One-Step Preparation of Hyperbranched Polyether Functionalized Graphene Oxide for Improved Corrosion Resistance of Epoxy Coatings
Abstract
1. Introduction
2. Experimental
2.1. Materials
2.2. Characterizations
2.3. Preparation of GO
2.4. Preparation of EHBPE
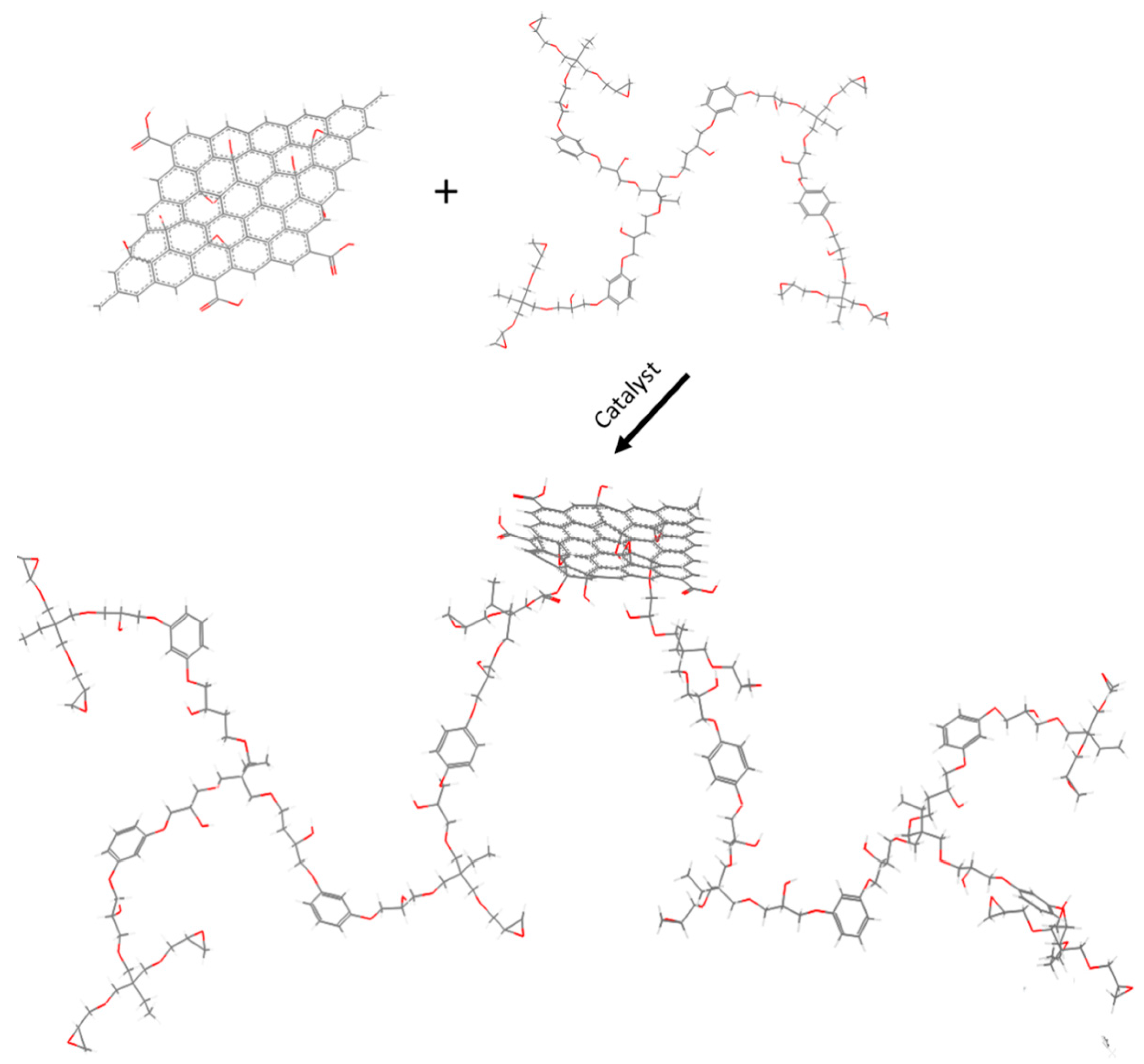
2.5. Preparation of EHBPE-GO
2.6. Preparation of EHBPE-GO/DGEBA Nanocomposite Epoxy Coating
2.7. Corrosion Testing
3. Result and Discussion
3.1. Characterization of EHBPE-GO
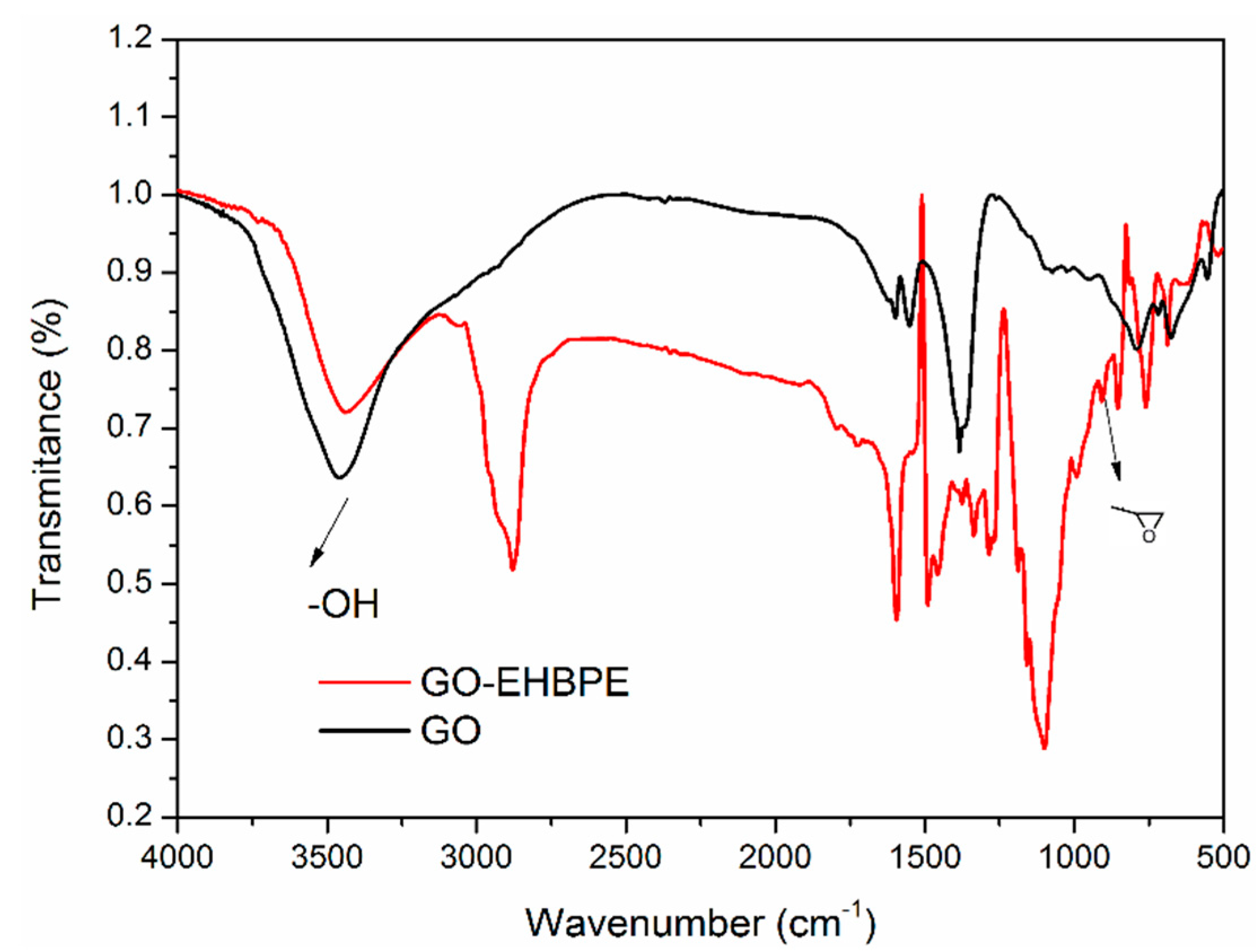
3.1.1. FTIR Characterization
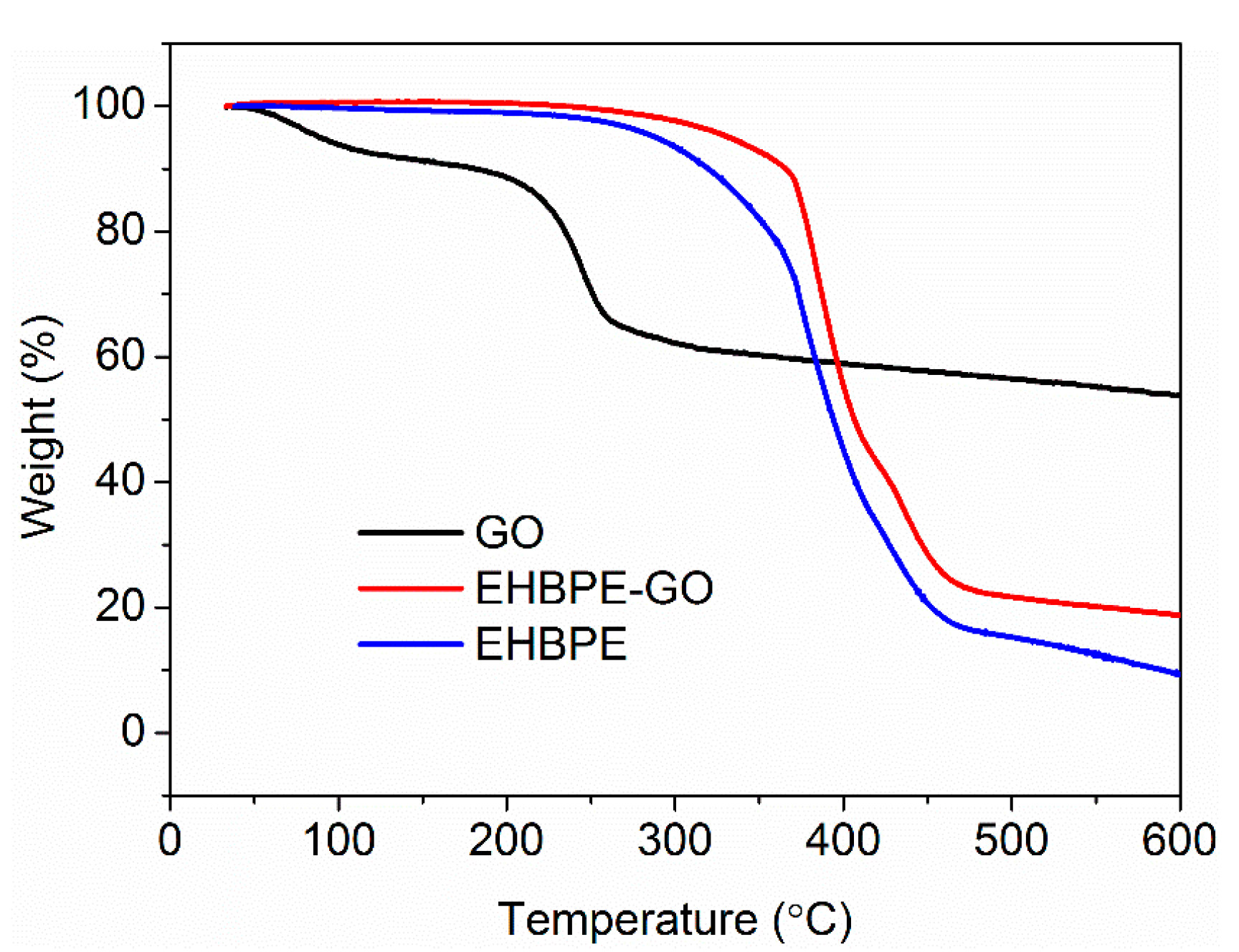
3.1.2. TGA Characterization
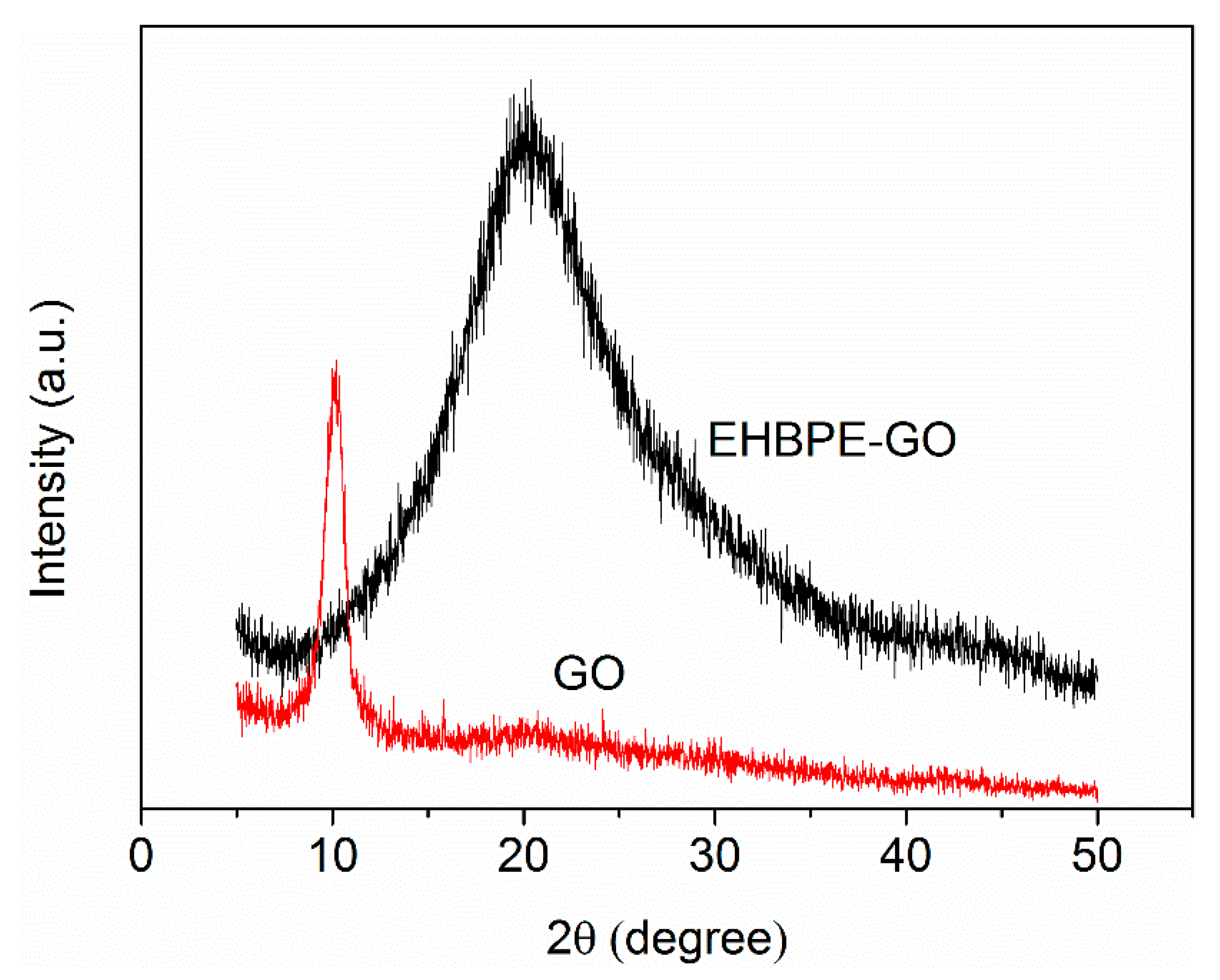
3.1.3. XRD Characterization
3.1.4. TEM Characterization
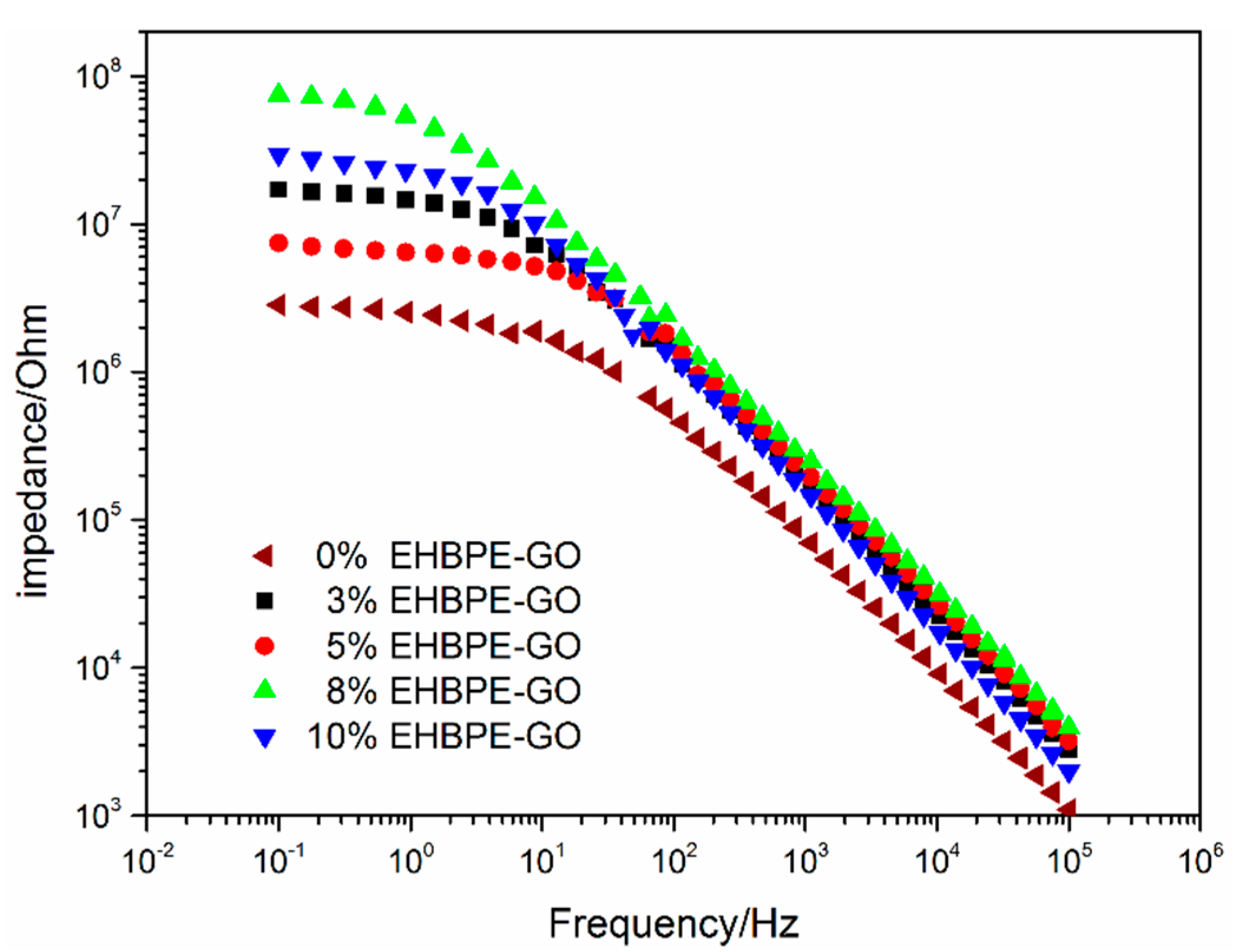
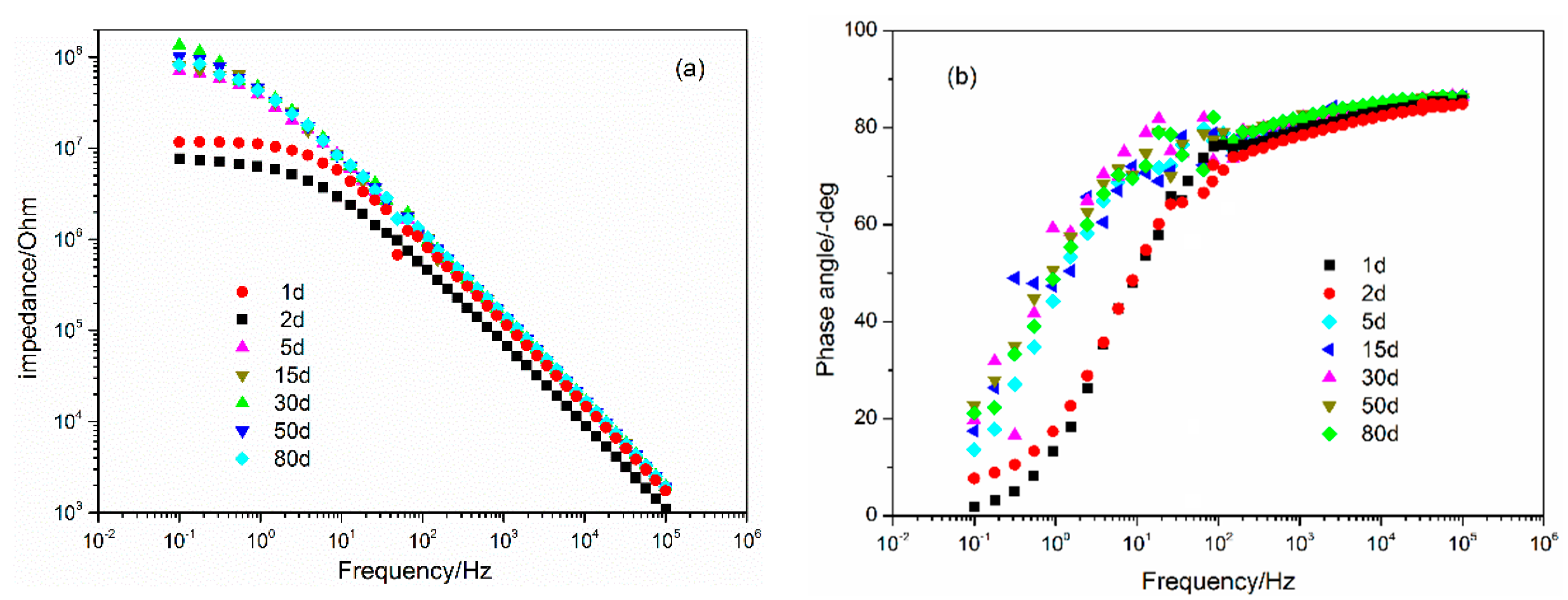
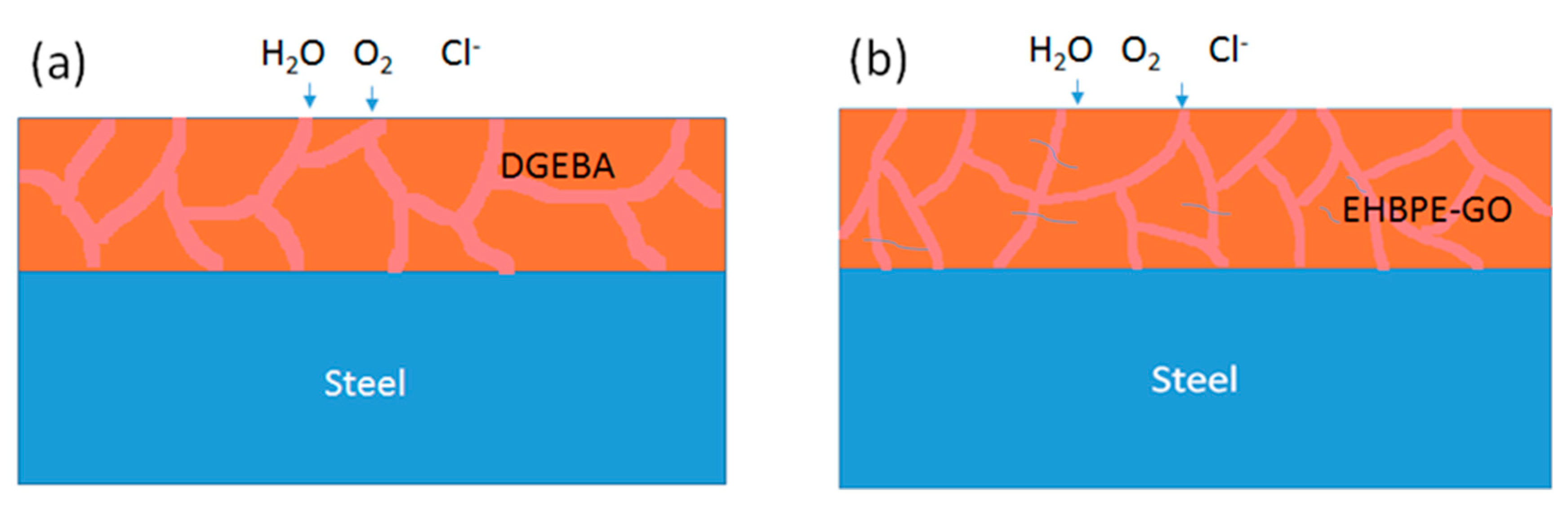
3.2. EIS Characterization
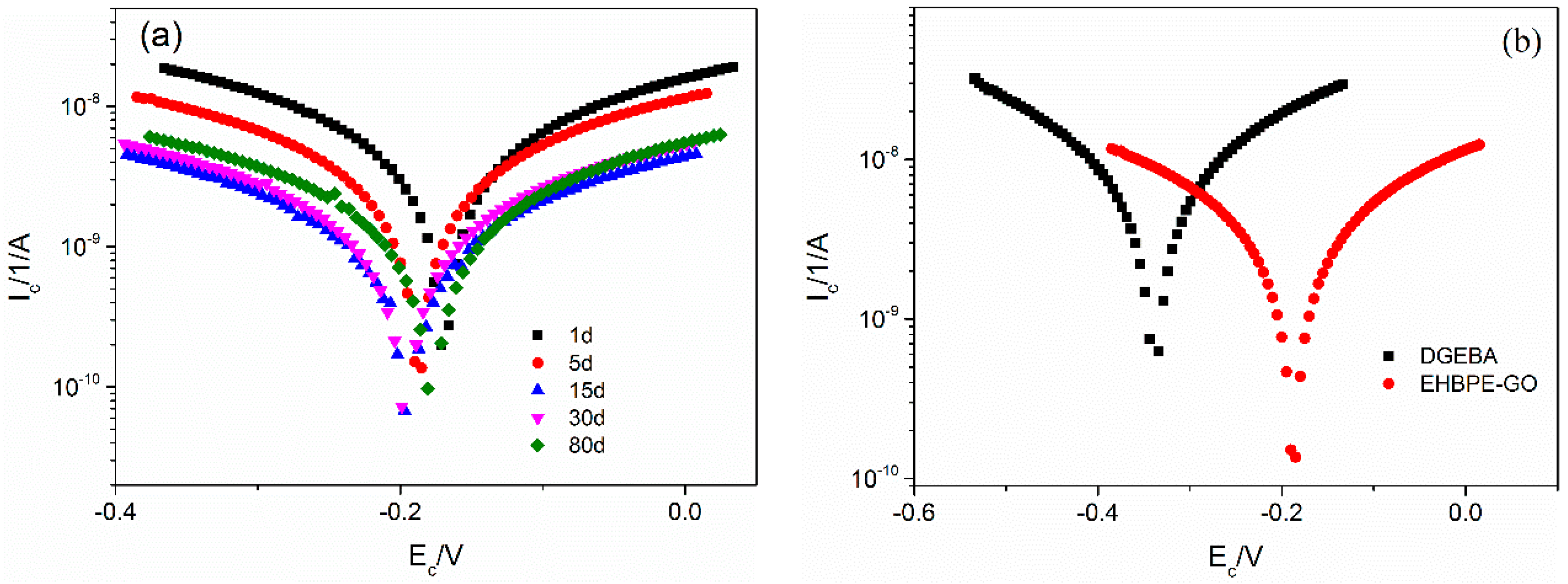
3.3. Potentiodynamic Polarization Test
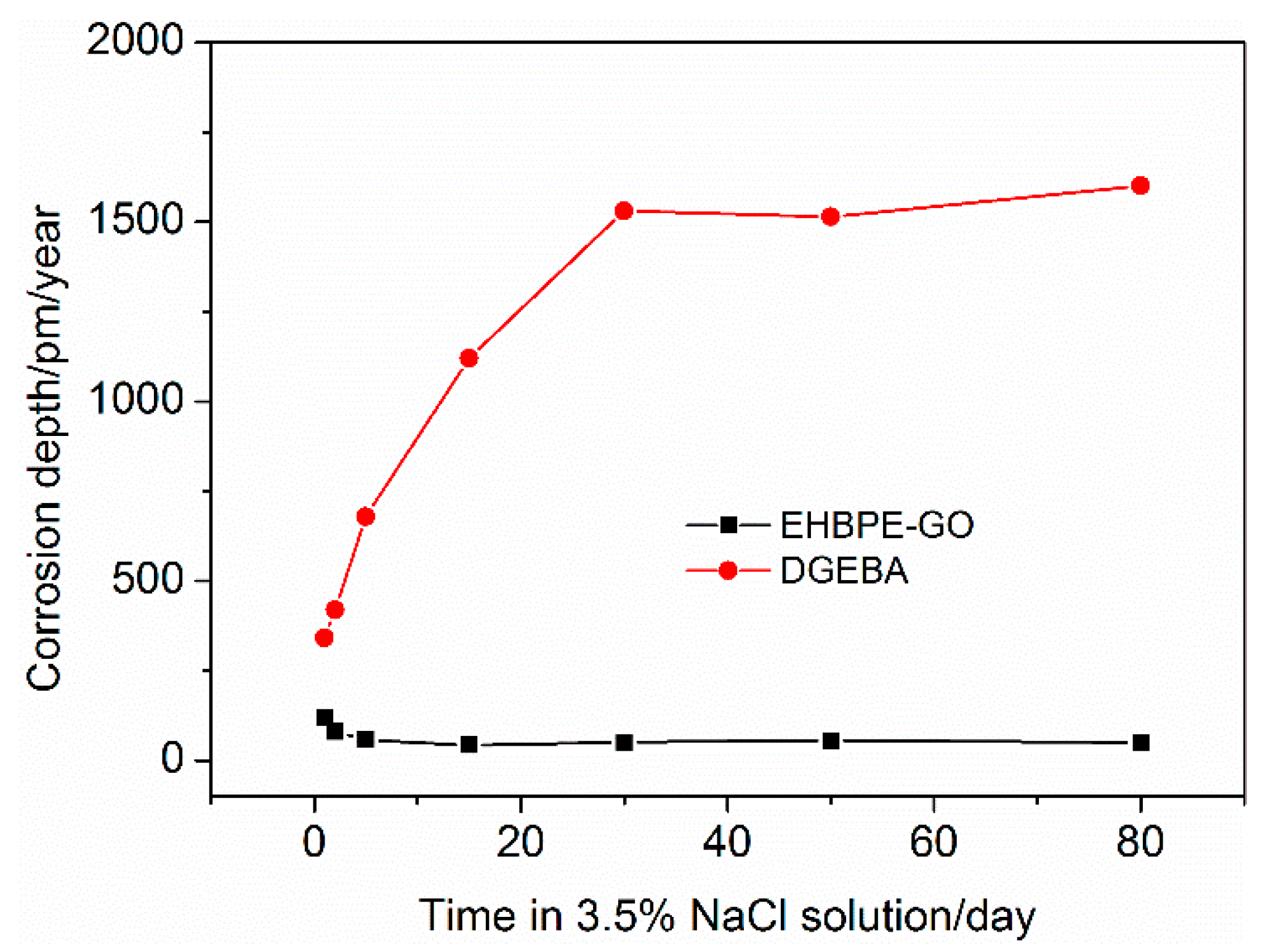
3.4. Corrosion Testing
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Messori, M.; Nobili, A.; Signorini, C.; Sola, A. Mechanical performance of epoxy coated AR-glass fabric Textile Reinforced Mortar: Influence of coating thickness and formulation. Compos. Part B Eng. 2018, 149, 135–143. [Google Scholar] [CrossRef]
