Can X-ray Diffraction Distinguish Natural from Anthropogenic Hematite? Replication of the Conversion of Natural Goethite in Both Furnace and Campfire
Abstract
1. Introduction
2. Materials and Methods
2.1. X-ray Diffractometry (XRD)
2.2. Ochre Samples
2.3. Sample Heating
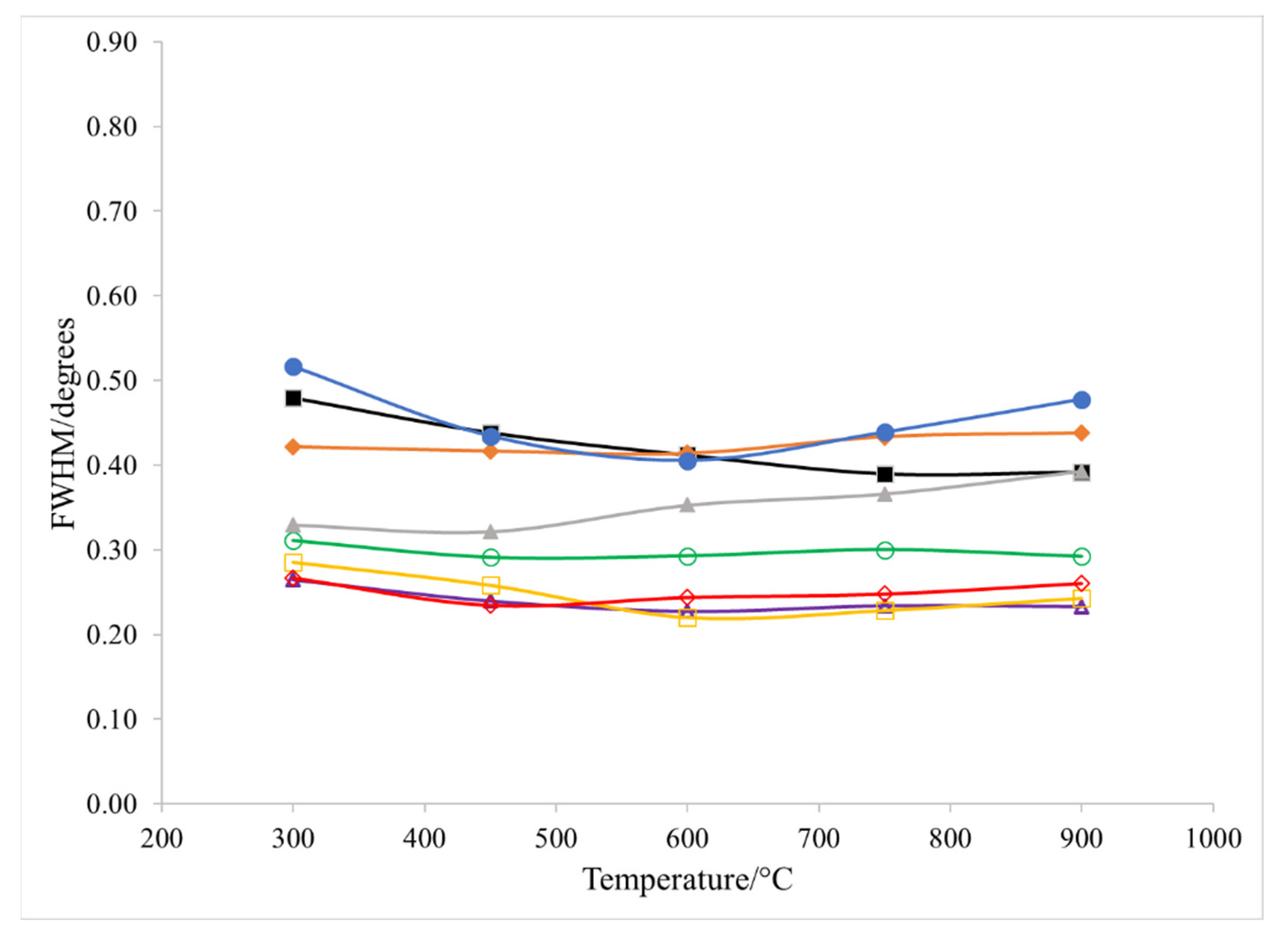
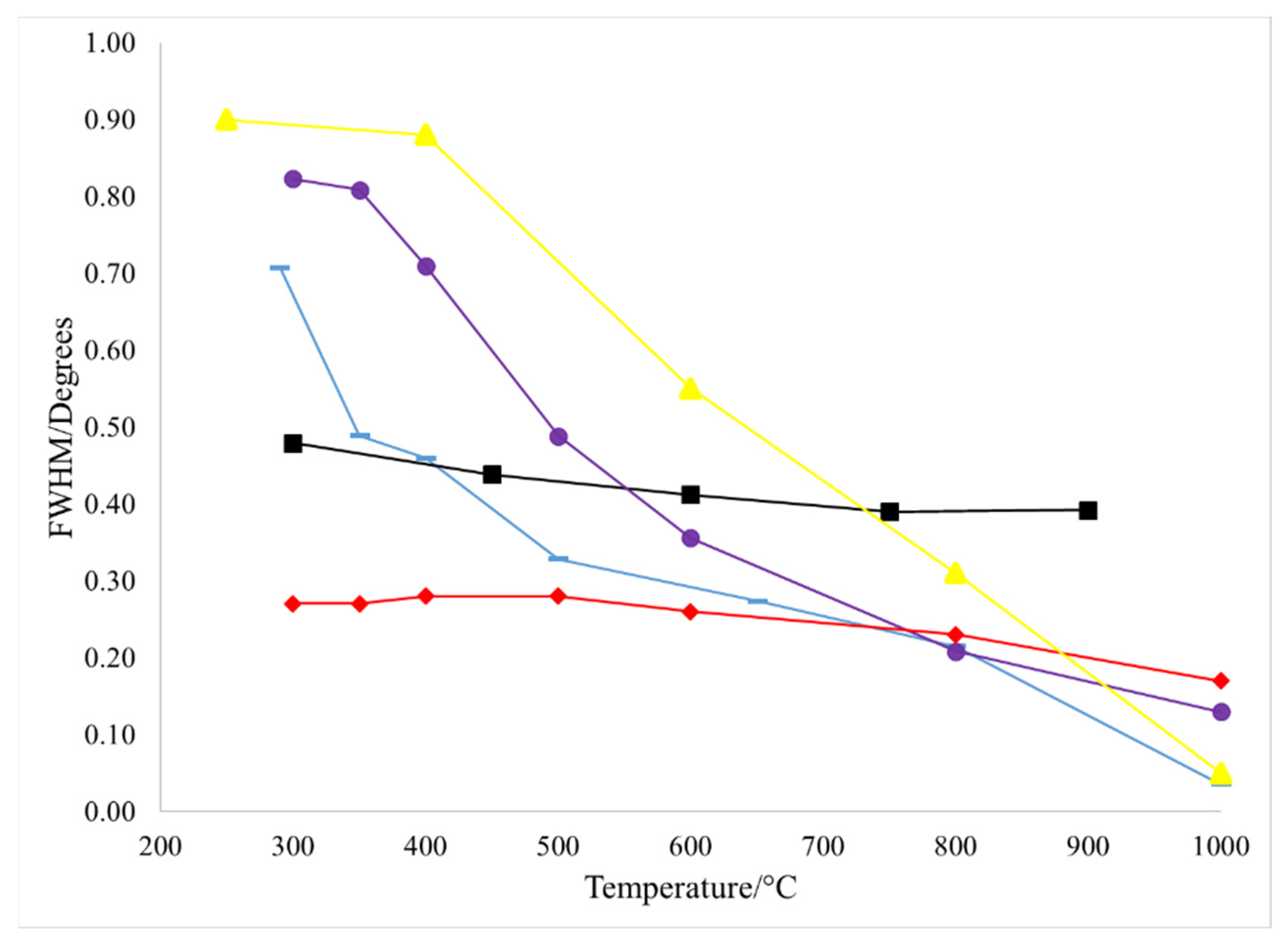
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Barham, L.S. Systematic pigment use in the Middle Pleistocene of South-Central Africa. Curr. Anthr. 2002, 43, 181–190. [Google Scholar] [CrossRef]
- Wadley, L. Putting ochre to the test: Replication studies of adhesives that may have been used for hafting tools in the Middle Stone Age. J. Hum. Evol. 2005, 49, 587–601. [Google Scholar] [CrossRef] [PubMed]
- Rifkin, R. Assessing the efficacy of red ochre as a prehistoric hide tanning ingredient. J. Afr. Archaeol. 2011, 9, 131–158. [Google Scholar] [CrossRef]
- Rifkin, R. Processing ochre in the Middle Stone Age: Testing the inference of prehistoric behaviours from actualistically derived experimental data. J. Anthropol. Archaeol. 2012, 31, 174–195. [Google Scholar] [CrossRef]
- Cavallo, G.; Fontana, F.; Gialanella, S.; Gonzato, F.; Riccardi, M.P.; Zorzin, R.; Peresani, M. Heat Treatment of Mineral Pigment During the Upper Palaeolithic in North-East Italy. Archaeometry 2018, 60, 1045–1061. [Google Scholar] [CrossRef]
- Dayet, L.; Le Bourdonnec, F.-X.; Daniel, F.; Porraz, G.; Texier, P.-J. Ochre provenance and procurement strategies during the middle stone age at Diepkloof Rock Shelter, South Africa. Archaeometry 2016, 58, 807–829. [Google Scholar] [CrossRef]
- Gialanella, S.; Belli, R.; Dalmeri, G.; Lonardelli, I.; Mattarelli, M.; Montagna, M.; Toniutti, L. Artificial or natural origin of hematite-based red pigments in archeological contexts: The case of Riparo Dalmeri (Trento, Italy). Archaeometry 2011, 53, 950–962. [Google Scholar] [CrossRef]
- Henshilwood, C.S.; D’Errico, F.; van Niekerk, K.L.; Coquinot, Y.; Jacobs, Z.; Lauritzen, S.-E.; Menu, M.; García-Moreno, R. A 100,000-year-old ochre-processing workshop at Blombos Cave, South Africa. Science 2011, 334, 219–222. [Google Scholar] [CrossRef]
