Chemical Vapor Deposition of TaC/SiC on Graphite Tube and Its Ablation and Microstructure Studies
Abstract
:1. Introduction
Aim and Objectives
2. Experimental
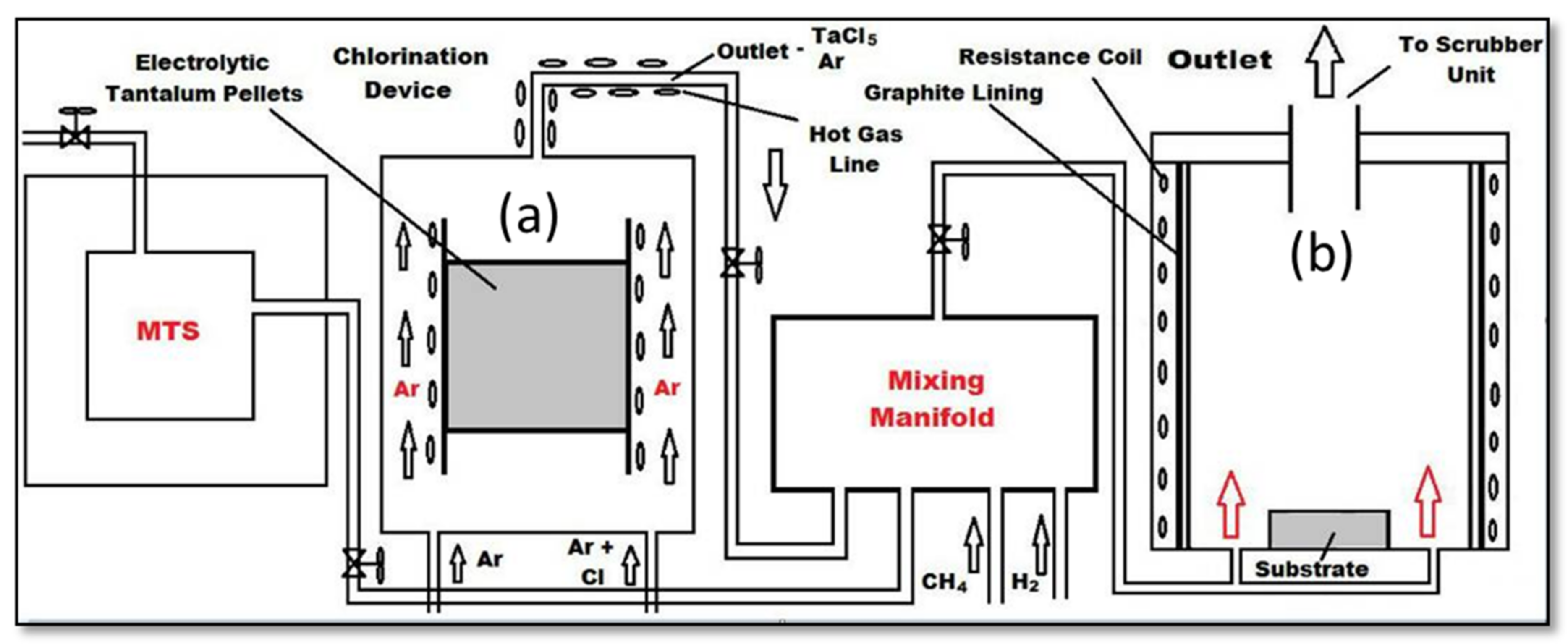
2.1. CVD Process Setup
