Packaging of Fresh Poultry Meat with Innovative and Sustainable ZnO/Pectin Bionanocomposite Films—A Contribution to the Bio and Circular Economy
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials and Reagents
2.2. Preparation of Passionfruit and Tomato Extract
2.3. Synthesis of ZnO Nanoparticles
2.4. Physicochemical Characterization of ZnO NPs
2.5. Preparation of Pectin Films
2.6. Physicochemical Characterization of Films
2.7. Use of Coatings in Fresh Poultry Meat
2.7.1. Antibacterial Properties of Films
2.7.2. Physicochemical Characterization of Poultry Meat
2.7.3. Migration of Zinc Oxide Nanoparticles to Poultry Meat
2.7.4. Poultry Meat Color
2.8. Statistical Analysis
3. Results and Discussion
3.1. ZnO Nanocomposite Film Characterization
3.2. Application of the Bionanocoatings in Fresh Poultry Meat
3.2.1. Microbiological Growth
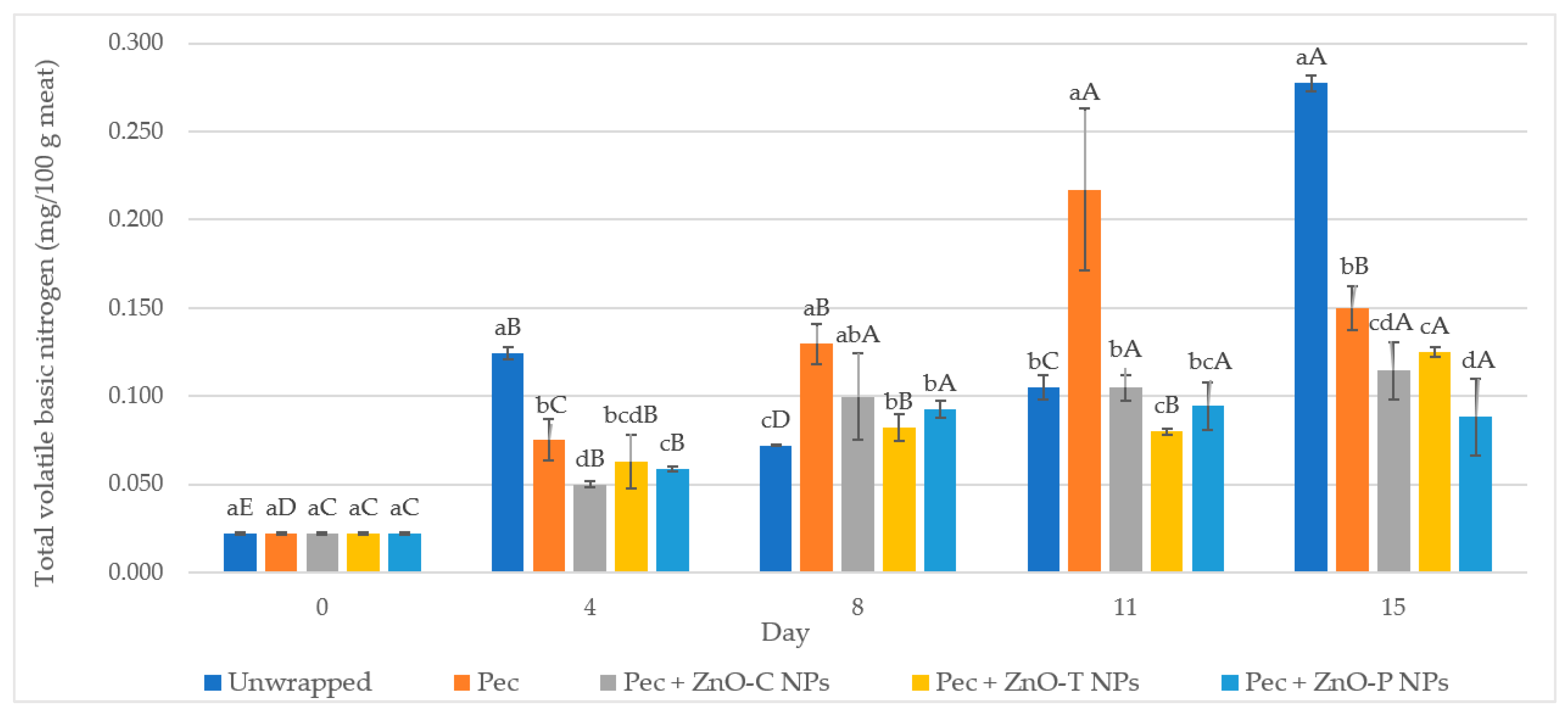
3.2.2. Physicochemical Characterization of Poultry Meat
3.2.3. Total Zinc Migration to Poultry Meat
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Corrado, S.; Sala, S. Food waste accounting along global and European food supply chains: State of the art and outlook. Waste Manag. 2018, 79, 120–131. [Google Scholar] [CrossRef]
- Alam, A.U.; Rathi, P.; Beshai, H.; Sarabha, G.K.; Deen, M.J. Fruit Quality Monitoring with Smart Packaging. Sensors 2021, 21, 1509. [Google Scholar] [CrossRef] [PubMed]
- Ncube, L.K.; Ude, A.U.; Ogunmuyiwa, E.N.; Zulkifli, R.; Beas, I.N. An overview of plastic waste generation and management in food packaging industries. Recycling 2021, 6, 12. [Google Scholar] [CrossRef]
- Pires, J.R.A.; Souza, V.G.L.; Fuciños, P.; Pastrana, L.; Fernando, A.L. Methodologies to assess the biodegradability of bio-based Polymers—Current knowledge and existing gaps. Polymers 2022, 14, 1359. [Google Scholar] [CrossRef] [PubMed]
- Galus, S.; Arik Kibar, A.E.; Gniewosz, M.; Kraśniewska, K. Novel materials in the preparation of edible films and coatings—A review. Coatings 2020, 10, 674. [Google Scholar] [CrossRef]
- Rosenboom, J.G.; Langer, R.; Traverso, G. Bioplastics for a circular economy. Nat. Rev. Mater. 2022, 7, 117–137. [Google Scholar] [CrossRef] [PubMed]
- Freitas, C.M.P.; Coimbra, J.S.R.; Souza, V.G.L.; Sousa, R.C.S. Structure and Applications of Pectin in Food, Biomedical, and Pharmaceutical Industry: A Review. Coatings 2021, 11, 922. [Google Scholar] [CrossRef]
- Rodrigues, C.; Souza, V.G.L.; Rashad, M.; Pari, L.; Outzourhit, A.; Fernando, A.L. Mucilage extraction from Opuntia spp. For production of biofilms. In Proceedings of the 27th European Biomass Conference and Exhibition, Lisbon, Portugal, 27–30 May 2019; pp. 1456–1459. [Google Scholar]
- Anugrah, D.S.B.; Alexander, H.; Pramitasari, R.; Hudiyanti, D.; Sagita, C.P. A Review of Polysaccharide-Zinc Oxide Nanocomposites as Safe Coating for Fruits Preservation. Coatings 2020, 10, 988. [Google Scholar] [CrossRef]
- Avramescu, S.M.; Butean, C.; Popa, C.V.; Ortan, A.; Moraru, I.; Temocico, G. Edible and Functionalized Films/Coatings—Performances and Perspectives. Coatings 2020, 10, 687. [Google Scholar] [CrossRef]
- Pires, J.; de Paula, C.D.; Souza, V.G.L.; Fernando, A.L.; Coelhoso, I. Understanding the barrier and mechanical behavior of different nanofillers in chitosan films for food packaging. Polymers 2021, 13, 721. [Google Scholar] [CrossRef]
