Itaconic Anhydride as a Bio-Based Compatibilizer for a Tung Oil-Based Thermosetting Resin Reinforced with Sand and Algae Biomass
Abstract
1. Introduction
2. Experimental
2.1. Materials
2.2. Methods
2.2.1. Algal Biomass
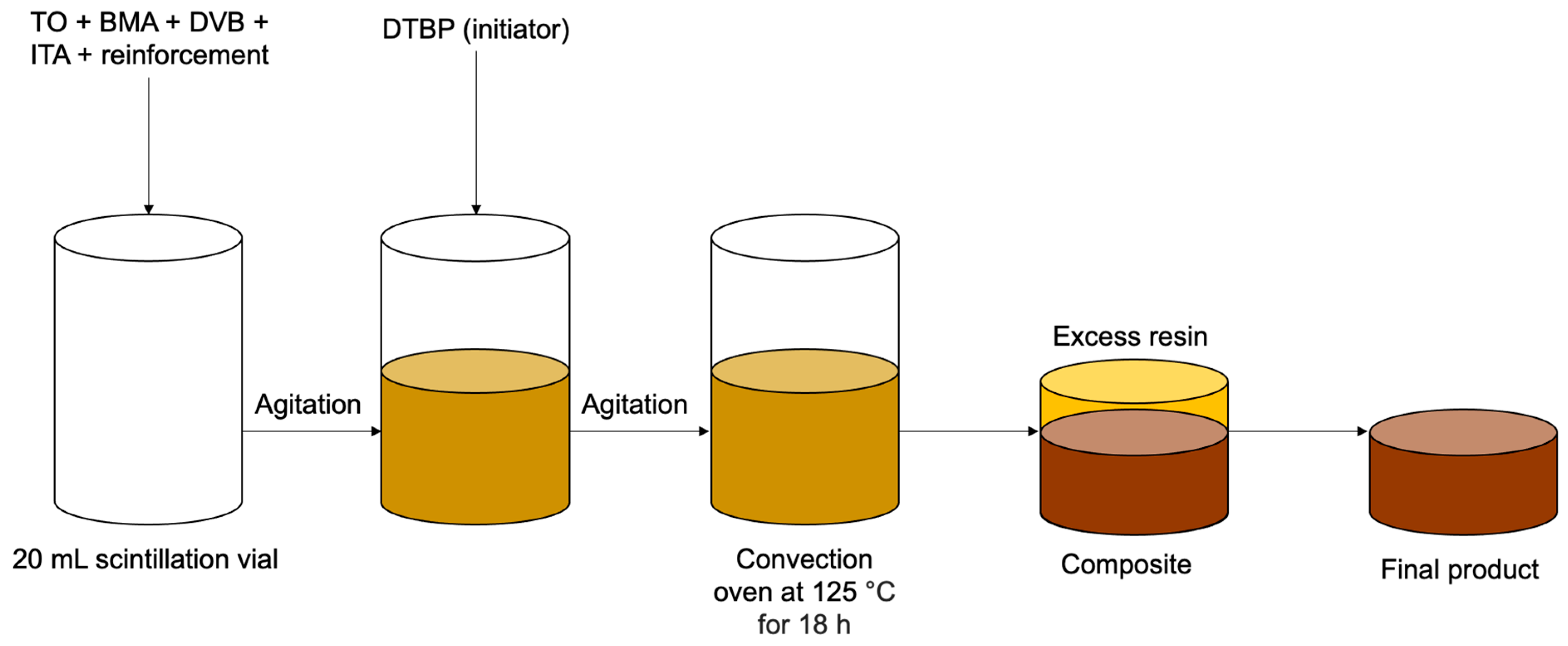
2.2.2. Preparation of Tung Oil-Based Composites
2.2.3. Characterization
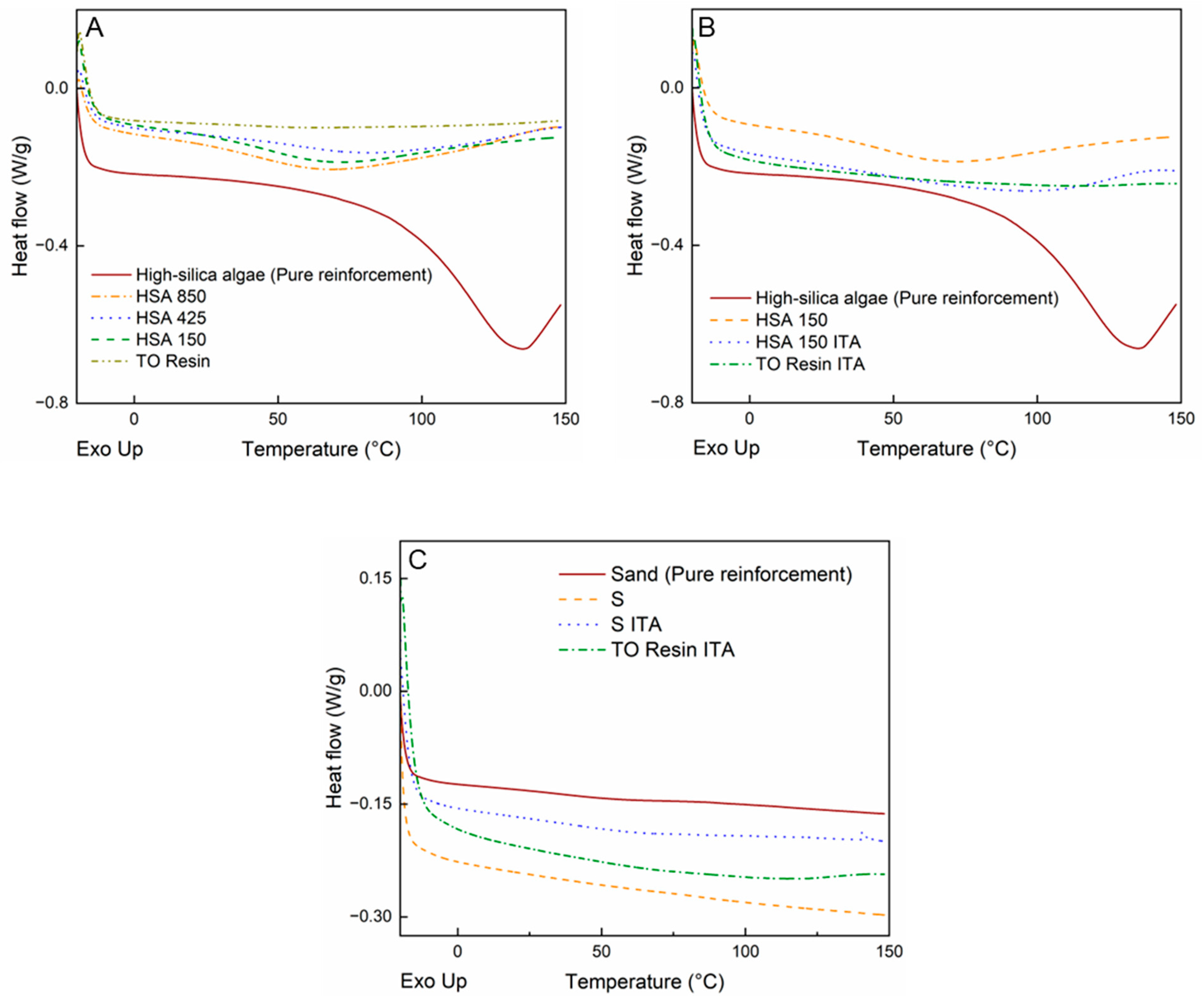
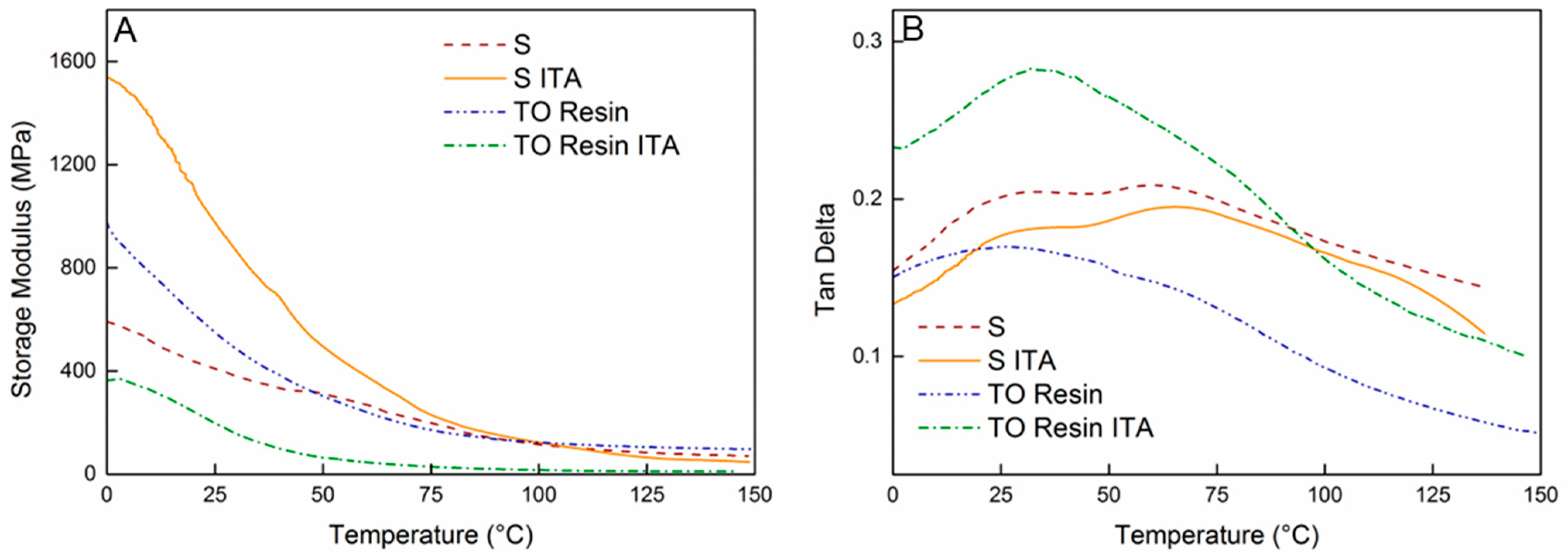
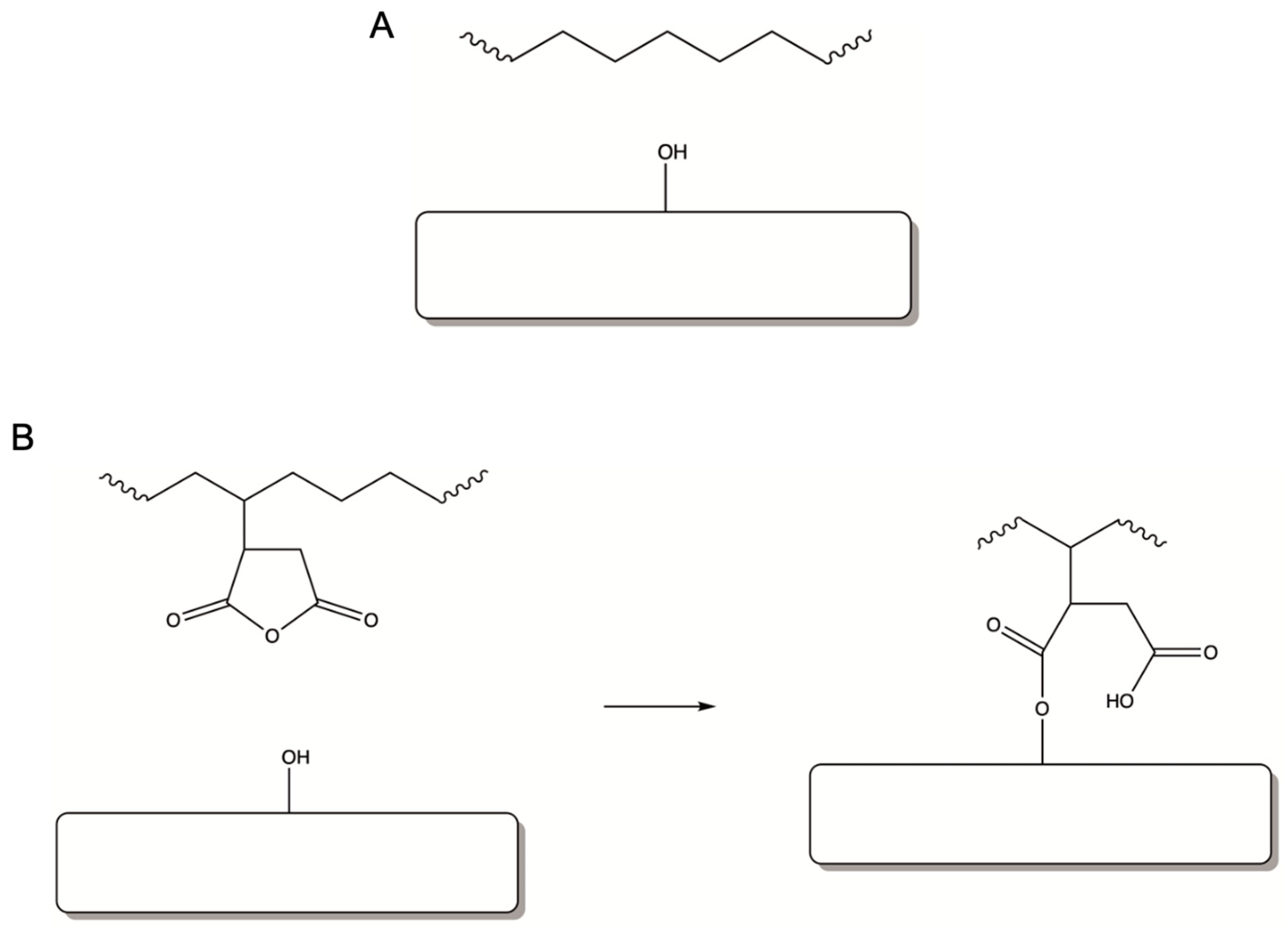
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Rosenboom, J.-G.; Langer, R.; Traverso, G. Bioplastics for a Circular Economy. Nat. Rev. Mater. 2022, 7, 117–137. [Google Scholar] [CrossRef]
- Gandini, A.; Lacerda, M.; Monomers, T. Macromolecular Materials from Renewable Resources: State of the Art and Perspectives. Molecules 2021, 27, 159. [Google Scholar] [CrossRef] [PubMed]
- Tremblay-Parrado, K.-K.; García-Astrain, C.; Avérous, L. Click Chemistry for the Synthesis of Biobased Polymers and Networks Derived from Vegetable Oils. Green Chem. 2021, 23, 4296–4327. [Google Scholar] [CrossRef]
- Silva, J.A.C.; Grilo, L.M.; Gandini, A.; Lacerda, T.M. The Prospering of Macromolecular Materials Based on Plant Oils within the Blooming Field of Polymers from Renewable Resources. Polymers 2021, 13, 1722. [Google Scholar] [CrossRef]
- Gomez, J.C.; Zakaria, R.; Aung, M.M.; Mokhtar, M.N.; Yunus, R. Synthesis and Characterization of Polyurethanes from Residual Palm Oil with High Poly-Unsaturated Fatty Acid Oils as Additive. Polymers 2021, 13, 4214. [Google Scholar] [CrossRef]
