1. Introduction
In recent years, lack of vitamin D
3 in humans worldwide has become a real problem. In fact, it is estimated that approximately one billion people suffer from vitamin D
3 deficiency due to less photosynthesis with certain skin types, aging, and scarce exposure to sunlight [
1]. Vitamin D
3 plays a major role in developing and maintaining the ossified skeleton by stabilizing the blood calcium level within the normal range [
2]. Moreover, vitamin D
3 deficiency can cause several diseases such as cancer, diabetes, hypertension metabolic syndrome, and other autoimmune and infectious diseases [
3].
Vitamin D
3 is synthesized in the human body through exposure to sunlight (specifically, ultraviolet B (UVB) radiation, 290–315 nm) and dietary intake. The daily recommended dose of vitamin D
3 is 10 μg; however, considering that egg yolk has 10 μg of vitamin D
3 per 100 g and fish oil has 5–10 μg per 100 g, this dose cannot realistically be met via synthesis through food [
4]. The most effective method to meet this dose is vitamin D
3 synthesis from UVB irradiation. However, UVB exposure risks DNA damage that is a major factor in the development of skin cancer [
5]. Furthermore, vitamin D
3 deficiencies could also be caused by low UVB exposure during winter and the lack of sunshine in certain areas with polar nights [
6]. Thus, although vitamin D
3 is mainly obtained through UVB exposure, this approach has some practical problems.
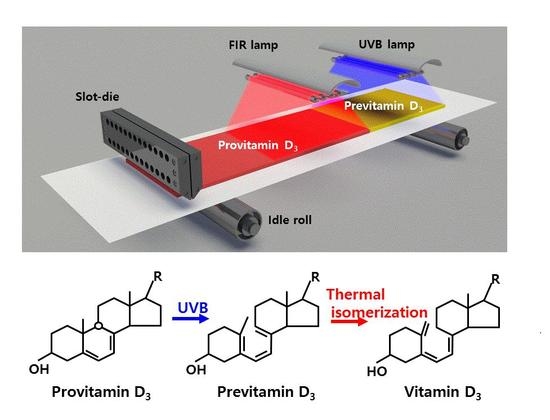
In this study, we introduce a novel previtamin D
3 patch containing 7-dehydrocholesterol (7-DHC) that produces vitamin D
3 under UVB exposure [
7]. We also introduce a new approach for mass-producing such patches through a roll-to-roll (R2R) process. Conventional advanced R2R processes [
8] are used for producing electrical devices such as transistors, sensors, and solar cells using materials such as conductors, semiconductors, and insulators. To the best of our knowledge, no study has applied R2R processes to the production of biomaterials. The coating and post-treatment approaches proposed in this study for composite films of a polymer and vitamin D
3 materials will open new prospects in the fields of bio- and green-manufacturing.
