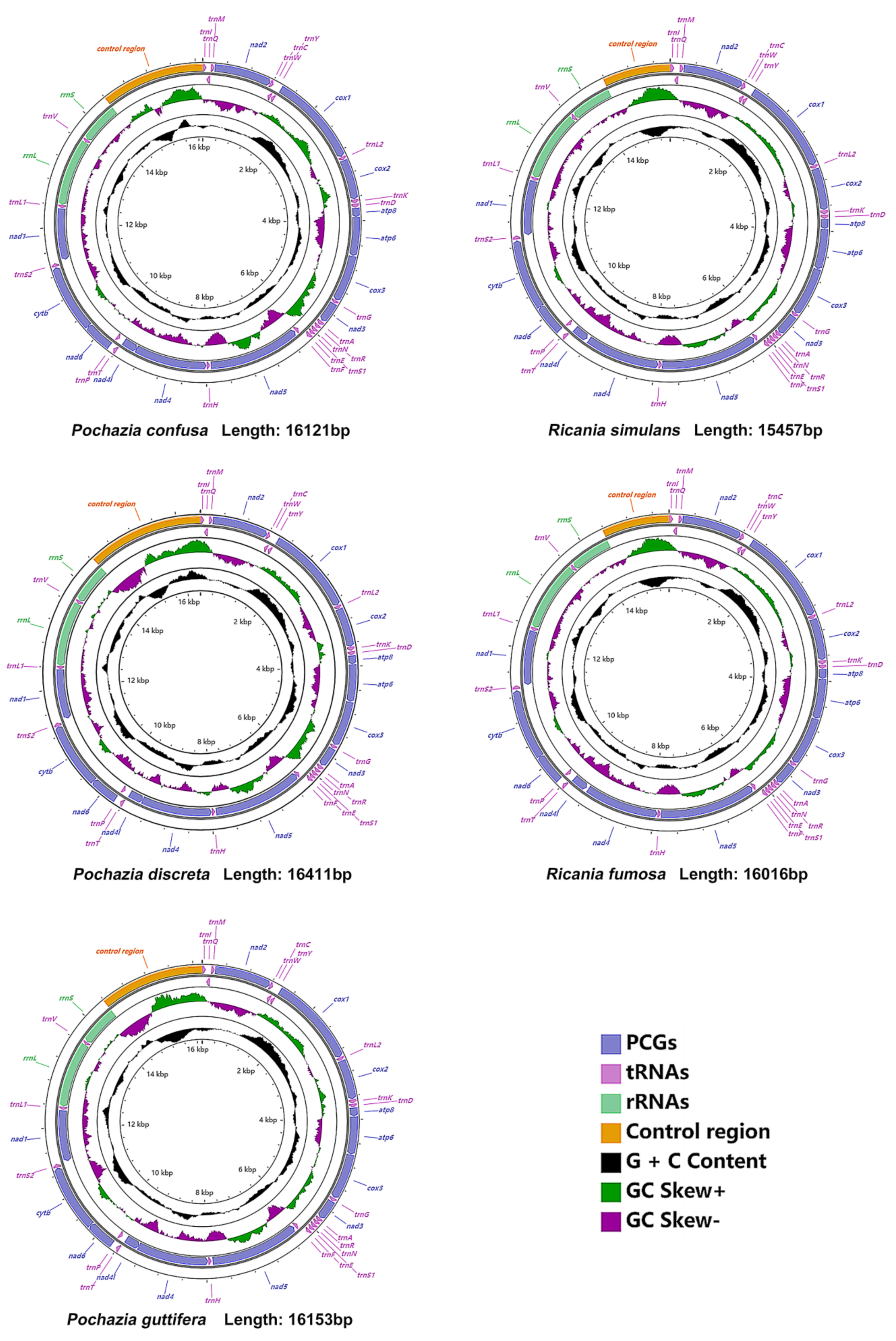
3.1. Mitogenome Organization and Base Composition
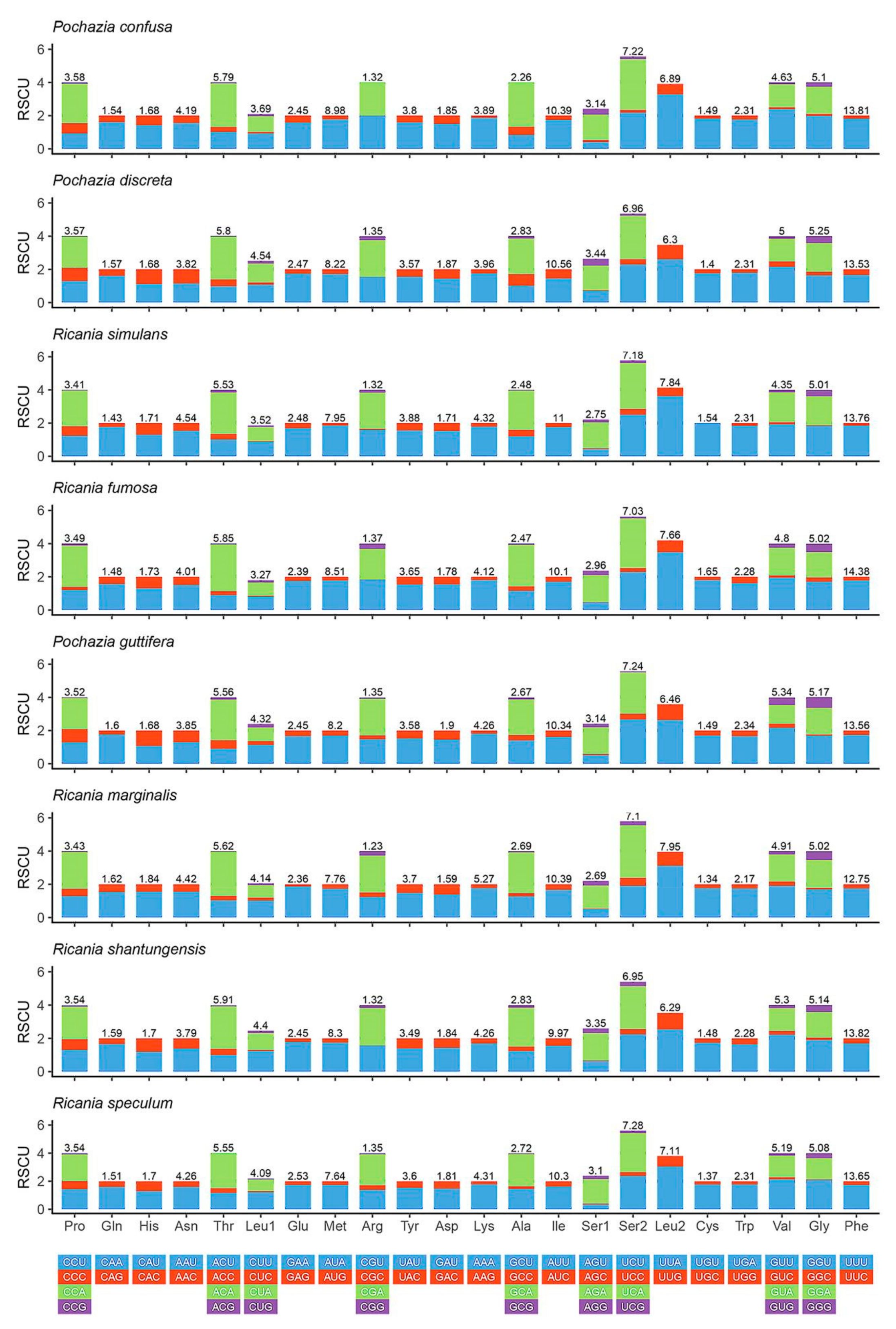
3.2. Protein-Coding Genes and Codon Usage
3.3. Transfer and Ribosomal RNA Genes
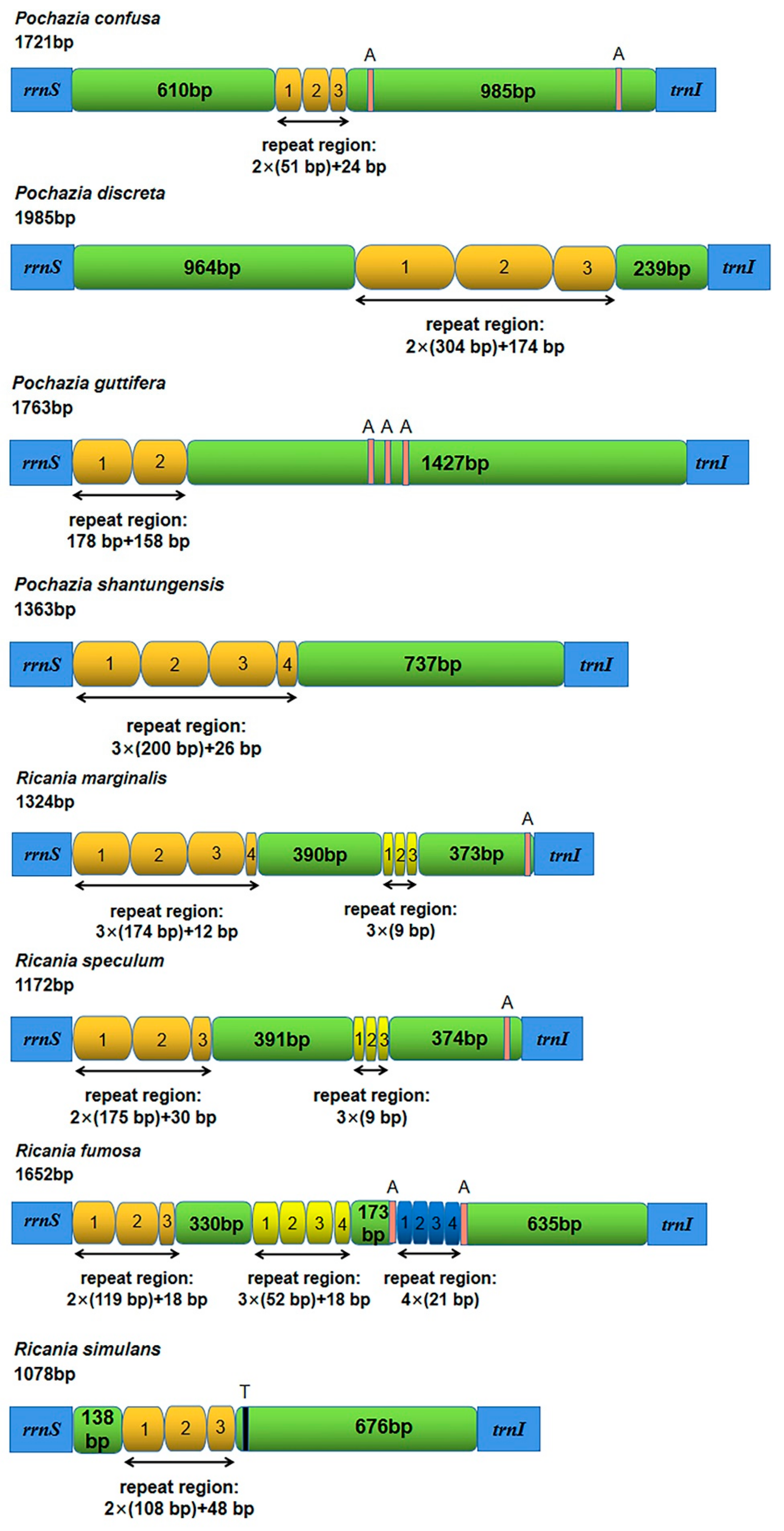
3.4. The Control Region
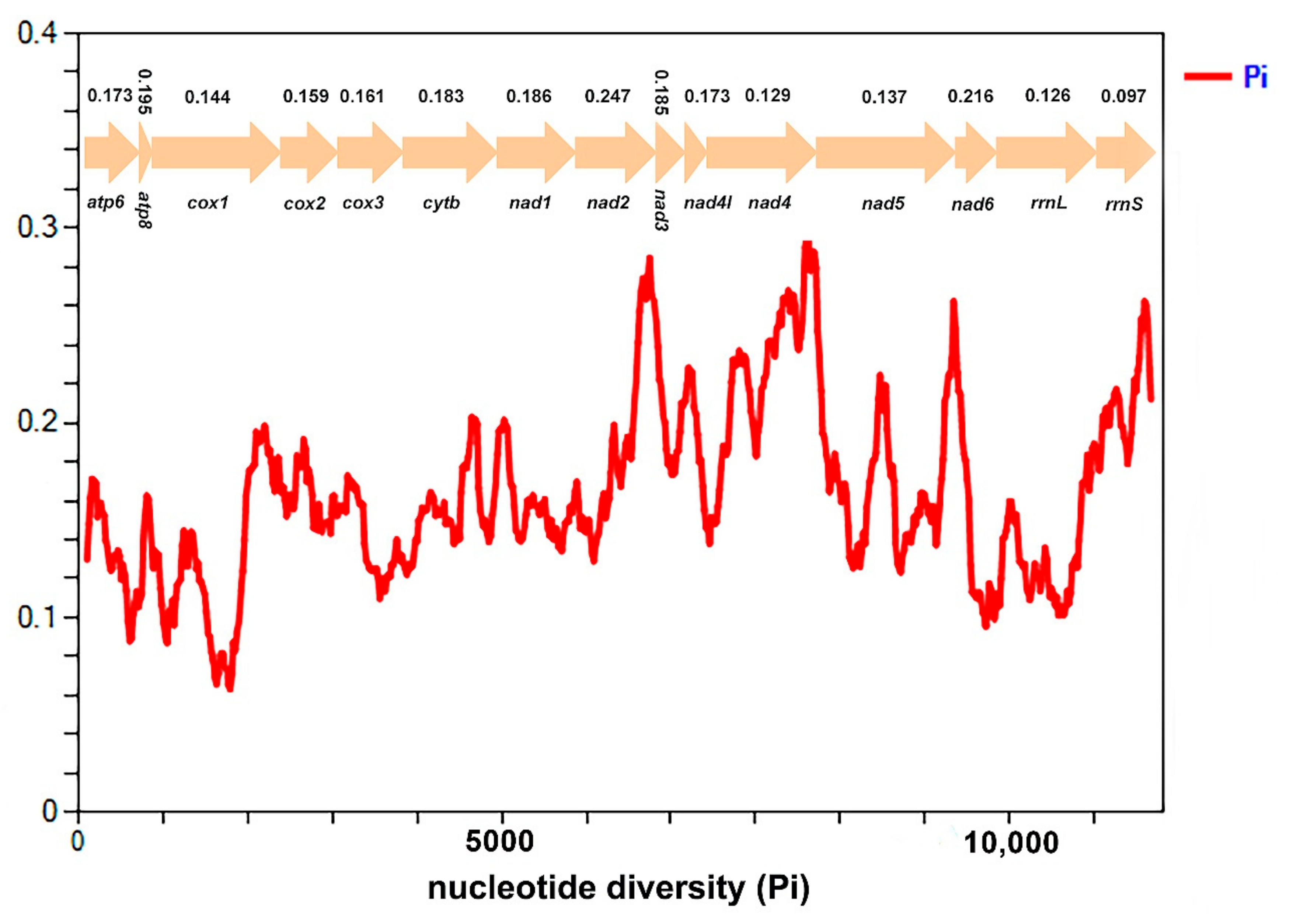
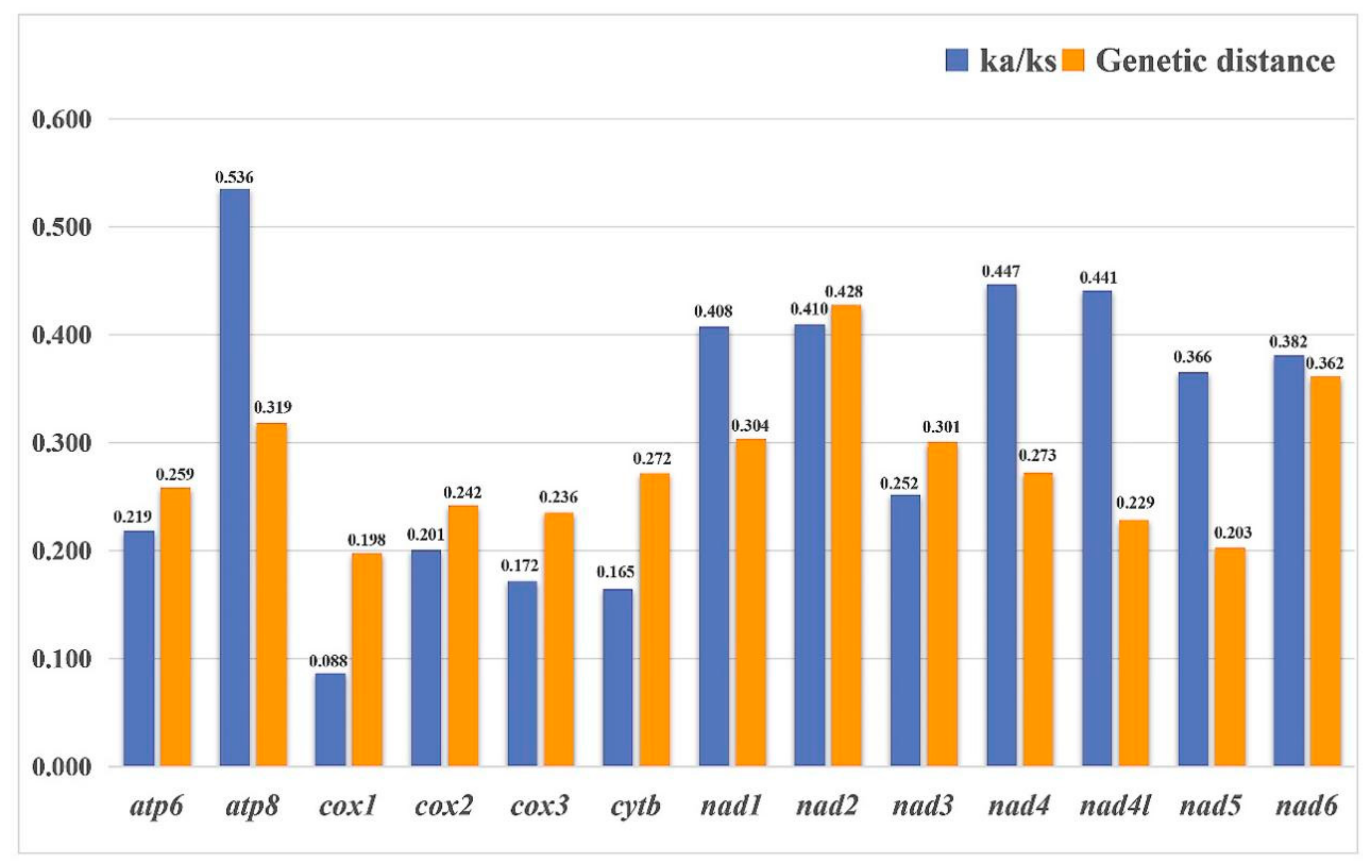
3.5. Nucleotide Diversity and Genetic Distance
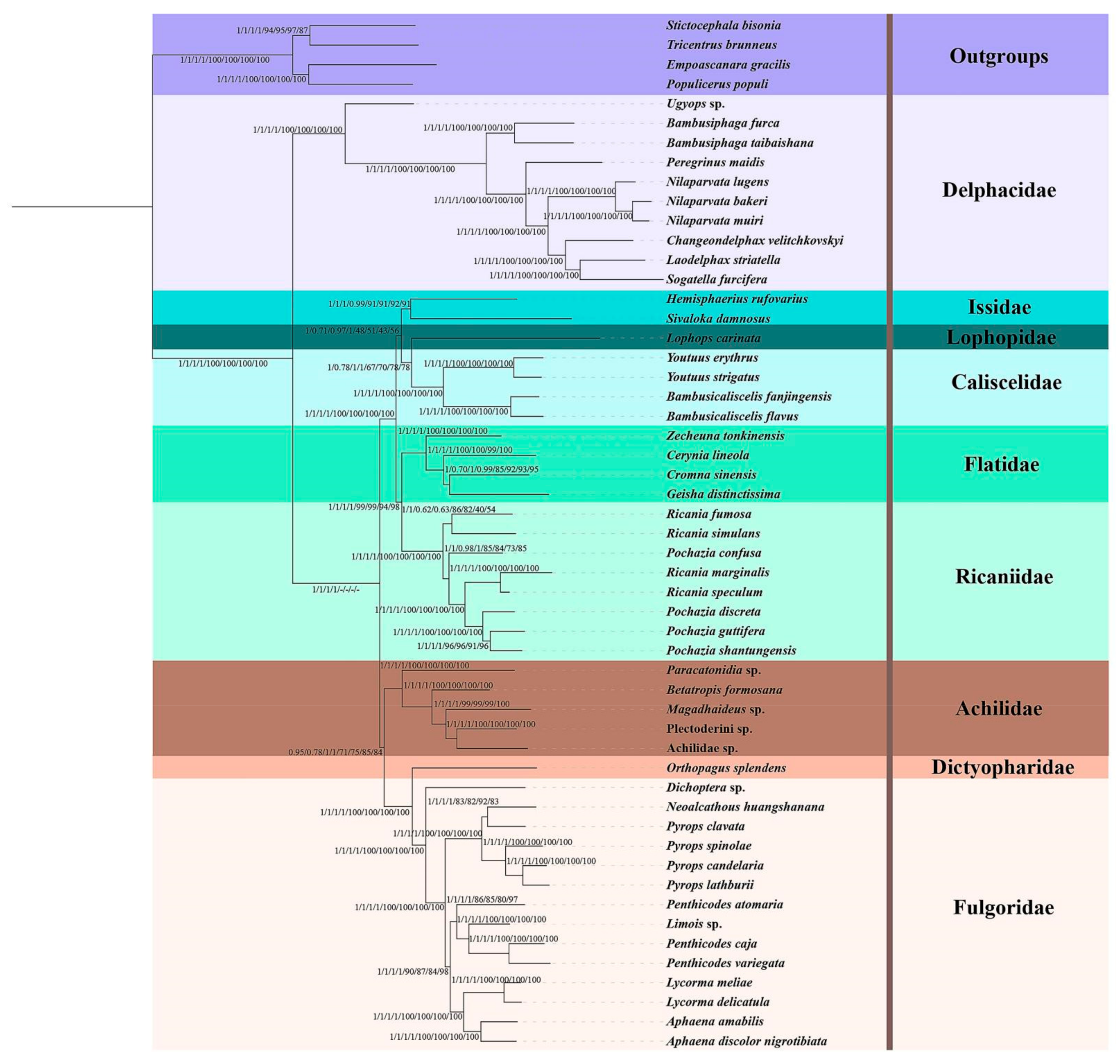
3.6. Phylogenetic Analyses
4. Discussion
4.1. Comparative Analysis of Ricaniid Mitogenomes
4.2. Nucleotide Diversity of Ricaniid Mitogenomes
4.3. Phylogeny
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bourgoin, T. FLOW (Fulgoromorpha Lists on the Web), Version 8. Available online: http://hemiptera-databases.org/flow/ (accessed on 19 August 2021).
- Rossi, E.; Stroiński, A.; Lucchi, A. Egg morphology, laying behavior and record of the host plants of Ricania speculum (Walker, 1851), a new alien species for Europe (Hemiptera: Ricaniidae). Zootaxa 2015, 4044, 93–104. [Google Scholar] [CrossRef]
- Bourgoin, T. Female genitalia in Hemiptera Fulgoromorpha, morphological and phylogenetic data. Ann. Soc. Entomol. Fr. 1993, 29, 225–244. [Google Scholar]
- Muir, F. On the classification of the Fulgoroidea (Homoptera). Ann. Mag. Nat. Hist. 1930, 10, 461–478. [Google Scholar] [CrossRef]
- Chen, S.; Yang, C.T. The metatarsi of the Fulgoroidea (Homoptera: Auchenorrhyncha). Chin. J. Entomol. 1995, 15, 257–269. [Google Scholar]
- Emeljanov, A.F. An attempt of construction of phylogenetic tree of the planthoppers (Homoptera, Cicadina). Entomol. Obozr. 1990, 69, 353–356. [Google Scholar]
- Yeh, W.B.; Yang, C.T.; Hui, C.F. A molecular phylogeny of planthoppers (Hemiptera: Fulgoroidea) inferred from mitochondrial 16S rDNA sequences. Zool. Stud. 2005, 44, 519–535. [Google Scholar]
- Urban, J.M.; Cryan, J.R. Evolution of the planthoppers (Insecta: Hemiptera: Fulgoroidea). Mol. Phylogenet. Evol. 2007, 42, 556–572. [Google Scholar] [CrossRef] [PubMed]
