The Role of Ammonium Chloride in the Powder Thermal Diffusion Alloying Process on a Magnesium Alloy
Abstract
:1. Introduction
2. Experimental Details
3. Results and Discussion
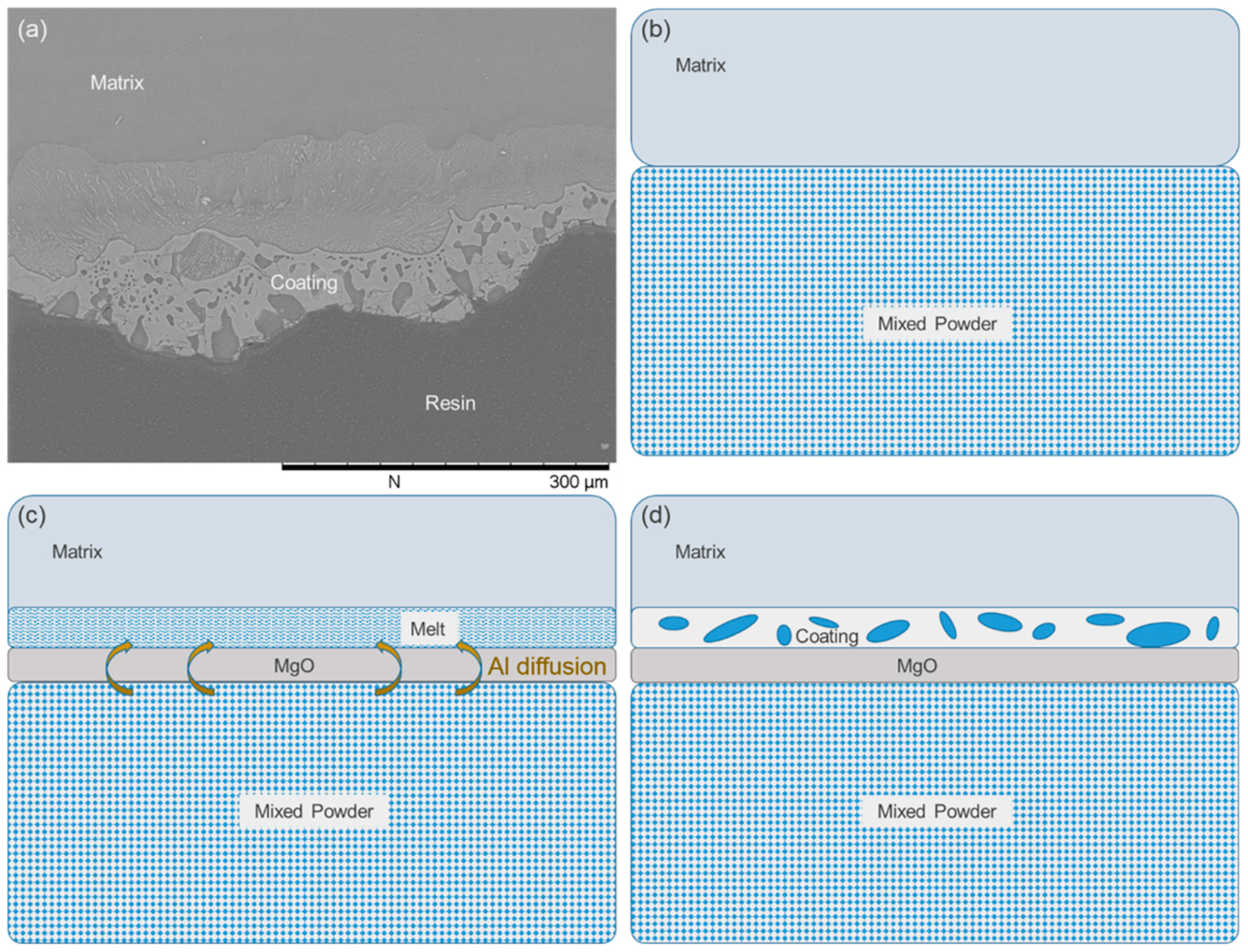
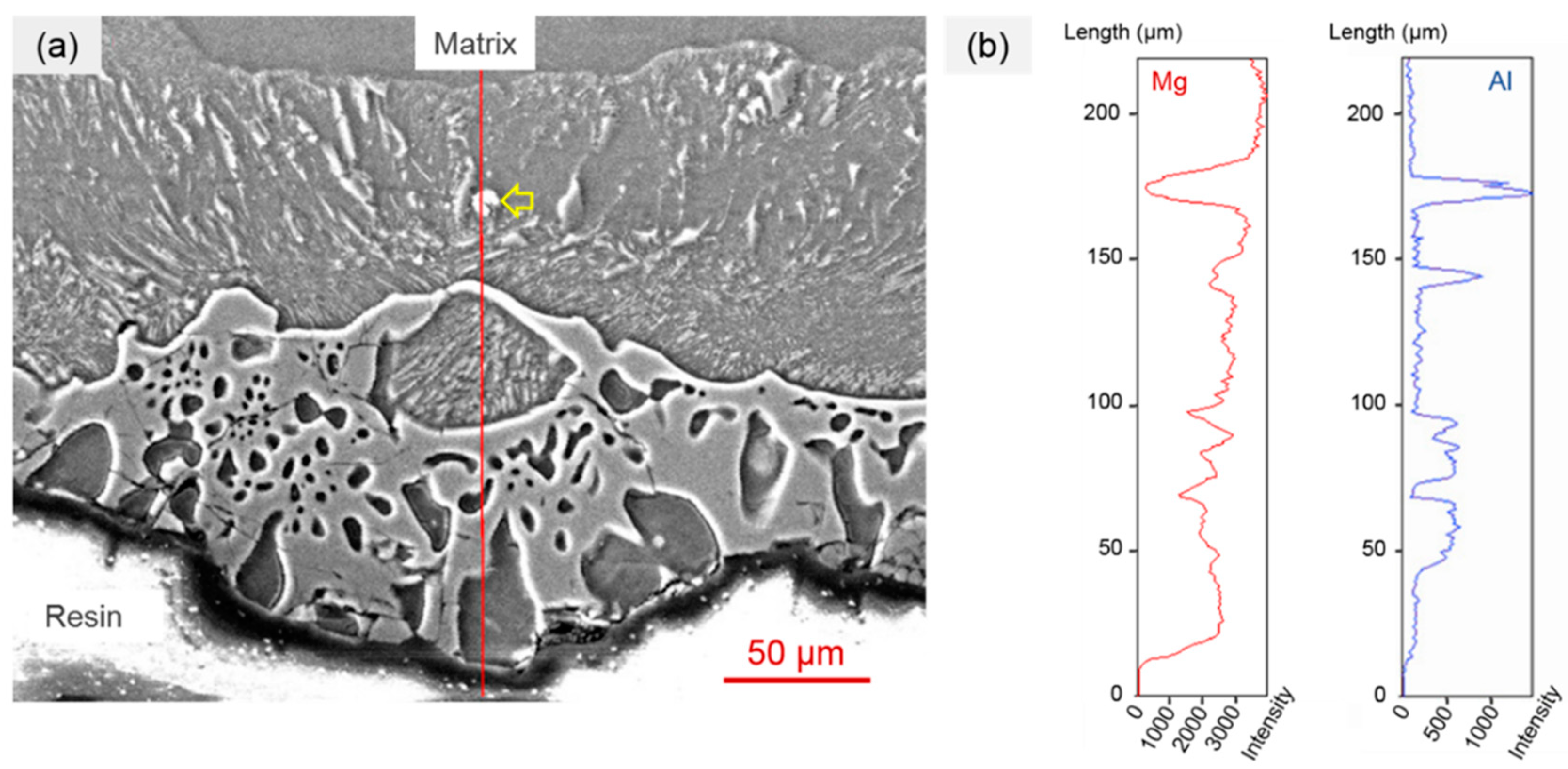
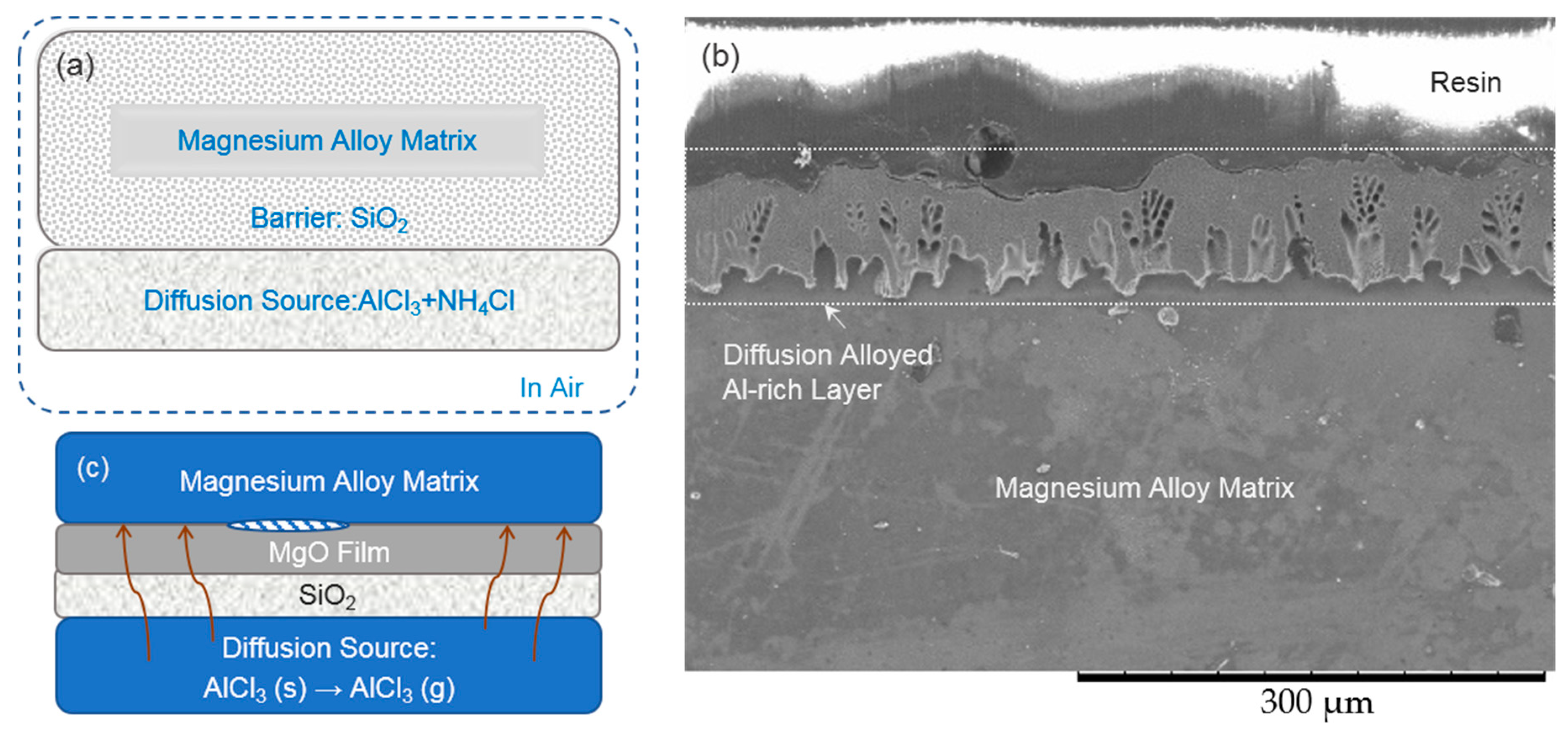
3.1. Formation of the Intermetallic Coating on the AZ91D Magnesium Alloy
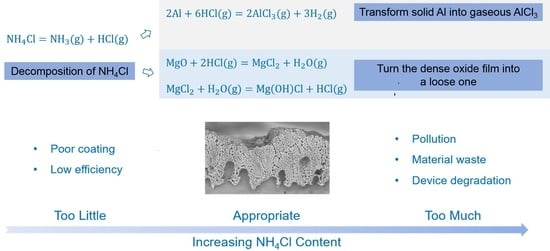
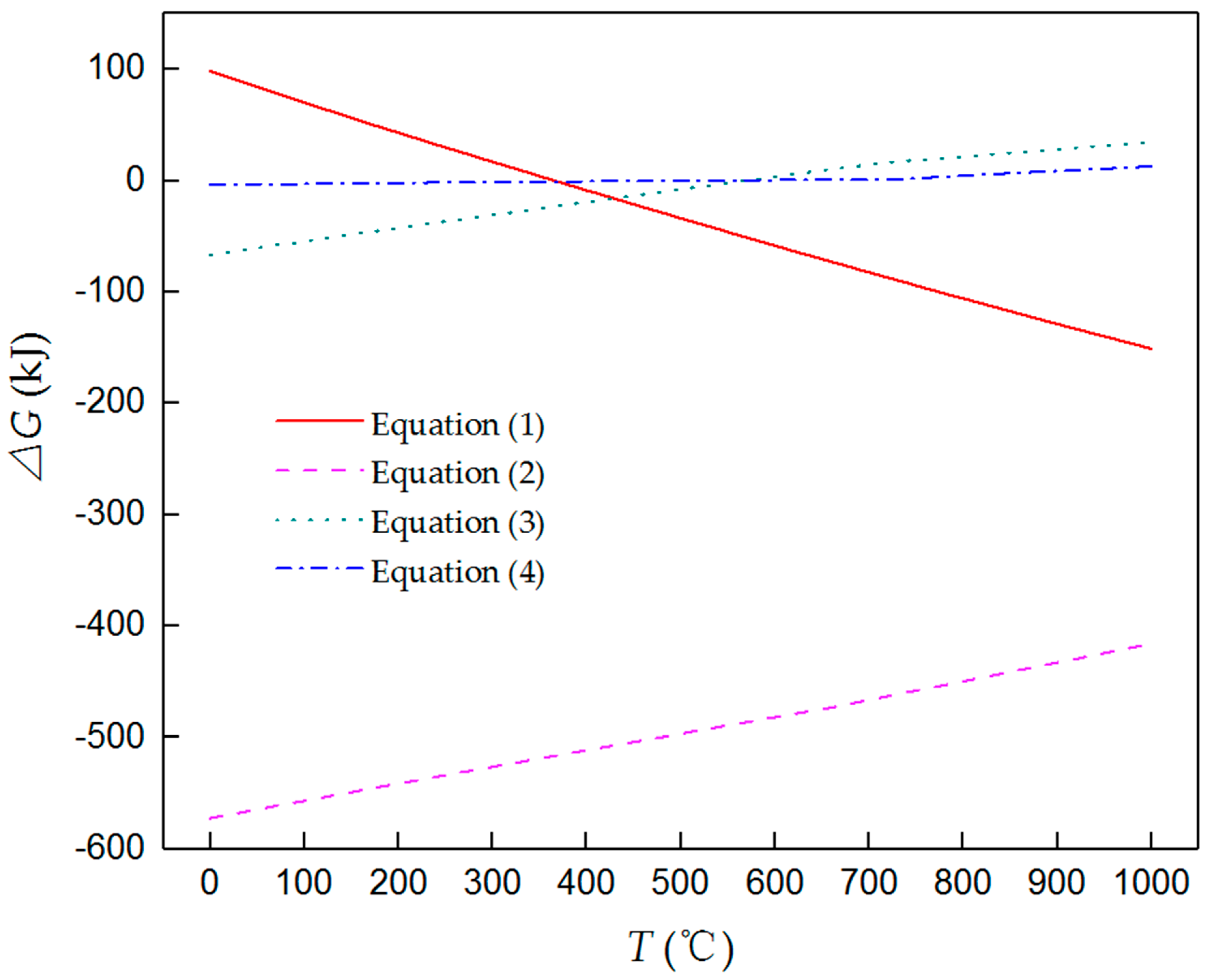
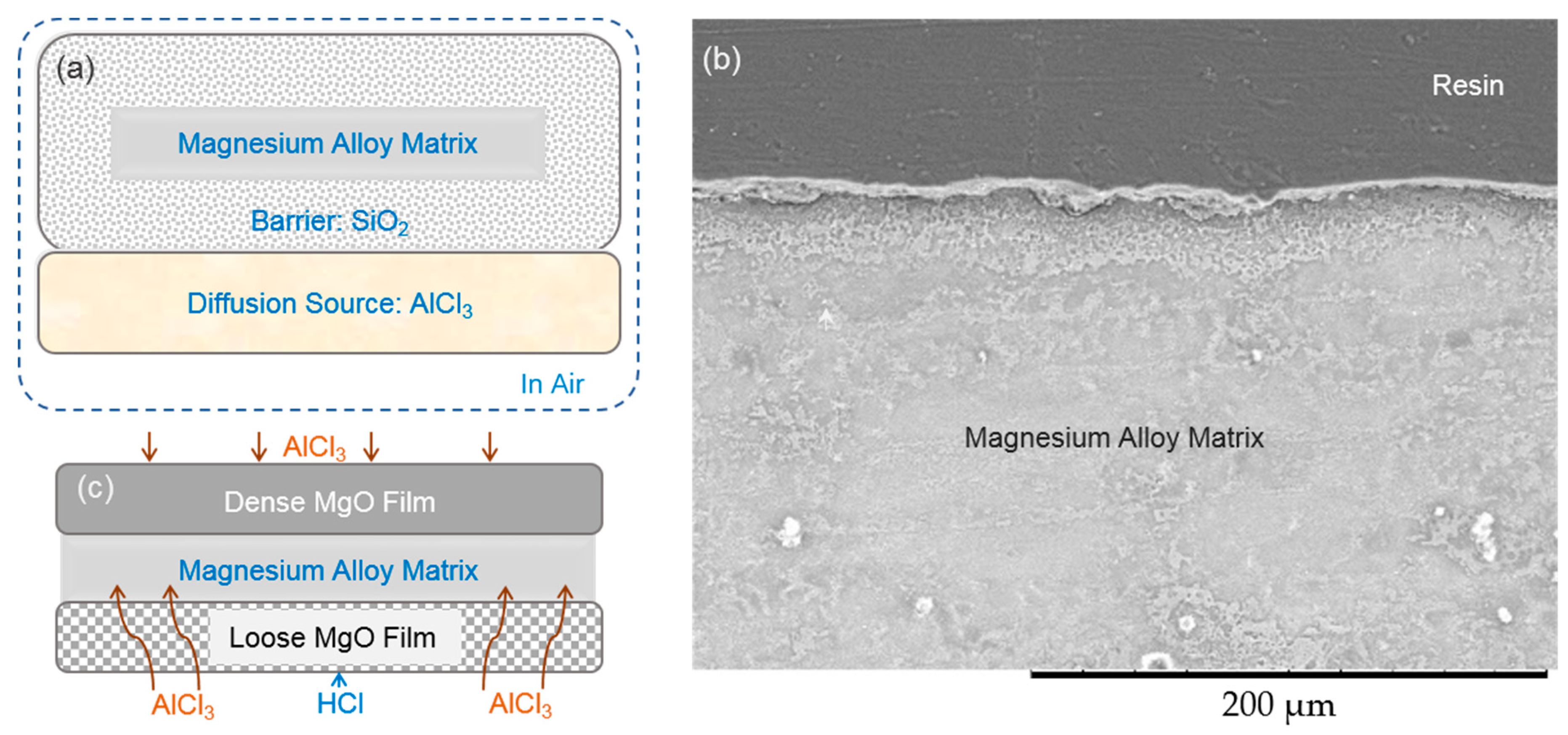
3.2. Ammonium Chloride Could Keep the Surface of the Magnesium Alloy Matrix Fresh
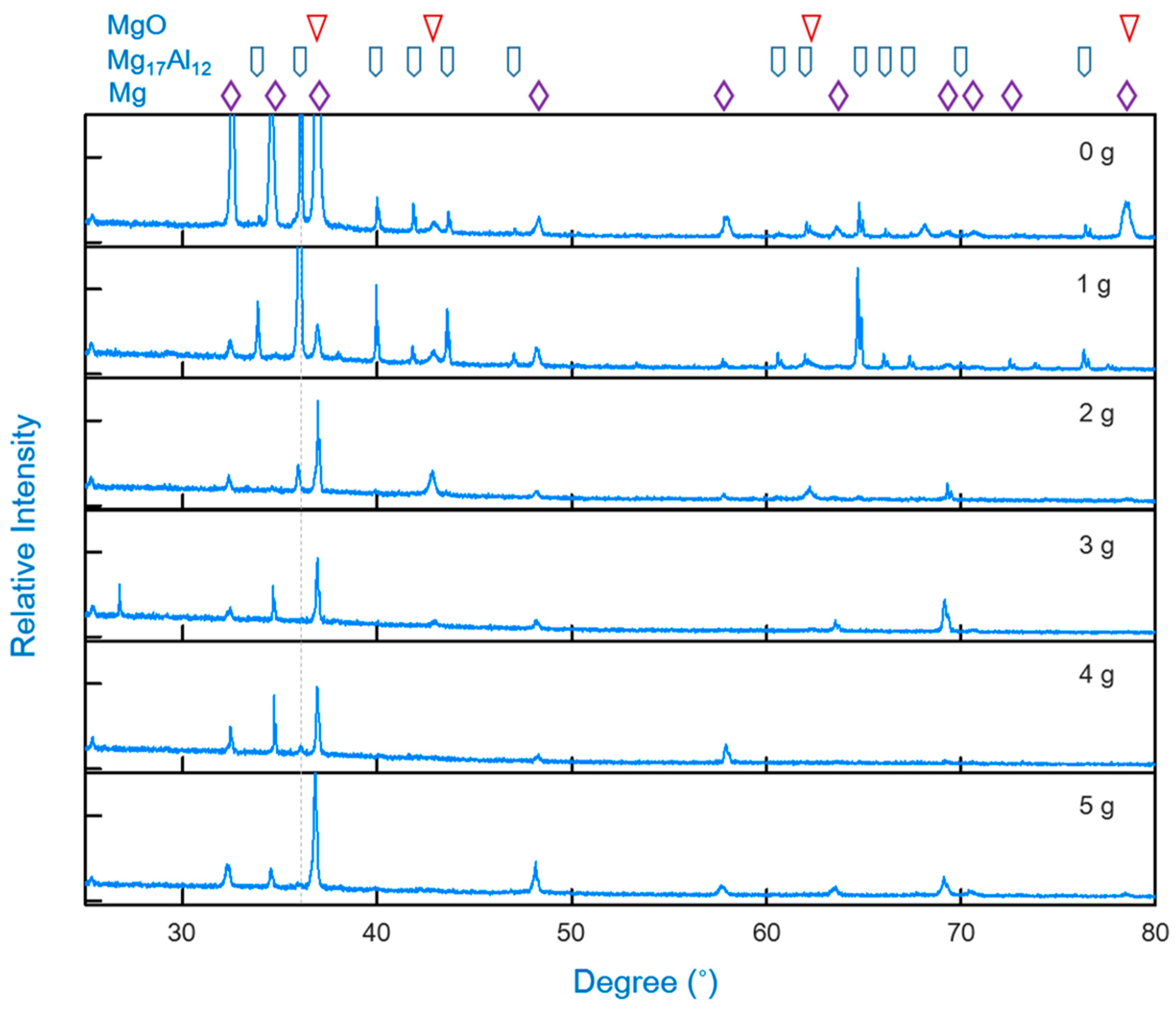
3.3. The Upper Content Limit of the Ammonium Chloride in a Diffusion Alloying Process
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Zhu, Y.Y.; Chang, W.H.; Zhang, S.F.; Song, Y.W.; Huang, H.D.; Zhao, R.F.; Li, G.Q.; Zhang, R.F.; Zhang, Y.J. Investigation