Construction of Flower-like FeCo2O4 Nanosheets on Ni Foam as Efficient Electrocatalyst for Oxygen Evolution Reaction
Abstract
:1. Introduction
2. Materials and Methods
2.1. Synthesis of FeCo2O4 /NF Composite
2.2. Characterization
2.3. Electrochemical Measurements
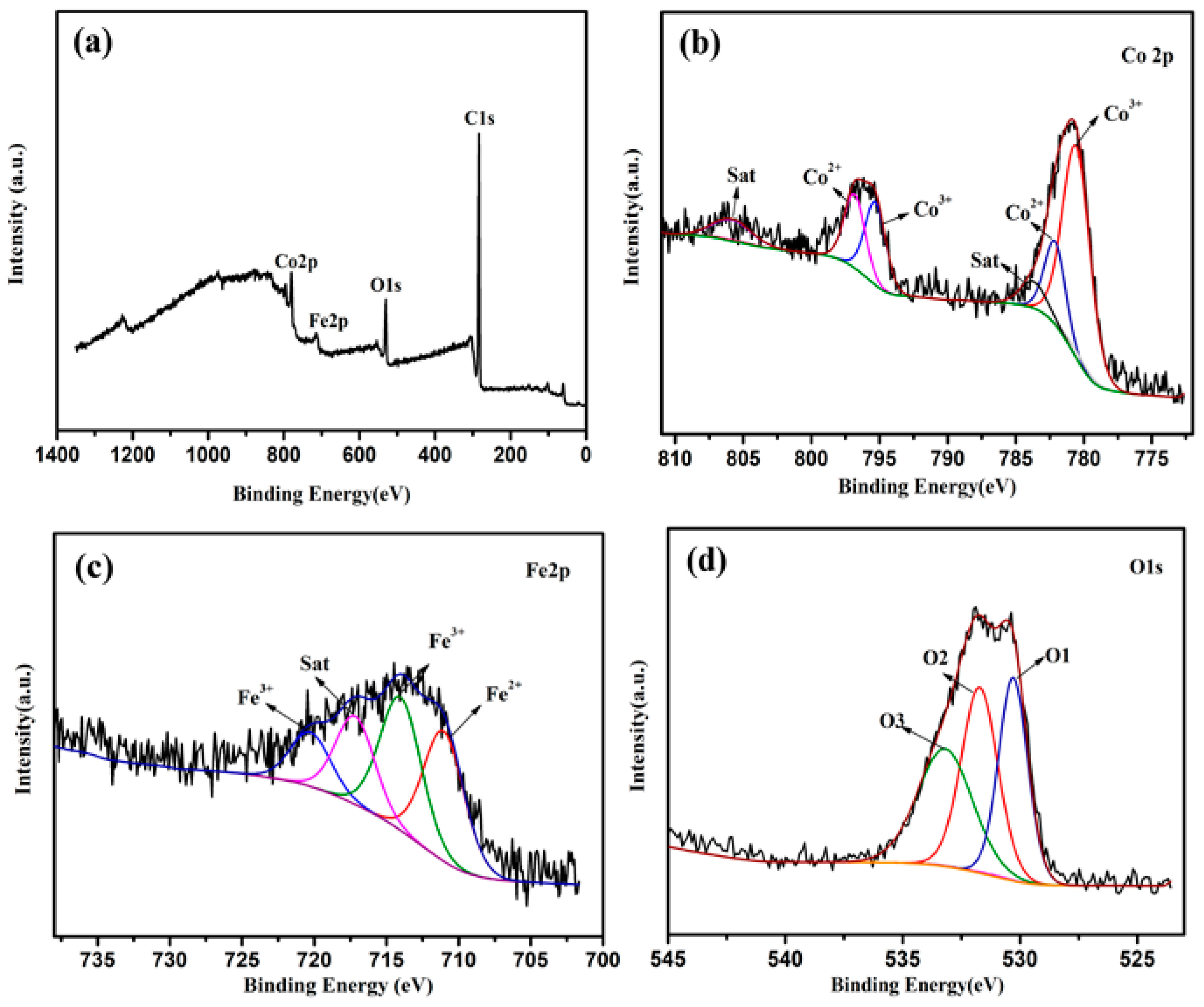
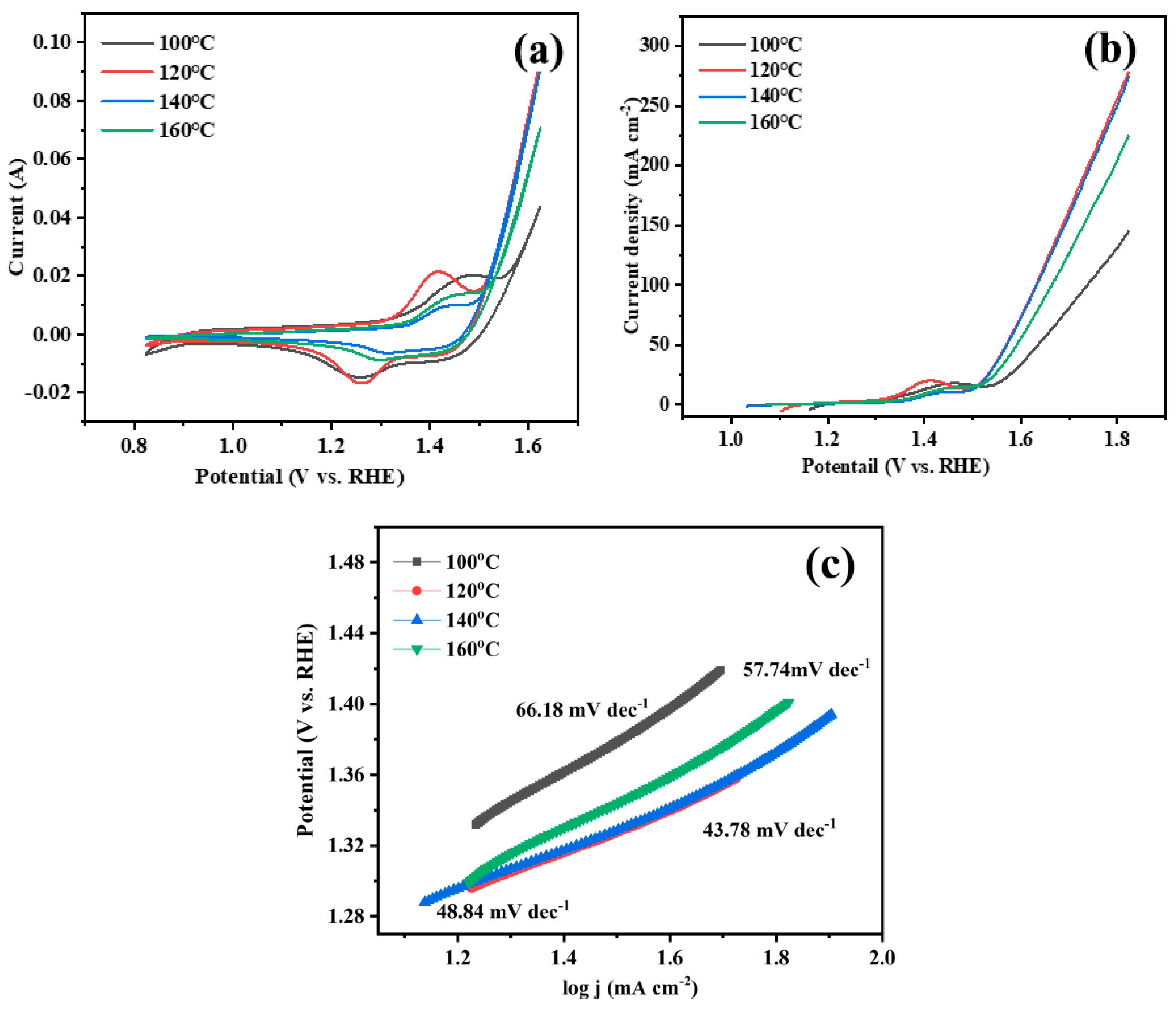
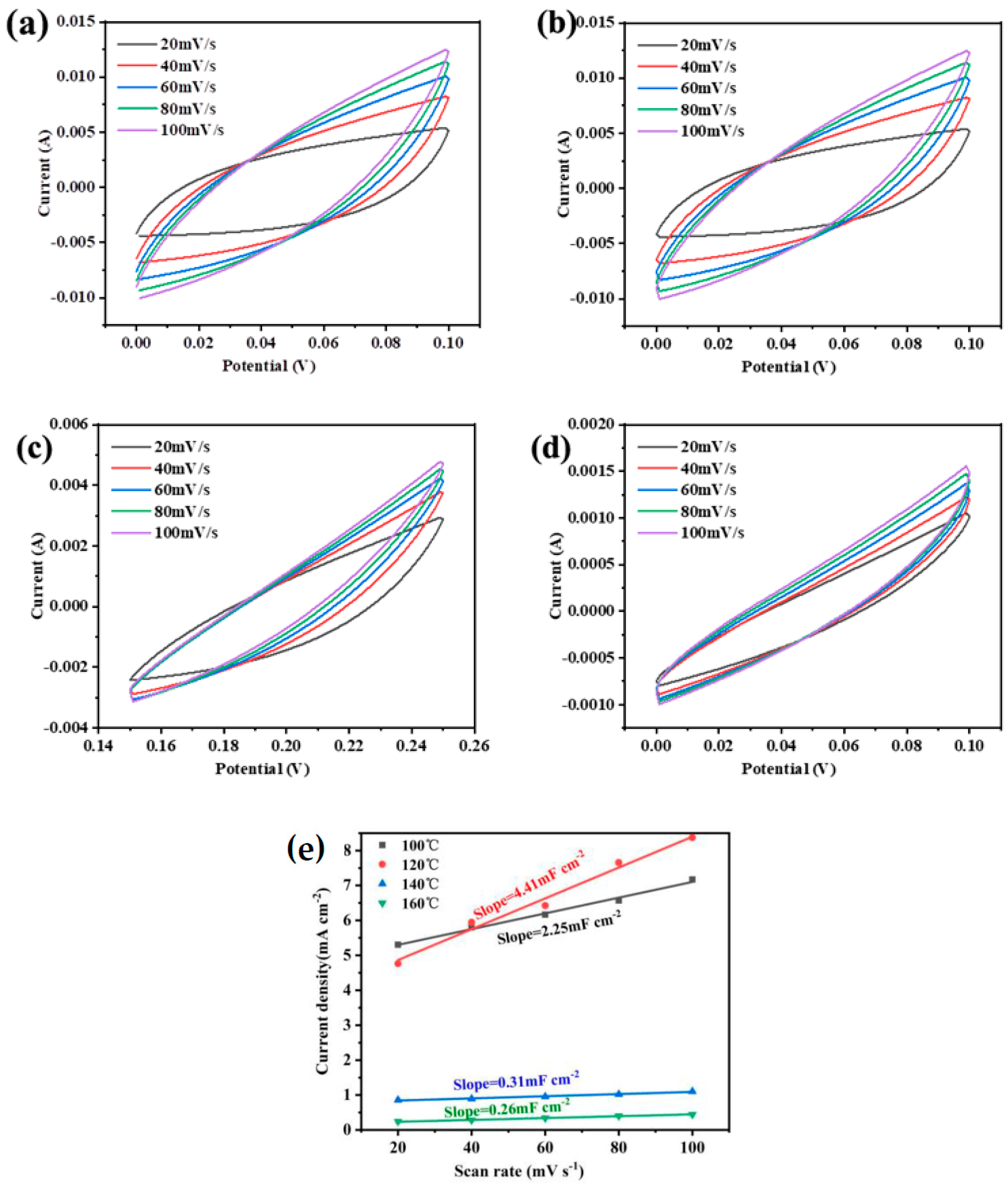
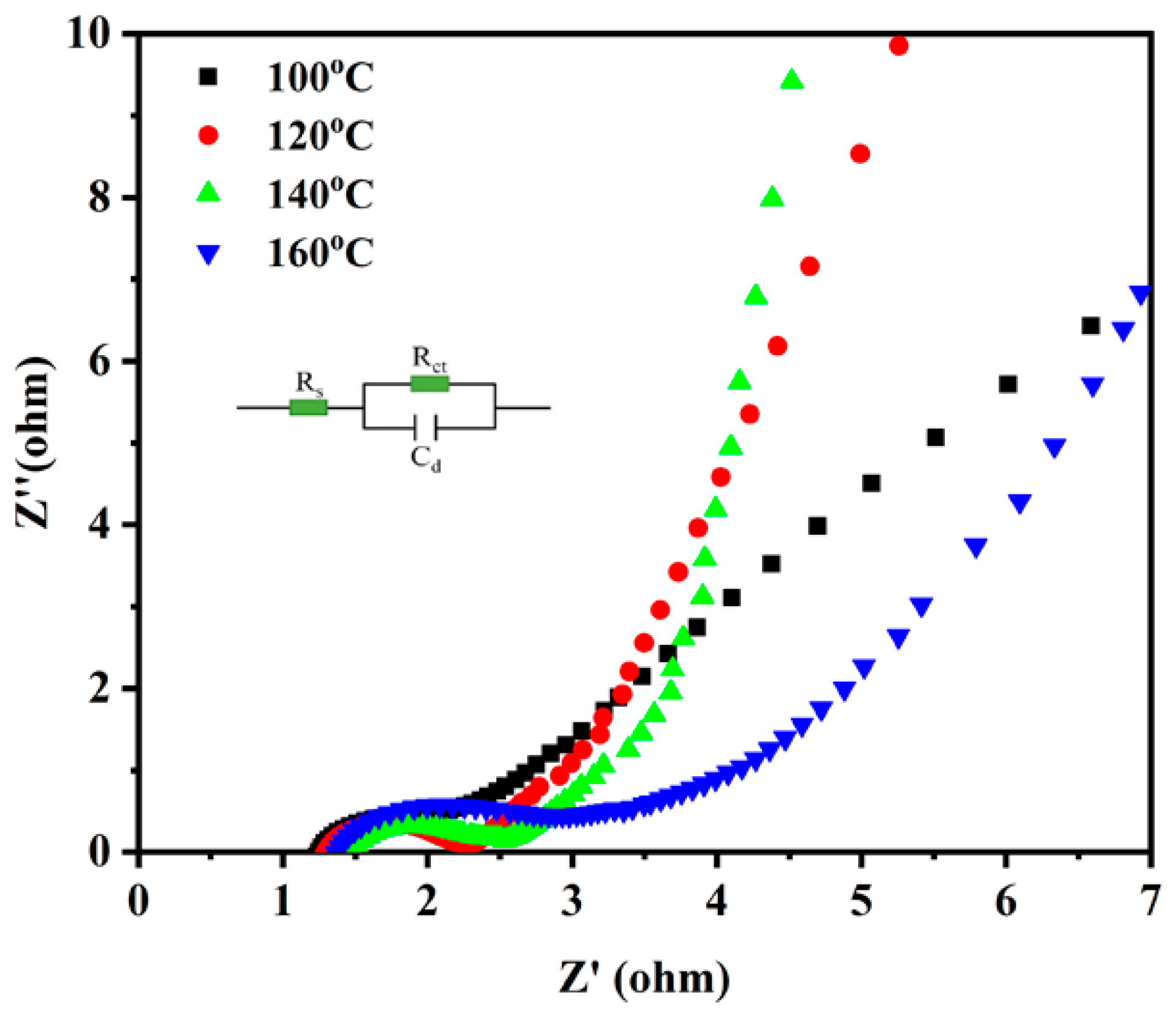
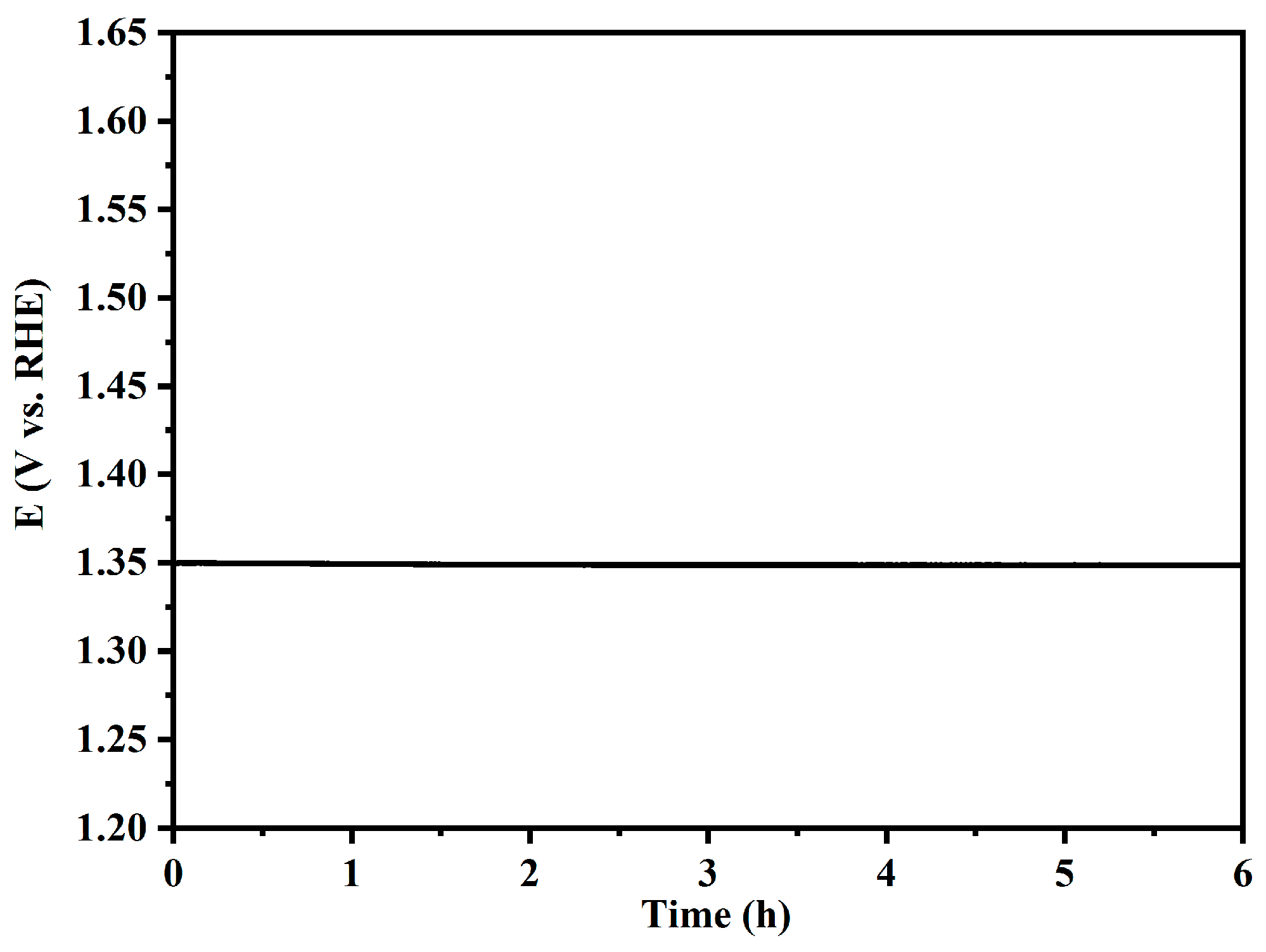
3. Results
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Gao, L.; Tang, C.; Liu, J.; He, L.; Wang, H.; Ke, Z.; Li, W.; Jiang, C.; He, D.; Cheng, L.; et al. Oxygen vacancy-induced electron density tuning of Fe3O4 for enhanced oxygen evolution catalysis. Energy Environ. Mater. 2021, 4, 392–398. [Google Scholar] [CrossRef]
- Suen, N.T.; Huang, S.F.; Quan, Q.; Zhang, N.; Xu, Y.J.; Chen, H.M. Electrocatalysisi for the oxygen evolution reaction: Recent development and future perspectives. Chem. Soc. Rev. 2017, 46, 337–365. [Google Scholar] [CrossRef] [PubMed]
- Mei, Y.J.; Feng, Y.B.; Zhang, C.X.; Qi, Q.; Hu, J. High-Entropy alloy with Mo-coordination as efficient electrocatalyst for oxygen evolution reaction. ACS Catal. 2022, 12, 10808–10817. [Google Scholar] [CrossRef]