- Song, N.; Liang, A.P. A preliminary molecular phylogeny of planthoppers (Hemiptera: Fulgoroidea) based on nuclear and mitochondrial DNA sequences. PLoS ONE 2013, 8, e58400. [Google Scholar] [CrossRef]
- Fletcher, M.J. A key to the genera of Ricaniidae (Hemiptera: Fulgoromorpha) recorded in Australia with notes on the Australian fauna, including a new species of Epithalamium Kirkaldy. Aust. J. Entomol. 2008, 47, 107–120. [Google Scholar] [CrossRef]
- Bu, C.P.; Liang, A.P. First record of the genus Aprivesa Melichar (Hemiptera, Fulgoromorpha) from South India, with description of one new species. Zookeys 2011, 81, 1–12. [Google Scholar] [CrossRef]
- Rahman, M.A.; Kwon, Y.J.; Suh, S.J.; Youn, Y.N.; Jo, S.H. The genus Pochazia Amyot and Serville (Hemiptera: Ricaniidae) from Korea, with a newly recorded species. J. Entomol. 2012, 9, 239–247. [Google Scholar] [CrossRef]
- Kwon, D.H.; Kim, S.J.; Kang, T.J.; Lee, J.H.; Kim, D.H. Analysis of the molecular phylogenetics and genetic structure of an invasive alien species, Ricania shantungensis, in Korea. J. Asia-Pac. Entomol. 2017, 20, 901–906. [Google Scholar] [CrossRef]
- Baek, S.; Lee, J.H.; Park, D.K.; Park, Y.; Kim, M.J.; Kim, K.; Seo, H.Y.; Choi, Y.S. Within-tree distribution of Ricania shantungensis (Hemiptera: Ricaniidae) eggs on chestnuts. J. Asia-Pac. Entomol. 2020, 23, 536–539. [Google Scholar] [CrossRef]
- Park, J.; Jung, S. On the scientific name of the invaded planthopper (Hemiptera: Fulgoroidea: Ricaniidae) in Korea. Korean J. Appl. Entomol. 2020, 59, 317–323. [Google Scholar] [CrossRef]
- Kang, J.Y.; An, I.; Park, S. The complete mitochondrial genome of Ricania shantungensis (Hemiptera: Ricaniidae) in Korea. Mitochondrial DNA Part B 2020, 5, 3813–3814. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Park, J.; Xi, H.; Lee, G.S.; Kim, I.; Park, J.; Lee, W. The complete mitochondrial genome of Ricania speculum (Walker, 1851) (Hemiptera: Ricaniidae): Investigation of intraspecific variations on mitochondrial genome. Mitochondrial DNA Part B 2020, 5, 3814–3816. [Google Scholar] [CrossRef]
