Circadian Clock Gene Period Contributes to Diapause via GABAeric-Diapause Hormone Pathway in Bombyx mori
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Silkworm Strain and Rearing
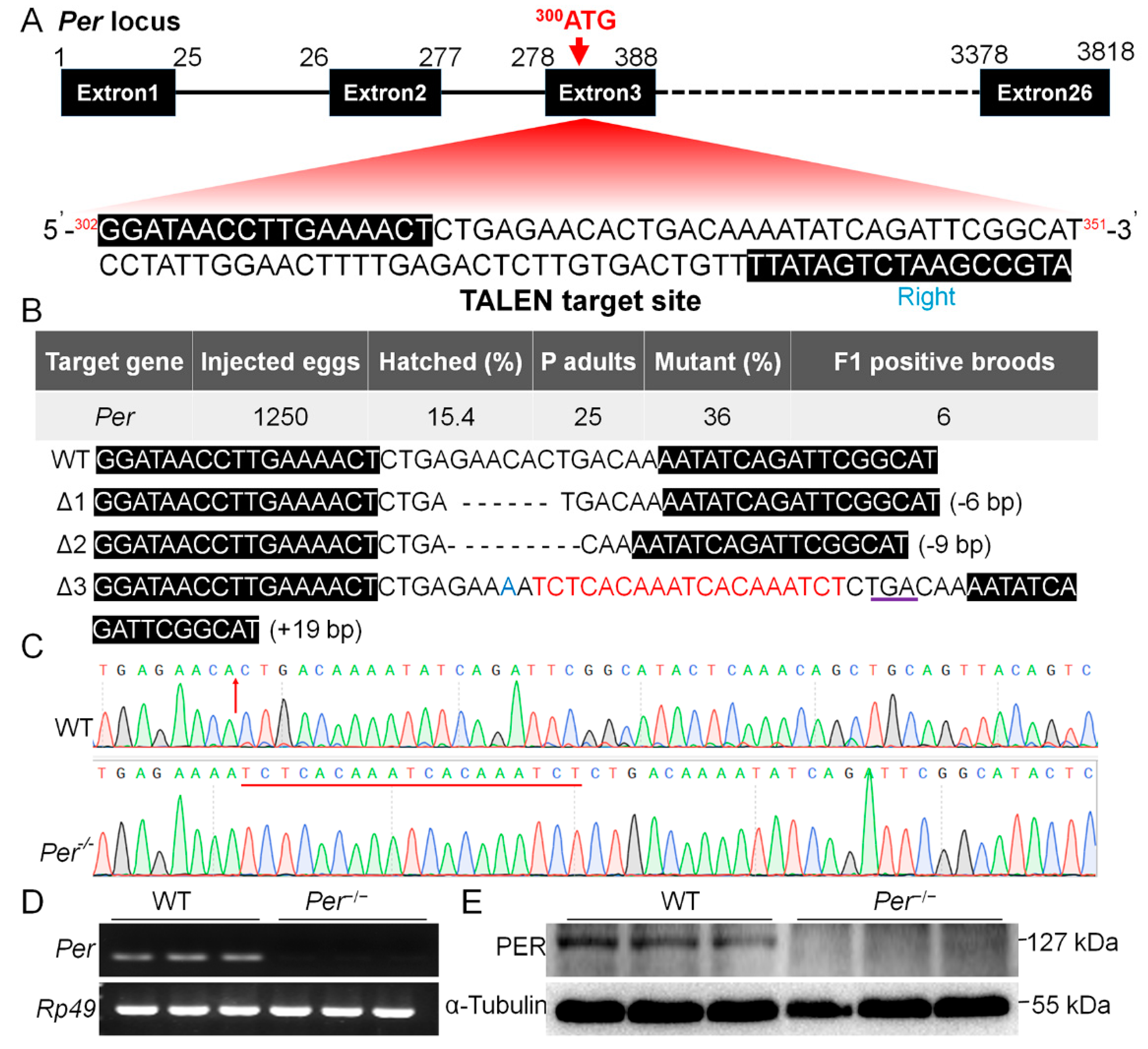
2.2. Plasmid Construction and Per Gene Knockout Mutant Screening
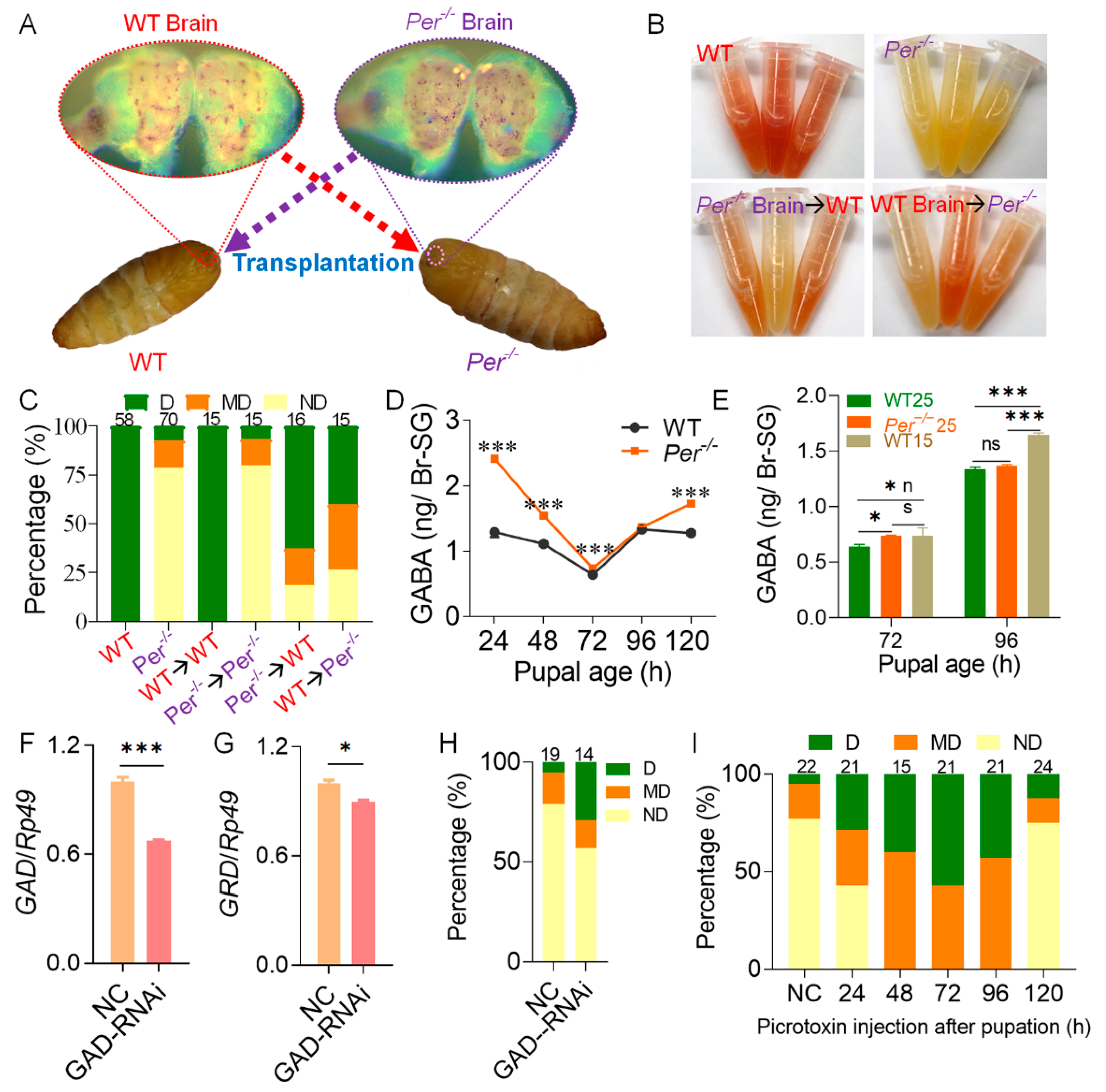
2.3. Brain Transplantation
2.4. Western Blot Analysis
2.5. Quantitative Real-Time PCR
2.6. RNA Interference
2.7. Liquid Chromatography-Mass Spectrometry/Mass Spectrometry (LC-MS/MS) Analysis
2.8. Ehrlich’s Diazo Reaction
2.9. Assay of Trehalose and Glycogen
2.10. Chromatin Immunoprecipitation and Sequencing (ChIP-Seq)
2.11. Dual Luciferase Reporter Assays
2.12. Statistical Analysis
3. Results
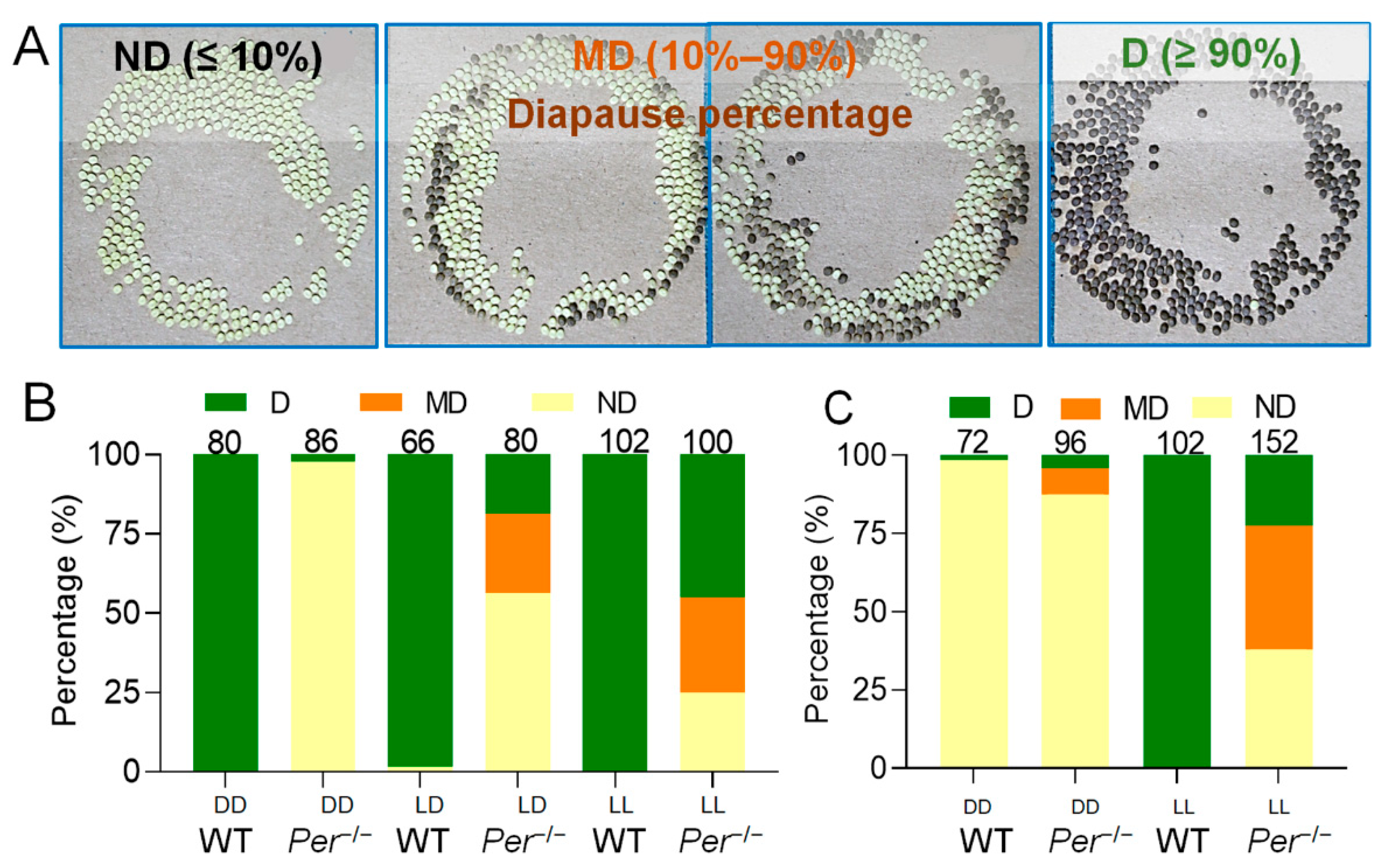
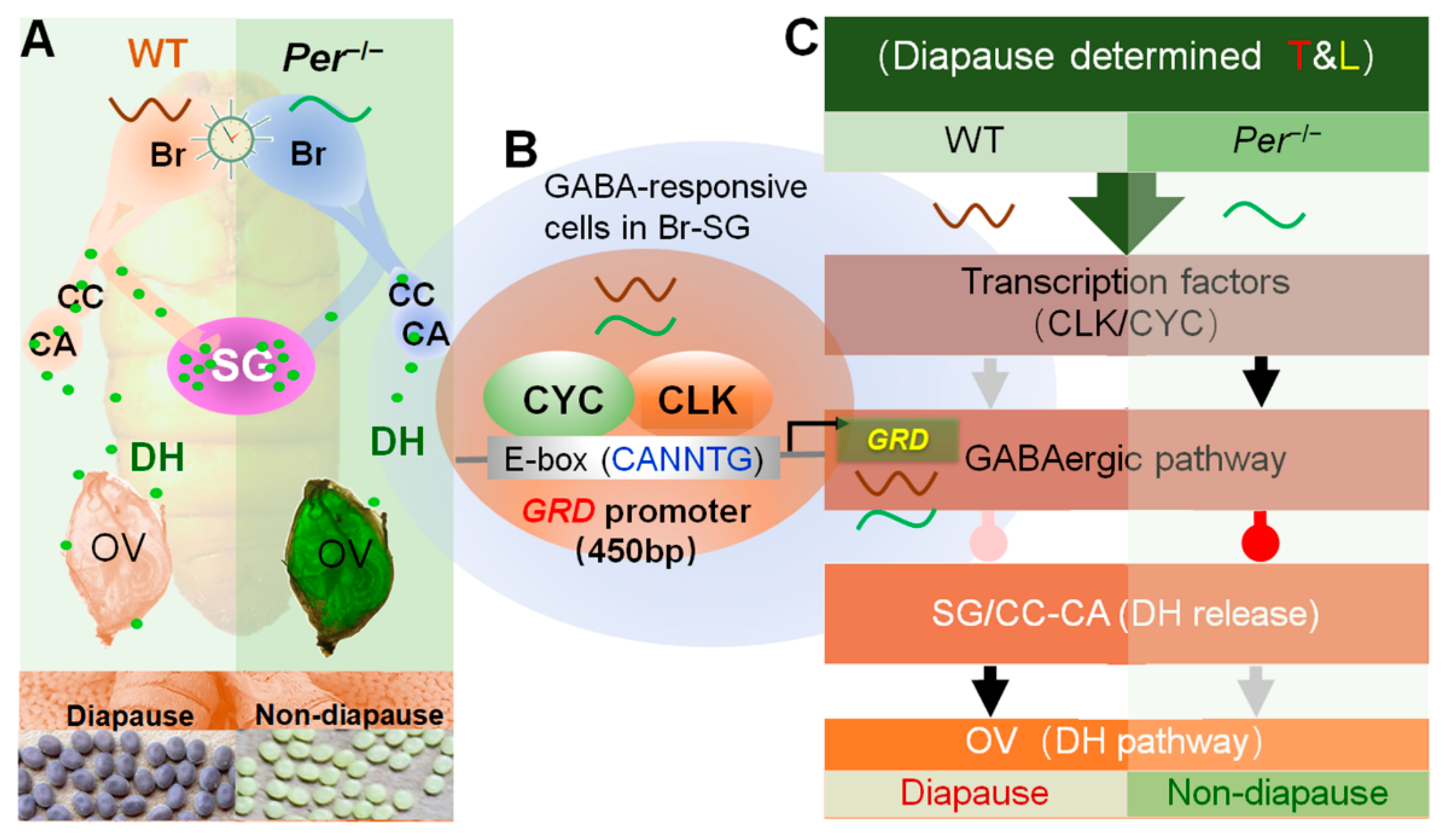
3.1. Per Expression Is Necessary for Diapause-Destiny in Bombyx mori
3.2. Deletion of Per Gene Inhibited the Secretion of DH through GABA Pathway in Brain
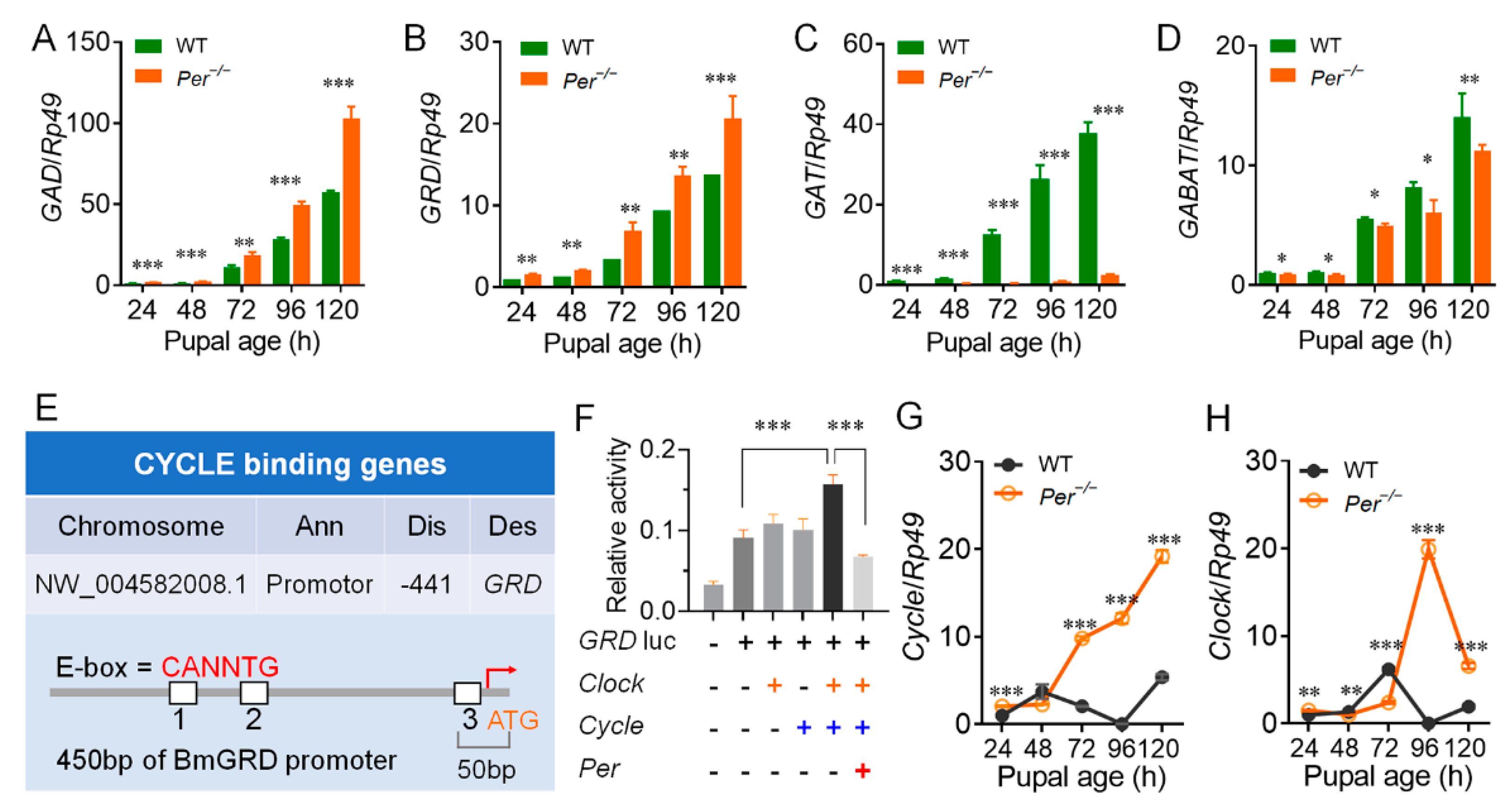
3.3. Per Directly Inhibited GABA Receptor GRD through Circadian Activators CYCLE and CLOCK
4. Discussion
- (1)
- Knockout of the circadian clock gene Per strongly changed the secretion and function of DH, which determined the diapause of silkworms.
- (2)
- The circadian clock of B. mori affected the DH levels and functions in the pupal stage through the GABA signaling pathway.
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sinturel, F.; Petrenko, V.; Dibner, C. Circadian Clocks Make Metabolism Run. J. Mol. Biol. 2020, 432, 3680–3699. [Google Scholar] [CrossRef]
- Sun, S.; Hanzawa, F.; Kim, D.; Umeki, M.; Nakajima, S.; Sakai, K.; Ikeda, S.; Mochizuki, S.; Oda, H. Circadian Rhythm-Dependent Induction of Hepatic Lipogenic Gene Expression in Rats Fed a High-Sucrose Diet. J. Biol. Chem. 2019, 294, 15206–15217. [Google Scholar] [CrossRef]
- Kumar Jha, P.; Challet, E.; Kalsbeek, A. Circadian Rhythms in Glucose and Lipid Metabolism in Nocturnal and Diurnal Mammals. Mol. Cell. Endocrinol. 2015, 418, 74–88. [Google Scholar] [CrossRef]
- Biancolin, A.D.; Martchenko, A.; Mitova, E.; Gurges, P.; Michalchyshyn, E.; Chalmers, J.A.; Doria, A.; Mychaleckyj, J.C.; Adriaenssens, A.E.; Reimann, F.; et al. The Core Clock Gene, Bmal1, and Its Downstream Target, the Snare Regulatory Protein Secretagogin, Are Necessary for Circadian Secretion of Glucagon-Like Peptide-1. Mol. Metab. 2020, 31, 124–137. [Google Scholar] [CrossRef]
- Walker II, W.H.; Walton, J.C.; DeVries, A.C.; Nelson, R.J. Circadian Rhythm Disruption and Mental Health. Transl. Psychiatry 2020, 10, 28. [Google Scholar] [CrossRef]