- Motamedi, M.; Ramezanzadeh, M.; Ramezanzadeh, B.; Saadatmandi, S. Enhancement of the active/passive anti-corrosion properties of epoxy coating via inclusion of histamine/zinc modified/reduced graphene oxide nanosheets. Appl. Surface Sci. 2019, 488, 77–91. [Google Scholar] [CrossRef]
- Dell’Erba, I.E.; Williams, R.J. Synthesis of oligomeric silsesquioxanes functionalized with (β-carboxyl)ester groups and their use as modifiers of epoxy networks. Eur. Polym. J. 2007, 43, 2759. [Google Scholar] [CrossRef]
- Ahmad, S.; Gupta, A.P.; Sharmin, E.; Alam, M.; Pandey, S.K. Synthesis, characterization and development of high performance siloxane-modified epoxy paints. Prog. Org. Coat. 2005, 54, 248. [Google Scholar] [CrossRef]
- Thomas, R.; Yumei, D.; Yuelong, H.; Le, Y.; Moldenaers, P.; Weimin, Y.; Czigany, T. Miscibility, morphology, thermal, and mechanical properties of a DGEBA based epoxy resin toughened with a liquid rubber. Polymer 2008, 49, 278. [Google Scholar] [CrossRef]
- Topsakal, M.; Şahin, H.; Ciraci, S. Graphene coatings: An efficient protection from oxidation. Phys. Rev. B Condens. Matter Mater. Phys. 2012, 85, 155445. [Google Scholar] [CrossRef]
- Su, Y.; Kravets, V.G.; Wong, S.L.; Waters, J.; Geim, A.K.; Nair, R.R. Impermeable barrier films and protective coatings based on reduced graphene oxide. Nat. Commun. 2014, 5, 4843. [Google Scholar] [CrossRef]
- Ghosh, S.; Bao, W.; Nika, D.L.; Subrina, S.; Pokatilov, E.P.; Lau, C.N.; Balandin, A.A. Dimensional crossover of thermal transport in few-layer graphene. Nat. Mater. 2010, 9, 555–558. [Google Scholar] [CrossRef]
