An Assessment of the Bactericidal and Virucidal Properties of ZrN-Cu Nanostructured Coatings Deposited by an Industrial PVD System
Abstract
1. Introduction
2. Materials and Methods
2.1. Synthesis of the Coatings
2.2. Analyses and Characterizations
2.3. Assessment of Bactericidal Properties
2.4. Assessment of Virucidal Properties
2.4.1. Virus and Cell Line
2.4.2. Plaque Assay
2.4.3. qRT-PCR Quantitation of SARS-CoV-2 Viral RNA
3. Results and Discussions
3.1. Mechanical Properties and Chemical Composition
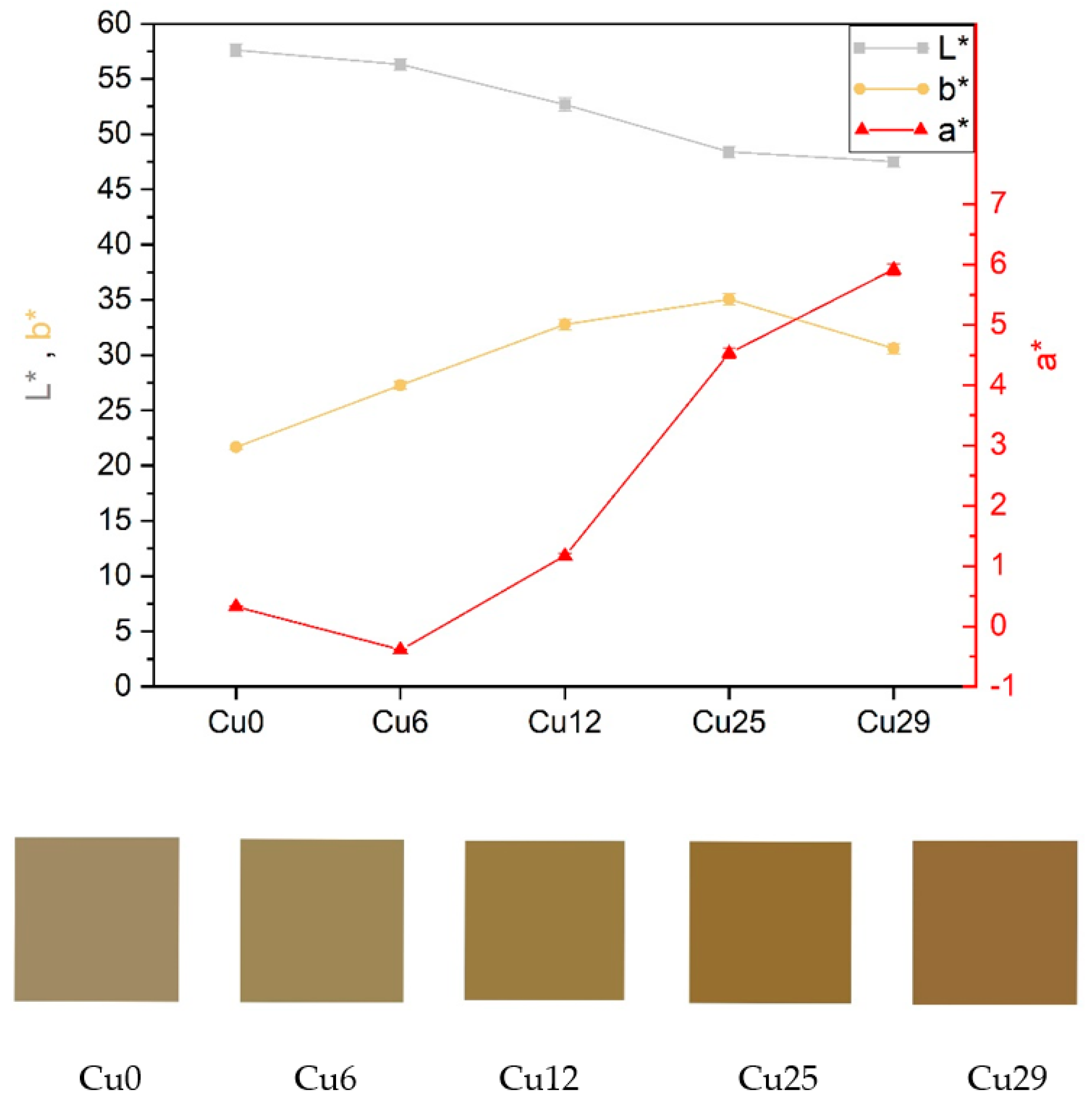
3.2. The Coloration of the Coatings
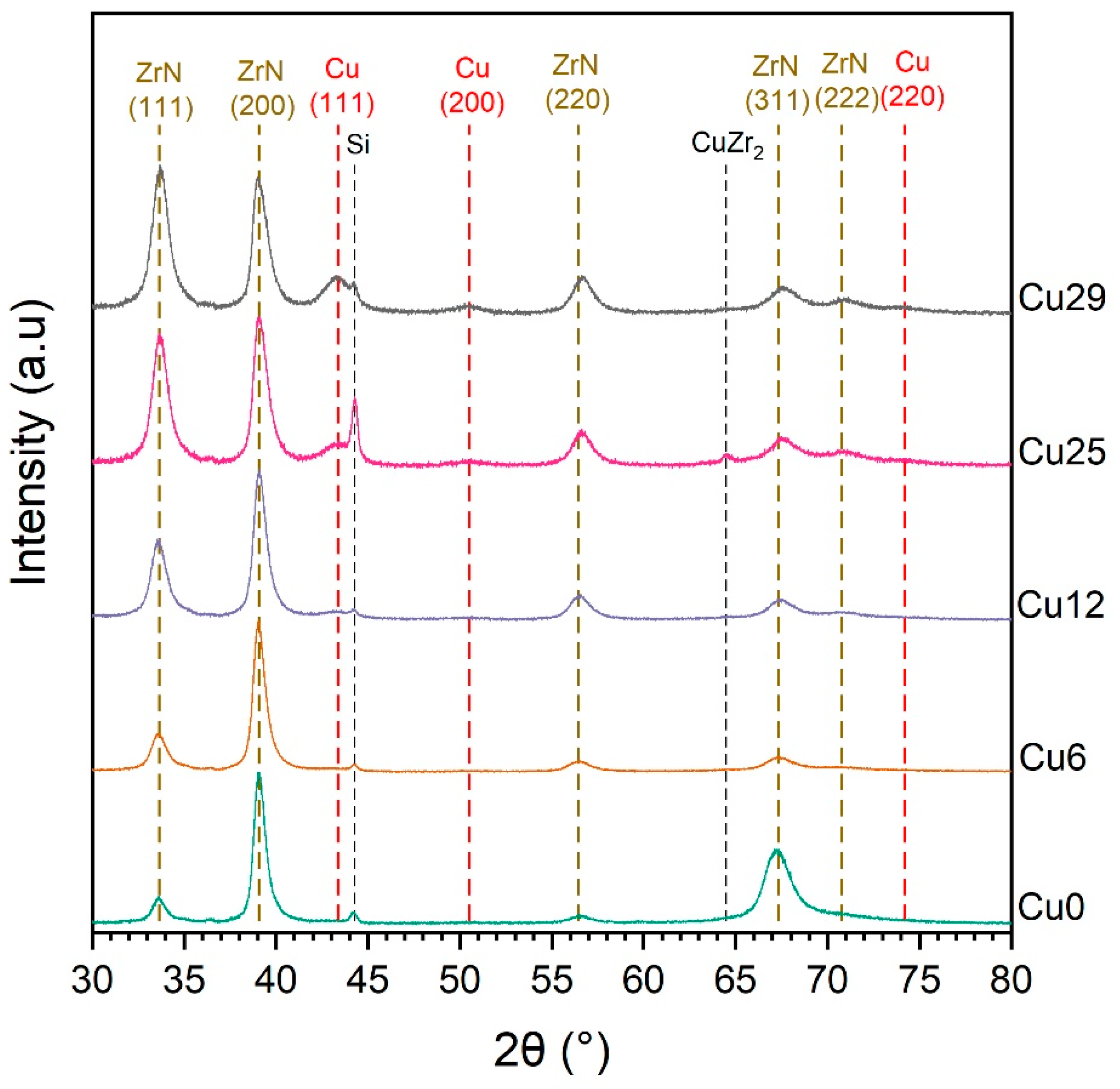
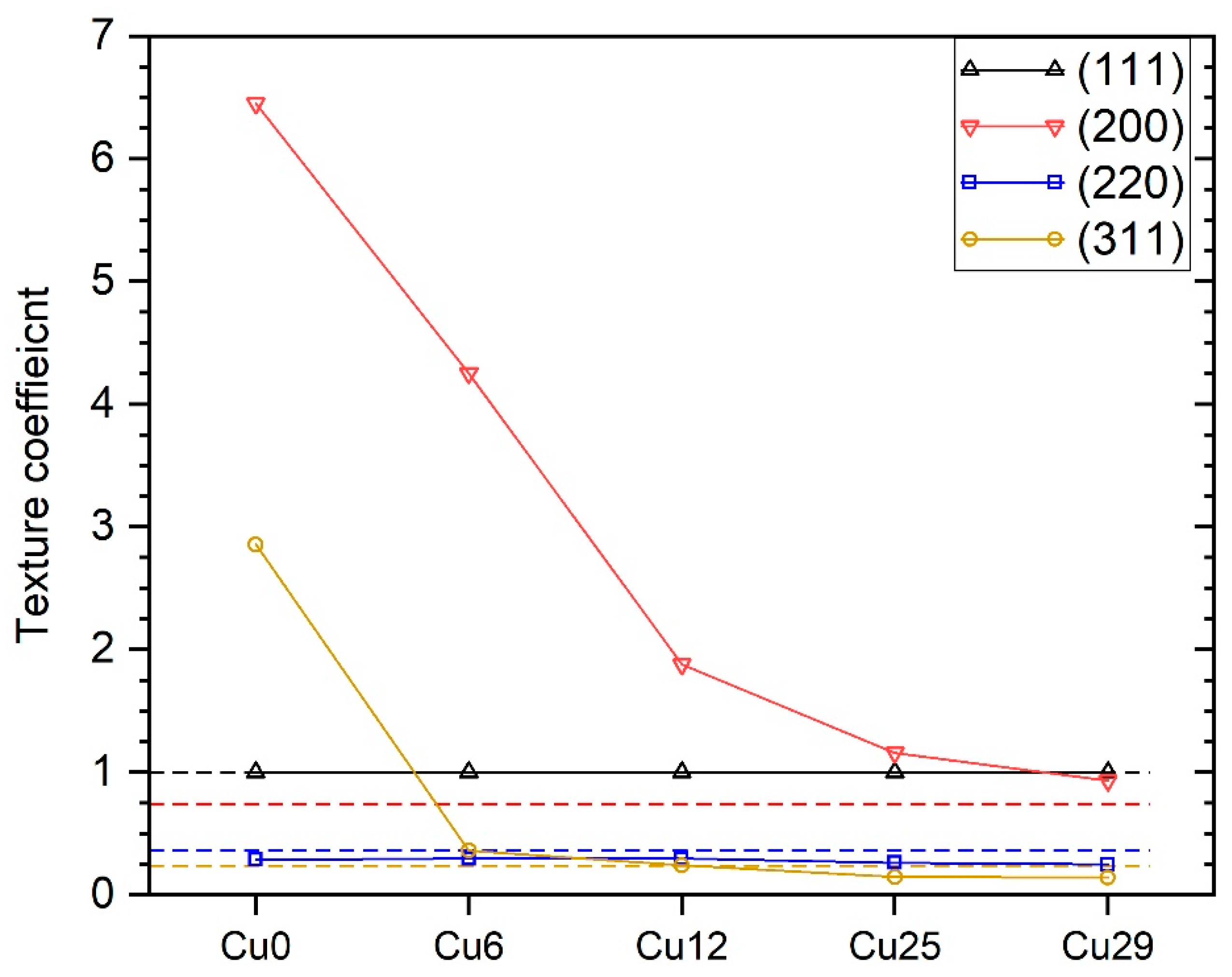
3.3. Microstructure
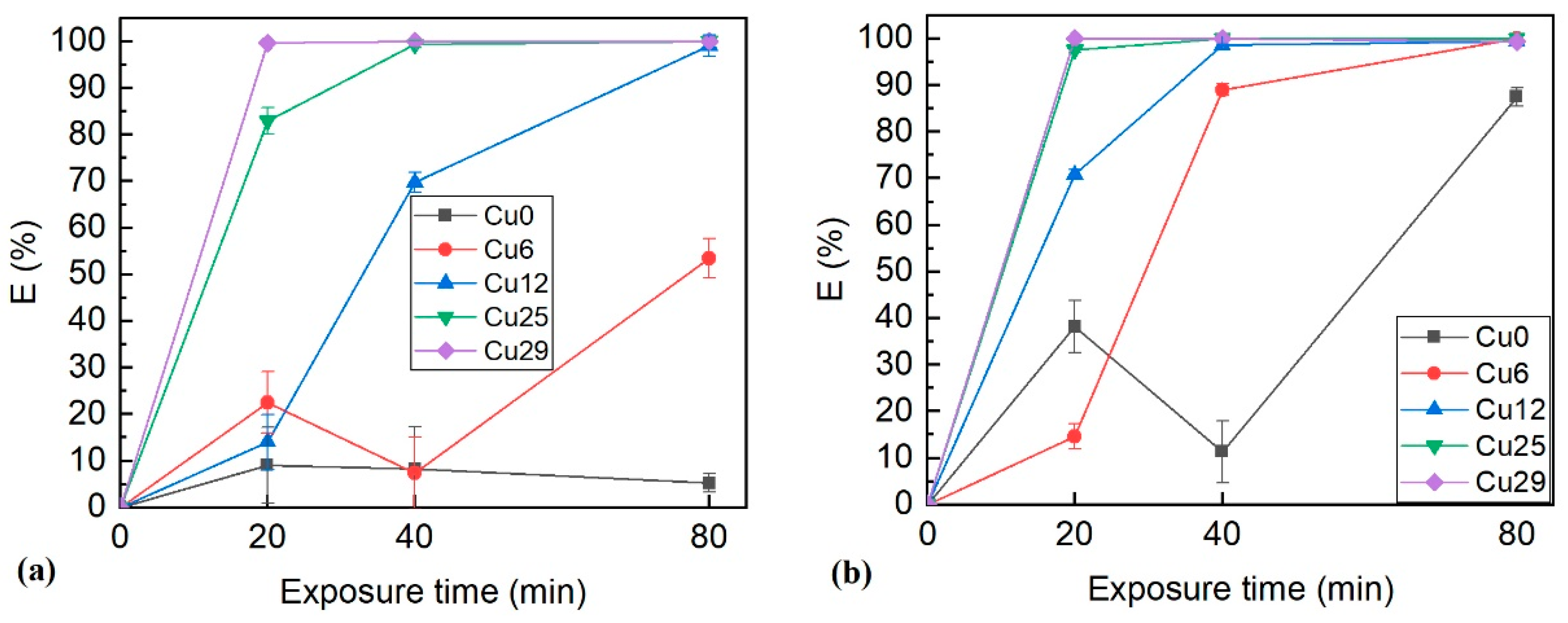
3.4. Bactericidal Properties
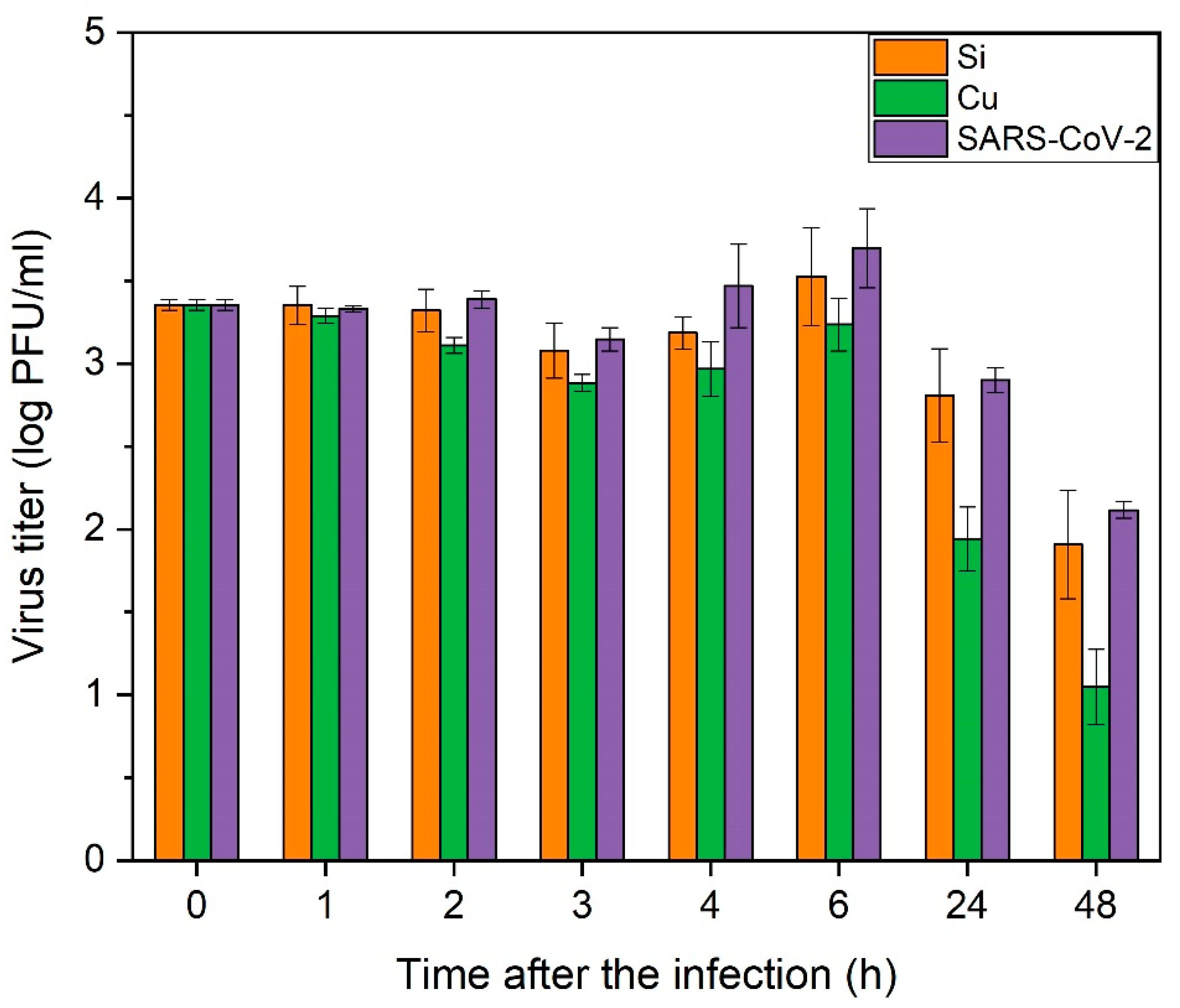
3.5. Virucidal Properties
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Potter, B.A.; Lob, M.; Mercaldo, R.; Hetzler, A.; Kaistha, V.; Khan, H.; Kingston, N.; Knoll, M.; Maloy-Franklin, B.; Melvin, K.; et al. A long-term study examining the antibacterial effectiveness of Agion silver zeolite technology on door handles within a college campus. Lett. Appl. Microbiol. 2015, 60, 120–127. [Google Scholar] [CrossRef] [PubMed]
- El-Rafie, M.H.; Ahmed, H.B.; Zahran, M.K. Characterization of nanosilver coated cotton fabrics and evaluation of its antibacterial efficacy. Carbohydr. Polym. 2014, 107, 174–181. [Google Scholar] [CrossRef] [PubMed]
- Page, K.; Wilson, M.; Parkin, I.P. Antimicrobial surfaces and their potential in reducing the role of the inanimate environment in the incidence of hospital-acquired infections. J. Mater. Chem. 2009, 19, 3818–3831. [Google Scholar] [CrossRef]
- Burke, G.H.; Butler, J.P. Analysis of the role of copper impregnated composite hard surfaces, bed linens and patient gowns in reducing healthcare-associated infection rates. Int. J. Infect. Control 2018, 14, 1–8. [Google Scholar] [CrossRef]
- Wickens, D.J.; West, G.; Kelly, P.J.; Verran, J.; Lynch, S.; Whitehead, K.A. Antimicrobial activity of nanocomposite zirconium nitride/silver coatings to combat external bone fixation pin infections. Int. J. Artif. Organs 2012, 35, 817–825. [Google Scholar] [CrossRef]
- Simoncic, B.; Tomsic, B. Structures of Novel Antimicrobial Agents for Textiles—A Review. Text. Res. J. 2010, 80, 1721–1737. [Google Scholar] [CrossRef]
