Antiviral Biodegradable Food Packaging and Edible Coating Materials in the COVID-19 Era: A Mini-Review
Abstract
:1. Introduction
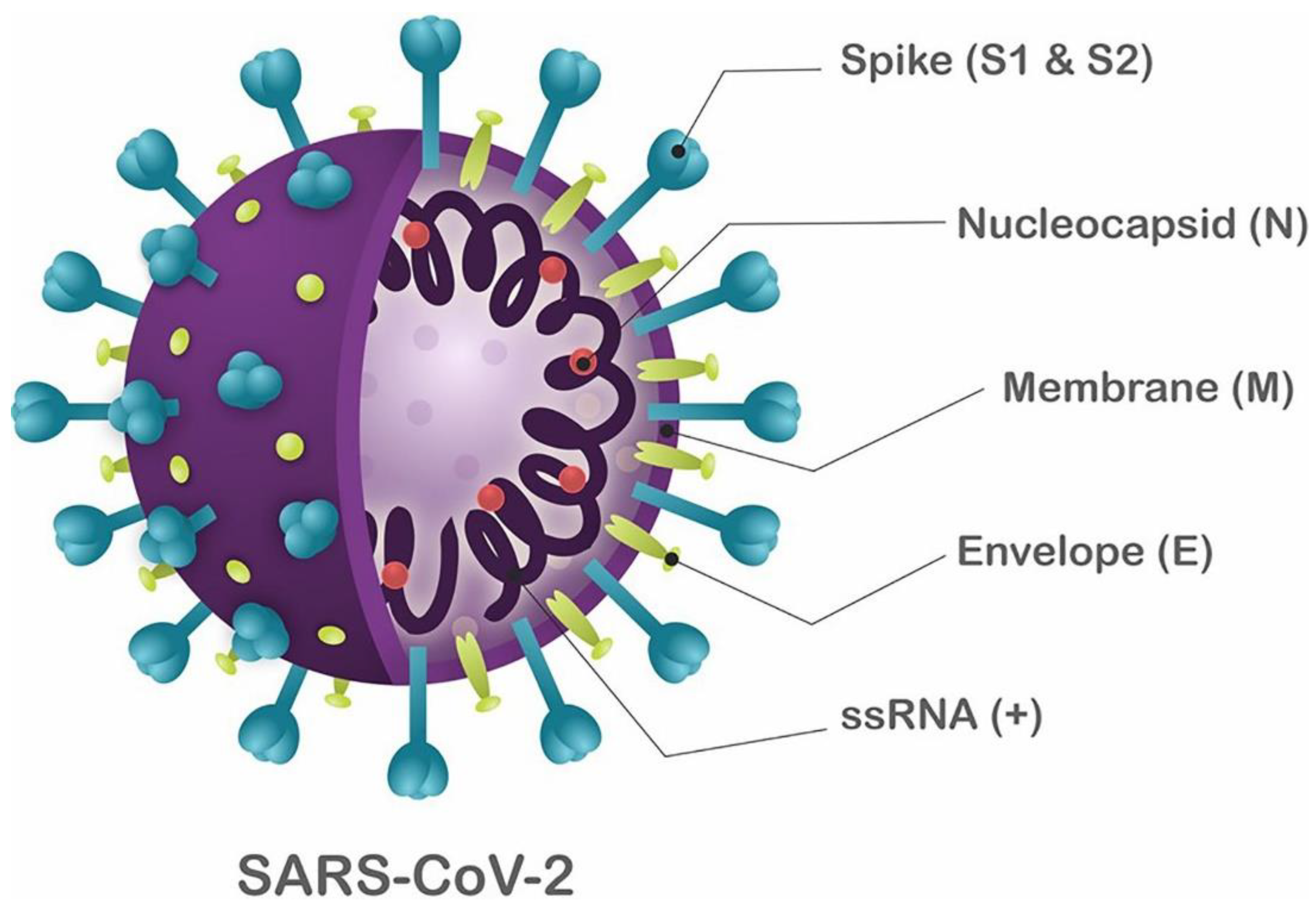
2. Virus Structure and Infection Mechanisms
3. Antiviral Materials and Mechanism of Action
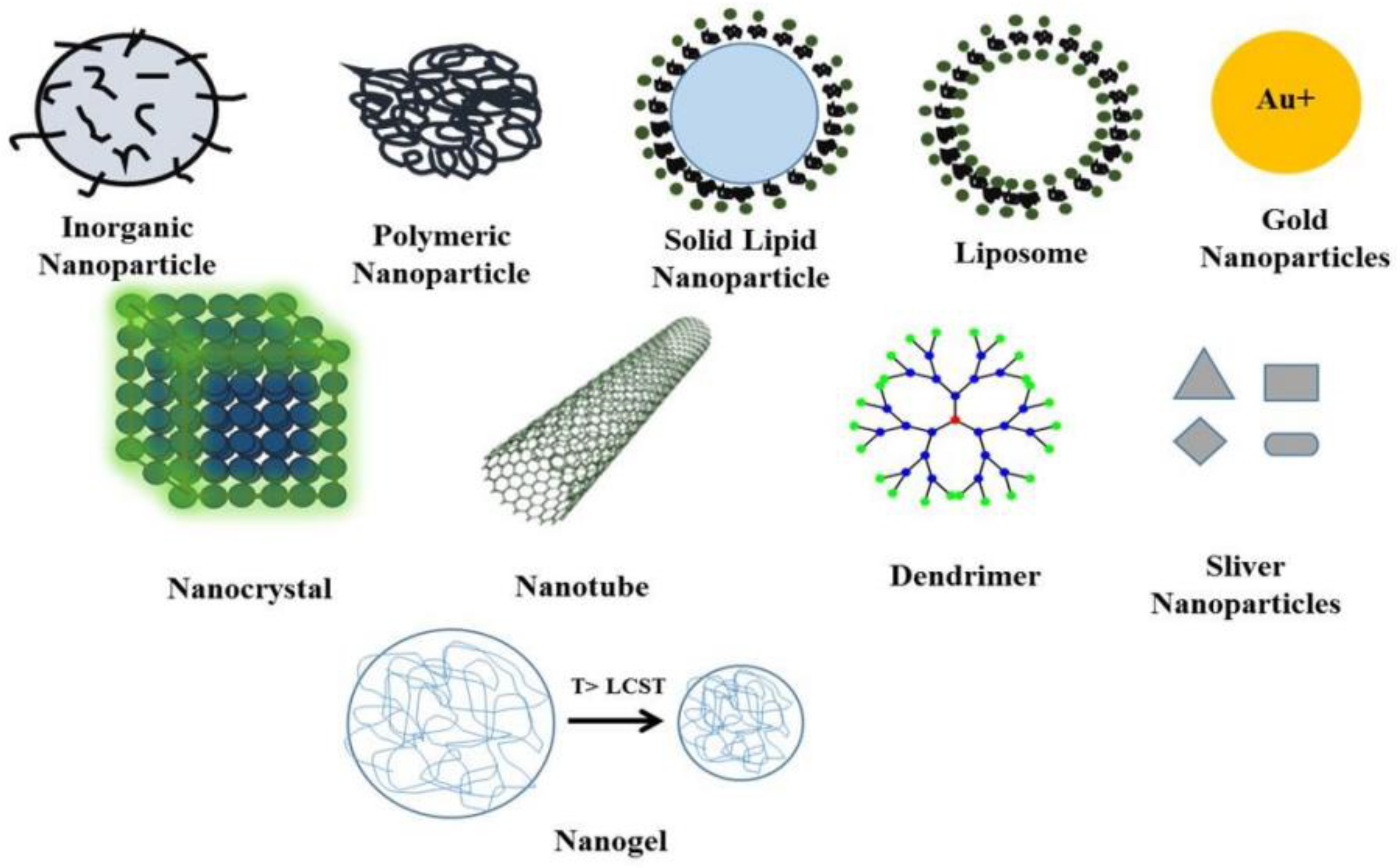
3.1. Nanomaterials
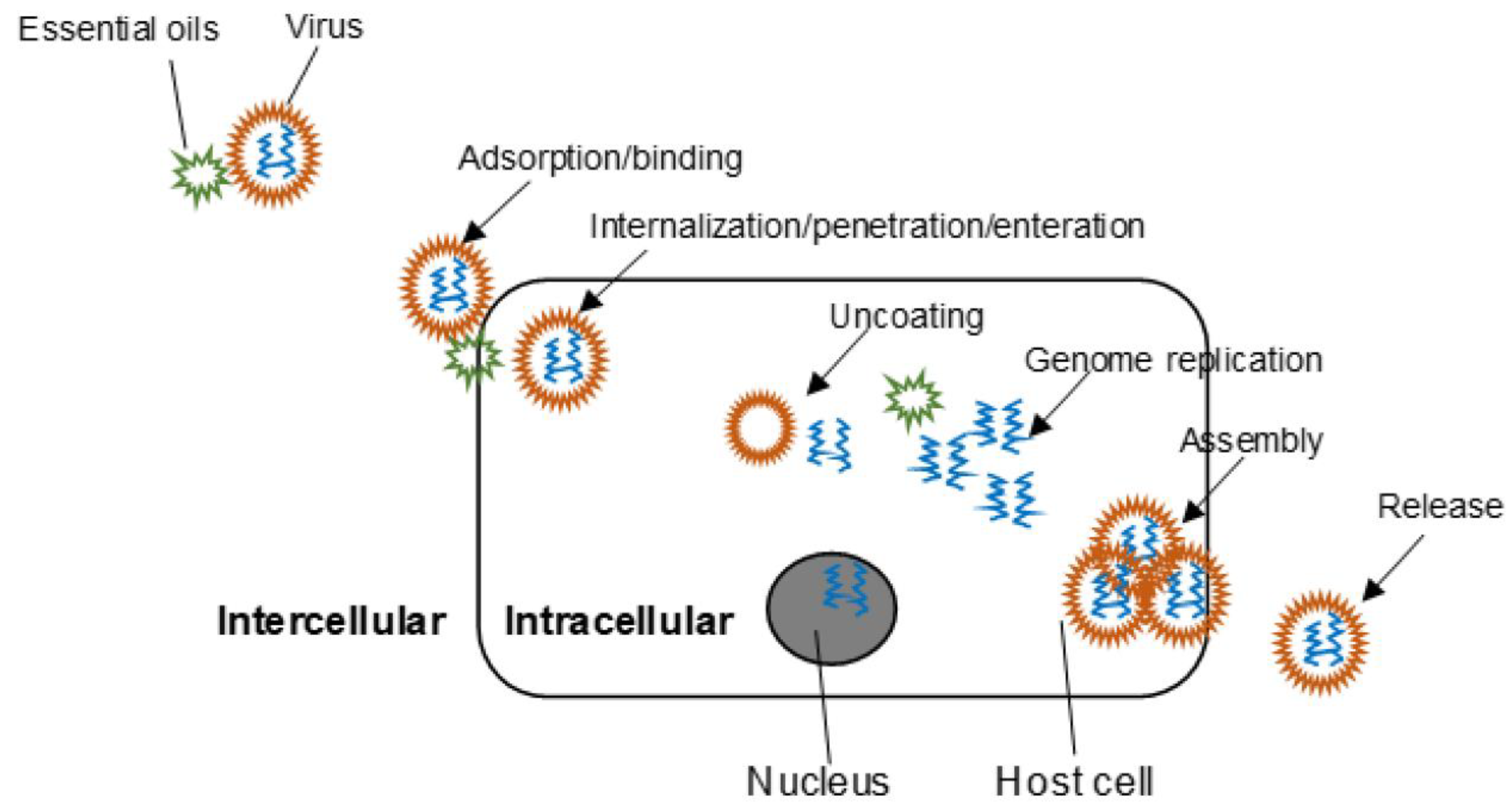
3.2. Natural Oils and Extracts
3.3. Biopolymers
4. Current Developments in Antiviral Food Packaging
| Components | Types of Packaging | Tested Against | Reference |
|---|---|---|---|
| Cinnamaldehyde Zein Polyhydroxybutyrate | Biodegradable multilayer system | Murine norovirus Feline calicivirus Hepatitis A virus | [94] |
| Chitosan Grape seed extract | Edible coatings and edible films | Murine norovirus (MNV-1) | [19] |
| ZnO Carvacrol Geraniol | Active external coating | Φ6 bacteriophage | [97] |
| Polyethylene film Rosemary, raspberry, and pomegranate extracts | Films covered with active coatings (functional food packaging) | Φ6 bacteriophage | [95,96] |
| Alginate Lipid Grape seed extract Green tea extract | Emulsified edible films | Murine norovirus Hepatitis A virus | [82] |
| Silver nanoparticles Silver nitrate Polyhydroxyalkanoates | Electrospun coating | Norovirus surrogates | [98] |
| Carrageenan Green tea extract | Edible coating | Murine norovirus Hepatitis A virus | [20] |
| Alginate Oleic acid Green tea extract | Edible coating | Human norovirus Hepatitis A virus | [102] |
| Persian gum Gelatin Allyl isothiocyanate | Antiviral edible coating | Murine norovirus | [103] |
5. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Wu, Y.-C.; Chen, C.-S.; Chan, Y.-J. The Outbreak of COVID-19: An Overview. J. Chin. Med. Assoc. 2020, 83, 217–220. [Google Scholar] [CrossRef] [PubMed]
