Pitted Corrosion Detection of Thermal Sprayed Metallic Coatings Using Fiber Bragg Grating Sensors
Abstract
:1. Introduction
2. Operational Principles
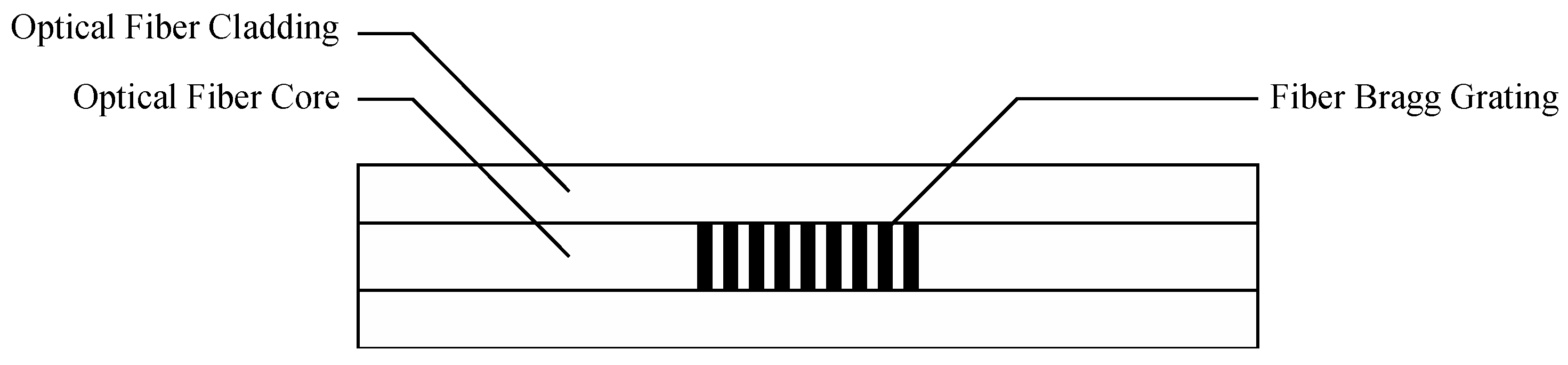
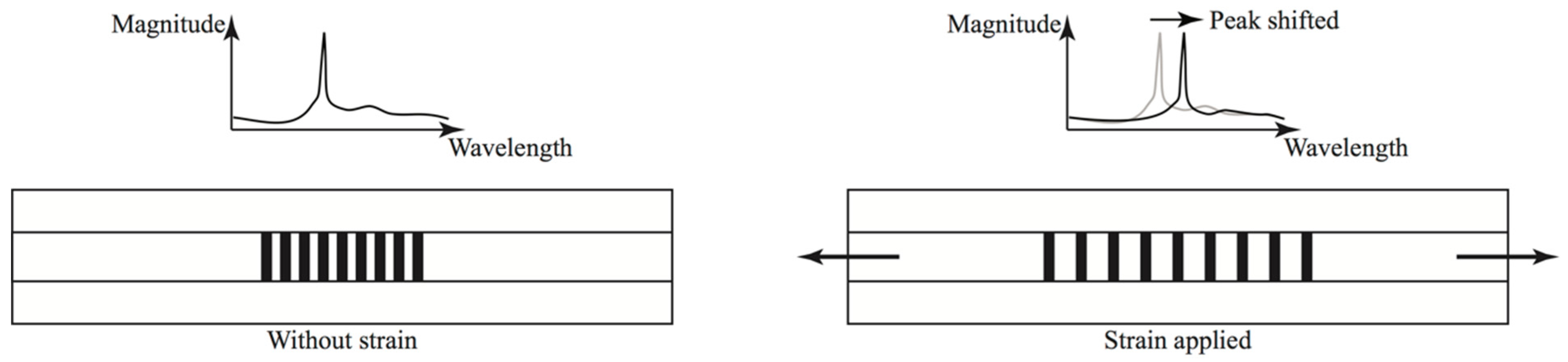
2.1. Principle of FBG Sensor
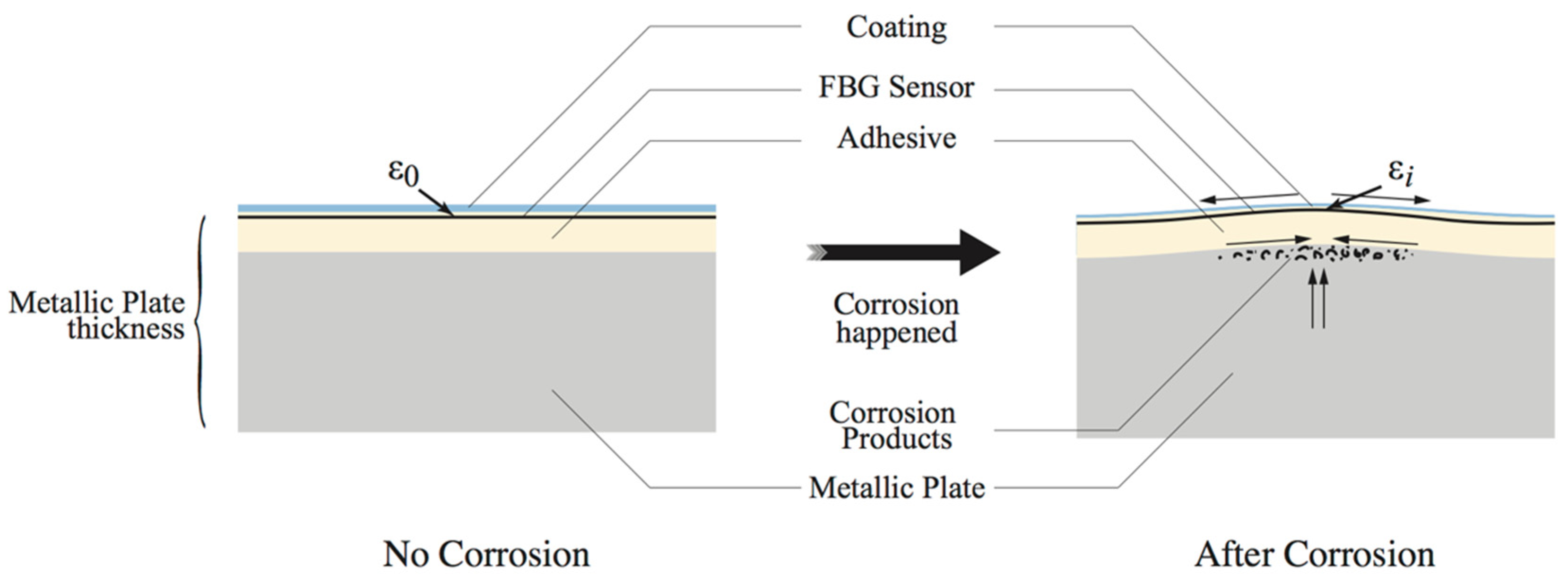
2.2. Operational Principle of the Corrosion and Crack Sensing in Coatings Using Embedded FBG Sensors
- The corrosion analyzed in this paper is pitted (localized) corrosion so its corrosion production is accumulated within a relatively small area comparing to the total span of the packaged FBG sensor;
- The expansion of corrosion productions mainly occurs in vertical direction.
