Hybrid Organic–Inorganic Materials Prepared by Sol–Gel and Sol–Gel-Coating Method for Biomedical Use: Study and Synthetic Review of Synthesis and Properties
Abstract
1. Introduction
- Antioxidants: Rich in antioxidants, plant extracts aid in the body’s defense against free radicals [21]. These substances have the ability to lower oxidative stress and shield cells from harm.
- Anti-inflammatory: A few plant extracts have been shown to have anti-inflammatory abilities, which may aid in lowering bodily inflammation. Treating inflammatory diseases and enhancing general health may benefit from this [22].
- Antimicrobial: Certain plant extracts may be able to combat bacteria, fungi, and other diseases thanks to antimicrobial qualities. These qualities can be used for sanitary and medical purposes [23].
- Anticancer: Research has been conducted on some plant extracts that may have the ability to stop the development of cancer cells or reduce their expansion [24].
- Antivirals: Certain plant extracts may include antiviral properties that aid in the defense against viral infections and bolster the immune system [25].
- Cardioprotective: By controlling blood pressure, lowering cholesterol, and exhibiting other cardioprotective properties, plant extracts may be beneficial to the heart and lower the risk of heart disease [26].
- Antidiabetics: A few plant extracts have the ability to control blood sugar levels, which may be advantageous for diabetic diseases [27].
- Neuroprotective: Certain plant extracts may have the ability to protect nerve cells and maintain brain health [28].
2. Biomaterials
2.1. Characteristics and Properties
- Biocompatibility: These materials need to work with the biological system without posing a risk of negative responses. To prevent the body from rejecting something or reacting in an inflammatory manner, biocompatibility is crucial. In fact, certain biomaterials have the ability to precisely control the biological response, affecting things like cell adhesion, cell division, and blood vessel creation [40].
- The capacity to disintegrate naturally over time, which might be favorable in some situations and allow for a progressive replacement of the biomaterial with the surrounding biological tissue. This is particularly crucial in situations when the material must permanently integrate with the body, such as in temporary applications [41].
- Mechanical characteristics: biomaterials need suitable mechanical characteristics to carry out their intended purpose. Orthopedic implants, for instance, need to be robust enough to endure the mechanical strain of the surrounding bone tissue [42].
- A wide range of materials, including composites, metals, polymers, and ceramics, may be used to create many kinds of biomaterials. Every type of material has distinct qualities that make it appropriate for particular uses. In fact, biomaterials are used in a wide range of applications, such as prosthetics, dental implants, medical devices, scaffolds for tissue regeneration, drug delivery systems, and much more [43,44].
- Continuous work is in progress because research on biomaterials is constantly evolving to improve the performance, safety, and durability of materials used in medicine and biology.
2.2. Different Classes
- Polymeric materials are separated into two categories: synthetic materials and natural materials. The former are taken out of biological materials including alginate, cellulose, and collagen [46,47]. Synthetic materials such as polyethylene, polyurethane, and polytetrafluoroethylene (PTFE) are illegal in laboratories.
- Bioinert materials, like Teflon, which do not significantly alter bodily processes [60].

2.3. Organic–Inorganic Hybrids
3. Sol–Gel Technique: An Innovative Process
3.1. From Concept to Application
3.2. Sol–Gel Process
- Solution preparation (Sol): the first step consists of the preparation of a solution containing inorganic precursors, such as metallic or alkylated salts. These precursors are soluble in an organic or aqueous solvent.
- Hydrolysis and condensation: the process of hydrolysis involves hydrogen atoms in water reacting with the oxides of inorganic precursors to break chemical bonds and generate hydrolyzed oxides. This occurs when the oxides or precursors are in the solution. The hydrolyzed oxides then proceed through condensation, where they join forces to create stronger connections. During this phase, an inorganic particle network forms in three dimensions, giving the gel its structure.
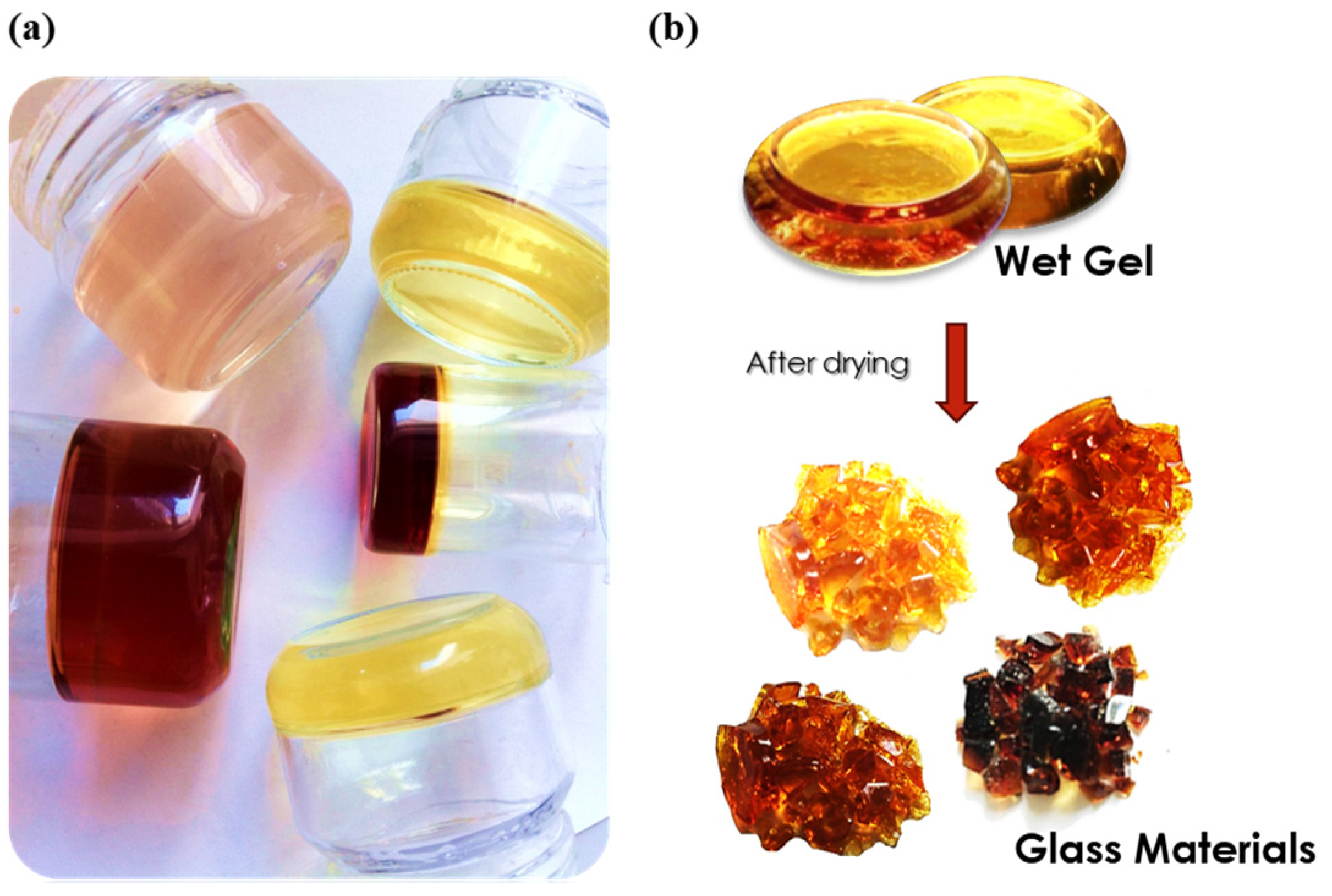
- Gelation: in the process of gelation, the liquid solution turns into a three-dimensional gel with the particles dispersed across a continuous matrix.
- Drying: the resultant gel can be dried in order to release any trapped water, creating xerogel, a porous solid substance.
- Subsequent treatments: the material can go through additional heat or chemical treatments to develop the structure and gain certain qualities like crystallinity, mechanical resistance, or porosity reduction, depending on the intended uses.

