Fabrication of Luminescent Antireflective Coatings with CaMoO4:Eu3+/Ag Composite Structure
Abstract
:1. Introduction
2. Materials and Methods
3. Results and Discussion
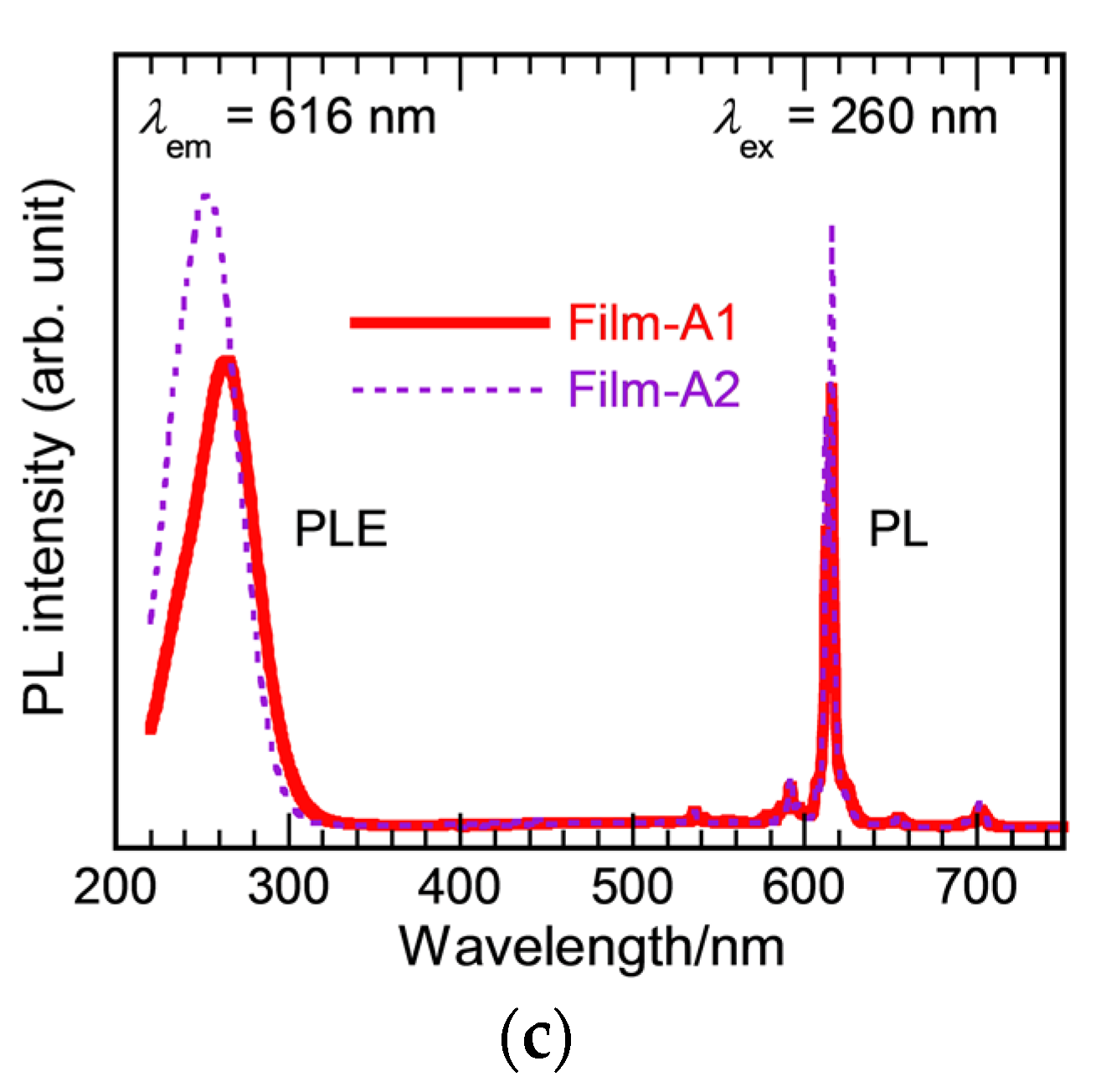
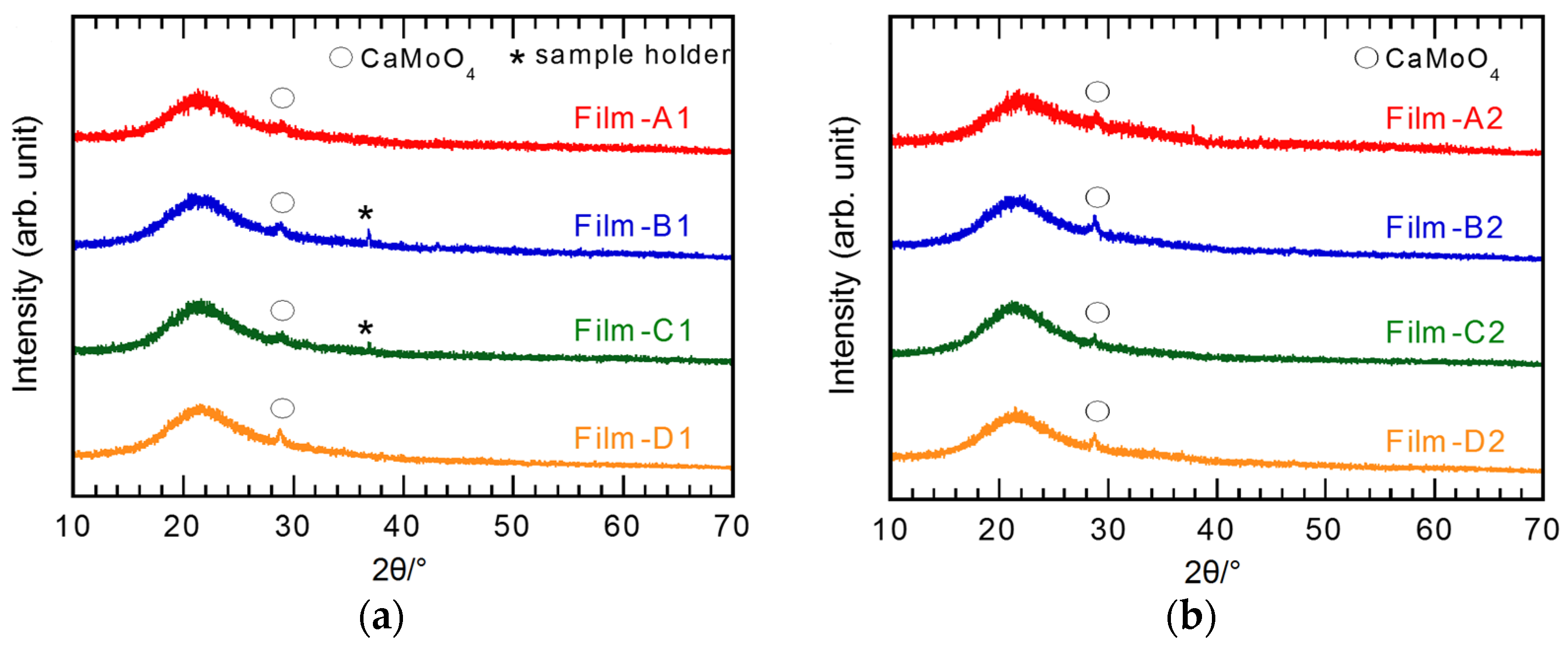
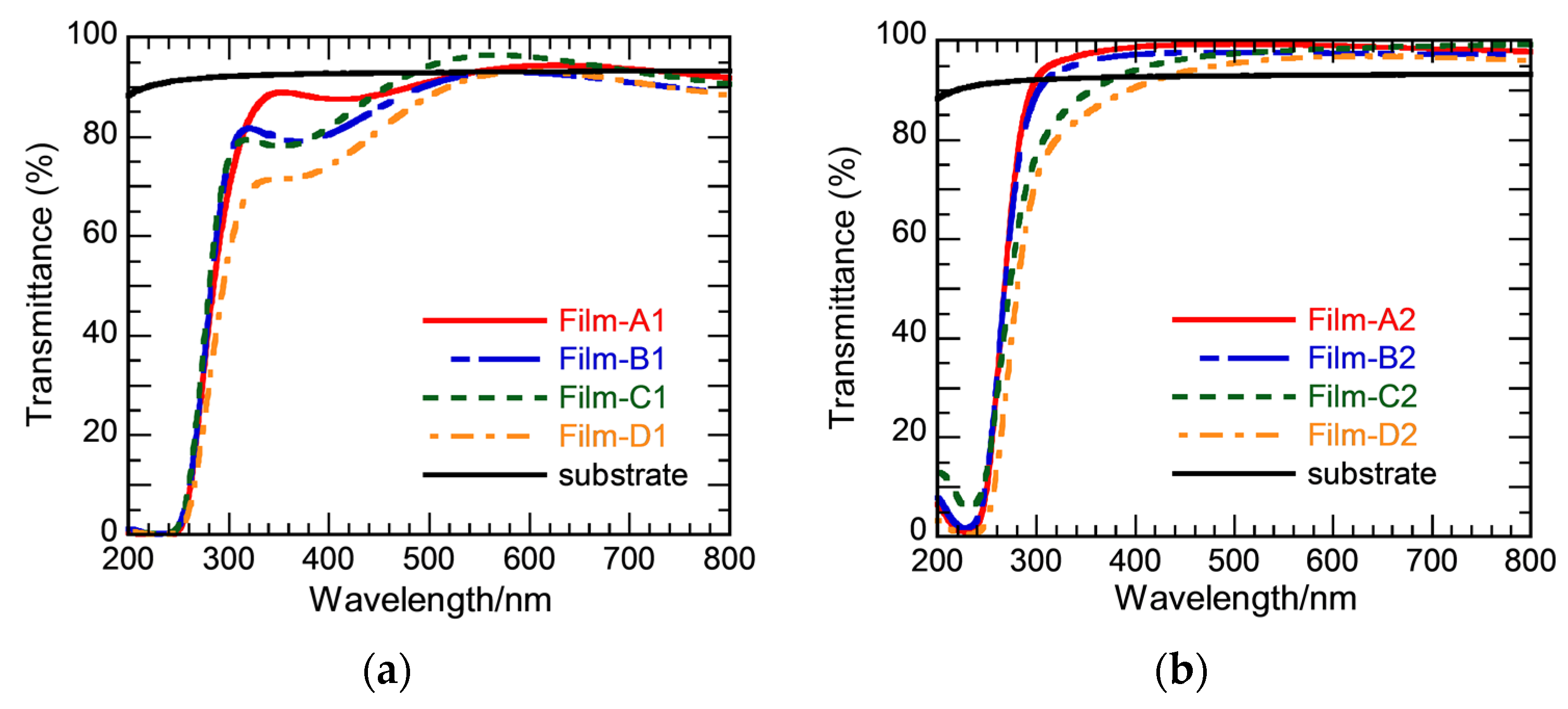
3.1. Effect of Hot Water Treatment
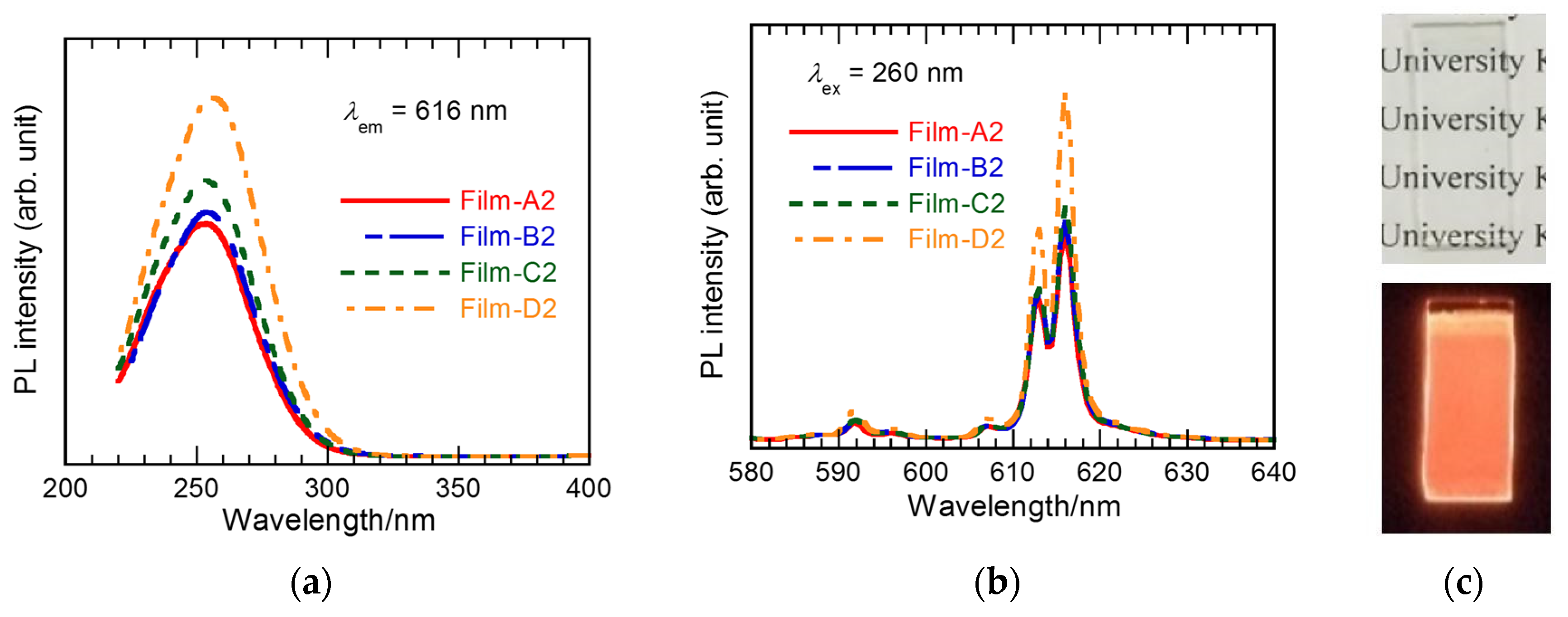
3.2. Effect of Silver Addition
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Chen, X.Y.; Zhang, Z.J.; Zhu, L.L.; Xu, M.; Wang, H.; Li, A.G.; Zhao, J.T. Preparation, and characterizations of a novel luminescence Lu2WO6:Eu3+ film as potential scintillator. Appl. Surf. Sci. 2014, 317, 730–736. [Google Scholar] [CrossRef]
- Huang, X.; Han, S.; Huang, W.; Liu, X. Enhancing solar cell efficiency: the search for luminescent materials as spectral converters. Chem. Soc. Rev. 2013, 42, 173–201. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Miyokawa, N.; Ide, K.; Hiramatsu, H.; Hosono, H.; Kamiya, T. Transparent amorphous oxide semiconductor thin film phosphor, In–Mg–O:Eu. J. Ceram. Soc. Jpn. 2016, 124, 532–535. [Google Scholar] [CrossRef]
- Kyomen, T.; Hanaya, M.; Takashima, H. Electroluminescence near interfaces between (Ca,Sr)TiO3:Pr phosphor and SnO2:Sb transparent conductor thin films prepared by sol–gel and spin-coating methods. J. Lumin. 2014, 149, 133–137. [Google Scholar] [CrossRef]