2.3. Sensor Design
3. Experimental Section
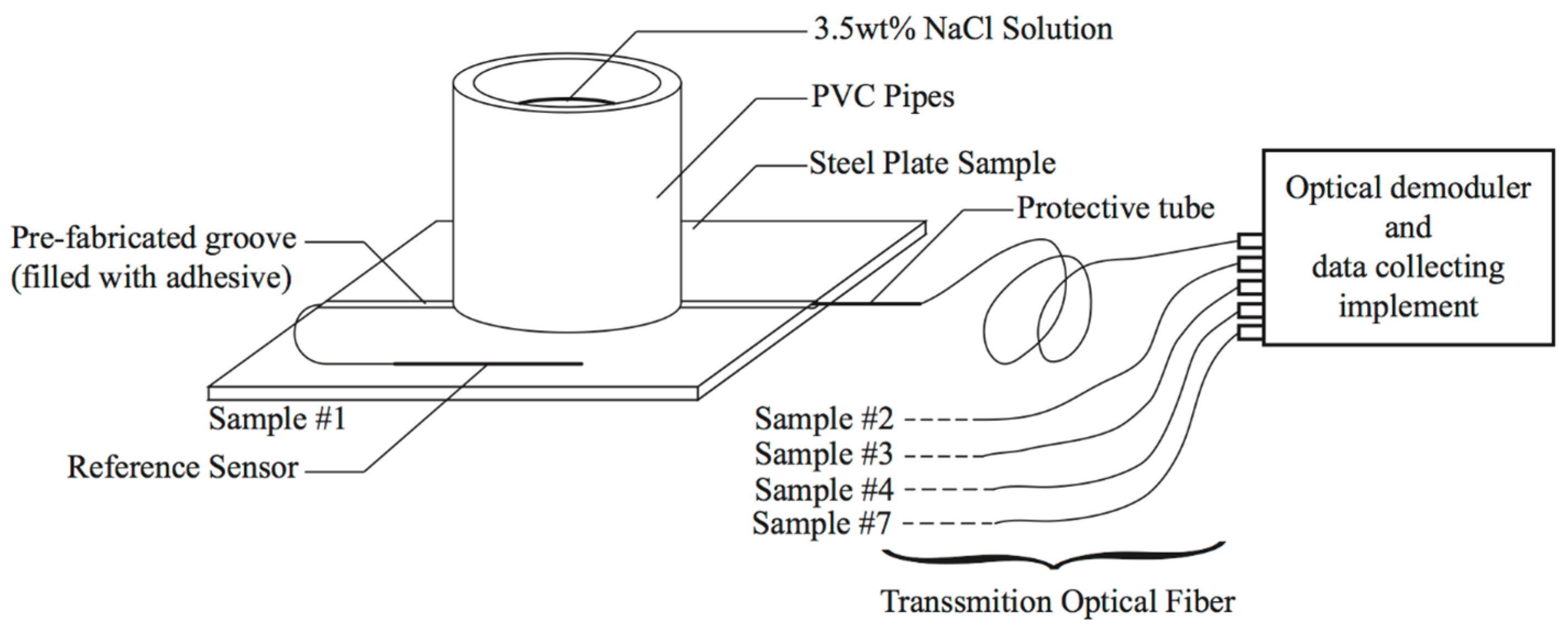
3.1. Sample Preparation
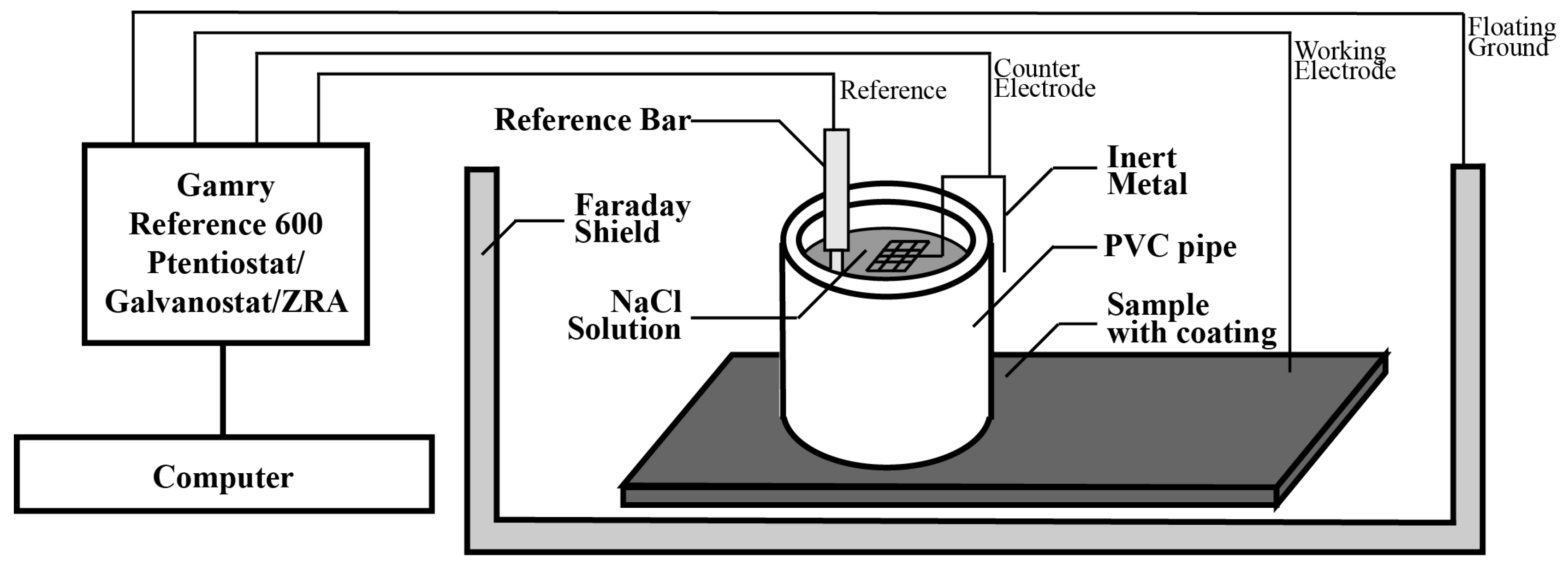
3.2. Corrosion Rate Measurement Using Electrochemical Approach
3.3. Experimental Setup for Accelerated Corrosion Test