3.3. Advantages and Disadvantages of the Sol–Gel Method
- Materials with particular qualities may be synthesized thanks to the exact control over the chemical composition of the final product provided by the sol–gel process.
- Many of the reactions involved in the sol–gel technique take place at relatively low temperatures, allowing for the production of materials in a more energetic and economical way than other synthesis methods. In particular, it is possible to incorporate vegetal drugs and thermolabile drugs into the inorganic matrix of interest [98].
- The versatility of the technique allows for the production of a wide range of materials, including glasses, ceramics, coatings, catalysts, and biomaterials.
- The material’s consistent particle distribution brought about by the gel’s production aids in the creation of a homogenous structure.
- The options for material design are increased by the integration of organic components into the inorganic matrix made possible by the sol–gel process.
- Lastly, by adjusting the process parameters, the porosity of the material may be regulated, producing porous materials like aerogels or xerogels [99].
- Production time and productivity may be impacted by the lengthy curing periods needed for some sol–gel techniques.
- Applying the sol–gel technology to large-scale industrial manufacturing can be difficult, and issues with the repeatability and consistency of the process may occur [100].
- Impurities in the surroundings or the solution may have a detrimental effect on the final product’s quality.
- When using exceptionally rare or pure materials, the cost of inorganic precursors might be high.
- Trying to create complicated objects or big structures might be limited by gel formation.
3.4. Combination of the Sol–Gel Method with Other Modern Techniques
3.5. Uses, Properties, and Comparisons between Sol–Gel Biomaterials
- -
- Creating biocompatible coatings on medical devices, implants, or prostheses. By enhancing the material’s contact with the surrounding biological tissues, these coatings lower the possibility of negative reactions and hasten the healing process [123].
- -
- The sol–gel matrix can be created to include medicines or other bioactive compounds. This makes it possible for medicinal compounds to be released gradually and under control, increasing treatment efficacy and minimizing negative effects [124].
- -
- Three-dimensional scaffolds can be built for tissue engineering using sol–gel biomaterials. These scaffolds aid in the repair of missing or injured tissue by offering short-term structural support for the proliferation and differentiation of cells [125].
- -
- The development of biomimetic sensors, which are able to identify particular chemical or biological substances, is made possible by the adaptability of the sol–gel process. In the medical field, these sensors can be applied for monitoring or diagnostic purposes [126].
- -
- The potential of sol–gel biomaterials for bone and dental tissue regeneration has been investigated. They can be made to resemble the properties of the extracellular matrix, which will promote the development of tooth or bone cells [127].
- -
- One of the most popular sol–gel biomaterials is silica-based sol–gel bio-glass [129]. It is utilized in coatings for dental and orthopedic implants and has strong biocompatibility.
- -
- Antibacterial coatings and dental prostheses are two uses for sol–gel titanium dioxide [130].
- -
- The sol–gel process can be used to create hydroxyapatite, a key element of the bone matrix that is used in bone regeneration [131].
- -
- Hydrogels made using the sol–gel method might act as scaffolds for tissue engineering or utilized as drug delivery systems [132].
4. Properties of Sol–Gel Materials
4.1. Bioactive Materials
4.2. Antioxidant Activity
4.3. Antibacterial Activities
4.4. Biocompatibility
5. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Eshraghi, A.A.; Gupta, C.; Ozdamar, O.; Balkany, T.J.; Truy, E.; Nazarian, R. Biomedical engineering principles of modern cochlear implants and recent surgical innovations. Anat. Rec. Adv. Integr. Anat. Evol. Biol. 2012, 295, 1957–1966. [Google Scholar] [CrossRef] [PubMed]
- Pecchia, L.; Pallikarakis, N.; Magjarevic, R.; Iadanza, E. Health technology assessment and biomedical engineering: Global trends, gaps and opportunities. Med. Eng. Phys. 2019, 72, 19–26. [Google Scholar] [CrossRef] [PubMed]
- Williams, D.F. On the nature of biomaterials. Biomaterials 2009, 30, 5897–5909. [Google Scholar] [CrossRef] [PubMed]
- Tathe, A.; Ghodke, M.; Nikalje, A.P. A brief review: Biomaterials and their application. Int. J. Pharm. Sci. 2010, 2, 19–23. [Google Scholar]
- Romanò, C.; Tsuchiya, H.; Morelli, I.; Battaglia, A.; Drago, L. Antibacterial coating of implants: Are we missing something? Bone Jt. Res. 2019, 8, 199–206. [Google Scholar] [CrossRef] [PubMed]
- Sahoo, P.; Das, S.K.; Paulo Davim, J. Tribology of Materials for Biomedical Applications. In Mechanical Behaviour of Biomaterials; Woodhead Publishing: Sawston, UK, 2019; pp. 1–45. [Google Scholar]
- Marin, E.; Boschetto, F.; Pezzotti, G. Biomaterials and biocompatibility: An historical overview. J. Biomed. Mater. Res. Part A 2020, 108, 1617–1633. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, M.Z.; Sarhan, A.A.; Yusuf, F.; Hamdi, M. Biomedical materials and techniques to improve the tribological, mechanical and biomedical properties of orthopedic implants—A review article. J. Alloys Compd. 2017, 714, 636–667. [Google Scholar] [CrossRef]
