Antifungal Composite Fibers Based on Cellulose and Betulin
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Dopes and Fiber Spinning (Films Formation)
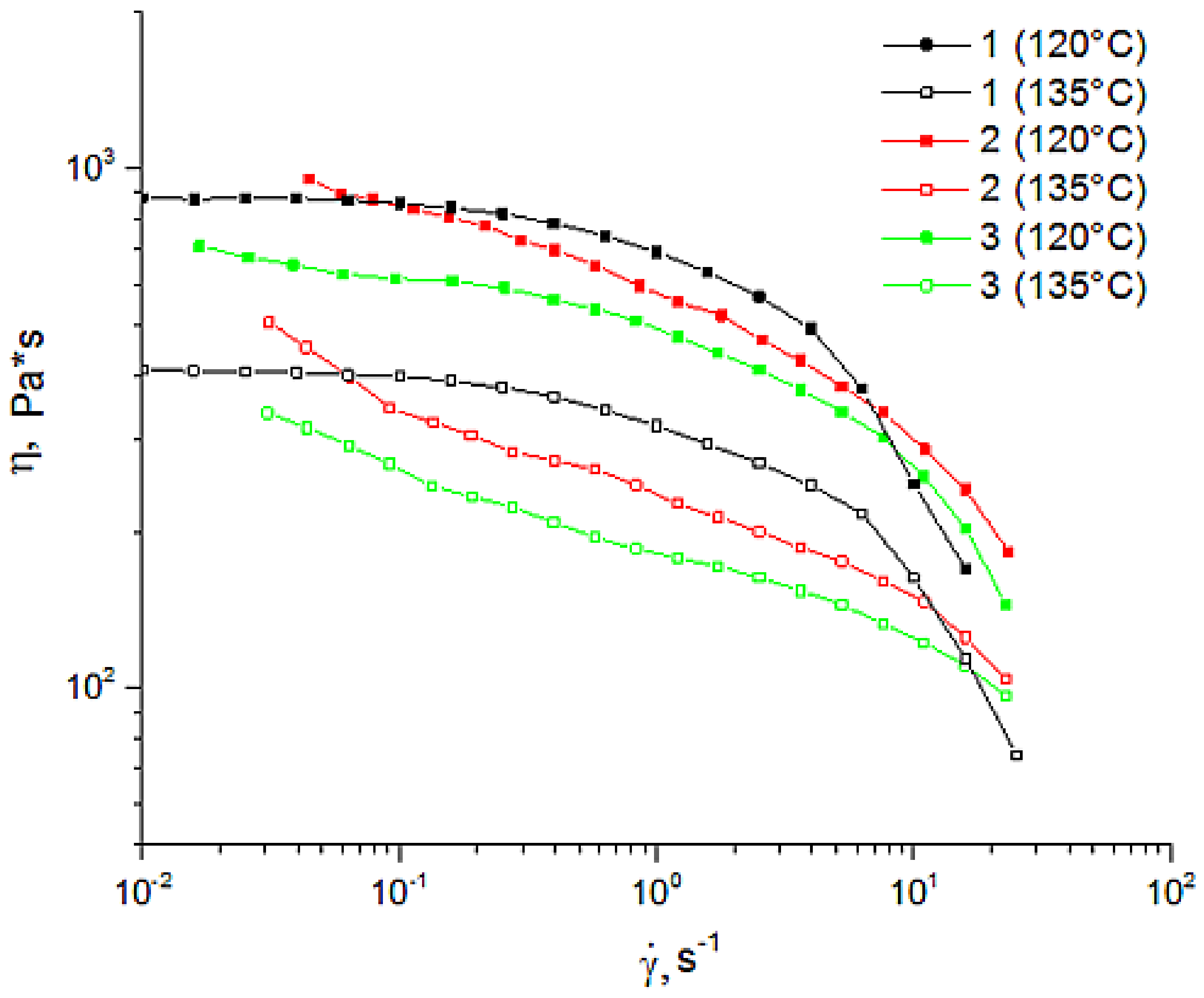
2.3. Rheology
2.4. Mechanical Test
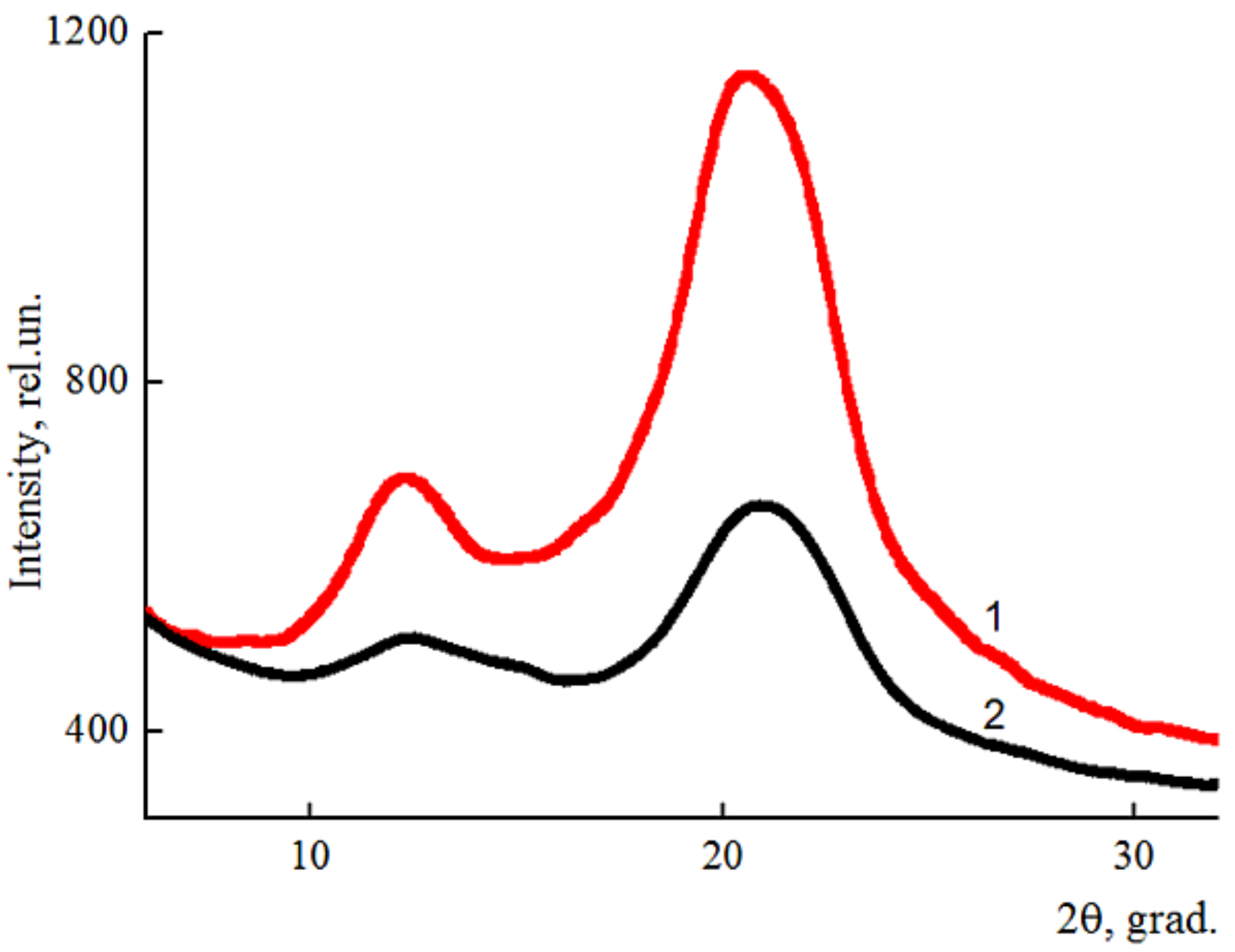
2.5. Wide-Angle X-ray Diffraction (WAXD)
2.6. Scanning Electron Microscopy (SEM)
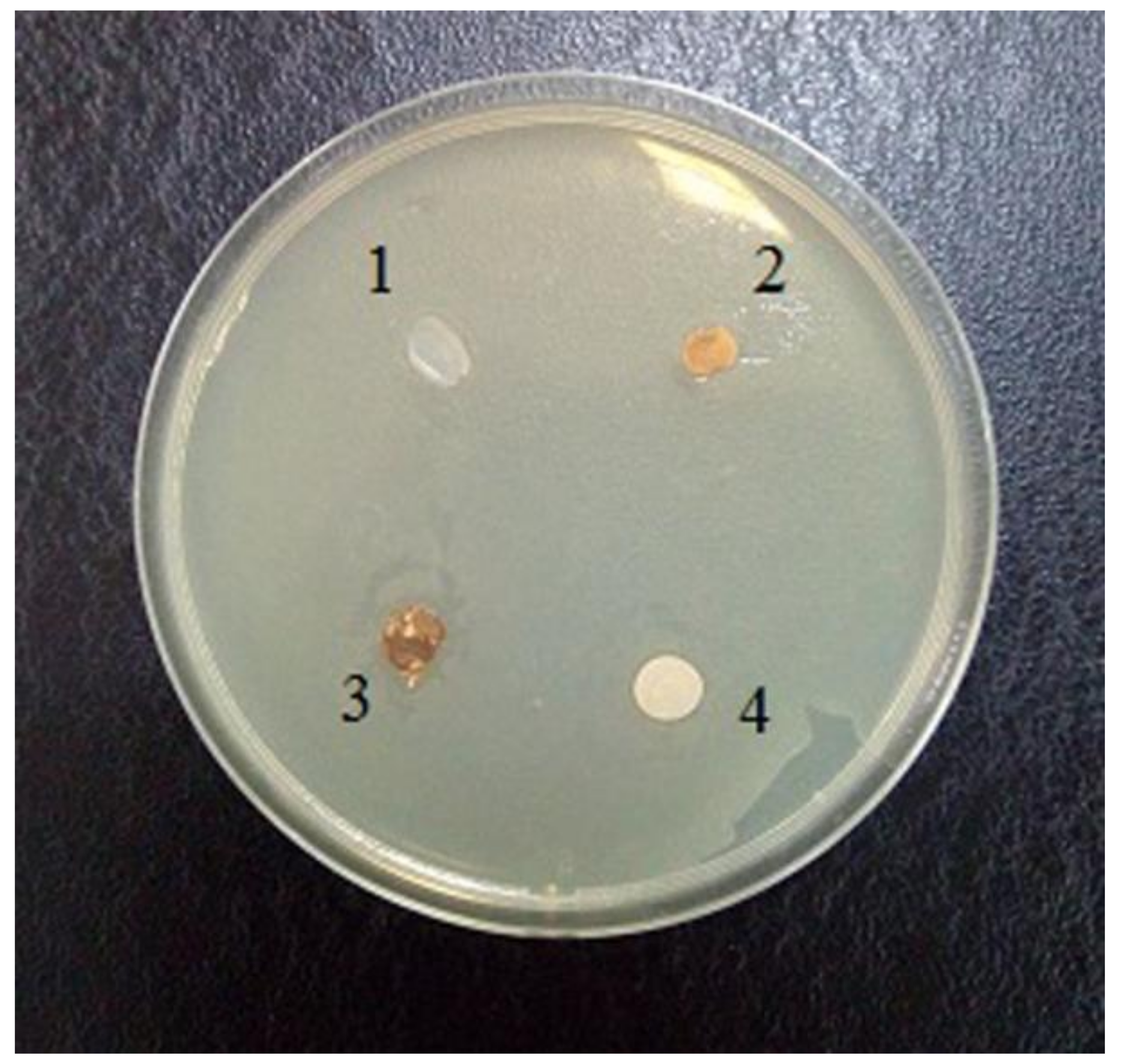
2.7. Fungal and Antimicrobial Resistance Tests
3. Results and Discussion
3.1. Morphology and Rheology
3.2. Mechanical Properties
3.3. Structure
3.4. Fungal Resistance
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- McCance, K.L.; Huether, S.E. Pathophysiology-E-Book: The Biologic Basis for Disease in Adults and Children (Pathophysiology the Biologic Basis), 7th ed.; Elsevier Health Sciences: London, UK, 2014; p. 1864. [Google Scholar]
- Voronchikhin, V.D.; Bondar, P.N. Evaluation of the fungi resistance of film-forming agents used to modify the surface of wood post 1. Fungi resistance of functional oligodenes. Hvojnye Boreal’noj Zony 2016, XXXIV, 117–120. (In Russian). Available online: https://elibrary.ru/item.asp?id=27541433 (accessed on 9 March 2018).
- Lazareva, S.V.; Smirnov, V.F.; Struchkova, I.V. Influence of the components of the culture medium on the phenoloxidase activity of the micromycetes Trichoderma viride and Trichderma Lignorum. Vestnik of Lobachevsky University of Nizhni Novgorod 2008, 1, 77–80. (In Russian). Available online: https://elibrary.ru/item.asp?id=10007939 (accessed on 9 March 2018).