- Grauzeliene, S.; Navaruckiene, A.; Skliutas, E.; Malinauskas, M.; Serra, A.; Ostrauskaite, J. Vegetable Oil-Based Thiol-Ene/Thiol-Epoxy Resins for Laser Direct Writing 3D Micro-/Nano-Lithography. Polymers 2021, 13, 872. [Google Scholar] [CrossRef]
- Abbasi, A.; Yahya, W.Z.N.; Nasef, M.M.; Moniruzzaman, M.; Ghumman, A.S.M. Copolymerization of Palm Oil with Sulfur Using Inverse Vulcanization to Boost the Palm Oil Industry. Polym. Polym. Compos. 2021, 29, S1446–S1456. [Google Scholar] [CrossRef]
- Alarcon, R.T.; Gaglieri, C.; Lamb, K.J.; Cavalheiro, É.T.G.; North, M.; Bannach, G. A New Acrylated Monomer from Macaw Vegetable Oil That Polymerizes without External Photoinitiators. J. Polym. Res. 2021, 28, 425. [Google Scholar] [CrossRef]
- Sain, S.; Åkesson, D.; Skrifvars, M. Synthesis and Properties of Thermosets from Tung Oil and Furfuryl Methacrylate. Polymers 2020, 12, 258. [Google Scholar] [CrossRef]
- Anbinder, S.; Meiorin, C.; Macchi, C.; Mosiewicki, M.A.; Aranguren, M.I.; Somoza, A. Structural Properties of Vegetable Oil Thermosets: Effect of Crosslinkers, Modifiers and Oxidative Aging. Eur. Polym. J. 2020, 124, 109470. [Google Scholar] [CrossRef]
- Sharma, V.; Kundu, P.P. Addition Polymers from Natural Oils—A Review. Prog. Polym. Sci. 2006, 31, 983–1008. [Google Scholar] [CrossRef]
- Todorovic, A.; Blößl, Y.; Oreski, G.; Resch-Fauster, K. High-Performance Composite with 100% Bio-Based Carbon Content Produced from Epoxidized Linseed Oil, Citric Acid and Flax Fiber Reinforcement. Compos. Part A Appl. Sci. Manuf. 2022, 152, 106666. [Google Scholar] [CrossRef]
- Seabra, C.P.; Sousa, A.C.; Bragancą, I.M.F.; Silva, C.M.A.; Robalo, M.P.; Loja, M.A.R.; Martins, P.A.F. On the Performance and Recyclability of a Green Composite Based on AESO Resin. J. Manuf. Mater. Process. 2020, 4, 65. [Google Scholar] [CrossRef]