- Souza, V.G.L.; Mello, I.P.; Khalid, O.; Pires, J.R.A.; Rodrigues, C.; Alves, M.M.; Santos, C.; Fernando, A.L.; Coelhoso, I. Strategies to Improve the Barrier and Mechanical Properties of Pectin Films for Food Packaging: Comparing Nanocomposites with Bilayers. Coatings 2022, 12, 108. [Google Scholar] [CrossRef]
- Souza, V.G.L.; Alves, M.M.; Santos, C.F.; Ribeiro, I.A.C.; Rodrigues, C.; Coelhoso, I.; Fernando, A.L. Biodegradable chitosan films with ZnO nanoparticles synthesized using food industry by-products—Production and characterization. Coatings 2021, 11, 646. [Google Scholar] [CrossRef]
- Al-Naamani, L.; Dobretsov, S.; Dutta, J. Chitosan-zinc oxide nanoparticle composite coating for active food packaging applications. Innov. Food Sci. Emerg. Technol. 2016, 38, 231–237. [Google Scholar] [CrossRef]
- Rezaei, F.; Shahbazi, Y. Shelf-life extension and quality attributes of sauced silver carp fillet: A comparison among direct addition, edible coating and biodegradable film. LWT 2018, 87, 122–133. [Google Scholar] [CrossRef]
- Ngo, T.M.P.; Dang, T.M.Q.; Tran, T.X.; Rachtanapun, P. Effects of zinc oxide nanoparticles on the properties of pectin/alginate edible films. Int. J. Polym. Sci. 2018, 2018, 5645797. [Google Scholar] [CrossRef]
- El Fawal, G.; Hong, H.; Song, X.; Wu, J.; Sun, M.; He, C.; Wang, H. Fabrication of antimicrobial films based on hydroxy-ethylcellulose and ZnO for food packaging application. Food Packag. Shelf Life 2020, 23, 100462. [Google Scholar] [CrossRef]
- Dwivedi, L.M.; Baranwal, K.; Gupta, S.; Mishra, M.; Sundaram, S.; Singh, V. Antibacterial nanostructures derived from oxidized sodium alginate-ZnO. Int. J. Biol. Macromol. 2020, 149, 1323–1330. [Google Scholar] [CrossRef] [PubMed]
- Singh, V.; Dwivedi, L.M.; Baranwal, K.; Asthana, S.; Sundaram, S. Oxidized guar gum–ZnO hybrid nanostructures: Synthesis, characterization and antibacterial activity. Appl. Nanosci. 2018, 8, 1149–1160. [Google Scholar] [CrossRef]
- Siddiqui, S.; Alrumman, S.A. Influence of nanoparticles on food: An analytical assessment. J. King Saud Univ. -Sci. 2021, 33, 101530. [Google Scholar] [CrossRef]
- Sun, Q.; Li, J.; Le, T. Zinc oxide nanoparticle as a novel class of antifungal agents: Current advances and future perspectives. J. Agric. Food Chem. 2018, 66, 11209–11220. [Google Scholar] [CrossRef]
- Malik, G.K.; Mitra, J. Zinc Oxide Nanoparticle Synthesis, Characterization, and Their Effect on Mechanical, Barrier, and Optical Properties of HPMC-Based Edible Film. Food Bioprocess Technol. 2021, 14, 441–456. [Google Scholar] [CrossRef]
- Dodero, A.; Alloisio, M.; Vicini, S.; Castellano, M. Preparation of composite alginate-based electrospun membranes loaded with ZnO nanoparticles. Carbohydr. Polym. 2020, 227, 115371. [Google Scholar] [CrossRef] [PubMed]
- Souza, V.G.L.; Rodrigues, C.; Valente, S.; Pimenta, C.; Pires, J.R.A.; Alves, M.M.; Fernando, A.L. Eco-friendly ZnO/Chitosan bionanocomposites films for packaging of fresh poultry meat. Coatings 2020, 10, 110. [Google Scholar] [CrossRef]
- Bandeira, M.; Giovanela, M.; Roesch-Ely, M.; Devine, D.M.; Crespo, J.S. Green synthesis of zinc oxide nanoparticles: A review of the synthesis methodology and mechanism of formation. Sustain. Chem. Pharm. 2020, 15, 100223. [Google Scholar] [CrossRef]
