Changes in Aphid—Plant Interactions under Increased Temperature
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Aphids
2.2. Host Plants
2.3. Entomological Experiments
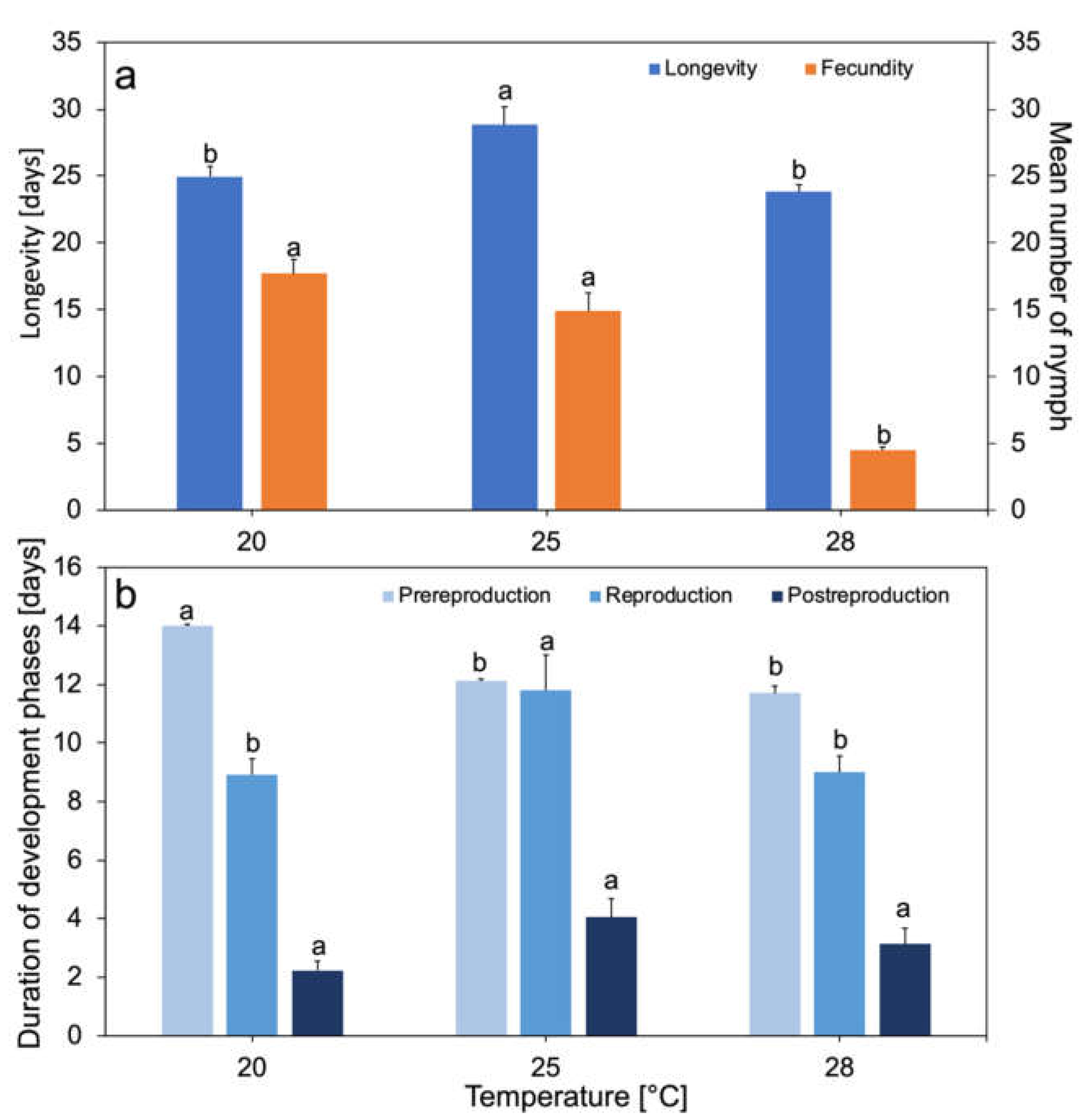
2.3.1. Longevity and Total Fecundity
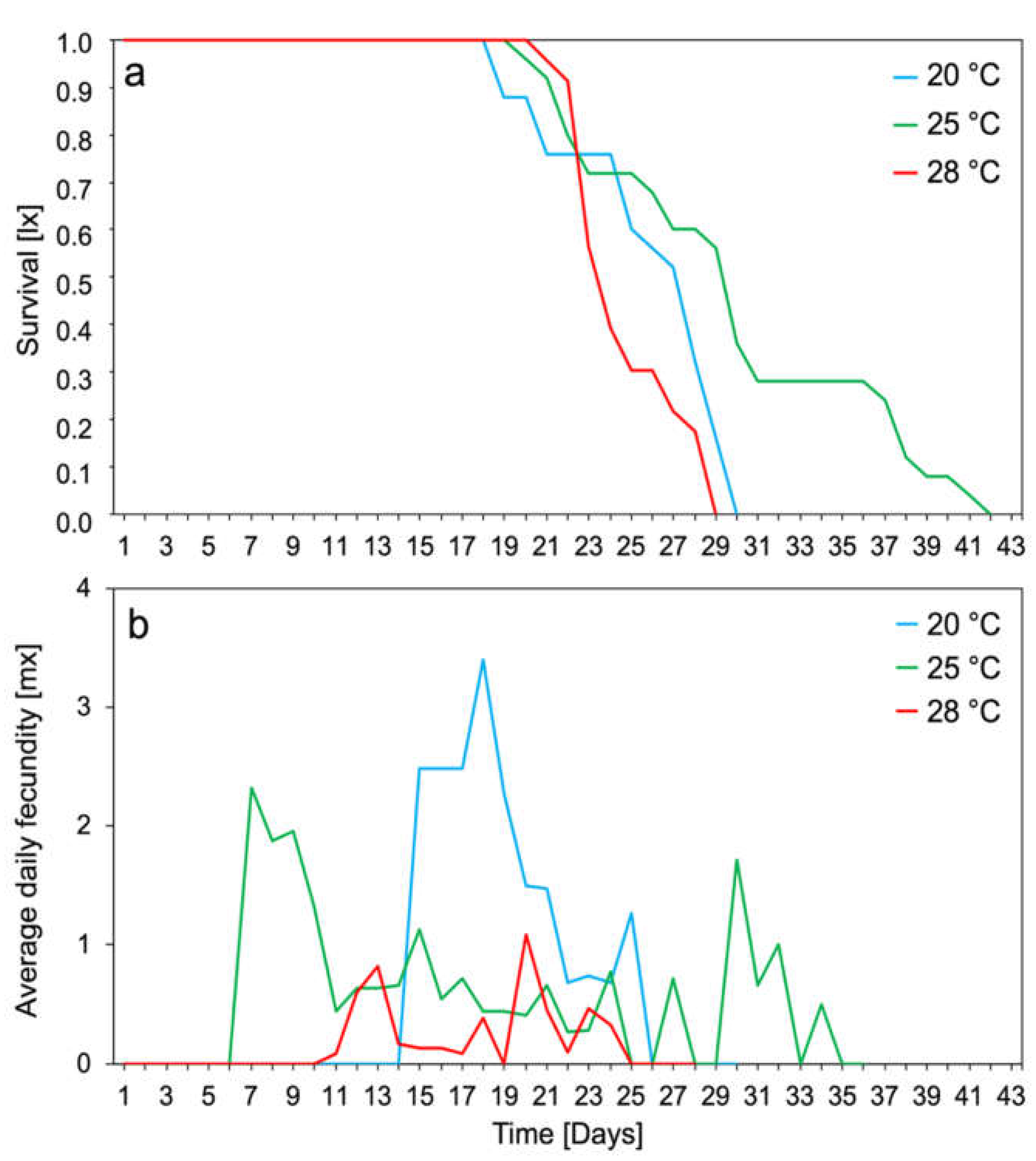
2.3.2. Demographic Parameters, Survival, and Average Daily Fecundity
- intrinsic rate of increase, rm = (lnMd × 0.738)/D,where D is the developmental period from birth to the beginning of the first reproduction (pre-reproductive period), and Md is the number of nymphs produced by the adult in the first D days of reproduction after the adult molt;
- net reproduction rates, Ro = Ʃ(lxmx),where lx and mx are cumulative daily survival and fecundity, respectively;
- finite rate of increase, λ = erm, where e is the base of the natural logarithm;
- mean generation time, T = ln Ro/rm;
- population doubling time, DT = ln 2/rm.
2.4. Biochemical Analyses
Effect of Temperature on the Enzymatic Activity in Aphid and Plant Tissues
2.5. Statistical Analyses
3. Results
3.1. Entomological Experiments
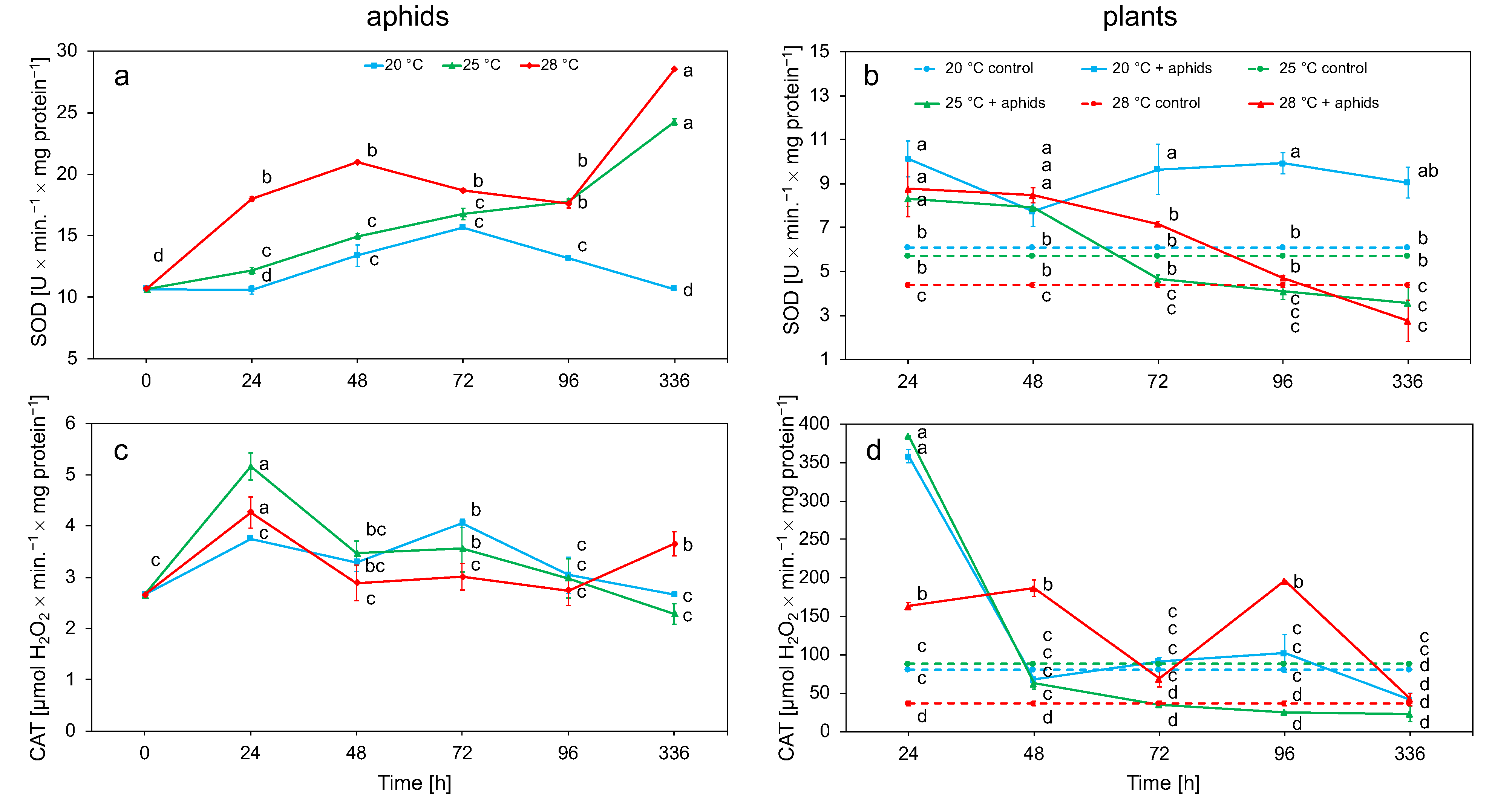
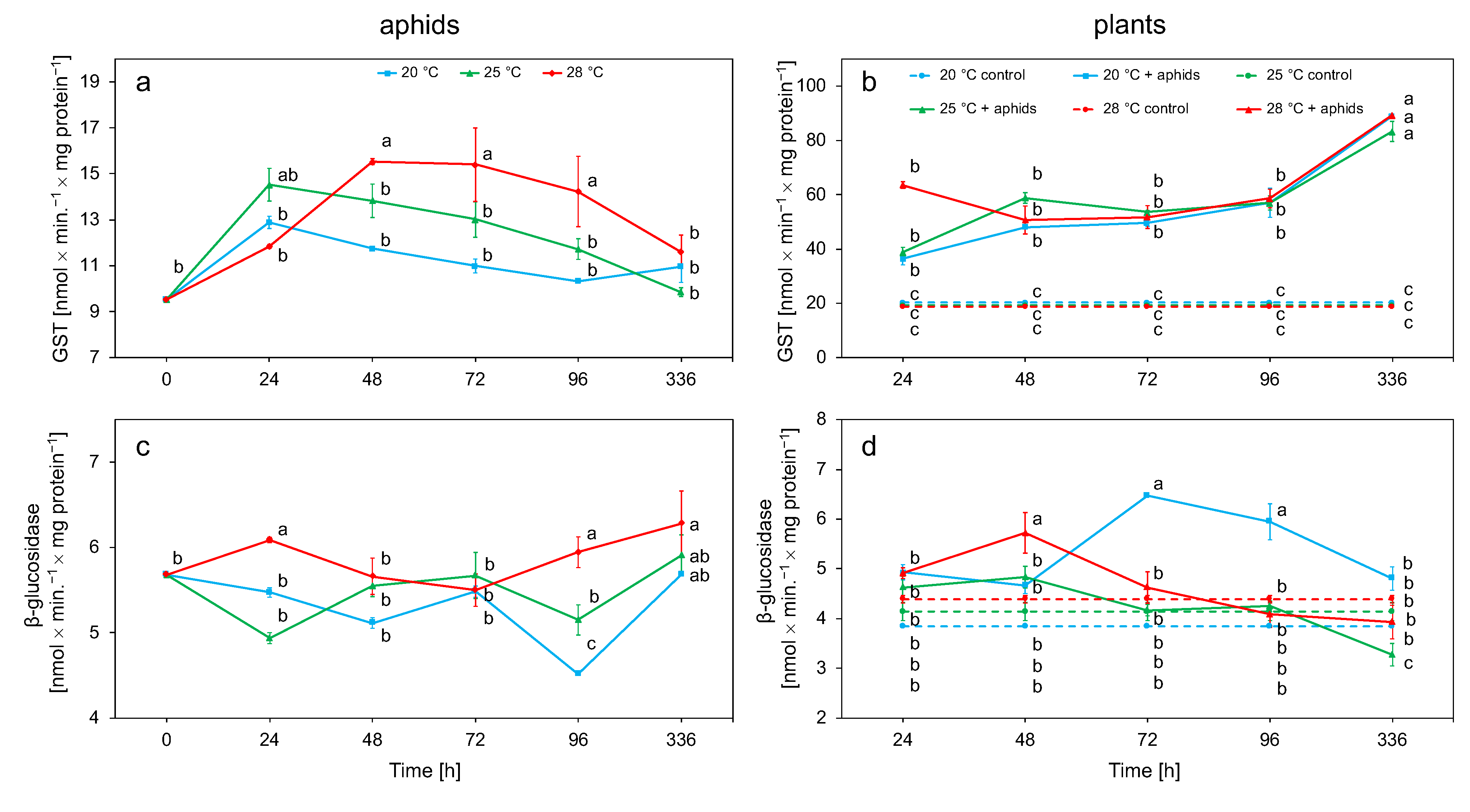
3.2. Biochemical Analyses
3.2.1. Superoxide Dismutase (SOD) and Catalase (CAT) Activity in Aphid and Plant Tissue
3.2.2. Glutathione S-Transferase (GST) and β-Glucosidase Activity in Aphid and Plant Tissue
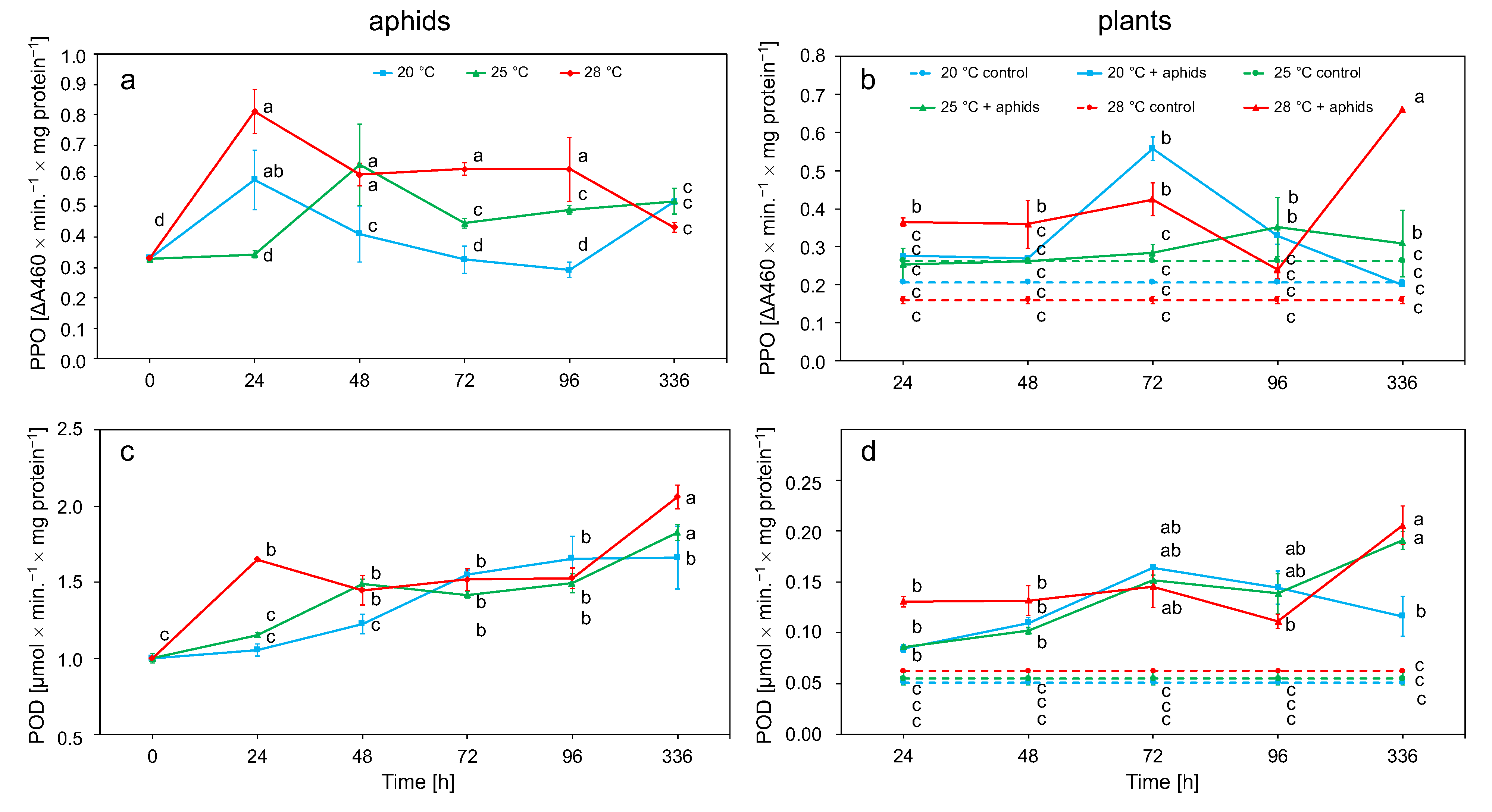
3.2.3. Polyphenol Oxidase (PPO) and Peroxidase (POD) Activity in Aphid and Plant Tissue
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bale, J.S.; Masters, G.J.; Hodkinson, I.D.; Awmack, C.; Bezemer, T.M.; Brown, V.K.; Butterfield, J.; Buse, A.; Coulson, J.C.; Farrar, J.; et al. Herbivory in global climate change research: Direct effects of rising temperature on insect herbivores. Glob. Chang. Biol. 2002, 8, 1–16. [Google Scholar] [CrossRef]
- Hullé, M.; Cœur d’Acier, A.; Bankhead-Dronnet, S.; Harrington, R. Aphids in the face of global changes. C. R. Biol. 2010, 333, 497–503. [Google Scholar] [CrossRef]
- Strathdee, A.T.; Bale, J.S.; Block, W.C.; Coulson, S.J.; Hodkinson, I.D.; Webb, N.R. Effects of temperature elevation on a field population of Acyrthosiphon svalbardicum (Hemiptera: Aphididae) on Spitsbergen. Oecologia 1993, 96, 457–465. [Google Scholar] [CrossRef] [PubMed]
- Mehrparvar, M.; Hatami, B. Effect of temperature on some biological parameters of an Iranian population of the Rose Aphid, Macrosiphum rosae (Hemiptera: Aphididae). Eur. J. Entomol. 2007, 104, 631–634. [Google Scholar] [CrossRef]
- Borowiak-Sobkowiak, B.; Durak, R. Biology and ecology of Appendiseta robiniae (Hemiptera: Aphidoidea)—An alien species in Europe. Open Life Sci. 2012, 7, 487–494. [Google Scholar] [CrossRef]
- Harrington, R.; Fleming, R.A.; Woiwod, I.P. Climate change impacts on insect management and conservation in temperate regions: Can they be predicted? Agric. For. Entomol. 2001, 3, 233–240. [Google Scholar] [CrossRef]