- Chaix, A.; Lin, T.; Le, H.D.; Chang, M.W.; Panda, S. Time-Restricted Feeding Prevents Obesity and Metabolic Syndrome in Mice Lacking a Circadian Clock. Cell Metab. 2019, 29, 303–319.e4. [Google Scholar] [CrossRef] [PubMed]
- Reinke, H.; Asher, G. Crosstalk between Metabolism and Circadian Clocks. Nat. Rev. Mol. Cell Biol. 2019, 20, 227–241. [Google Scholar] [CrossRef]
- Serin, Y.; Acar Tek, N. Effect of Circadian Rhythm on Metabolic Processes and the Regulation of Energy Balance. Ann. Nutr. Metab. 2019, 74, 322–330. [Google Scholar] [CrossRef]
- Panda, S. Circadian Physiology of Metabolism. Science 2016, 354, 1008–1015. [Google Scholar] [CrossRef]
- Sen, A.; Hoffmann, H.M. Role of Core Circadian Clock Genes in Hormone Release and Target Tissue Sensitivity in the Reproductive Axis. Mol. Cell. Endocrinol. 2020, 501, 110655. [Google Scholar] [CrossRef]
- Shimizu, T.; Hirai, Y.; Murayama, C.; Miyamoto, A.; Miyazaki, H.; Miyazaki, K. Circadian Clock Genes Per2 and Clock Regulate Steroid Production, Cell Proliferation, and Luteinizing Hormone Receptor Transcription in Ovarian Granulosa Cells. Biochem. Biophys. Res. Commun. 2011, 412, 132–135. [Google Scholar] [CrossRef]
- Lee, J.; Kim, M.S.; Li, R.; Liu, V.Y.; Fu, L.; Moore, D.D.; Ma, K.; Yechoor, V.K. Loss of Bmal1 Leads to Uncoupling and Impaired Glucose-Stimulated Insulin Secretion in Beta-Cells. Islets 2011, 3, 381–388. [Google Scholar] [CrossRef]
- Huang, J.; Zhong, Z.; Wang, M.; Chen, X.; Tan, Y.; Zhang, S.; He, W.; He, X.; Huang, G.; Lu, H.; et al. Circadian Modulation of Dopamine Levels and Dopaminergic Neuron Development Contributes to Attention Deficiency and Hyperactive Behavior. J. Neurosci. 2015, 35, 2572–2587. [Google Scholar] [CrossRef]
- Di Cara, F.; King-Jones, K. The Circadian Clock Is a Key Driver of Steroid Hormone Production in Drosophila. Curr. Biol. 2016, 26, 2469–2477. [Google Scholar] [CrossRef]
- Selcho, M.; Millan, C.; Palacios-Munoz, A.; Ruf, F.; Ubillo, L.; Chen, J.; Bergmann, G.; Ito, C.; Silva, V.; Wegener, C.; et al. Central and Peripheral Clocks Are Coupled by a Neuropeptide Pathway in Drosophila. Nat. Commun. 2017, 8, 15563. [Google Scholar] [CrossRef] [PubMed]
- Shiga, S.; Numata, H. Roles of Per Immunoreactive Neurons in Circadian Rhythms and Photoperiodism in the Blow Fly, Protophormia terraenovae. J. Exp. Biol. 2009, 212, 867–877. [Google Scholar] [CrossRef] [PubMed]
- Numata, H.; Hidaka, T. Termination of Adult Diapause by a Juvenile Hormone Analogue in the Bean Bug, Riptortus clavatus. Zool. Sci. 1984, 1, 751–754. [Google Scholar]
- Ikeno, T.; Tanaka, S.I.; Numata, H.; Goto, S.G. Photoperiodic Diapause under the Control of Circadian Clock Genes in an Insect. BMC Biol. 2010, 8, 116. [Google Scholar] [CrossRef]