- Alves, M.M.; Andrade, S.M.; Grenho, L.; Fernandes, M.H.; Santos, C.; Montemor, M.F. Influence of apple phytochemicals in ZnO nanoparticles formation, photoluminescence and biocompatibility for biomedical applications. Mater. Sci. Eng. C 2019, 101, 76–87. [Google Scholar] [CrossRef] [PubMed]
- ISO 4833-1:2013; Microbiology of the Food Chain—Horizontal Method for the Enumeration of Microorganisms—Part 1: Colony Count at 30 Degrees C by the Pour Plate Technique. ISO: Geneva, Switzerland, 2013.
- ISO 17410:2019; Microbiology of the Food Chain—Horizontal Method for the Enumeration of Psychrotrophic Microorganisms. ISO: Geneva, Switzerland, 2001.
- ISO 21528-2:2017; Microbiology of the Food Chain—Horizontal Method for the Detection and Enumeration of Enterobacteriaceae—Part 2: Colony-Count Technique. ISO: Geneva, Switzerland, 2017.
- Official Methods of Analysis of the Association of Official Analytical Chemists, 20th ed.; Association of the Official Analytical Chemists (AOAC): Rockville, MD, USA, 2016.
- Malle, P.; Poumeyrol, M. A New Chemical Criterion for the Quality Control of Fish: Trimethylamine/Total Volatile Basic Nitrogen (%). J. Food Prot. 1989, 52, 419–423. [Google Scholar] [CrossRef]
- Souza, V.G.L.; Pires, J.R.; Vieira, É.T.; Coelhoso, I.M.; Duarte, M.P.; Fernando, A.L. Shelf Life Assessment of Fresh Poultry Meat Packaged in Novel Bionanocomposite of Chitosan/Montmorillonite Incorporated with Ginger Essential Oil. Coatings 2018, 8, 177. [Google Scholar] [CrossRef]
- Vandecasteele, C.; Block, C.B. Modern Methods for Trace Element Determination; Wiley: Hoboken, NJ, USA, 1997; ISBN 978-0-471-97445-1. [Google Scholar]
- Souza, V.G.L.; Rodrigues, P.F.; Duarte, M.P.; Fernando, A.L. Antioxidant Migration Studies in Chitosan Films Incorporated with Plant Extracts. J. Renew. Mater. 2018, 6, 548–558. [Google Scholar] [CrossRef]
- Fonseca, A.M.A.; Geraldi, M.V.; Maróstica-Junior, M.R.; Silvestre, A.J.D.; Rocha, S.M. Purple passion fruit (Passiflora edulis f. edulis): A comprehensive review on the nutritional value, phytochemical profile and associated health effects. Food Res. Int. 2022, 160, 111665. [Google Scholar] [CrossRef]
- Chaudhary, P.; Sharma, A.; Singh, B.; Nagpal, A.K. Bioactivities of phytochemicals present in tomato. J. Food Sci. Technol. 2018, 55, 2833–2849. [Google Scholar] [CrossRef]
- Espitia, P.J.P.; Soares, N.d.F.F.; dos Reis Coimbra, J.S.; de Andrade, N.J.; Cruz, R.S.; Medeiros, E.A.A. Zinc Oxide Nanoparticles: Synthesis, Antimicrobial Activity and Food Packaging Applications. Food Bioprocess Technol. 2012, 5, 1447–1464. [Google Scholar] [CrossRef]
- Shankar, S.; Teng, X.; Li, G.; Rhim, J.W. Preparation, characterization, and antimicrobial activity of gelatin/ZnO nanocomposite films. Food Hydrocoll. 2015, 45, 264–271. [Google Scholar] [CrossRef]
- Malhotra, B.; Keshwani, A.; Kharkwal, H. Antimicrobial food packaging: Potential and pitfalls. Front. Microbiol. 2015, 6, 611. [Google Scholar] [CrossRef] [PubMed]
- Souza, V.G.L.; Pires, J.R.A.; Vieira, É.T.; Coelhoso, I.M.; Duarte, M.P.; Fernando, A.L. Activity of chitosan-montmorillonite bionanocomposites incorporated with rosemary essential oil: From in vitro assays to application in fresh poultry meat. Food Hydrocoll. 2019, 89, 241–252. [Google Scholar] [CrossRef]
- Kaewprachu, P.; Osako, K.; Benjakul, S.; Rawdkuen, S. Quality attributes of minced pork wrapped with catechin-lysozyme incorporated gelatin film. Food Packag. Shelf Life 2015, 3, 88–96. [Google Scholar] [CrossRef]