- Kiritani, K. Predicting impacts of global warming on population dynamics and distribution of arthropods in Japan. Popul. Ecol. 2006, 48, 5–12. [Google Scholar] [CrossRef]
- Margaritopoulos, J.T.; Tsitsipis, J.A.; Goudoudaki, S.; Blackman, R.L. Life cycle variation of Myzus persicae (Hemiptera: Aphididae) in Greece. Bull. Entomol. Res. 2002, 92, 309–319. [Google Scholar] [CrossRef]
- Danks, H.V. The elements of seasonal adaptations in insects. Can. Entomol. 2007, 139, 1–44. [Google Scholar] [CrossRef]
- Masters, G.J.; Brown, V.K.; Clarke, I.P.; Whittaker, J.B.; Hollier, J.A. Direct and indirect effects of climate change on insect herbivores: Auchenorrhyncha (Homoptera). Ecol. Entomol. 1998, 23, 45–52. [Google Scholar] [CrossRef]
- Miles, J.E.; Bale, J.S.; Hodkinson, I.D. Effects of temperature elevation on the population dynamics of the upland heather psyllid Strophingia ericae (Curtis) (Homoptera: Psylloidea). Glob. Chang. Biol. 1997, 3, 291–297. [Google Scholar] [CrossRef]
- Yamamura, K.; Yokozawa, M.; Nishimori, M.; Ueda, Y.; Yokosuka, T. How to analyze long-term insect population dynamics under climate change: 50-year data of three insect pests in paddy fields. Popul. Ecol. 2006, 48, 31–48. [Google Scholar] [CrossRef]
- Matsumura, T.; Matsumoto, H.; Hayakawa, Y. Heat stress hardening of oriental armyworms is induced by a transient elevation of reactive oxygen species during sublethal stress. Arch. Insect Biochem. Physiol. 2017, 96, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Fu, W.; Li, N.; Zhang, F.; Liu, T.-X. Antioxidant responses of Propylaea japonica (Coleoptera: Coccinellidae) exposed to high temperature stress. J. Insect Physiol. 2015, 73, 47–52. [Google Scholar] [CrossRef]
- Krishnan, N.; Kodrík, D.; Turanli, F.; Sehnal, F. Stage-specific distribution of oxidative radicals and antioxidant enzymes in the midgut of Leptinotarsa decemlineata. J. Insect Physiol. 2007, 53, 67–74. [Google Scholar] [CrossRef]
- Łukasik, I.; Goławska, S. Effect of host plant on levels of reactive oxygen species and antioxidants in the cereal aphids Sitobion avenae and Rhopalosiphum padi. Biochem. Syst. Ecol. 2013, 51, 232–239. [Google Scholar] [CrossRef]
- Kindlmann, P.; Dixon, A.F.G.; Michaud, J.P. Aphid Biodiversity under Environmental Change; Springer: Dordrecht, The Netherlands, 2010; ISBN 978-90-481-8600-6. [Google Scholar]
- Dahal, K.; Li, X.Q.; Tai, H.; Creelman, A.; Bizimungu, B. Improving potato stress tolerance and tuber yield under a climate change scenario—A current overview. Front. Plant Sci. 2019, 10. [Google Scholar] [CrossRef]
- Ding, X.; Jiang, Y.; Hao, T.; Jin, H.; Zhang, H.; He, L.; Zhou, Q.; Huang, D.; Hui, D.; Yu, J. Effects of heat shock onphotosynthetic properties, antioxidant enzyme activity, and downy mildew of cucumber (Cucumis sativus L.). PLoS ONE 2016, 11, e0152429. [Google Scholar] [CrossRef]
- Mittler, R. Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci. 2002, 7, 405–410. [Google Scholar] [CrossRef]