- Holland, C.; Numata, K.; Rnjak-Kovacina, J.; Seib, F.P. The Biomedical Use of Silk: Past, Present, Future. Adv. Healthc. Mater. 2019, 8, e1800465. [Google Scholar] [CrossRef] [PubMed]
- Peng, Z.; Yang, X.; Liu, C.; Dong, Z.; Wang, F.; Wang, X.; Hu, W.; Zhang, X.; Zhao, P.; Xia, Q. Structural and Mechanical Properties of Silk from Different Instars of Bombyx mori. Biomacromolecules 2019, 20, 1203–1216. [Google Scholar] [CrossRef]
- Meng, X.; Abdlli, N.; Wang, N.; Lu, P.; Nie, Z.; Dong, X.; Lu, S.; Chen, K. Effects of Ag Nanoparticles on Growth and Fat Body Proteins in Silkworms (Bombyx mori). Biol. Trace Elem. Res. 2017, 180, 327–337. [Google Scholar] [CrossRef]
- Panthee, S.; Paudel, A.; Hamamoto, H.; Sekimizu, K. Advantages of the Silkworm as an Animal Model for Developing Novel Antimicrobial Agents. Front. Microbiol. 2017, 8, 373. [Google Scholar] [CrossRef]
- Sato, A.; Sokabe, T.; Kashio, M.; Yasukochi, Y.; Tominaga, M.; Shiomi, K. Embryonic Thermosensitive Trpa1 Determines Transgenerational Diapause Phenotype of the Silkworm, Bombyx mori. Proc. Natl. Acad. Sci. USA 2014, 111, E1249–E1255. [Google Scholar] [CrossRef]
- Tsurumaki, J.; Ishiguro, J.; Yamanaka, A.; Endo, K. Effects of Photoperiod and Temperature on Seasonal Morph Development and Diapause Egg Oviposition in a Bivoltine Race (Daizo) of the Silkmoth, Bombyx mori L. J. Insect Physiol. 1999, 45, 101–106. [Google Scholar] [CrossRef]
- Kobayashi, J.; Ebinuma, H.; Kobayashi, M. Effect of Temperature on the Diapause Egg Production in the Tropical Race of the Silkworm, Bombyx mori. J. Sericultural Sci. Jpn. 1986, 55, 343–348. [Google Scholar]
- Sonobe, H.; Ohnishi, E. Accumulation of 3-Hydroxykynurenine in Ovarian Follicles in Relation to Diapause in the Silkworm, Bombyx mori L. Dev. Growth Differ. 1970, 12, 41–52. [Google Scholar] [CrossRef]
- Jiang, T.; Chen, Y.; Tan, Z.; Li, J.; Qian, P.; Tang, S.; Shen, X. Expression Analysis and Functional Identification of Several Genes Related to Diapause in Bombyx mori. Dev. Growth Differ. 2019, 61, 150–157. [Google Scholar] [CrossRef]
- Shen, Z.; Jiang, X.; Yan, L.; Chen, Y.; Wang, W.; Shi, Y.; Shi, L.; Liu, D.; Zhou, N. Structural Basis for the Interaction of Diapause Hormone with Its Receptor in the Silkworm, Bombyx mori. FASEB J. 2018, 32, 1338–1353. [Google Scholar] [CrossRef]
- Hasegawa, K. Studies on the Mode of Action of the Diapause Hormone in the Silkworm, Bombyx mori L. II. Content of Diapause Hormone in the Suboesophageal Ganglion. J. Exp. Biol. 1964, 41, 855–863. [Google Scholar] [CrossRef]
- Ichikawa, T.; Aoki, S.; Shimizu, I. Neuroendocrine Control of Diapause Hormone Secretion in the Silkworm, Bombyx mori. J. Insect Physiol. 1997, 43, 1101–1109. [Google Scholar]