- Sain, S.; Åkesson, D.; Skrifvars, M.; Roy, S. Hydrophobic Shape-Memory Biocomposites from Tung-Oil-Based Bioresin and Onion-Skin-Derived Nanocellulose Networks. Polymers 2020, 12, 2470. [Google Scholar] [CrossRef]
- Balanuca, B.; Komartin, R.S.; Necolau, M.I.; Damian, C.M.; Stan, R. Investigating the Synthesis and Characteristics of UV-Cured Bio-Based Epoxy Vegetable Oil-Lignin Composites Mediated by Structure-Directing Agents. Polymers 2023, 15, 439. [Google Scholar] [CrossRef]
- Biswas, E.; Silva, J.A.C.; Khan, M.; Quirino, R.L. Synthesis and Properties of Bio-Based Composites from Vegetable Oils and Starch. Coatings 2022, 12, 1119. [Google Scholar] [CrossRef]
- Gogoi, G.; Chowdhury, C.; Maji, T.K. Effect of Nanoclay on the Properties of Rosin Derivative Cross-Linked Green Composite Based on Chicken Feather Fiber and Modified Vegetable Oil. Polym. Eng. Sci. 2021, 61, 288–300. [Google Scholar] [CrossRef]
- Echeverri, D.A.; Inciarte, H.C.; Gómez, C.L.; Rios, L.A. Development of Glass Fiber/Unsaturated Polyester-like Resins Based on Modified Castor Oil. Iran. Polym. J. Engl. Ed. 2022, 31, 595–604. [Google Scholar] [CrossRef]
- Tang, Q.; Li, Q.; Pan, X.; Xi, Z.; Zhao, L. Poly(Acrylated Epoxidized Soybean Oil)-Modified Carbon Nanotubes and Their Application in Epoxidized Soybean Oil-Based Thermoset Composites. Polym. Compos. 2021, 42, 5774–5788. [Google Scholar] [CrossRef]
- Smith, M.; Payne, A.; Edwards, K.; Morris, S.; Beckler, B.; Quirino, R.L. Effect of Microwave Cure on the Thermo-Mechanical Properties of Tung Oil-Based/Carbon Nanotube Composites. Coatings 2015, 5, 557–575. [Google Scholar] [CrossRef]