- Gerhátová, Ž.; Paták, J.; Babincová, P.; Hudáková, M.; Palcut, M. Analysis of Biocompatible Metallic Materials used in Medicine. In Journal of Physics: Conference Series; IOP Publishing: Bristol, UK, 2024; Volume 2712, p. 012006. [Google Scholar]
- Catauro, M.; Barrino, F.; Dal Poggetto, G.; Crescente, G.; Piccolella, S.; Pacifico, S. New SiO2/caffeic acid hybrid materials: Synthesis, spectroscopic characterization, and bioactivity. Materials 2020, 13, 394. [Google Scholar] [CrossRef] [PubMed]
- Nassar, E.J.; Ciuffi, K.J.; Calefi, P.S.; Rocha, L.A.; De Faria, E.H.; Silva, M.L.; Fernandes, C.N. Biomaterials and sol–gel process: A methodology for the preparation of functional materials. In Biomaterials Science and Engineering; IntechOpen: London, UK, 2011. [Google Scholar]
- Simila, H.O.; Boccaccini, A.R. Sol-gel bioactive glass containing biomaterials for restorative dentistry: A review. Dent. Mater. 2022, 38, 725–747. [Google Scholar] [CrossRef]
- Brinker, C.J.; Scherer, G.W. Sol-Gel Science: The Physics and Chemistry of Sol-Gel Processing; Academic Press: Cambridge, MA, USA, 2013. [Google Scholar]
- Catauro, M.; Tranquillo, E.; Poggetto, G.D.; Naviglio, D.; Barrino, F. The Influence of Polymer on Fe (II) Citrate Release from Hybrid Materials Synthesized via Sol–Gel. Macromol. Symp. 2020, 389, 1900057. [Google Scholar] [CrossRef]
- Gallo, M.; Barrino, F.; Blanco, I.; Poggetto, G.D.; Ciaravolo, M.; Naviglio, D. Study of Bioactive Materials Containing New Complex of Iron (II) Citrate. Macromol. Symp. 2020, 389, 1900079. [Google Scholar] [CrossRef]
- Gomez-Romero, P. Hybrid organic–inorganic materials—In search of synergic activity. Adv. Mater. 2001, 13, 163–174. [Google Scholar] [CrossRef]
- Shchipunov, Y.A.; Karpenko, T.Y.Y.; Krekoten, A.V. Hybrid organic–inorganic nanocomposites fabricated with a novel biocompatible precursor using sol-gel processing. Compos. Interfaces 2005, 11, 587–607. [Google Scholar] [CrossRef]
- Pacifico, S.; Piccolella, S.; Barrino, F.; Catauro, M. Biomaterials containing the natural antioxidant quercetin: Synthesis and health benefits. Macromol. Symp. 2020, 389, 1900060. [Google Scholar] [CrossRef]
- Catauro, M.; Barrino, F.; Dal Poggetto, G.; Pacifico, F.; Piccolella, S.; Pacifico, S. Chlorogenic acid/PEG-based organic-inorganic hybrids: A versatile sol-gel synthesis route for new bioactive materials. Mater. Sci. Eng. C 2019, 100, 837–844. [Google Scholar] [CrossRef] [PubMed]
- Anulika, N.P.; Ignatius, E.O.; Raymond, E.S.; Osasere, O.I.; Abiola, A.H. The chemistry of natural product: Plant secondary metabolites. Int. J. Technol. Enhanc. Emerg. Eng. Res. 2016, 4, 1–9. [Google Scholar]
- Sindhi, V.; Gupta, V.; Sharma, K.; Bhatnagar, S.; Kumari, R.; Dhaka, N. Potential applications of antioxidants—A review. J. Pharm. Res. 2013, 7, 828–835. [Google Scholar] [CrossRef]
- Talhouk, R.S.; Karam, C.; Fostok, S.; El-Jouni, W.; Barbour, E.K. Anti-inflammatory bioactivities in plant extracts. J. Med. Food. 2007, 10, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, P.C.; Mau, J.L.; Huang, S.H. Antimicrobial effect of various combinations of plant extracts. Food Microbiol. 2001, 18, 35–43. [Google Scholar] [CrossRef]
- Solowey, E.; Lichtenstein, M.; Sallon, S.; Paavilainen, H.; Solowey, E.; Lorberboum-Galski, H. Evaluating medicinal plants for anticancer activity. Sci. World J. 2014, 2014, 721402. [Google Scholar] [CrossRef]
- Abad, M.J.; Bermejo, P.; Gonzales, E.; Iglesias, I.; Irurzun, A.; Carrasco, L. Antiviral activity of Bolivian plant extracts. Gen. Pharmacol. Vasc. Syst. 1999, 32, 499–503. [Google Scholar] [CrossRef] [PubMed]
- Munasinghe, T.C.J.; Seneviratne, C.K.; Thabrew, M.I.; Abeysekera, A.M. Antiradical and antilipoperoxidative effects of some plant extracts used by Sri Lankan traditional medical practitioners for cardioprotection. Phytother. Res. 2001, 15, 519–523. [Google Scholar] [CrossRef] [PubMed]
- Etxeberria, U.; de la Garza, A.L.; Campión, J.; Martínez, J.A.; Milagro, F.I. Antidiabetic effects of natural plant extracts via inhibition of carbohydrate hydrolysis enzymes with emphasis on pancreatic alpha amylase. Expert Opin. Ther. Targets 2012, 16, 269–297. [Google Scholar] [CrossRef] [PubMed]
- Aruoma, O.I.; Bahorun, T.; Jen, L.S. Neuroprotection by bioactive components in medicinal and food plant extracts. Mutat. Res. Rev. Mutat. Res. 2003, 544, 203–215. [Google Scholar] [CrossRef] [PubMed]
- Ciprioti, S.V.; Naviglio, D.; Gallo, M.; Barrino, F.; Catauro, M. Spectroscopic, Thermal Analysis and Bioactivity Study of New Ferrous Citrate Based Materials Prepared by Sol–Gel Method. Macromol. Symp. 2020, 389, 1900084. [Google Scholar] [CrossRef]
- Franks, W.; Schenker, I.; Schmutz, P.; Hierlemann, A. Impedance characterization and modeling of electrodes for biomedical applications. IEEE Trans. Biomed. Eng. 2005, 52, 1295–1302. [Google Scholar] [CrossRef] [PubMed]
- Catauro, M.; Tranquillo, E.; Barrino, F.; Dal Poggetto, G.; Blanco, I.; Cicala, G.; Ognibene, G.; Recca, G. Mechanical and thermal properties of fly ash-filled geopolymers. J. Therm. Anal. Calorim. 2019, 138, 3267–3276. [Google Scholar] [CrossRef]
- Catauro, M.; Barrino, F.; Scolaro, C.; Visco, A. Surface modifications induced in UHMWPE based nanocomposites during the ageing in simulated synovial fluid. Macromol. Symp. 2020, 389, 1900055. [Google Scholar] [CrossRef]
- Ingrassia, E.B.; Fiorentini, E.F.; Escudero, L.B. Hybrid biomaterials to preconcentrate and determine toxic metals and metalloids: A review. Anal. Bioanal. Chem. 2023, 415, 3073–3091. [Google Scholar] [CrossRef]
