Surface Functionalization of Activated Carbon with Phosphonium Ionic Liquid for CO2 Adsorption
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Synthesis
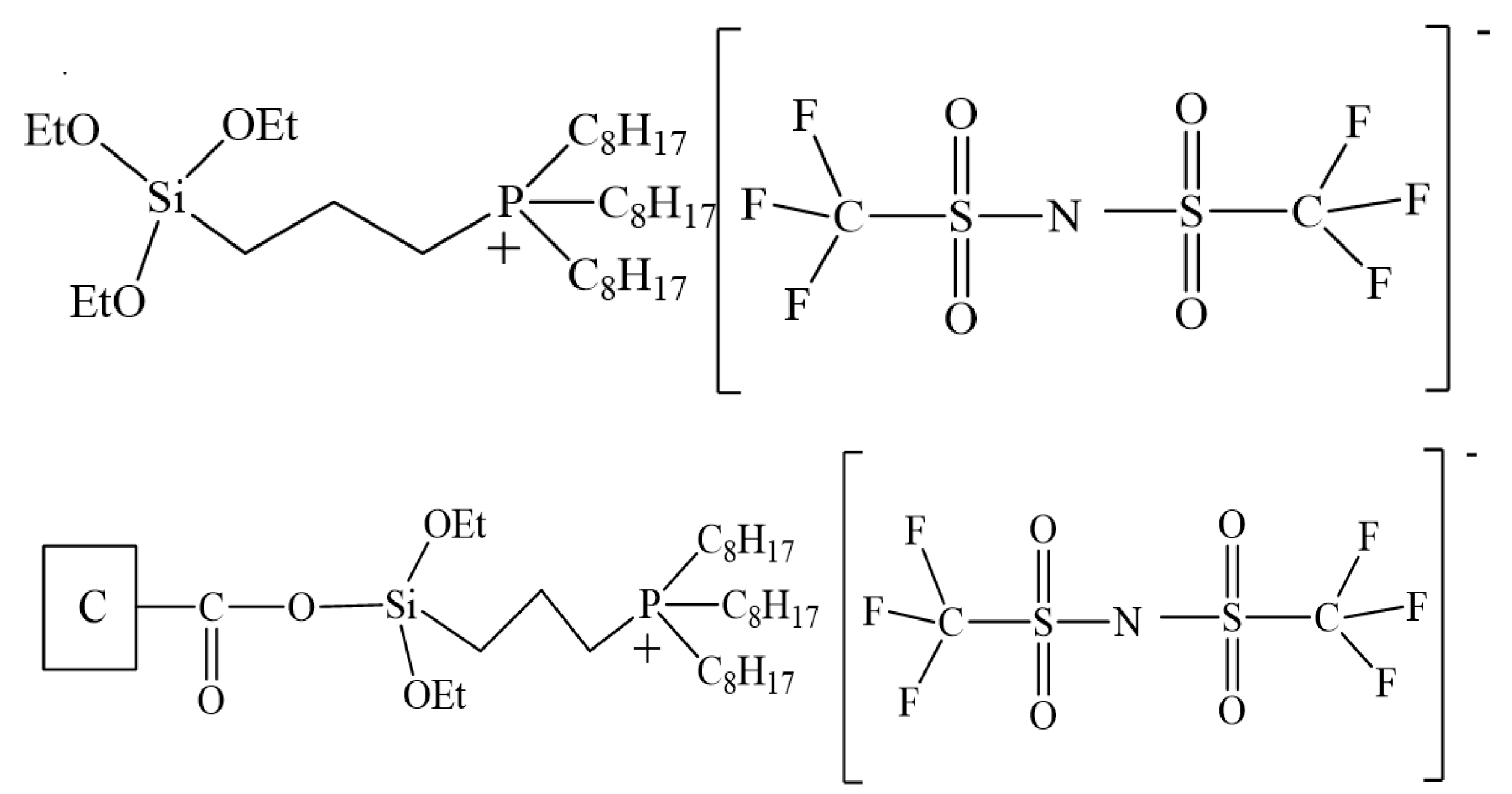
2.2.1. IL-Modified AC by Impregnated Method
2.2.2. IL-Modified AC by Grafting Method
2.3. Characterizations
2.3.1. Chemical Analysis
2.3.2. Evaluation of Pore Structure
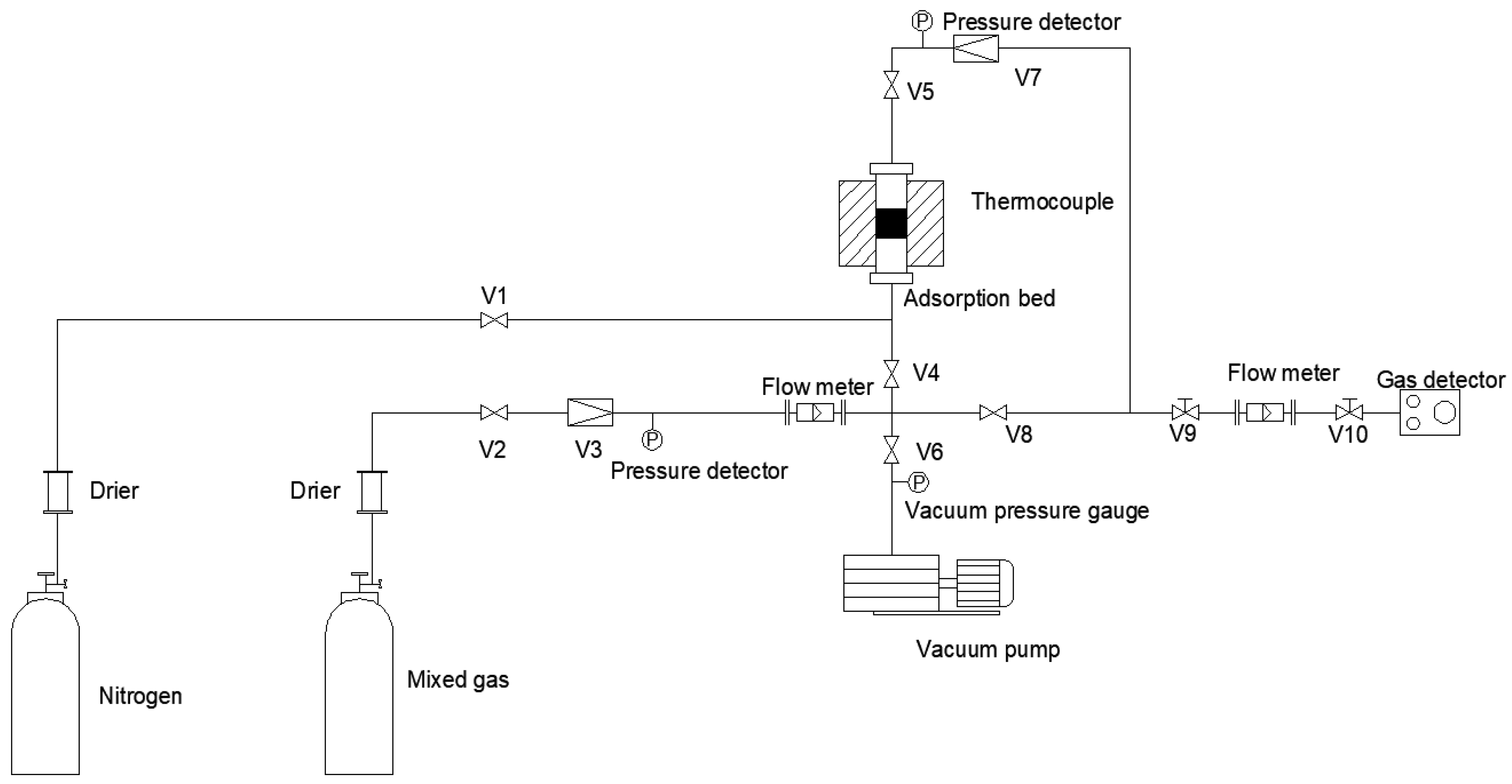
2.3.3. CO2 Adsorption Performance
3. Results and Discussion
3.1. Characterization
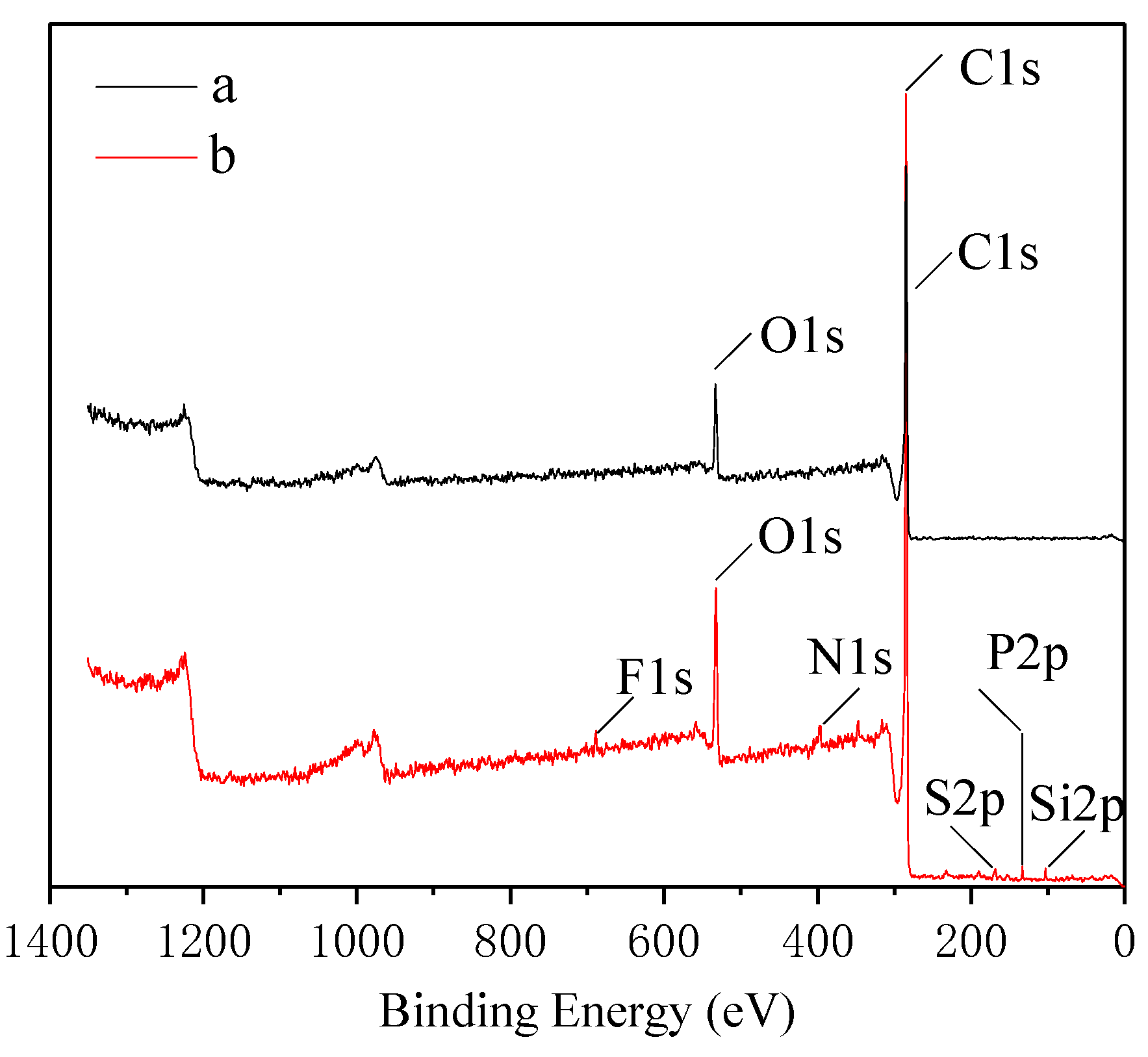
3.1.1. X-ray Photoelectron Spectroscopy Analysis
3.1.2. Thermogravimetric Analysis
3.1.3. Scanning Electron Microscope (SEM) Analysis
3.1.4. Evaluation of Pore Structure
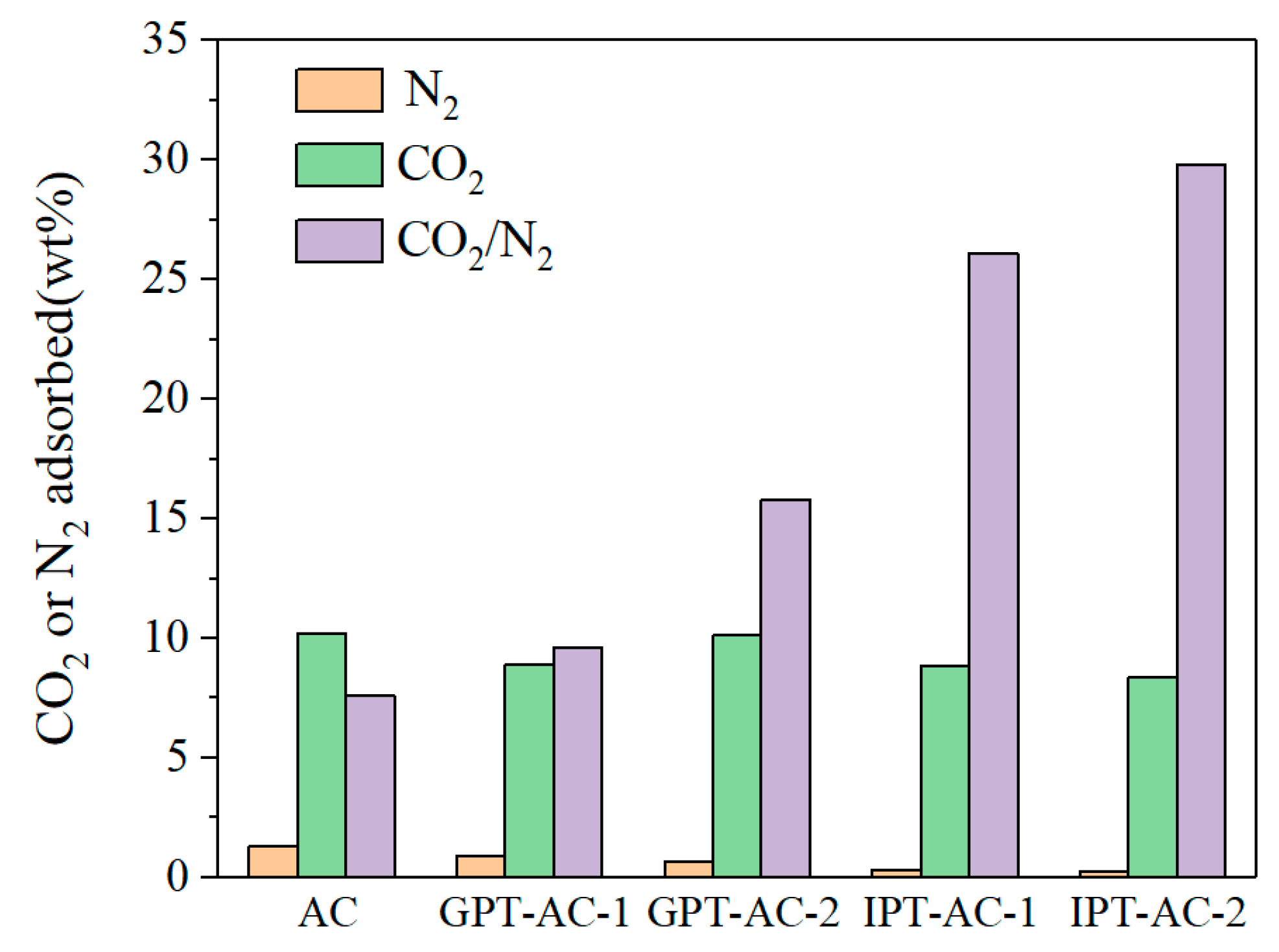
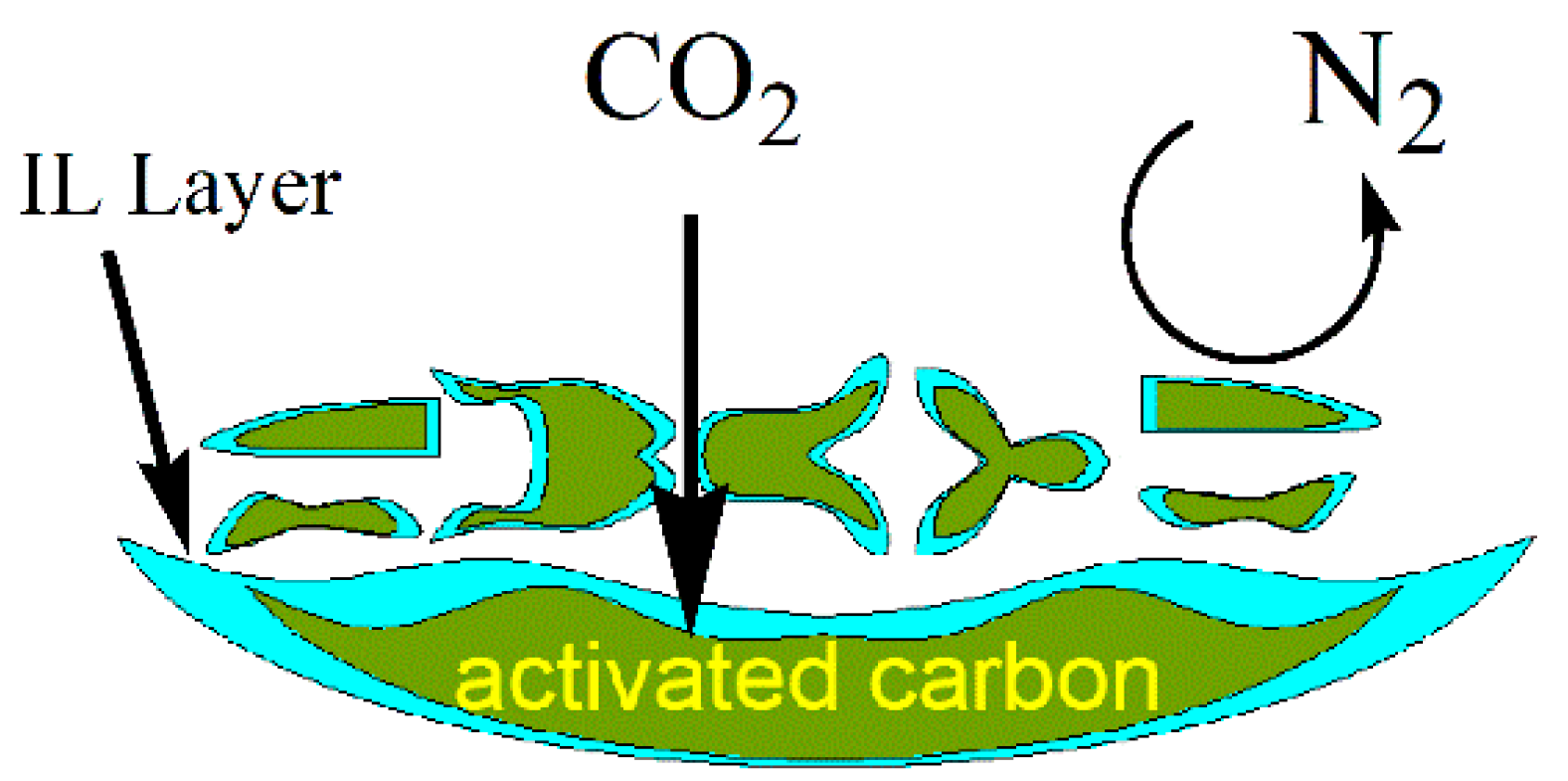
3.2. CO2/N2 Selectivity
3.3. Dynamic Adsorption Performance
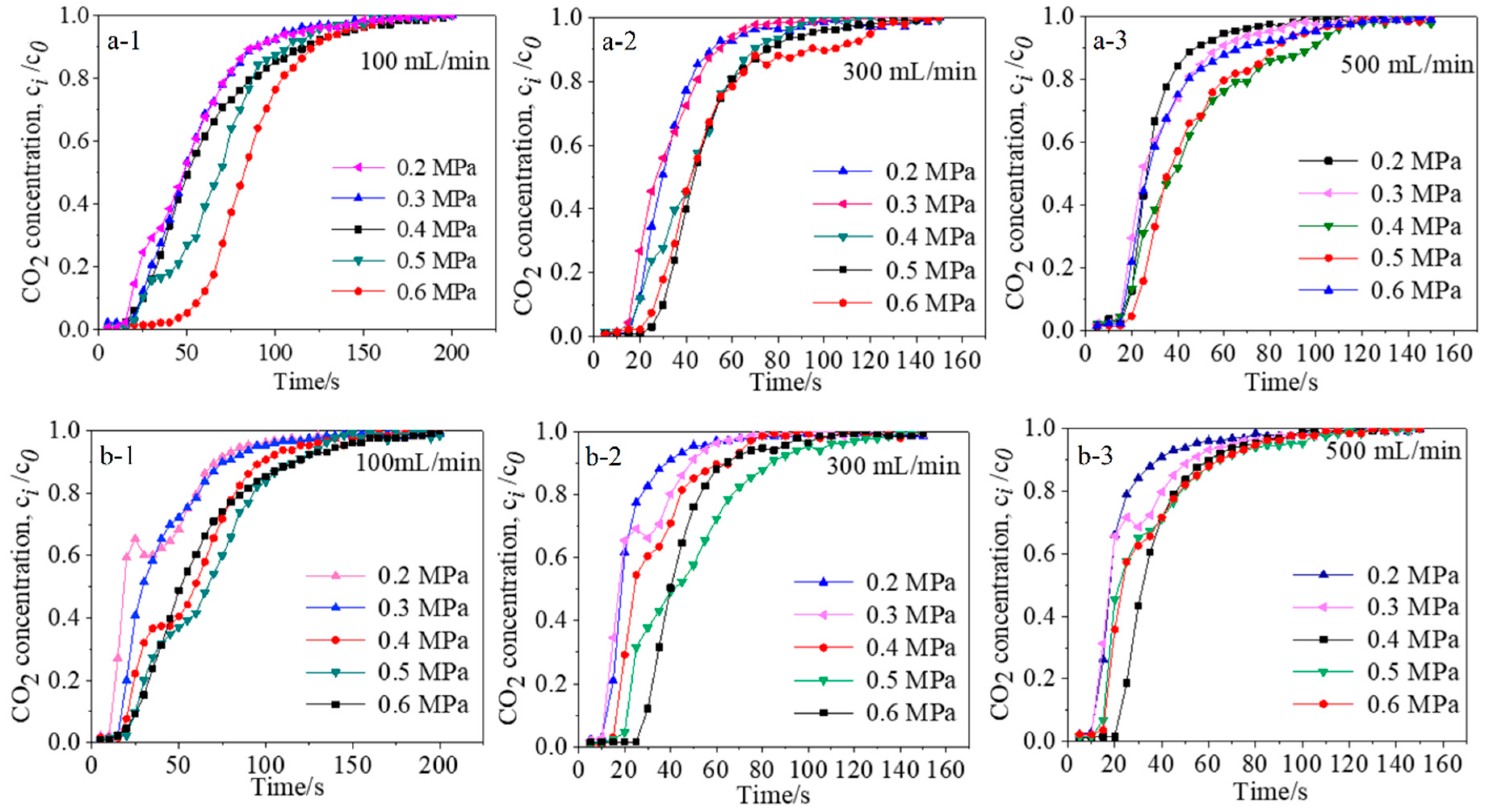
3.3.1. Effect of Flow Rate
3.3.2. Effect of Adsorption Pressure
3.3.3. Adsorption Capacity
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chaffee, A.L.; Knowles, G.P.; Liang, Z.; Zhang, J.; Xiao, P.; Webley, P.A. CO2 capture by adsorption: Materials and process development. Int. J. Greenh. Gas Control 2007, 1, 11–18. [Google Scholar] [CrossRef]
- Abbaspour Tamijani, A.; Ebrahimiaqda, E. Facet of rutile-structured GeO2: An ab initio investigation. Mol. Phys. 2017, 115, 1598–1605. [Google Scholar] [CrossRef]