on corrosion resistance and formation mechanism of a P–F–Zr contained micro-arc oxidation coating on AZ31B magnesium alloy using an orthogonal method. Coatings 2019, 9, 197. [Google Scholar] [CrossRef]
- Chen, J.; Kang, K.; Song, Y.W.; Han, E.H.; Ma, S.D.; Ao, J.Q. Corrosion behavior of hydrotalcite film on AZ31 alloy in simulated body fluid. Coatings 2019, 9, 113. [Google Scholar] [CrossRef]
- Lu, D.Z.; Huang, Y.L.; Jiang, Q.T.; Zheng, M.; Duan, J.Z.; Hou, B.R. An approach to fabricating protective coatings on a magnesium alloy utilising alumina. Surf. Coat. Technol. 2019, 367, 336–340. [Google Scholar] [CrossRef]
- Yang, H.Y.; Guo, X.W.; Wu, G.H.; Ding, W.J.; Birbilis, N. Electrodeposition of chemically and mechanically protective Al-coatings on AZ91D Mg alloy. Corros. Sci. 2011, 53, 381–387. [Google Scholar] [CrossRef]
- Xie, J.S.; Zhang, J.H.; Liu, S.J.; Li, Z.H.; Zhang, L.; Wu, R.Z.; Hou, L.G.; Zhang, M.L. Hydrothermal synthesis of protective coating on Mg alloy for degradable implant applications. Coatings 2019, 9, 160. [Google Scholar] [CrossRef]
- Zhang, X.C.; Wang, J.X.; Zhang, C.Y.; Liu, B.; Jiang, P.; Zhao, Y.; Buhe, B.; Zhang, T.; Meng, G.Z.; Wang, F.H. Formation process of an LDHs coating on magnesium alloy by a CO2 pressurization method. Coatings 2019, 9, 47. [Google Scholar] [CrossRef]
- Shaha, S.K.; Dayani, S.B.; Xue, Y.N.; Pang, X.; Jahed, H. Improving corrosion and corrosion-fatigue resistance of AZ31B cast Mg alloy using combined cold spray and top coatings. Coatings 2018, 8, 443. [Google Scholar] [CrossRef]
- Ma, Y.P.; Xu, K.W.; Wen, W.X.; He, X.P.; Liu, P.F. The effect of solid diffusion surface alloying on properties of ZM5 magnesium alloy. Surf. Coat. Technol. 2005, 190, 165–170. [Google Scholar] [CrossRef]