- Lee, S.Y.; Ahn, G.; Yoon, S.D. Preparation of niacinamide imprinted starch-based biomaterials for treating of hyperpigmentation. Int. J. Biol. Macromol. 2023, 232, 123382. [Google Scholar] [CrossRef]
- Divakaran, D.; Sriariyanun, M.; Basha, S.A.; Suyambulingam, I.; Sanjay, M.R.; Siengchin, S. Physico-chemical, thermal, and morphological characterization of biomass-based novel microcrystalline cellulose from Nelumbo nucifera leaf: Biomass to biomaterial approach. Biomass Convers. Biorefin. 2023, 1–15. [Google Scholar] [CrossRef]
- Xie, W.; Wei, X.; Kang, H.; Jiang, H.; Chu, Z.; Lin, Y.; Hou, Y.; Wei, Q. Static and Dynamic: Evolving Biomaterial Mechanical Properties to Control Cellular Mechanotransduction. Adv. Sci. 2023, 10, 2204594. [Google Scholar] [CrossRef]
- Rožanc, J.; Maver, U. Methods for Analyzing the Biological and Biomedical Properties of Biomaterials. Funct. Biomater. Des. Dev. Biotechnol. Pharmacol. Biomed. 2023, 1, 165–197. [Google Scholar]
- Gherman, S.P.; Biliuță, G.; Bele, A.; Ipate, A.M.; Baron, R.I.; Ochiuz, L.; Șpac, A.F.; Zavastin, D.E. Biomaterials Based on Chitosan and Polyvinyl Alcohol as a Drug Delivery System with Wound-Healing Effects. Gels 2023, 9, 122. [Google Scholar] [CrossRef]
- Al-Khalili, M.; Al-Habsi, N.; Al-Khusaibi, M.; Rahman, M.S. Proton, thermal and mechanical relaxation characteristics of a complex biomaterial (de-fatted date-pits) as a function of temperature. J. Therm. Anal. Calorim. 2023, 148, 3525–3534. [Google Scholar] [CrossRef]
- Karakullukcu, A.B.; Taban, E.; Ojo, O.O. Biocompatibility of biomaterials and test methods: A review. Mater. Test. 2023, 65, 545–559. [Google Scholar] [CrossRef]
- Binlateh, T.; Thammanichanon, P.; Rittipakorn, P.; Thinsathid, N.; Jitprasertwong, P. Collagen-based biomaterials in periodontal regeneration: Current applications and future perspectives of plant-based collagen. Biomimetics 2022, 7, 34. [Google Scholar] [CrossRef]
- Al-Shalawi, F.D.; Mohamed Ariff, A.H.; Jung, D.W.; Mohd Ariffin, M.K.A.; Seng Kim, C.L.; Brabazon, D.; Al-Osaimi, M.O. Biomaterials as Implants in the Orthopedic Field for Regenerative Medicine: Metal versus Synthetic Polymers. Polymers 2023, 15, 2601. [Google Scholar] [CrossRef]
- Abraham, A.M.; Venkatesan, S. A review on application of biomaterials for medical and dental implants. Proc. Inst. Mech. Eng. Part L J. Mater. Des. Appl. 2023, 237, 249–273. [Google Scholar] [CrossRef]
- Chi, M.; Yuan, B.; Xie, Z.; Hong, J. The innovative biomaterials and technologies for developing corneal endothelium tissue engineering scaffolds: A review and prospect. Bioengineering 2023, 10, 1284. [Google Scholar] [CrossRef]
- Patel, N.R.; Gohil, P.P. A review on biomaterials: Scope, applications & human anatomy significance. Int. J. Emerg. Technol. Adv. Eng. 2012, 2, 91–101. [Google Scholar]
- Kozlowska, J.; Stachowiak, N.; Sionkowska, A. The preparation and characterization of composite materials by incorporating microspheres into a collagen/hydroxyethyl cellulose matrix. Polym. Test. 2018, 69, 350–358. [Google Scholar] [CrossRef]
- Anghel, N.; Dinu, M.V.; Zaltariov, M.; Pamfil, D.; Spiridon, I. New cellulose-collagen-alginate materials incorporated with quercetin, anthocyanins and lipoic acid. Int. J. Biol. Macromol. 2021, 181, 30–40. [Google Scholar] [CrossRef]
- Catauro, M.; Barrino, F.; Blanco, I.; Piccolella, S.; Pacifico, S. Use of the sol–gel method for the preparation of coatings of titanium substrates with hydroxyapatite for biomedical application. Coatings 2020, 10, 203. [Google Scholar] [CrossRef]
- Bandyopadhyay, A.; Mitra, I.; Goodman, S.B.; Kumar, M.; Bose, S. Improving biocompatibility for next generation of metallic implants. Prog. Mater. Sci. 2023, 133, 101053. [Google Scholar] [CrossRef]
- Patil, N.A.; Kandasubramanian, B. Biological and mechanical enhancement of zirconium dioxide for medical applications. Ceram. Int. 2020, 46, 4041–4057. [Google Scholar] [CrossRef]
- Catauro, M.; Barrino, F.; Bononi, M.; Colombini, E.; Giovanardi, R.; Veronesi, P.; Tranquillo, E. Coating of titanium substrates with ZrO2 and ZrO2-SiO2 composites by sol-gel synthesis for biomedical applications: Structural characterization, mechanical and corrosive behavior. Coatings 2019, 9, 200. [Google Scholar] [CrossRef]
- Saba, N.; Jawaid, M. A review on thermomechanical properties of polymers and fibers reinforced polymer composites. J. Ind. Eng. Chem. 2018, 67, 1–11. [Google Scholar] [CrossRef]
- Gupta, M.K.; Srivastava, R.K. Mechanical properties of hybrid fibers-reinforced polymer composite: A review. Polym. Plast. Technol. Eng. 2016, 55, 626–642. [Google Scholar] [CrossRef]
- Si, Y.; Wang, L.; Wang, X.; Tang, N.; Yu, J.; Ding, B. Ultrahigh-water-content, superelastic, and shape-memory nanofiber-assembled hydrogels exhibiting pressure-responsive conductivity. Adv. Mater. 2017, 29, 1700339. [Google Scholar] [CrossRef]
- Choudhury, N.A.; Sampath, S.; Shukla, A.K. Hydrogel-polymer electrolytes for electrochemical capacitors: An overview. Energy Environ. Sci. 2009, 2, 55–67. [Google Scholar] [CrossRef]
- Mitzi, D.B. Thin-film deposition of organic-inorganic hybrid materials. Chem. Mater. 2001, 13, 3283–3298. [Google Scholar] [CrossRef]
- Barrino, F.; La Rosa-Ramírez, D.; Schiraldi, C.; López-Martínez, J.; Samper, M.D. Preparation and Characterization of New Bioplastics Based on Polybutylene Succinate (PBS). Polymers 2023, 15, 1212. [Google Scholar] [CrossRef]
- Agustini, T.W.; Suzery, M.; Sutrisnanto, D.; Ma’ruf, W.F. Comparative study of bioactive substances extracted from fresh and dried Spirulina sp. Procedia Environ. Sci. 2015, 23, 282–289. [Google Scholar] [CrossRef]
