Induction of Genomic Instability in a Primary Human Fibroblast Cell Line Following Low-Dose Alpha-Particle Exposure and the Potential Role of Exosomes
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Cell Culture
2.2. Exosome Isolation
2.3. Exosome Characterisation
2.4. Exosome Transfer for Bystander Experiments (Exosome Bystander)
2.5. Irradiation
2.5.1. Alpha-Particle Irradiation
2.5.2. X-ray
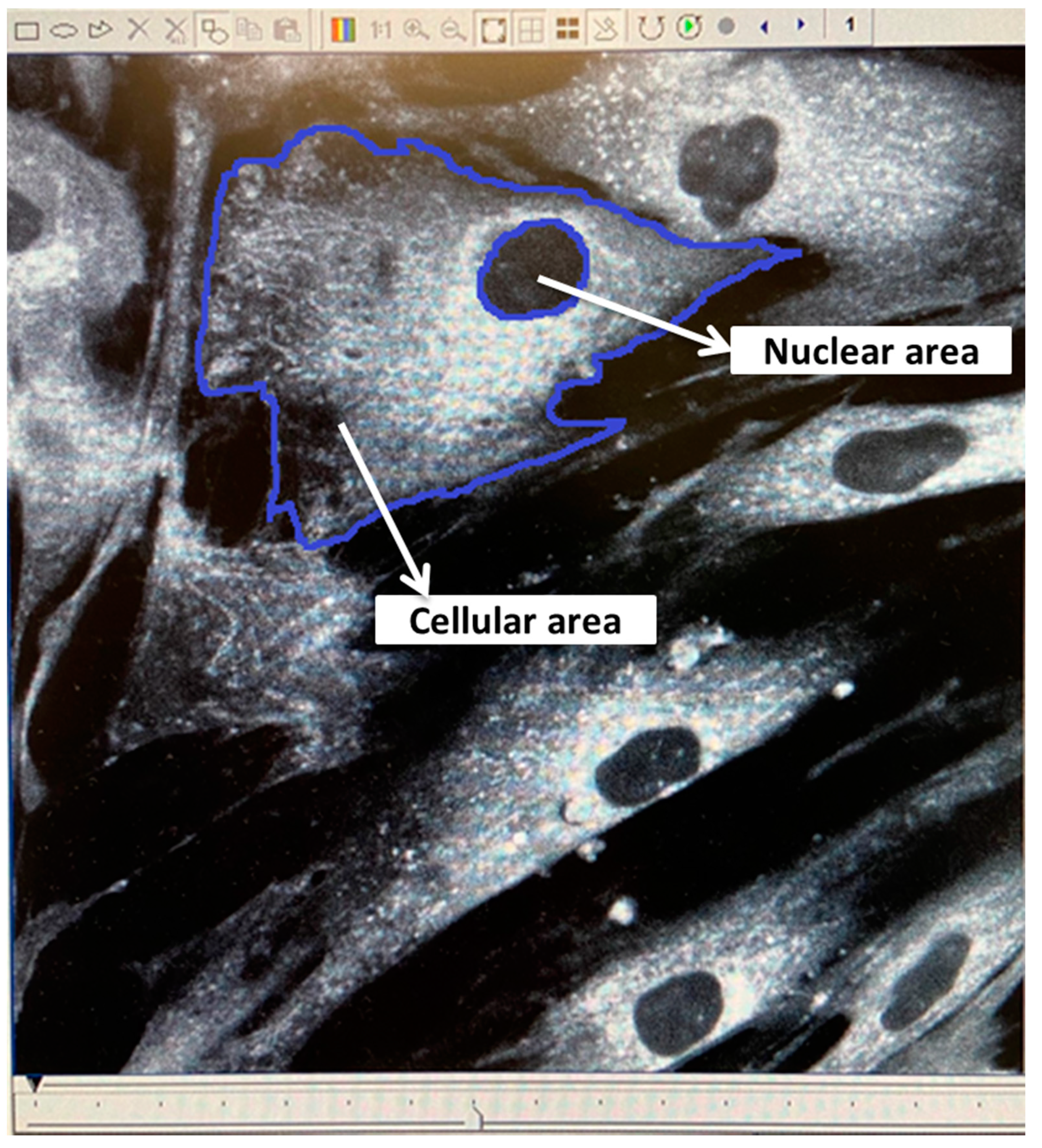
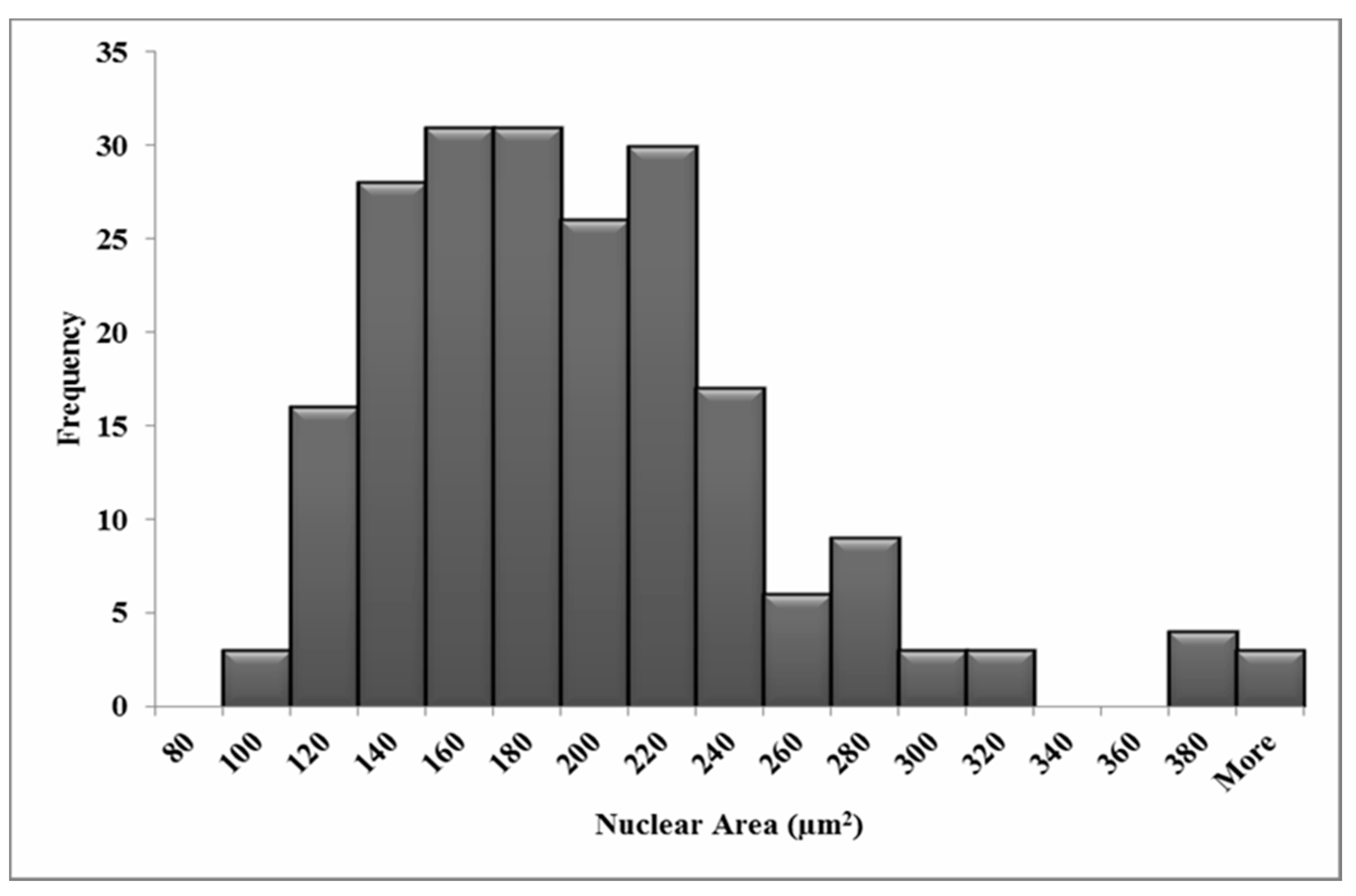
2.6. Measurements of Nuclear and Cellular Area
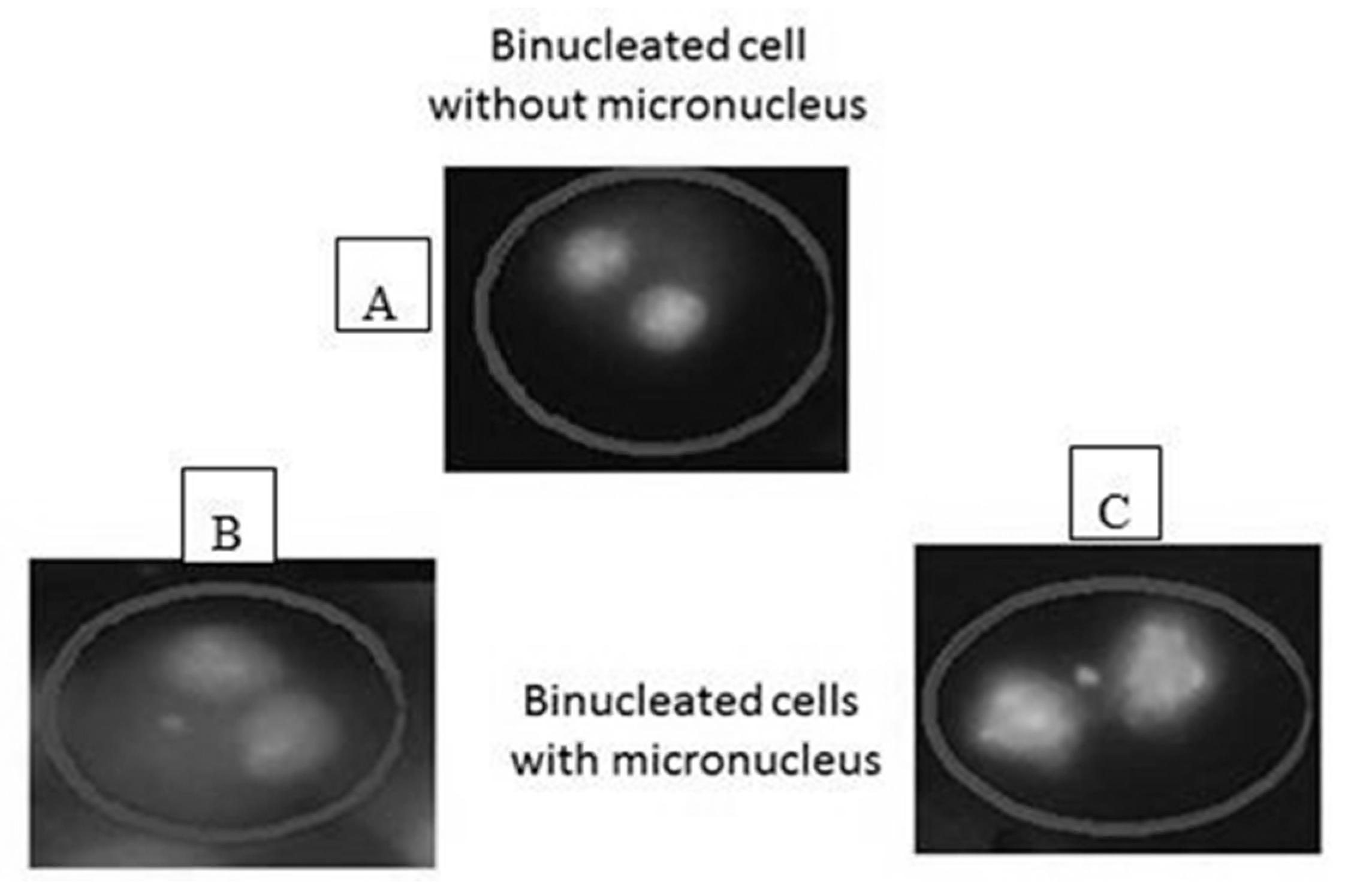
2.7. Micronucleus Assay
2.8. Staining of Slides
2.9. Scoring Micronuclei (MN)
2.10. Alkaline Comet Assay (Single Cell Gel Electrophoresis)
2.11. Statistical Analyses
3. Results
3.1. Calculating the Average Number of Alpha-Particle Track Traversals per Nucleus and per Cell
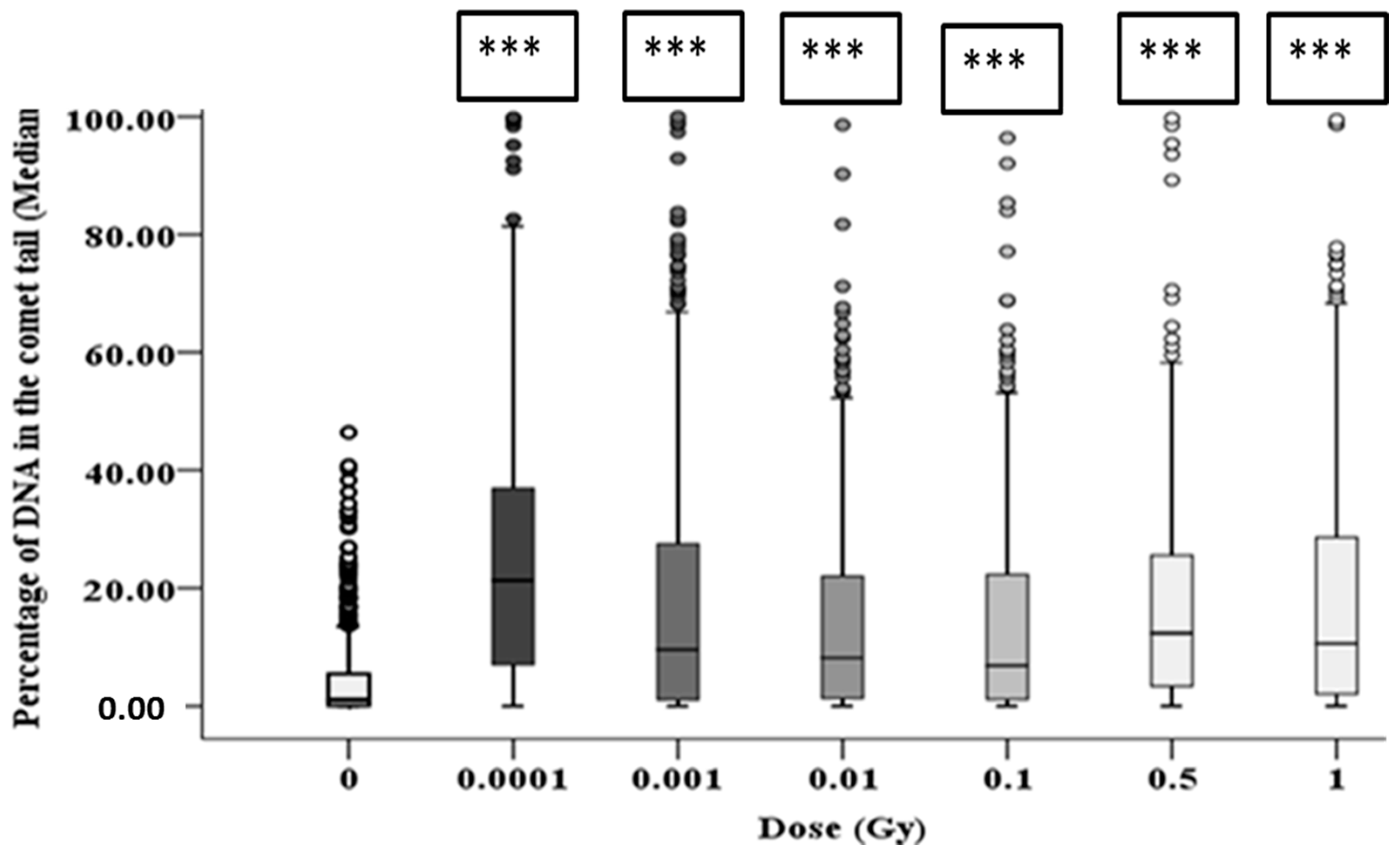
3.2. DNA Damage in HF19 Either Directly Irradiated or Bystander Cells Exposed to Exosomes from the Irradiated Cell Population
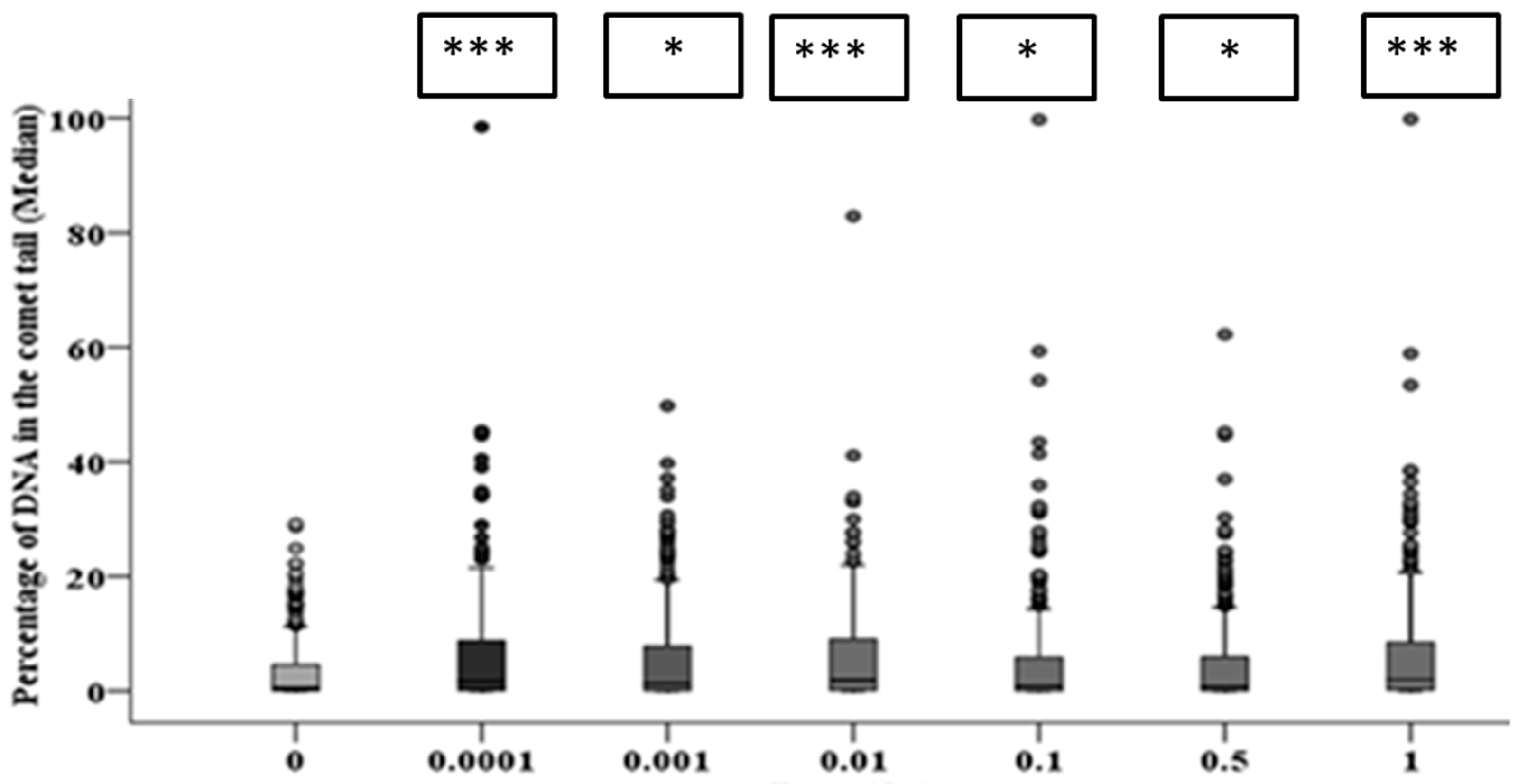
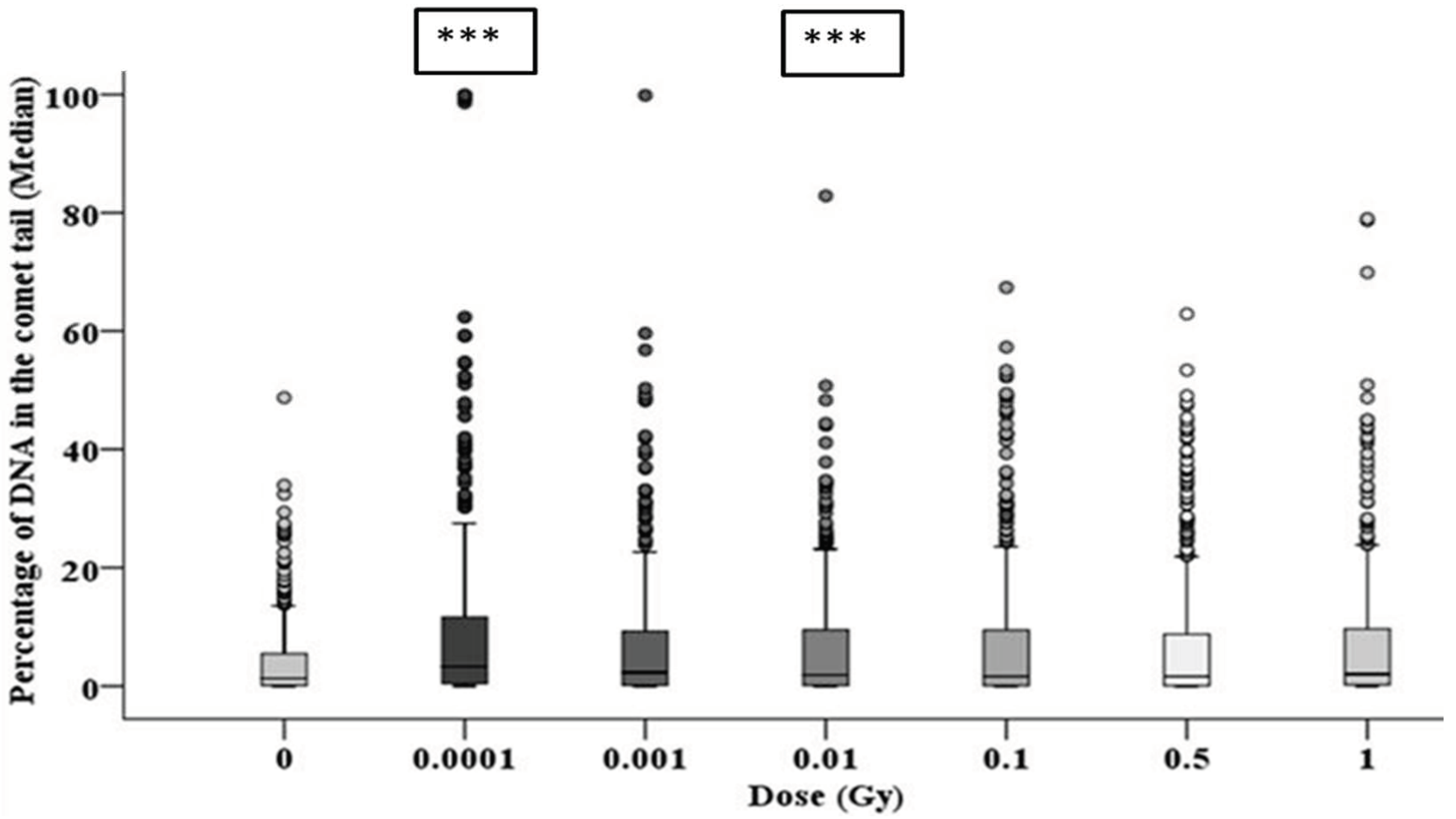