- Lee, C.; Wei, X.; Kysar, J.W.; Hone, J. Measurement of the Elastic Properties and Intrinsic Strength of Monolayer Graphene. Science 2008, 321, 385–388. [Google Scholar] [CrossRef]
- Wang, Y.; Chen, S.; Chen, X.; Lu, Y.; Miao, M.; Zhang, D. Controllability of epoxy equivalent weight and performance of hyperbranched epoxy resins. Compos. Part B Eng. 2019, 160, 615–625. [Google Scholar] [CrossRef]
- Miao, X.; Xing, A.; Yang, W.; He, L.; Meng, Y.; Li, X. Synthesis and characterization of hyperbranched polyether/DGEBA hybrid coatings. React. Funct. Polym. 2018, 122, 116–122. [Google Scholar] [CrossRef]
- Zhang, M.; Yan, H.; Yang, X.; Liu, C. Effect of functionalized graphene oxide with a hyperbranched cyclotriphosphazene polymer on mechanical and thermal properties of cyanate ester composites. RSC Adv. 2014, 4, 45930–45938. [Google Scholar] [CrossRef]
- Yan, H.; Li, S.; Jia, Y.; Ma, X. Hyperbranched polysiloxane grafted graphene for improved tribological performance of bismaleimide composites. RSC Adv. 2015, 5, 12578–12582. [Google Scholar] [CrossRef]
- Wu, C.; Huang, X.; Wang, G.; Wu, X.; Yang, K.; Li, S.; Jiang, P. Hyperbranched-polymer functionalization of graphene sheets for enhanced mechanical and dielectric properties of polyurethane composites. J. Mater. Chem. 2012, 22, 7010–7019. [Google Scholar] [CrossRef]