- Catauro, M.; Barrino, F.; Blanco, I.; Dal Poggetto, G.; Piccolella, S.; Crescente, G.; Pacifico, S. Bioactivity of chlorogenic acid/SiO2/PEG composite synthesized via sol-gel. Mater. Today Proc. 2021, 34, 99–102. [Google Scholar] [CrossRef]
- Wei, Q.; Becherer, T.; Angioletti-Uberti, S.; Dzubiella, J.; Wischke, C.; Neffe, A.T.; Lendlein, A.; Ballauff, M.; Haag, R. Protein interactions with polymer coatings and biomaterials. Angew. Chem. Int. Ed. 2014, 53, 8004–8031. [Google Scholar] [CrossRef] [PubMed]
- Loy, D.A. Hybrid organic–inorganic materials. MRS Bull. 2001, 26, 364–367. [Google Scholar] [CrossRef]
- Judeinstein, P.; Sanchez, C. Hybrid organic–inorganic materials: A land of multidisciplinarity. J. Mater. Chem. 1996, 6, 511–525. [Google Scholar] [CrossRef]
- Mammeri, F.; Le Bourhis, E.; Rozes, L.; Sanchez, C. Mechanical properties of hybrid organic–inorganic materials. J. Mater. Chem. 2005, 15, 3787–3811. [Google Scholar] [CrossRef]
- Pandey, S.; Mishra, S.B. Sol–gel derived organic–inorganic hybrid materials: Synthesis, characterizations and applications. J. Sol-Gel Sci. Technol. 2011, 59, 73–94. [Google Scholar] [CrossRef]
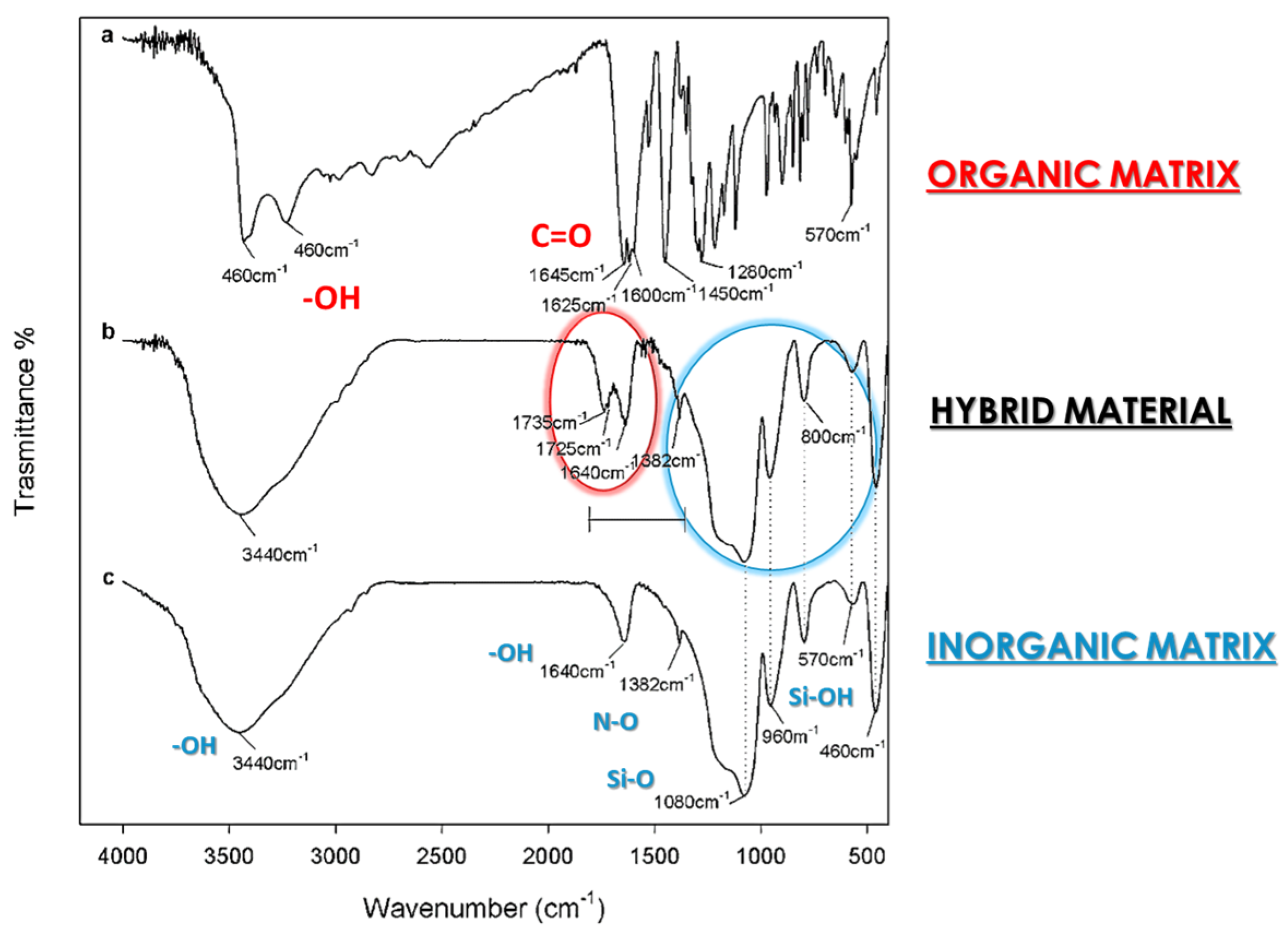
- Schmitt, J.; Flemming, H.C. FTIR-spectroscopy in microbial and material analysis. Int. Biodeter. Biodegr. 1998, 41, 1–11. [Google Scholar] [CrossRef]
- Han, Y.H.; Taylor, A.; Mantle, M.D.; Knowles, K.M. Sol–gel-derived organic–inorganic hybrid materials. J. Non-Cryst. Solids 2007, 353, 313–320. [Google Scholar] [CrossRef]
- Prati, S.; Joseph, E.; Sciutto, G.; Mazzeo, R. New advances in the application of FTIR microscopy and spectroscopy for the characterization of artistic materials. Acc. Chem. Res. 2010, 43, 792–801. [Google Scholar] [CrossRef] [PubMed]
- Criado, M.; Sobrados, I.; Sanz, J. Polymerization of hybrid organic–inorganic materials from several silicon compounds followed by TGA/DTA, FTIR and NMR techniques. Prog. Org. Coat. 2014, 77, 880–891. [Google Scholar] [CrossRef]
- Al Zoubi, W.; Kamil, M.P.; Fatimah, S.; Nashrah, N.; Ko, Y.G. Recent advances in hybrid organic-inorganic materials with spatial architecture for state-of-the-art applications. Prog. Mater. Sci. 2020, 112, 100663. [Google Scholar] [CrossRef]
- Vallet-Regí, M.; Colilla, M.; González, B. Medical applications of organic–inorganic hybrid materials within the field of silica-based bioceramics. Chem. Soc. Rev. 2011, 40, 596–607. [Google Scholar] [CrossRef] [PubMed]
- Tranquillo, E.; Barrino, F.; Dal Poggetto, G.; Blanco, I. Sol–gel synthesis of silica-based materials with different percentages of PEG or PCL and high chlorogenic acid content. Materials 2019, 12, 155. [Google Scholar] [CrossRef]
- John, Ł. Selected developments and medical applications of organic–inorganic hybrid biomaterials based on functionalized spherosilicates. Mater. Sci. Eng. C 2018, 88, 172–181. [Google Scholar] [CrossRef]
- Pandey, S.; Mishra, S.B. Bioceramics: Silica-Based Organic-Inorganic Hybrid Materials for Medical Applications. In Nanomedicine for Drug Delivery and Therapeutics; Wiley Online Library: Hoboken, NJ, USA, 2013; pp. 135–161. [Google Scholar]
- Catauro, M.; Tranquillo, E.; Barrino, F.; Blanco, I.; Dal Poggetto, F.; Naviglio, D. Drug release of hybrid materials containing Fe (II) citrate synthesized by sol-gel technique. Materials 2018, 11, 2270. [Google Scholar] [CrossRef]
- Pierre, A.C. Introduction to Sol-Gel Processing; Springer Nature: Berlin/Heidelberg, Germany, 2020. [Google Scholar]
- Ivicheva, S.N.; Ovsyannikov, N.A.; Lysenkov, A.S.; Klimashin, A.A.; Kargin, Y.F. Sol–gel synthesis of oxonitridoaluminosilicates (SiAlON). Russ. J. Inorg. Chem. 2020, 65, 1820–1830. [Google Scholar] [CrossRef]
- Sakka, S. Handbook of Sol-Gel Science and Technology. 1. Sol-Gel Processing; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2005; Volume 1. [Google Scholar]
- Dislich, H. Sol-Gel 1984→2004 (?). J. Non-Cryst. Solids 1985, 73, 599–612. [Google Scholar] [CrossRef]
- Brunauer, S.; Emmett, P.H.; Teller, E. Adsorption of gases in multimolecular layers. J. Am. Chem. Soc. 1938, 60, 309–319. [Google Scholar] [CrossRef]
- Depagne, C.; Roux, C.; Coradin, T. How to design cell-based biosensors using the sol–gel process. Anal. Bioanal. Chem. 2011, 400, 965–976. [Google Scholar] [CrossRef] [PubMed]
- Cauqui, M.A.; Rodriguez-Izquierdo, J.M. Application of the sol-gel methods to catalyst preparation. J. Non-Cryst. Solids 1992, 147, 724–738. [Google Scholar] [CrossRef]
- Omri, A.; Benzina, M.; Bennour, F. Industrial application of photocatalysts prepared by hydrothermal and sol–gel methods. J. Ind. Eng. Chem. 2015, 21, 356–362. [Google Scholar] [CrossRef]
- Hench, L.L.; West, J.K. The sol-gel process. Chem. Rev. 1990, 90, 33–72. [Google Scholar] [CrossRef]
- Klein, L.C. Sol-gel processing of silicates. Annu. Rev. Mater. Sci. 1985, 15, 227–248. [Google Scholar] [CrossRef]
- Livage, J.; Sanchez, C.; Henry, M.; Doeuff, S. The chemistry of the sol-gel process. Solid State Ion. 1989, 32, 633–638. [Google Scholar] [CrossRef]
- Landau, M.V. Sol–gel process. In Handbook of Heterogeneous Catalysis: Online; Wiley-VCH: Weinheim, Germany, 2008; pp. 119–160. [Google Scholar] [CrossRef]
- Kamanina, O.A.; Saverina, E.A.; Rybochkin, P.V.; Arlyapov, V.A.; Vereshchagin, A.N.; Ananikov, V.P. Preparation of hybrid sol-gel materials based on living cells of microorganisms and their application in nanotechnology. Nanomaterials 2022, 12, 1086. [Google Scholar] [CrossRef]