2.1.1. Chlorination Reactor
2.1.2. CVD Reactor
2.1.3. Mixing Manifold
2.2. CVD Procedure
2.3. Characterization and Morphology
2.4. Ablation Test Using Lene Flame
3. Results and Discussion
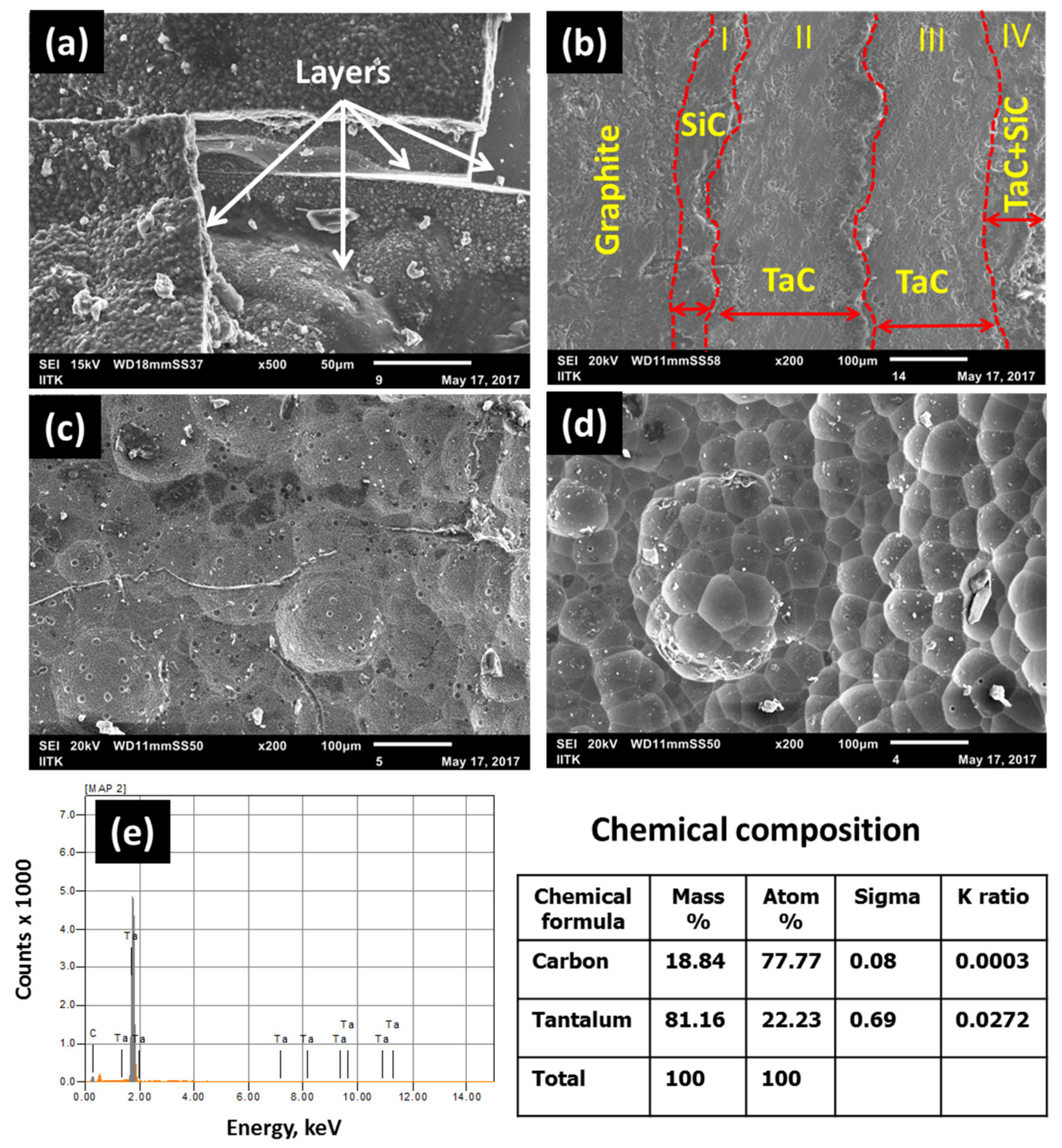
