Cinnamon Essential-Oil-Loaded Fish Gelatin–Cellulose Nanocrystal Films Prepared under Acidic Conditions
Abstract
1. Introduction
2. Materials and Methods
2.1. Raw Materials and Chemicals
2.2. Emulsion and Film Preparation
2.3. Film Characterization
2.3.1. Film Thickness
2.3.2. Optical Properties
2.3.3. Wetting Properties
2.3.4. Mechanical Properties
2.3.5. Fourier Transform Infrared (FT-IR) Measurement
2.3.6. Water Solubility (WS)
2.4. Statistical Analysis
3. Results and Discussion
3.1. Thickness Measurement
3.2. Optical Properties
3.3. Wetting Properties
3.4. Mechanical Properties
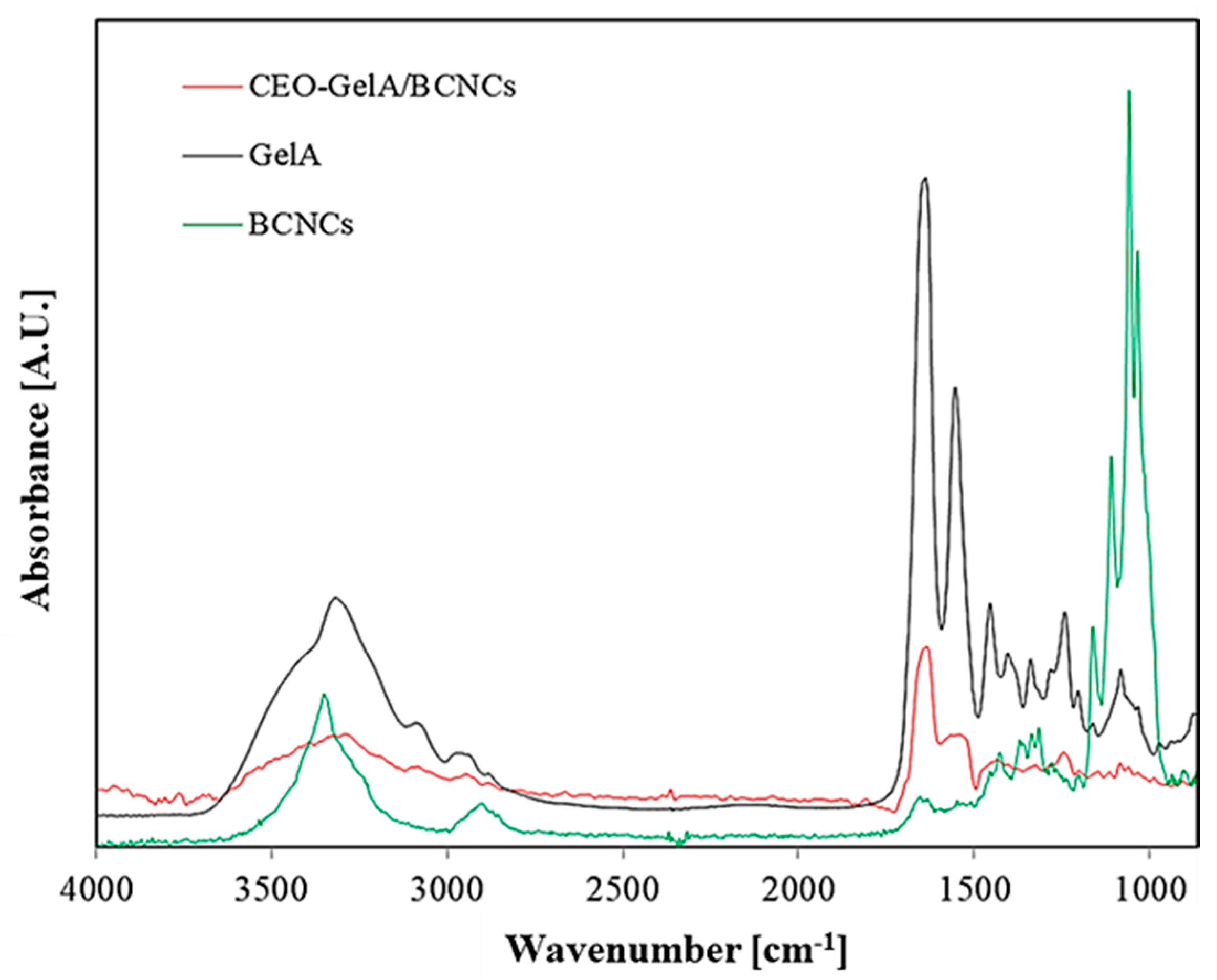
3.5. FTIR Measurements
3.6. WS Measurements
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Avena-Bustillos, R.; Chiou, B.S.; Olsen, C.; Bechtel, P.; Olson, D.; McHugh, T. Gelation, oxygen permeability, and mechanical properties of mammalian and fish gelatin films. J. Food Sci. 2011, 76, E519–E524. [Google Scholar] [CrossRef] [PubMed]
- Tongnuanchan, P.; Benjakul, S.; Prodpran, T. Properties and antioxidant activity of fish skin gelatin film incorporated with citrus essential oils. Food Chem. 2012, 134, 1571–1579. [Google Scholar] [CrossRef] [PubMed]
- Echegaray, M.; Mondragon, G.; Loli, M.; González, A.; Peña-Rodriguez, C.; Arbelaiz, A. Physicochemical and mechanical properties of gelatin reinforced with nanocellulose and montmorillonite. J. Renew. Mater. 2016, 4, 206. [Google Scholar] [CrossRef]
- Kanmani, P.; Rhim, J.-W. Physical, mechanical and antimicrobial properties of gelatin based active nanocomposite films containing AgNPs and nanoclay. Food Hydrocoll. 2014, 35, 644–652. [Google Scholar] [CrossRef]
- Tongnuanchan, P.; Benjakul, S.; Prodpran, T.; Pisuchpen, S.; Osako, K. Mechanical, thermal and heat sealing properties of fish skin gelatin film containing palm oil and basil essential oil with different surfactants. Food Hydrocoll. 2016, 56, 93–107. [Google Scholar] [CrossRef]
- Martucci, J.F.; Ruseckaite, R.A. Biodegradation of three-layer laminate films based on gelatin under indoor soil conditions. Polym. Degrad. Stab. 2009, 94, 1307–1313. [Google Scholar] [CrossRef]
- Mu, C.; Guo, J.; Li, X.; Lin, W.; Li, D. Preparation and properties of dialdehyde carboxymethyl cellulose crosslinked gelatin edible films. Food Hydrocoll. 2012, 27, 22–29. [Google Scholar] [CrossRef]