- Livak, K.J.; Schmittgen, T.D. Analysis of Relative Gene Expression Data Using Real-Time Quantitative Pcr and the 2(-Delta Delta C(T)) Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Heid, C.A.; Stevens, J.; Livak, K.J.; Williams, P.M. Real Time Quantitative Pcr. Genome Res. 1996, 6, 986–994. [Google Scholar] [CrossRef]
- Barca-Mayo, O.; Pons-Espinal, M.; Follert, P.; Armirotti, A.; Berdondini, L.; De Pietri Tonelli, D. Astrocyte Deletion of Bmal1 Alters Daily Locomotor Activity and Cognitive Functions via GABA Signalling. Nat. Commun. 2017, 8, 14336. [Google Scholar] [CrossRef]
- Xu, H.; Wang, Z.; Zhu, L.; Sui, Z.; Bi, W.; Liu, R.; Bi, K.; Li, Q. Targeted Neurotransmitters Profiling Identifies Metabolic Signatures in Rat Brain by Lc-Ms/Ms: Application in Insomnia, Depression and Alzheimer’s Disease. Molecules 2018, 23, 2375. [Google Scholar] [CrossRef]
- Hasegawa, K.; Shimizu, I. GABAergic Control of the Release of Diapause Hormone from the Suboesophageal Ganglion of the Silkworm, Bombyx mori. J. Insect Physiol. 1990, 36, 909–915. [Google Scholar] [CrossRef]
- Shimizu, I.; Matsui, T.; Hasegawa, K. Possible Involvement of GABAergic Neurons in Regulation of Diapause Hormone Secretion in the Silkworm, Bombyx mori. Zool. Sci. 1989, 6, 809–812. [Google Scholar]
- Symons, P.H. The Modifed Reaction Diazo Reaction in the Diagnosis of Typhoid Fever. S. Afr. Med. J. 1932, 6, 594–597. [Google Scholar]
- Hughes, M.E.; Hogenesch, J.B.; Kornacker, K. Jtk_Cycle: An Efficient Non-Parametric Algorithm for Detecting Rhythmic Components in Genome-Scale Data Sets. J. Biol. Rhythm. 2010, 25, 372–380. [Google Scholar] [CrossRef]
- Omura, S.; Numata, H.; Goto, S.G. Circadian Clock Regulates Photoperiodic Responses Governed by Distinct Output Pathways in the Bean Bug, Riptortus pedestris. Biol. Rhythm Res. 2016, 47, 937–945. [Google Scholar] [CrossRef]
- Ikeno, T.; Numata, H.; Goto, S.G. Circadian Clock Genes Period and Cycle Regulate Photoperiodic Diapause in the Bean Bug Riptortus Pedestris Males. J. Insect Physiol. 2011, 57, 935–938. [Google Scholar] [CrossRef] [PubMed]
- Saunders, D.S.; Gilbert, L.I. Regulation of Ovarian Diapause in Drosophila melanogaster by Photoperiod and Moderately Low Temperature. J. Insect Physiol. 1990, 36, 195–200. [Google Scholar] [CrossRef]
- Meuti, M.E.; Stone, M.; Ikeno, T.; Denlinger, D.L. Functional Circadian Clock Genes Are Essential for the Overwintering Diapause of the Northern House Mosquito, Culex pipiens. J. Exp. Biol. 2015, 218, 412–422. [Google Scholar] [CrossRef]