- Corum, W.K.; Abbaspour Tamijani, A.; Mason, E.S. Density functional theory study of arsenate adsorption onto alumina surfaces. Minerals 2018, 8, 91. [Google Scholar] [CrossRef]
- Jones, C.W. CO2 capture from dilute gases as a component of modern global carbon management. Annu. Rev. Chem. Biomol. Eng. 2011, 2, 31–52. [Google Scholar] [CrossRef] [PubMed]
- El Gamal, M.; Mousa, H.A.; El-Naas, M.H.; Zacharia, R.; Judd, S. Bio-regeneration of activated carbon: A comprehensive review. Sep. Purif. Technol. 2018, 197, 345–359. [Google Scholar] [CrossRef]
- Pevida, C.; Plaza, M.G.; Arias, B.; Fermoso, J.; Rubiera, F.; Pis, J.J. Surface modification of activated carbons for CO2 capture. Appl. Surf. Sci. 2008, 254, 7165–7172. [Google Scholar] [CrossRef]
- Shafeeyan, M.S.; Daud, W.M.A.W.; Houshmand, A.; Shamiri, A. A review on surface modification of activated carbon for carbon dioxide adsorption. J. Anal. Appl. Pyrolysis 2010, 89, 143–151. [Google Scholar] [CrossRef]
- Rehman, A.; Park, M.; Park, S.J. Current progress on the surface chemical modification of carbonaceous materials. Coatings 2019, 9, 103. [Google Scholar] [CrossRef]
- Durán, I.; Álvarez-Gutiérrez, N.; Rubiera, F.; Pevida, C. Biogas purification by means of adsorption on pine sawdust-based activated carbon: Impact of water vapor. Chem. Eng. J. 2018, 353, 197–207. [Google Scholar] [CrossRef]
