High-Temperature Corrosion of AlCrSiN Film in Ar-1%SO2 Gas
Abstract
:1. Introduction
2. Experimental Section
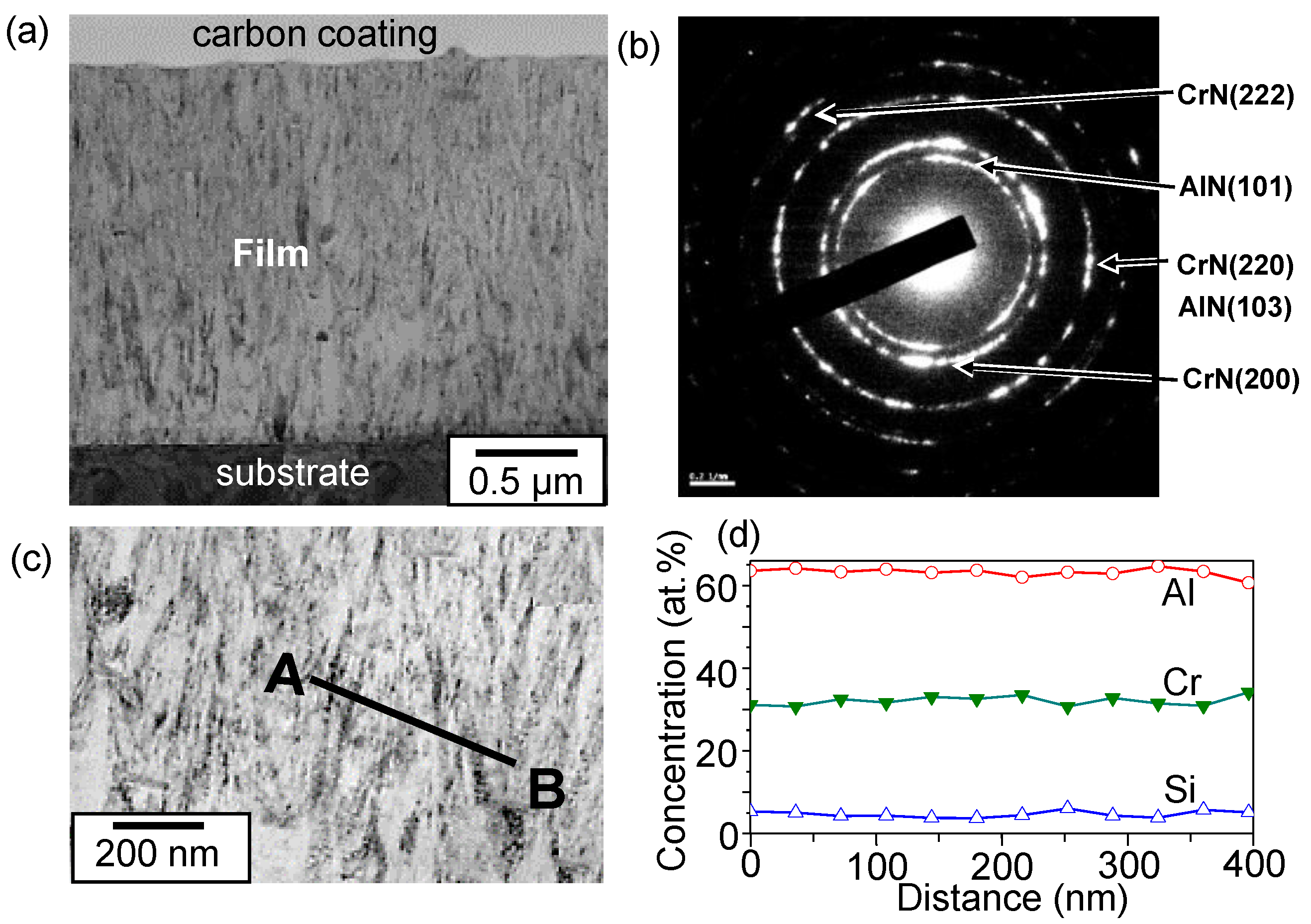
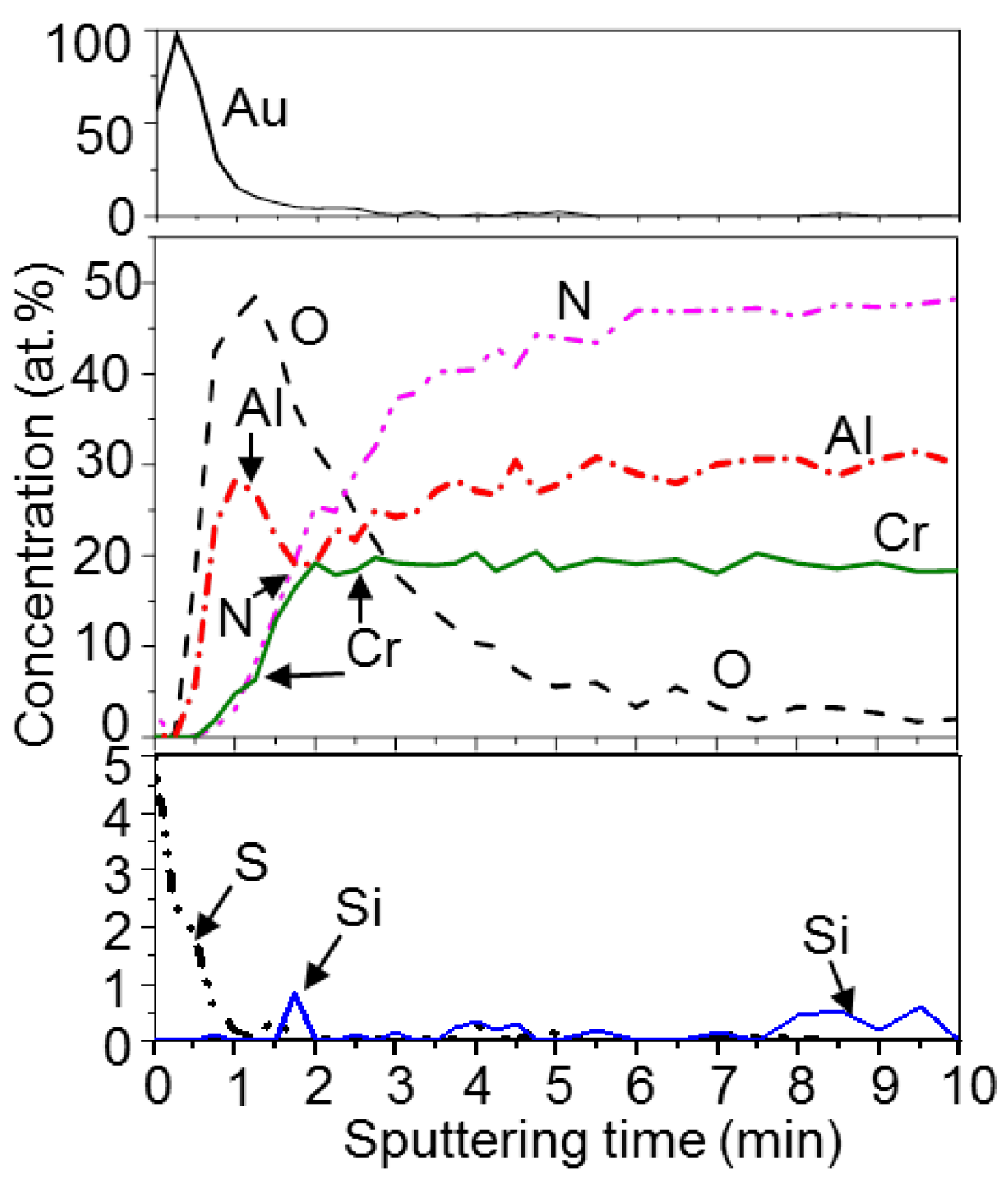
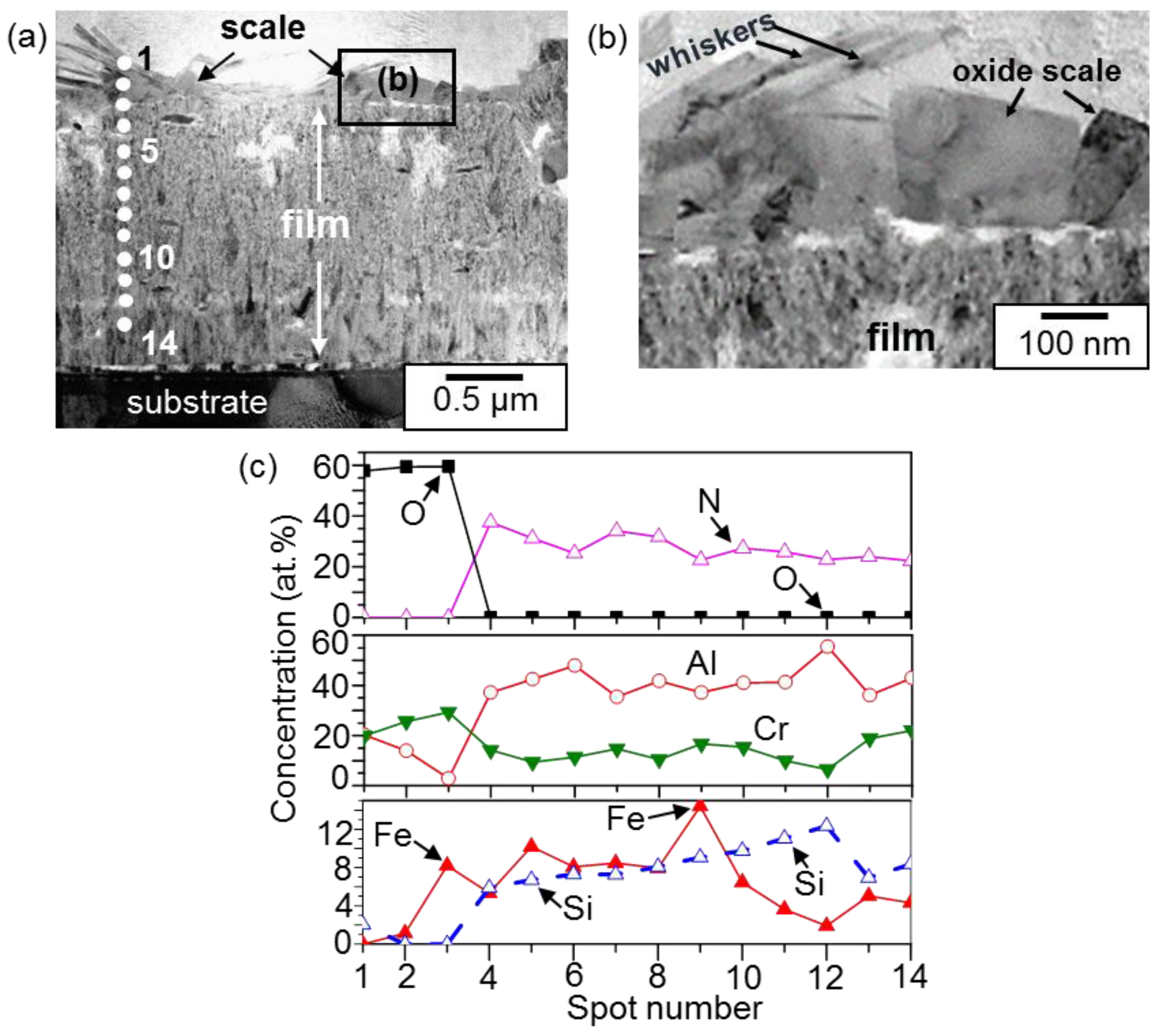
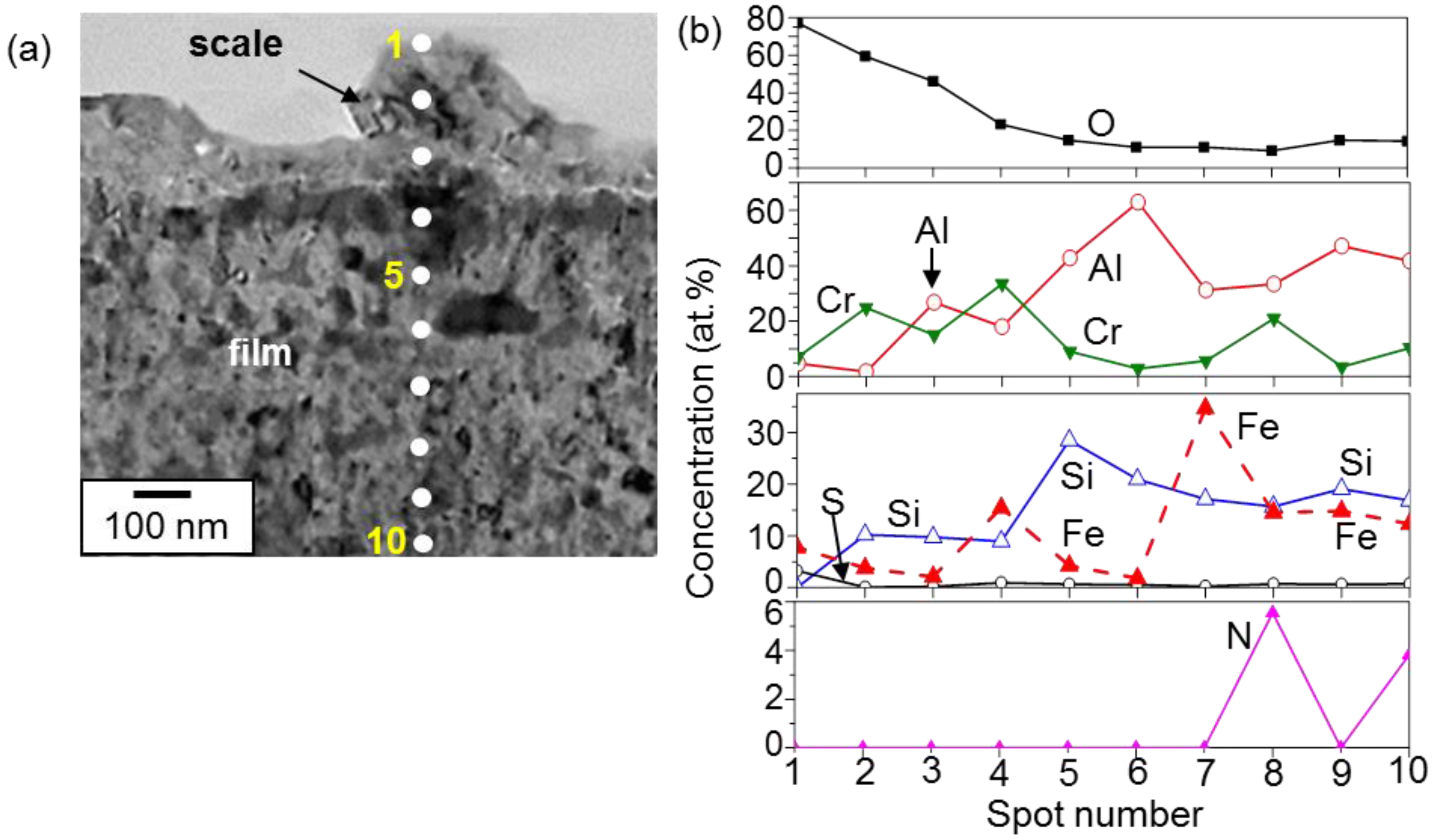
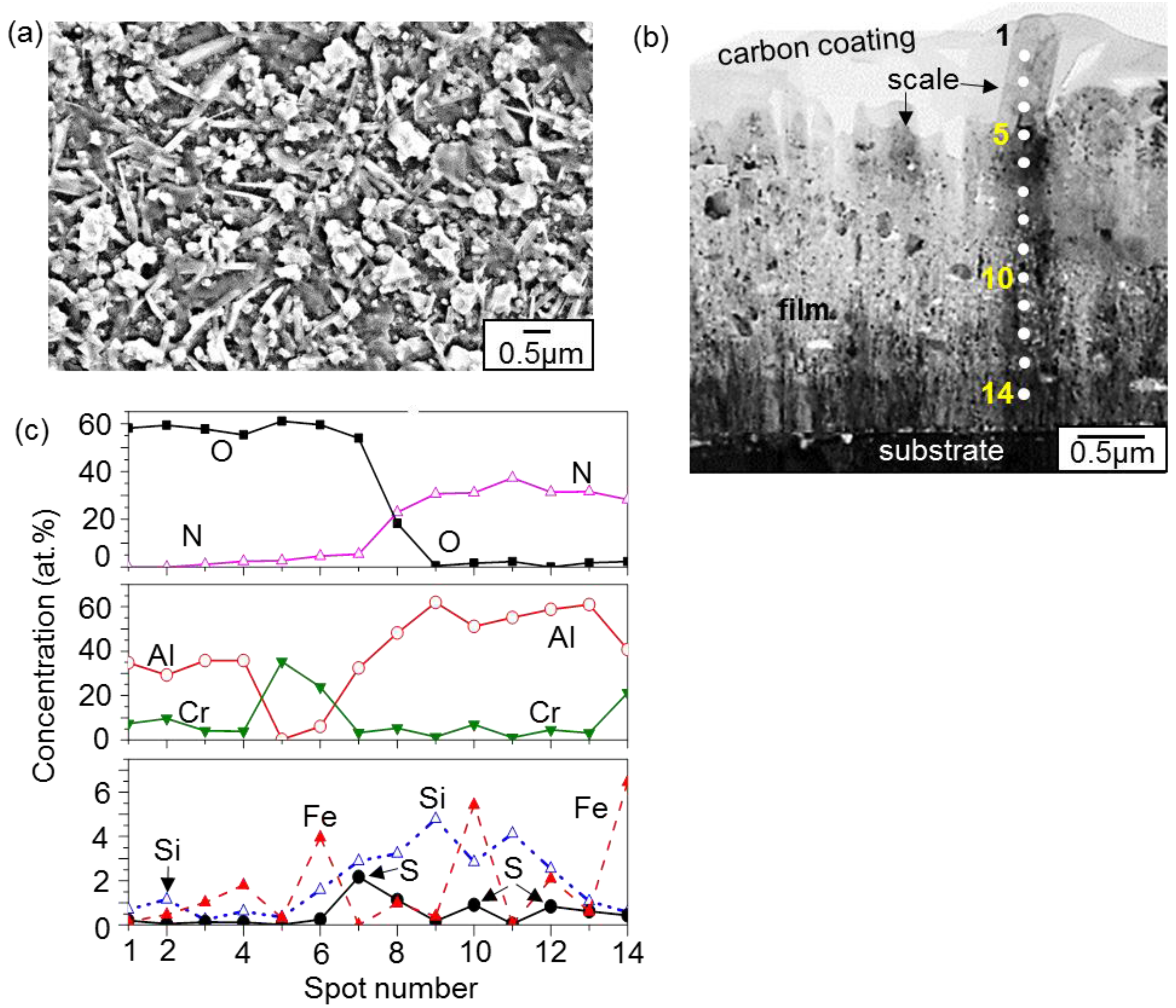
3. Results and Discussion
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lin, C.Y.; Lu, F.H. Oxidation behavior of AlN films at high temperature under controlled atmosphere. J. Eur. Ceram. Soc. 2008, 28, 691–698. [Google Scholar] [CrossRef]
- Fox-Rabinovich, G.S.; Beake, B.D.; Endrino, J.L.; Veldhuis, S.C.; Parkinson, R.; Shuster, L.S.; Migranov, M.S. Effect of mechanical properties measured at room and elevated temperatures on the wear resistance of cutting tools with TiAlN and AlCrN coatings. Surf. Coat. Technol. 2006, 200, 5738–5742. [Google Scholar] [CrossRef]