- Qi, Z.; Tan, Y.; Gao, L.; Zhang, C.; Wang, L.; Xiao, C. Effects of hyperbranched polyamide functionalized graphene oxide on curing behaviour and mechanical properties of epoxy composites. Polym. Test. 2018, 71, 145–155. [Google Scholar] [CrossRef]
- Mahapatra, S.; Ramasamy, M.; Yoo, H.; Cho, J. A reactive graphene sheet in situ functionalized hyperbranched polyurethane for high performance shape memory material. RSC Adv. 2014, 4, 15146–15153. [Google Scholar] [CrossRef]
- Ding, R.; Jiang, J.; Gui, T. Study of impedance model and water transport behavior of modified solvent-free epoxy anticorrosion coating by EIS. J. Coat. Technol. Res. 2016, 13, 501–515. [Google Scholar] [CrossRef]
- Lakshmi, R.V.; Aruna, S.T.; Anandan, C.; Bera, P.; Sampath, S. EIS and XPS studies on the self-healing properties of Ce-modified silica-alumina hybrid coatings: Evidence for Ce(III) migration. Surf. Coat. Technol. 2017, 309, 363. [Google Scholar] [CrossRef]
- Shi, X.; Nguyen, T.A.; Suo, Z.; Liu, Y.; Avci, R. Effect of nanoparticles on the anticorrosion and mechanical properties of epoxy coating. Surf. Coat. Technol. 2009, 204, 237. [Google Scholar] [CrossRef]
- Yuan, X.; Yue, Z.F.; Chen, X.; Wen, S.F.; Li, L.; Feng, T. EIS study of effective capacitance and water uptake behaviors of silicone-epoxy hybrid coatings on mild steel. Prog. Org. Coat. 2015, 86, 41. [Google Scholar] [CrossRef]