- Teng, Y.; Huo, Y.Q.; Li, S.T.; Niu, X.; Fan, N.; Su, Z. A zipper-like NiCo2O4/Ni(OH)2 growing on multifunctional nickel foam with excellent capacitive performance. J. Alloys Compd. 2019, 784, 712–719. [Google Scholar] [CrossRef]
- He, D.P.; Wang, M.Z.; Wang, X.; Feng, S.Y.; Chen, J.; Jiang, P. Construction of ZnCo2O4 nanowire arrays 3D binder-free electrode with highly catalytic activity towards glucose oxidation. J. Solid State Chem. 2020, 284, 121214. [Google Scholar] [CrossRef]
- Lin, Y.N.; Yang, G.J.; Fu, Y.; Zhu, B.; Zhao, J.; Li, J. CoO/MnO heterostructure on three-dimensional nickel foam as efficient electrocatalyst for oxygen evolution reaction. J. Phys. Chem. Solids 2022, 160, 110373. [Google Scholar] [CrossRef]
- Zhao, L.; Yang, D.; Ma, L.L.; Feng, X.T.; Ding, H.M. An efficient heterogeneous catalyst of FeCo2O4/g-C3N4 composite for catalytic peroxymonosulfate oxidation of organic pollutants under visible light. Colloids Surf. A 2021, 610, 125725. [Google Scholar] [CrossRef]
- Liu, J.; Nan, Y.; Chang, X.X.; Li, X.Z.; Fang, Y.Y.; Liu, Y.; Tang, Y.; Wang, X.; Li, R.; Ma, J.T. Hierarchical nitrogen-enriched porous carbon materials derived from Schiff-base networks supported FeCo2O4 nanoparticles for efficient water oxidation. Int. J. Hydrogen Energy 2017, 42, 10802–10812. [Google Scholar] [CrossRef]