- Xu, Y.; Dai, S.; Zhang, H.; Bi, L.; Jiang, J.; Chen, Y. Reprocessable, Self-Adhesive, and Recyclable Carbon Fiber-Reinforced Composites Using a Catalyst-Free Self-Healing Bio-Based Vitrimer Matrix. ACS Sustain. Chem. Eng. 2021, 9, 16281–16290. [Google Scholar] [CrossRef]
- Chen, B.; Liao, M.; Sun, J.; Shi, S. A Novel Biomass Polyurethane-Based Composite Coating with Superior Radiative Cooling, Anti-Corrosion and Recyclability for Surface Protection. Prog. Org. Coat. 2023, 174, 107250. [Google Scholar] [CrossRef]
- Kim, K.M.; Kim, H.; Kim, H.J. Enhancing Thermo-Mechanical Properties of Epoxy Composites Using Fumed Silica with Different Surface Treatment. Polymers 2021, 13, 2691. [Google Scholar] [CrossRef]
- Chen, W.; Wu, Z.; Xie, Y.; He, X.; Su, Y.; Qin, Y.; Tang, D.; Oh, S.K. Fabrication of Silane and Nano-Silica Composite Modified Bio-Based WPU and Its Interfacial Bonding Mechanism with Cementitious Materials. Constr. Build. Mater. 2023, 371, 130819. [Google Scholar] [CrossRef]
- Alameri, I.; Oltulu, M. Mechanical Properties of Polymer Composites Reinforced by Silica-Based Materials of Various Sizes. Appl. Nanosci. 2020, 10, 4087–4102. [Google Scholar] [CrossRef]
- Nartop, D.; Kazak, Ç. Synthesis and Characterization of Novel Polystyrene-Silica Composites Containing Azomethine. J. Mol. Struct. 2021, 1227, 129705. [Google Scholar] [CrossRef]
- Slieptsova, I.; Savchenko, B.; Sova, N.; Slieptsov, A. Polymer Sand Composites Based on the Mixed and Heavily Contaminated Thermoplastic Waste. IOP Conf. Ser. Mater. Sci. Eng. 2016, 111, 012027. [Google Scholar] [CrossRef]