- Peña, C.; Mondragon, G.; Algar, I.; Mondragon, I.; Martucci, J.; Ruseckaite, R. Gelatin films: Renewable resource for food packaging. In Gelatin: Production, Applications and Health Implications; Nova Science Publishers: New York, NY, USA, 2013; pp. 1–15. [Google Scholar]
- Regenstein, J.; Chaudry, M. Kosher and halal issues pertaining to edible films and coatings. In Protein-Based Films and Coatings; Gennadios, A., Ed.; CRC Press: New York, NY, USA, 2002; pp. 601–620. [Google Scholar]
- Pérez-Gago, M.B.; Krochta, J. Protein-based films and coatings. In Edible Coatings and Films to Improve Food Quality; CRC Press: Boca Raton, FL, USA, 2012; pp. 13–77. [Google Scholar]
- Hosseini, S.F.; Rezaei, M.; Zandi, M.; Ghavi, F.F. Preparation and functional properties of fish gelatin–chitosan blend edible films. Food Chem. 2013, 136, 1490–1495. [Google Scholar] [CrossRef]
- Hosseini, S.F.; Rezaei, M.; Zandi, M.; Farahmandghavi, F. Development of bioactive fish gelatin/chitosan nanoparticles composite films with antimicrobial properties. Food Chem. 2016, 194, 1266–1274. [Google Scholar] [CrossRef]
- Scartazzini, L.; Tosati, J.; Cortez, D.; Rossi, M.; Flôres, S.; Hubinger, M.; Di Luccio, M.; Monteiro, A. Gelatin edible coatings with mint essential oil (Mentha arvensis): Film characterization and antifungal properties. J. Food Sci. Technol. 2019, 56, 4045–4056. [Google Scholar] [CrossRef]
- Wu, J.; Sun, X.; Guo, X.; Ge, S.; Zhang, Q. Physicochemical properties, antimicrobial activity and oil release of fish gelatin films incorporated with cinnamon essential oil. Aquac. Fish. 2017, 2, 185–192. [Google Scholar] [CrossRef]
- Rashidi, M.J.; Nasiraie, L.R.; Zomorrodi, S.; Jafarian, S. Development and characterization of novel active opopanax gum and gelatin bio-nanocomposite film containing zinc oxide nanoparticles and peppermint essential oil. J. Food Meas. Charact. 2023, 17, 1953–1961. [Google Scholar] [CrossRef]
- Moghadam, F.A.M.; Khoshkalampour, A.; Moghadam, F.A.M.; PourvatanDoust, S.; Naeijian, F.; Ghorbani, M. Preparation and physicochemical evaluation of casein/basil seed gum film integrated with guar gum/gelatin based nanogel containing lemon peel essential oil for active food packaging application. Int. J. Biol. Macromol. 2023, 224, 786–796. [Google Scholar] [CrossRef]
- Sadat Razavi, M.; Golmohammadi, A.; Nematollahzadeh, A.; Ghanbari, A.; Davari, M.; Carullo, D.; Farris, S. Production of Innovative essential oil-based emulsion coatings for fungal growth control on postharvest fruits. Foods 2022, 11, 1602. [Google Scholar] [CrossRef] [PubMed]