- Zhang, Y.; Holzwarth, N.A.W.; Williams, R.T. Electronic band structures of the scheelite materials CaMoO4, CaWO4, PbMoO4, and PbWO4. Phys. Rev. B 1998, 57, 12738–12750. [Google Scholar] [CrossRef]
- Thongtem, T.; Kungwankunakorn, S.; Kuntalue, B.; Phuruangrat, A.; Thongtem, S. Luminescence and absorbance of highly crystalline CaMoO4, SrMoO4, CaWO4 and SrWO4 nanoparticles synthesized by co-precipitation method at room temperature. J. Alloys Compd. 2010, 506, 475–481. [Google Scholar] [CrossRef]
- Bonner, G.M.; Pask, H.M.; Lee, A.J.; Kemp, A.J.; Wang, J.; Zhang, H.; Omatsu, T. Measurement of thermal lensing in a CW BaWO4 intracavity Raman laser. Opt. Express 2012, 20, 9810–9818. [Google Scholar] [CrossRef] [PubMed]
- De Azevedo Marques, A.P.; Longo, V.M.; de Melo, D.M.A.; Pizani, P.S.; Leite, E.R.; Varela, J.A.; Longo, E. Shape controlled synthesis of CaMoO4 thin films and their photoluminescence property. J. Solid State Chem. 2008, 181, 1249–1257. [Google Scholar] [CrossRef]
- Raju, G.S.R.; Pavitra, E.; Ko, Y.H.; Yu, J.S. A facile and efficient strategy for the preparation of stable CaMoO4 spherulites using ammonium molybdate as a molybdenum source and their excitation induced tunable luminescent properties for optical applications. J. Mater. Chem. 2012, 22, 15562–15569. [Google Scholar] [CrossRef]
- Dutta, S.; Som, S.; Sharma, S.K. Excitation spectra and luminescence decay analysis of K+ compensated Dy3+ doped CaMoO4 phosphors. RSC Adv. 2015, 5, 7380–7387. [Google Scholar] [CrossRef]