- Zahran, R.R. Effect of Sand Addition on the Tensile Properties of Compression Moulded Sand/Polyethylene Composite System. Mater. Lett. 1998, 34, 161–167. [Google Scholar] [CrossRef]
- Aston, J.E.; Wahlen, B.D.; Davis, R.W.; Siccardi, A.J.; Wendt, L.M. Application of Aqueous Alkaline Extraction to Remove Ash from Algae Harvested from an Algal Turf Scrubber. Algal Res. 2018, 35, 370–377. [Google Scholar] [CrossRef]
- Kartik, A.; Akhil, D.; Lakshmi, D.; Panchamoorthy Gopinath, K.; Arun, J.; Sivaramakrishnan, R.; Pugazhendhi, A. A Critical Review on Production of Biopolymers from Algae Biomass and Their Applications. Bioresour. Technol. 2021, 329, 124868. [Google Scholar] [CrossRef]
- Bohre, A.; Modak, A.; Chourasia, V.; Ram Jadhao, P.; Sharma, K.; Kishore Pant, K. Recent Advances in Supported Ionic Liquid Catalysts for Sustainable Biomass Valorisation to High-Value Chemicals and Fuels. Chem. Eng. J. 2022, 450, 138032. [Google Scholar] [CrossRef]
- Di Mauro, C.; Genua, A.; Mija, A. Fully Bio-Based Reprocessable Thermosetting Resins Based on Epoxidized Vegetable Oils Cured with Itaconic Acid. Ind. Crops Prod. 2022, 185, 115116. [Google Scholar] [CrossRef]
- Murawski, A.; Quirino, R.L. Bio-Based Composites Withnhanced Matrix-Reinforcement Interactionsrom the Polymerizationf α-Eleostearic Acid. Coatings 2019, 9, 447. [Google Scholar] [CrossRef]
- Johns, A.; Morris, S.; Edwards, K.; Quirino, R.L. Asolectin from Soybeans as a Natural Compatibilizer for Cellulose-Reinforced Biocomposites from Tung Oil. J. Appl. Polym. Sci. 2015, 132, 41833. [Google Scholar] [CrossRef]