3.3. Early and Late DNA Damage in HF19 Cells Post Low and High Doses of Alpha-Particle Irradiation: Comet Assay
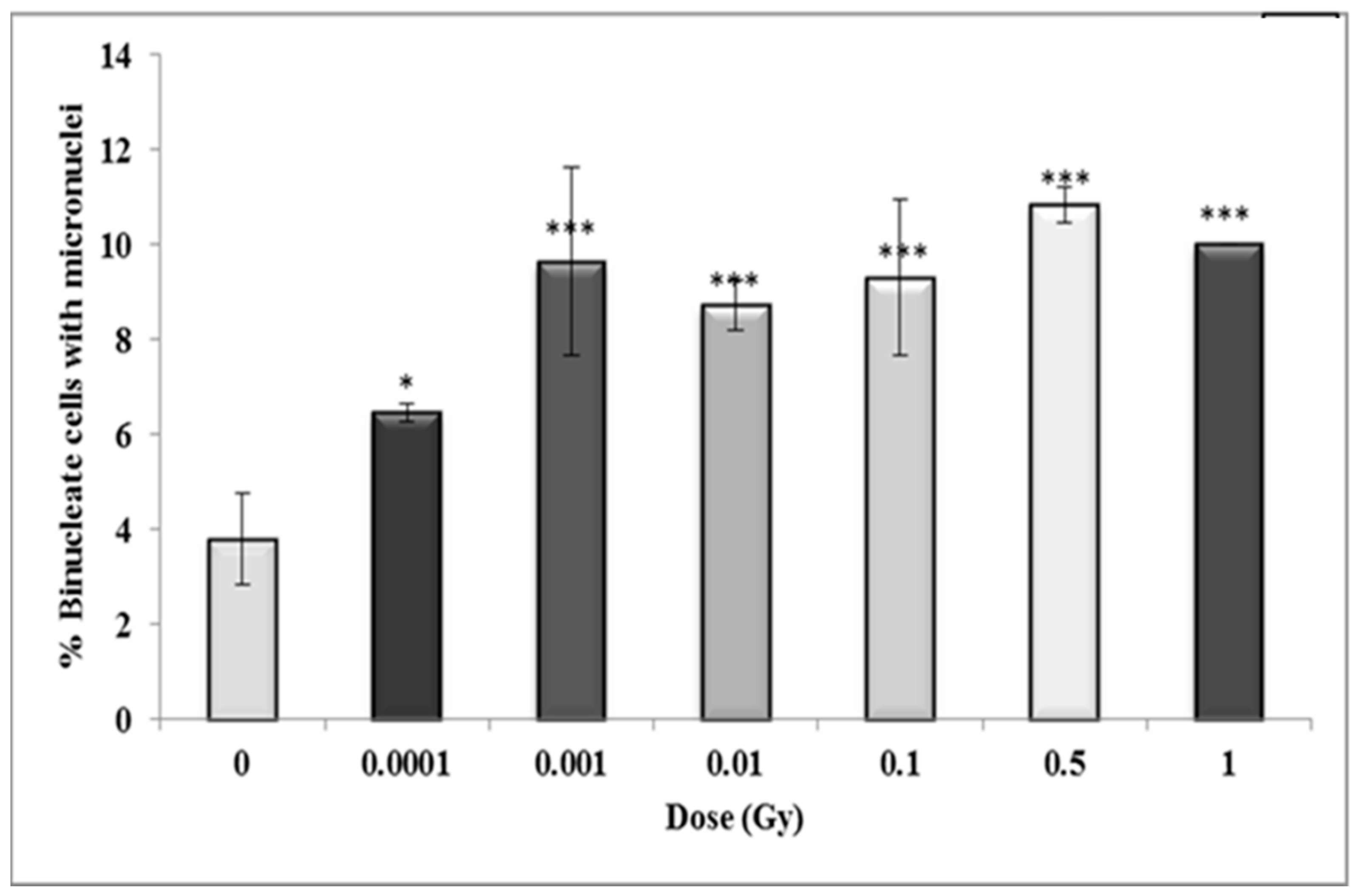
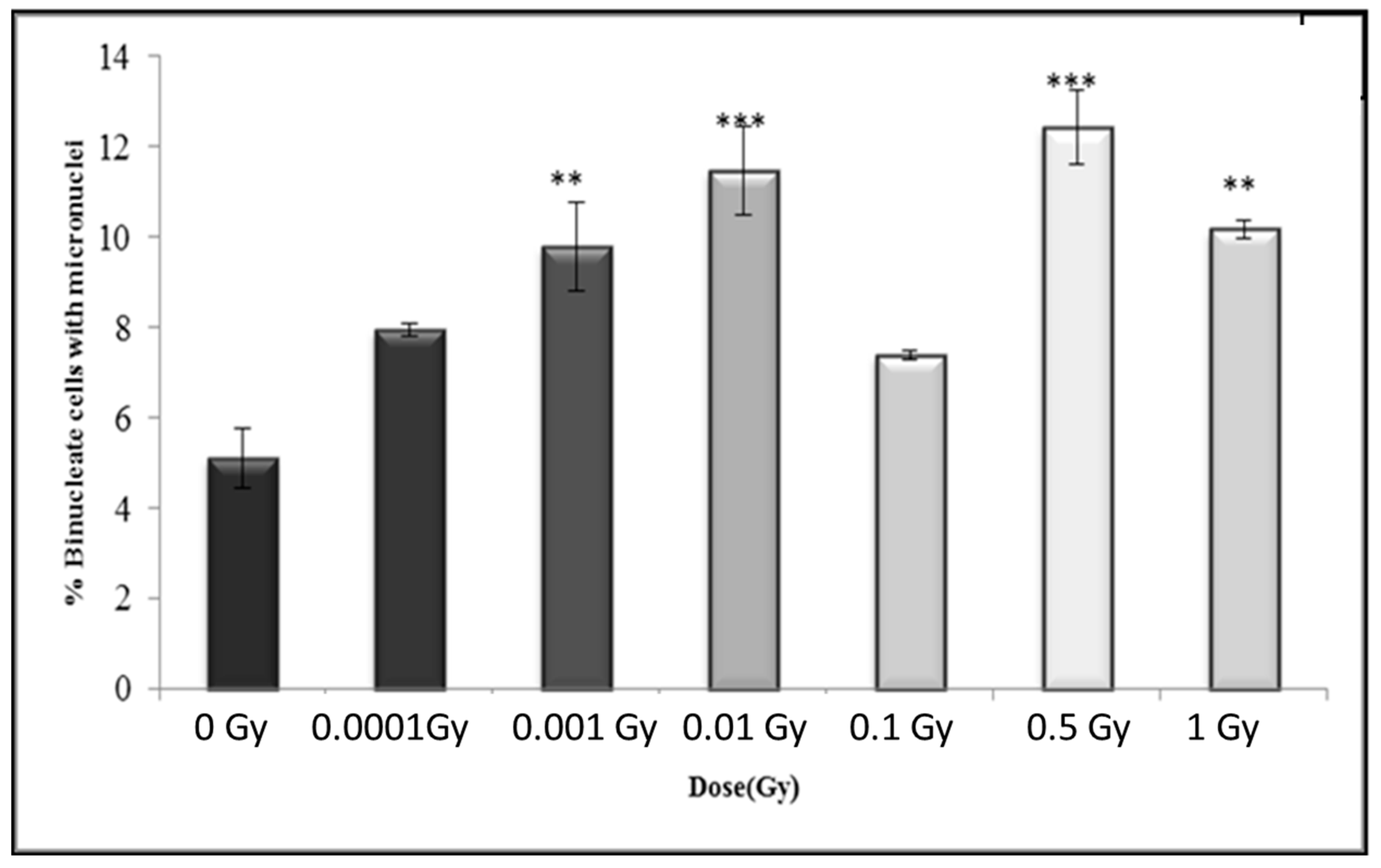
3.4. Early and Late Induction of Micronuclei in Binucleate HF 19 Cells Post Low and High Doses of Alpha-Particle Irradiation: Micronucleus Assay
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Milner, J.; Shrubsole, C.; Das, P.; Jones, B.; Ridley, I.; Chalabi, Z.; Hamilton, I.; Armstrong, B.; Davies, M.; Wilkinson, P. Home energy efficiency and radon related risk of lung cancer: Modelling study. BMJ 2014, 348, f7493. [Google Scholar] [CrossRef] [PubMed]
- Dionian, J.; Muirhead, C.R.; Wan, S.L.; Wrixon, A.D. The Risks of Leukaemia and Other Cancers in Thurso from Radiation Exposure; National Radiological Protection Board: Chilton, UK, 1986; p. 37. [Google Scholar]
- Maalouf, B.N.; Sartor, O.; Hauck, C.R.; Macklis, R. Targeted use of Alpha Particles: Current Status in Cancer Therapeutics. J. Nucl. Med. Radiat. Ther. 2012, 3. [Google Scholar] [CrossRef]