- Zhang, Y.; Ji, X.; Xie, Y.; Lu, X. Screening of conventional ionic liquids for carbon dioxide capture and separation. Appl. Energy 2016, 162, 1160–1170. [Google Scholar] [CrossRef]
- Aghaie, M.; Rezaei, N.; Zendehboudi, S. A systematic review on CO2 capture with ionic liquids: Current status and future prospects. Renew. Sustain. Energy Rev. 2018, 96, 502–525. [Google Scholar] [CrossRef]
- Zhang, X.; Zhang, X.; Dong, H.; Zhao, Z.; Zhang, S.; Huang, Y. Carbon capture with ionic liquids: Overview and progress. Energy Environ. Sci. 2012, 5, 6668–6681. [Google Scholar] [CrossRef]
- Hemp, S.T.; Zhang, M.; Allen, M.H., Jr.; Cheng, S.; Moore, R.B.; Long, T.E. Comparing ammonium and phosphonium polymerized ionic liquids: Thermal analysis, conductivity, and morphology. Macromol. Chem. Phys. 2013, 214, 2099–2107. [Google Scholar] [CrossRef]
- Ficke, L.E.; Novak, R.R.; Brennecke, J.F. Thermodynamic and thermophysical properties of ionic liquid + water systems. J. Chem. Eng. Data 2010, 55, 4946–4950. [Google Scholar] [CrossRef]
- Neves, C.M.S.S.; Carvalho, P.J.; Freire, M.G.; Coutinho, J.A.P. Thermophysical properties of pure and water-saturated tetradecyltrihexylphosphonium-based ionic liquids. J. Chem. Thermodyn. 2011, 43, 948–957. [Google Scholar] [CrossRef]