4. Experimental Results and Discussion
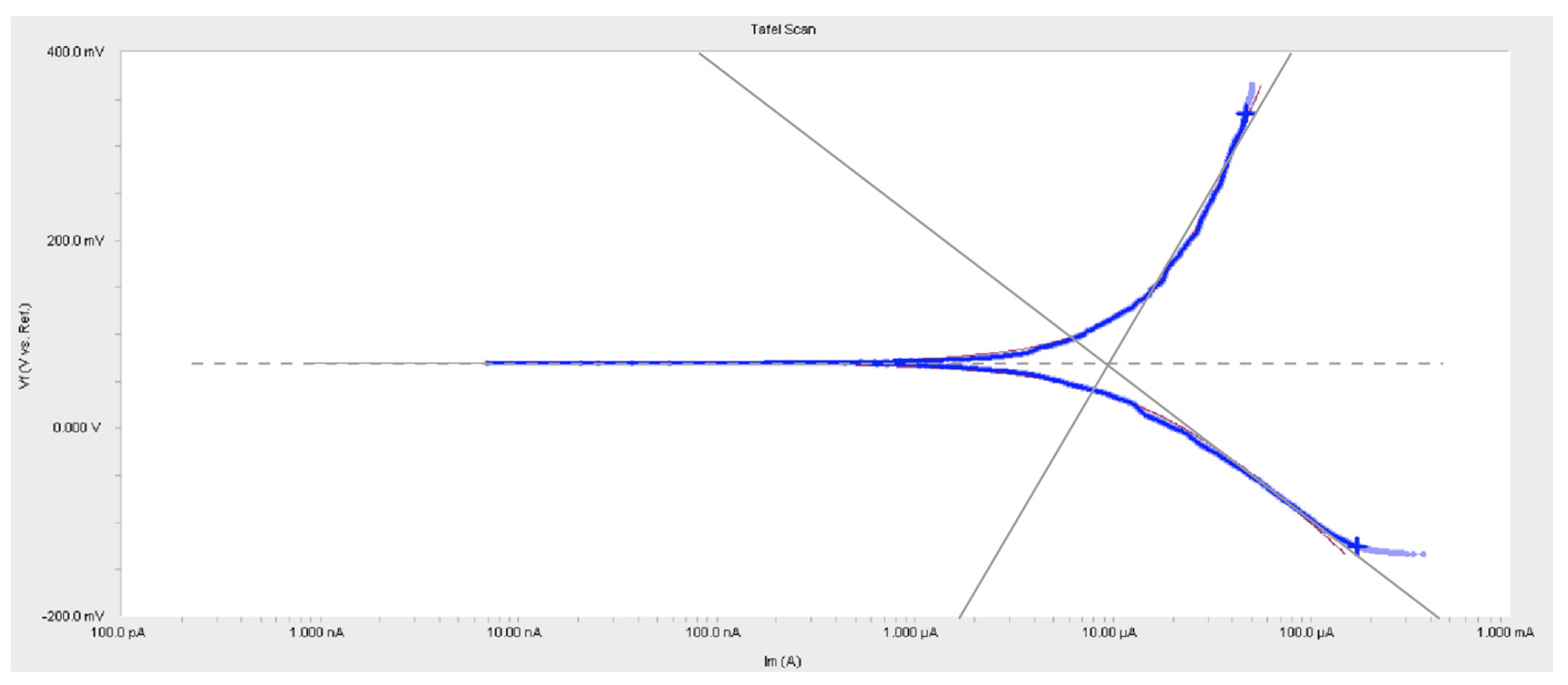
4.1. Experimental Results from Electrochemical Method
4.2. Experimental Results from Accelerated Corrosion Tests Using Embedded FBG Sensors