- Hodgskiss, T. Identifying grinding, scoring and rubbing use-wear on experimental ochre pieces. J. Archaeol. Sci. 2010, 37, 3344–3358. [Google Scholar] [CrossRef]
- Hodgskiss, T.; Wadley, L. How people used ochre at Rose Cottage Cave, South Africa: Sixty thousand years of evidence from the Middle Stone Age. PLoS ONE 2017, 12, e0176317. [Google Scholar] [CrossRef]
- MacDonald, B.L.; Fox, W.; Dubreuil, L.; Beddard, J.; Pidruczny, A. Iron oxide geochemistry in the Great Lakes Region (North America): Implications for ochre provenance studies. J. Archaeol. Sci. Rep. 2018, 19, 476–490. [Google Scholar] [CrossRef]
- MacDonald, B.L.; Stalla, D.; He, X.; Maschmann, M.R.; Emerson, D.; White, T.A. Reconstructing rock art Fe-oxide pigment pyrotechnology using in situ SEM heating experiments. Microsc. Microanal. 2018, 24, 2124–2125. [Google Scholar] [CrossRef]
- MacDonald, B.L.; Chatters, J.C.; Reinhardt, E.G.; Devos, F.; Meacham, S.; Rissolo, D.; Rock, B.; Le Maillot, C.; Stalla, D.; Marino, M.D.; et al. Paleoindian ochre mines in the submerged caves of the Yucatán Peninsula, Quintana Roo, Mexico. Sci. Advant. 2020, 6, eaba1219. [Google Scholar] [CrossRef] [PubMed]
- Pomiès, M.; Morin, G.; Vignaud, C. XRD study of the goethite-hematite transformation: Application to the identification of heated prehistoric pigments. Eur. J. Solid State Inorg. Chem. 1998, 35, 9–25. [Google Scholar] [CrossRef]
- Pradeau, J.-V.; Salomon, H.; Bon, F.; Mensan, R.; Lejay, M.; Regert, M. Les matières colorantes sur le site aurignacien de plein air de Régismont-le-Haut (Poilhes, Hérault). Bull. De La Soc. Préhistorique Fr. 2014, 111, 631–658. [Google Scholar] [CrossRef][Green Version]
- Rosso, D.E.; Martí, P.; D’Errico, F. Middle Stone Age Ochre Processing and Behavioural Complexity in the Horn of Africa: Evidence from Porc-Epic Cave, Dire Dawa, Ethiopia. PLoS ONE 2016, 11, e0164793. [Google Scholar] [CrossRef]
- Salomon, H.; Goemaere, E.; Billard, C.; Dreesen, R.; Bosquet, D.; Hamon, C.; Jadin, I. Analyse critique du protocole de caracterisation des hematites oolithiques mis en place dans le cadre du projet collectif de recherché sur L’origine des hematites oolithiques durant la prehistoire recent entre I’Eifel (DE) et la Normandie (FR). Anthropol. Et Praehist. 2016, 125, 225–246. [Google Scholar]