- Mukai, A.; Goto, S.G. The Clock Gene Period Is Essential for the Photoperiodic Response in the Jewel Wasp Nasonia vitripennis (Hymenoptera: Pteromalidae). Appl. Entomol. Zool. 2016, 51, 185–194. [Google Scholar] [CrossRef]
- Saunders, D.S.; Henrich, V.C.; Gilbert, L.I. Induction of Diapause in Drosophila melanogaster: Photoperiodic Regulation and the Impact of Arrhythmic Clock Mutations on Time Measurement. Proc. Natl. Acad. Sci. USA 1989, 86, 3748–3752. [Google Scholar] [CrossRef]
- Saunders, D.S. Dormancy, Diapause, and the Role of the Circadian System in Insect Photoperiodism. Annu. Rev. Entomol. 2020, 65, 373–389. [Google Scholar] [CrossRef]
- Zhu, L.; Tian, Z.; Guo, S.; Liu, W.; Zhu, F.; Wang, X.P. Circadian Clock Genes Link Photoperiodic Signals to Lipid Accumulation During Diapause Preparation in the Diapause-Destined Female Cabbage Beetles Colaphellus bowringi. Insect Biochem. Mol. Biol. 2019, 104, 1–10. [Google Scholar] [CrossRef]
- Cavaletto, G.; Faccoli, M.; Marini, L.; Mazzon, L. Emergence Phenology and Temperature Effect on the Post-Diapause Egg Development in the Bush Cricket Barbitistes vicetinus (Orthoptera, Tettigoniidae). Bull. Entomol. Res. 2020, 110, 161–168. [Google Scholar] [CrossRef]
- Ahmadi, F.; Moharramipour, S.; Mikani, A. The Effect of Temperature and Photoperiod on Diapause Induction in Pupae of Scrobipalpa ocellatella (Lepidoptera: Gelechiidae). Environ. Entomol. 2018, 47, 1314–1322. [Google Scholar] [CrossRef] [PubMed]
- Denlinger, D.L. Regulation of Diapause. Annu. Rev. Entomol. 2002, 47, 93–122. [Google Scholar] [CrossRef]
- Shiomi, K.; Takasu, Y.; Kunii, M.; Tsuchiya, R.; Mukaida, M.; Kobayashi, M.; Sezutsu, H.; Ichida Takahama, M.; Mizoguchi, A. Disruption of Diapause Induction by Talen-Based Gene Mutagenesis in Relation to a Unique Neuropeptide Signaling Pathway in Bombyx. Sci. Rep. 2015, 5, 15566. [Google Scholar] [CrossRef]
- Hasegawa, K.; Yamashita, O. Studies on the Mode of Action of the Diapause Hormone in the Silkworm, Bombyx mori L. Vi.The Target Organ of the Diapause Hormone. J. Exp. Biol. 1965, 43, 271–277. [Google Scholar] [CrossRef]
- Fukuda, S. The Production of the Diapause Eggs by Transplanting the Subesophageal Ganglion in the Silkworm. Proc. Jpn. Acad. 1951, 27, 672–677. [Google Scholar] [CrossRef]
- Hasegawa, K. Studies on the Voltinism in the Silkworm, Bombyx mori L., with Special Reference to the Organs Concerning Determination of Voltinism (a Preliminary Note). Proc. Jpn. Acad. 1951, 27, 667–671. [Google Scholar] [CrossRef][Green Version]
- Cox, K.H.; Takahashi, J.S. Circadian Clock Genes and the Transcriptional Architecture of the Clock Mechanism. J. Mol. Endocrinol. 2019, 63, R93–R102. [Google Scholar] [CrossRef]