- Xie, W.; Ji, X.; Feng, X.; Lu, X. Mass-transfer rate enhancement for CO2 separation by ionic liquids: Theoretical study on the mechanism. AIChE J. 2015, 61, 4437–4444. [Google Scholar] [CrossRef]
- Ren, J.; Li, Z.; Chen, Y.; Yang, Z.; Lu, X. Supported ionic liquid sorbents for CO2 capture from simulated flue-gas. Chin. J. Chem. Eng. 2018, 26, 2377–2384. [Google Scholar] [CrossRef]
- Zulkurnai, N.Z.; Ali, U.M.; Ibrahim, N.; Abdul Manan, N.S. Carbon Dioxide (CO2) adsorption by activated carbon functionalized with deep eutectic solvent (DES). IOP Conf. Ser. Mater. Sci. Eng. 2017, 206, 012001. [Google Scholar] [CrossRef]
- D’Alessandro, D.M.; Smit, B.; Long, J.R. Carbon dioxide capture: Prospects for new materials. Angew. Chem. Int. Ed. 2010, 49, 6058–6082. [Google Scholar] [CrossRef]
- Lin, K.Y.A.; Park, A.H.A. Effects of bonding types and functional groups on CO2 capture using novel multiphase systems of liquid-like nanoparticle organic hybrid materials. Environ. Sci. Technol. 2011, 45, 6633–6639. [Google Scholar] [CrossRef] [PubMed]
- Kinik, F.P.; Altintas, C.; Balci, V.; Koyuturk, B.; Uzun, A.; Keskin, S. [BMIM][PF6] incorporation doubles CO2 selectivity of ZIF-8: Elucidation of interactions and their consequences on performance. ACS Appl. Mater. Interfaces 2016, 8, 30992–31005. [Google Scholar] [CrossRef]