- Łukasik, I.; Goławska, S.; Wójcicka, A.; Goławski, A. Effect of host plants on antioxidant system of pea aphid Acyrthosiphon pisum. Bull. Insectol. 2011, 64, 153–158. [Google Scholar]
- Almeselmani, M.; Deshmukh, P.S.; Sairam, R.K.; Kushwaha, S.R.; Singh, T.P. Protective role of antioxidant enzymes under high temperature stress. Plant Sci. 2006, 171, 382–388. [Google Scholar] [CrossRef]
- Francis, F.; Vanhaelen, N.; Haubruge, E. Glutathione S-transferases in the adaptation to plant secondary metabolites in the Myzus persicae aphid. Arch. Insect Biochem. Physiol. 2005, 58, 166–174. [Google Scholar] [CrossRef]
- Després, L.; David, J.P.; Gallet, C. The evolutionary ecology of insect resistance to plant chemicals. Trends Ecol. Evol. 2007, 22, 298–307. [Google Scholar] [CrossRef]
- Nianiou-Obeidat, I.; Madesis, P.; Kissoudis, C.; Voulgari, G.; Chronopoulou, E.; Tsaftaris, A.; Labrou, N.E. Plant glutathione transferase-mediated stress tolerance: Functions and biotechnological applications. Plant Cell Rep. 2017, 36, 791–805. [Google Scholar] [CrossRef] [PubMed]
- Pentzold, S.; Zagrobelny, M.; Rook, F.; Bak, S. How insects overcome two-component plant chemical defence: Plant β-glucosidases as the main target for herbivore adaptation. Biol. Rev. 2014, 89, 531–551. [Google Scholar] [CrossRef]
- Chrzanowski, G.; Leszczyński, B.; Czerniewicz, P.; Sytykiewicz, H.; Matok, H.; Krzyzanowski, R.; Sempruch, C. Effect of phenolic acids from black currant, sour cherry and walnut on grain aphid (Sitobion avenae F.) development. Crop Prot. 2012, 35, 71–77. [Google Scholar] [CrossRef]
- Pourcel, L.; Routaboul, J.M.; Cheynier, V.; Lepiniec, L.; Debeaujon, I. Flavonoid oxidation in plants: From biochemical properties to physiological functions. Trends Plant Sci. 2007, 12, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Blackman, R.L.; Eastop, V.F. Aphids on the World’s Trees: An Identification and Information Guide; CABI: Wallingford, UK, 1994; ISBN 0851988776. [Google Scholar]
- Mehrparvar, M.; Mansouri, S.M.; Hatami, B. Some bioecological aspects of the rose aphid, Macrosiphum rosae (Hemiptera: Aphididae) and its natural enemies. Acta Univ. Sapientiae Agric. Environ. 2016, 8, 74–88. [Google Scholar] [CrossRef]
- Olmez, S.; Bayhan, E.; Ulusoy, M.R. Effect of different temperatures on the biological parameters of Macrosiphum rosae (L.) (Homoptera: Aphididae). J. Plant Dis. Prot. 2003, 110, 203–208. [Google Scholar]
- Golizadeh, A.; Jafari-Behi, V.; Razmjou, J.; Naseri, B.; Hassanpour, M. Population Growth Parameters of Rose Aphid, Macrosiphum rosae (Hemiptera: Aphididae) on Different Rose Cultivars. Neotrop. Entomol. 2017, 46, 100–106. [Google Scholar] [CrossRef]
- Koricheva, J.; Larsson, S.; Haukioja, E. Insect performance on experimentally stressed woody plants: A meta-analysis. Annu. Rev. Entomol. 1998, 43, 195–216. [Google Scholar] [CrossRef] [PubMed]
- Dampc, J.; Kula-Maximenko, M.; Molon, M.; Durak, R. Enzymatic defense response of apple aphid Aphis pomi to increased temperature. Insects 2020, 11, 436. [Google Scholar] [CrossRef]