- Chen, N.H.; Chung, C.J.; Chiang, C.C.; Chen, K.C.; He, J.L. Antimicrobial copper-containing titanium nitride coatings Co-deposited by arc ion plating/magnetron sputtering for protective and decorative purposes. Surf. Coat. Technol. 2014, 253, 83–88. [Google Scholar] [CrossRef]
- Kelly, P.J.; Li, H.; Benson, P.S.; Whitehead, K.A.; Verran, J.; Arnell, R.D.; Iordanova, I. Comparison of the tribological and antimicrobial properties of CrN/Ag, ZrN/Ag, TiN/Ag, and TiN/Cu nanocomposite coatings. Surf. Coat. Technol. 2010, 205, 1606–1610. [Google Scholar] [CrossRef]
- Zhao, L.; Chu, P.K.; Zhang, Y.; Wu, Z. Antibacterial coatings on titanium implants. J. Biomed. Mater. Res. Part B Appl. Biomater. 2009, 91, 470–480. [Google Scholar] [CrossRef]
- Qin, S.; Xu, K.; Nie, B.; Ji, F.; Zhang, H. Approaches based on passive and active antibacterial coating on titanium to achieve antibacterial activity. J. Biomed. Mater. Res. Part A 2018, 106, 2531–2539. [Google Scholar] [CrossRef]
- Tian, X.B.; Wang, Z.M.; Yang, S.Q.; Luo, Z.J.; Fu, R.K.Y.; Chu, P.K. Antibacterial copper-containing titanium nitride films produced by dual magnetron sputtering. Surf. Coat. Technol. 2007, 201, 8606–8609. [Google Scholar] [CrossRef]
- Wang, J.; Zhao, B.; Li, G.; Yang, K.; Yu, J.; Yang, X.; Zhan, D. Study of TiCu/TiCuN multilayer films with antibacterial activity. Mater. Technol. 2020, 35, 475–482. [Google Scholar] [CrossRef]
- Mejía, H.D.; Echavarría, A.M.; Bejarano G., G. Influence of Ag-Cu nanoparticles on the microstructural and bactericidal properties of TiAlN(Ag,Cu) coatings for medical applications deposited by Direct Current (DC) magnetron sputtering. Thin Solid Film. 2019, 687, 137460. [Google Scholar] [CrossRef]
- Sharifahmadian, O.; Salimijazi, H.R.; Fathi, M.H.; Mostaghimi, J.; Pershin, L. Relationship between surface properties and antibacterial behavior of wire arc spray copper coatings. Surf. Coat. Technol. 2013, 233, 74–79. [Google Scholar] [CrossRef]
- Birkett, M.; Dover, L.; Lukose, C.C.; Zia, A.W.; Tambuwala, M.M.; Serrano-Aroca, Á. Recent Advances in Metal-Based Antimicrobial Coatings for High-Touch Surfaces. Int. J. Mol. Sci. 2022, 23, 1162. [Google Scholar] [CrossRef]
- Wang, N.; Ferhan, A.R.; Yoon, B.K.; Jackman, J.A.; Cho, N.J.; Majima, T. Chemical design principles of next-generation antiviral surface coatings. Chem. Soc. Rev. 2021, 50, 9741–9765. [Google Scholar] [CrossRef]