- Li, S.S.; Sirisomboonchai, S.; Yoshida, A.; An, X.W.; Hao, X.G.; Abudulac, A.; Guan, G.Q. Bifunctional CoNi/CoFe2O4/Ni foam electrodes for efficient overall water splitting at a high current density. J. Mater. Chem. A 2018, 6, 19221–19230. [Google Scholar] [CrossRef]
- Yan, W.N.; Yang, Z.R.; Bian, W.Y.; Yang, R.Z. FeCo2O4/hollow graphene spheres hybrid with enhanced electrocatalytic activities for oxygen reduction and oxygen evolution reaction. Carbon 2015, 92, 74–83. [Google Scholar] [CrossRef]
- Zhao, D.P.; Dai, M.Z.; Zhao, Y.; Liu, H.Q.; Liu, Y.; Wu, X. Improving electrocatalytic activities of FeCo2O4@FeCo2S4@PPy electrodes by surface/interface regulation. Nano Energy 2020, 72, 104715. [Google Scholar] [CrossRef]
- Gao, G.; Wang, K.; Wang, X.T. 2D/2D core/shell structure of FeCo2O4@NiMn LDH for efficient oxygen evolution reaction. J. Alloys Compd. 2023, 937, 168478. [Google Scholar] [CrossRef]
- Gao, S.Q.; Fan, J.C.; Liao, H.W.; Xiao, G.C.; Li, S.D.; Cui, K.X.; Niu, C.Q.; Luo, W.B.; Chao, Z.S. Three dimensional FeCo2O4 nanosheets for integrated all-solid-state supercapacitors and electrochemical energy-saving H2 production. Mater. Chem. Phys. 2022, 287, 126332. [Google Scholar] [CrossRef]
- Zhang, L.J.; Yuan, H.C.; Li, X.; Wang, Y. Hydrothermal synthesis of NiCo2O4@NiCo2O4 core-shell nanostructures anchored on Ni foam for efficient oxygen evolution reactions catalysts. Coatings 2022, 12, 1240. [Google Scholar] [CrossRef]
