Sensitive Fingerprint Detection Using Biocompatible Mesoporous Silica Nanoparticle Coating on Non-Porous Surfaces
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Sample Collection
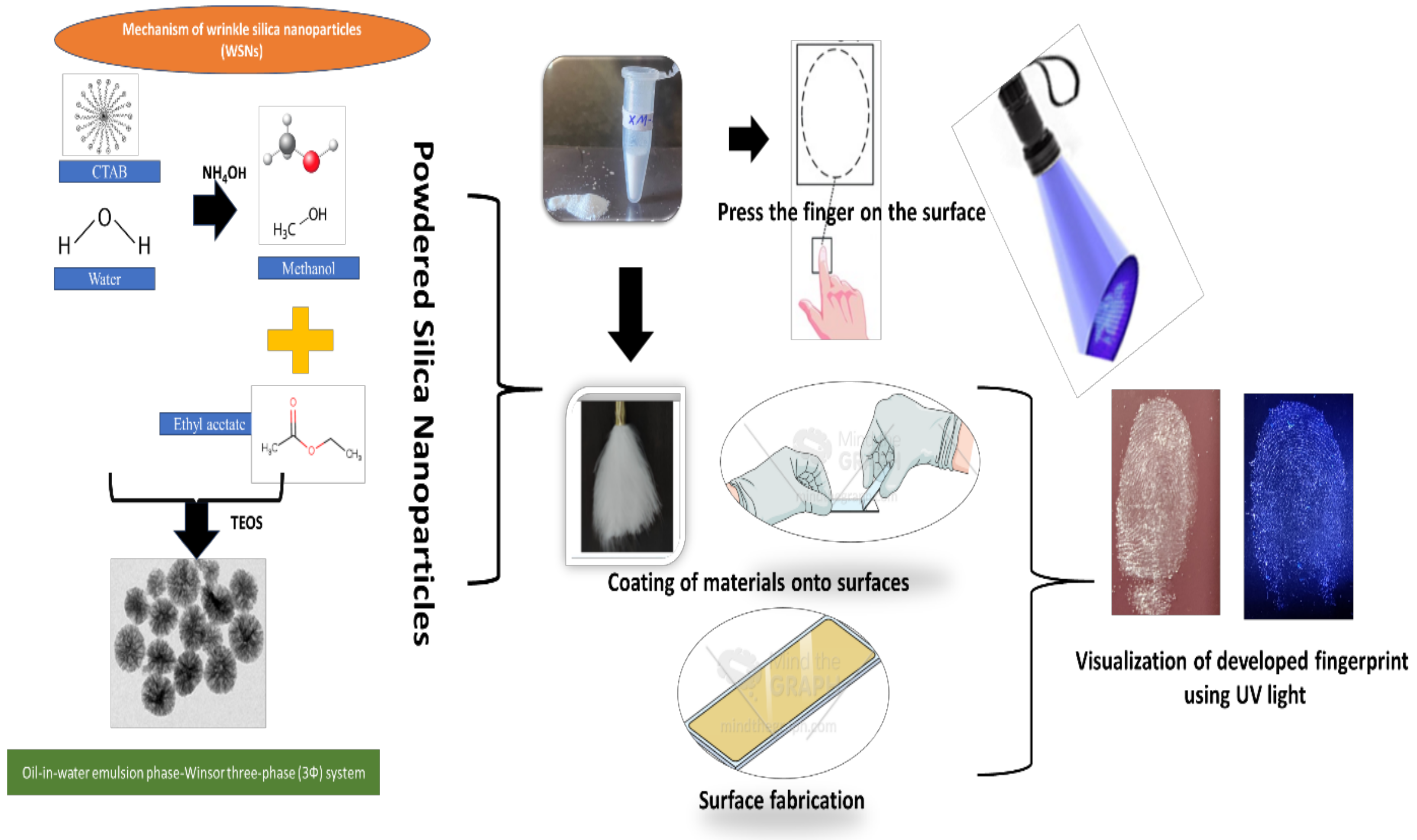
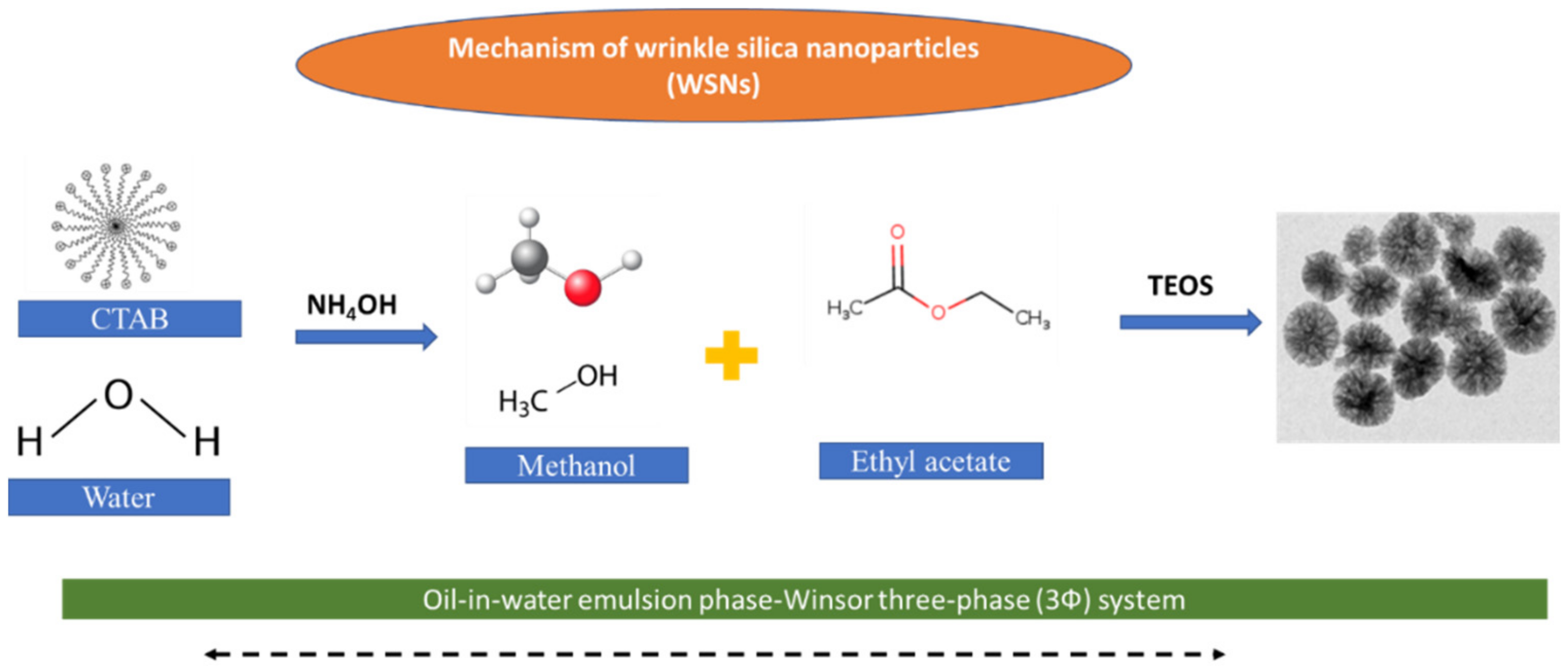
2.3. Synthesis of Mesoporous Silica Nanoparticles (MSNPs)
2.4. Characterization of Synthesized Nanoparticles
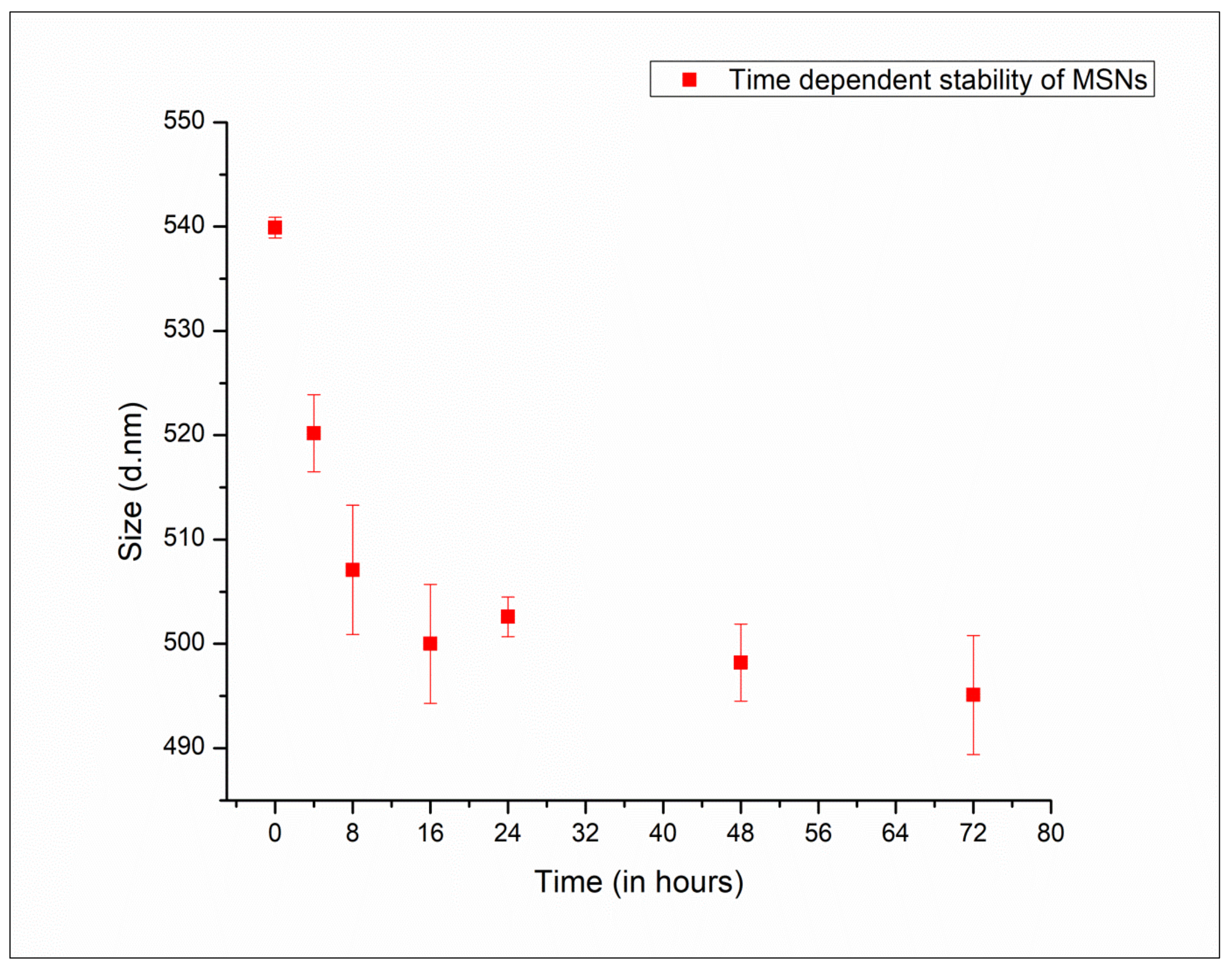
2.5. Assessment of Post-Synthesis Stability of MSNPs
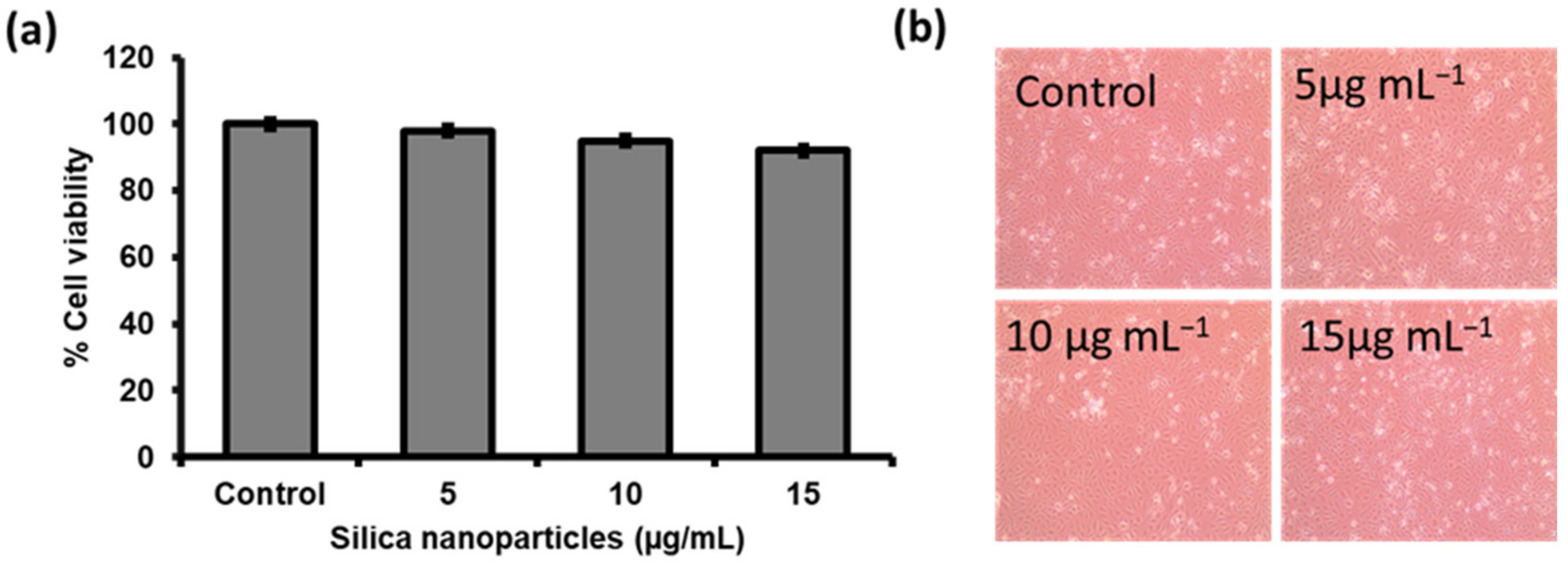
2.6. Cytotoxicity of the Synthesized MSNPs via Cell Viability Analysis
Cell Lines and Culturing
2.7. Application of MSNPs for Detection of Fingerprints
Detection Procedure for Fingerprints
3. Results
3.1. Synthesis and Characterization of the Synthesized MSNPs
3.2. Stability Studies of MSNPs via Transformative Changes in Particles during Storage
3.3. Development of Quiescent Finger Impressions Using MSNPs
3.4. Development of Latent Fingerprints by Using the MSNPs
3.5. Biocompatibility/Toxicity Measurements
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Crispino, F.; Houck, M.M. Principles of Forensic Science; Elsevier Ltd.: Amsterdam, The Netherlands, 2013. [Google Scholar]
- Kücken, M.; Newell, A.C. Fingerprint Formation. J. Theor. Biol. 2005, 235, 71–83. [Google Scholar] [CrossRef] [PubMed]
- Bhati, K. Role of Nanoparticles in Latent Fingerprinting: An Update. Lett. Appl. NanoBioSci. 2020, 9, 1427–1443. [Google Scholar] [CrossRef]
- Kim, B.-I.; AfsaráUddin, M.; YoungáWoo, H. Others Surfactant Chemistry for Fluorescence Imaging of Latent Fingerprints using Conjugated Polyelectrolyte Nanoparticles. Chem. Commun. 2015, 51, 13634–13637. [Google Scholar]