- Quirino, R.L.; Larock, R.C. Rice Hull Biocomposites, Part 2: Effect of the Resin Composition on the Properties of the Composite. J. Appl. Polym. Sci. 2011, 121, 2050–2059. [Google Scholar] [CrossRef]
- Conti Silva, J.A.; Walton, H.; Dever, S.; Kardel, K.; Martins Lacerda, T.; Lopes Quirino, R. Itaconic Anhydride as a Green Compatibilizer in Composites Prepared by the Reinforcement of a Tung Oil-Based Thermosetting Resin with Miscanthus, Pine Wood, or Algae Biomass. Coatings 2023, 13, 25. [Google Scholar] [CrossRef]
- Xiao, R.; Yang, W.; Cong, X.; Dong, K.; Xu, J.; Wang, D.; Yang, X. Thermogravimetric Analysis and Reaction Kinetics of Lignocellulosic Biomass Pyrolysis. Energy 2020, 201, 117537. [Google Scholar] [CrossRef]
- Blivi, A.S.; Benhui, F.; Bai, J.; Kondo, D.; Bédoui, F. Experimental Evidence of Size Effect in Nano-Reinforced Polymers: Case of Silica Reinforced PMMA. Polym. Test. 2016, 56, 337–343. [Google Scholar] [CrossRef]
- Silverstein, R.M.; Webster, F.X. Spectrometric Identification of Organic Compounds, 6th ed.; John Wiley & Sons: New York, NY, USA, 1998. [Google Scholar]




| Reinforcement | Sample Name | Reinforcement Particle Size | ITA wt.% |
|---|---|---|---|
| - a | TO Resin | - | - |
| - a | TO Resin ITA | - | 10 |
| Sand | S | <500 μm | - |
| Sand | S ITA | <500 μm | 10 |
| High-silica algae | HSA 850 | 425–850 μm | - |
| High-silica algae | HSA 425 | 150–425 μm | - |
| High-silica algae | HSA 150 | <150 μm | - |
| High-silica algae | HSA 150 ITA | <150 μm | 10 |
| Sample | E′ at 25 °C (MPa) | Tg (°C) | E′ at Tg + 50 °C (MPa) |
|---|---|---|---|
| TO Resin [36] | 518 ± 108 | 26 ± 6 | 146 ± 52 |