4.3. Discussion and Data Analysis
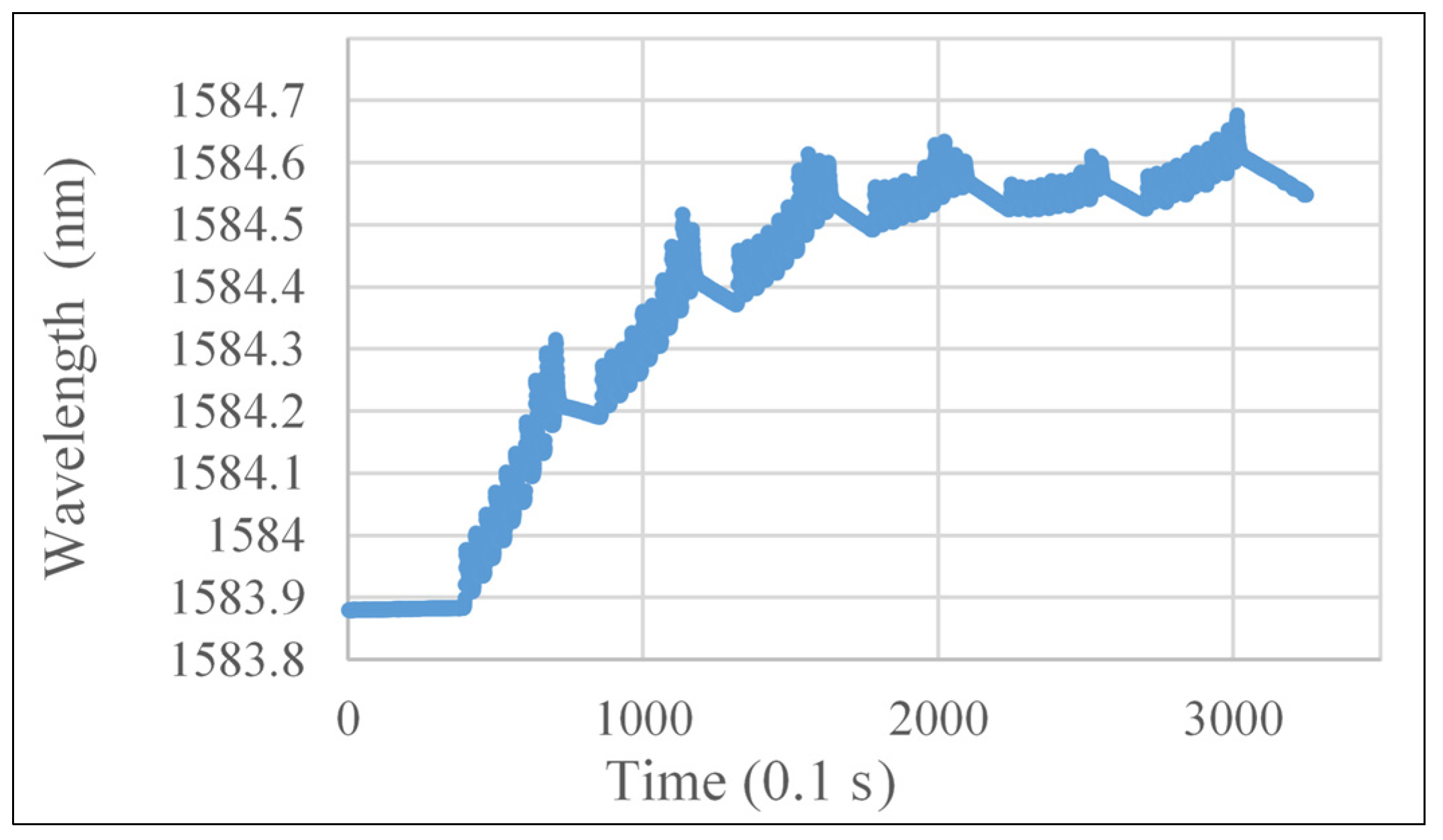
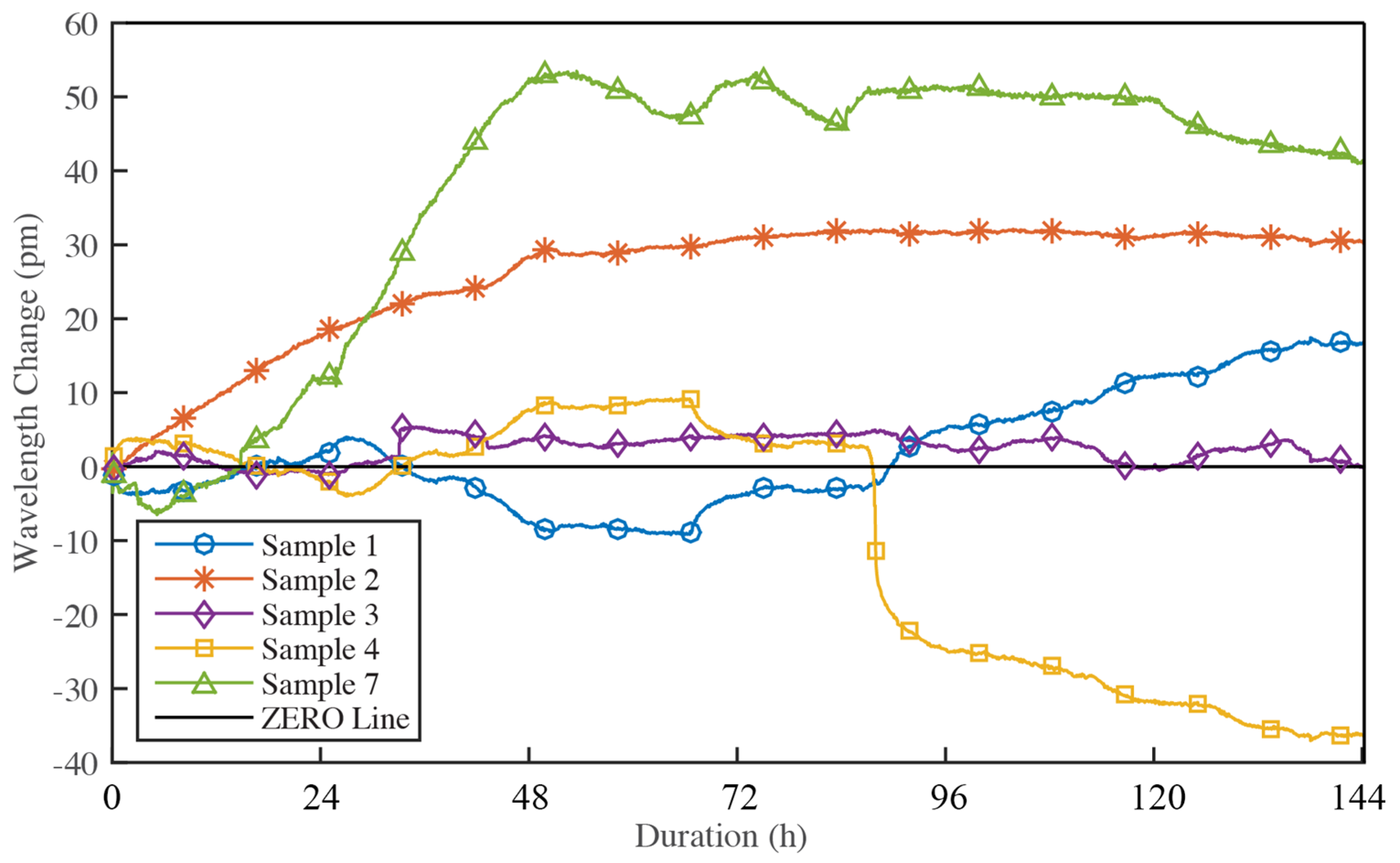
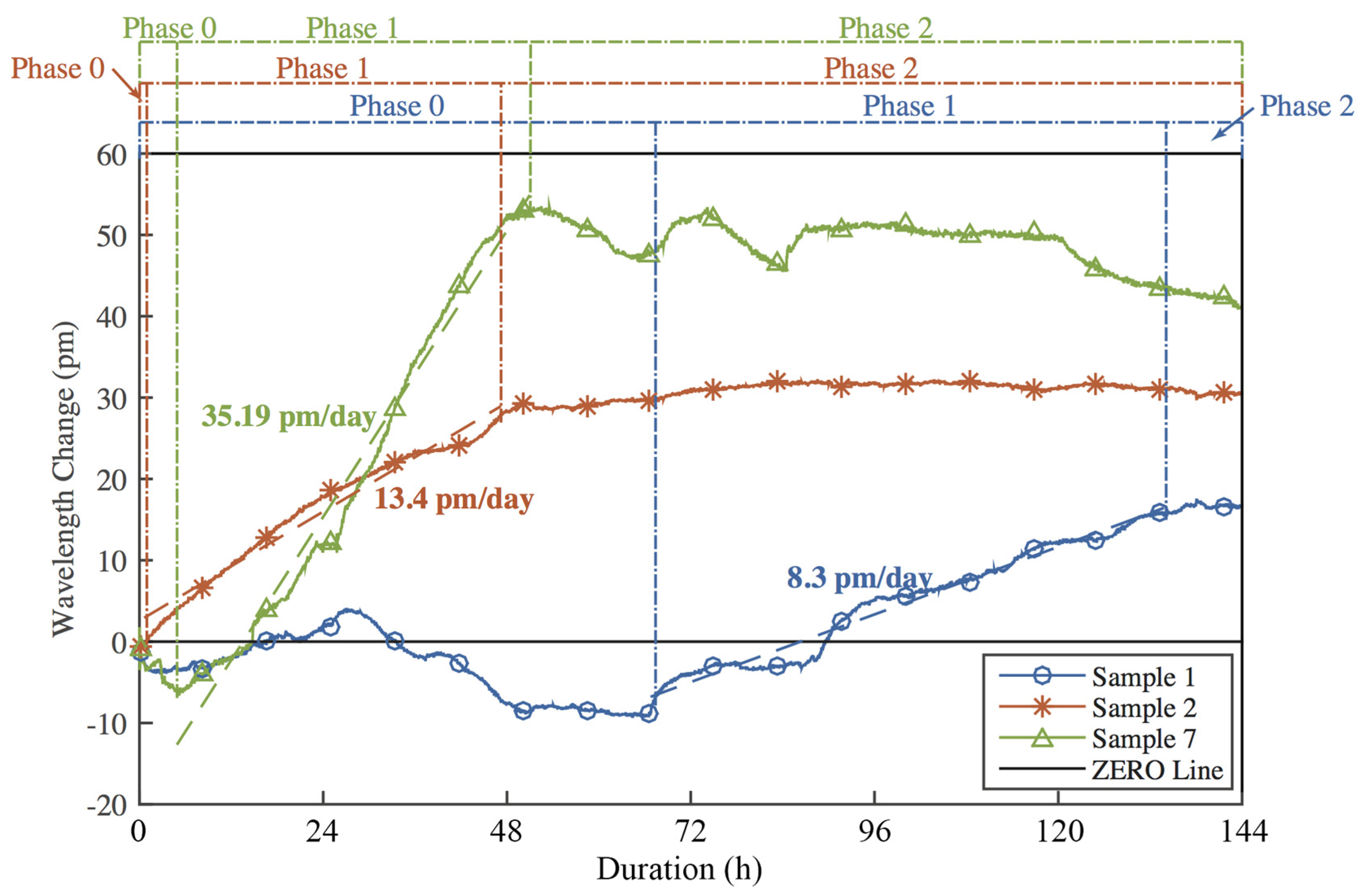
- In Phase 0 (short-term influences phase), the corrosion is initialized and corrosion products start to fill the pores between adhesive and the FBG sensors. As a result, compression strains are observed on FBG sensors, introducing a drop of Bragg wavelengths of all FBG sensors on all three samples shown in Figure 19.