- Mendoza-Viveros, L.; Bouchard-Cannon, P.; Hegazi, S.; Cheng, A.H.; Pastore, S.; Cheng, H.M. Molecular Modulators of the Circadian Clock: Lessons from Flies and Mice. Cell. Mol. Life Sci. 2017, 74, 1035–1059. [Google Scholar] [CrossRef]
- Millar, A.J. The Intracellular Dynamics of Circadian Clocks Reach for the Light of Ecology and Evolution. Annu. Rev. Plant Biol. 2016, 67, 595–618. [Google Scholar] [CrossRef]
- Kitagawa, N.; Shiomi, K.; Imai, K.; Niimi, T.; Yaginuma, T.; Yamashita, O. Establishment of a Sandwich Elisa System to Detect Diapause Hormone, and Developmental Profile of Hormone Levels in Egg and Subesophageal Ganglion of the Silkworm, Bombyx mori. Zool. Sci. 2005, 22, 213–221. [Google Scholar] [CrossRef]
- Yamashita, O. Diapause Hormone of the Silkworm, Bombyx mori: Structure, Gene Expression and Function. J. Insect Physiol. 1996, 42, 669–679. [Google Scholar] [CrossRef]
- Homma, T.; Watanabe, K.; Tsurumaru, S.; Kataoka, H.; Imai, K.; Kamba, M.; Niimi, T.; Yamashita, O.; Yaginuma, T. G Protein-Coupled Receptor for Diapause Hormone, an Inducer of Bombyx Embryonic Diapause. Biochem. Biophys. Res. Commun. 2006, 344, 386–393. [Google Scholar] [CrossRef]
- Kamei, Y.; Hasegawa, Y.; Niimi, T.; Yamashita, O.; Yaginuma, T. Trehalase-2 Protein Contributes to Trehalase Activity Enhanced by Diapause Hormone in Developing Ovaries of the Silkworm, Bombyx mori. J. Insect Physiol. 2011, 57, 608–613. [Google Scholar] [CrossRef]
- Su, Z.H.; Ikeda, M.; Sato, Y.; Saito, H.; Imai, K.; Isobe, M.; Yamashita, O. Molecular Characterization of Ovary Trehalase of the Silkworm, Bombyx mori and Its Transcriptional Activation by Diapause Hormone. Biochim. Biophys. Acta 1994, 1218, 366–374. [Google Scholar] [CrossRef]
- Shiomi, K.; Ishida, Y.; Ikeda, M.; Sato, Y.; Saito, H.; Imai, K.; Yamashita, O. Induction of Non-Diapause Eggs by Injection of Anti-Diapause Hormone Rabbit Serum into the Diapause Type of the Silkworm, Bombyx mori. J. Insect Physiol. 1994, 40, 693–699. [Google Scholar] [CrossRef]
- Gong, C.; Zeng, W.; Zhang, T.; Liu, R.; Ou, Y.; Ai, J.; Xiang, Z.; Xu, H. Effects of Transgenic Overexpression of Diapause Hormone and Diapause Hormone Receptor Genes on Non-Diapause Silkworm. Transgenic Res. 2017, 26, 807–815. [Google Scholar] [CrossRef]
- Hagino, A.; Kitagawa, N.; Imai, K.; Yamashita, O.; Shiomi, K. Immunoreactive Intensity of Fxprl Amide Neuropeptides in Response to Environmental Conditions in the Silkworm, Bombyx mori. Cell Tissue Res. 2010, 342, 459–469. [Google Scholar] [CrossRef] [PubMed]
- Shiomi, K.; Fujiwara, Y.; Yasukochi, Y.; Kajiura, Z.; Nakagaki, M.; Yaginuma, T. The Pitx Homeobox Gene in Bombyx mori: Regulation of Dh-Pban Neuropeptide Hormone Gene Expression. Mol. Cell. Neurosci. 2007, 34, 209–218. [Google Scholar] [CrossRef]
