Specific Bioelectrical Impedance Vector Analysis Identifies Body Fat Reduction after a Lifestyle Intervention in Former Elite Athletes
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants and Study Design
2.2. Anthropometry
2.3. Dual-Energy X-ray Absorptiometry
2.4. Bioelectrical Impedance Spectroscopy
2.5. Statistical Analysis
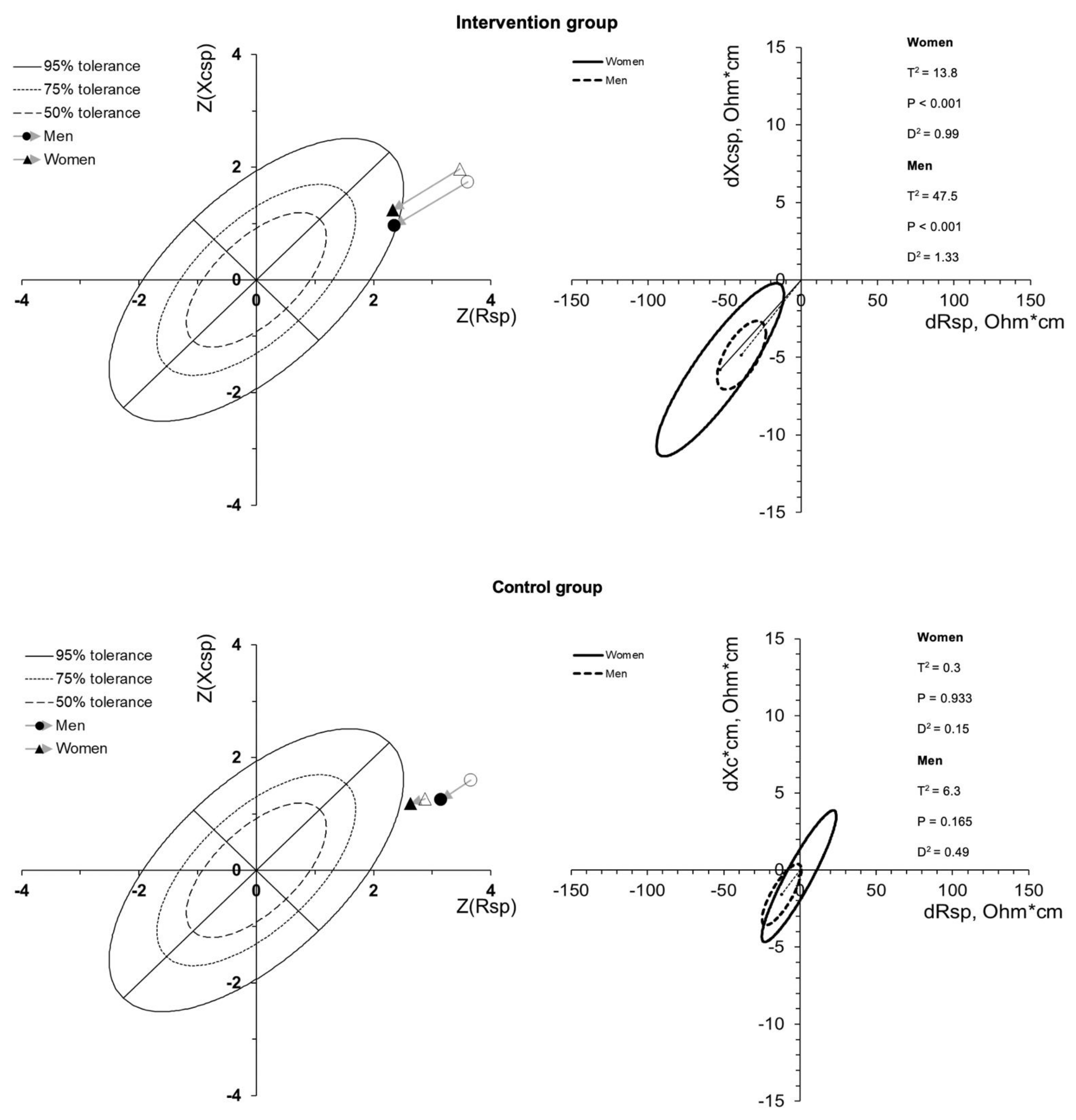
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Silva, A.M. Structural and functional body components in athletic health and performance phenotypes. Eur. J. Clin. Nutr. 2019, 73, 215–224. [Google Scholar] [CrossRef] [PubMed]
- Santanasto, A.J.; Goodpaster, B.H.; Kritchevsky, S.B.; Miljkovic, I.; Satterfield, S.; Schwartz, A.V.; Cummings, S.R.; Boudreau, R.M.; Harris, T.B.; Newman, A.B. Body Composition Remodeling and Mortality: The Health Aging and Body Composition Study. J. Gerontol. A. Biol. Sci. Med. Sci. 2017, 72, 513–519. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.M.; Pierson, R.N.J.; Heymsfield, S.B. The five-level model: A new approach to organizing body-composition research. Am. J. Clin. Nutr. 1992, 56, 19–28. [Google Scholar] [CrossRef]
- Lukaski, H.; Raymond-Pope, C.J. New Frontiers of Body Composition in Sport. Int. J. Sports Med. 2021. [Google Scholar] [CrossRef]
- Matias, C.N.; Campa, F.; Santos, D.A.; Lukaski, H.; Sardinha, L.B.; Silva, A.M. Fat-free Mass Bioelectrical Impedance Analysis Predictive Equation for Athletes using a 4-Compartment Model. Int. J. Sports Med. 2021, 42, 27–32. [Google Scholar] [CrossRef]
- Buffa, R.; Saragat, B.; Cabras, S.; Rinaldi, A.C.; Marini, E. Accuracy of specific BIVA for the assessment of body composition in the United States population. PLoS ONE 2013, 8, e58533. [Google Scholar] [CrossRef] [PubMed]
- Santos, D.A.; Dawson, J.A.; Matias, C.N.; Rocha, P.M.; Minderico, C.S.; Allison, D.B.; Sardinha, L.B.; Silva, A.M. Reference values for body composition and anthropometric measurements in athletes. PLoS ONE 2014, 9, e97846. [Google Scholar] [CrossRef] [PubMed]
- Campa, F.; Matias, C.N.M.; Teixeira, F.J.; Reis, J.F.; Valamatos, M.J.; Toselli, S.; Monteiro, C.P. Leucine metabolites do not induce changes in phase angle, bioimpedance vector analysis patterns, and strength in resistance trained men. Appl. Physiol. Nutr. Metab. 2020. [Google Scholar] [CrossRef]
- Ward, L.C. Bioelectrical impedance analysis for body composition assessment: Reflections on accuracy, clinical utility, and standardisation. Eur. J. Clin. Nutr. 2019, 73, 194–199. [Google Scholar] [CrossRef]