- Pulker, H.K. Coatings on Glass, 2nd ed.; Elsevier: Amsterdam, The Netherlands, 1999; pp. 433–440. [Google Scholar]
- Cavalli, E.; Bovero, E.; Belletti, A. Optical spectroscopy of CaMoO4:Dy3+ single crystals. J. Phys. Condens. Matter 2002, 14, 5221–5228. [Google Scholar] [CrossRef]
- Zheng, Q.; Zhang, D.; Huang, J.; Wang, Y.; Huang, F. Integrating surface textures on ZnO substrate for high light extraction efficiency light-emitting diode. J. Phys. Chem. C 2014, 118, 14894–14898. [Google Scholar] [CrossRef]
- Yang, J.; Luo, F.; Kao, T.S.; Li, X.; Ho, G.W.; Teng, J.; Luo, X.; Hong, M. Design and fabrication of broadband ultralow reflectivity black Si surfaces by laser micro/nanoprocessing. Light Sci. Appl. 2014, 3, e185. [Google Scholar] [CrossRef]
- Leem, J.W.; Kim, S.; Lee, S.H.; Rogers, J.A.; Kim, E.; Yu, J.S. Efficiency enhancement of organic solar cells using hydrophobic antireflective inverted moth-eye nanopatterned PDMS films. Adv. Energy Mater. 2014, 4, 1301315. [Google Scholar] [CrossRef]
- Kasugai, H.; Nagamatsu, K.; Miyake, Y.; Honshio, A.; Kawashima, T.; Iida, K.; Iwaya, M.; Kamiyama, S.; Amano, H.; Akasaki, I.; et al. Light extraction process in moth-eye structure. Phys. Status Solidi c 2006, 3, 2165–2168. [Google Scholar] [CrossRef]
- Rao, J.; Winfield, R.; Keeney, L. Moth-eye-structured light-emitting diodes. Opt. Commun. 2010, 283, 2446–2450. [Google Scholar] [CrossRef]