2. Results and Discussion
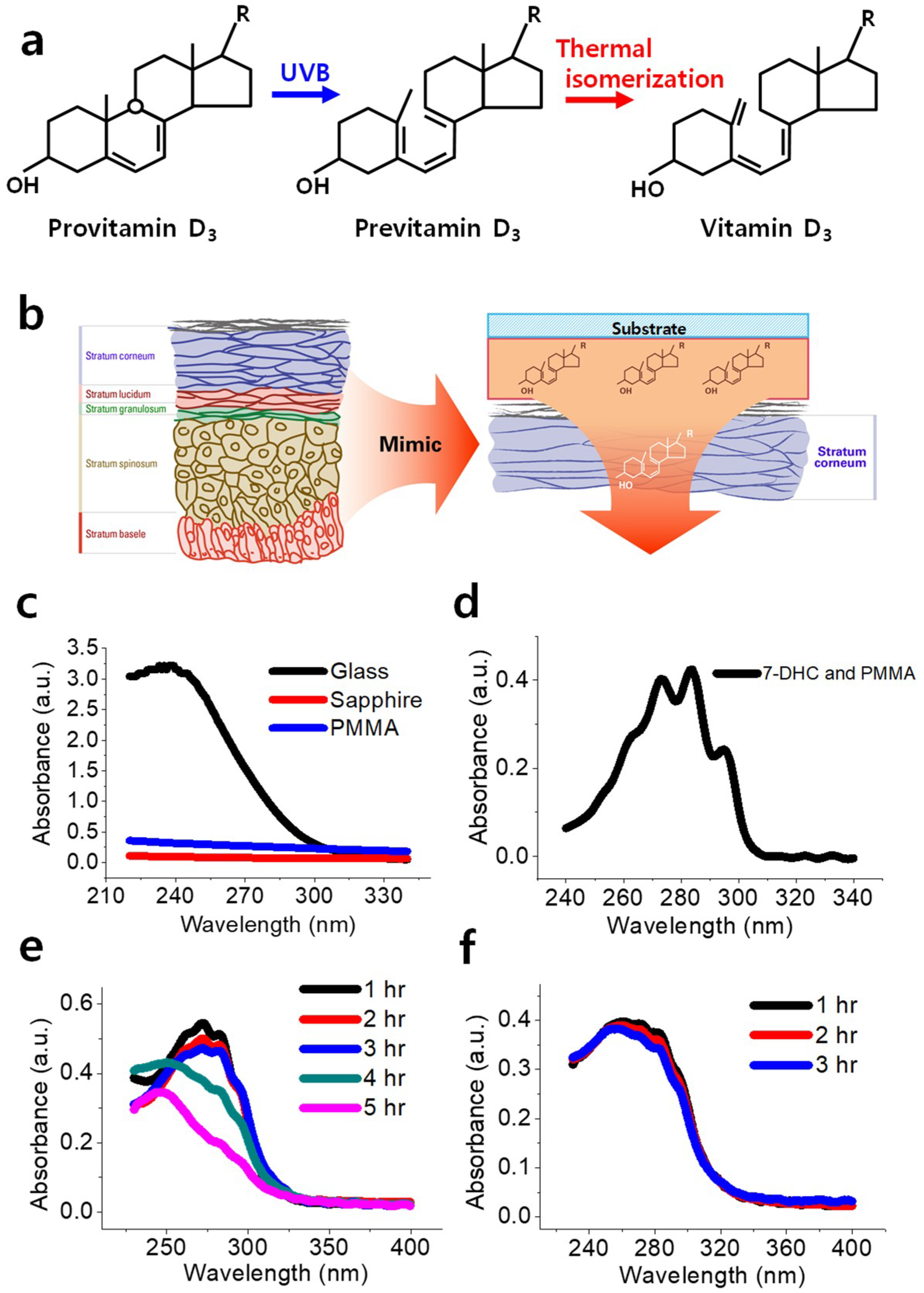
The vitamin D
3 formation mechanism is based on the transformation of D-dosimeters [
9]. A D-dosimeter is an in vitro model of vitamin D that measures the photodamage of DNA molecules following exposure to UVB radiation in the UV spectrum. 7-DHC, the provitamin D
3 used in this study, absorbs UVB radiation and converts to previtamin D
3 via the opening of its hexadiene ring, as shown in
Figure 1a. Then, the obtained previtamin D
3 converts to vitamin D
3 via thermal isomerization with an intramolecular H shift. Nevertheless, Jacobs and Havinnga reported side photoconversion of three isomers from previtamin D
3: lumisterol, toxysterol and tachysterol [
10]. These conversions can be seen in the UV absorption spectra [
11].
Human skin comprises an inner layer (dermis) and an outer layer (epidermis). The epidermis consists of five strata, as shown in
Figure 1b: the stratum corneum, stratum lucidum, stratum granulosum, stratum spinosum, and stratum basele [
12]. The two innermost strata, namely, the stratum spinosum and stratum basele, are known to contain a high concentration of 7-DHC.