- Zhou, P.; Yang, X.L.; Wang, X.G.; Hu, B.; Zhang, L.; Zhang, W.; Si, H.R.; Zhu, Y.; Li, B.; Huang, C.L.; et al. A Pneumonia Outbreak Associated with a New Coronavirus of Probable Bat Origin. Nature 2020, 579, 270–273. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kahn, J.S.; McIntosh, K. History and Recent Advances in Coronavirus Discovery. Pediatr. Infect. Dis. J. 2005, 24, S223–S227. [Google Scholar] [CrossRef]
- Yan, Y.; Shin, W.I.; Pang, Y.X.; Meng, Y.; Lai, J.; You, C.; Zhao, H.; Lester, E.; Wu, T.; Pang, C.H. The First 75 Days of Novel Coronavirus (SARS-CoV-2) Outbreak: Recent Advances, Prevention, and Treatment. Int. J. Environ. Res. Public Health 2020, 17, 2323. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De Oliveira, W.Q.; de Azeredo, H.M.C.; Neri-Numa, I.A.; Pastore, G.M. Food Packaging Wastes amid the COVID-19 Pandemic: Trends and Challenges. Trends Food Sci. Technol. 2021, 116, 1195–1199. [Google Scholar] [CrossRef] [PubMed]
- Klompas, M.; Baker, M.A.; Rhee, C. Airborne Transmission of SARS-CoV-2: Theoretical Considerations and Available Evidence. JAMA 2020, 324, 441–442. [Google Scholar] [CrossRef] [PubMed]
- Jyoti; Bhattacharya, B. Impact of COVID-19 in Food Industries and Potential Innovations in Food Packaging to Combat the Pandemic—A Review. Sci. Agropecu. 2021, 12, 133–140. [Google Scholar] [CrossRef]
- Fabra, M.J.; López-Rubio, A.; Lagaron, J.M. Biopolymers for Food Packaging Applications. In Smart Polymers And Their Applications; Woodhead Publishing: Sawston, UK, 2014; pp. 476–509. [Google Scholar] [CrossRef]
- Grujić, R.; Vujadinović, D.; Savanović, D. Biopolymers as Food Packaging Materials. In Advances In Applications of Industrial Biomaterials; Springer: Cham, Switzerland, 2017; pp. 139–160. [Google Scholar] [CrossRef]
