Influence of Film Quality on Power Conversion Efficiency in Perovskite Solar Cells
Abstract
1. Introduction
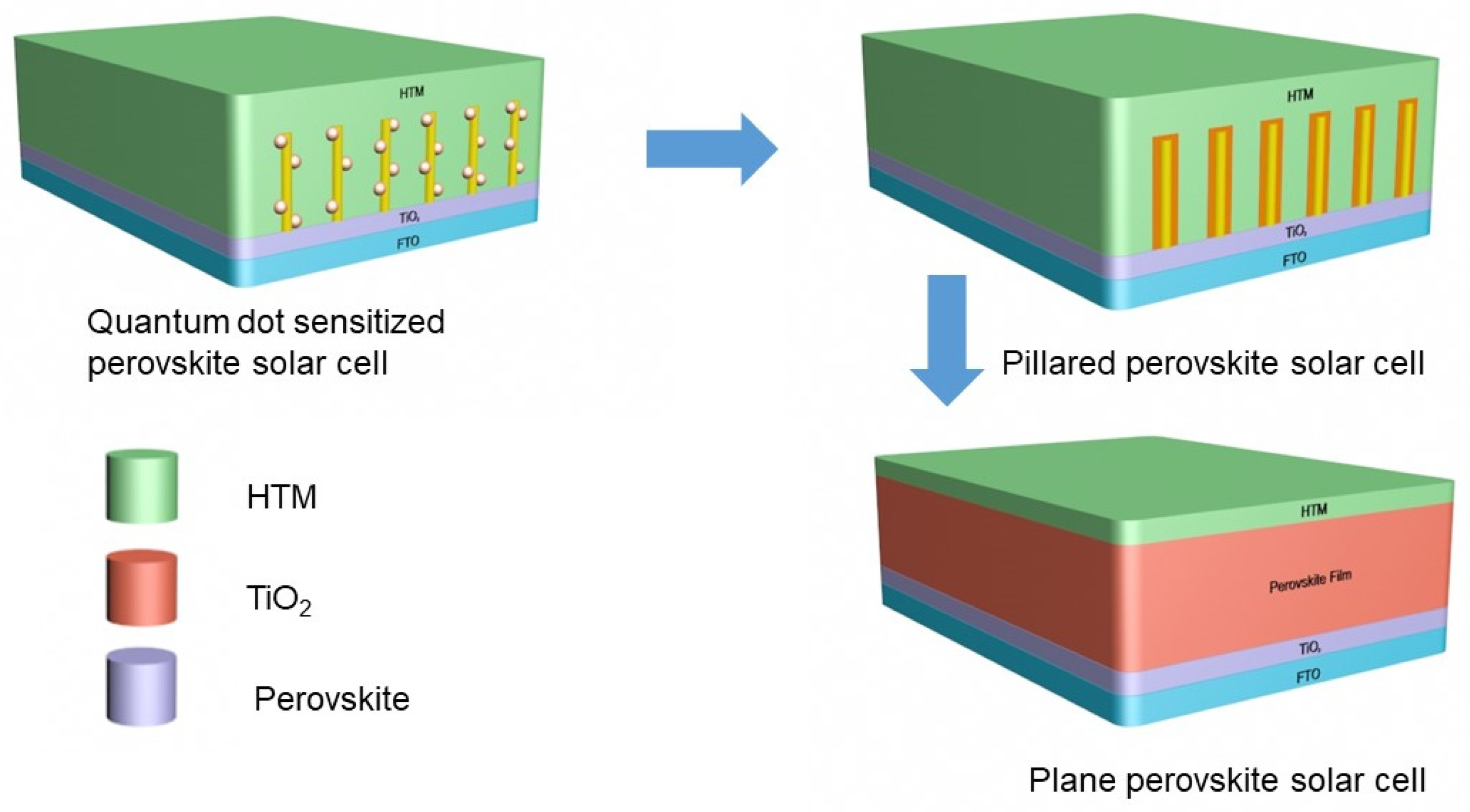
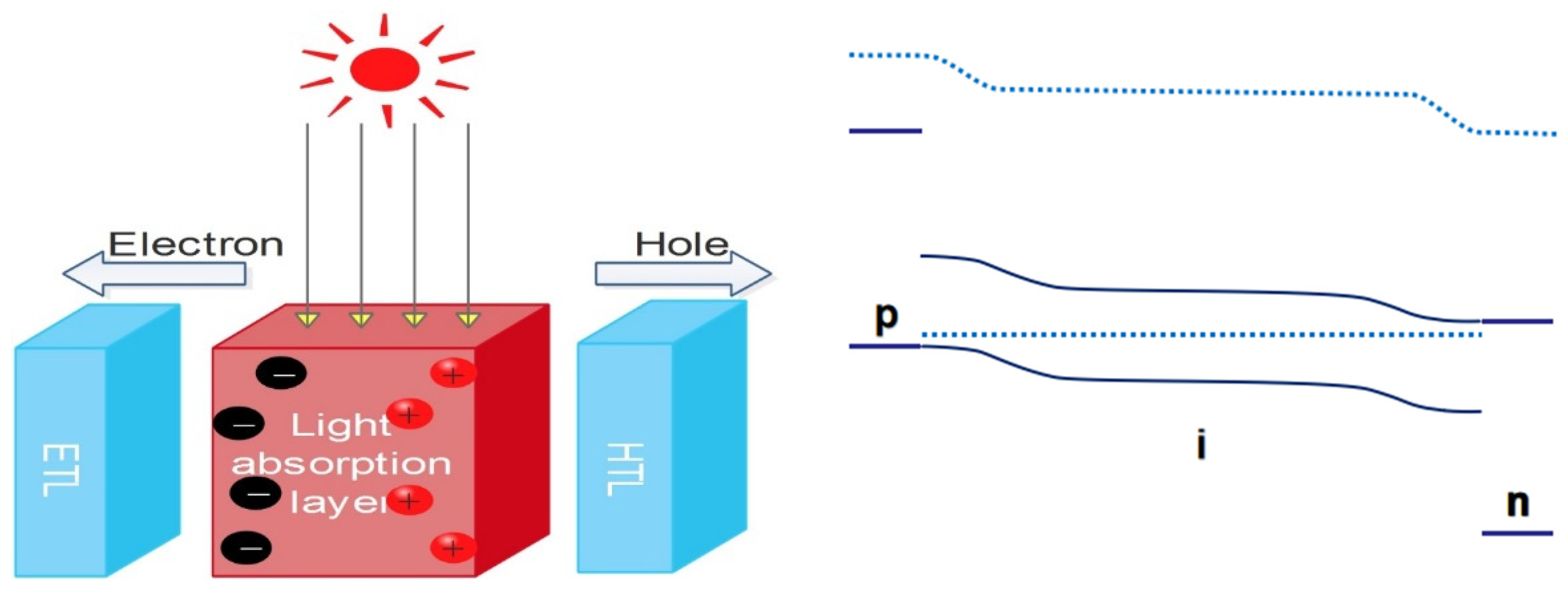
2. The Role of Film Quality in Perovskite Solar Cells
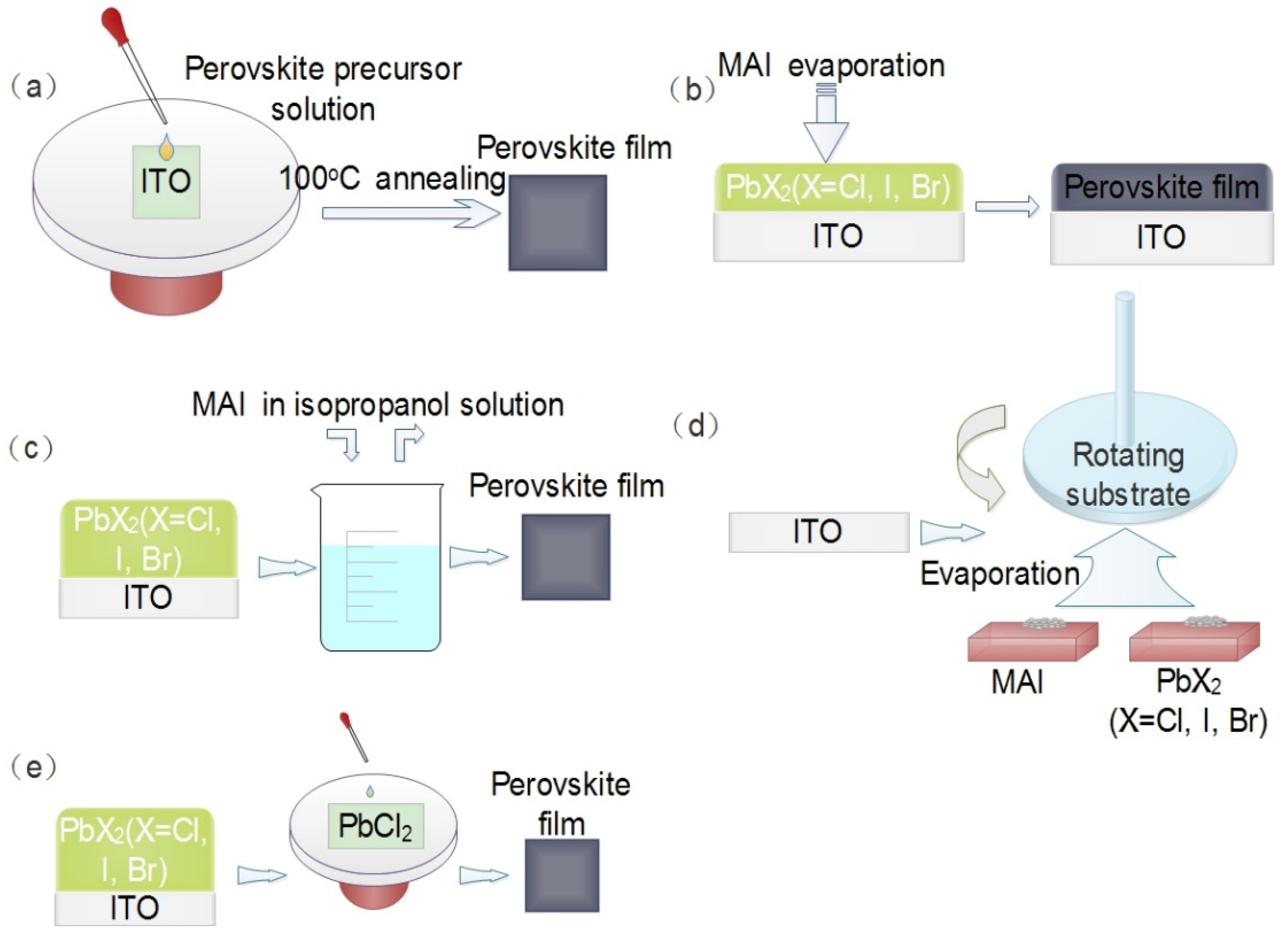
3. Fabrication of Perovskite Film
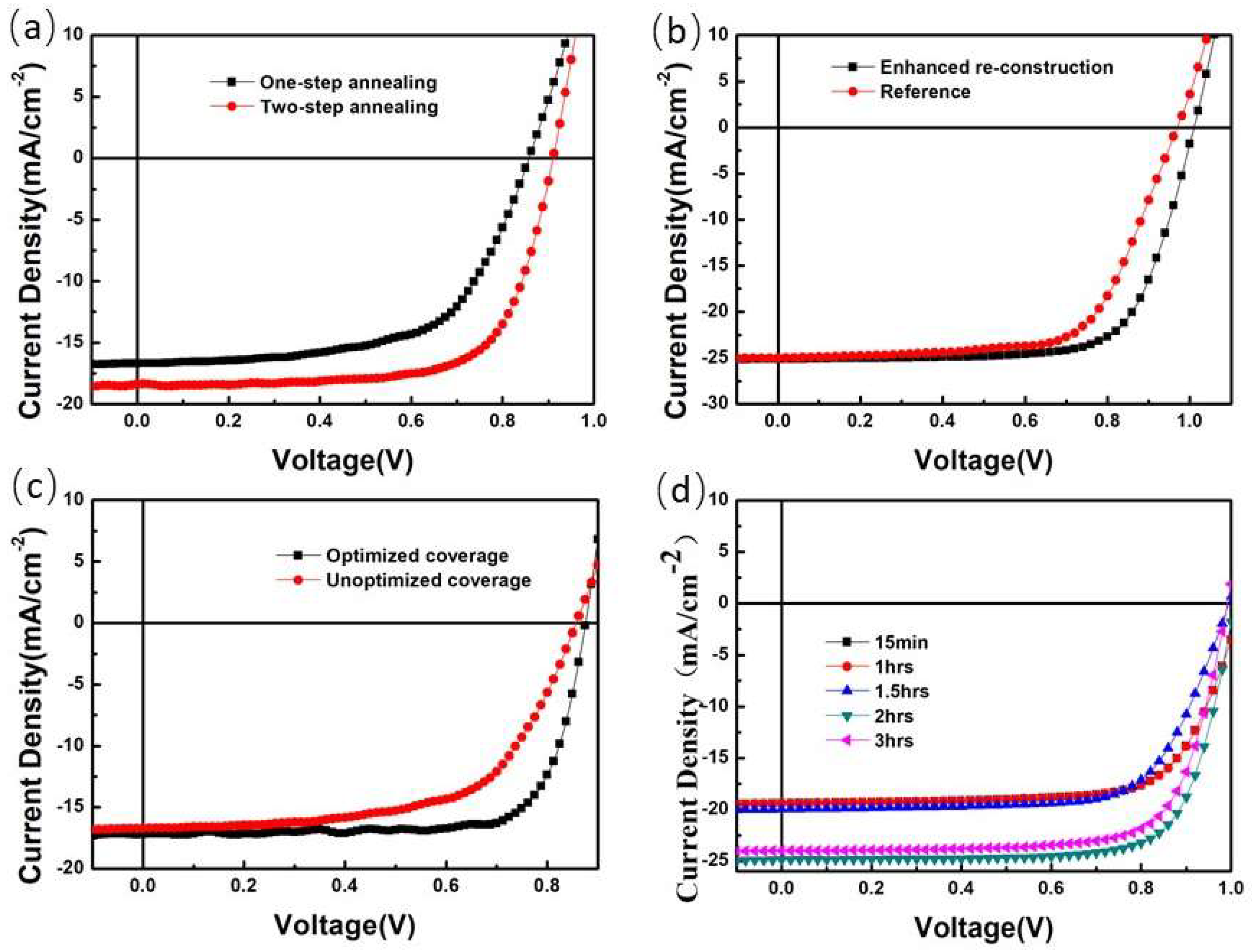
4. State and Development of Perovskite Film
5. Conclusions
Funding
Conflicts of Interest
References
- NREL Efficiency Chart. Available online: https://www.nrel.gov/pv/assets/pdfs/best-res earch-cell-efficiencies.20190802.pdf (accessed on 31 August 2019).
- Huang, H.; Wang, M.; Ding, L.; Deng, J.; Yao, X. Efficiency enhancement of the MAPbI3−xClx based perovskite solar cell by a two-step annealing procedure. Semicond. Sci. Technol. 2016, 31, 025009. [Google Scholar] [CrossRef]
- Huang, H.; Wang, M.; Ding, L.; Igbari, F.; Yao, X. Efficiency enhancement of MAPbI3−xClx based perovskite solar cell by modifying the TiO2 interface with Silver Nanowires. Sol. Energy 2016, 130, 273–280. [Google Scholar] [CrossRef]
- Huang, H.; Wang, M.; Ding, L.; Yang, Z.; Zhang, K. Hydrobromic acid assisted crystallization of MAPbI3−xClx for enhanced power conversion efficiency in perovskite solar cells. RSC Adv. 2016, 6, 55720–55725. [Google Scholar] [CrossRef]
- Huang, H.; Wang, M.; Li, J.; Yang, Z.; Yao, X. BCP influenced crystallization of MAPbI3−xClx for enhanced power conversion efficiency and stability in perovskite solar cell. Org. Electron. 2018, 52, 130–137. [Google Scholar] [CrossRef]
- Snaith, H.J. Perovskites: The emergence of a new era for low-cost, high-efficiency solar cells. J. Phys. Chem. Lett. 2013, 4, 3623–3630. [Google Scholar] [CrossRef]
