Recent Development in Phosphonic Acid-Based Organic Coatings on Aluminum
Abstract
:1. Introduction
2. Characterization of the Phosphonic Acid-Modified Aluminum Surfaces
2.1. Surface Morphology
2.2. Presence, Composition, and Stability of Layer
2.3. Orientation of Molecules and Binding Mode to Aluminum
3. Corrosion Evaluation Methods
3.1. Electrochemical Measurements
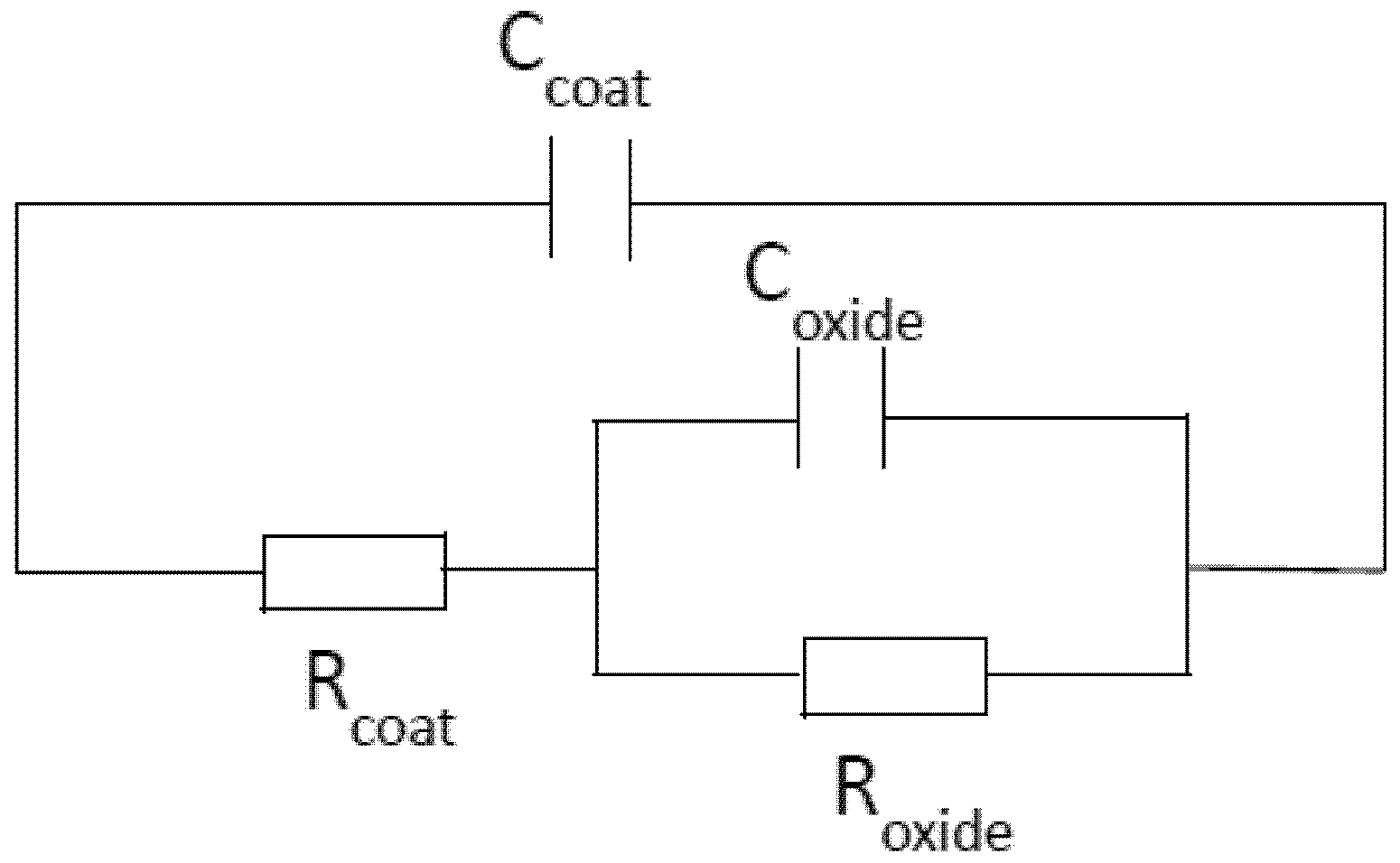
3.1.1. Electrochemical Impedance Spectroscopy (EIS)
3.1.2. Electrochemical Noise Method (ENM)
3.1.3. Potentiodynamic Polarization Measurements
3.1.4. Localized Electrochemical Techniques
3.2. Conventional Measurements
3.2.1. Immersion Test
3.2.2. Salt Spray Test
3.3. Spectral Analysis
3.4. Surface Analysis
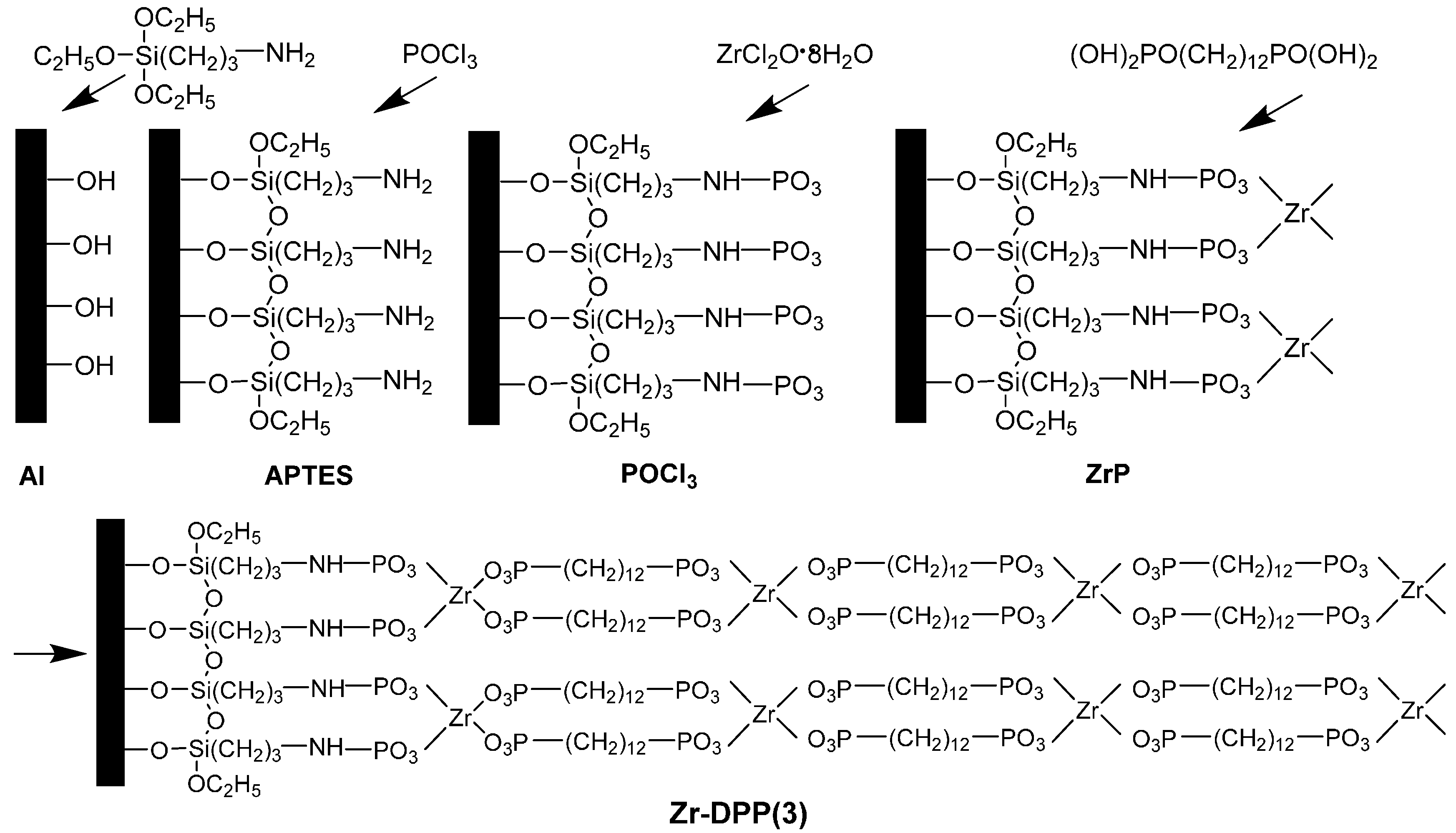
4. Aluminum Surface Treatments with Phosphonic Acids
4.1. Phosphonic Acids Used as Pre-Coating
4.1.1. Phenylphosphonic Acid (PPA)
4.1.2. Vinylphosphonic Acid (VPA)
4.1.3. 1,12-Dodecyldiphosphonic Acid (DDP)
4.1.4. Amino Trimethylene Phosphonic Acid (ATMP)
4.1.5. Ethylenediamine Tetra Methylene Phosphonic Acid (EDTPO)
4.1.6. 1,2-Diaminoethanetetrakis-Methylenephosphonic Acid (DETAPO)
4.1.7. (12-Ethylamino-Dodecyl)-Phosphonic Acid
4.1.8. Aminopropyl Phosphonate (APP)
4.1.9. ω-(3-Phenylpyrrol-1-ylalkyl) Phosphonic Acid (Cn-Ph-P)
4.1.10. ω-(2,5-Dithienylpyrrol-1-yl-alkyl) Phosphonic Acid (SNS-n-P)
4.1.11. (1H,1H, 2H,2H-Heptadecafluorodec-1-yl) Phosphonic Acid (HDF-PA)
4.1.12. Poly (Vinyl Phosphonic Acid) (PvPA)
4.2. Phosphates Used as Pre-Coating
4.2.1. Monostearyl Acid Phosphate (MSAP)
4.2.2. Phosphoric Acid Mono-(12-Hydroxy-Dodecyl) Ester
4.2.3. Fluoro Alkyl Phosphates (Zonyl UR)
4.3. Coatings Dissolved in Paints that Contain Phosphonic Acids or Phosphates
4.3.1. Phenylphosphonic Acid (PPA)
4.3.2. 2-(Phosphonooxy) Benzoic Acid (Fosfosal)
4.3.3. Phosphonosuccinic Acid (PPSA)
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Twite, R.L.; Bierwagen, G.P. Review of alternatives to chromate for corrosion protection of aluminum aerospace alloys. Prog. Org. Coat. 1998, 33, 91–100. [Google Scholar] [CrossRef]
- Liu, Y.; Sun, D.; You, H.; Chung, J.S. Corrosion resistance properties of organic–inorganic hybrid coatings on 2024 aluminum alloy. Appl. Surf. Sci. 2005, 246, 82–89. [Google Scholar] [CrossRef]
- Bulusu, A.; Paniagua, S.A.; MacLeod, B.A.; Sigdel, A.K.; Berry, J.J.; Olson, D.C.; Marder, S.R.; Graham, S. Efficient modification of metal oxide surfaces with phosphonic acids by spray coating. Langmuir 2013, 29, 3935–3942. [Google Scholar] [CrossRef] [PubMed]