- Adeyeye, O.A.; Sadiku, E.R.; Babu Reddy, A.; Ndamase, A.S.; Makgatho, G.; Sellamuthu, P.S.; Perumal, A.B.; Nambiar, R.B.; Fasiku, V.O.; Ibrahim, I.D.; et al. The Use of Biopolymers in Food Packaging. In Green Biopolymers and Their Nanocomposites; Springer: Singapore, 2019; pp. 137–158. [Google Scholar] [CrossRef]
- Porta, R.; Sabbah, M.; Di Pierro, P. Biopolymers as Food Packaging Materials. Int. J. Mol. Sci. 2020, 21, 4942. [Google Scholar] [CrossRef]
- Priyadarshi, R.; Roy, S.; Ghosh, T.; Biswas, D.; Rhim, J.-W. Antimicrobial Nanofillers Reinforced Biopolymer Composite Films for Active Food Packaging Applications—A Review. Sustain. Mater. Technol. 2021, e00353. [Google Scholar] [CrossRef]
- Priyadarshi, R.; Rhim, J.-W. Chitosan-Based Biodegradable Functional Films for Food Packaging Applications. Innov. Food Sci. Emerg. Technol. 2020, 62, 102346. [Google Scholar] [CrossRef]
- Shankar, S.; Rhim, J.-W. Antimicrobial Wrapping Paper Coated with a Ternary Blend of Carbohydrates (Alginate, Carboxymethyl Cellulose, Carrageenan) and Grapefruit Seed Extract. Carbohydr. Polym. 2018, 196, 92–101. [Google Scholar] [CrossRef] [PubMed]
- Balasubramaniam, B.; Prateek; Ranjan, S.; Saraf, M.; Kar, P.; Singh, S.P.; Thakur, V.K.; Singh, A.; Gupta, R.K. Antibacterial and Antiviral Functional Materials: Chemistry and Biological Activity toward Tackling COVID-19-like Pandemics. ACS Pharmacol. Transl. Sci. 2020, 4, 8–54. [Google Scholar] [CrossRef] [PubMed]
- Randazzo, W.; Fabra, M.J.; Falcó, I.; López-Rubio, A.; Sánchez, G. Polymers and Biopolymers with Antiviral Activity: Potential Applications for Improving Food Safety. Compr. Rev. Food Sci. Food Saf. 2018, 17, 754–768. [Google Scholar] [CrossRef] [Green Version]