- Song, X.; Segura-Egea, J.J.; Díaz-Cuenca, A. Sol–Gel technologies to obtain advanced bioceramics for dental therapeutics. Molecules 2023, 28, 6967. [Google Scholar] [CrossRef]
- Aparicio, M.; Jitianu, A.; Klein, L.C. Sol-Gel Processing for Conventional and Alternative Energy; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2012. [Google Scholar]
- Dislich, H. Sol-gel: Science, processes and products. J. Non-Cryst. Solids 1986, 80, 115–121. [Google Scholar] [CrossRef]
- Gonçalves, M.C. Sol-gel silica nanoparticles in medicine: A natural choice. Design, synthesis and products. Molecules 2018, 23, 2021. [Google Scholar] [CrossRef] [PubMed]
- Löbmann, P. Antireflective coatings by sol–gel processing: Commercial products and future perspectives. J. Sol-Gel Sci. Technol. 2017, 83, 291–295. [Google Scholar] [CrossRef]
- Attia, Y.A. Sol-Gel Processing and Applications; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2012. [Google Scholar]
- Mackenzie, J.D. Applications of the sol-gel process. J. Non-Cryst. Solids 1988, 100, 162–168. [Google Scholar] [CrossRef]
- Dehghanghadikolaei, A.; Ansary, J.; Ghoreishi, R. Sol-gel process applications: A mini-review. Proc. Nat. Res. Soc. 2018, 2, 02008–02029. [Google Scholar] [CrossRef]
- Aegerter, M.A.; Mennig, M. Sol-Gel Technologies for Glass Producers and Users; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2013. [Google Scholar]
- Dimesso, L. Pechini processes: An alternate approach of the sol–gel method, preparation, properties, and applications. In Handbook of Sol-Gel Science and Technology; Springer: Berlin/Heidelberg, Germany, 2016; Volume 2, pp. 1–22. [Google Scholar]
- Tshikovhi, A.; Koao, L.F.; Malevu, T.D.; Linganiso, E.C.; Motaung, T.E. Dopants concentration on the properties of various host materials by sol-gel method: Critical review. Results Mater. 2023, 19, 100447. [Google Scholar] [CrossRef]
- Tinoco Navarro, L.K.; Jaroslav, C. Enhancing Photocatalytic Properties of TiO2 Photocatalyst and Heterojunctions: A Comprehensive Review of the Impact of Biphasic Systems in Aerogels and Xerogels Synthesis, Methods, and Mechanisms for Environmental Applications. Gels 2023, 9, 976. [Google Scholar] [CrossRef] [PubMed]
- Innocenzi, P. Sol-gel processing for advanced ceramics, a perspective. Open Ceram. 2023, 16, 100477. [Google Scholar] [CrossRef]
- Kumar, A.; Yadav, N.; Bhatt, M.; Mishra, N.K.; Chaudhary, P.; Singh, R. Sol-gel derived nanomaterials and it’s applications: A review. Res. J. Chem. Sci. 2015, 5, 1–8. [Google Scholar]
- Gvishi, R. Fast sol–gel technology: From fabrication to applications. J. Sol-Gel Sci. Technol. 2009, 50, 241–253. [Google Scholar] [CrossRef]
- Chaijaruwanich, A. Coating techniques for biomaterials: A review. CMUJ Nat. Sci. 2011, 10, 39–50. [Google Scholar]
- Harun, W.S.W.; Asri, R.I.M.; Alias, J.; Zulkifli, F.H.; Kadirgama, K.; Ghani, S.A.C.; Shariffuddin, J.H.M. A comprehensive review of hydroxyapatite-based coatings adhesion on metallic biomaterials. Ceram. Int. 2018, 44, 1250–1268. [Google Scholar] [CrossRef]
- Tranquillo, E.; Bollino, F. Surface modifications for implants lifetime extension: An overview of sol-gel coatings. Coatings 2020, 10, 589. [Google Scholar] [CrossRef]
- Innocenzi, P.C.; Guglielmi, M.; Gobbin, M.; Colombo, P. Coating of metals by the sol-gel dip-coating method. J. Eur. Ceram. 1992, 10, 431–436. [Google Scholar] [CrossRef]
- Faustini, M.; Louis, B.; Albouy, P.A.; Kuemmel, M.; Grosso, D. Preparation of sol−gel films by dip-coating in extreme conditions. J. Phys. Chem. C 2010, 114, 7637–7645. [Google Scholar] [CrossRef]
- Mechiakh, R.; Sedrine, N.B.; Chtourou, R.; Bensaha, R. Correlation between microstructure and optical properties of nano-crystalline TiO2 thin films prepared by sol–gel dip coating. Appl. Surf. Sci. 2010, 257, 670–676. [Google Scholar] [CrossRef]
- Jokinen, M.; Pätsi, M.; Rahiala, H.; Peltola, T.; Ritala, M.; Rosenholm, J.B. Influence of sol and surface properties on in vitro bioactivity of sol-gel-derived TiO2 and TiO2-SiO2 films deposited by dip-coating method. J. Biomed. Mater. Res. 1998, 42, 295–302. [Google Scholar] [CrossRef]
- Mendhe, A.C. Spin Coating: Easy Technique for Thin Films. In Simple Chemical Methods for Thin Film Deposition: Synthesis and Applications; Springer Nature: Singapore, 2023; pp. 387–424. [Google Scholar]
- Devi, K.P.; Goswami, P.; Chaturvedi, H. Fabrication of nanocrystalline TiO2 thin films using Sol-Gel spin coating technology and investigation of its structural, morphology and optical characteristics. Appl. Surf. Sci. 2022, 591, 153226. [Google Scholar] [CrossRef]
- Yepuri, V.; Satyanarayana, A. A novel methodology in fabricating dielectric reflectors for the desired wavelength spectrum using sol-gel spin coating technique. Opt. Mater. 2024, 147, 114770. [Google Scholar] [CrossRef]
- Maho, A.; Nayak, S.; Gillissen, F.; Cloots, R.; Rougier, A. Film Deposition of Electrochromic Metal Oxides through Spray Coating: A Descriptive Review. Coatings 2023, 13, 1879. [Google Scholar] [CrossRef]
- Pan, Z.; Guo, J.; Li, S.; Li, X.; Zhang, H. Properties of alumina coatings prepared on silica-based ceramic substrate by plasma spraying and sol-gel dipping methods. Ceram. Int. 2021, 47, 27453–27461. [Google Scholar] [CrossRef]
- Ilsatoham, M.I.; Alkian, I.; Azzahra, G.; Hidayanto, E.; Sutanto, H. Effect of substrate temperature on the properties of Bi2O3 thin films grown by sol-gel spray coating. Results Eng. 2023, 17, 100991. [Google Scholar] [CrossRef]
- Gvishi, R.; Sokolov, I. 3D sol–gel printing and sol–gel bonding for fabrication of macro-and micro/nano-structured photonic devices. J. Sol-Gel Sci. Technol. 2020, 95, 635–648. [Google Scholar] [CrossRef]