- Wyatt, I.J.; White, P.F. Simple estimation of intrinsic increase rates for aphids and tetranychid mites. J. Appl. Ecol. 1977, 14, 757. [Google Scholar] [CrossRef]
- Birch, L.C. The intrinsic rate of natural increase of an insect population. J. Anim. Ecol. 1948, 17, 15. [Google Scholar] [CrossRef]
- Wang, Y.; Oberley, L.W.; Murhammer, D.W. Antioxidant defense systems of two lipidopteran insect cell lines. Free Radic. Biol. Med. 2001, 30, 1254–1262. [Google Scholar] [CrossRef]
- Aebi, H. Catalase in vitro. In Methods in Enzymology; Elsevier: Amsterdam, The Netherlands, 1984; Volume 105, pp. 121–126. [Google Scholar] [CrossRef]
- Katagiri, C. α-D-Glucosidase in the serum of the american cockroach, Periplaneta americana. Insect Biochem. 1979, 9, 199–204. [Google Scholar] [CrossRef]
- Leszczynski, B.; Dixon, A.F.G. Resistance of cereals to aphids: The interaction between hydroxamic acids and glutathione S-transferases in the grain aphid Sitobion avenae (F.) (Hom., Aphididae). J. Appl. Entomol. 1992, 113, 61–67. [Google Scholar] [CrossRef]
- Miles, P.W. Studies on the salivary physiology of plant bugs: Oxidase activity in the salivary apparatus and saliva. J. Insect Physiol. 1964, 10, 121–129. [Google Scholar] [CrossRef]
- Laurema, S.; Varis, A.-L.; Miettinen, H. Studies on enzymes in the salivary glands of Lygus rugulipennis (Hemiptera, miridae). Insect Biochem. 1985, 15, 211–224. [Google Scholar] [CrossRef]
- Fehrmann, H.; Dimond, A.E. Peroxidase activity and Phytophthora resistance in different organs of the potato plant. Phytopathology 1969, 57, 69–72. [Google Scholar]
- Lowry, O.H.; Rosebrough, N.J.; Farr, A.L.; Randall, R.J. Protein measurement with Folin reagent. J. Biol. Chem. 1951, 193, 265–275. [Google Scholar] [CrossRef]
- Foyer, C.H.; Noctor, G. Oxygen processing in photosynthesis: Regulation and signalling. N. Phytol. 2000, 146, 359–388. [Google Scholar] [CrossRef]
- Bi, J.L.; Felton, G.W. Foliar oxidative stress and insect herbivory: Primary compounds, secondary metabolites, and reactive oxygen species as components of induced resistance. J. Chem. Ecol. 1995, 21, 1511–1530. [Google Scholar] [CrossRef] [PubMed]
- Thaler, J.S.; Bostock, R.M. Interactions between abscisic-acid-mediated responses and plant resistance to pathogens and insects. Ecology 2004, 85, 48–58. [Google Scholar] [CrossRef]
- Régnière, J.; Powell, J.; Bentz, B.; Nealis, V. Effects of temperature on development, survival and reproduction of insects: Experimental design, data analysis and modeling. J. Insect Physiol. 2012, 58, 634–647. [Google Scholar] [CrossRef]
- Mołoń, M.; Dampc, J.; Kula-Maximenko, M.; Zebrowski, J.; Mołoń, A.; Dobler, R.; Durak, R.; Skoczowski, A. Effects of temperature on lifespan of Drosophila melanogaster from different genetic backgrounds: Links between metabolic rate and longevity. Insects 2020, 11, 470. [Google Scholar] [CrossRef]