- Velliky, E.C.; Porr, M.; Conard, N.J. Ochre and pigment use at Hohle Fels cave: Results of the first systematic review of ochre and ochre-related artefacts from the Upper Palaeolithic in Germany. PLoS ONE 2018, 13, e0209874. [Google Scholar] [CrossRef]
- Villa, P.; Pollarolo, L.; Degano, I.; Birolo, L.; Pasero, M.; Biagioni, C.; Douka, K.; Vinciguerra, R.; Lucejko, J.J.; Wadley, L. A milk and ochre paint mixture used 49,000 years ago at Sibudu, South Africa. PLoS ONE 2015, 10, e0131273. [Google Scholar] [CrossRef]
- Wadley, L. Cemented ash as a receptacle or work surface for ochre powder production at Sibudu, South Africa, 58,000 years ago. J. Archaeol. Sci. 2010, 37, 2397–2406. [Google Scholar] [CrossRef]
- Watts, I. The pigments from Pinnacle Point Cave 13B, Western Cape, South Africa. J. Hum. Evol. 2010, 59, 392–411. [Google Scholar] [CrossRef] [PubMed]
- Wojcieszak, M.; Hodgskiss, T.; Colomban, P.; Wadley, L. Finding chemical and physical evidence of heat treatment of ochre by using non-destructive methods: A preliminary study. In The Exploitation of Raw Materials in Prehistory: Sourcing, Processing and Distribution; Pereira, T., Terradas, X., Bicho, N., Eds.; Cambridge Scholars Publishing: Newcastle upon Tyne, UK, 2017; pp. 587–600. [Google Scholar]
- Wojcieszak, M.; Wadley, L. A Raman micro-spectroscopy study of 77,000 to 71,000 year old ochre processing tools from Sibudu, KwaZulu-Natal, South Africa. Herit. Sci. 2019, 7, 24. [Google Scholar] [CrossRef]
- Helwig, K. A note on burnt yellow earth pigments: Documentary sources and scientific analysis. Stud. Conserv. 1997, 42, 181–188. [Google Scholar]
- Bentsen, S.E. Controlling the heat: An experimental approach to Middle Stone Age pyrotechnology. South Afr. Archaeol. Bull. 2011, 68, 137–145. [Google Scholar]
- Wadley, L. Post-depositional heating may cause over-representation of red-coloured ochre in Stone Age sites. South Afr. Archaeol. Bull. 2009, 64, 166–171. [Google Scholar]
- Onoratini, G.; Perinet, G. Données minéralogiques sur les colorants rouges préhistoriques de Provence: Démonstration que certains d’ entre eux ont été obtenus par calcination de goethite. C. R. Acad. Sc. Paris 1985, 301, 119–124. [Google Scholar]