- Song, N.; Liang, A.P.; Bu, C.P. A molecular phylogeny of Hemiptera inferred from mitochondrial genome sequences. PLoS ONE 2012, 7, e48778. [Google Scholar] [CrossRef]
- Kearse, M.; Moir, R.; Wilson, A.; Havas, S.S.; Cheung, M.; Sturrock, S.; Buxton, S.; Cooper, A.; Markowitz, S.; Duran, C.; et al. Geneious Basic: An integrated and extendable desktop software platform for the organization and analysis of sequence data. Bioinformatics 2012, 28, 1647–1649. [Google Scholar] [CrossRef]
- Grant, J.R.; Stothard, P. The CG View Server: A comparative genomics tool for circular genomes. Nucleic Acids Res. 2008, 36, W181–W184. [Google Scholar] [CrossRef]
- Bernt, M.; Donath, A.; Jühling, F.; Externbrink, F.; Florentz, C.; Fritzsch, G.; JoernPütz, J.; Middendorf, M.; Stadler, P.F. MITOS: Improved de novo metazoan mitochondrial genome annotation. Mol. Phylogenet. Evol. 2013, 69, 313–319. [Google Scholar] [CrossRef]
- Zhang, D.; Gao, F.; Jakovlić, I.; Zou, H.; Zhang, J.; Li, W.X.; Wang, G.T. PhyloSuite: An integrated and scalable desktop platform for streamlined molecular sequence data management and evolutionary phylogenetics studies. Mol. Ecol. Resour. 2020, 20, 348–355. [Google Scholar] [CrossRef] [PubMed]
- Benson, G. Tandem repeats finder: A program to analyze DNA sequences. Nucleic Acids Res. 1999, 27, 573–580. [Google Scholar] [CrossRef] [PubMed]
- Rozas, J.; Ferrer-Mata, A.; Sánchez-DelBarrio, J.C.; Guirao-Rico, S.; Librado, P.; Ramos-Onsins, S.E.; Sánchez-Gracia, A. DnaSP 6: DNA sequence polymorphism analysis of large datasets. Mol. Biol. Evol. 2017, 34, 3299–3302. [Google Scholar] [CrossRef]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular evolutionary genetics analysis across computing platforms. Mol. Biol. Evol. 2018, 35, 1547–1549. [Google Scholar] [CrossRef]
- Katoh, K.; Standley, D.M. MAFFT multiple sequence alignment software version 7: Improvements in performance and usability. Mol. Biol. Evol. 2013, 30, 772–780. [Google Scholar] [CrossRef]