- Sato, Y.; Oguchi, M.; Menjo, N.; Imai, K.; Saito, H.; Ikeda, M.; Isobe, M.; Yamashita, O. Precursor Polyprotein for Multiple Neuropeptides Secreted from the Suboesophageal Ganglion of the Silkworm Bombyx mori: Characterization of the Cdna Encoding the Diapause Hormone Precursor and Identification of Additional Peptides. Proc. Natl. Acad. Sci. USA 1993, 90, 3251–3255. [Google Scholar] [CrossRef]
- Matsutani, K.; Sonobe, H. Control of Diapause-Factor Secretion from the Suboesophageal Ganglion in the Silkworm, Bombyx mori: The Roles of the Protocerebrum and Tritocerebrum. J. Insect Physiol. 1987, 33, 279–285. [Google Scholar] [CrossRef]
- Sonobe, H.; Hiyama, Y.; Keino, H. Changes in the Amount of the Diapause Factor in the Subesophageal Ganglion During Development of the Silkworm, Bombyx mori. J. Insect Physiol. 1977, 23, 633–637. [Google Scholar] [CrossRef]
- Noguchi, H.; Hayakawa, Y. Dopamine Is a Key Factor for the Induction of Egg Diapause of the Silkworm, Bombyx mori. Eur. J. Biochem. 2001, 268, 774–780. [Google Scholar] [CrossRef]
- Tsuchiya, R.; Kaneshima, A.; Kobayashi, M.; Yamazaki, M.; Takasu, Y.; Sezutsu, H.; Tanaka, Y.; Mizoguchi, A.; Shiomi, K. Maternal GABAergic and Gnrh/Corazonin Pathway Modulates Egg Diapause Phenotype of the Silkworm Bombyx mori. Proc. Natl. Acad. Sci. USA 2021, 118, e2020028118. [Google Scholar] [CrossRef]
- Erlander, M.G.; Tillakaratne, N.J.; Feldblum, S.; Patel, N.; Tobin, A.J. Two Genes Encode Distinct Glutamate Decarboxylases. Neuron 1991, 7, 91–100. [Google Scholar] [CrossRef]
- Mustard, J.A.; Jones, L.; Wright, G.A. GABA Signaling Affects Motor Function in the Honey Bee. J. Insect Physiol. 2020, 120, 103989. [Google Scholar] [CrossRef]
- Enomoto, Y.; Nt An, P.; Yamaguchi, M.; Fukusaki, E.; Shimma, S. Mass Spectrometric Imaging of GABA in the Drosophila melanogaster Adult Head. Anal. Sci. 2018, 34, 1055–1059. [Google Scholar] [CrossRef]
- Fei, H.; Chow, D.M.; Chen, A.; Romero-Calderon, R.; Ong, W.S.; Ackerson, L.C.; Maidment, N.T.; Simpson, J.H.; Frye, M.A.; Krantz, D.E. Mutation of the Drosophila Vesicular GABA Transporter Disrupts Visual Figure Detection. J. Exp. Biol. 2010, 213, 1717–1730. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ono, D.; Honma, K.I.; Yanagawa, Y.; Yamanaka, A.; Honma, S. GABA in the Suprachiasmatic Nucleus Refines Circadian Output Rhythms in Mice. Commun. Biol. 2019, 2, 232. [Google Scholar] [CrossRef]
- Hosie, A.M.; Aronstein, K.; Sattelle, D.B.; Ffrench-Constant, R.H. Molecular Biology of Insect Neuronal GABA Receptors. Trends Neurosci. 1997, 20, 578–583. [Google Scholar] [CrossRef]