- Balagna, C.; Francese, R.; Perero, S.; Lembo, D.; Ferraris, M. Nanostructured composite coating endowed with antiviral activity against human respiratory viruses deposited on fibre-based air filters. Surf. Coat. Technol. 2021, 409, 126873. [Google Scholar] [CrossRef]
- Mittireddi, R.T.; Patel, N.M.; Gautam, A.R.S.; Soppina, V.; Panda, E. Non-stoichiometric amorphous TiOx as a highly reactive, transparent anti-viral surface coating. J. Alloys Compd. 2021, 881, 160610. [Google Scholar] [CrossRef]
- Meister, T.L.; Fortmann, J.; Breisch, M.; Sengstock, C.; Steinmann, E.; Köller, M.; Pfaender, S.; Ludwig, A. Copper and silver thin film systems display differences in antiviral and antibacterial properties-implications for the prevention of SARS-CoV-2 infections. arXiv 2021, arXiv:2106.11606. [Google Scholar]
- Butot, S.; Baert, L.; Zubera, A.S. Assessment of Antiviral Coatings for High-Touch Surfaces by Using Human Coronaviruses Hcov-229e and Sars-Cov-2. Appl. Environ. Microbiol. 2021, 87, e01098-21. [Google Scholar] [CrossRef]
- Rakowska, P.D.; Tiddia, M.; Faruqui, N.; Bankier, C.; Pei, Y.; Pollard, A.J.; Zhang, J.; Gilmore, I.S. Antiviral surfaces and coatings and their mechanisms of action. Commun. Mater. 2021, 2, 53. [Google Scholar] [CrossRef]
- Basak, S.; Packirisamy, G. Nano-based antiviral coatings to combat viral infections. Nano-Struct. Nano-Objects 2020, 24, 100620. [Google Scholar] [CrossRef]
- Luo, J.; Hein, C.; Mücklich, F.; Solioz, M. Killing of bacteria by copper, cadmium, and silver surfaces reveals relevant physicochemical parameters. Biointerphases 2017, 12, 020301. [Google Scholar] [CrossRef] [PubMed]
- Vincent, M.; Duval, R.E.; Hartemann, P.; Engels-Deutsch, M. Contact killing and antimicrobial properties of copper. J. Appl. Microbiol. 2018, 124, 1032–1046. [Google Scholar] [CrossRef] [PubMed]
- Shimazaki, T.; Miyamoto, H.; Ando, Y.; Noda, I.; Yonekura, Y.; Kawano, S.; Miyazaki, M.; Mawatari, M.; Hotokebuchi, T. In vivo antibacterial and silver-releasing properties of novel thermal sprayed silver-containing hydroxyapatite coating. J. Biomed. Mater. Res. Part B Appl. Biomater. 2010, 92, 386–389. [Google Scholar] [CrossRef]
- Ju, H.; Yu, D.; Yu, L.; Ding, N.; Xu, J.; Zhang, X.; Zheng, Y.; Yang, L.; He, X. The influence of Ag contents on the microstructure, mechanical and tribological properties of ZrN-Ag films. Vacuum 2018, 148, 54–61. [Google Scholar] [CrossRef]