- Sha-Tshibey Tshibangu, D.; Liyongo Inkoto, C.; T Tshibangu, D.S.; Matondo, A.; Lengbiye, E.M.; Inkoto, C.L.; Ngoyi, E.M.; Kabengele, C.N.; Bongo, G.N.; Gbolo, B.Z.; et al. Possible Effect of Aromatic Plants and Essential Oils against COVID-19: Review of Their Antiviral Activity. J. Complement. Altern. Med. Res. 2020, 11, 10–22. [Google Scholar] [CrossRef]
- Parham, S.; Kharazi, A.Z.; Bakhsheshi-Rad, H.R.; Nur, H.; Ismail, A.F.; Sharif, S.; Ramakrishna, S.; Berto, F. Antioxidant, Antimicrobial and Antiviral Properties of Herbal Materials. Antioxidants 2020, 9, 1309. [Google Scholar] [CrossRef] [PubMed]
- Amankwaah, C.; Li, J.; Lee, J.; Pascall, M.A. Development of Antiviral and Bacteriostatic Chitosan-Based Food Packaging Material with Grape Seed Extract for Murine Norovirus, Escherichia coli, and Listeria innocua Control. Food Sci. Nutr. 2020, 8, 6174–6181. [Google Scholar] [CrossRef]
- Falcó, I.; Randazzo, W.; Sánchez, G.; López-Rubio, A.; Fabra, M.J. On the Use of Carrageenan Matrices for the Development of Antiviral Edible Coatings of Interest in Berries. Food Hydrocoll. 2019, 92, 74–85. [Google Scholar] [CrossRef]
- Dung, T.T.N.; Nam, V.N.; Nhan, T.T.; Ngoc, T.T.B.; Minh, L.Q.; Nga, B.T.T.; Phan Le, V.; Quang, D.V. Silver Nanoparticles as Potential Antiviral Agents against African Swine Fever Virus. Mater. Res. Express 2020, 6, 1250g9. [Google Scholar] [CrossRef]
- Shu, F.; Jiang, B.; Yuan, Y.; Li, M.; Wu, W.; Jin, Y.; Xiao, H. Biological Activities and Emerging Roles of Lignin and Lignin-Based Products—A Review. Biomacromolecules 2021, 22, 4905–4918. [Google Scholar] [CrossRef]
- Santoyo, S.; Jaime, L.; García-Risco, M.R.; Lopez-Hazas, M.; Reglero, G. Supercritical Fluid Extraction as an Alternative Process to Obtain Antiviral Agents from Thyme Species. Ind. Crops Prod. 2014, 52, 475–480. [Google Scholar] [CrossRef]