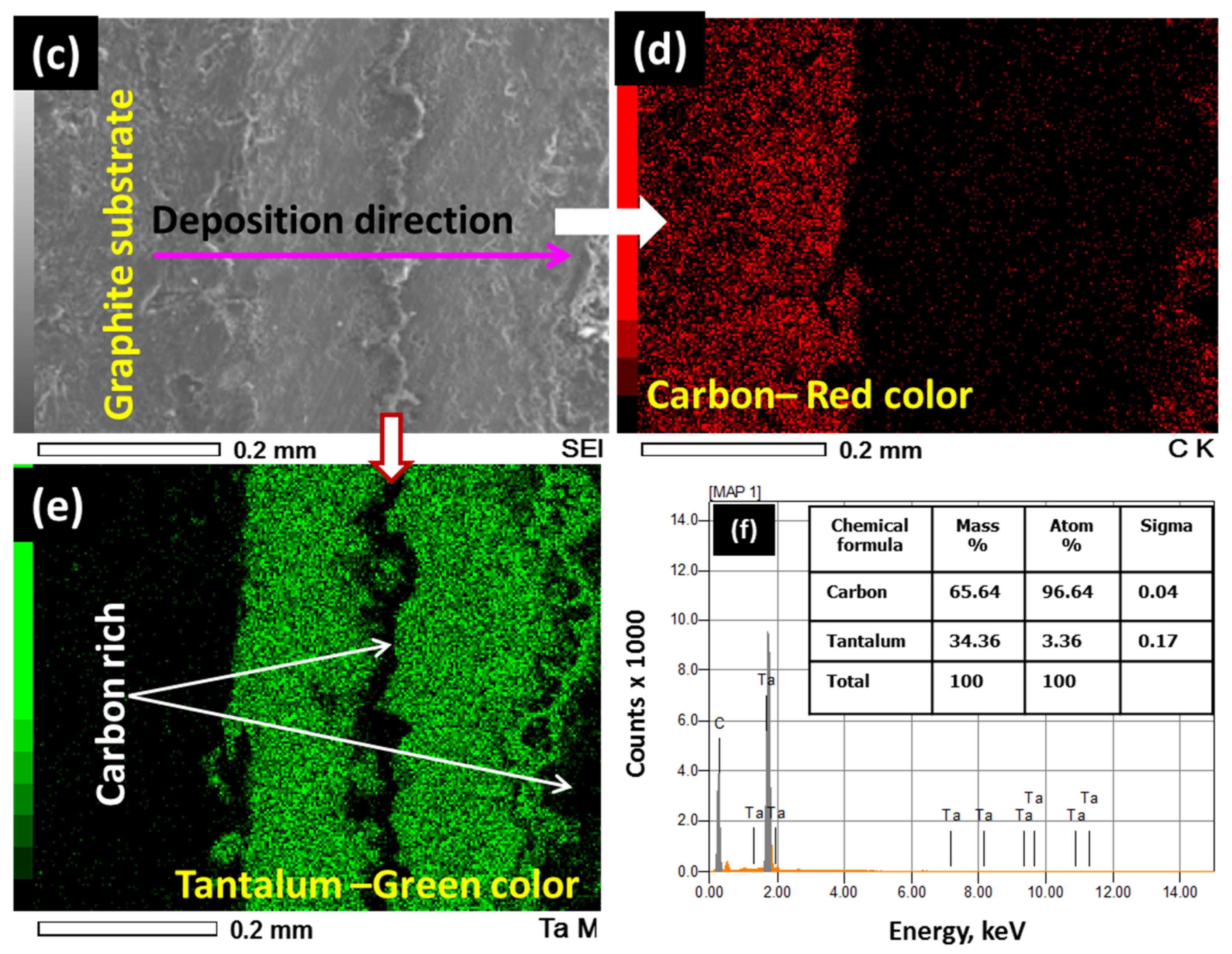
3.1. Coatings on Tubular Graphite Substrates
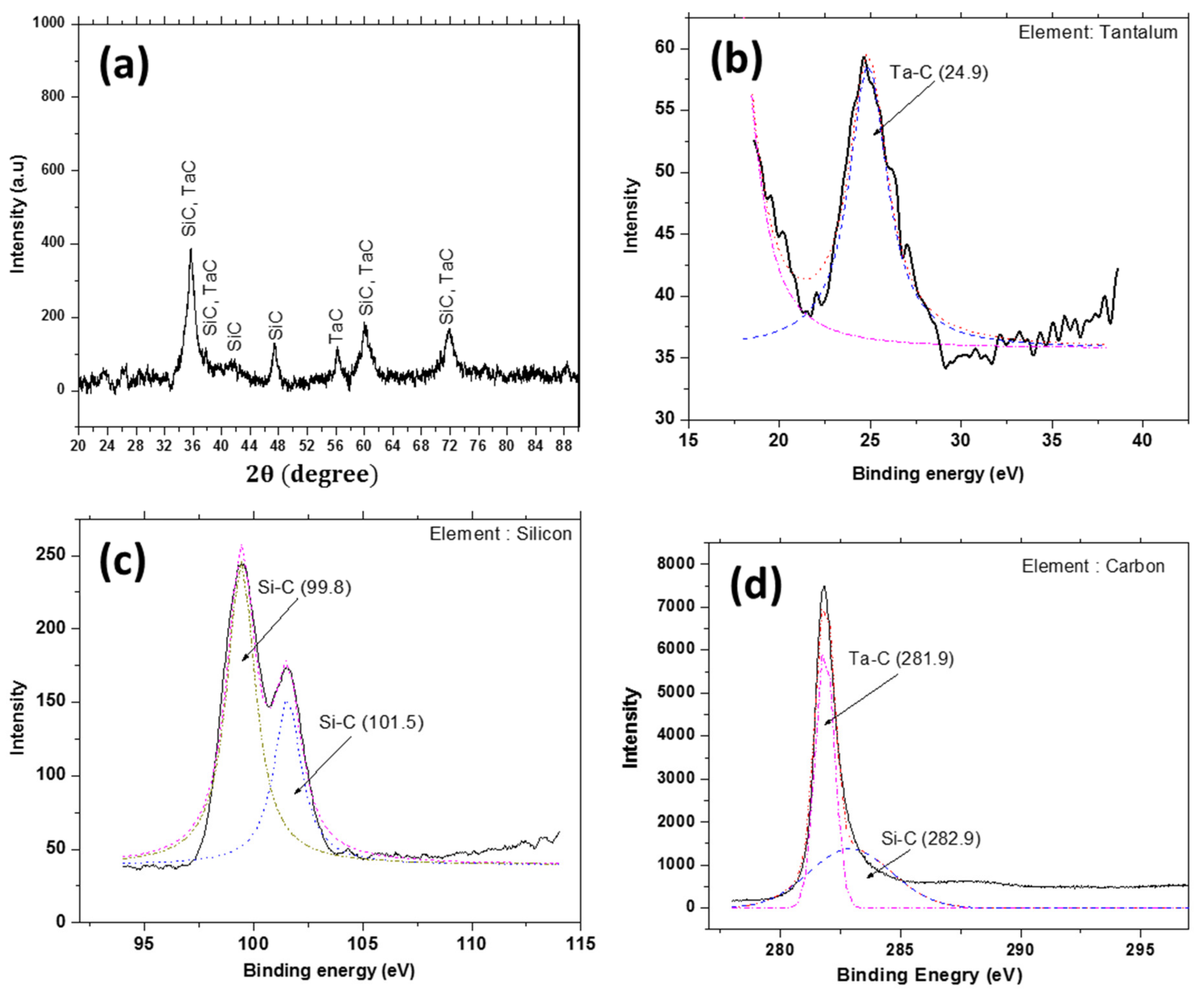
3.2. XRD and XPS Analysis of the As-Deposited Coating
3.3. Characterization after Thermal Shock and Ablation