- Major, G.H.; Farley, N.; Sherwood, P.M.A.; Linford, M.R.; Terry, J.; Fernandez, V.; Artyushkova, K. Practical guide for curve fitting in X-ray photoelectron spectroscopoy. J. Vac. Sci. Technol. A 2020, 38, 061203. [Google Scholar] [CrossRef]
- Li, S.S.; Wang, Y.; Sun, J.L.; Xu, C.J.; Chen, H.Y. Simple preparation of porous FeCo2O4 microspheres and nanosheets for advanced asymmetric suepercapacitors. ACS Appl. Energy Mater. 2020, 3, 11307–11317. [Google Scholar] [CrossRef]
- Wang, X.H.; Xing, J.J.; Fu, X.L. Cu-doped NiFe layered double hydroxide nanosheets on nickel foam as an effective and durable electrocatalyst for oxygen evolution reaction. Int. J. Mod. Phys. B 2023, 37, 2350260. [Google Scholar] [CrossRef]
- Dong, F.; Liu, X.H.; Irfan, M.; Yang, L.; Li, S.L.; Ding, J.; Li, Y.; Khan, I.U.; Zhang, P.P. Macaroon-like FeCo2O4 modified activated carbon anode for enhancing power generation in direct glucose fuel cell. Int. J. Hydrogen Energy 2019, 44, 8178–8187. [Google Scholar] [CrossRef]
- Liu, L.; Zhang, H.; Mu, Y.; Yang, J.; Wang, Y.; Yang, J.; Wang, Y. Porous Iron cobaltate nanoneedles array on nickel foam as anode materials for lithium-ion batteries with enhanced electrochemical performance. ACS Appl. Mater. Interfaces 2016, 8, 1351–1359. [Google Scholar] [CrossRef]