- Durak, R.; Węgrzyn, E.; Leniowski, K. Do all aphids benefit from climate warming? An effect of temperature increase on a native species of temperate climatic zone Cinara juniperi. Ethol. Ecol. Evol. 2015, 28, 1–14. [Google Scholar] [CrossRef]
- Durak, R.; Dampc, J.; Dampc, J. Role of temperature on the interaction between Japanese quince Chaenomeles japonica and herbivorous insect Aphis pomi (Hemiptera: Aphidoidea). Environ. Exp. Bot. 2020, 176, 104100. [Google Scholar] [CrossRef]
- Davis, J.A.; Radcliffe, E.B.; Ragsdale, D.W. Effects of high and fluctuating temperatures on Myzus persicae (Hemiptera: Aphididae). Environ. Entomol. 2006, 35, 1461–1468. [Google Scholar] [CrossRef]
- Kuo, M.H.; Chiu, M.C.; Perng, J.J. Temperature effects on life history traits of the corn leaf aphid, Rhopalosiphum maidis (Homoptera: Aphididae) on corn in Taiwan. Appl. Entomol. Zool. 2006, 41, 171–177. [Google Scholar] [CrossRef]
- Chiu, M.-C.; Chen, Y.-H.; Kuo, M.-H. The effect of experimental warming on a low-latitude aphid, Myzus varians. Entomol. Exp. Appl. 2012, 142, 216–222. [Google Scholar] [CrossRef]
- Kikuchi, Y.; Tada, A.; Musolin, D.L.; Hari, N.; Hosokawa, T.; Fujisaki, K.; Fukatsu, T. Collapse of insect gut symbiosis under simulated climate change. MBio 2016, 7, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Ohtaka, C.; Ishikawa, H. Effects of heat treatment on the symbiotic system of an aphid mycetocyte. Symbiosis 1991, 11, 19–30. [Google Scholar]
- Dunbar, H.E.; Wilson, A.C.C.; Ferguson, N.R.; Moran, N.A. Aphid thermal tolerance is governed by a point mutation in bacterial symbionts. PLoS Biol. 2007, 5, e96. [Google Scholar] [CrossRef] [PubMed]
- Abdelsalam, S.A.; Awad, A.M.A.; Abdelrahman, M.A.A.; Nasser, M.A.K.; Abdelhamid, N.M.R. Antioxidant defense response of the green peach aphid, Myzus persicae against secondary metabolites of the host plants cumin, anise, and coriander. J. Agric. Sci. Technol. 2016, 18, 1583–1592. [Google Scholar]
- Ali, A.; Rashid, M.A.; Huang, Q.Y.; Wong, C.; Lei, C.-L. Response of antioxidant enzymes in Mythimna separata (Lepidoptera: Noctuidae) exposed to thermal stress. Bull. Entomol. Res. 2017, 107, 382–390. [Google Scholar] [CrossRef]
- Ferry, N.; Stavroulakis, S.; Guan, W.; Davison, G.M.; Bell, H.A.; Weaver, R.J.; Down, R.E.; Gatehouse, J.A.; Gatehouse, A.M.R. Molecular interactions between wheat and cereal aphid (Sitobion avenae): Analysis of changes to the wheat proteome. Proteomics 2011, 11, 1985–2002. [Google Scholar] [CrossRef]
- Mai, V.C.; Bednarski, W.; Borowiak-Sobkowiak, B.; Wilkaniec, B.; Samardakiewicz, S.; Morkunas, I. Oxidative stress in pea seedling leaves in response to Acyrthosiphon pisum infestation. Phytochemistry 2013, 93, 49–62. [Google Scholar] [CrossRef]
- Moloi, M.J.; van der Westhuizen, A.J. Antioxidative enzymes and the Russian wheat aphid (Diuraphis noxia) resistance response in wheat (Triticum aestivum). Plant Biol. 2008, 10, 403–407. [Google Scholar] [CrossRef]