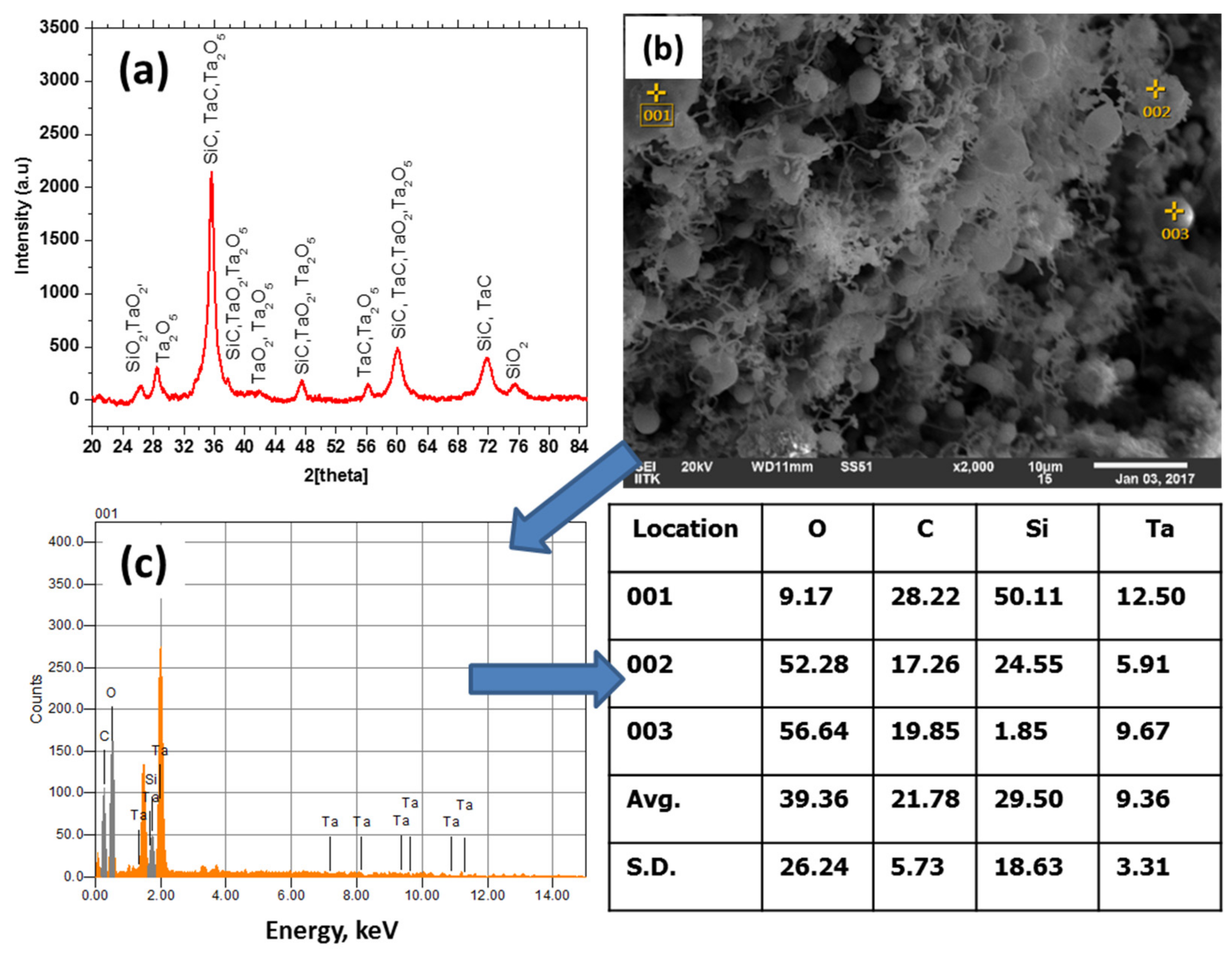
3.4. Morphology and Phase Analysis of the Ablated Specimens
4. Conclusions
- Chlorination of tantalum chips at ≈550 °C has resulted in the in situ synthesis of TaCl5. Based on TaCl5-CH4-Ar and CH3SiCl3-H2-Ar reactive systems, the crack-free multilayer coating of TaC and SiC is successfully developed on a graphite tube by CVD at 1050–1150 °C and 50–100 mbar. XRD and XPS studies confirm the formation of TaC and SiC.