- Bakhshandeh, E.; Jannesari, A.; Ranjbar, Z.; Sobhani, S.; Saeb, M.R. Anti-corrosion hybrid coatings based on epoxy–silica nano-composites: Toward relationship between the morphology and EIS data. Prog. Org. Coat. 2014, 77, 1169. [Google Scholar] [CrossRef]
- Hummers, W.S., Jr.; Offeman, R.E. Preparation of Graphitic Oxide. J. Am. Chem. Soc. 1958, 80, 1339. [Google Scholar] [CrossRef]
- Xu, Y.X.; Bai, H.; Lu, G.W.; Li, C.; Shi, G.Q. Flexible Graphene Films via the Filtration of Water-Soluble Noncovalent Functionalized Graphene Sheets. J. Am. Chem. Soc. 2008, 130, 5856. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Huang, X.; Xie, L.; Wu, X.; Yu, J.; Jiang, P. Morphology-controllable graphene–TiO2 nanorod hybrid nanostructures for polymer composites with high dielectric performance. J. Mater. Chem. 2011, 21, 17729. [Google Scholar] [CrossRef]
- Miao, X.; Guo, Y.; He, L.; Meng, Y.; Li, X. Rheological behaviors of a series of hyperbranched polyethers. Chin. J. Polym. Sci. 2015, 33, 1574–1585. [Google Scholar] [CrossRef]
- Bao, C.L.; Guo, Y.Q.; Song, L.; Kan, Y.C.; Qian, X.D.; Hu, Y. In situ preparation of functionalized graphene oxide/epoxy nanocomposites with effective reinforcements. J. Mater. Chem. 2011, 21, 13290–13298. [Google Scholar] [CrossRef]
- Zhou, T.; Li, F.; Fan, Y.; Song, W.; Mu, X.; Zhang, H.; Wang, Y. Hydrogen-bonded dimer stacking induced emission of aminobenzoic acid compounds. Chem. Commun. 2009, 22, 3199–3201. [Google Scholar] [CrossRef]