- Durak, R.; Bednarski, W.; Formela-Luboińska, M.; Woźniak, A.; Borowiak-Sobkowiak, B.; Durak, T.; Dembczyński, R.; Morkunas, I. Defense responses of Thuja orientalis to infestation of anholocyclic species aphid Cinara tujafilina. J. Plant Physiol. 2019, 232, 160–170. [Google Scholar] [CrossRef] [PubMed]
- Grinnan, R.; Carter, T.E.; Johnson, M.T.J. Effects of drought, temperature, herbivory, and genotype on plant–insect interactions in soybean (Glycine max). Arthropod. Plant. Interact. 2013, 7, 201–215. [Google Scholar] [CrossRef]
- Dat, J.F.; Lopez-Delgado, H.; Foyer, C.H.; Scott, I.M. Effects of salicylic acid on oxidative stress and thermotolerance in tobacco. J. Plant Physiol. 2000, 156, 659–665. [Google Scholar] [CrossRef]
- Morkunas, I.; Mai, V.C.; Gabryś, B. Phytohormonal signaling in plant responses to aphid feeding. Acta Physiol. Plant. 2011, 33, 2057–2073. [Google Scholar] [CrossRef]
- Czerniewicz, P.; Sytykiewicz, H.; Durak, R.; Borowiak-Sobkowiak, B.; Chrzanowski, G. Role of phenolic compounds during antioxidative responses of winter triticale to aphid and beetle attack. Plant Physiol. Biochem. 2017, 118, 529–540. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Wen, D.; Sun, A.; Han, X.; Zhang, J.; Wang, Z.; Yin, Y. Differential activity and expression of antioxidant enzymes and alteration in osmolyte accumulation under high temperature stress in wheat seedlings. J. Cereal Sci. 2014, 60, 653–659. [Google Scholar] [CrossRef]
- Taggar, G.K.; Gill, R.S.; Gupta, A.K.; Sandhu, J.S. Fluctuations in peroxidase and catalase activities of resistant and susceptible black gram (Vigna mungo (L.) Hepper) genotypes elicited by Bemisia tabaci (Gennadius) feeding. Plant Signal. Behav. 2012, 7, 1321–1329. [Google Scholar] [CrossRef]
- Shao, Y.; Guo, M.; He, X.; Fan, Q.; Wang, Z.; Jia, J.; Guo, J. Constitutive H2O2 is involved in sorghum defense against aphids. Braz. J. Bot. 2019, 42, 271–281. [Google Scholar] [CrossRef]
- Durak, R.; Molon, M.; Durak, T.; Chrzanowski, G. The enzymatic markers of the adaptation of Cinara tujafilina to changing the host plant. Ethol. Ecol. Evol. 2018, 30, 416–429. [Google Scholar] [CrossRef]
- Sprawka, I.; Goławska, S.; Goławski, A.; Chrzanowski, G.; Czerniewicz, P.; Sytykiewicz, H. Entomotoxic action of jackbean lectin (Con A) in bird cherry-oat aphid through the effect on insect enzymes. J. Plant Interact. 2014, 9, 425–433. [Google Scholar] [CrossRef]
- Jena, K.; Kumar Kar, P.; Kausar, Z.; Babu, C.S. Effects of temperature on modulation of oxidative stress and antioxidant defenses in testes of tropical tasar silkworm Antheraea mylitta. J. Therm. Biol. 2013, 38, 199–204. [Google Scholar] [CrossRef]
- Kang, Z.-W.; Liu, F.-H.; Liu, X.; Yu, W.-B.; Tan, X.-L.; Zhang, S.-Z.; Tian, H.-G.; Liu, T.-X. The potential coordination of the heat-shock proteins and antioxidant enzyme genes of Aphidius gifuensis in response to thermal stress. Front. Physiol. 2017, 8, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Łukasik, I. Effect of host plant alternation on some adaptive enzymes of the bird cherry-oat aphid, Rhopalosiphum padi (L.). J. Pest Sci. 2009, 82, 203–209. [Google Scholar] [CrossRef]