- Pires, J.R.A.; de Souza, V.G.L.; Fernando, A.L. Chitosan/montmorillonite bionanocomposites incorporated with rosemary and ginger essential oil as packaging for fresh poultry meat. Food Packag. Shelf Life 2018, 17, 142–149. [Google Scholar] [CrossRef]
- Bonilla, J.; Vargas, M.; Atarés, L.; Chiralt, A. Effect of Chitosan Essential Oil Films on the Storage-Keeping Quality of Pork Meat Products. Food Bioprocess Technol. 2014, 7, 2443–2450. [Google Scholar] [CrossRef]
- The Commision of the European Communities. Commission Regulation (EC) No 2073/2005 of 15 November 2005 on Microbiological Criteria for Foodstuffs; Elsevier: Amsterdam, The Netherlands, 2005; Volume L338. [Google Scholar]
- Coma, V. Bioactive packaging technologies for extended shelf life of meat-based products. Meat Sci. 2008, 78, 90–103. [Google Scholar] [CrossRef]
- Economou, T.; Pournis, N.; Ntzimani, A.; Savvaidis, I.N. Nisin-EDTA treatments and modified atmosphere packaging to increase fresh chicken meat shelf-life. Food Chem. 2009, 114, 1470–1476. [Google Scholar] [CrossRef]
- Alves, V.D.; Costa, N.; Coelhoso, I.M. Barrier properties of biodegradable composite films based on kappa-carrageenan/pectin blends and mica flakes. Carbohydr. Polym. 2010, 79, 269–276. [Google Scholar] [CrossRef]
- Cerisuelo, J.P.; Alonso, J.; Aucejo, S.; Gavara, R.; Hernández-Muñoz, P. Modifications induced by the addition of a nanoclay in the functional and active properties of an EVOH film containing carvacrol for food packaging. J. Memb. Sci. 2012, 423–424, 247–256. [Google Scholar] [CrossRef]
- Rodrigues, C.; Souza, V.G.L.; Coelhoso, I.; Fernando, A.L. Bio-Based Sensors for Smart Food Packaging—Current Applications and Future Trends. Sensors 2021, 21, 2148. [Google Scholar] [CrossRef] [PubMed]
- Hari, K.D.; Garcia, C.V.; Shin, G.H.; Kim, J.T. Improvement of the UV barrier and antibacterial properties of crosslinked pectin/zinc oxide bionanocomposite films. Polymers 2021, 13, 2403. [Google Scholar] [CrossRef]
- Jalal, M.; Ansari, M.A.; Ali, S.G.; Khan, H.M.; Rehman, S. Anticandidal activity of bioinspired ZnO NPs: Effect on growth, cell morphology and key virulence attributes of Candida species. Artif. Cells Nanomed. Biotechnol. 2018, 46, 912–925. [Google Scholar] [CrossRef]
- Sharaby, M.R.; Soliman, E.A.; Abdel-Rahman, A.B.; Osman, A.; Khalil, R. Novel pectin-based nanocomposite film for active food packaging applications. Sci. Rep. 2022, 12, 20673. [Google Scholar] [CrossRef] [PubMed]
- Mohammadi, H.; Kamkar, A.; Misaghi, A.; Zunabovic-Pichler, M.; Fatehi, S. Nanocomposite films with CMC, okra mucilage, and ZnO nanoparticles: Extending the shelf-life of chicken breast meat. Food Packag. Shelf Life 2019, 21, 100330. [Google Scholar] [CrossRef]
- Mikus, M.; Galus, S. Food Coating–Materials, Methods and Applications in Food Industry. Food Sci. Technol. Qual. 2020, 125, 5–24. [Google Scholar] [CrossRef]
- Suo, B.; Li, H.; Wang, Y.; Li, Z.; Pan, Z.; Ai, Z. Effects of ZnO nanoparticle-coated packaging film on pork meat quality during cold storage. J. Sci. Food Agric. 2016, 97, 2023–2029. [Google Scholar] [CrossRef]
- Eldaly, E.A.; Fikry, A.; Mahmoud, A.; Abobakr, H.M. Preservative Effect of Chitosan Coating on Shelf Life and Sensory Properties of Chicken Fillets during Chilled Storage. J. Nutr. Food Secur. 2018, 3, 139–148. Available online: https://jnfs.ssu.ac.ir/article-1-195-en.pdf (accessed on 23 June 2023).