- Francisco, R.; Matias, C.N.; Santos, D.A.; Campa, F.; Minderico, C.S.; Rocha, P.; Heymsfield, S.B.; Lukaski, H.; Sardinha, L.B.; Silva, A.M. The Predictive Role of Raw Bioelectrical Impedance Parameters in Water Compartments and Fluid Distribution Assessed by Dilution Techniques in Athletes. Int. J. Environ. Res. Public Health 2020, 17, 759. [Google Scholar] [CrossRef]
- Moon, J.R. Body composition in athletes and sports nutrition: An examination of the bioimpedance analysis technique. Eur. J. Clin. Nutr. 2013, 67, S54. [Google Scholar] [CrossRef] [PubMed]
- Piccoli, A.; Rossi, B.; Pillon, L.; Bucciante, G. A new method for monitoring body fluid variation by bioimpedance analysis: The RXc graph. Kidney Int. 1994, 46, 534–539. [Google Scholar] [CrossRef] [PubMed]
- Silva, A.M.; Nunes, C.L.; Matias, C.N.; Rocha, P.M.; Minderico, C.S.; Heymsfield, S.B.; Lukaski, H.; Sardinha, L.B. Usefulness of raw bioelectrical impedance parameters in tracking fluid shifts in judo athletes. Eur. J. Sport Sci. 2020, 20, 734–743. [Google Scholar] [CrossRef] [PubMed]
- Campa, F.; Matias, C.N.; Marini, E.; Heymsfield, S.B.; Toselli, S.; Sardinha, L.B.; Silva, A.M. Identifying athlete body fluid changes during a competitive season with bioelectrical impedance vector analysis. Int. J. Sports Physiol. Perform. 2020, 15, 361–367. [Google Scholar] [CrossRef]
- Khalil, S.F.; Mohktar, M.S.; Ibrahim, F. The theory and fundamentals of bioimpedance analysis in clinical status monitoring and diagnosis of diseases. Sensors 2014, 14, 10895–10928. [Google Scholar] [CrossRef]
- Marini, E.; Campa, F.; Buffa, R.; Stagi, S.; Matias, C.N.; Toselli, S.; Sardinha, L.B.; Silva, A.M. Phase angle and bioelectrical impedance vector analysis in the evaluation of body composition in athletes. Clin. Nutr. 2020, 39, 447–454. [Google Scholar] [CrossRef]
- Stagi, S.; Irurtia, A.; Rosales Rafel, J.; Cabras, S.; Buffa, R.; Carrasco-Marginet, M.; Castizo-Olier, J.; Marini, E. Segmental body composition estimated by specific BIVA and dual-energy X-ray absorptiometry. Clin. Nutr. 2021, 40, 1621–1627. [Google Scholar] [CrossRef]
- Silva, A.M.; Nunes, C.L.; Matias, C.N.; Jesus, F.; Francisco, R.; Cardoso, M.; Santos, I.; Carraça, E.V.; Silva, M.N.; Sardinha, L.B.; et al. Champ4life Study Protocol: A One-Year Randomized Controlled Trial of a Lifestyle Intervention for Inactive Former Elite Athletes with Overweight/Obesity. Nutrients 2020, 12, 286. [Google Scholar] [CrossRef]