- Mehrabadi, M.; Bandani, A.R.; Kwon, O. Biochemical characterization of digestive α-d-glucosidase and β-d-glucosidase from labial glands and midgut of wheat bug Eurygaster maura (Hemiptera: Scutelleridae). Entomol. Res. 2011, 41, 81–87. [Google Scholar] [CrossRef]
- Sytykiewicz, H.; Chrzanowski, G.; Czerniewicz, P.; Sprawka, I.; Łukasik, I.; Goławska, S.; Sempruch, C. Expression profiling of selected glutathione transferase genes in Zea mays (L.) seedlings infested with cereal aphids. PLoS ONE 2014, 9, e111863. [Google Scholar] [CrossRef]
- Moran, P.J.; Thompson, G.A. Molecular responses to aphid feeding in Arabidopsis in relation to plant defense pathways. Plant Physiol. 2001, 125, 1074–1085. [Google Scholar] [CrossRef]
- Morant, A.V.; Bjarnholt, N.; Kragh, M.E.; Kjærgaard, C.H.; Jørgensen, K.; Paquette, S.M.; Piotrowski, M.; Imberty, A.; Olsen, C.E.; Møller, B.L.; et al. The β-glucosidases responsible for bioactivation of hydroxynitrile glucosides in Lotus japonicus. Plant Physiol. 2008, 147, 1072–1091. [Google Scholar] [CrossRef]
- Tjallingii, W.F.; Esch, T.H. Fine structure of aphid stylet routes in plant tissues in correlation with EPG signals. Physiol. Entomol. 1993, 18, 317–328. [Google Scholar] [CrossRef]
- Urbanska, A.; Freddy Tjallingii, W.; Dixon, A.F.G.; Leszczynski, B. Phenol oxidising enzymes in the grain aphid’s saliva. Entomol. Exp. Appl. 1998, 86, 197–203. [Google Scholar] [CrossRef]
- Lattanzio, V.; Lattanzio, V.M.T.; Cardinali, A. Phytochemistry: Advances in Research; Research Signpost: Trivandrum, India, 2006; Volume 661, ISBN 8130800349. [Google Scholar]
- Cai, Q.-N.; Han, Y.; Cao, Y.-Z.; Hu, Y.; Zhao, X.; Bi, J.-L. Detoxification of gramine by the cereal aphid Sitobion avenae. J. Chem. Ecol. 2009, 35, 320–325. [Google Scholar] [CrossRef]
- Morkunas, I.; Gmerek, J. The possible involvement of peroxidase in defense of yellow lupine embryo axes against Fusarium oxysporum. J. Plant Physiol. 2007, 164, 185–194. [Google Scholar] [CrossRef]
- Park, S.-J.; Huang, Y.; Ayoubi, P. Identification of expression profiles of sorghum genes in response to greenbug phloem-feeding using cDNA subtraction and microarray analysis. Planta 2006, 223, 932–947. [Google Scholar] [CrossRef]
- Morkunas, I.; Woźniak, A.; Formela, M.; Mai, V.C.; Marczak, Ł.; Narożna, D.; Borowiak-Sobkowiak, B.; Kühn, C.; Grimm, B. Pea aphid infestation induces changes in flavonoids, antioxidative defence, soluble sugars and sugar transporter expression in leaves of pea seedlings. Protoplasma 2016, 253, 1063–1079. [Google Scholar] [CrossRef]
- Argandoña, V.H.; Chaman, M.; Cardemil, L.; Muñoz, O.; Zúñiga, G.E.; Corcuera, L.J. Ethylene production and peroxidase activity in aphid-infested barley. J. Chem. Ecol. 2001, 27, 53–68. [Google Scholar] [CrossRef] [PubMed]
- Dowd, P.F.; Johnson, E.T.; Pinkerton, T.S. Identification and properties of insect resistance-associated maize anionic peroxidases. Phytochemistry 2010, 71, 1289–1297. [Google Scholar] [CrossRef] [PubMed]