- Budrikis, Z. Getting a Grip on Fingerprints. Nat. Rev. Phys. 2021, 3, 5. [Google Scholar] [CrossRef]
- Gupta, A. Evaluation of Correlation between Dental Caries and Fingerprints in Known Subjects. J. Adv. Med. Dent. Sci. Res. 2019, 7, 236–239. [Google Scholar]
- Wang, M.; Li, M.; Yu, A.; Zhu, Y.; Yang, M.; Mao, C. Fluorescent Nanomaterials for the Development of Latent Fingerprints in Forensic Sciences. Adv. Funct. Mater. 2017, 27, 1606243. [Google Scholar] [CrossRef]
- Gao, F.; Lv, C.; Han, J.; Li, X.; Wang, Q.; Zhang, J.; Chen, C.; Li, Q.; Sun, X.; Zheng, J.; et al. CdTe-Montmorillonite Nanocomposites: Control Synthesis, UV Radiation-Dependent Photoluminescence, and Enhanced Latent Fingerprint Detection. J. Phys. Chem. C 2011, 115, 21574–21583. [Google Scholar] [CrossRef]
- Lee, J.; Kim, J.; Heo, I.; Kim, S.J.; Jang, S.; Lee, H.-Y.; Jang, K.-S.; Yang, C.-S.; Lee, Y.; Yoo, W.C.; et al. One-Pot Bifunctionalization of Silica Nanoparticles Conjugated with Bioorthogonal Linkers: Application in Dual-Modal Imaging. Biomater. Sci. 2022, 10, 3540–3546. [Google Scholar] [CrossRef]
- Çetli, E.; Özkoçak, V.; Tatar, D. The Role of Silica Nanoparticle in Fingerprint Visualization Studies. Aksaray Univ. J. Sci. Eng. 2022, 6, 27–41. [Google Scholar] [CrossRef]
- Rajan, R.; Zakaria, Y.; Shamsuddin, S.; Nik Hassan, N.F. Fluorescent Variant of Silica Nanoparticle Powder Synthesised from Rice Husk for Latent Fingerprint Development. Egypt. J. Forensic Sci. 2019, 9, 50. [Google Scholar] [CrossRef]