In this study, we mimic the biomechanism of the epidermis by developing a polymer-mixed previtamin D
3 layer on a flexible polyethylene terephthalate (PET) substrate that can serve as a vitamin D
3 patch under UVB irradiation (
Figure 1b). We call this product V-Skin. Then, the coated 7-DHC and polymer layer on the PET substrate was exposed to UVB radiation in the laboratory. V-Skin has a previtamin D
3 layer on the PET substrate, which is the preceding dosimeter of vitamin D
3. V-Skin can be attached to the human body and can produce vitamin D
3 via thermal isomerization induced by body heat (
Figure 1b).
Figure 1c shows the absorbance spectra of sapphire, PMMA-coated sapphire, and glass. Sapphire glass was used as the base substrate owing to its clear absorbance spectra at UV wavelengths. In contrast, the glass substrate showed a relatively high absorbance rate in the desired ranges, making it unsuitable for use as a base substrate. Further, the PMMA-coated sapphire substrate showed an increase in absorbance rate up to 0.359, however, it did not show significant peak changes in the wavelength range of 220–340 nm. Finally, the coated layer with 7-DHC and PMMA showed clear peaks at 261, 273, 283, and 295 nm that corresponded to 7-DHC, as shown in
Figure 1d. Galkin and Terenetskaya reported the shift and change in absorbance peaks from 7-DHC to previtamin D
3 [
9].
Figure 1e shows the transformation of the absorption spectrum of 7-DHC under UVB irradiation for a few hours. The four peaks in the absorbance spectra shifted from 270 to 250 nm and gradually disappeared after 4–5 h of UVB exposure; this verified the presence of previtamin D
3 and was consistent with the plateau changes reported by Galkin and Terenetskaya [
9]. Lastly, the obtained previtamin D
3 was treated at 40 °C for 1–3 h, as shown in
Figure 1f. However, no specific change was seen. This suggested that the optical measurement does not confirm vitamin D
3 production as reported in previous studies [
9,
13].
The previtamin D
3 film can be upscaled by an R2R manufacturing process [