- Sumption, N.; Goodhead, D.T.; Anderson, R.M. Alpha-Particle-Induced Complex Chromosome Exchanges Transmitted through Extra-Thymic Lymphopoiesis In Vitro Show Evidence of Emerging Genomic Instability. PLoS ONE 2015, 10, e0134046. [Google Scholar] [CrossRef] [PubMed]
- Hazelton, W.D.; Luebeck, E.G.; Heidenreich, W.F.; Moolgavkar, S.H. Computational Approach for Determining the Spectrum of DNA Damage Induced by Ionizing Radiation. Radiat. Res. 2001, 156, 577–583. [Google Scholar] [CrossRef]
- Goodhead, D.T. Energy deposition stochastics and track structure: What about the target? Radiat. Prot. Dosim. 2006, 122, 3–15. [Google Scholar] [CrossRef]
- Dobbs, T.A.; Palmer, P.; Maniou, Z.; Lomax, M.E.; O’Neill, P. Interplay of two major repair pathways in the processing of complex double-strand DNA breaks. DNA Repair 2008, 7, 1372–1383. [Google Scholar] [CrossRef]
- Anderson, R.M.; Stevens, D.L.; Goodhead, D.T. M-FISH analysis shows that complex chromosome aberrations induced by α-particle tracks are cumulative products of localized rearrangements. Proc. Natl. Acad. Sci. USA 2002, 99, 12167–12172. [Google Scholar] [CrossRef]