- Hirmke, J.; Zhang, M.X.; St John, D.H. Influence of chemical composition of Mg alloys on surface alloying by diffusion coating. Metall. Mater. Trans. A 2012, 43, 1621–1628. [Google Scholar] [CrossRef]
- He, M.F.; Liu, L.; Wu, Y.T.; Tang, Z.X.; Hu, W.B. Improvement of the properties of AZ91D magnesium alloy by treatment with a molten AlCl3–NaCl salt to form an Mg-Al intermetallic surface layer. J. Coat. Technol. Res. 2009, 6, 407–411. [Google Scholar] [CrossRef]
- Mola, R. The properties of Mg protected by Al- and Al/Zn-enriched layers containing intermetallic phases. J. Mater. Res. 2015, 30, 3682–3691. [Google Scholar] [CrossRef]
- Zhong, C.; Liu, F.; Wu, Y.T.; Le, J.J.; Liu, L.; He, M.F.; Zhu, J.C.; Hu, W.B. Protective diffusion coatings on magnesium alloys: A review of recent developments. J. Alloys Compd. 2012, 520, 11–21. [Google Scholar] [CrossRef]
- Chang, H.W.; Zhang, M.X.; Atrens, A.; Huang, H. Nanomechanical properties of Mg-Al intermetallic compounds produced by packed powder diffusion coating (PPDC) on the surface of AZ91E. J. Alloys Compd. 2014, 587, 527–532. [Google Scholar] [CrossRef]
- Shigematsu, I.; Nakamura, M.; Saitou, N.; Shimojima, K. Surface treatment of AZ91D magnesium alloy by aluminum diffusion coating. J. Mater. Sci. Lett. 2000, 19, 473–475. [Google Scholar] [CrossRef]
- Lu, D.Z.; Zhang, Q.C.; Wang, X.T.; Yang, L.H.; Ma, X.M.; Wang, W.C.; Huang, Y.L. Intermetallic layer obtained by the compact powder diffusion alloying method on AZ91D magnesium alloy in air. Surf. Coat. Technol. 2017, 309, 986–993. [Google Scholar] [CrossRef]
- Lu, D.Z.; Zhang, Q.C.; Wang, W.C.; Guan, F.; Ma, X.M.; Yang, L.H.; Wang, X.T.; Huang, Y.L.; Hou, B.R. Effect of cooling rate and the original