- Kim, H.; Lim, K.-G.; Lee, T.-W. Planar heterojunction organometal halideperovskite solar cells: Roles of interfacial layers. Energy Environ. Sci. 2016, 9, 12–30. [Google Scholar] [CrossRef]
- Lee, J.-W.; Kim, H.-S.; Park, N.-G. Lewis acid-base adduct approach for high efficiency perovskite solar cells. Acc. Chem. Res. 2016, 49, 311–319. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Liu, C.; Du, P.; Zheng, J.; Gong, X. Bulk heterojunction perovskitehybrid solar cells with large fill factor. Energy Environ. Sci. 2015, 8, 1245–1255. [Google Scholar] [CrossRef]
- Calado, P.; Telford, A.M.; Bryant, D.; Li, X.; Nelson, J.; O’Regan, B.C.; Barnes, P.R. Evidence for ion migration in hybrid perovskite solar cells with minimal hysteresis. Nat. Commun. 2016, 7, 13831. [Google Scholar] [CrossRef]
- Im, J.-H.; Lee, C.-R.; Lee, J.-W.; Park, S.-W.; Park, N.-G. 6.5% efficient perovskite quantum-dot-sensitized solar cell. Nanoscale 2011, 3, 4088–4093. [Google Scholar] [CrossRef]
- Moon, H.G.; Shim, Y.-S.; Kim, D.H.; Jeong, H.Y.; Jeong, M.; Jung, J.Y.; Han, S.M.; Kim, J.K.; Kim, J.-S.; Park, H.-H.; et al. Self-activated ultrahigh chemosensitivity of oxide thin film nanostructures for transparent sensors. Sci. Rep. 2012, 2, 588. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.M.; Teuscher, J.; Miyasaka, T.; Murakami, T.N.; Snaith, H.J. Efficient hybrid solar cells based on meso-superstructured organometal halide perovskites. Science 2012, 338, 643–647. [Google Scholar] [CrossRef]