- Bhati, K.; Tripathy, D.; Chhabra, P. Role of Fluorescent Substances in Development of Latent Fingerprints: A Review. Indian J. Forensic Med. Pathol. 2021, 14, 351–358. [Google Scholar] [CrossRef]
- Lee, P.L.T.; Kanodarwala, F.K.; Lennard, C.; Spindler, X.; Spikmans, V.; Roux, C.; Moret, S. Latent Fingermark Detection Using Functionalised Silicon Oxide Nanoparticles: Investigation into Novel Application Procedures. Forensic Sci. Int. 2022, 335, 111275. [Google Scholar] [CrossRef] [PubMed]
- Leśniewski, A. Hybrid Organic–Inorganic Silica Based Particles for Latent Fingermarks Development: A Review. Synth. Met. 2016, 222, 124–131. [Google Scholar] [CrossRef]
- Kim, Y.J.; Jung, H.S.; Lim, J.; Ryu, S.J.; Lee, J.K. Rapid Imaging of Latent Fingerprints Using Biocompatible Fluorescent Silica Nanoparticles. Langmuir 2016, 32, 8077–8083. [Google Scholar] [CrossRef] [PubMed]
- Sawhney, S.; Bhati, K.; Chhabra, P.; Tripathy, D. Role of Nanotechnology in Techniques in Fingerprints Enhancement. Indian J. Forensic Med. Pathol. 2021, 14, 288–294. [Google Scholar] [CrossRef]
- Zhang, M.; Ou, Y.; Du, X.; Li, X.; Huang, H.; Wen, Y.; Zhang, X. Systematic Study of Dye Loaded Small Mesoporous Silica Nanoparticles for Detecting Latent Fingerprints on Various Substrates. J. Porous Mater. 2017, 24, 13–20. [Google Scholar] [CrossRef]
- Rajan, R.; Zakaria, Y.; Shamsuddin, S.; Nik Hassan, N.F. Robust Synthesis of Mono-Dispersed Spherical Silica Nanoparticle from Rice Husk for High-Definition Latent Fingermark Development. Arab. J. Chem. 2020, 13, 8119–8132. [Google Scholar] [CrossRef]