| TO Resin ITA [36] | 232 ± 118 | 32 ± 5 | 26 ± 16 |
| S a | 479 ± 70 | 67 ± 1 | 134 ± 59 |
| S ITA | 922 ± 67 | 60 ± 6 | 219 ± 187 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Silva, J.A.C.; Dever, S.; Siccardi, A.; Snelling, D.; Al Qabani, I.; Thompson, S.; Goldberg, K.; Baudoin, G.; Martins Lacerda, T.; Quirino, R.L. Itaconic Anhydride as a Bio-Based Compatibilizer for a Tung Oil-Based Thermosetting Resin Reinforced with Sand and Algae Biomass. Coatings 2023, 13, 1188. https://doi.org/10.3390/coatings13071188
Silva JAC, Dever S, Siccardi A, Snelling D, Al Qabani I, Thompson S, Goldberg K, Baudoin G, Martins Lacerda T, Quirino RL. Itaconic Anhydride as a Bio-Based Compatibilizer for a Tung Oil-Based Thermosetting Resin Reinforced with Sand and Algae Biomass. Coatings. 2023; 13(7):1188. https://doi.org/10.3390/coatings13071188
Chicago/Turabian StyleSilva, Julio Antonio Conti, Seth Dever, Anthony Siccardi, Drew Snelling, Ibrahim Al Qabani, Scott Thompson, Karin Goldberg, Genevieve Baudoin, Talita Martins Lacerda, and Rafael Lopes Quirino. 2023. "Itaconic Anhydride as a Bio-Based Compatibilizer for a Tung Oil-Based Thermosetting Resin Reinforced with Sand and Algae Biomass" Coatings 13, no. 7: 1188. https://doi.org/10.3390/coatings13071188
APA StyleSilva, J. A. C., Dever, S., Siccardi, A., Snelling, D., Al Qabani, I., Thompson, S., Goldberg, K., Baudoin, G., Martins Lacerda, T., & Quirino, R. L. (2023). Itaconic Anhydride as a Bio-Based Compatibilizer for a Tung Oil-Based Thermosetting Resin Reinforced with Sand and Algae Biomass. Coatings, 13(7), 1188. https://doi.org/10.3390/coatings13071188