- Vishnu, V.; Kumar, N.; Narayanasamy, S. Development of Fe3O4/CAC nanocomposite for the effective removal of contaminants of emerging concerns (Ce3+) from water: An ecotoxocological assessment. Environ. Pollut. 2021, 285, 117326. [Google Scholar] [CrossRef]
- Yamashita, T.; Hayes, P. Analysis of XPS spectra of Fe2+ and Fe3+ ions in oxide materials. Appl. Surf. Sci. 2008, 254, 2441–2449. [Google Scholar] [CrossRef]
- Ni, L.; Zhou, J.H.; Chen, N.N.; Li, X.G.; Xu, S.C.; Zhang, L.; Lu, C.L.; Chen, J.; Xu, L.; Hou, W.H. In situ direct growth of flower-like hierarchical architecture of CoNi-layered double hydroxide on Ni foam as an efficient self-supported oxygen evolution electrocatalyst. Int. J. Hydrogen Energy 2020, 45, 22788–22796. [Google Scholar] [CrossRef]
- Harada, M.; Kotegawa, F.; Kuwa, M. Structural changes of spinel MCo2O4(M=Mn, Fe, Co, Ni and Zn) electrocatalysts during the oxygen evolution reaction investigated by in stiu X-ray Adsorption spectroscopy. ACS Appl. Energy Mater. 2022, 5, 278–294. [Google Scholar] [CrossRef]
- Zhang, J.Y.; Sun, X.; Liu, Y.; Hou, L.R.; Yuan, C.Z. Design and construction of bi-metal MOF-derived yolk-shell Ni2P/ZnNp2 hollow microspheres for efficient electrocatalytic oxygen evolution. Mater. Chem. Front. 2020, 4, 1366. [Google Scholar] [CrossRef]