- Queffélec, C.; Petit, M.; Janvier, P.; Knight, D.A.; Bujoli, B. Surface modification using phosphonic acids and esters. Chem. Rev. 2012, 112, 3777–3807. [Google Scholar] [CrossRef] [PubMed]
- Pellerite, M.J.; Dunbar, T.D.; Boardman, L.D.; Wood, E.J. Effects of fluorination on self-assembled monolayer formation from alkanephosphonic acids on aluminum: Kinetics and structure. J. Phys. Chem. B 2003, 107, 11726–11736. [Google Scholar] [CrossRef]
- Attavar, S.; Diwekar, M.; Linford, M.R.; Davis, M.A.; Blair, S. Passivation of aluminum with alkyl phosphonic acids for biochip applications. Appl. Surf. Sci. 2010, 256, 7146–7150. [Google Scholar] [CrossRef]
- Hoque, E.; DeRose, J.A.; Kulik, G.; Hoffmann, P.; Mathieu, H.J.; Bhushan, B. Alkylphosphonate modified aluminum oxide surfaces. J. Phys. Chem. B 2006, 110, 10855–10861. [Google Scholar] [CrossRef] [PubMed]
- Shida, A.; Sugimura, H.; Futsuhara, M.; Takai, O. Zirconium-phosphate films self-assembled on aluminum substrate toward corrosion protection. Surf. Coat. Technol. 2003, 169–170, 686–690. [Google Scholar] [CrossRef]
- Liu, X.F. Filiform corrosion attack on pretreated aluminum alloy with tailored surface of epoxy coating. Corros. Sci. 2007, 49, 3494–3513. [Google Scholar] [CrossRef]
- Jaehne, E.; Oberoi, S.; Adler, H.-J.P. Ultra thin layers as new concepts for corrosion inhibition and adhesion promotion. Prog. Org. Coat. 2008, 61, 211–223. [Google Scholar] [CrossRef]
- He, L.; Zhao, Y.; Xing, L.; Liu, P.; Wang, Z.; Zhang, Y.; Liu, X. Preparation of phosphonic acid functionalized graphene oxide-modified aluminum powder with enhanced anticorrosive properties. Appl. Surf. Sci. 2017, 411, 235–239. [Google Scholar] [CrossRef]
- Shepard, M.J.; Comer, J.R.; Young, T.L.; McNatt, J.S.; Espe, M.P.; Ramsier, R.D.; Robinson, T.R.; Nelson, L.Y. Organophosphate Adsorption on Metal Oxide Surfaces; VSP BV: Oud-Beijerland, The Netherlands, 2004; pp. 225–239. [Google Scholar]
- Ulman, A. Formation and structure of self-assembled monolayers. Chem. Rev. 1996, 96, 1533–1554. [Google Scholar] [CrossRef] [PubMed]
- Whitten, M.C.; Burke, V.J.; Neuder, H.A.; Lin, C.-T. Simultaneous acid catalysis and in situ phosphatization using a polyester-melamine paint: A surface phosphatization study. Ind. Eng. Chem. Res. 2003, 42, 3671–3679. [Google Scholar] [CrossRef]
- Dalmoro, V.; Santos, J.H.Z.; Azambuja, D.S. Corrosion behavior of AA2024-T3 alloy treated with phosphonate-containing TEOS. J. Solid State Electrochem. 2012, 16, 403–414. [Google Scholar] [CrossRef]