- Amjadi, S.; Emaminia, S.; Nazari, M.; Davudian, S.H.; Roufegarinejad, L.; Hamishehkar, H. Application of Reinforced ZnO Nanoparticle-Incorporated Gelatin Bionanocomposite Film with Chitosan Nanofiber for Packaging of Chicken Fillet and Cheese as Food Models. Food Bioprocess Technol. 2019, 12, 1205–1219. [Google Scholar] [CrossRef]
- Meindrawan, B.; Putri, S.; Susanto, C.S.; Ofe, O.; Mangindaan, D.; Ayman, A.; Kasih, T.P. Bionanocomposite of Gelatin–ZnO Nanoparticles as Potential Edible Coating for Broiler Chicken Fillet. Macromol. Symp. 2020, 391, 1900165. [Google Scholar] [CrossRef]
- Naveen, Z.; Eswara, R.B.; Mallika, E.N.; Sreenivasa, R.T.; Nath, D.N.; Prasad, T.N.V.K.V. Evaluating the NanoZinc Oxide Composite Polymer Films Developed for Packaging Chicken Meat against pH Variations during Refrigerated Storage. Indian Vet. J. 2022, 99, 75–79. [Google Scholar]
- Khulal, U.; Zhao, J.; Hu, W.; Chen, Q. Intelligent evaluation of total volatile basic nitrogen (TVB-N) content in chicken meat by an improved multiple level data fusion model. Sens. Actuators B Chem. 2017, 238, 337–345. [Google Scholar] [CrossRef]
- Pellissery, A.J.; Vinayamohan, P.G.; Amalaradjou, M.A.R.; Venkitanarayanan, K. Spoilage bacteria and meat quality. In Meat Quality Analysis; Academic Press: Cambridge, MA, USA, 2019; pp. 307–334. [Google Scholar] [CrossRef]
- Kolakowska, A.; Bartosz, G. Oxidation of Food Components: An Introduction. In Food Oxidants and Antioxidants Chemical, Biological and Functional Properties; Bartosz, G., Ed.; CRC Press: Boca Raton, FL, USA, 2014; p. 550. ISBN 978-1-4398-8242-9. [Google Scholar]
- Baek, S.K.; Song, K. Bin Development of Gracilaria vermiculophylla extract films containing zinc oxide nanoparticles and their application in smoked salmon packaging. LWT-Food Sci. Technol. 2018, 89, 269–275. [Google Scholar] [CrossRef]
- Panea, B.; Ripoll, G.; González, J.; Fernández-Cuello, Á.; Albertí, P. Effect of nanocomposite packaging containing different proportions of ZnO and Ag on chicken breast meat quality. J. Food Eng. 2014, 123, 104–112. [Google Scholar] [CrossRef]
- Szabo, K.; Dulf, F.V.; Diaconeasa, Z.; Vodnar, D.C. Antimicrobial and antioxidant properties of tomato processing byproducts and their correlation with the biochemical composition. LWT 2019, 116, 108558. [Google Scholar] [CrossRef]
- López-Vargas, J.H.; Fernández-López, J.; Pérez-Álvarez, J.A.; Viuda-Martos, M. Chemical, physico-chemical, technological, antibacterial and antioxidant properties of dietary fiber powder obtained from yellow passion fruit (Passiflora edulis var. flavicarpa) co-products. Food Res. Int. 2013, 51, 756–763. [Google Scholar] [CrossRef]
- Kenar, M.; Ozogul, F.; Kuley, E. Effects of rosemary and sage tea extracts on the sensory, chemical and microbiological changes of vacuum-packed and refrigerated sardine (Sardina pilchardus) fillets. Int. J. Food Sci. Technol. 2010, 45, 2366–2372. [Google Scholar] [CrossRef]
- Minolta Precise Color Communication: Color Control from Perception to Instrumentation; Minolta Co., Ltda.: Tokyo, Japan, 2007.
- Souza, V.G.L.; Ribeiro-Santos, R.; Rodrigues, P.F.; Otoni, C.G.; Duarte, M.P.; Coelhoso, I.M.; Fernando, A.L. Nanomaterial migration from composites into food matrices. In Composite Materials for Food Packaging; Cirillo, G., Kozlowski, M.A., Spizzirri, U.G., Eds.; Scrivener Publishing LLC: Beverly, MA, USA, 2018; p. 465. ISBN 9781119160205. [Google Scholar]
- European Food Safety Authority (EFSA). Safety assessment of the substance zinc oxide, nanoparticles, for use in food contact materials. EFSA J. 2017, 14, 4408. [Google Scholar] [CrossRef]
- INSA PortFIR-Composition of Poultry Meat (Skinless Chicken Breast, Raw). Available online: https://portfir-insa.min-saude.pt/foodcomp/food?22601 (accessed on 5 January 2023).