- Hill, M.A. Radiation Track Structure: How the Spatial Distribution of Energy Deposition Drives Biological Response. Clin. Oncol. 2020, 32, 75–83. [Google Scholar] [CrossRef]
- National Research Council. Health Effects of Exposure to Radon (BEIR VI); The National Academies Press: Washington, DC, USA, 1999. [CrossRef]
- Kadhim, M.; Salomaa, S.; Wright, E.; Hildebrandt, G.; Belyakov, O.V.; Prise, K.M.; Little, M.P. Non-targeted effects of ionising radiation—Implications for low dose risk. Mutat. Res. Mutat. Res. 2013, 752, 84–98. [Google Scholar] [CrossRef]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef]
- Kadhim, M.A.; Marsden, S.J.; Malcolmson, A.M.; Folkard, M.; Goodhead, D.T.; Prise, K.M.; Michael, B.D. Long-term genomic instability in human lymphocytes induced by single-particle irradiation. Radiat. Res. 2001, 155, 122–126. [Google Scholar] [CrossRef]
- Kadhim, M.A.; Macdonald, D.A.; Goodhead, D.T.; Lorimore, S.A.; Marsden, S.J.; Wright, E.G. Transmission of chromosomal instability after plutonium α-particle irradiation. Nature 1992, 355, 738–740. [Google Scholar] [CrossRef] [PubMed]