- Fielding, G.A.; Roy, M.; Bandyopadhyay, A.; Bose, S. Antibacterial and biological characteristics of silver containing and strontium doped plasma sprayed hydroxyapatite coatings. Acta Biomater. 2012, 8, 3144–3152. [Google Scholar] [CrossRef]
- Chen, W.; Liu, Y.; Courtney, H.S.; Bettenga, M.; Agrawal, C.M.; Bumgardner, J.D.; Ong, J.L. In vitro anti-bacterial and biological properties of magnetron co-sputtered silver-containing hydroxyapatite coating. Biomaterials 2006, 27, 5512–5517. [Google Scholar] [CrossRef]
- Kelly, P.J.; Li, H.; Whitehead, K.A.; Verran, J.; Arnell, R.D.; Iordanova, I. A study of the antimicrobial and tribological properties of TiN/Ag nanocomposite coatings. Surf. Coat. Technol. 2009, 204, 1137–1140. [Google Scholar] [CrossRef]
- Musil, J.; Zítek, M.; Fajfrlík, K.; Čerstvý, R. Flexible antibacterial Zr-Cu-N thin films resistant to cracking. J. Vac. Sci. Technol. A. 2016, 34, 021508. [Google Scholar] [CrossRef]
- Stranak, V.; Wulff, H.; Rebl, H.; Zietz, C.; Arndt, K.; Bogdanowicz, R.; Nebe, B.; Bader, R.; Podbielski, A.; Hubicka, Z.; et al. Deposition of thin titanium-copper films with antimicrobial effect by advanced magnetron sputtering methods. Mater. Sci. Eng. C 2011, 31, 1512–1519. [Google Scholar] [CrossRef]
- Kumar, D.D.; Kaliaraj, G.S. Multifunctional zirconium nitride/copper multilayer coatings on medical grade 316L SS and titanium substrates for biomedical applications. J. Mech. Behav. Biomed. Mater. 2018, 77, 106–115. [Google Scholar] [CrossRef] [PubMed]
- Hadrup, N.; Sharma, A.K.; Loeschner, K. Toxicity of silver ions, metallic silver, and silver nanoparticle materials after in vivo dermal and mucosal surface exposure: A review. Regul. Toxicol. Pharmacol. 2018, 98, 257–267. [Google Scholar] [CrossRef]
- Ji, M.K.; Park, S.W.; Lee, K.; Kang, I.C.; Yun, K.D.; Kim, H.S.; Lim, H.P. Evaluation of antibacterial activity and osteoblast-like cell viability of TiN, ZrN and (Ti1-xZrx)N coating on titanium. J. Adv. Prosthodont. 2015, 7, 166–171. [Google Scholar] [CrossRef] [PubMed]
- Brunello, G.; Brun, P.; Gardin, C.; Ferroni, L.; Bressan, E.; Meneghello, R.; Zavan, B.; Sivolella, S. Biocompatibility and antibacterial properties of zirconium nitride coating on titanium abutments: An in vitro study. PLoS ONE 2018, 13, e0199591. [Google Scholar] [CrossRef]