- Talavera, G.; Castresana, J. Improvement of phylogenies after removing divergent and ambiguously aligned blocks from protein sequence alignments. Syst. Biol. 2007, 56, 564–577. [Google Scholar] [CrossRef]
- Kück, P.; Meid, S.A.; Groß, C.; Wägele, J.W.; Misof, B. AliGROOVE—Visualization of heterogeneous sequence divergence within multiple sequence alignments and detection of inflated branch support. BMC Bioinform. 2014, 15, 294. [Google Scholar] [CrossRef]
- Lanfear, R.; Frandsen, P.B.; Wright, A.M.; Senfeld, T.; Calcott, B. PartitionFinder 2: New methods for selecting partitioned models of evolution for molecular and morphological phylogenetic analyses. Mol. Biol. Evol. 2017, 34, 772–773. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, L.T.; Schmidt, H.A.; Haeseler, A.V.; Minh, B.Q. IQ−TREE: A fast and effective stochastic algorithm for estimating maximum−likelihood phylogenies. Mol. Biol. Evol. 2015, 32, 268–274. [Google Scholar] [CrossRef] [PubMed]
- Ronquist, F.; Teslenko, M.; Mark, P.V.D.; Ayres, D.L.; Darling, A.; Höhna, S.; Larget, B.; Liu, L.; Suchard, M.A.; Huelsenbeck, J.P. MrBayes 3.2: Efficient Bayesian phylogenetic inference and model choice across a large model space. Syst. Biol. 2012, 61, 539–542. [Google Scholar] [CrossRef] [PubMed]
- Miller, M.; Pfeiffer, W.T.; Schwartz, T. Creating the CIPRES science gateway for inference of large phylogenetic trees. In Proceedings of the Gateway Computing Environments Workshop (GCE), New Orleans, LA, USA, 14 November 2010; Institute of Electrical and Electronics Engineers (IEEE): New Orleans, LA, USA, 2010; pp. 1–8. [Google Scholar] [CrossRef]
- Wang, W.Q.; Huang, Y.X.; Bartlett, C.R.; Zhou, F.M.; Meng, R.; Qin, D.Z. Characterization of the complete mitochondrial genomes of two species of the genus Aphaena Guérin-Méneville (Hemiptera: Fulgoridae) and its phylogenetic implications. Int. J. Biol. Macromol. 2019, 141, 29–40. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.Q.; Zhang, H.; Constant, J.; Bartlett, C.R.; Qin, D.Z. Characterization, comparative analysis and phylogenetic implications of mitogenomes of Fulgoridae (Hemiptera: Fulgoromorpha). Genes 2021, 12, 1185. [Google Scholar] [CrossRef]