- Hei, T.K.; Zhou, H.; Ivanov, V.N.; Hong, M.; Lieberman, H.B.; Brenner, D.J.; Amundson, S.A.; Geard, C.R. Mechanism of radiation-induced bystander effects: A unifying model. J. Pharm. Pharmacol. 2008, 60, 943–950. [Google Scholar] [CrossRef] [PubMed]
- Barker, H.E.; Paget, J.T.E.; Khan, A.A.; Harrington, K.J. The tumour microenvironment after radiotherapy: Mechanisms of resistance and recurrence. Nat. Rev. Cancer 2015, 15, 409–425. [Google Scholar] [CrossRef] [PubMed]
- Lorimore, S.A.; Kadhim, M.A.; Pocock, D.A.; Papworth, D.; Stevens, D.L.; Goodhead, D.T.; Wright, E.G. Chromosomal instability in the descendants of unirradiated surviving cells after α-particle irradiation. Proc. Natl. Acad. Sci. USA 1998, 95, 5730–5733. [Google Scholar] [CrossRef]
- Kadhim, M.; Lorimore, S.; Hepburn, M.; Goodhead, D.; Buckle, V.; Wright, E. α-particle-induced chromosomal instability in human bone marrow cells. Lancet 1994, 344, 987–988. [Google Scholar] [CrossRef]
- Morgan, W.F. Non-targeted and delayed effects of exposure to ionizing radiation: II. Radiation-induced genomic instability and bystander effects in vivo, clastogenic factors and transgenerational effects. Radiat. Res. 2003, 159, 581–596. [Google Scholar] [CrossRef]