- Ruckart, K.N.; O’Brien, R.A.; Woodard, S.M.; West, K.N.; Glover, T.G. Porous solids impregnated with task-specific ionic liquids as composite sorbents. J. Phys. Chem. C 2015, 119, 20681–20697. [Google Scholar] [CrossRef]
- Erto, A.; Silvestre-Albero, A.; Silvestre-Albero, J.; Rodríguez-Reinoso, F.; Balsamo, M.; Lancia, A.; Montagnaro, F. Carbon-supported ionic liquids as innovative adsorbents for CO2 separation from synthetic flue-gas. J. Colloid Interface Sci. 2015, 448, 41–50. [Google Scholar] [CrossRef]
- Zhu, J.; Xin, F.; Huang, J.; Dong, X.; Liu, H. Adsorption and diffusivity of CO2 in phosphonium ionic liquid modified silica. Chem. Eng. J. 2014, 246, 79–87. [Google Scholar] [CrossRef]
- El-Hendawy, A.N.A. Influence of HNO3 oxidation on the structure and adsorptive properties of corncob-based activated carbon. Carbon 2003, 41, 713–722. [Google Scholar] [CrossRef]
- Green, M.D.; Salas-de la Cruz, D.; Ye, Y.; Layman, J.M.; Elabd, Y.A.; Winey, K.I.; Long, T.E. Alkyl-substituted N-vinylimidazolium polymerized ionic liquids: Thermal properties and ionic conductivities. Macromol. Chem. Phys. 2011, 212, 2522–2528. [Google Scholar] [CrossRef]
- Liu, H.; Huang, J.; Pendleton, P. Experimental and modelling study of CO2 absorption in ionic liquids containing Zn (II) ions. Energy Procedia 2011, 4, 59–66. [Google Scholar] [CrossRef]