14]. The R2R system comprises numerous rollers linked by a flexible substrate (web) [
15].
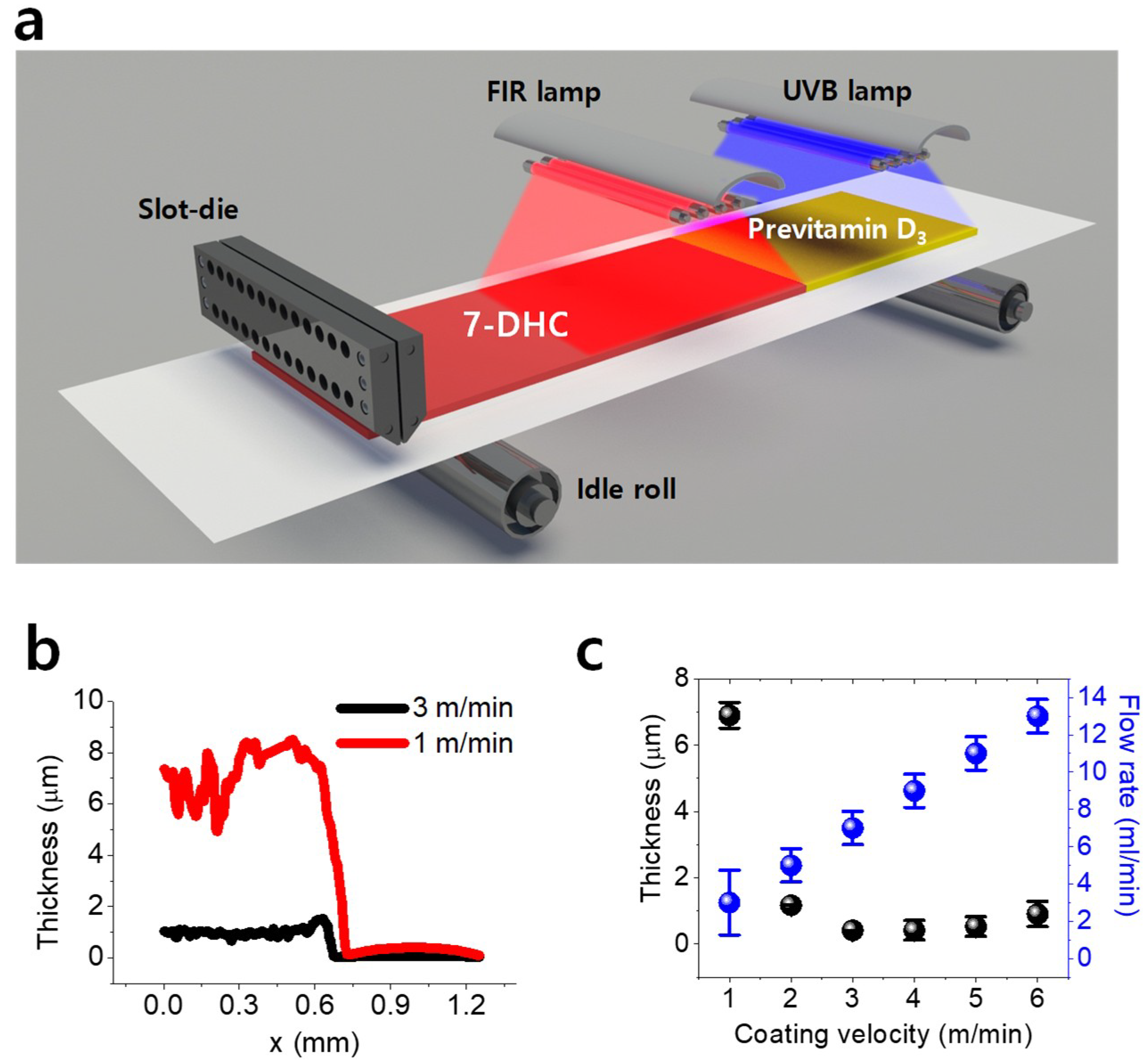
Figure 2a shows the proposed fabrication scheme that consists of the following three steps: R2R slot-die coating of synthesized solution, far-infrared (FIR) drying for solvent evaporation, and UVB irradiation for converting 7-DHC to previtamin D
3. UVB irradiation was conducted offline using a UVB lamp owing to equipment limitations. The R2R system (SND Corporation Co., Ltd., Incheon, Korea) used in the study included an unwinder, outfeeder, offset pivot guider, dancer, load cell (L/C), FIR dryer, accumulator, proportional-integral-derivative (PID) controller, outfeeder, and rewinder, as shown in
Figure S1.
The process conditions of slot-die coating can be used to control the thickness of the coated layer and thereby increase the yield rate of previtamin D
3. The dominant parameters affecting the thickness are known to be the liquid viscosity, liquid surface tension, liquid solid content, coating velocity, flow rate, and coating gap [
16]. In this experiment, the capillary number is higher than 0.02 owing to the use of PMMA with an 18% concentration in the coating solution; therefore, the coating window is inappropriate for the use of the viscocapillary model [
17]. Moreover, the minimum thickness can be obtained only by using a vacuum box in the system, and the general large-area coating machine does not have a vacuum box. Therefore, the wet thickness is always greater than the estimated thickness. To obtain a predictable thickness when using a high-viscosity solution without a vacuum box, the thickness can be predicted based on the mass conservation law, as shown in Equation (1) [
18,
19]:
Here,
tdry,
fr,
n,
w,
V, ρ
l, and ρ
s denote the dry thickness of a coated layer, flow rate, number of coating strips, width of a coating strip, coating velocity, density of the solid in a liquid, and density of the solid in a dried film, respectively. The coating velocity and flow rate were selected as process parameters in this study.