- Ramoul, C.; Beliardouh, N.E.; Bahi, R.; Nouveau, C.; Djahoudi, A.; Walock, M.J. Surface performances of PVD ZrN coatings in biological environments. Tribol.-Mater. Surf. Interfaces 2019, 13, 12–19. [Google Scholar] [CrossRef]
- Peng, C.; Zhao, Y.; Jin, S.; Wang, J.; Liu, R.; Liu, H.; Shi, W.; Kolawole, S.K.; Ren, L.; Yu, B.; et al. Antibacterial TiCu/TiCuN Multilayer Films with Good Corrosion Resistance Deposited by Axial Magnetic Field-Enhanced Arc Ion Plating. ACS Appl. Mater. Interfaces 2019, 11, 125–136. [Google Scholar] [CrossRef] [PubMed]
- Baer, A.; Kehn-Hall, K. Viral concentration determination through plaque assays: Using traditional and novel overlay systems. J. Vis. Exp. 2014, 93, e52065. [Google Scholar] [CrossRef]
- Audronis, M.; Jimenez, O.; Leyland, A.; Matthews, A. The morphology and structure of PVD ZrN-Cu thin films. J. Phys. D Appl. Phys. 2009, 42, 085308. [Google Scholar] [CrossRef]
- Jimenez, O.; Audronis, M.; Baker, M.A.; Matthews, A.; Leyland, A. Structure and mechanical properties of nitrogen-containing Zr-Cu based thin films deposited by pulsed magnetron sputtering. J. Phys. D Appl. Phys. 2008, 41, 155301. [Google Scholar] [CrossRef]
- Li, J.; Zhang, H.; Fan, A.; Tang, B. Tribological properties characterization of Ti/Cu/N Thin films prepared by DC magnetron sputtering on titanium alloy. Surf. Coat. Technol. 2016, 294, 30–35. [Google Scholar] [CrossRef]
- Zeman, P.; Čerstvý, R.; Mayrhofer, P.H.; Mitterer, C.; Musil, J. Structure and properties of hard and superhard Zr-Cu-N nanocomposite coatings. Mater. Sci. Eng. A 2000, 289, 189–197. [Google Scholar] [CrossRef]
- Slater, J.C. Atomic radii in crystals. J. Chem. Phys. 1964, 41, 3199–3204. [Google Scholar] [CrossRef]
- Chen, T.; Yu, L.; Ju, H.; Xu, J.; Koyama, S. Influence of Cu Content on the Microstructure, Mechanical, and Tribological Properties of ZrN-Cu Films. Nano 2018, 13, 1850035. [Google Scholar] [CrossRef]
- Pierson, J.F.; Wiederkehr, D.; Billard, A. Reactive magnetron sputtering of copper, silver, and gold. Thin Solid Film. 2005, 478, 196–205. [Google Scholar] [CrossRef]
- Nowakowska-Langier, K.; Chodun, R.; Minikayev, R.; Okrasa, S.; Strzelecki, G.W.; Wicher, B.; Zdunek, K. Copper nitride layers synthesized by pulsed magnetron sputtering. Thin Solid Film. 2018, 645, 32–37. [Google Scholar] [CrossRef]
- Liu, Z.Q.; Wang, W.J.; Wang, T.M.; Chao, S.; Zheng, S.K. Thermal stability of copper nitride films prepared by rf magnetron sputtering. Thin Solid Film. 1998, 325, 55–59. [Google Scholar] [CrossRef]