- World Medical Association. World Medical Association Declaration of Helsinki: Ethical principles for medical research involving human subjects. JAMA 2013, 310, 2191–2194. [Google Scholar] [CrossRef]
- Lohman, T.; Roche, A.F.; Martorell, R. Anthropometric Standardization Reference Manual; Human Kinetics Books: Champaign, IL, USA, 1988. [Google Scholar]
- Santos, D.A.; Gobbo, L.A.; Matias, C.N.; Petroski, E.L.; Goncalves, E.M.; Cyrino, E.S.; Minderico, C.S.; Sardinha, L.B.; Silva, A.M. Body composition in taller individuals using DXA: A validation study for athletic and non-athletic populations. J. Sports Sci. 2013, 31, 405–413. [Google Scholar] [CrossRef]
- Matias, C.N.; Santos, D.A.; Gonçalves, E.M.; Fields, D.A.; Sardinha, L.B.; Silva, A.M. Is bioelectrical impedance spectroscopy accurate in estimating total body water and its compartments in elite athletes? Ann. Hum. Biol. 2013, 40, 152–156. [Google Scholar] [CrossRef]
- Campa, F.; Matias, C.; Gatterer, H.; Toselli, S.; Koury, J.C.; Andreoli, A.; Melchiorri, G.; Sardinha, L.B.; Silva, A.M. Classic Bioelectrical Impedance Vector Reference Values for Assessing Body Composition in Male and Female AthleteS. Int. J. Environ. Res. Public Health 2019, 16, 5066. [Google Scholar] [CrossRef]
- Toselli, S.; Marini, E.; Maietta Latessa, P.; Benedetti, L.; Campa, F. Maturity Related Differences in Body Composition Assessed by Classic and Specific Bioimpedance Vector Analysis among Male Elite Youth Soccer Players. Int. J. Environ. Res. Public Health 2020, 17, 729. [Google Scholar] [CrossRef]
- Kasper, A.M.; Langan-Evans, C.; Hudson, J.F.; Brownlee, T.E.; Harper, L.D.; Naughton, R.J.; Morton, J.P.; Close, G.L. Come Back Skinfolds, All Is Forgiven: A Narrative Review of the Efficacy of Common Body Composition Methods in Applied Sports Practice. Nutrients 2021, 13, 1075. [Google Scholar] [CrossRef]
- Boileau, R.A.; Horswill, C.A. Body composition in sports: Measurement and applications for weight gain and loss. In Exercise and Sport Science; Garret, W.E., Jr., Kinkendall, D.T., Eds.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2000; pp. 319–338. [Google Scholar]
- Bosch, T.A.; Burruss, T.P.; Weir, N.L.; Fielding, K.A.; Engel, B.E.; Weston, T.D.; Dengel, D.R. Abdominal body composition differences in NFL football players. J. Strength Cond. Res. 2014, 28, 3313–3319. [Google Scholar] [CrossRef]