- Xu, S.Y.; Long, J.K.; Chen, X.S. Comparative analysis of the complete mitochondrial genomes of five Achilidae species (Hemiptera: Fulgoroidea) and other Fulgoroidea reveals conserved mitochondrial genome organization. PeerJ 2019, 7, e6659. [Google Scholar] [CrossRef] [PubMed]
- Ai, D.Q.; Peng, L.F.; Qin, D.Z.; Zhang, Y.L. Characterization of three complete mitogenomes of Flatidae (Hemiptera: Fulgoroidea) and compositional heterogeneity analysis in the planthoppers’ mitochondrial phylogenomics. Int. J. Mol. Sci. 2021, 22, 5586. [Google Scholar] [CrossRef] [PubMed]
- Gong, N.; Yang, L.; Chen, X.S. Structural features and phylogenetic implications of four new mitogenomes of Caliscelidae (Hemiptera: Fulgoromorpha). Int. J. Mol. Sci. 2021, 22, 1348. [Google Scholar] [CrossRef]
- Ceotto, P.; Kergoat, G.J.; Rasplus, J.Y.; Bourgoin, T. Molecular phylogenetics of cixiid planthoppers (Hemiptera: Fulgoromorpha): New insights from combined analyses of mitochondrial and nuclear genes. Mol. Phylogenet. Evol. 2008, 48, 667–678. [Google Scholar] [CrossRef]
- Urban, J.M.; Cryan, J.R. Entomologically famous, evolutionarily unexplored: The first phylogeny of the lanternfly family Fulgoridae (Insecta: Hemiptera: Fulgoroidea). Mol. Phylogenet. Evol. 2009, 50, 471–484. [Google Scholar] [CrossRef]
- Huang, Y.X.; Zheng, L.F.; Bartlett, C.R.; Qin, D.Z. Resolving phylogenetic relationships of Delphacini and Tropidocephalini (Hemiptera: Delphacidae: Delphacinae) as inferred from four genetic loci. Sci. Rep. 2017, 7, 3319. [Google Scholar] [CrossRef]
- Urban, J.M.; Bartlett, C.R.; Cryan, J.R. Evolution of Delphacidae (Hemiptera: Fulgoroidea): Combined-evidence phylogenetics reveals importance of grass host shifts. Syst. Entomol. 2010, 35, 678–691. [Google Scholar] [CrossRef]
- Wang, M.L.; Zhang, Y.L.; Bourgoin, T. Planthopper family Issidae (Insecta: Hemiptera: Fulgoromorpha): Linking molecular phylogeny with classification. Mol. Phylogenet. Evol. 2016, 105, 224–234. [Google Scholar] [CrossRef]