- Wang, M.; Li, M.; Yu, A.; Wu, J.; Mao, C. Rare Earth Fluorescent Nanomaterials for Enhanced Development of Latent Fingerprints. ACS Appl. Mater. Interfaces 2015, 7, 28110–28115. [Google Scholar] [CrossRef]
- Yang, Y.; Liu, R.; Cui, Q.; Xu, W.; Peng, R.; Wang, J.; Li, L. Red-Emissive Conjugated Oligomer/Silica Hybrid Nanoparticles with High Affinity and Application for Latent Fingerprint Detection. Colloids Surf. A Physicochem. Eng. Asp. 2019, 565, 118–130. [Google Scholar] [CrossRef]
- Kwon, D.; Cha, B.G.; Cho, Y.; Min, J.; Park, E.-B.; Kang, S.-J.; Kim, J. Extra-Large Pore Mesoporous Silica Nanoparticles for Directing in Vivo M2 Macrophage Polarization by Delivering IL-4. Nano Lett. 2017, 17, 2747–2756. [Google Scholar] [CrossRef]
- Kim, J.; Lee, J.E.; Lee, J.; Yu, J.H.; Kim, B.C.; An, K.; Hwang, Y.; Shin, C.-H.; Park, J.-G.; Kim, J.; et al. Magnetic Fluorescent Delivery Vehicle Using Uniform Mesoporous Silica Spheres Embedded with Monodisperse Magnetic and Semiconductor Nanocrystals. J. Am. Chem. Soc. 2006, 128, 688–689. [Google Scholar] [CrossRef] [PubMed]
- Cha, B.G.; Jeong, J.H.; Kim, J. Extra-Large Pore Mesoporous Silica Nanoparticles Enabling Co-Delivery of High Amounts of Protein Antigen and Toll-like Receptor 9 Agonist for Enhanced Cancer Vaccine Efficacy. ACS Cent. Sci. 2018, 4, 484–492. [Google Scholar] [CrossRef] [PubMed]
- Sevimli, F.; Yilmaz, A. Surface Functionalization of SBA-15 Particles for Amoxicillin Delivery. Microporous Mesoporous Mater. 2012, 158, 281–291. [Google Scholar] [CrossRef]