- Pomiès, M.P.; Menu, M.; Vignaud, C. Red Paleolithic pigments: Natural hematite or heated goethite? Archaeometry 1999, 41, 275–285. [Google Scholar] [CrossRef]
- Pomiès, M.P.; Menu, M.; Vignaud, C. TEM observations of goethite dehydration: Application to archaeological samples. J. Eur. Ceram. Soc. 1999, 19, 1605–1614. [Google Scholar]
- Pomiès, M.P.; Barbaza, M.; Menu, M.; Vignaud, C. Preparation des pigments rouges prehistoriques par chauffage. L’Anthroplogie 1999, 103, 503–518. [Google Scholar]
- Schwertmann, U.; Cornell, R.M. Iron Oxides in the Laboratory, Preparation and Characterization; VCH: Weinheim, Germany, 1991. [Google Scholar]
- Löffler, L.; Mader, W. Anisotropic X-ray broadening and twin formation in hematite derived from natural and synthetic goethite. J. Eur. Ceram. Soc. 2006, 26, 131–139. [Google Scholar] [CrossRef]
- Cavallo, G.; Ischia, G.; Zorzin, R.; Gialanella, S. Experimental analysis on natural heated goethite from ponte di Veja (Mt Lessini, NE Italy). J. Archaeol. Sci. Rep. 2021, 36, 102871. [Google Scholar] [CrossRef]
- Salomon, H.; Vignaud, C.; Coquinot, Y.; Beck LStringer, C.; Strivay, D.; D’Errico, F. Selection and heating of colouring materials in the Mousterian level of Es-Skhul (c. 100, 000 years BP, Mount Carmel, Israel). Archaeometry 2012, 54, 698–722. [Google Scholar] [CrossRef]
- Salomon, H.; Vignaud, C.; Lahlil, S.; Menguy, N. Solutrean and Magdalenian ferruginous rocks heat-treatment: Accidental and/or deliberate action? J. Archaeol. Sci. 2015, 55, 100–112. [Google Scholar] [CrossRef]
- Cavallo, G.; Riccardi, M.P.; Zorzin, R. Powder diffraction of yellow and red natural earths from Lessini Mountains in NE Italy. Powder Diffr. 2015, 30, 122–129. [Google Scholar] [CrossRef]
- Cavallo, G.; Fontana, F.; Gonzato, F.; Guerreschi, A.; Riccardi, M.P.; Sardelli, G.; Zorzin, R. Sourcing and processing of ochre during the late upper Palaeolithic at Tagliente rock-shelter (NE Italy) based on conventional X-ray powder diffraction analysis. Archaeol. Anthr. Sci. 2015, 9, 763–775. [Google Scholar] [CrossRef]