- Bourhis, E.L.; Goudeau, P.; Staia, M.H.; Carrasquero, E.; Puchi-Cabrera, E.S. Mechanical properties of hard AlCrN-based coated substrates. Surf. Coat. Technol. 2009, 203, 2961–2968. [Google Scholar] [CrossRef]
- Willmann, H.; Mayrhofer, P.H.; Persson, P.O.A.; Reiter, A.E.; Hultman, L.; Mitterer, C. Thermal stability of Al–Cr–N hard coatings. Scr. Mater. 2006, 54, 1847–1851. [Google Scholar] [CrossRef]
- Ding, X.Z.; Zeng, X.T. Structural, mechanical and tribological properties of CrAlN coatings deposited by reactive unbalanced magnetron sputtering. Surf. Coat. Technol. 2005, 200, 1372–1376. [Google Scholar] [CrossRef]
- Reiter, A.E.; Derflinger, V.H.; Hanselmann, B.; Bachmann, T.; Sartory, B. Investigation of the properties of Al1−xCrxN coatings prepared by cathodic arc evaporation. Surf. Coat. Technol. 2005, 200, 2114–2122. [Google Scholar] [CrossRef]
- Kawate, M.; Hashimoto, A.K.; Suzuki, T. Oxidation resistance of CrAlN and TiAlN films. Surf. Coat. Technol. 2003, 165, 163–167. [Google Scholar] [CrossRef]
- Hirai, M.; Ueno, Y.; Suzuki, T.; Jiang, W.; Grigoriu, C.; Yatsui, K. Characteristics of (Cr1−x, Alx)N films prepared by pulsed laser deposition. Jpn. J. Appl. Phys. 2001, 40, 1056–1060. [Google Scholar] [CrossRef]
- Banakh, O.; Schmid, P.E.; Sanjinés, R.; Lévy, F. High-temperature oxidation resistance of Cr1−xAlxN thin films deposited by reactive magnetron sputtering. Surf. Coat. Technol. 2003, 163, 57–61. [Google Scholar] [CrossRef]
- Wu, W.; Chen, W.; Yang, S.; Lin, Y.; Zhang, S.; Cho, T.Y.; Lee, G.H.; Kwon, S.C. Design of AlCrSiN multilayer and nanocomposite coating for HSS cutting tools. Appl. Surf. Sci. 2015, 351, 803–810. [Google Scholar] [CrossRef]
- Soldán, J.; Neidhardt, J.; Sartory, B.; Kaindl, R.; Čerstvý, R.; Mayrhofer, P.H.; Tessadri, R.; Polcik, P.; Lechthaler, M.; Mitterer, C. Structure–property relations of arc-evaporated Al–Cr–Si–N coatings. Surf. Coat. Technol. 2008, 202, 3555–3562. [Google Scholar] [CrossRef]