- Liu, F.; Wang, J.; Huang, P.; Zhang, Q.; Deng, J.; Cao, Q.; Jia, J.; Cheng, J.; Fang, Y.; Deng, D.Y.B.; et al. Outside-in Stepwise Functionalization of Mesoporous Silica Nanocarriers for Matrix Type Sustained Release of Fluoroquinolone Drugs. J. Mater. Chem. B 2015, 3, 2206–2214. [Google Scholar] [CrossRef] [PubMed]
- Wardhani, G.A.P.K.; Nurlela, N.; Azizah, M. Silica Content and Structure from Corncob Ash with Various Acid Treatment (HCl, HBr, and Citric Acid). Molekul 2017, 12, 174–181. [Google Scholar] [CrossRef]
- Thuadaij, N.; Nuntiya, A. Preparation of Nanosilica Powder from Rice Husk Ash by Precipitation Method. Chiang Mai J. Sci. 2008, 35, 206–211. [Google Scholar]
- Bogeshwaran, K.; Kalaivani, R.; Ashraf, S.; Manikandan, G.N.; Prabhu, G.E. Production of Silica from Rice Husk. Int. J. ChemTech Res. 2014, 6, 4337–4345. [Google Scholar]
- Sargazi, S.; Laraib, U.; Barani, M.; Rahdar, A.; Fatima, I.; Bilal, M.; Pandey, S.; Sharma, R.K.; Kyzas, G.Z. Recent Trends in Mesoporous Silica Nanoparticles of Rode-like Morphology for Cancer Theranostics: A review. J. Mol. Struct. 2022, 1261, 132922. [Google Scholar] [CrossRef]
- Liu, X.L.; Liu, J.; Yang, H.M.; Huang, B.; Zeng, G.H. Design of a High-performance Graphene/SiO2-Ag Periodic Grating/MoS2 Surface Plasmon Resonance Sensor. Appl. Opt. 2022, 61, 6752–6760. [Google Scholar] [CrossRef]
- Huang, W.; Li, X.; Wang, H.; Xu, X.; Liu, H.; Wang, G. Synthesis of Amphiphilic Silica Nanoparticles for Latent Fingerprint Detection. Anal. Lett. 2015, 48, 1524–1535. [Google Scholar] [CrossRef]