- Schnitzler, P.; Schön, K.; Reichling, J. Antiviral Activity of Australian Tea Tree Oil and Eucalyptus Oil against Herpes Simplex Virus in Cell Culture. Pharmazie 2001, 56, 343–347. [Google Scholar] [PubMed]
- Vicidomini, C.; Roviello, V.; Roviello, G.N. Molecular Basis of the Therapeutical Potential of Clove (Syzygium aromaticum L.) and Clues to Its Anti-COVID-19 Utility. Molecules 2021, 26, 1880. [Google Scholar] [CrossRef] [PubMed]
- Brown, T.A. Gene Cloning and DNA Analysis: An Introduction; Brown, T.A., Ed.; Blackwell Science: Oxford, UK, 2001. [Google Scholar]
- Gelderblom, H.R. Structure and Classification of Viruses. In Medical Microbiology; Baron, S., Ed.; University of Texas Medical Branch at Galveston: Galveston, TX, USA, 1996; ISBN 0963117211. [Google Scholar]
- Zhu, N.; Zhang, D.; Wang, W.; Li, X.; Yang, B.; Song, J.; Zhao, X.; Huang, B.; Shi, W.; Lu, R.; et al. A Novel Coronavirus from Patients with Pneumonia in China, 2019. N. Engl. J. Med. 2020, 382, 727–733. [Google Scholar] [CrossRef] [PubMed]
- Zhong, N.S.; Zheng, B.J.; Li, Y.M.; Poon, L.L.M.; Xie, Z.H.; Chan, K.H.; Li, P.H.; Tan, S.Y.; Chang, Q.; Xie, J.P.; et al. Epidemiology and Cause of Severe Acute Respiratory Syndrome (SARS) in Guangdong, People’s Republic of China, in February 2003. Lancet 2003, 362, 1353–1358. [Google Scholar] [CrossRef] [Green Version]
- Zaki, A.M.; Van Boheemen, S.; Bestebroer, T.M.; Osterhaus, A.D.M.E.; Fouchier, R.A.M. Isolation of a Novel Coronavirus from a Man with Pneumonia in Saudi Arabia. N. Engl. J. Med. 2012, 367, 1814–1820. [Google Scholar] [CrossRef]
- Goldsmith, C.S.; Tatti, K.M.; Ksiazek, T.G.; Rollin, P.E.; Comer, J.A.; Lee, W.W.; Rota, P.A.; Bankamp, B.; Bellini, W.J.; Zaki, S.R. Ultrastructural Characterization of SARS Coronavirus. Emerg. Infect. Dis. 2004, 10, 320. [Google Scholar] [CrossRef]