- Kumar, K.; Amaresan, N.; Bhagat, S.; Madhuri, K.; Srivastava, R.C. Isolation and Characterization of Trichoderma spp. for Antagonistic Activity Against Root Rot and Foliar Pathogens. Indian J. Microbiol. 2012, 52, 137–144. [Google Scholar] [CrossRef] [PubMed]
- Skryabin, G.K.; Golovlev, E.L.; Klesov, A.A. Problems of Bioconversion of Plant Raw Materials; Nauka: Moscow, Russia, 1986; p. 292. (In Russian) [Google Scholar]
- Eremenko, A.M.; Petrik, I.S.; Smirnova, N.P.; Rudenko, A.V.; Marikvas, Y.S. Antibacterial and Antimycotic Activity of Cotton Fabrics, Impregnated with Silver and Binary Silver/Copper Nanoparticles. Nanoscale Res. Lett. 2016, 11, 28. [Google Scholar] [CrossRef] [PubMed]
- Kislitsyn, A.N. Extractive substances birch bark: Isolation, composition, properties, application. Himiya Drev. 1994, 3, 3–28. (In Russian) [Google Scholar]
- Zheng, C.; Li, D.; Ottenhall, A.; Ek, M. Cellulose fiber based fungal and water resistant insulation materials. Holzforschung 2017, 71, 1–7. [Google Scholar] [CrossRef]
- Dzubak, P.; Hajduch, M.; Vydra, D.; Hustova, A.; Kvasnica, M.; Biedermann, D.; Markova, L.; Urban, M.; Sarek, J. Pharmacological activities of natural triterpenoids and their therapeutic implications. Nat. Prod. Rep. 2006, 23, 394–411. [Google Scholar] [CrossRef] [PubMed]
- Kazakova, O.B.; Giniyatullina, G.V.; Yamansarov, E.Y.; Tolstikov, G.A. Betulin and ursolic acid synthetic derivatives as inhibitors of papilloma virus. Bioorg. Med. Chem. Lett. 2010, 20, 4088–4090. [Google Scholar] [CrossRef] [PubMed]
- Guo, M.; Li, W.; Zhang, Z.; Qiu, C.; Li, C.; Deng, G. Betulin suppresses S. aureus-induced mammary gland inflammatory injury by regulating PPAR-γ in mice. Int. Immunopharmacol. 2015, 29, 824–831. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.; Li, H.; Qui, J.; Feng, H. Betulin protects mice from bacterial pneumonia and acute lung injury. Microb. Pathog. 2014, 75, 21–28. [Google Scholar] [CrossRef] [PubMed]
- Huang, T.; Li, D.; Ek, M. Water repellency improvement of cellulosic textile fibers by betulin and a betulin-based copolymer. Cellulose 2018, 1–14. [Google Scholar] [CrossRef]
- Makarov, I.S.; Golova, L.K.; Kuznetsova, L.K.; Bondarenko, G.N.; Skvortsov, I.Yu.; Mironova, M.V.; Bermeshev, M.V. Composite fibers based on cellulose and tetraethoxysilane: Preparation, structure and properties. Fibre Chem. 2017, 49, 101–107. [Google Scholar] [CrossRef]
- Chulovskaya, S.A.; Garas'ko, E.V.; Kuz’min, S.M.; Semenov, YA.S.; Parfenyuk, V.I. Fungicidal properties of a cellulose-based composite material with zeolite additives. Khimija Rastitel’nogo Syr’ja 2012, 3, 49–53. (In Russian). Available online: https://elibrary.ru/item.asp?id=18807214 (accessed on 9 March 2018).