- George, J.; Hatna, S. High performance edible nanocomposite films containing bacterial cellulose nanocrystals. Carbohydr. Polym. 2012, 87, 2031–2037. [Google Scholar] [CrossRef]
- Dehnad, D.; Emam-Djomeh, Z.; Mirzaei, H.; Jafari, S.-M.; Dadashi, S. Optimization of physical and mechanical properties for chitosan–nanocellulose biocomposites. Carbohydr. Polym. 2014, 105, 222–228. [Google Scholar] [CrossRef]
- Karimi, S.; Dufresne, A.; Tahir, M.P.; Karimi, A.; Abdulkhani, A. Biodegradable starch-based composites: Effect of micro and nanoreinforcements on composite properties. J. Mater. Sci. 2014, 49, 4513–4521. [Google Scholar] [CrossRef]
- Mondragon, G.; Peña-Rodriguez, C.; González, A.; Eceiza, A.; Arbelaiz, A. Bionanocomposites based on gelatin matrix and nanocellulose. Eur. Polym. J. 2015, 62, 1–9. [Google Scholar] [CrossRef]
- Razavi, M.S.; Golmohammadi, A.; Nematollahzadeh, A.; Rovera, C.; Farris, S. Cinnamon essential oil encapsulated into a fish gelatin-bacterial cellulose nanocrystals complex and active films thereof. Food Biophys. 2022, 17, 38–46. [Google Scholar] [CrossRef]
- Taokaew, S.; Seetabhawang, S.; Siripong, P.; Phisalaphong, M. Biosynthesis and characterization of nanocellulose-gelatin films. Materials 2013, 6, 782–794. [Google Scholar] [CrossRef]
- Yao, J.; Chen, S.; Chen, Y.; Wang, B.; Pei, Q.; Wang, H. Macrofibers with high mechanical performance based on aligned bacterial cellulose nanofibers. ACS Appl. Mater. Interfaces 2017, 9, 20330–20339. [Google Scholar] [CrossRef]
- Lin, K.-W.; Lin, H.-Y. Quality characteristics of chinese-style meatball containing bacterial cellulose (Nata). J. Food Sci. 2004, 69, SNQ107–SNQ111. [Google Scholar] [CrossRef]
- Razavi, M.S.; Golmohammadi, A.; Nematollahzadeh, A.; Fiori, F.; Rovera, C.; Farris, S. Preparation of cinnamon essential oil emulsion by bacterial cellulose nanocrystals and fish gelatin. Food Hydrocoll. 2020, 109, 106111. [Google Scholar] [CrossRef]
- Rovera, C.; Ghaani, M.; Santo, N.; Trabattoni, S.; Olsson, R.T.; Romano, D.; Farris, S. Enzymatic hydrolysis in the green production of bacterial cellulose nanocrystals. ACS Sustain. Chem. Eng. 2018, 6, 7725–7734. [Google Scholar] [CrossRef]
- Designation D 1003-00; Standard Test Method for Haze and Luminous Transmittance of Transparent Plastics. American Society for Testing and Materials: Philadelphia, PA, USA, 2000.