- Hu, J.M.; Zhang, J.Q.; Cao, C.N. Oxygen evolution reaction on IrO2 based DSA type electrodes:kinetics analysis of Tafel lines and EIS. Int. J. Hydrogen Energy 2003, 29, 791–797. [Google Scholar] [CrossRef]
- Shrestha, N.K.; Patil, S.A.; Han, J.H.; Cho, S.; Inamdar, A.I.; Kim, H.; Im, H. Chemical etching induced microporous nickel backbones decorated with metallic Fe@hydroxide nanocatalysts: An efficient and sustainable OER anode toward industrial alkaline water-splitting. J. Mater. Chem. A 2022, 10, 8989–9000. [Google Scholar] [CrossRef]
- Shi, H.; Zha, Q.; Ni, Y. Fe-doped (Ni, Mn)Co2O4 nanorod arrays on Ni foam as highly efficient electrocatalyst for oxygen evolution reaction in alkaline and neutral conditions with superb long-term stability. J. Alloys Compd. 2022, 904, 164052. [Google Scholar] [CrossRef]
- Alegre, C.; Busacca, C.; Di Blasi, A.; Di Blasi, O.; Aricò, A.S.; Antonucci, V.; Baglio, V. Toward more efficient and stable bifunctional electrocatalysts for oxygen electrodes using FeCo2O4/carbon nanofiber prepared by electrospinning. Mater. Today Energy 2020, 18, 100508. [Google Scholar] [CrossRef]
- Chen, S.; Yu, C.; Cao, Z.F.; Huang, X.P.; Wang, S.; Zhong, H. Trimetallic NiFeCr-LDH/MoS2 composites as novel electrocatalyst for OER. Int. J. Hydrogen Energy 2021, 46, 7037–7046. [Google Scholar] [CrossRef]