Table S1 summarizes the operation conditions of the R2R slot-die coating process. The estimated thickness was in the range of 2–7 μm, and it decreased with increasing speed, as shown in
Figure S1. The flow rate conditions for a coating width of 130 mm affected the thickness reduction at high speed.
Figure 2b shows the measured thickness of the coated layer under coating velocities of 1 and 3 m/min. The corresponding obtained coated layer thicknesses were 6.9 and 0.4 μm, respectively. The surface morphology of the coated layer with coating velocity of 3 m/min was better than that with one of 1 m/min. Generally, a uniform coated surface is obtained at high speed [
20]. Furthermore, all experimental results (
Figure 2c) indicated that the coated layer thickness decreased with increasing coating speed; this agrees with the estimated result shown in
Figure S2. However, unexpectedly, the thickness variation at low speeds remained stable because the meniscus was stable in a high-viscosity liquid [
21].
The previously tested sapphire glass is rigid; therefore, it was replaced with a PET film. However, when PMMA and 7-DHC were coated on the PET film, the absorbance of the UV region varied greatly, as shown in
Figure 3a. This is due to the high absorbance of the PET film at UV wavelengths (see
Figure S3) [
22]. When the absorbance is measured, scattering occurs in the PET film when UV light passes from the dispenser to the detector. In the R2R-based experiment, all conditions were set the same as those in the previous lab-scale experiment, and the amount of vitamin D
3 on the films was quantified using high-performance liquid chromatography (HPLC) measurements [
23]. A window with dimension of 120 mm × 30 mm was cut in the rewound roll to allow UVB irradiation of the 7-DHC and PMMA film. The samples were exposed under a UVB lamp in a darkroom for 5 h, which is the optimal condition shown in
Figure 1e. After exposure, the samples were heated at 40 °C for 2 h following the conditions recommended by Galkin and Ternetskaya [
9]. The coated film should be in the liquid state for HPLC measurement; therefore, the PMMA film was immersed in toluene to separate it from the PET substrate and then dissolved by stirring. Finally, commercial oral vitamin D