- Stumpf, M.; Travitzky, N.; Greil, P.; Fey, T. Sol-gel infiltration of complex cellular indirect 3D printed alumina. J. Eur. Ceram. 2018, 38, 3603–3609. [Google Scholar] [CrossRef]
- Zhu, Y.; Di, W.; Song, M.; Chitrakar, B.; Liu, Z. Correlating 3D printing performance with sol-gel transition based on thermo-responsive k-carrageenan affected by fructose. J. Food Eng. 2023, 340, 111316. [Google Scholar] [CrossRef]
- Echalier, C.; Levato, R.; Mateos-Timoneda, M.A.; Castaño, O.; Déjean, S.; Garric, X.; Pinese, C.; Noel, D.; Engel, E.; Martinez, J.; et al. Modular bioink for 3D printing of biocompatible hydrogels: Sol–gel polymerization of hybrid peptides and polymers. RSC Adv. 2017, 7, 12231–12235. [Google Scholar] [CrossRef]
- Gupta, R.; Kumar, A. Bioactive materials for biomedical applications using sol–gel technology. Biomed. Mater. 2008, 3, 034005. [Google Scholar] [CrossRef] [PubMed]
- Lei, Q.; Guo, J.; Noureddine, A.; Wang, A.; Wuttke, S.; Brinker, C.J.; Zhu, W. Sol–gel-based advanced porous silica materials for biomedical applications. Adv. Funct. Mater. 2020, 30, 1909539. [Google Scholar] [CrossRef]
- Balamurugan, A.; Sockalingum, G.; Michel, J.; Fauré, J.; Banchet, V.; Wortham, L.; Bouthors, S.; Laurent-Maquin, D.; Balossier, G. Synthesis and characterisation of sol gel derived bioactive glass for biomedical applications. Mater. Lett. 2006, 60, 3752–3757. [Google Scholar] [CrossRef]
- Jaafar, A.; Hecker, C.; Árki, P.; Joseph, Y. Sol-gel derived hydroxyapatite coatings for titanium implants: A review. Bioengineering 2020, 7, 127. [Google Scholar] [CrossRef]
- Azadani, R.N.; Sabbagh, M.; Salehi, H.; Cheshmi, A.; Raza, A.; Kumari, B.; Erabi, G. Sol-gel: Uncomplicated, routine and affordable synthesis procedure for utilization of composites in drug delivery. J. Compos. Compd. 2021, 3, 57–70. [Google Scholar]
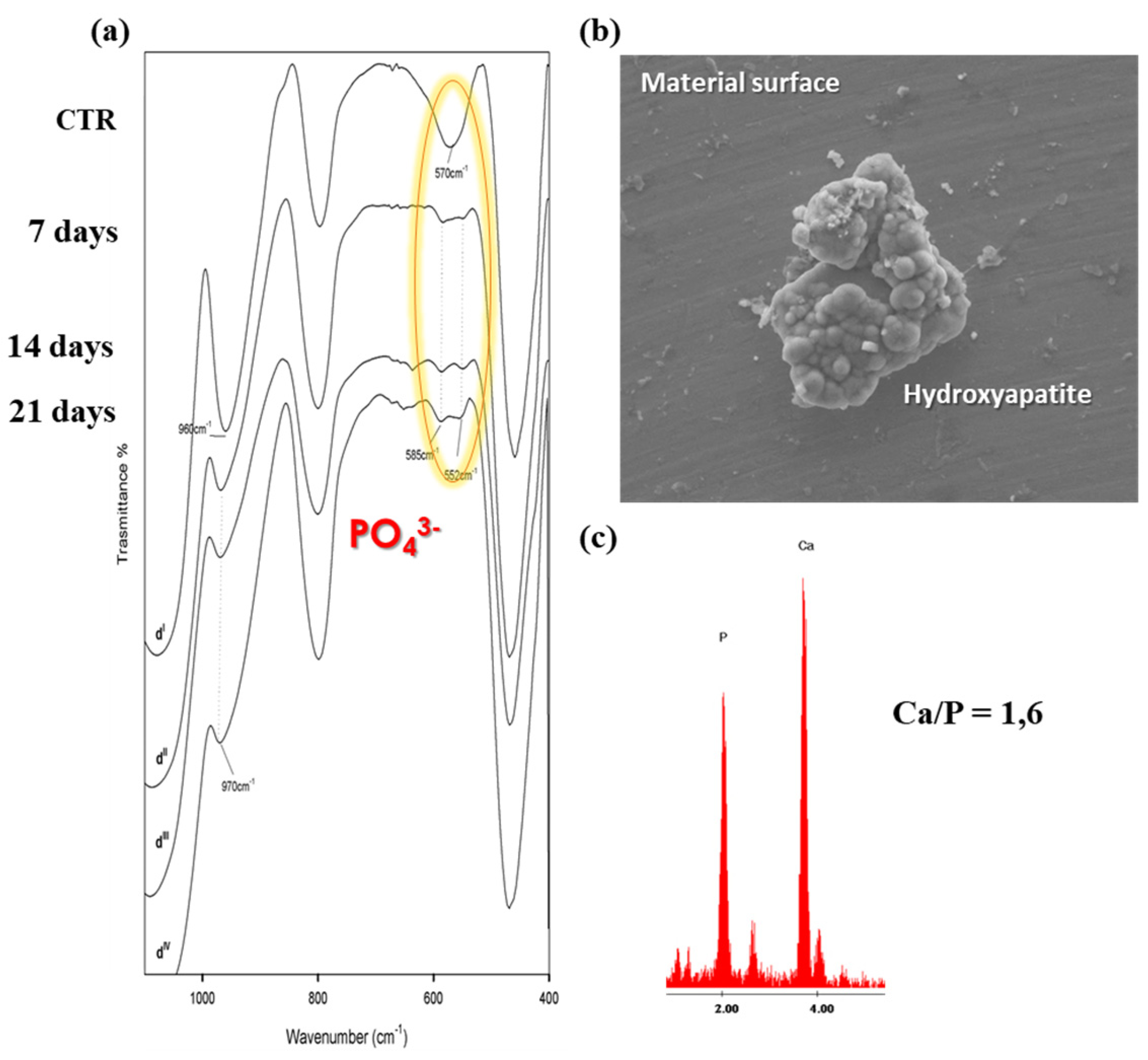
- Arcos, D.; Vallet-Regí, M. Sol–gel silica-based biomaterials and bone tissue regeneration. Acta Biomater. 2010, 6, 2874–2888. [Google Scholar] [CrossRef] [PubMed]
- Shchipunov, Y. Biomimetic Sol–Gel Chemistry to Tailor Structure, Properties, and Functionality of Bionanocomposites by Biopolymers and Cells. Materials 2024, 17, 224. [Google Scholar] [CrossRef] [PubMed]
- Farano, V.; Maurin, J.C.; Attik, N.; Jackson, P.; Grosgogeat, B.; Gritsch, K. Sol–gel bioglasses in dental and periodontal regeneration: A systematic review. J. Biomed. Mater. Res. Part B Appl. Biomater. 2019, 107, 1210–1227. [Google Scholar] [CrossRef]
- Fernández-Hernán, J.P.; Torres, B.; López, A.J.; Rams, J. The role of the sol-gel synthesis process in the biomedical field and its use to enhance the performance of bioabsorbable magnesium implants. Gels 2022, 8, 426. [Google Scholar] [CrossRef] [PubMed]
- Al-Harbi, N.; Mohammed, H.; Al-Hadeethi, Y.; Bakry, A.S.; Umar, A.; Hussein, M.A.; Abbassy, M.A.; Vaidya, K.G.; Al Berakdar, G.; Mkawi, E.M.; et al. Silica-based bioactive glasses and their applications in hard tissue regeneration: A review. Pharmaceuticals 2021, 14, 75. [Google Scholar] [CrossRef]
- Horkavcova, D.; Novak, P.; Fialova, I.; Černý, M.; Jablonska, E.; Lipov, J.; Ruml, T.; Helebrant, A. Titania sol-gel coatings containing silver on newly developed TiSi alloys and their antibacterial effect. Mater. Sci. Eng. C 2017, 76, 25–30. [Google Scholar] [CrossRef]
- Kaygili, O.; Dorozhkin, S.V.; Keser, S. Synthesis and characterization of Ce-substituted hydroxyapatite by sol–gel method. Mater. Sci. Eng. C 2014, 42, 78–82. [Google Scholar] [CrossRef] [PubMed]
- Belet, A.; Wolfs, C.; Mahy, J.G.; Poelman, D.; Vreuls, C.; Gillard, N.; Lambert, S.D. Sol-gel syntheses of photocatalysts for the removal of pharmaceutical products in water. Nanomaterials 2019, 9, 126. [Google Scholar] [CrossRef]
- Kokubo, T.; Takadama, H. Simulated body fluid (SBF) as a standard tool to test the bioactivity of implants. In Handbook of Biomineralization: Biological Aspects and Structure Formation; Wiley Online Library: Hoboken, NJ, USA, 2007; pp. 97–109. [Google Scholar]
- Kokubo, T.; Takadama, H. How useful is SBF in predicting in vivo bone bioactivity? Biomaterials 2006, 27, 2907–2915. [Google Scholar] [CrossRef]
- Takadama, H.; Hashimoto, M.; Mizuno, M.; Kokubo, T. Round-robin test of SBF for in vitro measurement of apatite-forming ability of synthetic materials. Phosphorus Res. Bull. 2004, 17, 119–125. [Google Scholar] [CrossRef] [PubMed]
- Kokubo, T.; Yamaguchi, S. Simulated body fluid and the novel bioactive materials derived from it. J. Biomed. Mater. Res. 2019, 107, 968–977. [Google Scholar] [CrossRef]
- Catauro, M.; Barrino, F.; Dal Poggetto, G.; Milazzo, M.; Blanco, I.; Ciprioti, S.V. Structure, drug absorption, bioactive and antibacterial properties of sol-gel SiO2/ZrO2 materials. Ceram. Int. 2020, 46, 29459–29465. [Google Scholar] [CrossRef]
- Rizzotto, F.; Vasiljevic, Z.Z.; Stanojevic, G.; Dojcinovic, M.P.; Jankovic-Castvan, I.; Vujancevic, J.D.; Tadic, N.B.; Brankovic, G.O.; Magniez, A.; Vidic, J.; et al. Antioxidant and cell-friendly Fe2TiO5 nanoparticles for food packaging application. Food Chem. 2022, 390, 133198. [Google Scholar] [CrossRef] [PubMed]
- Sivakanthan, S.; Rajendran, S.; Gamage, A.; Madhujith, T.; Mani, S. Antioxidant and antimicrobial applications of biopolymers: A review. Food Res. Int. 2020, 136, 109327. [Google Scholar] [CrossRef]
- Catauro, M.; Barrino, F.; Dal Poggetto, G.; Crescente, G.; Piccolella, S.; Pacifico, S. Chlorogenic acid entrapped in hybrid materials with high PEG content: A strategy to obtain antioxidant functionalized biomaterials? Materials 2019, 12, 148. [Google Scholar] [CrossRef]