- Silva, A.M.; Matias, C.N.; Nunes, C.L.; Santos, D.A.; Marini, E.; Lukaski, H.C.; Sardinha, L.B. Lack of agreement of in vivo raw bioimpedance measurements obtained from two single and multi-frequency bioelectrical impedance devices. Eur. J. Clin. Nutr. 2019, 73, 1077–1083. [Google Scholar] [CrossRef]
- Dellinger, J.R.; Johnson, B.A.; Benavides, M.L.; Moore, M.L.; Stratton, M.T.; Harty, P.S.; Siedler, M.R.; Tinsley, G.M. Agreement of bioelectrical resistance, reactance, and phase angle values from supine and standing bioimpedance analyzers. Physiol. Meas. 2021. [Google Scholar] [CrossRef]
- Campa, F.; Toselli, S.; Mazzilli, M.; Gobbo, L.A.; Coratella, G. Assessment of Body Composition in Athletes: A Narrative Review of Available Methods with Special Reference to Quantitative and Qualitative Bioimpedance Analysis. Nutrients 2021, 13, 1620. [Google Scholar] [CrossRef]
- Stratton, M.T.; Smith, R.W.; Harty, P.S.; Rodriguez, C.; Johnson, B.A.; Dellinger, J.R.; Williams, A.D.; White, S.J.; Benavides, M.L.; Tinsley, G.M. Longitudinal agreement of four bioimpedance analyzers for detecting changes in raw bioimpedance during purposeful weight gain with resistance training. Eur. J. Clin. Nutr. 2021. [Google Scholar] [CrossRef]

| Intervention (n 40) | Control (n 40) | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Men (n 27) | Women (n 13) | Whole Sample | Men (n 26) | Women (n 14) | Whole Sample | ES † | Interaction p-Value | ||
| Weight (kg) | PRE | 95.5 ± 14.9 | 83.4 ± 12.0 | 91.5 ± 15.7 | 94.6 ± 17.2 | 76.6 ± 11.6 | 88.3 ± 17.6 | −0.053 | <0.001 |
| POST | 90.3 ± 17.1 | 78.6 ± 11.9 | 86.6 ± 16.6 * | 95.0 ± 18.2 | 76.9 ± 11.1 | 88.7 ± 18.4 | |||
| BMI (kg/m2) | PRE | 31.7 ± 4.4 | 31.4 ± 3.5 | 31.6 ± 4.1 | 31.1 ± 5.4 | 29.4 ± 4.1 | 30.5 ± 5.0 | −0.051 | <0.001 |
| POST | 30.2 ± 4.7 | 29.7 ± 3.3 | 30.0 ± 4.3 * | 31.3 ± 5.7 | 29.4 ± 4.1 | 30.6 ± 5.2 | |||
| FM (kg) | PRE | 28.0 ± 7.7 | 35.6 ± 6.5 | 30.4 ± 8.1 | 28.0 ± 11.3 | 30.7 ± 6.7 | 28.9 ± 9.9 | 0.06 | <0.001 |
| POST | 24.2 ± 7.8 | 31.9 ± 7.0 | 26.6 ± 8.3 * | 28.6 ± 13.1 | 30.8 ± 6.5 | 29.4 ± 11.2 | |||
| FM (%) | PRE | 29.1 ± 5.7 | 42.5 ± 3.1 | 33.5 ± 8.1 | 28.7 ± 6.4 | 39.8 ± 3.8 | 32.6 ± 7.7 | 0.058 | <0.001 |
| POST | 26.5 ± 5.4 | 40.2 ± 4.1 | 30.9 ± 8.2 * | 29.0 ± 7.1 | 39.8 ± 3.8 | 32.8 ± 8.0 | |||
| FFM (kg) | PRE | 67.5 ±10.9 | 47.8 ± 5.7 | 61.1 ± 13.3 | 66.6 ± 7.6 | 45.9 ± 5.9 | 59.4 ± 12.2 | −0.093 | 0.054 |
| POST | 66.1 ± 11.3 | 46.7 ± 5.2 | 60.0 ± 13.3 * | 66.4 ± 7.4 | 46.1 ± 5.5 | 59.3 ± 11.9 | |||
| Rsp (Ω·cm) | PRE | 436.7 ± 49.6 | 529.3 ± 40.0 | 466.8 ± 63.7 | 438.6 ± 54.1 | 501.2 ± 78.9 | 460.5 ± 69.7 | 0.12 | 0.001 |