- By depositing SiC as transition layers between the graphite and TaC, the stress concentration could be released and crack-free coating could be obtained. The coating remained intact even after the ablation test under oxyacetylene flame. At 1150 °C the thickness of the coating was about 600 μm, while at 1050 °C it was about 400 μm for the same duration and flow conditions.
- The oxyacetylene flame ablation of the TaC coating is mainly an oxidation process where surface TaC is oxidized to TaO2 and Ta2O2, while SiC oxided SiO2 and acted as protective liquid film to prevent further oxidation of the graphite underneath.
- The uniform deposition of TaC on the cylindrical graphite tube has established the process for generating a CVD coating of TaC on similar geometrical featured propulsion systems such as graphite throat inserts, as well as the chambers and nozzles of rocket motors.
Acknowledgments
Author Contributions
Conflicts of Interest
References
- McKee, D.W. Oxidation Protection of Carbon Materials, Chemistry and Physics of Carbon; Thrower, P.A., Ed.; Marcel Dekker: New York, NY, USA, 1991; Volume 23, pp. 173–232. [Google Scholar]
- Kumar, S.; Shekar, K.C.; Jana, B.; Manocha, L.M.; Prasad, N.E. C/C and C/SiC Composites for Aerospace Applications. In Aerospace Materials and Material Technologies; Part of the series Indian Institute of Metals Series; Springer: Singapore, Singapore, 2016; pp. 343–369. [Google Scholar]
- Paul, A.; Jayaseelan, D.D.; Venugopal, S.; Zapata-Solvas, E.; Binner, J.G.P.; Vaidhyanathan, B.; Heaton, A.; Brown, P.M.; Lee, W.E. UHTC composites for hypersonic applications. Am. Ceram. Soc. Bull. 2012, 91, 22–28. [Google Scholar]
- Yang, Y.; Li, K.; Li, H. Ablation behavior of HfC coating prepared by supersonic plasma spraing for SiC-coated C/C composites. Adv. Compos. Lett. 2015, 24, 107–112. [Google Scholar]
- Hu, M.H.; Li, K.Z.; Li, H.J.; Wang, B.; Ma, H.L. Double layer ZrSi2-ZrC-SiC/SiC oxidation protective coating for carbon/ carbon composites. Surf. Eng. 2014, 31, 335–341. [Google Scholar] [CrossRef]
- Ren, X.; Li, H.; Fu, Q.; Li, K. Ultra-high temperature ceramic TaB2-TaC-SiC coating for oxidation protection of SiC-coated carbon/carbon composites. Ceram. Int. 2014, 40, 9419–9425. [Google Scholar] [CrossRef]
- Boisselier, G.; Maury, F.; Schuster, F. SiC coatings grown by liquid injection chemical vapor deposition using single source metal-organic precursors. Surf. Coat. Technol. 2013, 215, 152–160. [Google Scholar] [CrossRef]
- Chen, S.A.; Li, G.D.; Hu, H.F.; Li, Y.; Mei, M. Microstructure and properties of ablative C/ZrC-SiC composites prepared by reactive melt infiltration of zirconium and vapour silicon infiltration. Ceram. Int. 2017, 43, 3439–3442. [Google Scholar] [CrossRef]
- Li, G.-D.; Xiong, X.; Huang, K.-L. Ablation mechanism of TaC coating fabricated by chemical vapor deposition on carbon-carbon composites. Trans. Nonferr. Met. Soc. China 2009, 19, s689–s695. [Google Scholar] [CrossRef]
- Dua, A.K.; George, V.C. TaC coatings prepared by hot filament chemical vapour deposition: Characterization and properties. Thin Solid Films 1994, 247, 34–38. [Google Scholar] [CrossRef]
- Bruno, T.S.; Anthony, C.; Paul, Y.; Sanjay, B.; Paul, J.; Lynn, D.; Joseph, Z. Net Mold Tantalum Carbide Rocket Nozzle Throat. U.S. Patent 6,673,449, 6 January 2004. [Google Scholar]