- Mellado-Vázquez, R.; García-Hernández, M.; López-Marure, A.; López-Camacho, P.Y.; Morales-Ramírez, Á.D.J.; Beltrán-Conde, H.I. Sol-gel synthesis and antioxidant properties of yttrium oxide nanocrystallites incorporating P-123. Materials 2014, 7, 6768–6778. [Google Scholar] [CrossRef] [PubMed]
- Martysiak-Żurowska, D.; Wenta, W. A comparison of ABTS and DPPH methods for assessing the total antioxidant capacity of human milk. Acta Sci. Pol. Technol. 2012, 11, 83–89. [Google Scholar]
- Floegel, A.; Kim, D.O.; Chung, S.J.; Koo, S.I.; Chun, O.K. Comparison of ABTS/DPPH assays to measure antioxidant capacity in popular antioxidant-rich US foods. J. Food Compos. Anal. 2011, 24, 1043–1048. [Google Scholar] [CrossRef]
- Shalaby, E.A.; Shanab, S.M. Comparison of DPPH and ABTS Assays for Determining Antioxidant Potential of Water and Methanol Extracts of Spirulina platensis; NISCAIR-CSIR: New Delhi, India, 2013. [Google Scholar]
- Kawashita, M.; Tsuneyama, S.; Miyaji, F.; Kokubo, T.; Kozuka, H.; Yamamoto, K. Antibacterial silver-containing silica glass prepared by sol–gel method. Biomaterials 2000, 21, 393–398. [Google Scholar] [CrossRef]
- Jeon, H.J.; Yi, S.C.; Oh, S.G. Preparation and antibacterial effects of Ag–SiO2 thin films by sol–gel method. Biomaterials 2003, 24, 4921–4928. [Google Scholar] [CrossRef]
- Jaiswal, S.; McHale, P.; Duffy, B. Preparation and rapid analysis of antibacterial silver, copper and zinc doped sol–gel surfaces. Colloids Surf. B 2012, 94, 170–176. [Google Scholar] [CrossRef] [PubMed]
- Mortazavi, V.; Nahrkhalaji, M.M.; Fathi, M.H.; Mousavi, S.B.; Esfahani, B.N. Antibacterial effects of sol-gel-derived bioactive glass nanoparticle on aerobic bacteria. J. Biomed. Mater. Res. 2010, 94, 160–168. [Google Scholar] [CrossRef] [PubMed]
- Ielo, I.; Giacobello, F.; Castellano, A.; Sfameni, S.; Rando, G.; Plutino, M.R. Development of antibacterial and antifouling innovative and eco-sustainable sol–gel based materials: From marine areas protection to healthcare applications. Gels 2021, 8, 26. [Google Scholar] [CrossRef] [PubMed]
- Catauro, M.; Tranquillo, E.; Poggetto, G.D.; Naviglio, S.; Barrino, F. Antibacterial properties of sol–gel biomaterials with different percentages of PEG or PCL. Macromol. Symp. 2020, 389, 1900056. [Google Scholar] [CrossRef]
- Nablo, B.J.; Rothrock, A.R.; Schoenfisch, M.H. Nitric oxide-releasing sol–gels as antibacterial coatings for orthopedic implants. Biomaterials 2005, 26, 917–924. [Google Scholar] [CrossRef] [PubMed]
- Paşahan, A.; Sevimli, R.; Kıvılcım, N.; Karaca Açarı, İ.; Erenler, A.Ş.; Sezer, S.; Durmaz, H.T.; Hüz, M.; Ünver, T.; Seçkin, T.; et al. Preparation, characterization, and biocompatibility of chondroitin sulfate-based sol-gel coatings and investigation of their effects on osseointegration improvement. Int. J. Polym. Mater. 2023, 72, 1510–1528. [Google Scholar] [CrossRef]
- Simila, H.O.; Boccaccini, A.R. Sol-gel synthesis of lithium doped mesoporous bioactive glass nanoparticles and tricalcium silicate for restorative dentistry: Comparative investigation of physico-chemical structure, antibacterial susceptibility and biocompatibility. Front. Bioeng. Biotechnol. 2023, 11, 1065597. [Google Scholar] [CrossRef]
- Mahdi, B.; Rouabah, F. Effect of Titanium Dioxide Nanoparticles on the Properties of Poly (Vinyl Alcohol)/Silica Hybrid Films Prepared by the Sol-Gel Method. Nano Hybrids Compos. 2023, 38, 63–79. [Google Scholar] [CrossRef]
- Ebrahimi, M.; Manafi, S.; Sharifianjazi, F. The effect of Ag2O and MgO dopants on the bioactivity, biocompatibility, and antibacterial properties of 58S bioactive glass synthesized by the sol-gel method. J. Non-Cryst. Solids 2023, 606, 122189. [Google Scholar] [CrossRef]
- Bahati, D.; Bricha, M.; El Mabrouk, K. Synthesis, characterization, and in vitro apatite formation of strontium-doped sol-gel-derived bioactive glass nanoparticles for bone regeneration applications. Ceram. Int. 2023, 49, 23020–23034. [Google Scholar] [CrossRef]
- Tarzanagh, Y.J.; Seifzadeh, D.; Rajabalizadeh, Z.; Habibi-Yangjeh, A.; Khodayari, A.; Sohrabnezhad, S. Sol-gel/MOF nanocomposite for effective protection of 2024 aluminum alloy against corrosion. Surf. Coat. Technol. 2024, 380, 125038. [Google Scholar] [CrossRef]
- Chelu, M.; Musuc, A.M. Advanced biomedical applications of multifunctional natural and synthetic biomaterials. Processes 2023, 11, 2696. [Google Scholar] [CrossRef]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barrino, F. Hybrid Organic–Inorganic Materials Prepared by Sol–Gel and Sol–Gel-Coating Method for Biomedical Use: Study and Synthetic Review of Synthesis and Properties. Coatings 2024, 14, 425. https://doi.org/10.3390/coatings14040425
Barrino F. Hybrid Organic–Inorganic Materials Prepared by Sol–Gel and Sol–Gel-Coating Method for Biomedical Use: Study and Synthetic Review of Synthesis and Properties. Coatings. 2024; 14(4):425. https://doi.org/10.3390/coatings14040425
Chicago/Turabian StyleBarrino, Federico. 2024. "Hybrid Organic–Inorganic Materials Prepared by Sol–Gel and Sol–Gel-Coating Method for Biomedical Use: Study and Synthetic Review of Synthesis and Properties" Coatings 14, no. 4: 425. https://doi.org/10.3390/coatings14040425
APA StyleBarrino, F. (2024). Hybrid Organic–Inorganic Materials Prepared by Sol–Gel and Sol–Gel-Coating Method for Biomedical Use: Study and Synthetic Review of Synthesis and Properties. Coatings, 14(4), 425. https://doi.org/10.3390/coatings14040425