- Jalili, M.; Rostami, M.; Ramezanzadeh, B. Surface modification of aluminum flakes with amino trimethylene phosphonic acid: Studying the surface characteristics and corrosion behavior of the pigment in the epoxy coating. Corrosion 2015, 71, 628–640. [Google Scholar] [CrossRef]
- Dalmoro, V.; Aleman, C.; Ferreira, C.A.; dos Santos, J.H.Z.; Azambuja, D.S.; Armelin, E. The influence of organophosphonic acid and conducting polymer on the adhesion and protection of epoxy coating on aluminium alloy. Prog. Org. Coat. 2015, 88, 181–190. [Google Scholar] [CrossRef]
- Blajiev, O.L.; Ithurbide, A.; Hubin, A.; Van Haesendonck, C.; Terryn, H. XPS study of the assembling morphology of 3-hydroxy-3-phosphono-butyric acid tert-butyl ester on variously pretreated Al surfaces. Prog. Org. Coat. 2008, 63, 272–281. [Google Scholar] [CrossRef]
- Lee, J.; Bong, J.; Ha, Y.-G.; Park, S.; Ju, S. Durability of self-assembled monolayers on aluminum oxide surface for determining surface wettability. Appl. Surf. Sci. 2015, 330, 445–448. [Google Scholar] [CrossRef]
- Davies, P.R.; Newton, N.G. The chemisorption of organophosphorus compounds at an Al (111) surface. Appl. Surf. Sci. 2001, 181, 296–306. [Google Scholar] [CrossRef]
- Hauffman, T.; Hubin, A.; Terryn, H. Study of the self-assembling of n-octylphosphonic acid layers on aluminum oxide from ethanolic solutions. Surf. Interface Anal. 2013, 45, 1435–1440. [Google Scholar] [CrossRef]
- Sheffer, M.; Groysman, A.; Starosvetsky, D.; Savchenko, N.; Mandler, D. Anion embedded sol-gel films on al for corrosion protection. Corros. Sci. 2004, 46, 2975–2985. [Google Scholar] [CrossRef]
- Iribarren-Mateos, J.I.; Buj-Corral, I.; Vivancos-Calvet, J.; Aleman, C.; Iribarren, J.I.; Armelin, E. Silane and epoxy coatings: A bilayer system to protect aa2024 alloy. Prog. Org. Coat. 2015, 81, 47–57. [Google Scholar] [CrossRef]
- Van den Brand, J.; Van Gils, S.; Beentjes, P.C.J.; Terryn, H.; Sivel, V.; de Wit, J.H.W. Improving the adhesion between epoxy coatings and aluminum substrates. Prog. Org. Coat. 2004, 51, 339–350. [Google Scholar] [CrossRef]
- Essahli, M.; El Asri, M.; Boulahna, A.; Zenkouar, M.; Viguier, M.; Hervaud, Y.; Boutevin, B. New alkylated and perfluoroalkylated phosphonic acids: Synthesis, adhesive and water-repellent properties on aluminum substrates. J. Fluorine Chem. 2006, 127, 854–860. [Google Scholar] [CrossRef]
- Kowalik, T.; Adler, H.J.P.; Plagge, A.; Stratmann, M. Ultrathin layers of phosphorylated cellulose derivatives on aluminium surfaces. Macromol. Chem. Phys. 2000, 201, 2064–2069. [Google Scholar] [CrossRef]
- Adler, H.-J.P.; Jaehne, E.; Stratmann, M.; Grundmeier, G. New concepts for corrosion inhibition and adhesion promotion. Proc. Annu. Meet. Tech. Program FSCT 2002, 80, 528–545. [Google Scholar]