- Santos, I.D.A.; Grosche, V.R.; Bergamini, F.R.G.; Sabino-Silva, R.; Jardim, A.C.G. Antivirals Against Coronaviruses: Candidate Drugs for SARS-CoV-2 Treatment? Front. Microbiol. 2020, 11, 1818. [Google Scholar] [CrossRef]
- Jazie, A.A.; Albaaji, A.J.; Abed, S.A. A Review on Recent Trends of Antiviral Nanoparticles and Airborne Filters: Special Insight on COVID-19 Virus. Air Qual. Atmos. Health 2021, 14, 1811–1824. [Google Scholar] [CrossRef]
- Gurunathan, S.; Qasim, M.; Choi, Y.; Do, J.T.; Park, C.; Hong, K.; Kim, J.H.; Song, H. Antiviral Potential of Nanoparticles—Can Nanoparticles Fight Against Coronaviruses? Nanomaterials 2020, 10, 1645. [Google Scholar] [CrossRef]
- Akbarzadeh, A.; Kafshdooz, L.; Razban, Z.; Dastranj Tbrizi, A.; Rasoulpour, S.; Khalilov, R.; Kavetskyy, T.; Saghfi, S.; Nasibova, A.N.; Kaamyabi, S.; et al. An Overview Application of Silver Nanoparticles in Inhibition of Herpes Simplex Virus. Artif. Cells Nanomed. Biotechnol. 2017, 46, 263–267. [Google Scholar] [CrossRef]
- Lin, Z.; Li, Y.; Guo, M.; Xu, T.; Wang, C.; Zhao, M.; Wang, H.; Chen, T.; Zhu, B. The Inhibition of H1N1 Influenza Virus-Induced Apoptosis by Silver Nanoparticles Functionalized with Zanamivir. RSC Adv. 2017, 7, 742–750. [Google Scholar] [CrossRef] [Green Version]
- Kim, J.; Yeom, M.; Lee, T.; Kim, H.O.; Na, W.; Kang, A.; Lim, J.W.; Park, G.; Park, C.; Song, D.; et al. Porous Gold Nanoparticles for Attenuating Infectivity of Influenza A Virus. J. Nanobiotechnol. 2020, 18, 1–11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, M.Y.; Yang, J.A.; Jung, H.S.; Beack, S.; Choi, J.E.; Hur, W.; Koo, H.; Kim, K.; Yoon, S.K.; Hahn, S.K. Hyaluronic Acid-Gold Nanoparticle/Interferon α Complex for Targeted Treatment of Hepatitis C Virus Infection. ACS Nano 2012, 6, 9522–9531. [Google Scholar] [CrossRef] [PubMed]