- Février, B.; Raposo, G. Exosomes: Endosomal-Derived Vesicles Shipping Extracellular Messages. Curr. Opin. Cell Biol. 2004, 16, 415–421. [Google Scholar] [CrossRef]
- Shim, G.; Normil, M.D.; Testard, I.; Hempel, W.M.; Ricoul, M.; Sabatier, L. Comparison of Individual Radiosensitivity to γ-Rays and Carbon Ions. Front. Oncol. 2016, 6, 137. [Google Scholar] [CrossRef]
- Yang, M.; Li, Y.; Chilukuri, K.; Brady, O.A.; Boulos, M.I.; Kappes, J.C.; Galileo, D.S. L1 stimulation of human glioma cell motility correlates with FAK activation. J. Neuro-Oncol. 2011, 105, 27–44. [Google Scholar] [CrossRef]
- Jella, K.K.; Rani, S.; O’Driscoll, L.; McClean, B.; Byrne, H.J.; Lyng, F. Exosomes Are Involved in Mediating Radiation Induced Bystander Signaling in Human Keratinocyte Cells. Radiat. Res. 2014, 181, 138–145. [Google Scholar] [CrossRef]
- Kadhim, M.; Bright, S.; Al-Mayah, A.; Goodwin, E. The Emerging Role of Exosomes in the Biological Processes Initiated by Ionizing Radiation. Available online: https://three.jsc.nasa.gov/articles/Exosomes_Kadhim.pdf (accessed on 5 October 2020).