- Li, G.; Wang, Q.; Jiang, T.; Luo, J.; Rao, M.; Peng, Z. Roll-up effect of sulfur dioxide adsorption on zeolites FAU 13X and LTA 5A. Adsorption 2017, 23, 699–710. [Google Scholar] [CrossRef]
- Kaur, B.; Gupta, R.K.; Bhunia, H. Chemically activated nanoporous carbon adsorbents from waste plastic for CO2 capture: Breakthrough adsorption study. Microporous Mesoporous Mater. 2019, 282, 146–158. [Google Scholar] [CrossRef]
- Bernard, F.L.; Duczinski, R.B.; Rojas, M.F.; Fialho, M.C.C.; Carreño, L.Á.; Chaban, V.V.; Vecchia, F.D.; Einloft, S. Cellulose based poly(ionic liquids): Tuning cation-anion interaction to improve carbon dioxide sorption. Fuel 2018, 211, 76–86. [Google Scholar] [CrossRef]
- Huang, G.G.; Liu, Y.F.; Wu, X.X.; Cai, J.J. Activated carbons prepared by the KOH activation of a hydrochar from garlic peel and their CO2 adsorption performance. New Carbon Mater. 2019, 34, 247–257. [Google Scholar] [CrossRef]
- Wu, F.C.; Wu, P.H.; Tseng, R.L.; Juang, R.S. Description of gas adsorption isotherms on activated carbons with heterogeneous micropores using the Dubinin–Astakhov equation. J. Taiwan Inst. Chem. Eng. 2014, 45, 1757–1763. [Google Scholar] [CrossRef]