- In Phase 1 (high corrosion rate phase), oxygen surrounded at corrosion area is consumed and more oxygen is rapidly absorbed in water, which results in high corrosion rate of the material. Due to principles discussed in Section 2, corrosion products tend to lift the embedded FBG sensors as a simply supported beam, causing an increase in Bragg wavelengths following Equations (3)–(5) in Section 2. Thus, the slope of Bragg wavelength change in Phase 1 reflects the production rate of corrosion products, which is the expected corrosion rate in Equation (6). Sample #7 with bare steel has a big corrosion rate slope of 35.19 pm/day during Phase 1. Samples #1 and #2 with thermal sprayed composite coatings have a smaller corrosion rate slope of 8.3 and 13.4 pm/day in Phase 1, respectively. This result indicates that the thermal sprayed composite coating used in this study has a higher corrosion resistance when compared with bare steel. To estimate the corrosion related parameters in Equation (6), we take a look at the corrosion rate of Sample #7, the bare steel without coating. The measured corrosion rate of the bare steel using electrochemical method yield to 1.5 mil/year and the corrosion rate slope of the Bragg wavelength change of the embedded FBG is 35.19 pm/day. Thus, the sensitivity of the embedded FBG sensor for corrosion rate measurements, α, can be determined as
5. Conclusions
- A simply supported beam theory can be used to analyze the operational principle of the response of an embedded FBG sensor to corrosion developed in or under coatings.
- The embedded FBG sensors can successfully measure the corrosion progressing of the thermal sprayed coatings and the bare steel through monitoring the Bragg wavelength changes of the FBG sensors.
- Accelerated corrosion tests showed a three-phase phenomenon of the corrosion process of the thermal sprayed composite coatings used in this study and the corrosion rate can be calculated through the slope of Phase 1. The obtained corrosion rate of 0.354 and 0.571 mil/year for thermal sprayed coating matches well with that from the electrochemical method of 0.5054 mil/year.
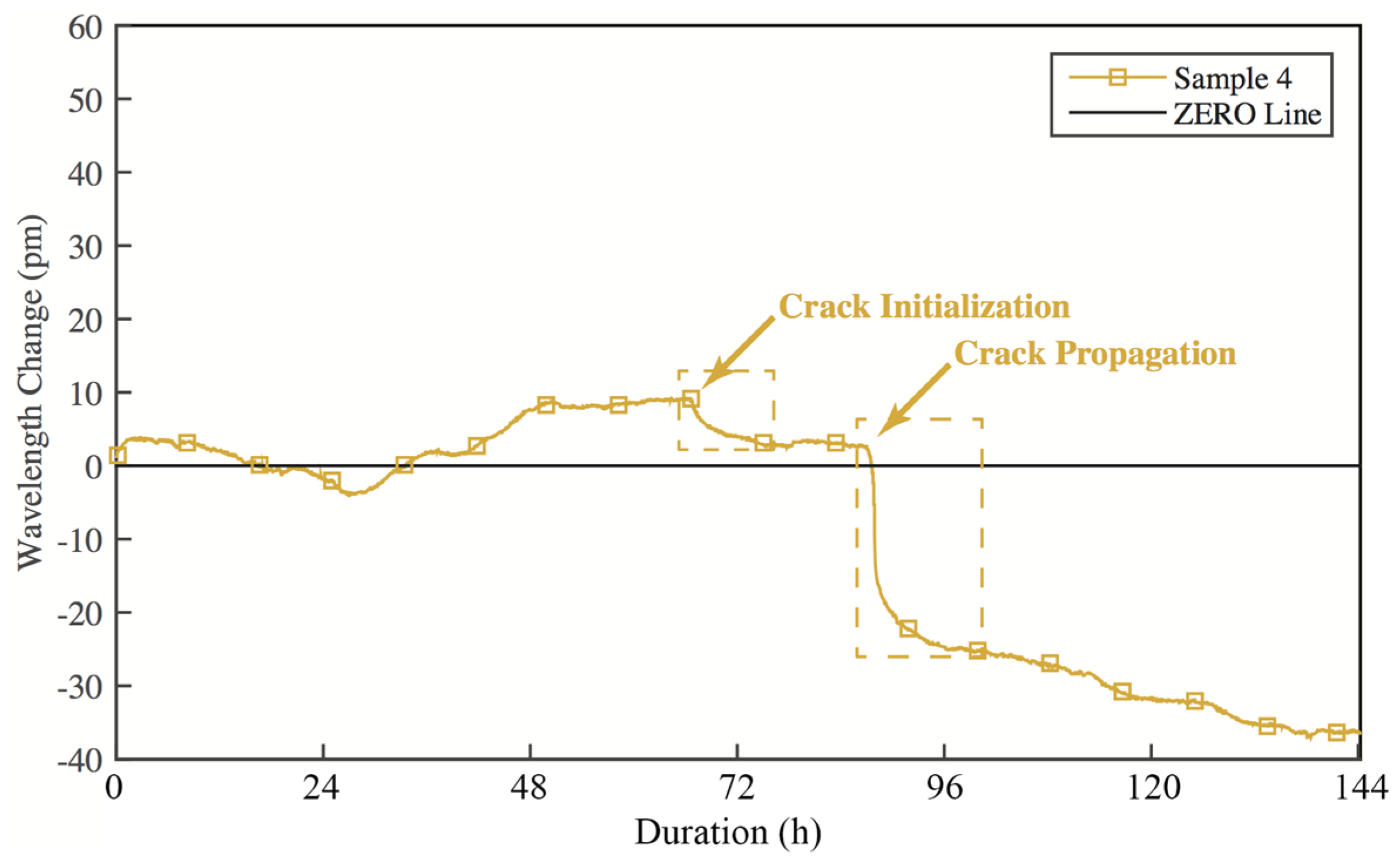
- The embedded FBG sensors can identify the corrosion induced cracks in coating successfully as shown from the laboratory tests.