3 (Solar D liquid 400, 0.278 mg/mL, Viva Pharmaceutical Inc., Richmond, BC, Canada) liquid was used as a standard calibration curve for the peak area as a concentration of the standard sample, as shown in
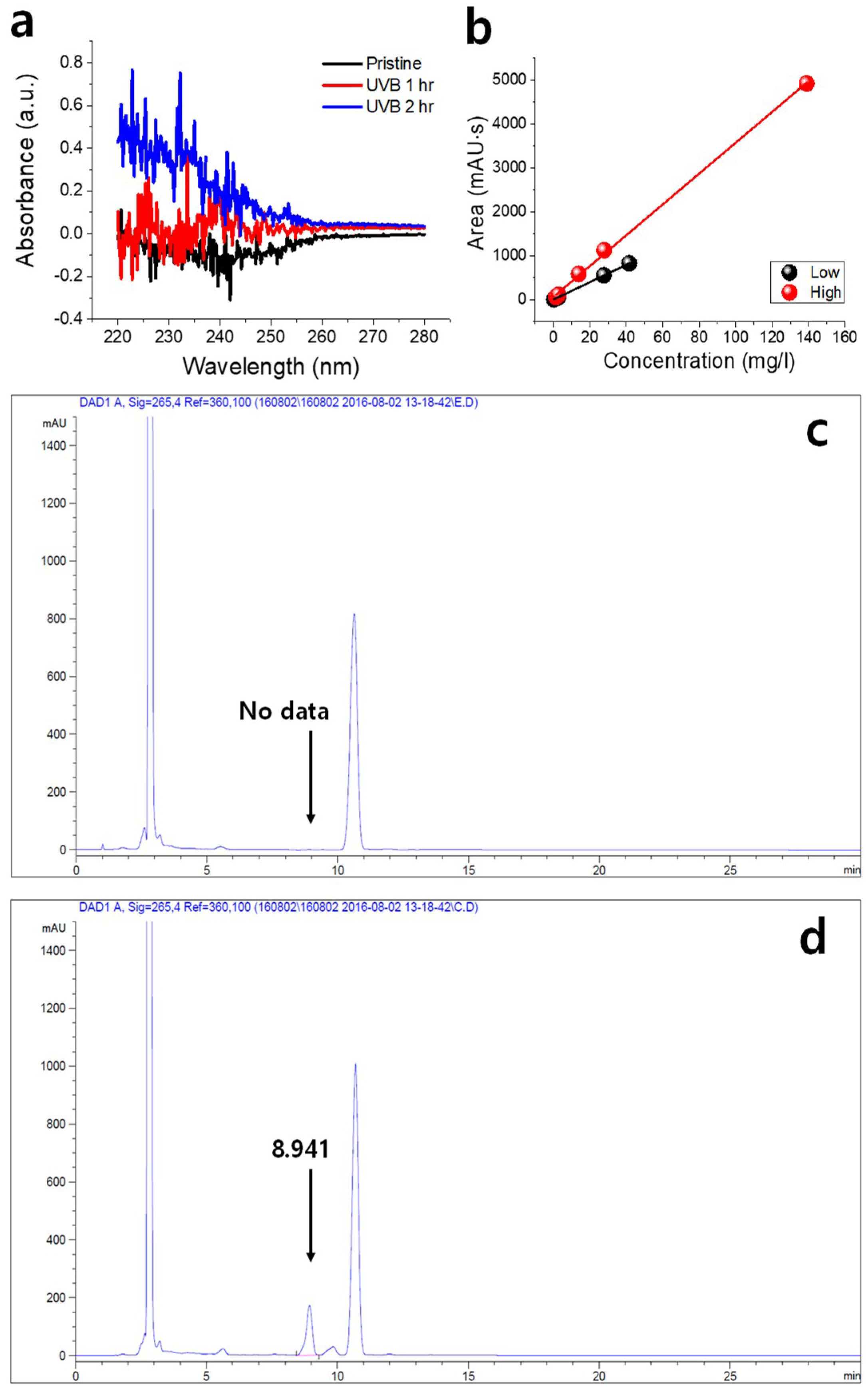
Figure 3b (see
Figure S4 and Table S2 for a photograph of the prepared samples and HPLC of commercial vitamin D
3, respectively). The calibration curves were obtained for each experiment. “Low” was measured for low-thickness samples (thickness: 0.7 μm) and “High,” for high-thickness samples (thickness: 6.9 μm). Further, the dilution rate was doubled for high-thickness samples. The equations of the fitted curves for the “High” and “Low” sets are
y = 35.0675
x + 65.7071 and
y = 19.8042
x + 3.6455 (
y: area,
x: concentration), respectively.
Figures S5a and S6a show the HPLC data of the standard sample in each experiment. As expected, the peak of the standard sample did not appear in the HPLC result of the coating film without UVB exposure (
Figure 3c).
Table 1 lists the test report of HPLC measurement for samples with thicknesses of 6.9 and 0.7 μm. UVB-irradiated samples corresponded to retention times of 8.9 and 6 min for each standard sample. The variation of the peak areas is due to the thickness difference and sheet area difference during sampling. The difference in retention time was due to the difference in dilution rate between the samples measured on different days [
24].
Figure 3d shows the measured HPLC data with retention time of 8.941 min and area of 2795.98 mAU·s, which is the representative sample with the highest peak (Sample 2). As a result, it can be seen that vitamins are formed after UVB exposure; this is confirmed by detection in the standard sample with the same retention time. On the contrary, small peaks were detected in samples with low thickness. However, the vitamin D
3 observed in the film may be part of the vitamin D
3 converted from previtamin D
3. This is because the complete coated 7-DHC and PMMA films may not have been subjected to the UVB irradiation or heating processes.