- Li, G.D.; Xiong, X.; Huang, B.-Y. Microstructure characteristic and formation mechanism of crack-free TaC coating on C/C composite. Trans. Nonferr. Met. Soc. China 2005, 15, 1206–1213. [Google Scholar]
- Li, G.D.; Xiong, X.; Huang, B.-Y.; Huang, K.-L. Structural characteristics and formation mechanisms of crack-free multilayer TaC/SiC coatings on carbon-carbon composites. Trans. Nonferr. Met. Soc. China 2008, 18, 255–261. [Google Scholar] [CrossRef]
- Verdon, C.; Szwedek, O.; Jacques, S.; Allemand, A.; Le Petitcorps, Y. Hafnium and silicon carbide multilayer coatings for the protection of carbon composites. Surf. Coat. Technol. 2013, 230, 124–129. [Google Scholar] [CrossRef]
- Yasuaki, H.; William, D.; Russel, M. Temperature dependence of nucleation density of chemical vapor deposition diamond. Jpn. J. Appl. Phys. 1992, 31, L193–L196. [Google Scholar]
- Khyzhun, O.Y.; Sinelnichenko, A.K.; Kolyagin, V.A. NIST XPS Database. Available online: https://srdata.nist.gov/xps/XPSDetailPage.aspx?AllDataNo=94091 (accessed on 15 June 2017).
- Ramqvist, L. Charge transfer in transition metal carbides and related compounds studied by ESCAJ. Phys. Chem. Solids 1969, 30, 1835. [Google Scholar] [CrossRef]
- Galuska, A.A.; Uht, J.C.; Marquez, N. Reactive and nonreactive ion mixing of Ti films on carbon substrates. J. Vac. Sci. Technol. 1988, A6, 110–122. [Google Scholar] [CrossRef]
- Aoyama, T.; Sugil, T.; Ito, T. Determination of band line-up in β-SiC/Si hetero-junction for Si-HBT’s. Appl. Surf. Sci. 1989, 41, 584–586. [Google Scholar]
- Niles, D.W.; Hochst, H.; Zajac, G.W.; Fleisch, T.H.; Johnson, B.C.; Meese, J.M. The interface formation and thermal stability of Ag overlayers grown on cubic SiC (100). J. Vac. Sci. Technol. 1988, A6, 1584–1588. [Google Scholar] [CrossRef]
- Mag-isa, A.E.; Kim, J.-H.; Oh, C.-S. Measurements of the in-plane coefficient of thermal expansion of free standing single-crystal natural graphite. Mater. Lett. 2016, 171, 312–331. [Google Scholar] [CrossRef]
- Lespade, P.; Richet, N.; Goursat, P. Oxidation resistance of HfB2-SiC composites for protection of carbon-based materials. Acta Astronaut. 2007, 60, 858–864. [Google Scholar] [CrossRef]
- Pienti, L.; Sciti, D.; Silvestroni, L.; Cecere, A.; Savino, R. Ablation tests on HfC- and TaC-based ceramics for aeropropulsive applications. J. Eur. Ceram. Soc. 2015, 35, 1401–1411. [Google Scholar] [CrossRef]



© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kumar, S.; Mondal, S.; Kumar, A.; Ranjan, A.; Prasad, N.E. Chemical Vapor Deposition of TaC/SiC on Graphite Tube and Its Ablation and Microstructure Studies. Coatings 2017, 7, 101. https://doi.org/10.3390/coatings7070101
Kumar S, Mondal S, Kumar A, Ranjan A, Prasad NE. Chemical Vapor Deposition of TaC/SiC on Graphite Tube and Its Ablation and Microstructure Studies. Coatings. 2017; 7(7):101. https://doi.org/10.3390/coatings7070101
Chicago/Turabian StyleKumar, Suresh, Samar Mondal, Anil Kumar, Ashok Ranjan, and Namburi Eswara Prasad. 2017. "Chemical Vapor Deposition of TaC/SiC on Graphite Tube and Its Ablation and Microstructure Studies" Coatings 7, no. 7: 101. https://doi.org/10.3390/coatings7070101