- Takahama, U. Oxidation of vacuolar and apoplastic phenolic substrates by peroxidase: Physiological significance of the oxidation reactions. Phytochem. Rev. 2004, 3, 207–219. [Google Scholar] [CrossRef]
- Mahalingam, R.; Fedoroff, N. Stress response, cell death and signalling: The many faces of reactive oxygen species. Physiol. Plant. 2003, 119, 56–68. [Google Scholar] [CrossRef]
| Temperature | 20 °C | 25 °C | 28 °C |
|---|---|---|---|
| Intrinsic rate of increase (rm) | 0.16 | 0.16 | 0.09 |
| Net reproductive rate (Ro) | 17.63 | 14.76 | 4.08 |
| Finite rate of increase (λ) | 1.17 | 1.17 | 1.09 |
| Generation time (T) | 17.94 | 16.82 | 15.62 |
| Doubling time (DT) | 4.33 | 4.33 | 7.7 |
| SOD | CAT | GST | β-Glucosidase | PPO | POD | |
|---|---|---|---|---|---|---|
| Aphid Tissue | ||||||
| T | F(2,36) = 461.22 *** | F(2,36) = 1.68 * | F(2,36) = 2.86 * | F(2,36) = 4.74 ** | F(2,36) = 5.18 ** | F(2,36) = 5.23 ** |
| t | F(5,36) = 240.31 *** | F(5,36) = 12.26 *** | F(5,36) = 3.14 * | F(5,36) = 2.25 | F(5,36) = 3.37 ** | F(5,36) = 18.43 *** |
| T × t | F(10,36) = 75.69 *** | F(10,36) = 2.42 * | F(10,36) = 0.79 | F(10,36) = 1.07 | F(10,36) = 2.40* | F(10,36) = 2.27 * |
| Plant Tissue | ||||||
| T | F(2,60) = 26.28 *** | F(2,60) = 12.21 *** | F(2,60) = 0.58 ** | F(2,60) = 2.40 | F(2,60) = 0.85 | F(2,60) = 2.83 |
| t | F(4,60) = 5.60 *** | F(4,60) = 181.09 *** | F(4,60) = 7.98 *** | F(4,60) = 1.16 | F(4,60) = 2.21 | F(4,60) = 5.70 *** |
| a | F(1,60) = 21.02 *** | F(1,60) = 238.87 *** | F(1,60) = 467.29 *** | F(1,60) = 12.40 *** | F(1,60) = 43.59 *** | F(1,60) = 224.25 *** |
| T × t | F(8,60) = 1.69 | F(8,60) = 38.74 *** | F(8,60) = 2.13 * | F(8,60) = 1.84 | F(8,60) = 2.95 *** | F(8,60) = 1.29 |
| T × a | F(2,60) = 17.11 *** | F(2,60) = 41.26 *** | F(2,60) = 7.05 *** | F(2,60) = 7.01 *** | F(2,60) = 14.59 *** | F(2,60) = 0.14 |
| t × a | F(4,60) = 5.83 *** | F(4,60) = 181.09 *** | F(4,60) = 7.99 *** | F(4,60) = 1.16 | F(4,60) = 2.21 | F(4,60) = 5.70 *** |
| T × t × a | F(8,60) = 1.75 | F(8,60) = 38.74 *** | F(8,60) = 2.13 * | F(8,60) = 1.84 | F(8,60) = 2.95*** | F(8,60) = 1.29 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dampc, J.; Mołoń, M.; Durak, T.; Durak, R. Changes in Aphid—Plant Interactions under Increased Temperature. Biology 2021, 10, 480. https://doi.org/10.3390/biology10060480
Dampc J, Mołoń M, Durak T, Durak R. Changes in Aphid—Plant Interactions under Increased Temperature. Biology. 2021; 10(6):480. https://doi.org/10.3390/biology10060480
Chicago/Turabian StyleDampc, Jan, Mateusz Mołoń, Tomasz Durak, and Roma Durak. 2021. "Changes in Aphid—Plant Interactions under Increased Temperature" Biology 10, no. 6: 480. https://doi.org/10.3390/biology10060480
APA StyleDampc, J., Mołoń, M., Durak, T., & Durak, R. (2021). Changes in Aphid—Plant Interactions under Increased Temperature. Biology, 10(6), 480. https://doi.org/10.3390/biology10060480