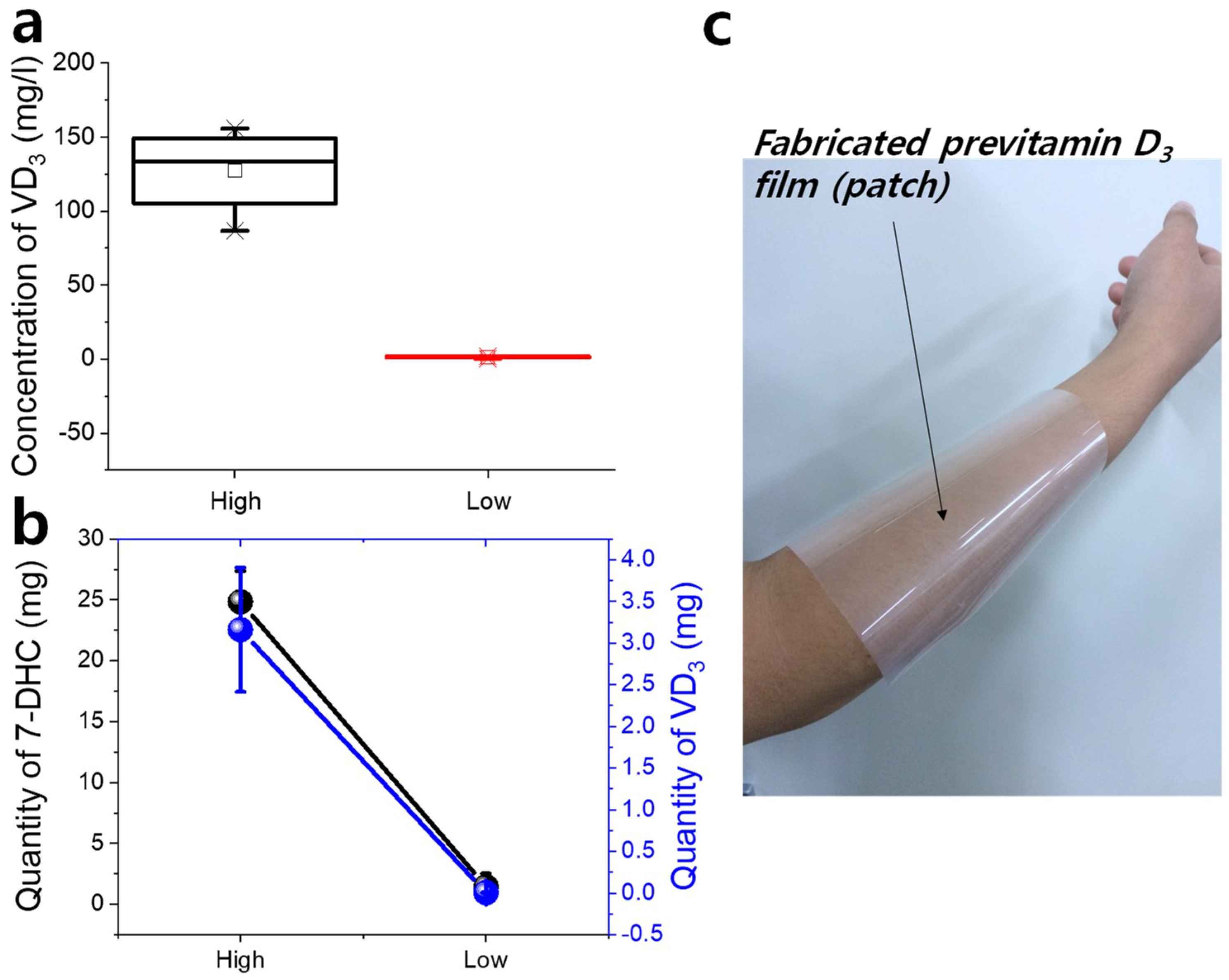
The actual vitamin D
3 concentration in the samples can be obtained, as shown in
Figure 4a, by using the calibration curves shown in
Figure 3b. The vitamin D
3 concentrations were obtained as 127 ± 30 and 1.42 ± 0.7 mg/L at thicknesses of 6.9 and 0.7 μm, respectively. This value is ~0.5–43% that of oral vitamin D
3. As mentioned earlier, this is not a low amount because vitamin D
3 formed from 7-DHC is more effective than that obtained through oral nutrients.