- Hofer, R.; Textor, M.; Spencer, N.D. Alkyl phosphate monolayers, self-assembled from aqueous solution onto metal oxide surfaces. Langmuir 2001, 17, 4014–4020. [Google Scholar] [CrossRef]
- Maxisch, M.; Thissen, P.; Giza, M.; Grundmeier, G. Interface chemistry and molecular interactions of phosphonic acid self-assembled monolayers on oxyhydroxide-covered aluminum in humid environments. Langmuir 2011, 27, 6042–6048. [Google Scholar] [CrossRef] [PubMed]
- Dalmoro, V.; dos Santos, J.H.Z.; Armelin, E.; Alemán, C.; Azambuja, D.S. Phosphonic acid/silica-based films: A potential treatment for corrosion protection. Corros. Sci. 2012, 60, 173–180. [Google Scholar] [CrossRef]
- Guerrero, G.; Chaplais, G.; Mutin, P.H.; Le Bideau, J.; Leclercq, D.; Vioux, A. Grafting of alumina by diphenylphosphinate coupling agents. Mater. Res. Soc. Symp. Proc. 2001, 628. [Google Scholar] [CrossRef]
- Guerrero, G.; Mutin, P.H.; Vioux, A. Organically modified aluminas by grafting and sol-gel processes involving phosphonate derivatives. J. Mater. Chem. 2001, 11, 3161–3165. [Google Scholar] [CrossRef]
- Mutin, P.H.; Guerrero, G.; Vioux, A. Organic-inorganic hybrid materials based on organophosphorus coupling molecules: From metal phosphonates to surface modification of oxides. C. R. Chim. 2003, 6, 1153–1164. [Google Scholar] [CrossRef]
- Textor, M.; Ruiz, L.; Hofer, R.; Rossi, A.; Feldman, K.; Hahner, G.; Spencer, N.D. Structural chemistry of self-assembled monolayers of octadecylphosphoric acid on tantalum oxide surfaces. Langmuir 2000, 16, 3257–3271. [Google Scholar] [CrossRef]
- Alexander, M.R.; Thompson, G.E.; Beamson, G. Characterization of the oxide/hydroxide surface of aluminium using x-ray photoelectron spectroscopy: A procedure for curve fitting the O 1s core level. Surf. Interface Anal. 2000, 29, 468–477. [Google Scholar] [CrossRef]
- Jeurgens, L.P.H.; Sloof, W.G.; Tichelaar, F.D.; Mittemeijer, E.J. Composition and chemical state of the ions of aluminium-oxide films formed by thermal oxidation of aluminium. Surf. Sci. 2002, 506, 313–332. [Google Scholar] [CrossRef]
- Paniagua, S.A.; Hotchkiss, P.J.; Jones, S.C.; Marder, S.R.; Mudalige, A.; Marrikar, F.S.; Pemberton, J.E.; Armstrong, N.R. Phosphonic acid modification of indium-tin oxide electrodes: Combined XPS/UPS/contact angle studies. J. Phys. Chem. C 2008, 112, 7809–7817. [Google Scholar] [CrossRef]