matrix on the thermal diffusion alloyed intermetallic layer on magnesium alloys. Mater. Des. 2017, 120, 75–82. [Google Scholar] [CrossRef]
- Xiang, Z.D.; Datta, P.K. Kinetics of low-temperature pack aluminide coating formation on alloy steels. Metall. Mater. Trans. A 2006, 37, 3359–3365. [Google Scholar] [CrossRef]
- Lu, T.; Yao, D.Z.; Zhou, C.G. Low-temperature formation of aluminide coatings on Ni-base superalloys by pack cementation process. Chin. J. Aeronaut 2010, 23, 381–385. [Google Scholar] [CrossRef]
- Hu, G.X.; Xu, Z.X.; Liu, J.J.; Li, Y. Microstructure and corrosion resistance of simultaneous Al–Fe coating on copper by pack cementation. Surf. Coat. Technol. 2009, 203, 3392–3397. [Google Scholar] [CrossRef]
- Park, H.H.; Lee, K.T.; Shin, H.S. Simultaneous chromizing-aluminizing diffusion coating of austenitic stainless steel by a two-step CVD process. Oxid. Met. 1998, 50, 377–387. [Google Scholar] [CrossRef]
- Monas, A.; Shchyglo, O.; Kim, S.J.; Yim, C.D.; Höche, D.; Steinbach, I. Divorced eutectic solidification of Mg-Al alloys. JOM 2015, 67, 1805–1811. [Google Scholar] [CrossRef]
- Tan, Q.Y.; Atrens, A.; Mo, N.; Zhang, M.X. Oxidation of magnesium alloys at elevated temperatures in air: A review. Corros. Sci. 2016, 112, 734–759. [Google Scholar] [CrossRef]
- Czerwinski, F. The reactive element effect on high-temperature oxidation of magnesium. Int. Mater. Rev. 2015, 60, 264–296. [Google Scholar] [CrossRef]




| Element (wt. %) | Al | Zn | Mn | Si | Fe | Cu | Ni | Mg |
|---|---|---|---|---|---|---|---|---|
| AZ91D | 9.1 | 0.52 | 0.26 | 0.03 | 0.001 | 0.014 | 0.001 | Balance |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lu, D.; Jiang, Q.; Zheng, M.; Zhang, J.; Huang, Y.; Hou, B. The Role of Ammonium Chloride in the Powder Thermal Diffusion Alloying Process on a Magnesium Alloy. Coatings 2019, 9, 252. https://doi.org/10.3390/coatings9040252
Lu D, Jiang Q, Zheng M, Zhang J, Huang Y, Hou B. The Role of Ammonium Chloride in the Powder Thermal Diffusion Alloying Process on a Magnesium Alloy. Coatings. 2019; 9(4):252. https://doi.org/10.3390/coatings9040252
Chicago/Turabian StyleLu, Dongzhu, Quantong Jiang, Meng Zheng, Jie Zhang, Yanliang Huang, and Baorong Hou. 2019. "The Role of Ammonium Chloride in the Powder Thermal Diffusion Alloying Process on a Magnesium Alloy" Coatings 9, no. 4: 252. https://doi.org/10.3390/coatings9040252
APA StyleLu, D., Jiang, Q., Zheng, M., Zhang, J., Huang, Y., & Hou, B. (2019). The Role of Ammonium Chloride in the Powder Thermal Diffusion Alloying Process on a Magnesium Alloy. Coatings, 9(4), 252. https://doi.org/10.3390/coatings9040252