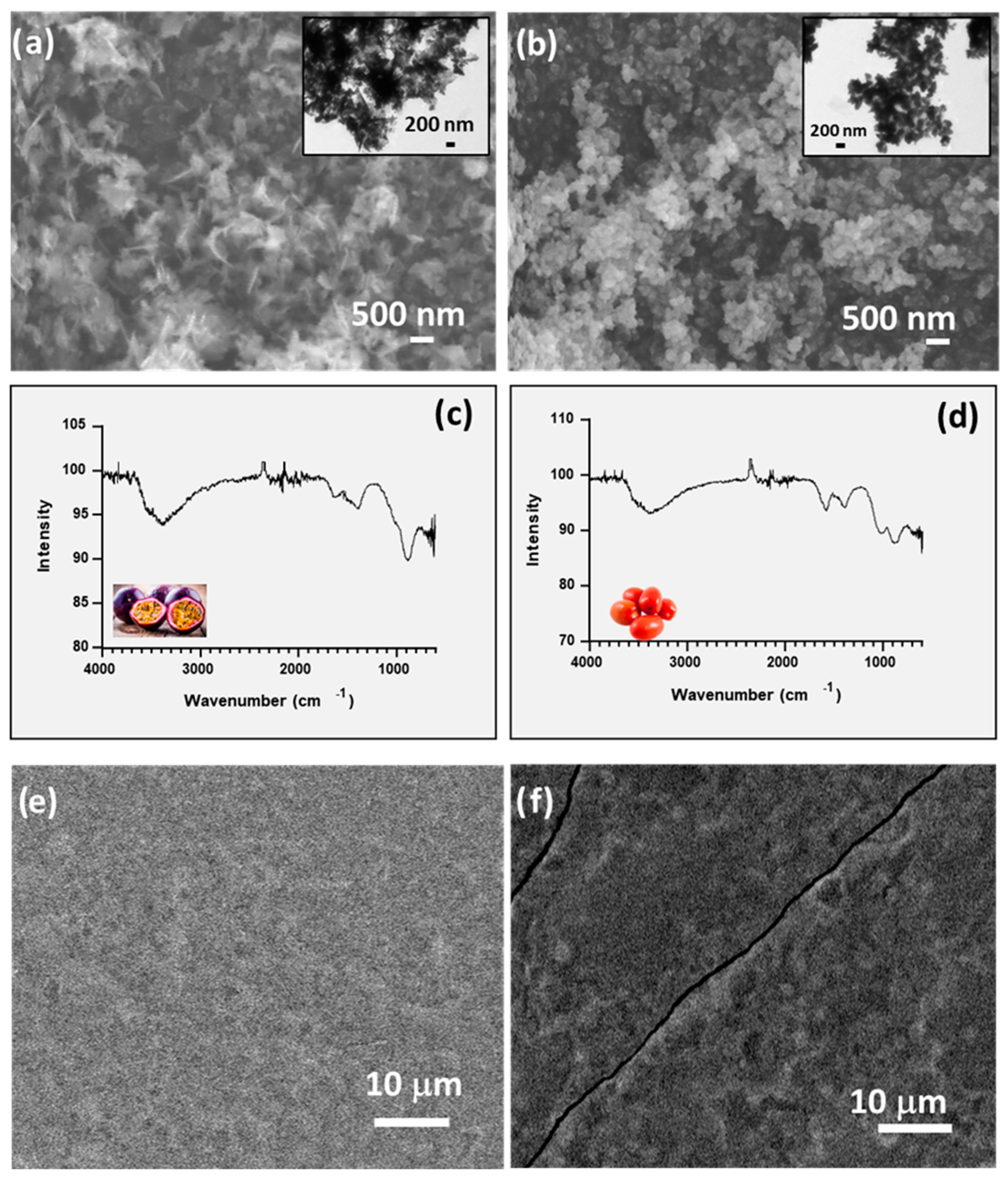
| Parameter | Day | Unwrapped | Pec | Pec + ZnO-C NPs | Pec + ZnO-T NPs | Pec + ZnO- P NPs |
|---|---|---|---|---|---|---|
| Total mesophilic aerobic microorganisms (Log CFU/g meat) | 0 | 4.86 ± 0.06 aD | 4.86 ± 0.06 aC | 4.86 ± 0.06 aC | 4.86 ± 0.06 aC | 4.86 ± 0.06 aC |
| 4 | 7.69 ± 0.08 aC | 7.29 ± 0.53 abcB | 7.02 ± 0.04 bB | 6.87 ± 0.02 cB | 6.93 ± 0.23 bcB | |
| 8 | 10.55 ± 0.78 aB | 9.59 ± 0.66 aA | 10.71 ± 1.49 aA | 10.49 ± 1.47 aA | 10.76 ± 1.55 aA | |
| 11 | 13.33 ± 0.57 aA | 9.60 ± 0.31 bA | 8.96 ± 0.33 bcA | 8.71 ± 0.29 cA | 8.94 ± 0.30 bcA | |
| 15 | 12.17 ± 1.18 aAB | 9.70 ± 1.37 aA | 9.74 ± 1.36 aA | 9.69 ± 1.29 aA | 10.00 ± 1.42 aA | |
| Total psychrotropic aerobic microorganisms (Log CFU/g meat) | 0 | 3.43 ± 0.20 aC | 3.43 ± 0.20 aC | 3.43 ± 0.20 aD | 3.43 ± 0.20 aC | 3.43 ± 0.20 aD |
| 4 | 6.99 ± 0.16 aB | 6.72 ± 1.29 aB | 7.09 ± 0.38 aC | 6.83 ± 0.46 aB | 7.05 ± 0.68 aC | |
| 8 | 10.51 ± 0.90 aA | 9.22 ± 0.78 abA | 9.82 ± 1.11 abAB | 9.84 ± 1.33 abA | 8.55 ± 0.19 bB | |
| 11 | 12.06 ± 0.63 aA | 8.22 ± 1.04 bAB | 8.48 ± 0.95 bB | 8.88 ± 0.08 bA | 8.91 ± 0.34 bB | |
| 15 | 11.51 ± 0.18 aA | 9.61 ± 1.34 bA | 10.43 ± 0.78 bA | 9.93 ± 1.32 bA | 10.50 ± 0.70 bA | |
| Enterobacteriaceae (Log CFU/g meat) | 0 | 2.36 ± 0.17 aE | 2.36 ± 0.17 aC | 2.36 ± 0.17 aD | 2.36 ± 0.17 aC | 2.36 ± 0.17 aD |
| 4 | 5.71 ± 0.10 aD | 5.86 ± 0.89 abB | 5.62 ± 0.03 aC | 5.34 ± 0.44 abB | 5.08 ± 0.35 bC | |
| 8 | 9.54 ± 0.06 aB | 8.17 ± 0.25 cA | 8.64 ± 0.01 bA | 7.85 ± 0.71 cdA | 7.57 ± 0.01 dA | |
| 11 | 11.19 ± 0.54 aA | 8.15 ± 0.11 bA | 7.80 ± 0.17 cB | 7.81 ± 0.33 bcA | 7.00 ± 0.20 dB | |
| 15 | 8.38 ± 0.38 abC | 8.36 ± 0.58 abA | 8.22 ± 0.72 abAB | 8.31 ± 0.21 aA | 7.39 ± 0.66 bAB |
| Parameter | Day | Unwrapped | Pec | Pec + ZnO-C NPs | Pec + ZnO-T NPs | Pec + ZnO- P NPs |
|---|---|---|---|---|---|---|
| Moisture (%) | 0 | 75.3 ± 0.3 aB | 75.3 ± 0.3 aA | 75.3 ± 0.3 aA | 75.3 ± 0.3 aA | 75.3 ± 0.3 aA |
| 4 | 75.0 ± 0.4 aB | 69.5 ± 1.5 cB | 71.0 ± 0.9 bcB | 72.2 ± 0.7 bB | 72.4 ± 1.6 bcB | |