- Van den Brand, J.; Sloof, W.G.; Terryn, H.; de Wit, J.H.W. Correlation between hydroxyl fraction and O/Al atomic ratio as determined from XPS spectra of aluminium oxide layers. Surf. Interface Anal. 2004, 36, 81–88. [Google Scholar] [CrossRef]
- Gouzman, I.; Dubey, M.; Carolus, M.D.; Schwartz, J.; Bernasek, S.L. Monolayer vs. multilayer self-assembled alkylphosphonate films: X-ray photoelectron spectroscopy studies. Surf. Sci. 2006, 600, 773–781. [Google Scholar] [CrossRef]
- Dalmoro, V.; dos Santos, J.H.Z.; Armelin, E.; Alemán, C.; Azambuja, D.S. A synergistic combination of tetraethylorthosilicate and multiphosphonic acid offers excellent corrosion protection to AA1100 aluminum alloy. Appl. Surf. Sci. 2013, 273, 758–768. [Google Scholar] [CrossRef]
- Phung, L.H.; Kleinert, H.; Fuessel, U.; Duc, L.M.; Rammelt, U.; Plieth, W. Influence of self-assembling adhesion promoter on the properties of the epoxy/aluminum interphase. Int. J. Adhes. Adhes. 2004, 25, 239–245. [Google Scholar] [CrossRef]
- Rossi, S.; Fedel, M.; Deflorian, F.; Vadillo, M.D. Localized electrochemical techniques: Theory and practical examples in corrosion studies. C. R. Chim. 2008, 11, 984–994. [Google Scholar] [CrossRef]
- Jabeera, B.; Shibli, S.M.A.; Anirudhan, T.S. Synergistic inhibitive effect of tartarate and tungstate in preventing steel corrosion in aqueous media. Appl. Surf. Sci. 2006, 252, 3520–3524. [Google Scholar] [CrossRef]
- Leidheiser, H., Jr. Towards a better understanding of corrosion beneath organic coatings. Corrosion 1983, 39, 189–201. [Google Scholar] [CrossRef]
- Zhou, L.X.; Chen, J.; Yang, Z.R. Methods for study and evaluation of anticorrosion performance of organic coatings. Corros. Sci. Prot. Tech. 2004, 16, 375–377. [Google Scholar]
- Bertocci, U.; Gabrielli, C.; Huet, F.; Keddam, M. Noise resistance applied to corrosion measurements. I. Theoretical analysis. J. Electrochem. Soc. 1997, 144, 31–37. [Google Scholar] [CrossRef]
- Bacon, R.C.; Smith, J.J.; Rugg, F.M. Electrolytic resistance in evaluating protective merit of coatings on metals. Ind. Eng. Chem. 1948, 40, 161–167. [Google Scholar] [CrossRef]
- Mills, D.; Jamali, S.; Tobiszewski, M.T. Developing electrochemical measurements in order to assess anti-corrosive coatings more effectively. Prog. Org. Coat. 2012, 74, 385–390. [Google Scholar] [CrossRef]
- Ohwaki, T.; Urushihara, W.; Kinugasa, J.; Noishiki, K. Aluminum Alloy Material and Plate Heat Exchanger with Superior Corrosion Resistance. US Patent 20100006277A1, 14 January 2010. [Google Scholar]
- Sheffer, M.; Groysman, A.; Mandler, D. Electrodeposition of sol-gel films on al for corrosion protection. Corros. Sci. 2003, 45, 2893–2904. [Google Scholar] [CrossRef]