- Roostaee, M.; Beitollahi, H.; Sheikhshoaie, I. Simultaneous Determination of Dopamine and Uric Acid in Real Samples Using a Voltammetric Nanosensor Based on Co-MOF, Graphene Oxide, and 1-Methyl-3-butylimidazolium Bromide. Micromachines 2022, 13, 1834. [Google Scholar] [CrossRef] [PubMed]
- Verma, R.K.; Nagar, V.; Aseri, V.; Mavry, B.; Pandit, P.P.; Chopade, R.L.; Singh, A.; Singh, A.; Yadav, V.L.; Pandey, K.; et al. Zinc oxide (ZnO) nanoparticles: Synthesis Properties and their Forensic Applications in Latent Fingerprints Development. Mater. Today Proceed. 2022, 69, 36–41. [Google Scholar] [CrossRef]
- Choi, M.J.; McDonagh, A.M.; Maynard, P.J.; Wuhrer, R. Preparation and evaluation of metal nanopowders for the detection of fingermarks on nonporous surfaces. J. Forensic Ident. 2006, 56, 756–768. [Google Scholar]
- Richardson, M. Comparison of the Effectiveness of Different Commercial Titanium Dioxides (TiO2) for White Powder Suspension on Rubber-based Adhesive Tapes. In Home Office Internal Report; HOSDB: St Albans, UK, 2006. [Google Scholar]


| SN | Nanoparticles | Surface | Material | Level-I (Pattern) | Level-II (Ridge Characteristics) |
|---|---|---|---|---|---|
| 1. | MSNPs | NON-POROUS | Silica phone cover | Visible | Visible |
| 2. | MSNPs | NON-POROUS | Plastic phone case | Visible | Visible |
| 3. | MSNPs | NON-POROUS | Glass slide | Visible | Visible |
| 4. | MSNPs | NON-POROUS | Stainless steel | Visible | Visible |
| 5. | MSNPs | NON-POROUS | Plastic calculator cover | Visible | Visible |
| 6. | MSNPs | NON-POROUS | Transparent poly bag | Not-Visible | Not-Visible |
| 7. | MSNPs | POROUS | Black paper | Not-Visible | Not-Visible |