- Rovera, C.; Türe, H.; Hedenqvist, M.S.; Farris, S. Water vapor barrier properties of wheat gluten/silica hybrid coatings on paperboard for food packaging applications. Food Packag. Shelf Life 2020, 26, 100561. [Google Scholar] [CrossRef]
- Designation D 882-18; Standard Test Method for Tensile Properties of Thin Plastic Sheeting. American Society for Testing and Materials: Philadelphia, PA, USA, 2018.
- Cozzolino, C.A.; Campanella, G.; Türe, H.; Olsson, R.T.; Farris, S. Microfibrillated cellulose and borax as mechanical, O2-barrier, and surface-modulating agents of pullulan biocomposite coatings on BOPP. Carbohydr. Polym. 2016, 143, 179–187. [Google Scholar] [CrossRef] [PubMed]
- Yao, Y.; Ding, D.; Shao, H.; Peng, Q.; Huang, Y. Antibacterial activity and physical properties of fish gelatin-chitosan edible films supplemented with D-Limonene. Int. J. Polym. Sci. 2017, 2017, 1837171. [Google Scholar] [CrossRef]
- Jamróz, E.; Konieczna-Molenda, A.; Para, A. Ternary potato starch-furcellaran-gelatin film–a new generation of biodegradable foils. Polimery 2017, 62, 673–679. [Google Scholar] [CrossRef]
- Kilinc, D.; Ocak, B.; Özdestan-Ocak, Ö. Preparation, characterization and antioxidant properties of gelatin films incorporated with Origanum onites L. essential oil. J. Food Meas. Charact. 2021, 15, 795–806. [Google Scholar] [CrossRef]
- Unalan, I.U.; Wan, C.; Figiel, Ł.; Olsson, R.T.; Trabattoni, S.; Farris, S. Exceptional oxygen barrier performance of pullulan nanocomposites with ultra-low loading of graphene oxide. Nanotechnology 2015, 26, 275703. [Google Scholar] [CrossRef]
- Su, J.-F.; Huang, Z.; Yuan, X.-Y.; Wang, X.-Y.; Li, M. Structure and properties of carboxymethyl cellulose/soy protein isolate blend edible films crosslinked by Maillard reactions. Carbohydr. Polym. 2010, 79, 145–153. [Google Scholar] [CrossRef]
- Vogler, E.A. Structure and reactivity of water at biomaterial surfaces. Adv. Colloid Interface Sci. 1998, 74, 69–117. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Liu, H.; Ge, S.; Wang, S.; Qin, Z.; Chen, L.; Zheng, Q.; Liu, Q.; Zhang, Q. The preparation, characterization, antimicrobial stability and in vitro release evaluation of fish gelatin films incorporated with cinnamon essential oil nanoliposomes. Food Hydrocoll. 2015, 43, 427–435. [Google Scholar] [CrossRef]
- Ramos, M.; Valdés, A.; Beltran, A.; Garrigós, M.C. Gelatin-based films and coatings for food packaging applications. Coatings 2016, 6, 41. [Google Scholar] [CrossRef]
- Said, N.S.; Sarbon, N.M. Physical and mechanical characteristics of gelatin-based films as a potential food packaging material: A review. Membranes 2022, 12, 442. [Google Scholar] [CrossRef]
- Nunes, J.C.; Melo, P.T.S.; Lorevice, M.V.; Aouada, F.A.; de Moura, M.R. Effect of green tea extract on gelatin-based films incorporated with lemon essential oil. J. Food Sci. Technol. 2021, 58, 1–8. [Google Scholar] [CrossRef]