- Ma, W.; Wu, H.; Higaki, Y.; Otsuka, H.; Takahara, A. A “non-sticky” superhydrophobic surface prepared by self-assembly of fluoroalkyl phosphonic acid on a hierarchically micro/nanostructured alumina gel film. Chem. Commun. 2012, 48, 6824–6826. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.; Han, M.; Ahn, Y. Fabrication of superhydrophobic metal surfaces with self-assembled monolayers of silane derivatives having inter-hydrogen bonding. Bull. Korean Chem. Soc. 2011, 32, 1091–1094. [Google Scholar] [CrossRef]
- Van, T.N.; Lee, Y.K.; Lee, J.; Park, J.Y. Tuning hydrophobicity of TiO2 layers with silanization and self-assembled nanopatterning. Langmuir 2013, 29, 3054–3060. [Google Scholar] [CrossRef] [PubMed]
- Akporiaye, D.; Stöcker, M. Solid-state n.m.r. and XRD study of the thermal stability of VPI-5: Assignment of 31P and 27Al MAS n.m.r. spectra. Zeolites 1992, 12, 351–359. [Google Scholar] [CrossRef]
- Gao, W.; Dickinson, L.; Grozinger, C.; Morin, F.G.; Reven, L. Self-assembled monolayers of alkylphosphonic acids on metal oxides. Langmuir 1996, 12, 6429–6435. [Google Scholar] [CrossRef]
- Gao, W.; Reven, L. Solid-state nmr studies of self-assembled monolayers. Langmuir 1995, 11, 1860–1863. [Google Scholar] [CrossRef]
- McNatt, J.S.; Morgan, J.M.; Farkas, N.; Ramsier, R.D.; Young, T.L.; Rapp-Cross, J.; Espe, M.P.; Robinson, T.R.; Nelson, L.Y. Sonication assisted growth of fluorophosphate films on alumina surfaces. Langmuir 2003, 19, 1148–1153. [Google Scholar] [CrossRef]
- Neff, G.A.; Page, C.J.; Meintjes, E.; Tsuda, T.; Pilgrim, W.C.; Roberts, N.; Warren, W.W. Hydrolysis of surface-bound phosphonate esters for the self-assembly of multilayer films: Use of solid state magic angle spinning 31p nmr as a probe of reactions on surfaces. Langmuir 1996, 12, 238–242. [Google Scholar] [CrossRef]
- Phung, L.H.; Kleinert, H.; Jansen, I.; Häßler, R.; Jähne, E. Improvement in strength of the aluminium/epoxy bonding joint by modification of the interphase. Macromol. Symp. 2004, 210, 349–358. [Google Scholar] [CrossRef]
- Lin, C.T.; Lin, P.; Hsiao, M.W.; Meldrum, D.A.; Martin, F.L. Chemistry of a single-step phosphate/paint system. Ind. Eng. Chem. Res. 1992, 31, 424–430. [Google Scholar] [CrossRef]
- Gan, S.; Solimeno, R.D.; Jones, F.N.; Hill, L.W. Recent studies of the curing of polyester-melamine enamels. Possible causes of overbake softening. Proceedings of Water-Borne and Higher-Solids Coatings Symposium, Hattiesburg, MS, USA, 1–3 February 1989; pp. 87–108. [Google Scholar]
| Characterization | Technique | Literature |
|---|---|---|