- Golova, L.K.; Borodina, O.E.; Kuznetsova, L.K.; Lyubova, T.A.; Krylova, T.B. The Solid-Phase MMO Process. Fibre Chem. 2000, 32, 243. [Google Scholar] [CrossRef]
- Rosenau, T.; Potthast, A.; Sixta, H.; Kosma, P. The chemistry of side reactions and byproduct formation in the system NMMO/cellulose (Lyocell process). Prog. Polym. Sci. 2001, 26, 1763–1837. [Google Scholar] [CrossRef]
- Plotnikova, E.P.; Golova, L.K.; Makarov, I.S.; Kulichikhin, V.G. Rheological properties of mixed solutions of cellulose and layered aluminosilicates in N-methylmorpholine-N-oxide. Polym. Sci. Ser. A 2013, 55, 258–267. [Google Scholar] [CrossRef]
- Kulichikhin, V.; Golova, L.; Makarov, I.; Bondarenko, G.; Makarova, V.; Ilyin, S.; Skvortsov, I.; Berkovich, A. Solutions of acrylonitrile copolymers in N-methylmorpholine-N-oxide: Structure, properties, fiber spinning. Eur. Polym. J. 2017, 92, 326–337. [Google Scholar] [CrossRef]
- Makarov, I.S.; Golova, L.K.; Kuznetsova, L.K.; Rebrov, A.V.; Berkovich, A.K.; Skvortsov, I.Yu.; Kulichikhin, V.G. Composite Fibers Based on Cellulose and Polyacrylonitrile Copolymers. Russ. J. Gen. Chem. 2017, 87, 1351–1356. [Google Scholar] [CrossRef]
- Wendler, F.; Meister, F.; Montigny, R.; Wagener, M. A New Antimicrobial ALCERU Fiber with Silver Nanoparticles. Fibers Text. East. Eur. 2007, 64, 41–45. [Google Scholar]
- Golova, L.K.; Romanov, V.V.; Balashova, O.B. Method for Producing a Solution for Spinning Fibers RF Patent 1645308, 1992. Available online: http://patents.su/3-1645308-sposob-polucheniya-rastvora-dlya-formovaniya-volokon.html (accessed on 9 March 2018).
- Egorov, N.S. Fundamentals of the doctrine of antibiotics. In Classical University Textbook, 6th ed.; Nauka: Moscow, Russia, 2004; p. 528. (In Russian) [Google Scholar]
- Mikhaylenko, M.A.; Shakhtshneyderb, T.P.; Brezgunova, M.E.; Drebuschak, V.A.; Kuznetsova, S.A.; Boldyrev, V.V. Obtaining and studying the physico-chemical properties of betulin solvates. Khimija Rastitel’nogo Syr’ja 2010, 2, 63–70. (In Russian) [Google Scholar]
- Makarov, I.S.; Golova, L.K.; Kuznetsova, L.K.; Antonov, S.V.; Kotsyuk, A.V.; Ignatenko, V.Ya.; Kulichikhin, V.G. Influence of Precipitation and Conditioning Baths on the Structure, Morphology, and Properties of Cellulose Films. Fibre Chem. 2016, 48, 298–305. [Google Scholar] [CrossRef]
- Blackburn, R. Biodegradable and Sustainable Fibres; CRC Press LLC: Boca Raton, FL, USA, 2005; p. 464. [Google Scholar]
- Firgoa, H.; Schuster, K.C.; Suchomel, F.; Männer, J.; Burrow, T.; Abu-Rous, M. The Functional Properties of TENCEL®—A Current Update. Lenzing. Berichte 2006, 85, 22–30. [Google Scholar]






| System | d, μm | σ 1, MPa | E 2, GPa | ε 3, % |
|---|---|---|---|---|
| 100% cellulose | 14–20 | 570–610 | 11–12 | 10–12 |
| 95% cellulose + 5% betulin | 11–15 | 440–520 | 14–20 | 6–8 |
| 90% cellulose + 10% betulin | 11–15 | 440–480 | 11–19 | 4–7 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Makarov, I.; Vinogradov, M.; Gromovykh, T.; Lutsenko, S.; Feldman, N.; Shambilova, G.; Sadykova, V. Antifungal Composite Fibers Based on Cellulose and Betulin. Fibers 2018, 6, 23. https://doi.org/10.3390/fib6020023
Makarov I, Vinogradov M, Gromovykh T, Lutsenko S, Feldman N, Shambilova G, Sadykova V. Antifungal Composite Fibers Based on Cellulose and Betulin. Fibers. 2018; 6(2):23. https://doi.org/10.3390/fib6020023
Chicago/Turabian StyleMakarov, Igor, Markel Vinogradov, Tatyana Gromovykh, Sergey Lutsenko, Nataliya Feldman, Gulbarshin Shambilova, and Vera Sadykova. 2018. "Antifungal Composite Fibers Based on Cellulose and Betulin" Fibers 6, no. 2: 23. https://doi.org/10.3390/fib6020023
APA StyleMakarov, I., Vinogradov, M., Gromovykh, T., Lutsenko, S., Feldman, N., Shambilova, G., & Sadykova, V. (2018). Antifungal Composite Fibers Based on Cellulose and Betulin. Fibers, 6(2), 23. https://doi.org/10.3390/fib6020023