- Arpi, N.; Hardianti, E. Preparation and characterization of biodegradable film based on skin and bone fish gelatin. IOP Conf. Ser. Earth Environ. Sci. 2018, 207, 012050. [Google Scholar] [CrossRef]
- Atmaka, W.; Yudhistira, B.; Putro, M. Characteristic study of chitosan addition in Tilapia (Oreochromis niloticus) bone based gelatin film. IOP Conf. Ser. Earth Environ. Sci. 2018, 142, 012028. [Google Scholar] [CrossRef]
- Jiang, M.; Liu, S.; Du, X.; Wang, Y. Physical properties and internal microstructures of films made from catfish skin gelatin and triacetin mixtures. Food Hydrocoll. 2010, 24, 105–110. [Google Scholar] [CrossRef]
- Jridi, M.; Abdelhedi, O.; Salem, A.; Kechaou, H.; Nasri, M.; Menchari, Y. Physicochemical, antioxidant and antibacterial properties of fish gelatin-based edible films enriched with orange peel pectin: Wrapping application. Food Hydrocoll. 2020, 103, 105688. [Google Scholar] [CrossRef]
- Lee, K.-Y.; Lee, J.-H.; Yang, H.-J.; Song, K.B. Characterization of a starfish gelatin film containing vanillin and its application in the packaging of crab stick. Food Sci. Biotechnol. 2016, 25, 1023–1028. [Google Scholar] [CrossRef]
- Suderman, N.; Isa, M.; Sarbon, N. Characterization on the mechanical and physical properties of chicken skin gelatin films in comparison to mammalian gelatin films. IOP Conf. Ser. Earth Environ. Sci. 2018, 440, 012033. [Google Scholar] [CrossRef]
- Sarbon, N.M.; Badii, F.; Howell, N.K. Preparation and characterisation of chicken skin gelatin as an alternative to mammalian gelatin. Food Hydrocoll. 2013, 30, 143–151. [Google Scholar] [CrossRef]
- Nur Hanani, Z.; Roos, Y.; Kerry, J. Fourier transform ınfrared (FTIR) spectroscopic analysis of biodegradable gelatin films immersed in water. In Proceedings of the 11th International Congress on Engineering and Food, ICEF11, Athens, Greece, 22–26 May 2011; Volume 5, pp. 6–9. [Google Scholar]
- Ratnasari, I.; Yuwono, S.; Nusyam, H.; Widjanarko, S. Extraction and characterization of gelatin from different fresh water fishes as alternative sources of gelatin. Int. Food Res. J. 2013, 20. [Google Scholar]
- Etxabide, A.; Leceta, I.; Cabezudo, S.; Guerrero, P.; de la Caba, K. Sustainable fish gelatin films: From food processing waste to compost. ACS Sustain. Chem. Eng. 2016, 4, 4626–4634. [Google Scholar] [CrossRef]
- Jeyaratnam, N.; Nour, A.H.; Kanthasamy, R.; Nour, A.H.; Yuvaraj, A.; Akindoyo, J.O. Essential oil from Cinnamomum cassia bark through hydrodistillation and advanced microwave assisted hydrodistillation. Ind. Crops Prod. 2016, 92, 57–66. [Google Scholar] [CrossRef]
- Hanani, Z.N.; Roos, Y.H.; Kerry, J.P. Use and application of gelatin as potential biodegradable packaging materials for food products. Int. J. Biol. Macromol. 2014, 71, 94–102. [Google Scholar] [CrossRef]