- Yu, L.L.; Cui, Y.J.; Lang, G.J.; Zhang, M.Y.; Zhang, C.X. The Ionotropic Γ-Aminobutyric Acid Receptor Gene Family of the Silkworm, Bombyx mori. Genome 2010, 53, 688–697. [Google Scholar] [CrossRef] [PubMed]
- Honma, S.; Katsuno, Y.; Shinohara, K.; Abe, H.; Honma, K. Circadian Rhythm and Response to Light of Extracellular Glutamate and Aspartate in Rat Suprachiasmatic Nucleus. Am. J. Physiol. 1996, 271, R579–R585. [Google Scholar] [CrossRef]
- Castañeda, T.R.; de Prado, B.M.; Prieto, D.; Mora, F. Circadian Rhythms of Dopamine, Glutamate and GABA in the Striatum and Nucleus Accumbens of the Awake Rat: Modulation by Light. J. Pineal Res. 2004, 36, 177–185. [Google Scholar] [CrossRef]
- Nair, B.; Johar, K.; Priya, A.; Wong-Riley, M.T. Specificity Protein 4 (Sp4) Transcriptionally Regulates Inhibitory GABAergic Receptors in Neurons. Biochim. Biophys. Acta 2016, 1863, 1–9. [Google Scholar] [CrossRef]
- Lonze, B.E.; Ginty, D.D. Function and Regulation of Creb Family Transcription Factors in the Nervous System. Neuron 2002, 35, 605–623. [Google Scholar] [CrossRef]
- Samaco, R.C.; Hogart, A.; LaSalle, J.M. Epigenetic Overlap in Autism-Spectrum Neurodevelopmental Disorders: Mecp2 Deficiency Causes Reduced Expression of Ube3a and Gabrb3. Hum. Mol. Genet. 2005, 14, 483–492. [Google Scholar] [CrossRef]
- Barker, J.S.; Hines, R.M. Regulation of GABAA Receptor Subunit Expression in Substance Use Disorders. Int. J. Mol. Sci. 2020, 21, 4445. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cui, W.-Z.; Qiu, J.-F.; Dai, T.-M.; Chen, Z.; Li, J.-L.; Liu, K.; Wang, Y.-J.; Sima, Y.-H.; Xu, S.-Q. Circadian Clock Gene Period Contributes to Diapause via GABAeric-Diapause Hormone Pathway in Bombyx mori. Biology 2021, 10, 842. https://doi.org/10.3390/biology10090842
Cui W-Z, Qiu J-F, Dai T-M, Chen Z, Li J-L, Liu K, Wang Y-J, Sima Y-H, Xu S-Q. Circadian Clock Gene Period Contributes to Diapause via GABAeric-Diapause Hormone Pathway in Bombyx mori. Biology. 2021; 10(9):842. https://doi.org/10.3390/biology10090842
Chicago/Turabian StyleCui, Wen-Zhao, Jian-Feng Qiu, Tai-Ming Dai, Zhuo Chen, Jiang-Lan Li, Kai Liu, Yu-Jun Wang, Yang-Hu Sima, and Shi-Qing Xu. 2021. "Circadian Clock Gene Period Contributes to Diapause via GABAeric-Diapause Hormone Pathway in Bombyx mori" Biology 10, no. 9: 842. https://doi.org/10.3390/biology10090842
APA StyleCui, W.-Z., Qiu, J.-F., Dai, T.-M., Chen, Z., Li, J.-L., Liu, K., Wang, Y.-J., Sima, Y.-H., & Xu, S.-Q. (2021). Circadian Clock Gene Period Contributes to Diapause via GABAeric-Diapause Hormone Pathway in Bombyx mori. Biology, 10(9), 842. https://doi.org/10.3390/biology10090842