- De Toro, J.; Herschlik, L.; Waldner, C.; Mongini, C. Emerging Roles of Exosomes in Normal and Pathological Conditions: New Insights for Diagnosis and Therapeutic Applications. Front. Immunol. 2015, 6, 203. [Google Scholar] [CrossRef] [PubMed]
- Cox, R.; Masson, W. X-ray survival curves of cultured human diploid fibroblasts. In Cell survival after low doses of radiation: Theoretical and clinical implications. In Proceedings of the Sixth L.H. Gray Conference, Bedford, London, 16–21 September 1974. [Google Scholar]
- Al-Mayah, A.H.J.; Irons, S.L.; Pink, R.C.; Carter, D.R.F.; Kadhim, M.A. Possible Role of Exosomes Containing RNA in Mediating Nontargeted Effect of Ionizing Radiation. Radiat. Res. 2012, 177, 539–545. [Google Scholar] [CrossRef] [PubMed]
- Al-Mayah, A.H.J.; Bright, S.; Bowler, D.A.; Slijepcevic, P.; Goodwin, E.; Kadhim, M. Exosome-Mediated Telomere Instability in Human Breast Epithelial Cancer Cells after X Irradiation. Radiat. Res. 2017, 187, 98–106. [Google Scholar] [CrossRef] [PubMed]
- Al-Mayah, A.; Bright, S.; Chapman, K.; Irons, S.; Luo, P.; Carter, D.; Goodwin, E.; Kadhim, M.A. The non-targeted effects of radiation are perpetuated by exosomes. Mutat. Res. Mol. Mech. Mutagen. 2015, 772, 38–45. [Google Scholar] [CrossRef]
- Bowler, D.A.; Moore, S.R.; Macdonald, D.A.; Smyth, S.H.; Clapham, P.; Kadhim, M.A. Bystander-mediated genomic instability after high LET radiation in murine primary haemopoietic stem cells. Mutat. Res. Mol. Mech. Mutagen. 2006, 597, 50–61. [Google Scholar] [CrossRef] [PubMed]
- Goodhead, D.; Bance, D.; Stretch, A.; Wilkinson, R. A Versatile Plutonium-238 Irradiator for Radiobiological Studies with α-particles. Int. J. Radiat. Biol. 1991, 59, 195–210. [Google Scholar] [CrossRef]
- Hill, M.A.; Herdman, M.T.; Stevens, D.L.; Jones, N.J.; Thacker, J.; Goodhead, D.T. Relative Sensitivities of Repair-Deficient Mammalian Cells for Clonogenic Survival after α-Particle Irradiation. Radiat. Res. 2004, 162, 667–676. [Google Scholar] [CrossRef]
- Erexson, G.; KligermaN, A.; Allen, J. Diaziquone-Induced Micronuclei in Cytochalasin B-Blocked Mouse Peripheral Blood Lymphocytes. Mutat. Res. 1987, 178, 117–122. [Google Scholar] [CrossRef]
- Erexson, G.L.; Kligerman, A.D. A modified mouse peripheral blood lymphocyte culture system for cytogenetic analysis. Environ. Mol. Mutagen. 1987, 10, 377–386. [Google Scholar] [CrossRef]
- Azzam, E.I.; De Toledo, S.M.; Spitz, D.R.; Little, J.B. Oxidative Metabolism Modulates Signal Transduction and Micronucleus Formation in Bystander Cells from α-Particle-Irradiated Normal Human Fibroblast Cultures. Cancer Res. 2002, 62, 5436–5442. [Google Scholar]
- Liao, W.; McNutt, M.A.; Zhu, W.-G. The comet assay: A sensitive method for detecting DNA damage in individual cells. Methods 2009, 48, 46–53. [Google Scholar] [CrossRef] [PubMed]
- Natarajan, M.; Gibbons, C.F.; Mohan, S.; Moore, S.; A Kadhim, M. Oxidative stress signalling: A potential mediator of tumour necrosis factor α-induced genomic instability in primary vascular endothelial cells. Br. J. Radiol. 2007, 80, S13–S22. [Google Scholar] [CrossRef] [PubMed]
- Olive, P.L.; Banáth, J.P. The comet assay: A method to measure DNA damage in individual cells. Nat. Protoc. 2006, 1, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Salkind, N.J.; Frey, B.B. Statistics for People Who (Think They) Hate Statistics; SAGE Publications: New York, NY, USA, 2019; pp. 400–472. [Google Scholar]
- Goodhead, D.T. Mechanisms for the biological effectiveness of high-LET radiations. J. Radiat. Res. 1999, 40, 1–13. [Google Scholar] [CrossRef]
- Azzam, E.I.; De Toledo, S.M.; Little, J.B. Direct evidence for the participation of gap junction-mediated intercellular communication in the transmission of damage signals from α-particle irradiated to nonirradiated cells. Proc. Natl. Acad. Sci. USA 2001, 98, 473–478. [Google Scholar] [CrossRef]
- Lin, Y.-F.; Nagasawa, H.; Little, J.B.; Kato, T.A.; Shih, H.-Y.; Xie, X.-J.; Wilson, P.F., Jr.; Brogan, J.R.; Kurimasa, A.; Chen, D.J.; et al. Differential Radiosensitivity Phenotypes of DNA-PKcs Mutations Affecting NHEJ and HRR Systems following Irradiation with Gamma-Rays or Very Low Fluences of Alpha Particles. PLoS ONE 2014, 9, e93579. [Google Scholar] [CrossRef]
- Kanagaraj, K.; Rajan, V.; Pandey, B.N.; Thayalan, K.; Venkatachalam, P. Primary and secondary bystander effect and genomic instability in cells exposed to high and low linear energy transfer radiations. Int. J. Radiat. Biol. 2019, 95, 1648–1658. [Google Scholar] [CrossRef]