- Haghighi, H.; De Leo, R.; Bedin, E.; Pfeifer, F.; Siesler, H.W.; Pulvirenti, A. Comparative analysis of blend and bilayer films based on chitosan and gelatin enriched with LAE (lauroyl arginate ethyl) with antimicrobial activity for food packaging applications. Food Packag. Shelf Life 2019, 19, 31–39. [Google Scholar] [CrossRef]
- Kumari, S.; Rath, P.; Kumar, A.S.H. Chitosan from shrimp shell (Crangon crangon) and fish scales (Labeorohita): Extraction and characterization Suneeta. Afr. J. Biotechnol. 2016, 15, 1258–1268. [Google Scholar]
- Hoque, M.S.; Benjakul, S.; Prodpran, T. Effect of heat treatment of film-forming solution on the properties of film from cuttlefish (Sepia pharaonis) skin gelatin. J. Food Eng. 2010, 96, 66–73. [Google Scholar] [CrossRef]
- Bergo, P.; Sobral, P.J.d.A. Effects of plasticizer on physical properties of pigskin gelatin films. Food Hydrocoll. 2007, 21, 1285–1289. [Google Scholar] [CrossRef]
- Jamili, S.; Sadeghi, H.; Rezayat, M.; Attar, H.; Kaymaram, F. Extraction and evaluation of gelatin from yellow fin tuna (Thunnus albacares) skin and prospect as an alternative to mammalian gelatin. Iran. J. Fish. Sci. 2019, 18, 903–914. [Google Scholar]
- Das, M.P.; Suguna, P.; Prasad, K.; Vijaylakshmi, J.; Renuka, M. Extraction and characterization of gelatin: A functional biopolymer. Int. J. Pharm. Pharm. Sci. 2017, 9, 239. [Google Scholar] [CrossRef]
- Chi, K.; Catchmark, J.M. The influences of added polysaccharides on the properties of bacterial crystalline nanocellulose. Nanoscale 2017, 9, 15144–15158. [Google Scholar] [CrossRef]
- Vasconcelos, N.F.; Feitosa, J.P.A.; da Gama, F.M.P.; Morais, J.P.S.; Andrade, F.K.; de Souza, M.D.S.M.; de Freitas Rosa, M. Bacterial cellulose nanocrystals produced under different hydrolysis conditions: Properties and morphological features. Carbohydr. Polym. 2017, 155, 425–431. [Google Scholar] [CrossRef] [PubMed]
- Lima, H.L.S.; Gonçalves, C.; Cerqueira, M.Â.; do Nascimento, E.S.; Gama, M.F.; Rosa, M.F.; Borges, M.d.F.; Pastrana, L.M.; Brígida, A.I.S. Bacterial cellulose nanofiber-based films incorporating gelatin hydrolysate from tilapia skin: Production, characterization and cytotoxicity assessment. Cellulose 2018, 25, 6011–6029. [Google Scholar] [CrossRef]
- Voronova, M.I.; Surov, O.V.; Guseinov, S.S.; Barannikov, V.P.; Zakharov, A.G. Thermal stability of polyvinyl alcohol/nanocrystalline cellulose composites. Carbohydr. Polym. 2015, 130, 440–447. [Google Scholar] [CrossRef]
- Núñez-Flores, R.; Giménez, B.; Fernández-Martín, F.; López-Caballero, M.; Montero, M.; Gómez-Guillén, M. Physical and functional characterization of active fish gelatin films incorporated with lignin. Food Hydrocoll. 2013, 30, 163–172. [Google Scholar] [CrossRef]
- Santos, T.M.; Men de Sá Filho, M.S.; Caceres, C.A.; Rosa, M.F.; Morais, J.P.S.; Pinto, A.M.; Azeredo, H.M. Fish gelatin films as affected by cellulose whiskers and sonication. Food Hydrocoll. 2014, 41, 113–118. [Google Scholar] [CrossRef]
- Cazón, P.; Velazquez, G.; Ramírez, J.A.; Vázquez, M. Polysaccharide-based films and coatings for food packaging: A review. Food Hydrocoll. 2017, 68, 136–148. [Google Scholar] [CrossRef]