- Qian, X.; Wang, N.; Li, Y.; Zhang, J.; Xu, Z.; Long, Y. Bioinspired multifunctional vanadium dioxide: Improved thermochromism and hydrophobicity. Langmuir 2014, 30, 10766–10771. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, N.; Tadanaga, K.; Matsuda, A.; Minami, T.; Tatsumisago, M. Anti-reflective coatings of flowerlike alumina on various glass substrates by the sol–gel process with the hot water treatment. J. Sol-Gel Sci. Technol. 2005, 33, 117–120. [Google Scholar] [CrossRef]
- Tadanaga, K.; Yamaguchi, N.; Uraoka, Y.; Matsuda, A.; Minami, T.; Tatsumisago, M. Anti-reflective properties of nano-structured alumina thin films on poly(methyl methacrylate) substrates by the sol–gel process with hot water treatment. Thin Solid Films 2008, 516, 4526–4529. [Google Scholar] [CrossRef]
- Tadanaga, K. Surface morphology control of thin films prepared by solution processes and its application. J. Ceram. Soc. Jpn. 2013, 121, 819–824. [Google Scholar] [CrossRef]
- Tanaka, S.; Fujihara, S. Luminescent antireflective coatings with disordered surface nanostructures fabricated by liquid processes. Langmuir 2011, 27, 2929–2935. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Wang, N.; Long, Y. Multifunctional overcoats on vanadium dioxide thermochromic thin films with enhanced luminous transmission and solar modulation, hydrophobicity and anti-oxidation. Appl. Surf. Sci. 2013, 283, 222–226. [Google Scholar] [CrossRef]