- Gibson, J.A.A.; Gromov, A.V.; Brandani, S.; Campbell, E.E.B. The effect of pore structure on the CO2 adsorption efficiency of polyamine impregnated porous carbons. Microporous Mesoporous Mater. 2015, 208, 129–139. [Google Scholar] [CrossRef]
- Lively, R.P.; Leta, D.P.; DeRites, B.A.; Chance, R.R.; Koros, W.J. Hollow fiber adsorbents for CO2 capture: Kinetic sorption performance. Chem. Eng. J. 2011, 171, 801–810. [Google Scholar] [CrossRef]
- Guo, Y.; Zhao, C.; Li, C.; Wu, Y. CO2 sorption and reaction kinetic performance of K2CO3/AC in low temperature and CO2 concentration. Chem. Eng. J. 2015, 260, 596–604. [Google Scholar] [CrossRef]
- Álvarez-Gutiérrez, N.; Gil, M.V.; Rubiera, F.; Pevida, C. Kinetics of CO2 adsorption on cherry stone-based carbons in CO2/CH4 separations. Chem. Eng. J. 2017, 307, 249–257. [Google Scholar] [CrossRef]
- Guo, Y.; Tan, C.; Wang, P.; Sun, J.; Yan, J.; Li, W.; Zhao, C.; Lu, P. Kinetic study on CO2 adsorption behaviors of amine-modified co-firing fly ash. J. Taiwan Inst. Chem. Eng. 2019, 96, 374–381. [Google Scholar] [CrossRef]



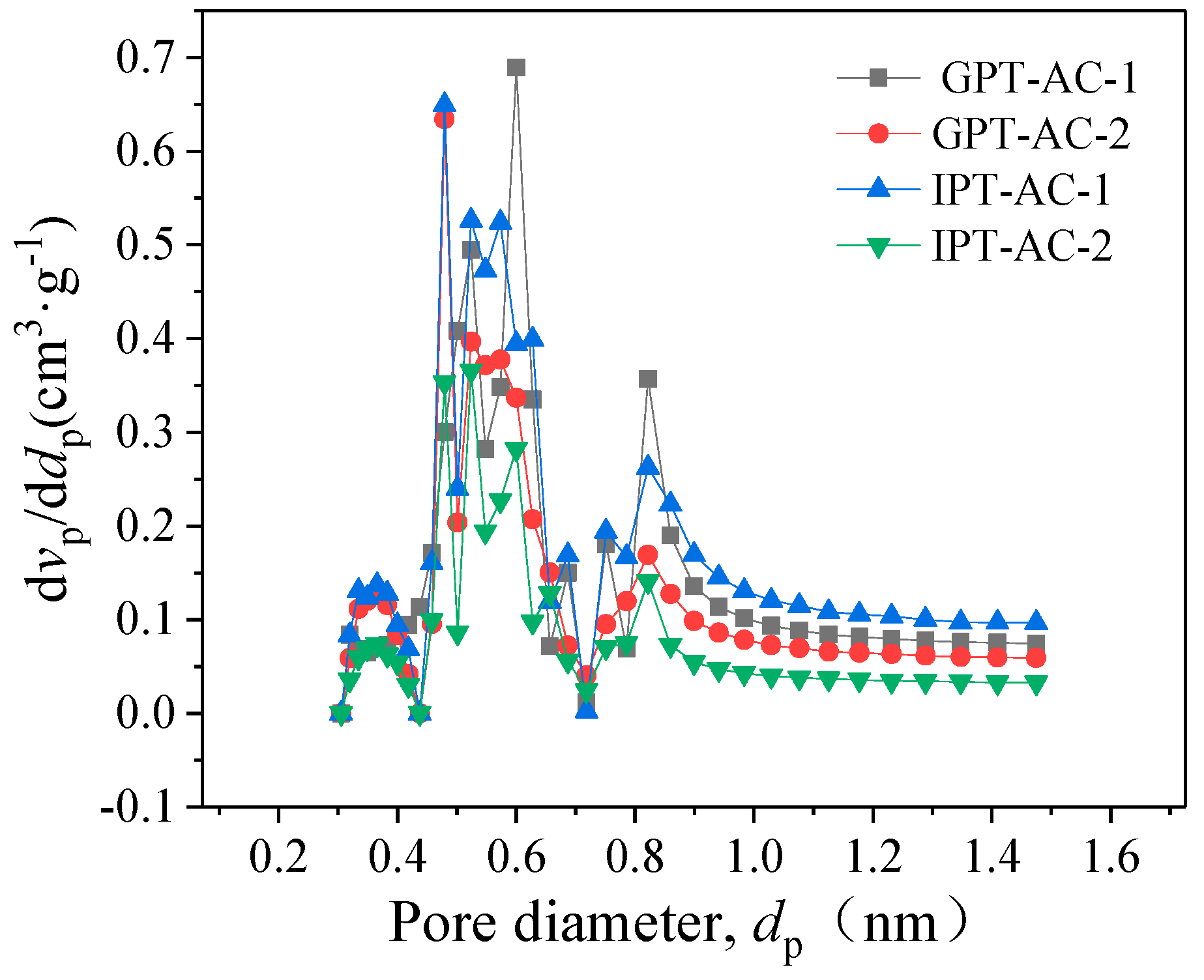

| Sample | Distribution of Pore Volume with Aperture-Width | CPV | CSA | |
|---|---|---|---|---|
| 0–2 (nm) | 2–24 (nm) | (cm3·g−1) | (m2·g−1) | |
| GPT-AC-1 | 72.775% | 27.225% | 0.318 | 502.0 |
| GPT-AC-2 | 70.574% | 29.426% | 0.334 | 549.6 |
| IPT-AC-1 | 65.825% | 34.175% | 0.263 | 431.3 |