| 8 | 75.2 ± 0.6 aB | 67.3 ± 2.6 bB | 70.1 ± 1.2 bBC | 70.2 ± 1.0 bC | 70.3 ± 1.3 bBC | |
| 11 | 76.9 ± 0.5 aA | 68.5 ± 1.4 bB | 68.8 ± 0.4 bC | 68.9 ± 1.4 bC | 68.9 ± 1.5 bC | |
| 15 | 76.2 ± 0.8 aAB | 66.5 ± 2.8 bcB | 65.6 ± 1.8 bcD | 65.9 ± 0.8 cD | 69.5 ± 2.3 bBC | |
| pH | 0 | 5.96 ± 0.03 aC | 5.96 ± 0.03 aA | 5.96 ± 0.03 aA | 5.96 ± 0.03 aA | 5.96 ± 0.03 aA |
| 4 | 6.07 ± 0.03 aB | 5.90 ± 0.06 bcA | 5.76 ± 0.03 dB | 5.83 ± 0.02 cB | 5.94 ± 0.02 aA | |
| 8 | 6.16 ± 0.06 aB | 5.39 ± 0.06 cB | 5.40 ± 0.07 cC | 5.60 ± 0.04 bC | 5.67 ± 0.13 bB | |
| 11 | 6.50 ± 0.40 aB | 5.30 ± 0.04 bB | 5.19 ± 0.02 cD | 5.29 ± 0.03 bD | 5.30 ± 0.06 bC | |
| 15 | 7.62 ± 0.15 aA | 5.32 ± 0.06 bB | 5.17 ± 0.07 cdD | 5.16 ± 0.02 dE | 5.25 ± 0.04 bcC | |
| Titratable acidity (% oleic acid equivalent) | 0 | 3.80 ± 0.03 aC | 3.80 ± 0.03 aC | 3.80 ± 0.03 aC | 3.80 ± 0.03 aE | 3.80 ± 0.03 aC |
| 4 | 4.10 ± 0.05 cB | 4.65 ± 0.33 aB | 4.02 ± 0.01 cB | 4.26 ± 0.03 bD | 3.93 ± 0.03 dB | |
| 8 | 4.27 ± 0.03 bA | 5.02 ± 0.73 abB | 4.31 ± 0.38 abAB | 4.60 ± 0.02 aC | 4.14 ± 0.54 abAB | |
| 11 | 3.26 ± 0.09 cD | 4.84 ± 0.66 abB | 4.88 ± 0.48 abA | 4.66 ± 0.00 aB | 4.27 ± 0.13 bA | |
| 15 | 2.88 ± 0.04 cE | 5.85 ± 0.02 aA | 4.71 ± 0.52 bA | 4.78 ± 0.05 bA | 4.71 ± 0.43 bA | |
| TBARS (mg malonaldehyde/kg meat) | 0 | 0.26 ± 0.14 aBC | 0.26 ± 0.14 aC | 0.26 ± 0.14 aB | 0.26 ± 0.14 aC | 0.26 ± 0.14 aC |
| 4 | 0.21 ± 0.05 cC | 0.53 ± 0.15 aBC | 0.36 ± 0.07 abB | 0.41 ± 0.06 aC | 0.31 ± 0.00 bC | |
| 8 | 0.27 ± 0.12 bBC | 0.42 ± 0.02 bC | 0.42 ± 0.04 bB | 0.37 ± 0.01 bC | 0.75 ± 0.20 aB | |
| 11 | 0.56 ± 0.23 bAB | 0.99 ± 0.00 aA | 0.44 ± 0.11 bB | 0.52 ± 0.03 bB | 0.59 ± 0.08 bB | |
| 15 | 0.75 ± 0.13 cA | 0.78 ± 0.11 cB | 0.94 ± 0.02 bA | 1.25 ± 0.30 abA | 1.68 ± 0.18 aA | |
| Hue angle (°) | 0 | 55 ± 2 aA | 55 ± 2 aAB | 55 ± 2 aB | 55 ± 2 aB | 55 ± 2 aC |
| 4 | 57 ± 2 abA | 54 ± 1 bB | 60 ± 3 aAB | 58 ± 3 abAB | 58 ± 3 abBC | |
| 8 | 54 ± 3 bA | 58 ± 1 abAB | 60 ± 2 abAB | 61 ± 3 abAB | 60 ± 1 aB | |