- Moeini, I.; Ahmadpour, M.; Mosavi, A.; Alharbi, N.; Gorji, N.E. Modeling the time-dependent characteristics of perovskite solar cells. Sol. Energy 2018, 170, 969–973. [Google Scholar] [CrossRef]
- Chen, S.; Shi, G. Two-dimensional materials for halide perovskite-based optoelectronic devices. Adv. Mater. 2017, 29, 1605448. [Google Scholar] [CrossRef]
- Huang, J.; Zhang, D.; Ding, L.; Zhang, F. Pb–Bi binary metal all-inorganic absorber layer for stability enhancement in perovskite solar cells. Adv. Mater. Interfaces 2019, 6, 1900517. [Google Scholar] [CrossRef]
- Zhou, H.; Chen, Q.; Li, G.; Luo, S.; Song, T.-B.; Duan, H.-S.; Hong, Z.; You, J.; Liu, Y.; Yang, Y. Interface engineering of highly efficient perovskite solar cells. Science 2014, 345, 542–546. [Google Scholar] [CrossRef]
- Eperon, G.E.; Burlakov, V.M.; Docampo, P.; Goriely, A.; Snaith, H.J. Morphological control for high performance, solution-processed planar heterojunction perovskite solar cells. Adv. Funct. Mater. 2014, 24, 151–157. [Google Scholar] [CrossRef]
- Ummadisingu, A.; Steier, L.; Seo, J.-Y.; Matsui, T.; Abate, A.; Tress, W.; Grätzel, M. The effect of illumination on the formation of metal halide perovskite films. Nature 2017, 545, 208–212. [Google Scholar] [CrossRef] [PubMed]
- Huang, F.; Dkhissi, Y.; Huang, W.; Xiao, M.; Benesperi, I.; Rubanov, S.; Zhu, Y.; Lin, X.; Jiang, L.; Zhou, Y.; et al. Gas-assisted preparation of lead iodide perovskite films consisting of a monolayer of single crystalline grains for high efficiency planar solar cells. Nano Energy 2014, 10, 10–18. [Google Scholar] [CrossRef]
- Jeon, N.J.; Noh, J.H.; Kim, Y.C.; Yang, W.S.; Ryu, S.; Seok, S.I. Solvent engineering for high-performance inorganic-organic hybrid perovskite solar cells. Nat. Mater. 2014, 13, 897–903. [Google Scholar] [CrossRef] [PubMed]
- Yin, X.; Chen, P.; Que, M.; Xing, Y.; Que, W.; Niu, C.; Shao, J. Highly efficient flexible perovskite solar cells using solution-derived NiOx hole contacts. ACS Nano. 2016, 10, 3630–3636. [Google Scholar] [CrossRef] [PubMed]
- Yin, X.; Que, M.; Xing, Y.; Liu, X.; Que, W.; Niu, C. Solution-induced morphology change of organic-inorganic hybrid perovskite films for high efficiency inverted planar heterojunction solar cells. Electrochim. Acta. 2016, 191, 750–757. [Google Scholar] [CrossRef]
- Li, G.; Zhang, T.; Zhao, Y. Hydrochloric acid accelerated formation of planar CH3NH3PbI3 perovskite with high humidity tolerance. J. Mater. Chem. A 2015, 3, 19674–19678. [Google Scholar] [CrossRef]
- Wang, F.; Yu, H.; Xu, H.; Zhao, N. HPbI3: A New Precursor Compound for Highly Efficient Solution-Processed Perovskite Solar Cells. Adv. Funct. Mater. 2015, 25, 1120–1126. [Google Scholar] [CrossRef]
- Chen, C.-C.; Hong, Z.; Li, G.; Chen, Q.; Zhou, H.; Yang, Y. One-step, low-temperature deposited perovskite solar cell utilizing small molecule additive. J. Photonics Energy 2015, 5, 057405. [Google Scholar] [CrossRef]
- Zhang, W.; Pathak, S.; Sakai, N.; Stergiopoulos, T.; Nayak, P.K.; Noel, N.K.; Haghighirad, A.A.; Burlakov, V.M.; deQuilettes, D.W.; Sadhanala, A.; et al. Enhanced optoelectronic quality of perovskite thin films with hypophosphorous acid for planar heterojunction solar cells. Nat. Commun. 2015, 6, 10030. [Google Scholar] [CrossRef] [PubMed]
- Liang, P.-W.; Liao, C.-Y.; Chueh, C.-C.; Zuo, F.; Williams, S.T.; Xin, X.-K.; Lin, J.; Jen, A.K.-Y. Additive enhanced crystallization of solution-processed perovskite for highly efficient planar-heterojunction solar cells. Adv. Mater. 2014, 26, 3748–3754. [Google Scholar] [CrossRef]
- Chang, C.-Y.; Chu, C.-Y.; Huang, Y.-C.; Huang, C.-W.; Chang, S.-Y.; Chen, C.-A.; Chao, C.-Y.; Su, W.-F. Tuning perovskite morphology by polymer additive for high efficiency solar cell. ACS Appl. Mater. Interfaces 2015, 7, 4955–4961. [Google Scholar] [CrossRef]
- Chiang, C.-H.; Wu, C.-G. Bulk heterojunction perovskite-PCBM solar cellswith high fill factor. Nat. Photonics 2016, 10, 196–201. [Google Scholar] [CrossRef]
- Song, X.; Wang, W.; Sun, P.; Ma, W.; Chen, Z.-K. Additive to regulate the perovskite crystal film growth in planar heterojunction solar cells. Appl. Phys. Lett. 2015, 106, 033901. [Google Scholar] [CrossRef]
- Zhao, Y.; Zhu, K. CH3NH3Cl-assisted one-step solution growth of CH3NH3PbI3: Structure, charge-carrier dynamics, and photovoltaic properties of perovskite solar cells. J. Phys. Chem. C 2014, 118, 9412–9418. [Google Scholar] [CrossRef]
- Wu, W.-Q.; Wang, Q.; Fang, Y.; Shao, Y.; Tang, S.; Deng, Y.; Lu, H.; Liu, Y.; Li, T.; Yang, Z.; et al. Molecular doping enabled scalable blading of efficient hole-transport-layer-free perovskite solar cells. Nat. Commun. 2018, 9, 1625. [Google Scholar] [CrossRef] [PubMed]
- Bi, C.; Shao, Y.; Yuan, Y.; Xiao, Z.; Wang, C.; Gao, Y.; Huang, J. Understanding the formation and evolution of interdiffusion grown organolead halide perovskite thin films by thermal annealing. J. Mater. Chem. A. 2014, 2, 18508–18514. [Google Scholar] [CrossRef]
- Leguy, A.M.A.; Hu, Y.; Campoy-Quiles, M.; Alonso, M.I.; Weber, O.J.; Azarhoosh, P.; van Schilfgaarde, M.; Weller, M.T.; Bein, T.; Nelson, J.; et al. Reversible hydration of CH3NH3PbI3 in Films, single crystals, and solar cells. Chem. Mater. 2015, 27, 3397–3407. [Google Scholar] [CrossRef]
- Liu, Y.; Zhang, Y.; Yang, Z.; Feng, J.; Xu, Z.; Li, Q.; Hu, M.; Ye, H.; Zhao, X.; Liu, M.; et al. Low-temperature-gradient crystallization for multi-inch high-quality perovskite single crystals for record performance photodetectors. Mater. Today 2019, 22, 67–75. [Google Scholar] [CrossRef]
- Zhang, J.; Bai, D.; Jin, Z.; Bian, H.; Wang, K.; Sun, J.; Wang, Q.; Liu, S. Solar cells: 3D-2D-0D Interface profling for record effciency all-inorganic CsPbBrI2 perovskite solar cells with superior stability. Adv. Energy Mater. 2018, 8, 1870067. [Google Scholar] [CrossRef]

| Additives | Information of Cell (Active Area/Structure) | Device Parameters | Efficiency (%) | Material | References/Year | |||
|---|---|---|---|---|---|---|---|---|
| Voc (V) | Fill Factor | Jsc (mA cm−2) | ||||||
| Assisted crystallization | HBr | 0.1 cm2/Planar | 0.94 | 0.77 | 21.71 | 15.76 | MAPbI3−xClx | [4]/2016 |
| HI | –/Planar | 0.95 | 0.65 | 23.8 | 15.4 | FAPbI3 | [25]/2015 | |
| HCl | 0.15 cm2/Planar | 1.02 | 0.70 | 20.08 | 14.28 | MAPbI3 | [24]/2015 | |
| H2O | 0.08 cm2/Planar | 0.95 | 0.78 | 21.67 | 16.06 | MAPbI3−xClx | [27]/2015 | |
| BmPyPhB | 0.1 cm2/Planar | 0.88 | 0.63 | 14.7 | 8.14 | MAPbI3 | [26]/2015 | |
| Lewis acid | 0.12 cm2/Planar | 1.03 | 0.66 | 19.42 | 13.2 | MAPbI3 | [14]/2016 | |
| DIO | 3.14 mm2/Planar | 0.92 | 0.73 | 17.5 | 11.8 | MAPbI3−xClx | [28]/2014 | |
| PEG | 0.09 cm2/Planar | 0.94 | 0.71 | 19.53 | 13.2 | MAPbI3−xClx | [29]/2015 | |
| Composite film | PCBM | –/Planar | 0.97 | 0.82 | 20.2 | 16 | MAPbI3 | [30]/2016 |
| CN | –/Planar | 0.85 | 0.64 | 16.7 | 8.9 | MAPbI3−xClx | [31]/2015 | |
| BCP | 0.1 cm2/Planar | 1.03 | 0.73 | 21.83 | 16.25 | MAPbI3−xClx | [5]/2016 | |
| A10C60 | 0.045 cm2/Planar | 0.88 | 0.83 | 19.54 | 14.34 | MAPbI3−xClx | [15]/2015 | |
| MACl | –/Planar | 1.02 | 0.58 | 20.36 | 12.1 | MAPbI3 | [32]/2015 | |
| MAI | 0.1 cm2/Planar | 0.91 | 0.71 | 19.7 | 12.72 | MAPbI3−xClx | [2]/2015 | |
| F4TCNQ | 0.08 cm2/Planar | 1.10 | 0.81 | 22.7 | 20.2 | MAPbI3 | [33]/2018 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, J.; Gao, C.; Zhang, D.; Tian, Q.; Zhang, F.; Liu, S. Influence of Film Quality on Power Conversion Efficiency in Perovskite Solar Cells. Coatings 2019, 9, 622. https://doi.org/10.3390/coatings9100622
Huang J, Gao C, Zhang D, Tian Q, Zhang F, Liu S. Influence of Film Quality on Power Conversion Efficiency in Perovskite Solar Cells. Coatings. 2019; 9(10):622. https://doi.org/10.3390/coatings9100622
Chicago/Turabian StyleHuang, Jin, Changji Gao, Dan Zhang, Qingwen Tian, Fanghui Zhang, and Shengzhong (Frank) Liu. 2019. "Influence of Film Quality on Power Conversion Efficiency in Perovskite Solar Cells" Coatings 9, no. 10: 622. https://doi.org/10.3390/coatings9100622
APA StyleHuang, J., Gao, C., Zhang, D., Tian, Q., Zhang, F., & Liu, S. (2019). Influence of Film Quality on Power Conversion Efficiency in Perovskite Solar Cells. Coatings, 9(10), 622. https://doi.org/10.3390/coatings9100622