- Rahmati, A.; Bidadi, H.; Ahmadi, K.; Hadian, F. Ti substituted nano-crystalline Cu3N thin films. J. Coat. Technol. Res. 2011, 8, 289–297. [Google Scholar] [CrossRef][Green Version]
- Maruyama, T.; Morishita, T. Copper nitride and tin nitride thin films for write-once optical recording media. Appl. Phys. Lett. 1996, 69, 890–891. [Google Scholar] [CrossRef]
- Maya, L. Deposition of crystalline binary nitride films of tin, copper, and nickel by reactive sputtering. J. Vac. Sci. Technol. A 1993, 11, 604–608. [Google Scholar] [CrossRef]
- Wang, D.; Nakamine, N.; Hayashi, Y. Properties of various sputter-deposited Cu–N thin films. J. Vac. Sci. Technol. A 1998, 16, 2084–2092. [Google Scholar] [CrossRef]
- Pierson, J.F. Structure and properties of copper nitride films formed by reactive magnetron sputtering. Vacuum 2002, 66, 59–64. [Google Scholar] [CrossRef]
- Yue, G.H.; Yan, P.X.; Wang, J. Study on the preparation and properties of copper nitride thin films. J. Cryst. Growth 2005, 274, 464–468. [Google Scholar] [CrossRef]
- Musil, J.; Zeman, P.; Hrubý, H.; Mayrhofer, P.H. ZrN/Cu nanocomposite film—A novel superhard material. Surf. Coat. Technol. 1999, 120, 179–183. [Google Scholar] [CrossRef]
- Schiøtz, J.; Vegge, T.; Di Tolla, F.D.; Jacobsen, K.W. Atomic-scale simulations of the mechanical deformation of nanocrystalline metals. Phys. Rev. B Condens. Matter Mater. Phys. 1999, 60, 11971–11983. [Google Scholar] [CrossRef]
- Zhang, S.; Sun, D.; Fu, Y.; Du, H. Recent advances of superhard nanocomposite coatings: A review. Surf. Coat. Technol. 2003, 167, 113–119. [Google Scholar] [CrossRef]
- Santo, C.E.; Taudte, N.; Nies, D.H.; Grass, G. Contribution of copper ion resistance to survival of Escherichia coli on metallic copper surfaces. Appl. Environ. Microbiol. 2008, 74, 977–986. [Google Scholar] [CrossRef]
- Kertzman, Z.; Marchal, J.; Suarez, M.; Staia, M.H.; Filip, P.; Kohli, P.; Aouadi, S.M. Mechanical, tribological, and biocompatibility properties of ZrN-Ag nanocomposite films. J. Biomed. Mater. Res. Part A 2008, 84, 1061–1067. [Google Scholar] [CrossRef] [PubMed]
- Wißmann, J.E.; Kirchhoff, L.; Brüggemann, Y.; Todt, D.; Steinmann, J.; Steinmann, E. Persistence of pathogens on inanimate surfaces: A narrative review. Microorganisms 2021, 9, 343. [Google Scholar] [CrossRef]