- Hao, C.; Wu, Y.; An, Y.; Cui, B.; Lin, J.; Li, X.; Wang, D.; Jiang, M.; Cheng, Z.; Hu, S. Interface-coupling of CoFe-LDH on MXene as high-performance oxygen evolution catalyst. Mater. Today Energy 2019, 12, 453–462. [Google Scholar] [CrossRef]
- Long, X.; Li, J.; Xiao, S.; Yan, K.Y.; Wang, Z.L.; Chen, H.N.; Yang, S.H. A strongly coupled graphene and FeNi double 372 hydroxide hybrids as an excellent electrocatalyst for the oxygen evolution reaction. Angew. Chem. Int. Ed. Engl. 2014, 53, 7584–7588. [Google Scholar] [CrossRef] [PubMed]
- Bi, Y.; Cai, Z.; Zhou, D.; Tian, Y.; Zhang, Q.; Zhang, Q.; Kuang, Y.; Li, Y.; Sun, X.; Duan, X. Understanding the incorporating effect of Co2+/Co3+ in NiFe-layered double hydroxide for electrocatalytic oxygen evolution reaction. J. Catal. 2018, 358, 100–107. [Google Scholar] [CrossRef]
- Magalhães, R.S.; Bargiela, P.; Rocha, M.G.C.; Gild, E.S.; Souza, A.R. Iron Cobaltite (FeCo2O4) nanocatalysts for water-oxidation: Effects of annealing temperature on catalytic properties. J. Braz. Chem. Soc. 2022, 33, 1163–1171. [Google Scholar] [CrossRef]



| Catalyst | Electrolyte (KOH) | Current Density (mA cm−2) | Overpotential for OER (mV) | References |
|---|---|---|---|---|
| IrO2 | 1 M | 50 | 354 | [25] |
| NF | 1 M | 50 | 480 | [26] |
| FeCo2O4@NPC-450 | 1 M | 10 | 330 | [8] |