- Tritremmel, C.; Daniel, R.; Lechthaler, M.; Polcik, P.; Mitterer, C. Influence of Al and Si content on structure and mechanical properties of arc evaporated Al–Cr–Si–N thin films. Thin Solid Films 2013, 534, 403–409. [Google Scholar] [CrossRef]
- Bobzin, K.; Bagcivan, N.; Immich, P.; Bolz, S.; Cremer, R.; Leyendecker, T. Mechanical properties and oxidation behaviour of (Al, Cr)N and (Al, Cr, Si)N coatings for cutting tools deposited by HPPMS. Thin Solid Films 2008, 517, 1251–1256. [Google Scholar] [CrossRef]
- Endrino, J.L.; Fox-Rabinovich, G.S.; Reiter, A.; Veldhuis, S.V.; Galindo, R.E.; Albella, J.M.; Marco, J.F. Oxidation tuning in AlCrN coatings. Surf. Coat. Technol. 2005, 201, 4505–4511. [Google Scholar] [CrossRef]
- Chen, H.W.; Chan, Y.C.; Lee, J.W.; Duh, J.G. Oxidation behavior of Si-doped nanocomposite CrAlSiN coatings. Surf. Coat. Technol. 2010, 205, 1189–1194. [Google Scholar] [CrossRef]
- Endrino, J.L.; Palacín, S.; Aguirre, M.H.; Gutiérrez, A.; Schäfers, F. Determination of the local environment of silicon and the microstructure of quaternary CrAl(Si)N films. Acta Mater. 2007, 55, 2129–2135. [Google Scholar] [CrossRef]
- Polcar, T.; Cavaleiro, A. High temperature properties of CrAlN, CrAlSiN and AlCrSiN coatings—Structure and oxidation. Mater. Chem. Phys. 2011, 129, 195–201. [Google Scholar] [CrossRef]
- Birks, N.; Meier, G.H.; Pettit, F.S. Introduction to the High-Temperature Oxidation of Metals, 2nd ed.; Cambridge University Press: Cambridge, UK, 2006. [Google Scholar]
- Grabke, H.J. High temperature corrosion in complex, multi-reactant gaseous environments. In High Temperature Materials Corrosion in Coal Gasification Atmospheres; Norton, J.F., Ed.; Elsevier Applied Science Publishers: London, UK, 1984; pp. 59–82. [Google Scholar]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yadav, P.; Lee, D.B.; Lin, Y.; Zhang, S.; Kwon, S.C. High-Temperature Corrosion of AlCrSiN Film in Ar-1%SO2 Gas. Coatings 2017, 7, 44. https://doi.org/10.3390/coatings7030044
Yadav P, Lee DB, Lin Y, Zhang S, Kwon SC. High-Temperature Corrosion of AlCrSiN Film in Ar-1%SO2 Gas. Coatings. 2017; 7(3):44. https://doi.org/10.3390/coatings7030044
Chicago/Turabian StyleYadav, Poonam, Dong Bok Lee, Yue Lin, Shihong Zhang, and Sik Chol Kwon. 2017. "High-Temperature Corrosion of AlCrSiN Film in Ar-1%SO2 Gas" Coatings 7, no. 3: 44. https://doi.org/10.3390/coatings7030044
APA StyleYadav, P., Lee, D. B., Lin, Y., Zhang, S., & Kwon, S. C. (2017). High-Temperature Corrosion of AlCrSiN Film in Ar-1%SO2 Gas. Coatings, 7(3), 44. https://doi.org/10.3390/coatings7030044