- Cavallo, G.; Fontana, F.; Gonzato, F.; Peresani, M.; Riccardi, M.P.; Zorzin, R. Textural, microstructural, and compositional characteristics of Fe-based geomaterials and Upper Paleolithic ochre in the Lessini Mountains. Northeast Italy: Implications for provenance studies. Geoarchaeology 2017, 32, 437–455. [Google Scholar] [CrossRef]
- Gialanella, S.; Girardi, F.; Ischia, G.; Lonardelli, I.; Mattarelli, M.; Montagna, M. On the goethite to hematite phase transformation. J. Therm. Anal. 2010, 102, 867–873. [Google Scholar] [CrossRef]
- Román, R.S.; Ruiz, M.D.L.; Juan-Juan, J.; Bañón, C.B.; Straus, L.G.; Morales, M.R.G. Sources of the ochres associated with the Lower Magdalenian “Red Lady” human burial and rock art in El Mirón Cave (Cantabria, Spain). J. Archaeol. Sci. Rep. 2019, 23, 265–280. [Google Scholar]
- Aldeias, V. Experimental approaches to archaeological fire features and their behavioral relevance. Curr. Anthropol. 2017, 58, S191–S205. [Google Scholar] [CrossRef]
- March, R.J.; Lucquin, A.; Joly, D.; Ferreri, J.C.; Muhieddine, M. Processes of formation and alteration of archaeological fire structures: Complexity viewed in the light of experimental approaches. J. Archaeol. Method Theory 2014, 21, 1–45. [Google Scholar] [CrossRef]
- Henshilwood, C.S.; Sealy, J.C.; Yates, R.; Cruz-Uribe, K.; Goldberg, P.; Grine, F.E.; Klein, R.G.; Poggenpoel, C.; van Niekerk, K.; Watts, I. Blombos Cave, Southern Cape, South Africa: Preliminary report on the 1992–1999 excavations of the Middle Stone Age levels. J. Archaeol. Sci. 2001, 28, 421–448. [Google Scholar] [CrossRef]
- Henshilwood, C.S.; D’Errico, F.; Van Niekerk, K.L.; Dayet, L.; Queffelec, A.; Pollarolo, L. An abstract drawing from the 73,000-year-old levels at Blombos Cave, South Africa. Nature 2018, 562, 115–118. [Google Scholar] [CrossRef]
- Wadley, L. Ochre crayons or waste products? Replications compared with MSA ‘crayons’ from Sibudu Cave, South Africa. Farming 2005, 3, 1–12. [Google Scholar] [CrossRef]
- Lorblanchet, M.; Labeau, M.; Vernet, J.L.; Fitte, P.; Valladas, H.; Cachier, H.; Arnold, M. Palaeolithic pigments in the Quercy, France. Rock Art Res. 1990, 7, 4–12. [Google Scholar]
- Lin, Z.; Natoli, J.M.; Picuri, J.C.; Shaw, S.E.; Bowyer, W.J. Replication of the conversion of goethite to hematite to make pigments in both furnace and campfire. J. Archaeol. Sci. Rep. 2021, 39, 103134. [Google Scholar] [CrossRef]