- Pillonnet, A.; Berthelot, A.; Pereira, A.; Benamara, O.; Derom, S.; des Francs, G.C.; Jurdyc, A.-M. Coupling distance between Eu3+ emitters and Ag nanoparticles. Appl. Phys. Lett. 2012, 100, 153115. [Google Scholar] [CrossRef]
- Lee, P.C.; Meisel, D. Adsorption and surface-enhanced Raman of dyes on silver and gold sols. J. Phys. Chem. 1982, 86, 3391–3395. [Google Scholar] [CrossRef]
- Sharma, V.K.; Yngard, R.A.; Lin, Y. Silver nanoparticles: green synthesis and their antimicrobial activities. Adv. Colloid Interf. Sci. 2009, 145, 83–96. [Google Scholar] [CrossRef] [PubMed]
- Gürmen, E.; Daniels, E.; King, J.S. Crystal structure refinement of SrMoO4, SrWO4, CaMoO4, and BaWO4 by neutron diffraction. J. Chem. Phys. 1971, 55, 1093–1097. [Google Scholar] [CrossRef]
- Parchur, A.K.; Ningthoujam, R.S.; Rai, S.B.; Okram, G.S.; Singh, R.A.; Tyagi, M.; Gadkari, S.C.; Tewari, R.; Vatsa, R.K. Luminescence properties of Eu3+ doped CaMoO4 nanoparticles. Dalton Trans. 2011, 40, 7595–7601. [Google Scholar] [CrossRef] [PubMed]
- Blass, G.; Grabmaier, B.C. Luminescent Materials; Springer-Verlag: Berlin, Germany, 1994; pp. 41–44. [Google Scholar]
- Laguna, M.; Nuñez, N.O.; Becerro, A.I.; Ocaña, M. Morphology control of uniform CaMoO4 microarchitectures and development of white light emitting phosphors by Ln doping (Ln = Dy3+, Eu3+). CrystEngComm 2017, 19, 1590–1600. [Google Scholar] [CrossRef]
- Paramelle, D.; Sadovoy, A.; Gorelik, S.; Free, P.; Hobley, J.; Fernig, D.G. A rapid method to estimate the concentration of citrate capped silver nanoparticles from UV-visible light spectra. Analyst 2014, 139, 4855–4861. [Google Scholar] [CrossRef] [PubMed]