- Gómez-Estaca, J.; De Lacey, A.L.; López-Caballero, M.; Gómez-Guillén, M.; Montero, P. Biodegradable gelatin–chitosan films incorporated with essential oils as antimicrobial agents for fish preservation. Food Microbiol. 2010, 27, 889–896. [Google Scholar] [CrossRef] [PubMed]
- Kavoosi, G.; Rahmatollahi, A.; Dadfar, S.M.M.; Purfard, A.M. Effects of essential oil on the water binding capacity, physico-mechanical properties, antioxidant and antibacterial activity of gelatin films. LWT-Food Sci. Technol. 2014, 57, 556–561. [Google Scholar] [CrossRef]

| Sample | δ (µm) | TT (%) | H (%) | θ (°) | WS (%) |
|---|---|---|---|---|---|
| Control | 24.20 ± 1.75 a | 90.55 ± 0.30 a | 0.92 ± 0.02 a | 78.26 ± 3.51 a | 52.08 ± 1.76 da |
| T1G | 26.90 ± 3.66 b | 90.51 ± 0.20 a | 0.96 ± 0.01 a | 95.97 ± 4.25 b | 31.90 ± 0.24 cb |
| T2G | 29.66 ± 1.75 c | 90.46 ± 0.40 a | 1.01 ± 0.02 a | 94.25 ± 0.69 b | 31.37 ± 0.51 bc |
| T3G | 31.33 ± 2.06 d | 90.37 ± 0.60 a | 1.07 ± 0.03 a | 97.19 ± 1.65 b | 30.45 ± 0.38 bcd |
| T4G | 36.62 ± 3.06 e | 90.32 ± 0.50 a | 1.16 ± 0.02 a | 98.50 ± 3.64 b | 29.45 ± 1.11 bcd |
| T5G | 32.00 ± 2.39 f | 90.29 ± 0.80 a | 2.58 ± 0.03 b | 97.40 ± 3.19 b | 29.12 ± 0.63 cd |
| T6G | 34.25 ± 1.66 g | 89.38 ± 0.60 a | 3.18 ± 0.04 c | 94.10 ± 0.84 b | 28.48 ± 0.52 d |
| Sample | TS (MPa) | EAB (%) | E (MPa) |
|---|---|---|---|
| Control | 8.98 ± 0.31 a | 0.84 ± 0.13 a | 316.03 ± 10.70 a |
| T1G | 7.78 ± 0.54 ab | 0.93 ± 0.05 a | 307.73 ± 12.03 a |
| T2G | 7.29 ± 0.37 b | 1.10 ± 0.13 ab | 297.22 ± 13.15 ab |
| T3G | 6.64 ± 0.74 b | 1.21 ± 0.32 ab | 284.70 ± 20.86 ab |
| T4G | 4.42 ± 0.58 c | 1.75 ± 0.30 b | 254.75 ± 18.76 ab |
| T5G | 2.56 ± 0.83 d | 2.90 ± 0.34 c | 226.95 ± 57.03 ac |
| T6G | 1.93 ± 0.20 d | 3.79 ± 0.47 d | 220.29 ± 27.22 c |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Golmohammadi, A.; Razavi, M.S.; Tahmasebi, M.; Carullo, D.; Farris, S. Cinnamon Essential-Oil-Loaded Fish Gelatin–Cellulose Nanocrystal Films Prepared under Acidic Conditions. Coatings 2023, 13, 1360. https://doi.org/10.3390/coatings13081360
Golmohammadi A, Razavi MS, Tahmasebi M, Carullo D, Farris S. Cinnamon Essential-Oil-Loaded Fish Gelatin–Cellulose Nanocrystal Films Prepared under Acidic Conditions. Coatings. 2023; 13(8):1360. https://doi.org/10.3390/coatings13081360
Chicago/Turabian StyleGolmohammadi, Abdollah, Mahsa Sadat Razavi, Mohammad Tahmasebi, Daniele Carullo, and Stefano Farris. 2023. "Cinnamon Essential-Oil-Loaded Fish Gelatin–Cellulose Nanocrystal Films Prepared under Acidic Conditions" Coatings 13, no. 8: 1360. https://doi.org/10.3390/coatings13081360
APA StyleGolmohammadi, A., Razavi, M. S., Tahmasebi, M., Carullo, D., & Farris, S. (2023). Cinnamon Essential-Oil-Loaded Fish Gelatin–Cellulose Nanocrystal Films Prepared under Acidic Conditions. Coatings, 13(8), 1360. https://doi.org/10.3390/coatings13081360