- Bai, Y.; Zhou, Y.; Liu, H.; Fang, L.; Liang, J.; Xiao, S. Glutathione-Stabilized Fluorescent Gold Nanoclusters Vary in Their Influences on the Proliferation of Pseudorabies Virus and Porcine Reproductive and Respiratory Syndrome Virus. ACS Appl. Nano Mater. 2018, 1, 969–976. [Google Scholar] [CrossRef]
- Priyadarshi, R.; Negi, Y.S. Effect of Varying Filler Concentration on Zinc Oxide Nanoparticle Embedded Chitosan Films as Potential Food Packaging Material. J. Polym. Environ. 2017, 25, 1087–1098. [Google Scholar] [CrossRef]
- Priyadarshi, R.; Kim, S.-M.; Rhim, J.-W. Carboxymethyl Cellulose-Based Multifunctional Film Combined with Zinc Oxide Nanoparticles and Grape Seed Extract for the Preservation of High-Fat Meat Products. Sustain. Mater. Technol. 2021, 29, e00325. [Google Scholar] [CrossRef]
- Ghaffari, H.; Tavakoli, A.; Moradi, A.; Tabarraei, A.; Bokharaei-Salim, F.; Zahmatkeshan, M.; Farahmand, M.; Javanmard, D.; Kiani, S.J.; Esghaei, M.; et al. Inhibition of H1N1 Influenza Virus Infection by Zinc Oxide Nanoparticles: Another Emerging Application of Nanomedicine. J. Biomed. Sci. 2019, 26, 1–10. [Google Scholar] [CrossRef]
- Tavakoli, A.; Ataei-Pirkooh, A.; Mm Sadeghi, G.; Bokharaei-Salim, F.; Sahrapour, P.; Kiani, S.J.; Moghoofei, M.; Farahmand, M.; Javanmard, D.; Monavari, S.H. Polyethylene Glycol-Coated Zinc Oxide Nanoparticle: An Efficient Nanoweapon to Fight against Herpes Simplex Virus Type 1. Nanomedicine 2018, 13, 2675–2690. [Google Scholar] [CrossRef]
- Ishida, T. Review on The Role of Zn2+ Ions in Viral Pathogenesis and the Effect of Zn2+ Ions for Host Cell-Virus Growth Inhibition. Am. J. Biomed. Sci. Res. 2019, 2, 28–37. [Google Scholar] [CrossRef] [Green Version]