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Glass, G.; Page, C.; Short, N. Factors affecting the corrosion rate of steel in carbonated mortars. Corros. Sci. 1991, 32, 1283–1294. [Google Scholar] [CrossRef]
- Southwell, C.; Bultman, J.; Alexander, A. Corrosion of Metals in Tropical Environments; Final Report of 16-Year Exposures; National Association of Corrosion Engineers: Houston, TE, USA, 1976. [Google Scholar]
- Andrade, C.; Alonso, C. Corrosion rate monitoring in the laboratory and on-site. Constr. Build. Mater. 1996, 10, 315–328. [Google Scholar] [CrossRef]
- Melchers, R.E.; Jeffrey, R. Early corrosion of mild steel in seawater. Corros. Sci. 2005, 47, 1678–1693. [Google Scholar] [CrossRef]
- Bach, F.-W.; Möhwald, K.; Laarmann, A.; Wenz, T. Modern Surface Technology; John Wiley & Sons: New York, NK, USA, 2006. [Google Scholar]
- Kendig, M.; Scully, J. Basic aspects of electrochemical impedance application for the life prediction of organic coatings on metals. Corrosion 1990, 46, 22–29. [Google Scholar] [CrossRef]
- Sørensen, P.A.; Kiil, S.; Dam-Johansen, K.; Weinell, C. Anticorrosive coatings: A review. J. Coat. Technol. Res. 2009, 6, 135–176. [Google Scholar] [CrossRef]
- Rout, T.; Jha, G.; Singh, A.; Bandyopadhyay, N.; Mohanty, O. Development of conducting polyaniline coating: A novel approach to superior corrosion resistance. Surf. Coat. Technol. 2003, 167, 16–24. [Google Scholar] [CrossRef]
- Hauert, R.; Patscheider, J. From alloying to nanocomposites—Improved performance of hard coatings. Adv. Eng. Mater. 2000, 2, 247–259. [Google Scholar] [CrossRef]
- Matthews, S.; James, B. Review of thermal spray coating applications in the steel industry: Part 1—Hardware in steel making to the continuous annealing process. J. Therm. Spray Technol. 2010, 19, 1267–1276. [Google Scholar] [CrossRef]
- Matthews, S.; James, B. Review of thermal spray coating applications in the steel industry: Part 2—Zinc pot hardware in the continuous galvanizing line. J. Therm. Spray Technol. 2010, 19, 1277–1286. [Google Scholar] [CrossRef]
- Davis, J.R. Handbook of Thermal Spray Technology; ASM International: Almere, The Netherlands, 2004. [Google Scholar]
- Fauchais, P.; Vardelle, A. Thermal Sprayed Coatings Used against Corrosion and Corrosive Wear; INTECH Open Access Publisher: Rijeka, Croatia, 2012. [Google Scholar]
- Shibli, S.; Meena, B.; Remya, R. A review on recent approaches in the field of hot dip zinc galvanizing process. Surf. Coat. Technol. 2015, 262, 210–215. [Google Scholar] [CrossRef]
- Tzimas, E.; Papadimitriou, G. Cracking mechanisms in high temperature hot-dip galvanized coatings. Surf. Coat. Technol. 2001, 145, 176–185. [Google Scholar] [CrossRef]
- Szymański, K.; Hernas, A.; Moskal, G.; Myalska, H. Thermally sprayed coatings resistant to erosion and corrosion for power plant boilers—A review. Surf. Coat. Technol. 2015, 268, 153–164. [Google Scholar] [CrossRef]
- Kuroda, S.; Kawakita, J.; Watanabe, M.; Katanoda, H. Warm spraying—A novel coating process based on high-velocity impact of solid particles. Sci. Technol. Adv. Mater. 2016, 9, 033002. [Google Scholar] [CrossRef] [PubMed]
- Mahbub, H. High Velocity Oxy-Fuel (HVOF) Thermal Spray Deposition of Functionally Graded Coatings; Dublin City University: Dublin, Ireland, 2005. [Google Scholar]
- Toma, D.; Brandl, W.; Marginean, G. Wear and corrosion behaviour of thermally sprayed cermet coatings. Surf. Coat. Technol. 2001, 138, 149–158. [Google Scholar] [CrossRef]
- Guilemany, J.; Fernandez, J.; Delgado, J.; Benedetti, A.V.; Climent, F. Effects of thickness coating on the electrochemical behaviour of thermal spray Cr3C2–NiCr coatings. Surf. Coat. Technol. 2002, 153, 107–113. [Google Scholar] [CrossRef]
- Miguel, J.; Guilemany, J.; Mellor, B.; Xu, Y. Acoustic emission study on WC-Co thermal sprayed coatings. Mater. Sci. Eng. A 2003, 352, 55–63. [Google Scholar] [CrossRef]
- Lin, C.-K.; Berndt, C. Measurement and analysis of adhesion strength for thermally sprayed coatings. J. Therm. Spray Technol. 1994, 3, 75–104. [Google Scholar] [CrossRef]
- Steffens, H.-D.; Crostack, H.-A. Methods based on ultrasound and optics for the non-destructive inspection of thermally sprayed coatings. Thin Solid Films 1981, 83, 325–342. [Google Scholar] [CrossRef]
- Rosa, G.; Oltra, R.; Nadal, M.-H. Evaluation of the coating–substrate adhesion by laser-ultrasonics: Modeling and experiments. J. Appl. Phys. 2002, 91, 6744–6753. [Google Scholar] [CrossRef]
- Bescond, C.; Kruger, S.; Lévesque, D.; Lima, R.; Marple, B. In situ simultaneous measurement of thickness, elastic moduli and density of thermal sprayed WC-Co coatings by laser-ultrasonics. J. Therm. Spray Technol. 2007, 16, 238–244. [Google Scholar] [CrossRef]