| 11 | 61 ± 5 abA | 59 ± 3 bAB | 63 ± 2 abA | 65 ± 3 abA | 66 ± 2 aA | |
| 15 | 54 ± 4 bA | 60 ± 4 abA | 62 ± 2 abA | 63 ± 2 aA | 62 ± 2 abAB |
| Sample | Zinc Content (mg Zn/kg Fresh Meat) |
|---|---|
| Initial zinc content—day 0 | 11.2 ± 2.4 B |
| Unwrapped—day 15 | 10.3 ± 2.7 B |
| Pec—day 15 | 15.2 ± 3.1 B |
| Pec + ZnO-CNPs—day 15 | 30.3 ± 4.1 A |
| Pec + ZnO-TNPs—day 15 | 35.2 ± 3.7 A |
| Pec + ZnO-PNPs—day 15 | 38.4 ± 4.8 A |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Przybyszewska, A.; Barbosa, C.H.; Pires, F.; Pires, J.R.A.; Rodrigues, C.; Galus, S.; Souza, V.G.L.; Alves, M.M.; Santos, C.F.; Coelhoso, I.; et al. Packaging of Fresh Poultry Meat with Innovative and Sustainable ZnO/Pectin Bionanocomposite Films—A Contribution to the Bio and Circular Economy. Coatings 2023, 13, 1208. https://doi.org/10.3390/coatings13071208
Przybyszewska A, Barbosa CH, Pires F, Pires JRA, Rodrigues C, Galus S, Souza VGL, Alves MM, Santos CF, Coelhoso I, et al. Packaging of Fresh Poultry Meat with Innovative and Sustainable ZnO/Pectin Bionanocomposite Films—A Contribution to the Bio and Circular Economy. Coatings. 2023; 13(7):1208. https://doi.org/10.3390/coatings13071208
Chicago/Turabian StylePrzybyszewska, Adrianna, Cássia H. Barbosa, Filipa Pires, João Ricardo Afonso Pires, Carolina Rodrigues, Sabina Galus, Victor Gomes Lauriano Souza, Marta M. Alves, Catarina F. Santos, Isabel Coelhoso, and et al. 2023. "Packaging of Fresh Poultry Meat with Innovative and Sustainable ZnO/Pectin Bionanocomposite Films—A Contribution to the Bio and Circular Economy" Coatings 13, no. 7: 1208. https://doi.org/10.3390/coatings13071208
APA StylePrzybyszewska, A., Barbosa, C. H., Pires, F., Pires, J. R. A., Rodrigues, C., Galus, S., Souza, V. G. L., Alves, M. M., Santos, C. F., Coelhoso, I., & Fernando, A. L. (2023). Packaging of Fresh Poultry Meat with Innovative and Sustainable ZnO/Pectin Bionanocomposite Films—A Contribution to the Bio and Circular Economy. Coatings, 13(7), 1208. https://doi.org/10.3390/coatings13071208