- Moon, I.K.; Lee, J.; Ruoff, R.S.; Lee, H. Reduced graphene oxide by chemical graphitization. Nat. Commun. 2010, 1, 73–78. [Google Scholar] [CrossRef]
- Baruah, P.; Karak, N. Bio-based tough hyperbranched epoxy/graphene oxide nanocomposite with enhanced biodegradability attribute. Polym. Degrad. Stabil. 2016, 129, 26–33. [Google Scholar] [CrossRef]
- Guan, L.Z.; Wan, Y.J.; Gong, L.X.; Yan, D.; Tang, L.C.; Wu, L.B.; Jiang, J.X.; Laia, G.Q. Toward effective and tunable interphases in graphene oxide/epoxy composites by grafting different chain lengths of polyetheramine onto graphene oxide. J. Mater. Chem. A 2014, 2, 15058–15069. [Google Scholar] [CrossRef]
- Liu, J.; Tao, L.; Yang, W.; Li, D.; Boyer, C.; Wuhrer, R.; Braet, F.; Davis, T.P. Synthesis, Characterization, and Multilayer Assembly of pH Sensitive Graphene/Polymer Nanocomposites. Langmuir 2010, 26, 10068–10075. [Google Scholar] [CrossRef] [PubMed]
- Zhan, Y.; Yang, X.; Guo, H.; Yang, J.; Meng, F.; Liu, X. Cross-linkable nitrile functionalized graphene oxide/poly (arylene ether nitrile) nanocomposite films with high mechanical strength and thermal stability. J. Mater. Chem. 2012, 22, 5602–5608. [Google Scholar] [CrossRef]
- Shau, S.M.; Juang, T.Y.; Lin, H.S.; Huang, C.L.; Hsieh, C.F.; Wu, J.Y.; Jeng, R.J. Individual graphene oxide platelets through direct molecular exfoliation with globular amphiphilic hyperbranched polymers. Polym. Chem. 2012, 3, 1249–1259. [Google Scholar] [CrossRef]