- Elias, M.; Chartier, C.; Prévot, G.; Garay, H.; Vignaud, C. The colour of ochres explained by their composition. Mater. Sci. Eng. B 2006, 127, 70–80. [Google Scholar] [CrossRef]
- Garofano, I.; Perez-Rodriguez, J.L.; Robador, M.D.; Duran, A. An innovative combination of non-invasive UV–Visible-FORS, XRD and XRF techniques to study Roman wall paintings from Seville, Spain. J. Cult. Herit. 2016, 22, 1028–1039. [Google Scholar] [CrossRef]
- Goss, C.J. The kinetics and reaction mechanism of the goethite to hematite transformation. Miner. Mag. 1987, 51, 437–451. [Google Scholar] [CrossRef]
- Lima-de-Faria, J. Dehydration of goethite and diaspore. Z. Für Krist. Bd. 1963, 119, 176–293. [Google Scholar] [CrossRef]
- Liu, H.; Chen, T.; Zou, X.; Qing, C.; Frost, R.L. Thermal treatment of natural goethite: Thermal transformation and physical properties. Thermochim. Acta 2013, 568, 115–121. [Google Scholar] [CrossRef]
- Botticelli, M.; Appolloni, G.; Candeias, A.; Mirão, J.; Maras, A. Synthesis of yellow and red ochre: A preliminary multianalytical, micro-invasive investigation on recipes and recognition criteria. Eur. Phys. J. Plus 2021, 136, 1–19. [Google Scholar] [CrossRef]
- Garilli, V.; Vita, G.; La Parola, V.; Vraca, M.P.; Giarrusso, R.; Rosina, P.; Bonfiglio, L.; Sineo, L. First evidence of Pleistocene ochre production from bacteriogenic iron oxides. A case study of the Upper Palaeolithic site at the San Teodoro Cave (Sicily, Italy). J. Archaeol. Sci. 2020, 123, 105221. [Google Scholar] [CrossRef]


| h k l | d/Å | 2θ/° |
|---|---|---|
| 0 1 2 | 3.705 | 23.96 |
| 1 0 4 | 2.713 | 32.96 |
| 1 1 0 | 2.528 | 35.42 |
| 1 1 3 | 2.215 | 40.66 |
| 0 2 4 | 1.846 | 49.31 |
| 1 1 6 | 1.699 | 53.88 |
| 2 1 4 | 1.489 | 62.26 |
| 3 0 0 | 1.456 | 63.82 |
| h k l | Natural a | Natural b | Natural c | Synthetic d | Synthetic e |
|---|---|---|---|---|---|
| 0 1 2 | 2.2 | 0.52 | 12 | 17 | 11 |
| 1 0 4 | 0.26 | 0.07 | 16 | 20. | 11 |
| 0 2 4 | 3.7 | 0.42 | 8.4 | 18 | 9.2 |
| 2 1 4 | −0.78 | −1.2 | 6.6 | 22 | 14 |
| average | 1.3 | −0.05 | 11 | 19 | 11 |
| 10-NFireP | 11-NFireP | |||
|---|---|---|---|---|
| h k l | FWHM/° | Temp./°C | FWHM/° | Temp./°C |
| 0 1 2 | 0.40 | 600 | 0.41 | 600 |