- Lanini, S.; D’Arezzo, S.; Puro, V.; Martini, L.; Imperi, F.; Piselli, P.; Montanaro, M.; Paoletti, S.; Visca, P.; Ippolito, G. Molecular epidemiology of a Pseudomonas aeruginosa hospital outbreak driven by a contaminated disinfectant-soap dispenser. PLoS ONE 2011, 6, e17064. [Google Scholar] [CrossRef]
- Dan, Z.G.; Ni, H.W.; Xu, B.F.; Xiong, J.; Xiong, P.Y. Microstructure and antibacterial properties of AISI 420 stainless steel implanted by copper ions. Thin Solid Film. 2005, 492, 93–100. [Google Scholar] [CrossRef]
- Chin, A.W.H.; Chu, J.T.S.; Perera, M.R.A.; Hui, K.P.Y.; Yen, H.L.; Chan, M.C.W.; Peiris, M.; Poon, L.L.M. Stability of SARS-CoV-2 in different environmental conditions. Lancet Microbe 2020, 1, e10. [Google Scholar] [CrossRef]
- Warnes, S.L.; Little, Z.R.; Keevil, C.W. Human coronavirus 229E remains infectious on common touch surface materials. MBio 2015, 6, e01697-15. [Google Scholar] [CrossRef] [PubMed]
- Warnes, S.L.; Keevil, C.W. Inactivation of Norovirus on Dry Copper Alloy Surfaces. PLoS ONE 2013, 8, e75017. [Google Scholar] [CrossRef]
- Warnes, S.L.; Summersgill, E.N.; Keevil, C.W. Inactivation of murine norovirus on a range of copper alloy surfaces is accompanied by loss of capsid integrity. Appl. Environ. Microbiol. 2015, 81, 1085–1091. [Google Scholar] [CrossRef] [PubMed]
- Sagripanti, J.L.; Routson, L.B.; Lytle, C.D. Virus inactivation by copper or iron ions alone and in the presence of peroxide. Appl. Environ. Microbiol. 1993, 59, 4374–4376. [Google Scholar] [CrossRef]
- Govind, V.; Bharadwaj, S.; Sai Ganesh, M.R.; Vishnu, J.; Shankar, K.V.; Shankar, B.; Rajesh, R. Antiviral properties of copper and its alloys to inactivate covid-19 virus: A review. BioMetals 2021, 34, 1217–1235. [Google Scholar] [CrossRef]
- Purniawan, A.; Lusida, M.I.; Pujiyanto, R.W.; Nastri, A.M.; Permanasari, A.A.; Harsono, A.A.H.; Oktavia, N.H.; Wicaksono, S.T.; Dewantari, J.R.; Prasetya, R.R.; et al. Synthesis and assessment of copper-based nanoparticles as a surface coating agent for antiviral properties against SARS-CoV-2. Sci. Rep. 2022, 12, 4835. [Google Scholar] [CrossRef]
- van Doremalen, N.; Bushmaker, T.; Morris, D.H.; Holbrook, M.G.; Gamble, A.; Williamson, B.N.; Tamin, A.; Harcourt, J.L.; Thornburg, N.J.; Gerber, S.I.; et al. Aerosol and Surface Stability of SARS-CoV-2 as Compared with SARS-CoV-1. N. Engl. J. Med. 2020, 382, 1564–1567. [Google Scholar] [CrossRef]