| Surface morphology | Scanning electron microscopy (SEM) | [9,14,15] |
| Atomic force microscopy (AFM) | [8,15] | |
| Presence, composition and stability of layer | X-ray photoelectron spectroscopy (XPS) | [6,7,8,11,16,17,18,19,20,21] |
| Auger electron spectroscopy (AES) | [22] | |
| Fourier-transform infrared spectroscopy (FTIR) | [8,11,14,16,17,23,24] | |
| Time-of-flight secondary ion mass spectrometry (ToF-SIMS) | [6] | |
| Water contact angle measurements (WCA) | [5,7,10,16,25,26] | |
| Orientation of molecules and binding mode to aluminum | Angle-resolved X-ray photoelectron spectroscopy (ARXPS) | [7,10,27,28] |
| Infrared reflection absorption spectroscopy at grazing angle (IRRAS) | [5,12,16,29,30,31,32,33] | |
| X-ray photoelectron spectroscopy (XPS) | [20,28,34] | |
| Solid-state 31P nuclear magnetic resonance (NMR) | [12,31,32,33] |
| Assignment | Binding Energy (eV) | Literature |
|---|---|---|
| Al 2p | [7,16,20,35,36] | |
| metal | 72.3–73.3 | |
| Al-oxide, Al-hydroxide | 74.2–76.2 | |
| Al-phosphonate | ~75.5 | |
| P 2p | [16,34,37] | |
| phosphonates | 133.3–134.2 | |
| P 2s | [20] | |
| P–C, P=O, P–O | 192.5–192.7 | |
| O 1s | [16,28,34,35,38,39] | |
| Al–oxide | 530.6–531.1 | |
| Al–hydroxide | 532.3–532.4 | |
| water | 534–535 | |
| P=O, P–O−, P–O–Metal | 531.4–532.1 | |
| P–OH, P–OR | 532.6–534.3 |
| Assignment | Peak Position (cm−1) | Literature |
|---|---|---|
| P–O sym. stretch | 910–960 | [5,24,27,30,37] |
| P–O asym. stretch | 1000–1040 | |
| P=O stretch | 1100–1250 | [8,11,12,16,24,27,30,31,32,37] |
| PO2− sym. stretch | 1000–1070 | [37] |
| PO2− asym. stretch | ~1160 | |
| PO32− sym. stretch | 960–1060 | [5,8,24,29,37] |
| PO32− asym. stretch | 1115–1140 | |
| P–OH stretch | 2500–2750 | [16,27,31] |
| Measurements | Examples | Literature |
|---|---|---|
| Electrochemical measurements | Electrochemical noise method (ENM) | [22] |
| Potentiodynamic polarization measurements | [14,15,17,22,30,40,41] | |
| Electrochemical impedance spectroscopy (EIS) | ||
| Localized electrochemical techniques | Scanning Kelvin Probe (SKP) and Scanning Vibrating Electrode Technique (SVET) | [42] |
| Conventional measurements | Immersion test | [14,23,41] |
| Salt spray test | [8,27] | |
| Weight loss treatment | [43] | |
| Spectral analysis | Infrared spectroscopy (IR) | [12] |
| Wettability | Contact angle measurements | [10,19] |
| Chemical Name | Structure | Abbreviation | Literature |
|---|---|---|---|
| Phenylphosphonic acid | 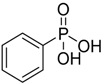 | PPA | [14,22] |
| Vinylphosphonic acid | 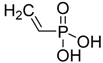 | VPA | [49] |