- Wang, J.Y.; Yang, S.Y.; Huang, Y.L.; Tien, H.W.; Chin, W.K.; Ma, C.C.M. Preparation and properties of graphene oxide/polyimide composite films with low dielectric constant and ultrahigh strength via in situpolymerization. J. Mater. Chem. 2011, 21, 13569–13575. [Google Scholar] [CrossRef]
- Li, R.; Liang, J.; Hou, Y.; Chu, Q. Enhanced corrosion performance of Zn coating by incorporating graphene oxide electrodeposited from deep eutectic solvent. RSC Adv. 2015, 5, 60698–60707. [Google Scholar] [CrossRef]
- Liang, J.; Srinivasan, P.B.; Blawert, C.; Dietzel, W. Influence of pH on the deterioration of plasma electrolytic oxidation coated AM50 magnesium alloy in NaCl solutions. Corros. Sci. 2010, 52, 540–547. [Google Scholar] [CrossRef]
- Yu, Z.; Lv, L.; Ma, Y.; Di, H.; He, Y. Covalent modification of graphene oxide by metronidazole for reinforced anti-corrosion properties of epoxy coatings. RSC Adv. 2016, 6, 18217–18226. [Google Scholar] [CrossRef]
- Kugler, S.; Kowalczyk, K.; Spychaj, T. Influence of synthetic and bio-based amine curing agents on properties of solventless epoxy varnishes and coatings with carbon nanofillers. Prog. Org. Coat. 2017, 109, 83–91. [Google Scholar] [CrossRef]

| Full Name | Abbreviation |
|---|---|
| Hyperbranched polyether functionalized graphene oxide | EHBPE-GO |
| Graphene oxide | GO |
| 1,1,1-Trihydroxymethylpropane triglycidyl ether | TMPGE |
| Tetrabutylammonium bromide | TBAB |