| IPT-AC-2 | 48.078% | 51.922% | 0.191 | 277.6 |
| Pressure (MPa) | GPT-AC-2 Adsorption (mmol·g−1) | IPT-AC-1 Adsorption (mmol·g−1) | GPT-AC-2 Adsorption (mmol·g−1) | IPT-AC-1 Adsorption (mmol·g−1) | GPT-AC-2 Adsorption (mmol·g−1) | IPT-AC-1 Adsorption (mmol·g−1) |
|---|---|---|---|---|---|---|
| 100 mL/min | 300 mL/min | 500 mL/min | ||||
| 0.2 | 1.105 | 0.872 | 0.843 | 0.731 | 0.748 | 0.681 |
| 0.3 | 1.134 | 0.893 | 0.877 | 0.752 | 0.824 | 0.723 |
| 0.4 | 1.278 | 1.181 | 1.157 | 0.815 | 1.112 | 0.925 |
| 0.5 | 1.383 | 1.323 | 1.268 | 1.106 | 1.221 | 0.839 |
| 0.6 | 1.746 | 1.240 | 1.335 | 1.133 | 0.856 | 0.832 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
He, X.; Zhu, J.; Wang, H.; Zhou, M.; Zhang, S. Surface Functionalization of Activated Carbon with Phosphonium Ionic Liquid for CO2 Adsorption. Coatings 2019, 9, 590. https://doi.org/10.3390/coatings9090590
He X, Zhu J, Wang H, Zhou M, Zhang S. Surface Functionalization of Activated Carbon with Phosphonium Ionic Liquid for CO2 Adsorption. Coatings. 2019; 9(9):590. https://doi.org/10.3390/coatings9090590
Chicago/Turabian StyleHe, Xiaodong, Jiamei Zhu, Hongmin Wang, Min Zhou, and Shuangquan Zhang. 2019. "Surface Functionalization of Activated Carbon with Phosphonium Ionic Liquid for CO2 Adsorption" Coatings 9, no. 9: 590. https://doi.org/10.3390/coatings9090590