- Tavakoli, A.; Hashemzadeh, M.S. Inhibition of Herpes Simplex Virus Type 1 by Copper Oxide Nanoparticles. J. Virol. Methods 2020, 275, 113688. [Google Scholar] [CrossRef]
- Mazurkow, J.M.; Yüzbasi, N.S.; Domagala, K.W.; Pfeiffer, S.; Kata, D.; Graule, T. Nano-Sized Copper (Oxide) on Alumina Granules for Water Filtration: Effect of Copper Oxidation State on Virus Removal Performance. Environ. Sci. Technol. 2020, 54, 1214–1222. [Google Scholar] [CrossRef] [PubMed]
- Merkl, P.; Long, S.; Mcinerney, G.M.; Sotiriou, G.A.; Ahonen, M.; Kogermann, K.; Pestryakov, A.; Mihailescu, I.N.; Papini, E. Antiviral Activity of Silver, Copper Oxide and Zinc Oxide Nanoparticle Coatings against SARS-CoV-2. Nanomaterials 2021, 11, 1312. [Google Scholar] [CrossRef] [PubMed]
- Sametband, M.; Kalt, I.; Gedanken, A.; Sarid, R. Herpes Simplex Virus Type-1 Attachment Inhibition by Functionalized Graphene Oxide. ACS Appl. Mater. Interfaces 2014, 6, 1228–1235. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.X.; Li, C.M.; Li, Y.F.; Wang, J.; Huang, C.Z. Synergistic Antiviral Effect of Curcumin Functionalized Graphene Oxide against Respiratory Syncytial Virus Infection. Nanoscale 2017, 9, 16086–16092. [Google Scholar] [CrossRef] [PubMed]
- Iannazzo, D.; Pistone, A.; Ferro, S.; De Luca, L.; Monforte, A.M.; Romeo, R.; Buemi, M.R.; Pannecouque, C. Graphene Quantum Dots Based Systems As HIV Inhibitors. Bioconjug. Chem. 2018, 29, 3084–3093. [Google Scholar] [CrossRef] [PubMed]
- Du, T.; Liang, J.; Dong, N.; Lu, J.; Fu, Y.; Fang, L.; Xiao, S.; Han, H. Glutathione-Capped Ag2S Nanoclusters Inhibit Coronavirus Proliferation through Blockage of Viral RNA Synthesis and Budding. ACS Appl. Mater. Interfaces 2018, 10, 4369–4378. [Google Scholar] [CrossRef]
- Pal, A.; Arshad, F.; Sk, M.P. