1. Introduction
Flake aluminum powders offer high spectral reflectance, good electric conductivity, excellent shielding capacity, and low cost. They are widely used in automobile metallized coatings, energy saving coatings, electromagnetic shielding coatings and infrared stealth coatings [
1,
2,
3,
4,
5,
6]. For the infrared stealth material, high concentrations of flakey aluminum powders can reduce infrared emissivity and restrain infrared radiation, but its high lightness and electrical conductivity challenge optics and radar stealth [
7,
8,
9]. To overcome the multispectral stealth problem, we previously reported a bimetallic composite material with a core shell structure by wrapping a thin magnetic Ni shell on the surface of flake Al powders [
9,
10]. The prepared Al/Ni core-shell pigments may combine the properties of the core and the shell materials to offer particular new functions.
Previous studies have shown that the optical, electrical, and magnetic properties of core-shell powders are associated with the relative content, thickness, and surface morphology of the shell layer. This can be influenced by the preparation process and method [
10,
11,
12,
13]. The galvanic displacement method is an effective method to prepare the bimetallic core-shell powder with controllability, high cladding rates, low costs, and simple processing [
14,
15,
16]. This method allows low activity Ni
2+ ions to be displaced by a highly active Al core at the solid-liquid interface. This ensures that the generated metal Ni is preferably deposited on the surface of the Al core rather than forming separate Ni particles. Thus, the quality and cladding rate of Al/Ni core-shell particles can be improved significantly.
However, the covering effect and morphology of Ni shell is significantly affected by the rate of the galvanic displacement reaction [
17,
18] to ultimately influence the properties of the Al/Ni core-shell powders. Here, the rate of the galvanic displacement reaction was controlled by changing the reactant concentration and order of addition. The effect of processing parameters on phase structure, morphology, optical, and antioxidant properties of Al/Ni core-shell pigments were investigated systematically.
2. Materials and Methods
2.1. Materials
Micrometer-scale flake aluminium pigments (particle size: ~20 μm) were procured from Toyo Aluminium K. K. (Zhaoqing, China). Nickel chloride (NiCl2), ammonium fluoride (NH4F), and ethanol were obtained from Sinopharm Chemical Reagent Co., Ltd. (Shanghai, China). All chemicals were A.R. grade.
2.2. Synthesis of Al/Ni Composite Material
A detailed galvanic displacement method has been reported in our previous work [
10]. In a typical synthesis, 2 g of flake Al powder was added to 200 mL of ethanol solution in a 500 mL conical flask and dispersed by ultrasonic vibration for 30 min. Then, 1.12 g of NH
4F was dissolved into 50 mL of a mixed solution of ethanol and distilled water with a volume ratio of 1:1 (this was Solution A). Next, NiCl
2 (2.11–5.28 g) was dissolved into 60 mL of ethanol solution to obtain Solution B.
To understand the mechanism of metallic Ni depositing on the surface of the flake Al core, two different preparation processes have been designed by changing the addition sequence of Solution A and Solution B. In this detailed processing step, Solution A (or Solution B) was quickly injected into the conical flask. After stirring for 30 min, Solution B (or Solution A) was added dropwise at 1 mL/min with a micro syringe pump under magnetic stirring (350 r/min) at 40 °C. The color of the powders varied from brilliant silver to dark gray after a few minutes. After another hour, the products were separated by centrifugation and washed with ethanol and distilled water several times and then dried at 80 °C for 12 h. This yielded black-gray Al/Ni core-shell pigments.
According to the above order of addition, the prepared samples were labeled as “NH4F→NiCl2” if Solution A was added first followed by dropwise addition of Solution B. Conversely, the samples were classified as “NiCl2→NH4F”.
2.3. Characterization
The samples were characterized by X-ray diffraction (XRD, D2 PHASER with Cu Kα radiation, Bruker, Karlsruhe, Germany) and field emission scanning electron microscopy (FESEM, QUANTA 250, Thermo Fisher Scientific, Waltham, MA, USA). Thermogravimetry and differential scanning calorimetry (TG-DSC) analysis was performed using a STA 449F3 instrument (NETZSCH, Selb, Germany) under air atmosphere from 30 to 780 °C at 10 °C/min. The specific surface area was measured via specific surface area analyzer (BET, ASAP 2020HD88, Micromeritics Instrument Ltd., Atlanta, GA, USA).
For optical characterization, paints containing Al/Ni composite pigments were prepared by mixing Al/Ni powder 16 wt. %, epoxy resin 12.5 wt. % and thinner 62.5 wt. %. Then the tested coating samples were painted by these mixtures onto microslides and cured at 60 °C for 12 h. The VIS/NIR reflection spectrum (380–2500 nm) was measured with a UV/VIS/NIR spectrophotometer (Lambda 750, PerkineElmer, Waltham, MA, USA). The CIE LAB color data (L*) were calculated from the visible light reflection spectrum by Color CIE software (PerkineElmer, CIE D65 photo source, and 10° observation angle; the calculated spectrum range was 400–700 nm). The infrared reflectance spectrum was measured with a Fourier transform infrared spectrometer (Tensor27, Bruker, Karlsruhe, Germany) with an integrating sphere attachment.
3. Results and Discussion
Figure 1 presents XRD patterns of the Al/Ni products obtained at different preparation processes and different Ni
2+:Al molar ratios. Only the Al phase (JCPDS, file No. 04-0787) and trace amounts of the Ni phase (JCPDS, file No. 04-0850) were obtained in the XRD spectrum at a Ni
2+:Al of 0.1:1. The intensity of the Ni peaks gradually strengthens as the molar ratio of Ni
2+: Al increases. This illustrates that Ni
2+ ions are successfully replaced by Al substances via the galvanic displacement reaction without any other reaction between Al and Ni. The high molar ratio of Ni
2+:Al can lead to more metallic Ni being generated on the surface of flake Al powders. This increases the relative Ni content in the shell.
Figure 1 shows that the Ni peak intensity of “NiCl
2→NH
4F” samples are higher than that of “NH
4F→NiCl
2” samples even with the same reaction concentration of Ni
2+. We concluded that the relative content of the Ni shell is highly dependent on the preparation process because the addition sequence of reactants, such as NH
4F and NiCl
2, would affect the rate of displacement.
For the reaction process of “NH
4F→NiCl
2” samples, the surface oxide layer of flake Al core is first removed by a high concentration of NH
4F solution via the following reaction:
Upon slow addition of NiCl
2 solution, the displacement reaction can be performed on all of the bare surfaces of the Al particles. The overall displacement reaction equation is:
However, the newly formed Ni layer insulates the Al core from the residual NiCl2 solution and obstructs the subsequent displacement reaction. If the concentration of Ni2+ is relatively low in the reaction system, then the slow displacement reaction rate inhibits the growth rate of the shell layer, and this leads to a low content of Ni in the shell.
In contrast, for the preparation of “NiCl2→NH4F” samples, while the flake Al particles are immersed in a high concentration of NiCl2, the displacement reaction is hampered by the Al2O3 layer. Once the NH4F solution is added dropwise, the Al2O3 layer is gradually dissolved, and the exposed surface of the Al substrate can react with Ni2+ ions to quickly form a Ni shell. The displacement reaction rate is accelerated by the high concentration of Ni2+, and the reaction time is extended due to the low removal rate of the Al2O3 layer. Thus, more metallic Ni can be deposited on the surface of flake Al powder.
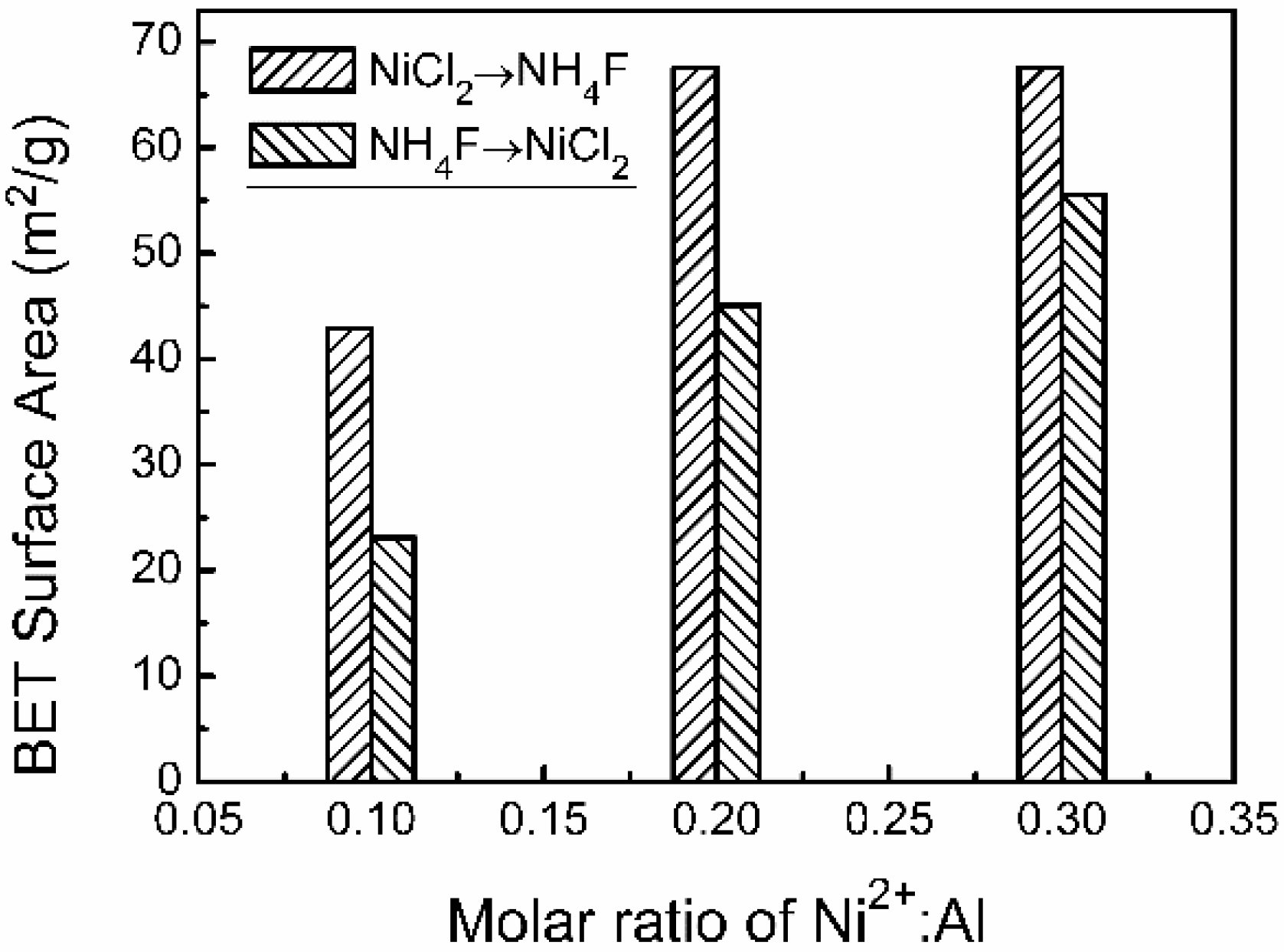
Figure 2 shows SEM images of the as-prepared Al/Ni composite powders obtained under different preparation process and Ni
2+:Al molar ratios. The surface of raw Al powders (
Figure 2a) is smooth and flat. After the galvanic displacement process, a nickel shell is formed on the Al substrate and the surface becomes rougher.
Figure 2b shows that a thin Ni layer is formed, and a small amount of nanoparticle is distributed on the surface of the composite particles. Compared with the sample in
Figure 2b, the Ni shell of the “NiCl
2→NH
4F” samples (
Figure 2e) becomes a granular film, and the surface contains some Ni nanoparticles when the ratio of Ni
2+:Al is 0.1:1. The surface roughness of the as-prepared compound particles is obviously enhanced. This results in a significant increase in the BET surface area from 24.1 to 42.9 m
2/g. It verifies that the surface morphology of the Ni shells has a corresponding change as a function of NH
4F and NiCl
2 addition order.
As shown in
Figure 2c, the quantity and size of the Ni nanoparticles on the surface of Ni shell gradually increases as the molar ratio of Ni
2+:Al increases—in particular, their shapes transform from nearly spherical into long graininess (
Figure 2c). These changes lead the BET surface area of the Al/Ni sample to reach 45.1 m
2/g (
Figure 3). Continued increases in the molar ratio of Ni
2+:Al to 0.3:1 (
Figure 2d) results in Ni particles that develop a flaky morphology. The transverse size reaches the microns scales although the number decreases. Meanwhile, the BET surface area increases to 55.5 m
2/g.
However, for the “NiCl
2→NH
4F” samples, the size of Ni particle remains constant with increasing Ni
2+ concentrations, but the particle number increases significantly (
Figure 2f). When the rate of Ni
2+:Al increased to 0.3:1 (
Figure 2g), it causes serious aggregation of Ni nanoparticles, and a small number of cubical particles are observed on the sample surface. However, the BET surface area is barely affected by the morphological change of the Ni shell.
The morphology of the Ni shell is strongly affected by the preparation conditions (Ni
2+:Al ratio and the addition sequence of reactant). This is because these changes the reactant concentration of Ni
2+. It is easier to form a relatively smooth and thin Ni shell through the heterogeneous nucleation at low Ni
2+ concentrations. However, the displacement reaction rate is accelerated with an increasing in the concentration of reactants. This increases the nucleation amount and inhibits growth of the Ni particles [
17].
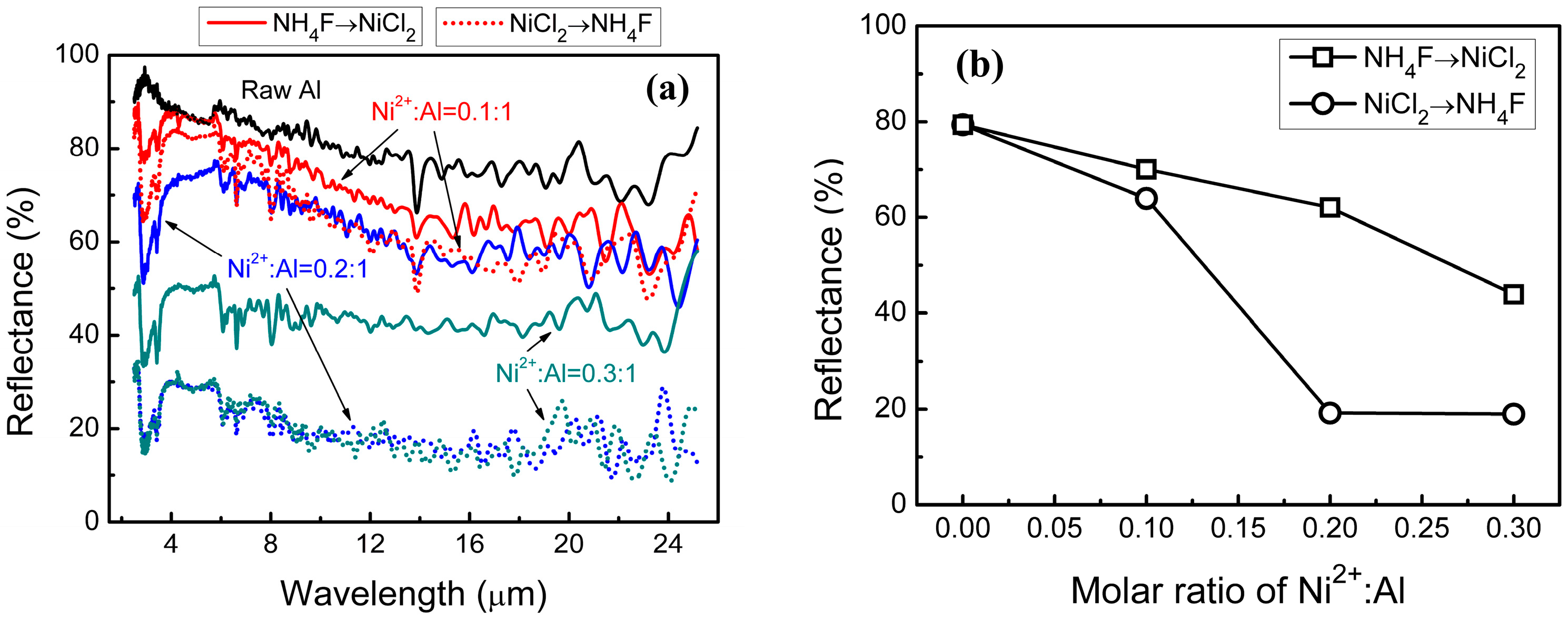
In order to research the influence of the Ni shell on the spectral reflectivity of flake Al powders, the VIS-NIR diffuse reflectance spectra of Al/Ni core-shell samples and raw Al powders were measured. Uncoated raw Al powders have extremely high reflectance value in both VIS (400–800 nm) and NIR (0.8–2.5 μm) wavebands (
Figure 4). The spectral reflectance is remarkably decreased by coating with the Ni shell on the surface of the flake Al pigments.
Regardless of the preparation process, the reflectance of Al/Ni samples declines gradually with the increasing Ni2+:Al; notably, the degree of decline is different. The “NH4F→NiCl2” samples at Ni2+:Al = 0.1:1 have a VIS and NIR reflectance about 29% and 25% lower, respectively. The VIS and NIR reflectance of “NiCl2→NH4F” samples reduces by 35% and 29%, respectively. Furthermore, the influence on the spectral reflectance is further exacerbated at higher ratios of Ni2+:Al.
According to our previous research results [
10,
11,
12], the incidence visible and infrared light is difficult to penetrate the Ni shell because of the low spectral transmittance of the metallic Ni in the VIS and NIR wavebands. This makes the reflectance of the Al/Ni core-shell samples more susceptible to the surface morphology of the Ni shell. The rough surface causes intense scatter of the incident rays and excellent extinction coefficients at VIS and NIR wavelengths. A stronger extinction ability can be obtained with a higher Ni shell roughness.
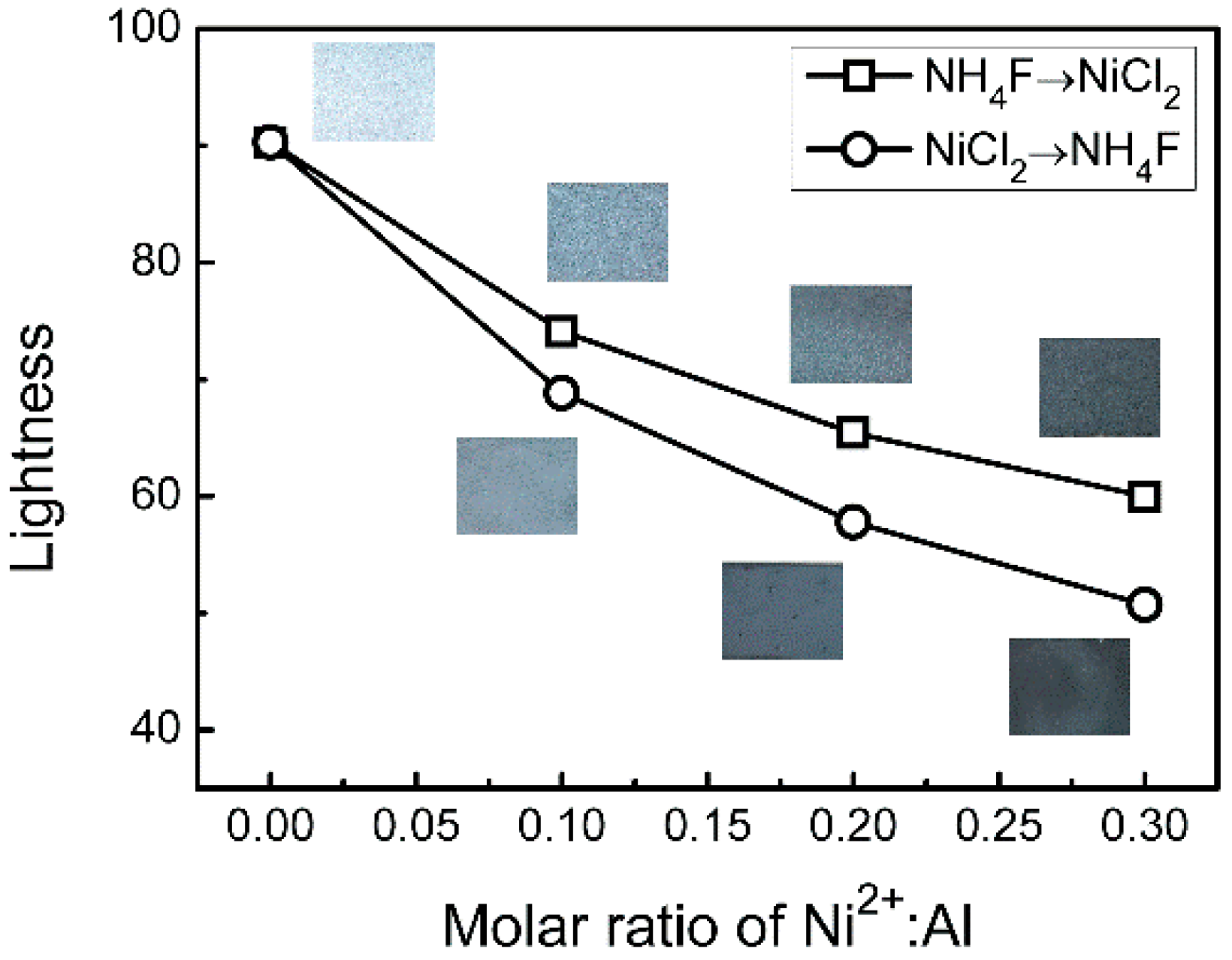
As shown in
Figure 5, the lightness value
L* of flake Al pigment can be significantly reduced via the cover treatment due to the close relationships between VIS reflectance and lightness. When the surface is only coated by the small number of metallic Ni (Ni
2+:Al = 0.1:1), the color of the flake pigments changes from brilliant silver to pale gray, and the lightness value
L* decreases by 16.2 and 21.4, respectively. The lightness declines gradually with increasing Ni
2+ content. If the ratio of Ni
2+:Al increases to 0.3:1, then the lightness decreases by 30.3 and 39.6, respectively, and the color becomes black gray.
The infrared reflectance spectra of coating samples prepared by raw Al and Al/Ni pigments are shown in
Figure 6. The reflectance between infrared reflectance spectra and VIS/NIR diffuse reflectance spectra are not approximately equal at λ = 2500 nm, which can be attributed to different test principle and instrumental error. Some characteristic infrared adsorption bands can be observed in the reflectance spectra. The absorption peaks at 2.85, 3.4, 6, 6.6 and 8 μm are attributed to the infrared spectra characteristics of the epoxy resin in the coating samples. It is similar to the VIS-NIR reflectance spectra that the infrared reflectance spectra can also be affected by the ratio of Ni
2+:Al and the preparation process. The reflectance gradually decreases with increasing Ni
2+ concentration—especially for coating samples consisting of “NiCl
2→NH
4F” pigments. Upon comparing
Figure 3 and
Figure 6b, we see that the variation in infrared reflectance is consistent with the BET surface area of the Al/Ni samples. The BET surface area data are directly related to the surface roughness, and the results show that the infrared reflectance is mainly influenced by the light scattering and absorption of the rough Ni shell.
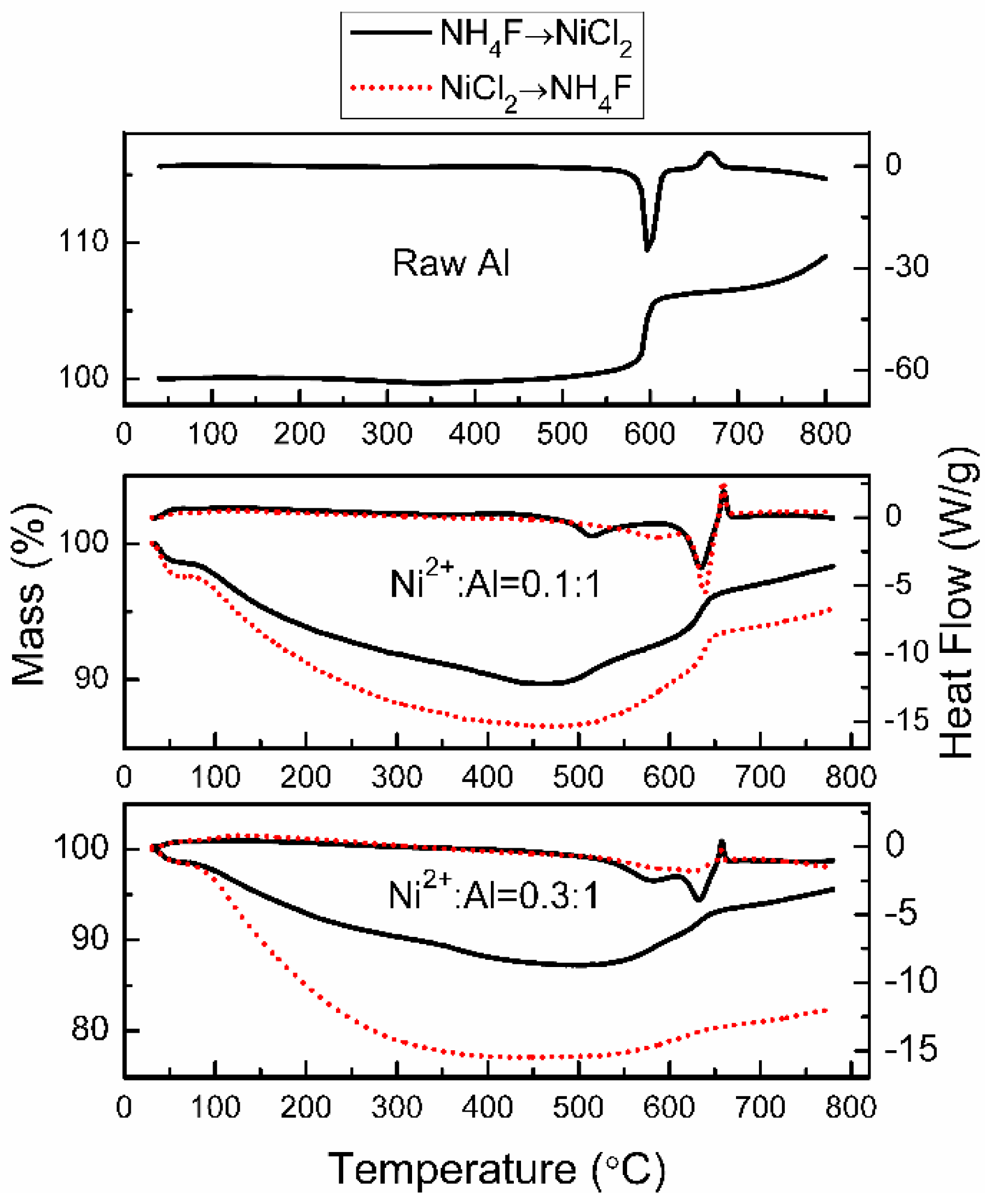
Figure 7 shows the thermal analysis curves of raw Al and Al/Ni products. TG analysis illustrates the weight of raw Al powders remains constant from 40 to 500 °C, but the weight rises sharply by about 5.2% when the temperature increases from 570 to 620 °C. DSC cure shows that there is a sharp exothermic peak at 596 °C due to the oxidation of aluminum. There is also a weak decalescence peak at 674 °C attributed to the crystalline phase transition of aluminum oxide [
10].
In contrast, the weight of the Al/Ni core-shell samples consistently decrease with increasing temperature from 40 to 490 °C because of the loss of water molecules and thermal decomposition of organic residues. When the temperature exceeds 500 °C, the mass of the Al/Ni samples starts to rise gradually—this eventually leads to a drastic increase at 630 °C. However, as the concentration of Ni2+ increases, this phenomenon gradually declines because of the increased thickness of Ni layer. The thick Ni shell helps to prevent Al core contact with the oxygen and protects it from high temperature oxidation.
In addition, the exothermic peak can be observed at about 630 °C from the DSC data. Compared with the raw flake Al powder, the oxidation temperature of the Al/Ni samples is delayed by 34 °C. This proves that the Ni shell has been successfully formed on the surface of flake Al powders via the galvanic displacement method. The compact Ni shell can prevent oxidation of flake aluminum core. Thus, the oxidation reaction is delayed. In contrast, the antioxidant property of Al core improves as the thickness of the Ni shell increased. The “NH4F→NiCl2” sample at Ni2+:Al = 0.3:1 has only a weak exothermic peak in the DSC curve. The antioxidant ability of the Al/Ni core-shell pigment is strongly affected by the content and surface morphology of the Ni shell. This is related to the preparation approach.