- Nayar, P.; Khanna, A. Optical properties and thermal stability of alumina films grown by pulsed laser deposition. Eur. Phys. J. Appl. Phys. 2013, 64, 10301. [Google Scholar] [CrossRef]
- Eihelbaum, M.; Rademann, K. Plasmonic enhancement or energy transfer? On the luminescence of gold-, silver-, and lanthanide-doped silicate glasses and its potential for light-emitting devices. Adv. Funct. Mater. 2009, 19, 2045–2052. [Google Scholar] [CrossRef]




© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Matsumoto, K.; Hagiwara, M.; Fujihara, S. Fabrication of Luminescent Antireflective Coatings with CaMoO4:Eu3+/Ag Composite Structure. Coatings 2017, 7, 74. https://doi.org/10.3390/coatings7060074
Matsumoto K, Hagiwara M, Fujihara S. Fabrication of Luminescent Antireflective Coatings with CaMoO4:Eu3+/Ag Composite Structure. Coatings. 2017; 7(6):74. https://doi.org/10.3390/coatings7060074
Chicago/Turabian StyleMatsumoto, Kazuhiro, Manabu Hagiwara, and Shinobu Fujihara. 2017. "Fabrication of Luminescent Antireflective Coatings with CaMoO4:Eu3+/Ag Composite Structure" Coatings 7, no. 6: 74. https://doi.org/10.3390/coatings7060074
APA StyleMatsumoto, K., Hagiwara, M., & Fujihara, S. (2017). Fabrication of Luminescent Antireflective Coatings with CaMoO4:Eu3+/Ag Composite Structure. Coatings, 7(6), 74. https://doi.org/10.3390/coatings7060074