| 8. | MSNPs | POROUS | Paper bag | Not-Visible | Not-Visible |
| SN | Surface Material | Stability/Shelf Life |
|---|---|---|
| 1 | Silica phone cover | <30 days |
| 2 | Plastic phone case | <15 days |
| 3 | Glass slide | <30 days |
| 4 | Stainless steel | <15 days |
| 5 | Plastic calculator cover | <20 days |
| 6 | Transparent poly bag | <30 days |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bhati, K.; Bajpai Tripathy, D.; Kumaravel, V.; Sudhani, H.P.K.; Ali, S.; Choudhary, R.; Shukla, S. Sensitive Fingerprint Detection Using Biocompatible Mesoporous Silica Nanoparticle Coating on Non-Porous Surfaces. Coatings 2023, 13, 268. https://doi.org/10.3390/coatings13020268
Bhati K, Bajpai Tripathy D, Kumaravel V, Sudhani HPK, Ali S, Choudhary R, Shukla S. Sensitive Fingerprint Detection Using Biocompatible Mesoporous Silica Nanoparticle Coating on Non-Porous Surfaces. Coatings. 2023; 13(2):268. https://doi.org/10.3390/coatings13020268
Chicago/Turabian StyleBhati, Kajol, Divya Bajpai Tripathy, Vignesh Kumaravel, Hemanth P. K. Sudhani, Sajad Ali, Rita Choudhary, and Shruti Shukla. 2023. "Sensitive Fingerprint Detection Using Biocompatible Mesoporous Silica Nanoparticle Coating on Non-Porous Surfaces" Coatings 13, no. 2: 268. https://doi.org/10.3390/coatings13020268
APA StyleBhati, K., Bajpai Tripathy, D., Kumaravel, V., Sudhani, H. P. K., Ali, S., Choudhary, R., & Shukla, S. (2023). Sensitive Fingerprint Detection Using Biocompatible Mesoporous Silica Nanoparticle Coating on Non-Porous Surfaces. Coatings, 13(2), 268. https://doi.org/10.3390/coatings13020268