- Gnezdilov, V.M.; Bourgoin, T.; Mozaffarian, F.; Manzari, S. Difficulties in building a molecular phylogeny of the issidoid planthopper lineages (Insecta: Hemiptera: Fulgoroidea). In Proceedings of the 1st Iranian International Congress of Entomology, Tehran, Iran, 29–31 August 2015; pp. 218–227. [Google Scholar]
- Gnezdilov, V.M.; Konstantinov, F.V.; Bodrov, S.Y. New insights into the molecular phylogeny and taxonomy of the family Issidae (Hemiptera: Auchenorrhyncha: Fulgoroidea). Proc. Zool. Inst. RAS 2020, 324, 146–161. [Google Scholar] [CrossRef][Green Version]
- Akiner, M.M.; Beris, F.S.; Seyis, F.; Ozturk, M.; Sevgili, H.; Demir, E. Annual variation of the Orosanga japonica Melichar 1898 (Hemiptera: Ricaniidae) populations in the eastern Black Sea region of Turkey and possible molecular separation with based on 28S rDNA sequences from other Ricaniidae groups. Plant Prot. Bull. 2019, 59, 11–19. [Google Scholar] [CrossRef]
- Bourgoin, T.; Gros, P.; Stroiński., A. Pochazia shantungensis (Chou & Lu, 1977), an important Asiatic invasive pest on fruit trees, first time reported from France (Hemiptera, Fulgoromorpha, Ricaniidae). Bull. Soc. Entomol. Fr. 2020, 125, 271–272. [Google Scholar] [CrossRef]

| Figure Taxon | Species | GenBank Number | References |
|---|---|---|---|
| Outgroups | |||
| Cicadellidae | Empoascanara gracilis Dworakowska | MT576649 | Unpublished |
| Populicerus populi (Linnaeus) | MH492318 | Wang et al., (2018) | |
| Membracidae | Stictocephala bisonia Kopp & Yonke | MW342606 | Yu et al., (2021) |
| Tricentrus brunneus Funkhouser | NC_044708 | Hu et al., (2019) | |
| Ingroup | |||
| Delphacidae | Ugyops sp. | MH352481 | Yu & Liang (2018) |
| Bambusiphaga furca Huang & Tian | NC_052689 | Huang et al., (2020) | |
| Bambusiphaga taibaishana Qin | NC_052690 | Huang et al., (2020) | |
| Changeondelphax velitchkovskyi (Melichar) | NC_037181 | Huang & Qin (2017) | |
| Peregrinus maidis (Ashmead) | NC_037182 | Huang & Qin (2017) | |