- Lishchynskyi, O.; Shymborska, Y.; Stetsyshyn, Y.; Raczkowska, J.; Skirtach, A.G.; Peretiatko, T.; Budkowski, A. Passive antifouling and active self-disinfecting antiviral surfaces. Chem. Eng. J. 2022, 446, 137048. [Google Scholar] [CrossRef]
- Bregnocchi, A.; Jafari, R.; Momen, G. Design strategies for antiviral coatings and surfaces: A review. Appl. Surf. Sci. Adv. 2022, 8, 100224. [Google Scholar] [CrossRef]
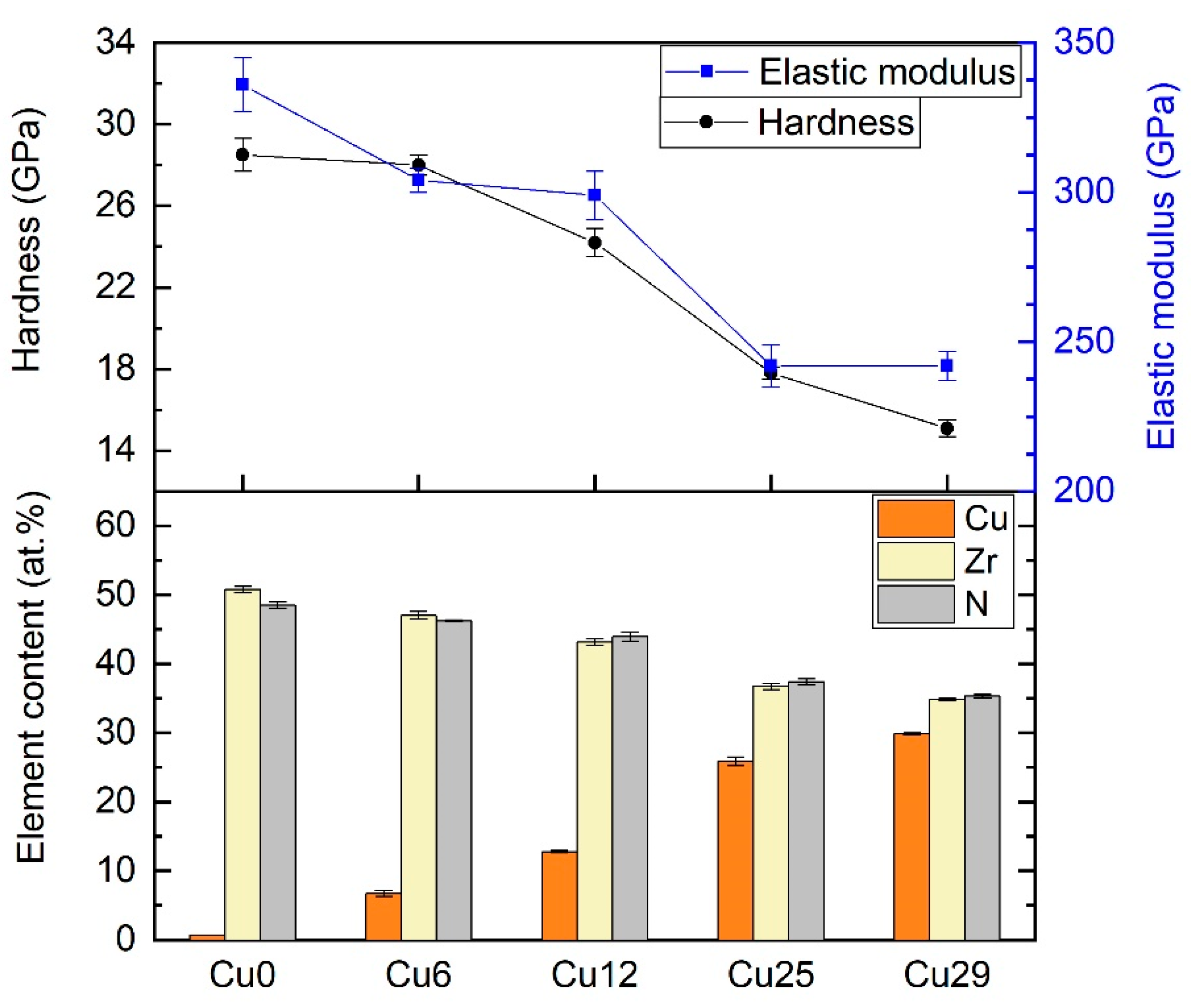




| Sample | Cu Target Power (kW) | Cu (at.%) | Zr (at.%) | N (at.%) | Deposition Rate (nm/min) | Hardness (GPa) | Elastic Modulus (GPa) |
|---|---|---|---|---|---|---|---|
| Cu0 | 0.5 | 0.6 | 50.7 | 48.7 | 19 | 28.5 ± 0.8 | 336 ± 9 |
| Cu6 | 1 | 6.7 | 47.1 | 46.2 | 19 | 28.0 ± 0.5 | 304 ± 4 |
| Cu12 | 1.3 | 12.8 | 43.2 | 44.0 | 21 | 24.2 ± 0.7 | 299 ± 8 |
| Cu25 | 2.2 | 25.8 | 36.9 | 37.3 | 17 | 17.8 ± 0.3 | 242 ± 7 |
| Cu29 | 3 | 29.8 | 34.9 | 35.3 | 22 | 15.1 ± 0.4 | 242 ± 5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Behrangi, S.; Sedláček, I.; Štěrba, J.; Suková, G.; Czigány, Z.; Buršíková, V.; Souček, P.; Sochora, V.; Balázsi, K.; Vašina, P. An Assessment of the Bactericidal and Virucidal Properties of ZrN-Cu Nanostructured Coatings Deposited by an Industrial PVD System. Coatings 2022, 12, 1330. https://doi.org/10.3390/coatings12091330
Behrangi S, Sedláček I, Štěrba J, Suková G, Czigány Z, Buršíková V, Souček P, Sochora V, Balázsi K, Vašina P. An Assessment of the Bactericidal and Virucidal Properties of ZrN-Cu Nanostructured Coatings Deposited by an Industrial PVD System. Coatings. 2022; 12(9):1330. https://doi.org/10.3390/coatings12091330
Chicago/Turabian StyleBehrangi, Sahand, Ivo Sedláček, Ján Štěrba, Gabriela Suková, Zsolt Czigány, Vilma Buršíková, Pavel Souček, Vjačeslav Sochora, Katalin Balázsi, and Petr Vašina. 2022. "An Assessment of the Bactericidal and Virucidal Properties of ZrN-Cu Nanostructured Coatings Deposited by an Industrial PVD System" Coatings 12, no. 9: 1330. https://doi.org/10.3390/coatings12091330
APA StyleBehrangi, S., Sedláček, I., Štěrba, J., Suková, G., Czigány, Z., Buršíková, V., Souček, P., Sochora, V., Balázsi, K., & Vašina, P. (2022). An Assessment of the Bactericidal and Virucidal Properties of ZrN-Cu Nanostructured Coatings Deposited by an Industrial PVD System. Coatings, 12(9), 1330. https://doi.org/10.3390/coatings12091330