| FeCo2O4@NiMn LDH | 1 M | 10 | 232 | [12] |
| Fe-doped (Ni,Mn)Co2O4/NF | 1 M | 10 | 242 | [27] |
| FeCo2O4/carbon nanofiber | 6 M | 10 | 130 | [28] |
| NiCo2+Fe LDH | 1 M | 10 | 290 | [29] |
| CoFe LDH | 1 M | 10 | 319 | [30] |
| FeCo2O4/NF-120 | 1 M | 10 | 124 | This work |
| 50 | 339 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, L.; Quan, Z.; Wang, Y.; Li, H.; Yang, X. Construction of Flower-like FeCo2O4 Nanosheets on Ni Foam as Efficient Electrocatalyst for Oxygen Evolution Reaction. Coatings 2023, 13, 1875. https://doi.org/10.3390/coatings13111875
Zhang L, Quan Z, Wang Y, Li H, Yang X. Construction of Flower-like FeCo2O4 Nanosheets on Ni Foam as Efficient Electrocatalyst for Oxygen Evolution Reaction. Coatings. 2023; 13(11):1875. https://doi.org/10.3390/coatings13111875
Chicago/Turabian StyleZhang, Lijuan, Zhonggui Quan, Yan Wang, Hangyang Li, and Xu Yang. 2023. "Construction of Flower-like FeCo2O4 Nanosheets on Ni Foam as Efficient Electrocatalyst for Oxygen Evolution Reaction" Coatings 13, no. 11: 1875. https://doi.org/10.3390/coatings13111875
APA StyleZhang, L., Quan, Z., Wang, Y., Li, H., & Yang, X. (2023). Construction of Flower-like FeCo2O4 Nanosheets on Ni Foam as Efficient Electrocatalyst for Oxygen Evolution Reaction. Coatings, 13(11), 1875. https://doi.org/10.3390/coatings13111875