| Sogatella furcifera (Horváth) | KC512915 | Zhang et al., (2014) | |
| Laodelphax striatella (Fallén) | JX880068 | Zhang et al., (2013) | |
| Nilaparvata bakeri (Muir) | NC_033388 | Unpublished | |
| Nilaparvata lugens (Stål) | NC_021748 | Unpublished | |
| Nilaparvata muiri China | JN563998 | Lv et al., (2015) | |
| Achilidae | Achilidae sp. | MH324929 | Xu et al., (2019) |
| Betatropis formosana Matsumura | MH324927 | Xu et al., (2019) | |
| Plectoderini sp. | MH324930 | Xu et al., (2019) | |
| Paracatonidia sp. | MH324931 | Xu et al., (2019) | |
| Magadhaideus sp. | MH324928 | Xu et al., (2019) | |
| Dictyopharidae | Orthopagus splendens (Germar) | MW441850 | Unpublished |
| Fulgoridae | Aphaena amabilis (Hope) | NC_045075 | Wang et al., (2019) |
| Aphaena discolor nigrotibiata Schmidt | MN025523 | Wang et al., (2019) | |
| Lycorma delicatula (White) | MN607209 | Unpublished | |
| Lycorma meliae Kato | MT079725 | Du et al., (2021) | |
| Penthicodes atomaria (Weber) | MW662662 | Wang et al., (2021) | |
| Penthicodes variegata (Guérin-Méneville) | MW662664 | Wang et al., (2021) | |
| Penthicodes caja (Walker) | MW662663 | Wang et al., (2021) | |
| Limois sp. | MW662660 | Wang et al., (2021) | |
| Neoalcathous huangshanana Wang & Huang | MW662661 | Wang et al., (2021) | |
| Dichoptera sp. | MW662659 | Wang et al., (2021) | |
| Pyrops candelaria (Linné) | MW355618 | Duan & Hu (2021) | |
| Pyrops clavatus (Westwood) | MW662665 | Wang et al., (2021) | |
| Pyrops lathburii (Kirby) | MW662666 | Wang et al., (2021) | |
| Pyrops spinolae (Westwood) | MW662667 | Wang et al., (2021) | |
| Issidae | Sivaloka damnosus (Chou & Lu) | NC_014286 | Song et al., (2010) |
| Hemisphaerius rufovarius Walker | MT210096 | Yang et al., (2020) | |
| Lophopidae | Lophops carinata (Kirby) | NC_053739 | Xu & Chen (2021) |
| Caliscelidae | Bambusicaliscelis fanjingensis Chen & Zhang | MW281859 | Gong et al., (2021) |
| Bambusicaliscelis flavus Chen & Gong | MW281858 | Gong et al., (2021) | |
| Youtuus erythrus Gong, Yang & Chen | MW281861 | Gong et al., (2021) | |