- Dröge, W. Free Radicals in the Physiological Control of Cell Function. Physiol. Rev. 2002, 82, 47–95. [Google Scholar] [CrossRef]
- Wilkins, R.C.; Rodrigues, M.A.; Beaton-Green, L.A. The application of imaging flow cytometry to high-throughput biodosimetry. Genome Integr. 2017, 8, 7. [Google Scholar] [CrossRef][Green Version]
- Iyer, R.; Lehnert, B.E. Factors Underlying the Cell Growth-Related Bystander Responses to α Particles. Cancer Res. 2000, 60, 1290–1298. [Google Scholar]
- Wuttke, K.; Müller, W.-U.; Streffer, C. The sensitivity of the in vitro cytokinesis-blocked micronucleus assay in lymphocytes for different and combined radiation qualities. Strahlenther. und Onkol. 1998, 174, 262–268. [Google Scholar] [CrossRef] [PubMed]
- Portess, D.I.; Bauer, G.; Hill, M.A.; O’Neill, P. Low-Dose Irradiation of Nontransformed Cells Stimulates the Selective Removal of Precancerous Cells via Intercellular Induction of Apoptosis. Cancer Res. 2007, 67, 1246–1253. [Google Scholar] [CrossRef] [PubMed]
- Abdelrazzak, A.B.; Stevens, D.L.; Bauer, G.; O’Neill, P.; Hill, M.A. The Role of Radiation Quality in the Stimulation of Intercellular Induction of Apoptosis in Transformed Cells at Very Low Doses. Radiat. Res. 2011, 176, 346–355. [Google Scholar] [CrossRef]
- Morgan, W.F. Non-Targeted and Delayed Effects of Exposure to Ionizing Radiation: I. Radiation-Induced Genomic Instability and Bystander Effects in Vitro. Radiat. Res. 2003, 159, 567–580. [Google Scholar] [CrossRef]
- Mavragani, I.V.; Nikitaki, Z.; Kalospyros, S.A.; Cetin, Z. Ionizing Radiation and Complex DNA Damage: From Prediction to Detection Challenges and Biological Significance. Cancers 2019, 11, 1789. [Google Scholar] [CrossRef]
- Moore, S.R.; Marsden, S.; Macdonald, D.; Mitchell, S.; Folkard, M.; Michael, B.; Goodhead, D.T.; Prise, K.M.; Kadhim, M.A. Genomic Instability in Human Lymphocytes Irradiated with Individual Charged Particles: Involvement of Tumor Necrosis Factor α in Irradiated Cells but not Bystander Cells. Radiat. Res. 2005, 163, 183–190. [Google Scholar] [CrossRef]
- Wrigh, M.A.K.S.J.M.E.G. Radiation-induced chromosomal instability in human fibroblasts: Temporal effects and the influence of radiation quality. Int. J. Radiat. Biol. 1998, 73, 143–148. [Google Scholar] [CrossRef]
- A Ghandhi, S.; Yaghoubian, B.; Amundson, S.A. Global gene expression analyses of bystander and alpha particle irradiated normal human lung fibroblasts: Synchronous and differential responses. BMC Med Genom. 2008, 1, 63. [Google Scholar] [CrossRef]
- Belli, M.; Tabocchini, M.A. Ionizing Radiation-Induced Epigenetic Modifications and Their Relevance to Radiation Protection. Int. J. Mol. Sci. 2020, 21, 5993. [Google Scholar] [CrossRef]




| Dose (Gy) | Calculated Average α-Particle Track Traversals per Cell (% of Cells Traversed by 1 or More Tracks) | Calculated Average α-Particle Track Traversals per Nucleus (% of Nuclei Traversed by 1 or More Tracks) |
|---|---|---|
| 0.0001 | 0.008 (0.80%) | 0.00098 (0.098%) |
| 0.001 | 0.080 (7.73%) | 0.0098 (0.967%) |
| 0.01 | 0.804 (55.3%) | 0.0976 (9.3%) |
| 0.1 | 8.04 (100%) | 0.976 (62.3%) |
| 0.5 | 40.2 (100%) | 4.88 (99.2%) |
| 1 | 80.4 (100%) | 9.76 (100%) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Elbakrawy, E.M.; Mayah, A.; Hill, M.A.; Kadhim, M. Induction of Genomic Instability in a Primary Human Fibroblast Cell Line Following Low-Dose Alpha-Particle Exposure and the Potential Role of Exosomes. Biology 2021, 10, 11. https://doi.org/10.3390/biology10010011
Elbakrawy EM, Mayah A, Hill MA, Kadhim M. Induction of Genomic Instability in a Primary Human Fibroblast Cell Line Following Low-Dose Alpha-Particle Exposure and the Potential Role of Exosomes. Biology. 2021; 10(1):11. https://doi.org/10.3390/biology10010011
Chicago/Turabian StyleElbakrawy, Eman Mohammed, Ammar Mayah, Mark A. Hill, and Munira Kadhim. 2021. "Induction of Genomic Instability in a Primary Human Fibroblast Cell Line Following Low-Dose Alpha-Particle Exposure and the Potential Role of Exosomes" Biology 10, no. 1: 11. https://doi.org/10.3390/biology10010011
APA StyleElbakrawy, E. M., Mayah, A., Hill, M. A., & Kadhim, M. (2021). Induction of Genomic Instability in a Primary Human Fibroblast Cell Line Following Low-Dose Alpha-Particle Exposure and the Potential Role of Exosomes. Biology, 10(1), 11. https://doi.org/10.3390/biology10010011