| 1,12-dodecyldiphosphonic acid | 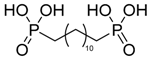 | DDP | [8] |
| Amino trimethylene phosphonic acid | 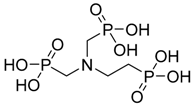 | ATMP | [15] |
| Ethylenediamine tetra methylene phosphonic acid |  | EDTPO | [17,30,40] |
| 1,2-diaminoethanetetrakis-methylenephosphonic acid | 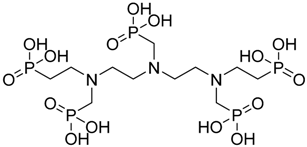 | DETAPO | [23] |
| Monostearyl acid phosphate |  | MSAP | [12] |
| Phosphoric acid mono-(12-hydroxy-dodecyl) ester | 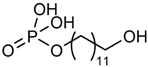 | – | [41] |
| (12-ethylamino-dodecyl)-phosphonic acid |  | – | [27] |
| Aminopropyl phosphonate | 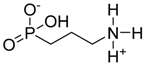 | APP | [9,11] |
| ω-(3-phenylpyrrol-1-ylalkyl) phosphonic acid | 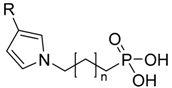 | Cn-Ph-P | [10] |
| ω-(2,5-Dithienylpyrrol-1-yl-alky) phosphonic acid | 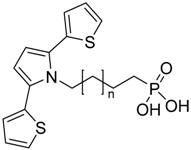 | SNS-n-P | [10] |
| 2-(Phosphonooxy) benzoic acid |  | Fosfosal | [14] |
| Phosphonosuccinic acid | 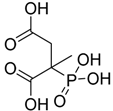 | PPSA | [14] |
| (1H,1H, 2H,2H-heptadecafluorodec-1-yl) phosphonic acid | 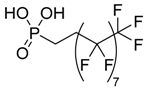 | HDF-PA | [19] |
| Fluoro alkyl phosphates |  | Zonyl UR | [12] |
| Rx PO(OH)y | |||
| R = CF3(CF2CF2)z(CH2CH2O) | |||
| x = 1, 2 or 3, x + y = 3, z = 1 to 7 | |||
| Poly (vinyl phosphonic acid) |  | PvPA | [24] |
| Sol-Gel Chemistry | PTMOS | PTMOS | PTMOS + TMA | PTMOS | TEOS | TEOS | TEOS + TMA |
|---|---|---|---|---|---|---|---|
| Anion | – | PPA | PPA | TBPA | – | PPA | PPA |
| Concentration | DSC (Tg) | EIS (Ω·cm2) | Saltwater Immersion | Pencil Hardness |
|---|---|---|---|---|
| 1% | 35 to 59 °C | 109–107 | – | 4H |
| 0.75% | 34 to 55 °C | 109 | Ø 3 mm blister | 4H |
| 0.5% | 18 to 48 °C | 109 | – | HB |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, R.; Rupper, P.; Gaan, S. Recent Development in Phosphonic Acid-Based Organic Coatings on Aluminum. Coatings 2017, 7, 133. https://doi.org/10.3390/coatings7090133
Zhao R, Rupper P, Gaan S. Recent Development in Phosphonic Acid-Based Organic Coatings on Aluminum. Coatings. 2017; 7(9):133. https://doi.org/10.3390/coatings7090133
Chicago/Turabian StyleZhao, Ruohan, Patrick Rupper, and Sabyasachi Gaan. 2017. "Recent Development in Phosphonic Acid-Based Organic Coatings on Aluminum" Coatings 7, no. 9: 133. https://doi.org/10.3390/coatings7090133
APA StyleZhao, R., Rupper, P., & Gaan, S. (2017). Recent Development in Phosphonic Acid-Based Organic Coatings on Aluminum. Coatings, 7(9), 133. https://doi.org/10.3390/coatings7090133