| 1 0 4 | 0.32 | <300 | 0.40 | >900 |
| 0 2 4 | 0.36 | >900 | 0.49 | 350 |
| 2 1 4 | 0.51 | <300 | 0.47 | <300 |
| Sample | Heat | Time/h | Temp/°C | Powder or Lump |
|---|---|---|---|---|
| 14-NFireP | Fire 2 | 2 | 300–600 | Powder |
| 20-NFireP | Fire 3 | 2 | 250–600 | Powder |
| 27-NFireL | Fire 4 | 2 | 300–500 | Lump |
| 28-NFireL | Fire 5 | 1.5 | 600–750 | Lump |
| 36-NFurnL | Furnace | 1 | 600 | Lump |
| 37-NFurnL | Furnace | 1 | 600 | Lump |
| h k l | 14-NFireP | 20-NFireP | 27-NFireL | 28-NFireL |
|---|---|---|---|---|
| 0 1 2 | 500 | 350 | 350 | >900 |
| 1 0 4 | >900 | >900 | 750 | >900 |
| 0 2 4 | NP | >900 | 350 | 500 |
| 2 1 4 | <300 | <300 | <300 | <300 |
| h k l | 5-NFurnP | 36-NFurnL | 37-NFurnL |
|---|---|---|---|
| 0 1 2 | 0.412 | 0.371 | 0.398 |
| 1 0 4 | 0.414 | 0.465 | 0.204 |
| 0 2 4 | 0.405 | 0.425 | 0.494 |
| 2 1 4 | 0.353 | 0.573 | 0.430 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Picuri, J.C.; Natoli, J.M.; Shaw, S.E.; Shyam, S.P.; VanHoesen, S.R.; Lin, Z.; Bowyer, W.J. Can X-ray Diffraction Distinguish Natural from Anthropogenic Hematite? Replication of the Conversion of Natural Goethite in Both Furnace and Campfire. Colorants 2022, 1, 363-375. https://doi.org/10.3390/colorants1030022
Picuri JC, Natoli JM, Shaw SE, Shyam SP, VanHoesen SR, Lin Z, Bowyer WJ. Can X-ray Diffraction Distinguish Natural from Anthropogenic Hematite? Replication of the Conversion of Natural Goethite in Both Furnace and Campfire. Colorants. 2022; 1(3):363-375. https://doi.org/10.3390/colorants1030022
Chicago/Turabian StylePicuri, Jules C., Julia M. Natoli, Sophia E. Shaw, Shruthi P. Shyam, Stephen R. VanHoesen, Zhenyu Lin, and Walter J. Bowyer. 2022. "Can X-ray Diffraction Distinguish Natural from Anthropogenic Hematite? Replication of the Conversion of Natural Goethite in Both Furnace and Campfire" Colorants 1, no. 3: 363-375. https://doi.org/10.3390/colorants1030022
APA StylePicuri, J. C., Natoli, J. M., Shaw, S. E., Shyam, S. P., VanHoesen, S. R., Lin, Z., & Bowyer, W. J. (2022). Can X-ray Diffraction Distinguish Natural from Anthropogenic Hematite? Replication of the Conversion of Natural Goethite in Both Furnace and Campfire. Colorants, 1(3), 363-375. https://doi.org/10.3390/colorants1030022