Figure 4b shows the amount of vitamin D
3 that was generated from the 7-DHC film. The previously prepared sample had area of 120 mm × 30 mm and multiplying it by the coated thickness gives the volume of each sample. The obtained amount of vitamin D
3 in high- and low-thickness samples was 3.16 ± 0.746 mg and 3 ± 2 μg, respectively. This value was obtained from 7-DHC amounts of 24.8 ± 1.44 and 2.52 ± 1.08 mg, respectively, contained in the specific volume before UVB irradiation. The rate of conversion of 7-DHC to vitamin D
3 was 12.7% in a thick sample; this is much higher than the rate of 0.24% seen in a low-thickness sample. The low rate in the low-thickness samples is attributed to environmental factors such as air and moisture acting on the outer layer.
4. Materials and Method
The coating solution was synthesized by mixing 1 wt.% 7-DHC [C27H44O, Sigma-Aldrich Co., LLC., Saint Louis, MO, USA], 18 wt.% PMMA [[CH2C(CH3)(CO2CH3)]n, average Mw: ~120,000, Sigma-Aldrich Co., LLC.], 8.1 wt.% ethanol [CH3CH2OH, Sigma-Aldrich Co., LLC.], and 72.9 wt.% acetone [CH3COCH3, Sigma-Aldrich Co., LLC.]. For lab-scale validation, the coating solution was coated on a sapphire substrate with increasing spinning velocity up to 3000 rpm by using a commercial spin coater (ACE-2000, Dong Ah Trade Corp Co., Ltd., Seoul, Korea). The coated layer was dried at 60 °C for 30 min in a vacuum oven. Subsequently, a piece of the coated substrate was irradiated by a UVB lamp (TL T12 40W/01 RS UVB, Philips Inc., Amsterdam, The Netherlands) in a darkroom. The absorbance of the coated layer was measured using a UV/Vis spectrophotometer (Optizen 2120 UV, Mecasys Co., Ltd., Daejeon, Korea). In the R2R experiment, a slot-die coater (Naraenanotech Co., Ltd., Yongin, Korea) was used to obtain coating width of up to 130 mm. The PET films used had a width of 150 mm and a thickness of 100 μm (SH-34, SKC Co., Ltd., Seoul, Korea). The operating tension was set to 10 N, the coating gap was 100 μm, and the printing velocity varied from 1 to 6 m/min. The 7-DHC coated layer was dried at 60 °C through the dryer length of 18 m and was rewound. The coated thickness was measured using an interferometer (NV-2000, Nanosystems Co., Ltd., Daejeon, Korea). HPLC results were obtained using the 1260 Infinity HPLC device (Agilent Technologies Inc., Santa Clara, CA, USA). The column used, column temperature, mobile phase, flow rate, detector, and injection amount were Eclipse XDE C18 (4.6 mm × 250 mm × 5 µm), 40 °C, isocratic (acetonitrile:methanol = 6:4), 1 mL/min, diode array detector (265 nm), and 10 μL, respectively. The measurements were performed at the Korea Polymer Testing and Research Institute (KOPTRI, Seoul), Ltd., an internationally recognized measurement institute.