- Lakestani, F.; Coste, J.-F.; Denis, R. Application of ultrasonic Rayleigh waves to thickness measurement of metallic coatings. NDT E Int. 1995, 28, 171–178. [Google Scholar] [CrossRef]
- Friebele, E.J. Fiber Bragg grating strain sensors: Present and future applications in smart structures. Opt. Photonics News 1998, 9, 33. [Google Scholar] [CrossRef]
- Moyo, P.; Brownjohn, J.; Suresh, R.; Tjin, S. Development of fiber Bragg grating sensors for monitoring civil infrastructure. Eng. Struct. 2005, 27, 1828–1834. [Google Scholar] [CrossRef]
- Guo, Z.-S. Strain and temperature monitoring of asymmetric composite laminate using FBG hybrid sensors. Struct. Health Monit. 2007, 6, 191–197. [Google Scholar] [CrossRef]
- Zhang, B.; Kahrizi, M. High-temperature resistance fiber Bragg grating temperature sensor fabrication. IEEE Sens. J. 2007, 7, 586–591. [Google Scholar] [CrossRef]
- Dewynter-Marty, V.; Ferdinand, P.; Bocherens, E.; Carbone, R.; Beranger, H.; Bourasseau, S. Embedded fiber Bragg grating sensors for industrial composite cure monitoring. J. Intell. Mater. Syst. Struct. 1998, 9, 785–787. [Google Scholar] [CrossRef]
- Betz, D.; Staszewski, W.; Thursby, G.; Culshaw, B. Multi-functional fibre Bragg grating sensors for fatigue crack detection in metallic structures. Proc. Inst. Mech. Eng. G J. Aerosp. Eng. 2006, 220, 453–461. [Google Scholar] [CrossRef]
- Kuang, K.; Cantwell, W.; Thomas, C. Crack detection and vertical deflection monitoring in concrete beams using plastic optical fibre sensors. Meas. Sci. Technol. 2003, 14, 205. [Google Scholar] [CrossRef]
- Kirkby, E.; de Oliveira, R.; Michaud, V.; Manson, J. Impact localisation with FBG for a self-healing carbon fibre composite structure. Compos. Struct. 2011, 94, 8–14. [Google Scholar] [CrossRef]
- Tsuda, H.; Lee, J.-R.; Guan, Y.; Takatsubo, J. Investigation of fatigue crack in stainless steel using a mobile fiber Bragg grating ultrasonic sensor. Opt. Fiber Technol. 2007, 13, 209–214. [Google Scholar] [CrossRef]
- Zheng, Z.; Sun, X.; Lei, Y. Monitoring corrosion of reinforcement in concrete structures via fiber Bragg grating sensors. Front. Mech. Eng. China 2009, 4, 316–319. [Google Scholar] [CrossRef]
- Gao, J.; Wu, J.; Li, J.; Zhao, X. Monitoring of corrosion in reinforced concrete structure using Bragg grating sensing. NDT E Int. 2011, 44, 202–205. [Google Scholar] [CrossRef]
- Hassan, M.R.A.; Bakar, M.H.A.; Dambul, K.; Adikan, F.R.M. Optical-based sensors for monitoring corrosion of reinforcement rebar via an etched cladding Bragg grating. Sensors 2012, 12, 15820–15826. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-R.; Yun, C.-Y.; Yoon, D.-J. A structural corrosion-monitoring sensor based on a pair of prestrained fiber Bragg gratings. Meas. Sci. Technol. 2009, 21, 017002. [Google Scholar] [CrossRef]
- Hu, W.; Cai, H.; Yang, M.; Tong, X.; Zhou, C.; Chen, W. Fe–C-coated fibre Bragg grating sensor for steel corrosion monitoring. Corros. Sci. 2011, 53, 1933–1938. [Google Scholar] [CrossRef]
- Fontana, M.; Greene, N. Corrosion Engineering, 3rd ed.; McGraw-Hill Book Company: New York, NY, USA, 1987. [Google Scholar]
- Gangopadhyay, T.K.; Majumder, M.; Chakraborty, A.K.; Dikshit, A.K.; Bhattacharya, D.K. Fibre Bragg grating strain sensor and study of its packaging material for use in critical analysis on steel structure. Sens. Actuators Phys. 2009, 150, 78–86. [Google Scholar] [CrossRef]
- Popov, B.; White, R. Electrochemical and Corrosion Experimental Techniques. Notes USC, Gamry Instruments Technical Report. Available online: https://www.gamry.com/application-notes/corrosion-coatings/basics-of-electrochemical-corrosion-measurements/ (accessed on 22 February 2017).










| Sample Number | Anodic Tafel Constant, βa, (V/Decade) | Cathodic Tafel Constant, βa, (V/Decade) | Polarization Resistance (kΩ) | Corrosion Current (amps) | Corrosion Rate (mil/Year) |
|---|---|---|---|---|---|
| Sample #4 | 0.5348 | 0.2047 | 2.3 | 2.798 × 10−5 | 0.5054 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Deng, F.; Huang, Y.; Azarmi, F.; Wang, Y. Pitted Corrosion Detection of Thermal Sprayed Metallic Coatings Using Fiber Bragg Grating Sensors. Coatings 2017, 7, 35. https://doi.org/10.3390/coatings7030035
Deng F, Huang Y, Azarmi F, Wang Y. Pitted Corrosion Detection of Thermal Sprayed Metallic Coatings Using Fiber Bragg Grating Sensors. Coatings. 2017; 7(3):35. https://doi.org/10.3390/coatings7030035
Chicago/Turabian StyleDeng, Fodan, Ying Huang, Fardad Azarmi, and Yechun Wang. 2017. "Pitted Corrosion Detection of Thermal Sprayed Metallic Coatings Using Fiber Bragg Grating Sensors" Coatings 7, no. 3: 35. https://doi.org/10.3390/coatings7030035
APA StyleDeng, F., Huang, Y., Azarmi, F., & Wang, Y. (2017). Pitted Corrosion Detection of Thermal Sprayed Metallic Coatings Using Fiber Bragg Grating Sensors. Coatings, 7(3), 35. https://doi.org/10.3390/coatings7030035