| Youtuus strigatus Gong, Yang & Chen | MW281860 | Gong et al., (2021) | |
| Flatidae | Cromna sinensis (Walker) | MW872012 | Ai et al., (2021) |
| Cerynia lineola Melichar | MW872011 | Ai et al., (2021) | |
| Geisha distinctissima (Walker) | FJ230961 | Song & Liang (2009) | |
| Zecheuna tonkinensis Zia | MW872013 | Ai et al., (2021) | |
| Ricaniidae | Pochazia confusa Distant | MZ617458 | This study |
| Pochazia discreta Melichar | MZ673797 | This study | |
| Pochazia guttifera Walker | MZ617457 | This study | |
| Pochazia shantungensis (Chou & Lu) | MT898421 | Kang et al., (2020) | |
| Ricania speculum (Walker) | MT834932 | Lee et al., (2020) | |
| Ricania fumosa (Walker) | MZ617460 | This study | |
| Ricania marginalis (Walker) | NC_019597 | Song et al., (2012) | |
| Ricania simulans (Walker) | MZ617459 | This study |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, H.; Fang, W.; Zhao, X.; Jiang, X.; Stroiński, A.; Qin, D. Comparative Analysis of the Complete Mitochondrial Genomes of Five Species of Ricaniidae (Hemiptera: Fulgoromorpha) and Phylogenetic Implications. Biology 2022, 11, 92. https://doi.org/10.3390/biology11010092
Zhang H, Fang W, Zhao X, Jiang X, Stroiński A, Qin D. Comparative Analysis of the Complete Mitochondrial Genomes of Five Species of Ricaniidae (Hemiptera: Fulgoromorpha) and Phylogenetic Implications. Biology. 2022; 11(1):92. https://doi.org/10.3390/biology11010092
Chicago/Turabian StyleZhang, Huan, Wei Fang, Xiaoyun Zhao, Xin Jiang, Adam Stroiński, and Daozheng Qin. 2022. "Comparative Analysis of the Complete Mitochondrial Genomes of Five Species of Ricaniidae (Hemiptera: Fulgoromorpha) and Phylogenetic Implications" Biology 11, no. 1: 92. https://doi.org/10.3390/biology11010092
APA StyleZhang, H., Fang, W., Zhao, X., Jiang, X., Stroiński, A., & Qin, D. (2022). Comparative Analysis of the Complete Mitochondrial Genomes of Five Species of Ricaniidae (Hemiptera: Fulgoromorpha) and Phylogenetic Implications. Biology, 11(1), 92. https://doi.org/10.3390/biology11010092