| POST | 397.6 ± 50.3 | 476.0 ± 71.1 | 423.1 ± 68.0 * | 422.5 ± 57.5 | 489.7 ± 67.4 | 446.0 ± 68.4 * | |||
| Xcsp (Ω·cm) | PRE | 54.7 ± 7.1 | 58.7 ± 7.0 | 56.0 ± 7.2 | 53.9 ± 8.6 | 53.1 ± 6.8 | 53.6 ± 7.9 | −0.105 | 0.009 |
| POST | 49.9 ± 8.6 | 52.9 ± 9.2 | 50.8 ± 8.8 * | 51.6 ± 7.5 | 51.4 ± 8.1 | 51.5 ± 7.6 * | |||
| VLsp (Ω·cm) | PRE | 440.0 ± 49.8 | 532.6 ± 40.0 | 470.1 ± 63.8 | 442.0 ± 54.4 | 504.6 ± 80.3 | 463.9 ± 70.4 | 0.123 | 0.001 |
| POST | 400.8 ± 50.6 | 478.9 ± 71.5 | 426.2 ± 68.2 * | 426.4 ± 58.8 | 492.4 ± 67.7 | 449.5 ± 69.0 * | |||
| PhA (º) | PRE | 7.2 ± 0.7 | 6.3 ± 0.7 | 6.9 ± 0.8 | 7.0 ± 0.8 | 6.1 ± 0.4 | 6.7 ± 0.8 | −0.257 | 0.964 |
| POST | 7.1 ± 0.9 | 6.3 ± 0.6 | 6.9 ± 0.9 | 7.0 ± 0.9 | 6.0 ± 0.6 | 6.7 ± 0.9 | |||
| Independent Variable | R2 | SEE | β | 95% CI | p-Value |
|---|---|---|---|---|---|
| Δ Specific vector length | 0.246 | 2.36 | 0.33 | 0.020, 0.046 | <0.001 |
| Model 1 | 0.373 | 2.12 | 0.23 | 0.009, 0.037 | 0.002 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Campa, F.; Matias, C.N.; Nunes, C.L.; Monteiro, C.P.; Francisco, R.; Jesus, F.; Marini, E.; Sardinha, L.B.; Martins, P.; Minderico, C.; et al. Specific Bioelectrical Impedance Vector Analysis Identifies Body Fat Reduction after a Lifestyle Intervention in Former Elite Athletes. Biology 2021, 10, 524. https://doi.org/10.3390/biology10060524
Campa F, Matias CN, Nunes CL, Monteiro CP, Francisco R, Jesus F, Marini E, Sardinha LB, Martins P, Minderico C, et al. Specific Bioelectrical Impedance Vector Analysis Identifies Body Fat Reduction after a Lifestyle Intervention in Former Elite Athletes. Biology. 2021; 10(6):524. https://doi.org/10.3390/biology10060524
Chicago/Turabian StyleCampa, Francesco, Catarina Nunes Matias, Catarina L. Nunes, Cristina P. Monteiro, Rubén Francisco, Filipe Jesus, Elisabetta Marini, Luís B. Sardinha, Paulo Martins, Cláudia Minderico, and et al. 2021. "Specific Bioelectrical Impedance Vector Analysis Identifies Body Fat Reduction after a Lifestyle Intervention in Former Elite Athletes" Biology 10, no. 6: 524. https://doi.org/10.3390/biology10060524
APA StyleCampa, F., Matias, C. N., Nunes, C. L., Monteiro, C. P., Francisco, R., Jesus, F., Marini, E., Sardinha, L. B., Martins, P., Minderico, C., & Silva, A. M. (2021). Specific Bioelectrical Impedance Vector Analysis Identifies Body Fat Reduction after a Lifestyle Intervention in Former Elite Athletes. Biology, 10(6), 524. https://doi.org/10.3390/biology10060524