| Diglycidyl ether of bisphenol A | DGEBA |
| Sample Code | Ec (V) | Ic (nA·cm2) | Rp (MΩ·cm2) |
|---|---|---|---|
| DGEBA | −0.23 | 10.90 | 5.13 |
| EHBPE-GO/DGEBA | −0.18 | 0.14 | 0.64 |
| Sample Code | 5% NaOH (day) | 10% H2SO4 (day) | H2O (day) | Salt Spray Test (day) |
|---|---|---|---|---|
| DGEBA | 80 | 8 | 80 | 80 |
| EHBPE-GO/DGEBA | 80 | 12 | 80 | 70 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Miao, X.; Xing, A.; He, L.; Meng, Y.; Li, X. One-Step Preparation of Hyperbranched Polyether Functionalized Graphene Oxide for Improved Corrosion Resistance of Epoxy Coatings. Coatings 2019, 9, 844. https://doi.org/10.3390/coatings9120844
Miao X, Xing A, He L, Meng Y, Li X. One-Step Preparation of Hyperbranched Polyether Functionalized Graphene Oxide for Improved Corrosion Resistance of Epoxy Coatings. Coatings. 2019; 9(12):844. https://doi.org/10.3390/coatings9120844
Chicago/Turabian StyleMiao, Xuepei, An Xing, Lifan He, Yan Meng, and Xiaoyu Li. 2019. "One-Step Preparation of Hyperbranched Polyether Functionalized Graphene Oxide for Improved Corrosion Resistance of Epoxy Coatings" Coatings 9, no. 12: 844. https://doi.org/10.3390/coatings9120844
APA StyleMiao, X., Xing, A., He, L., Meng, Y., & Li, X. (2019). One-Step Preparation of Hyperbranched Polyether Functionalized Graphene Oxide for Improved Corrosion Resistance of Epoxy Coatings. Coatings, 9(12), 844. https://doi.org/10.3390/coatings9120844





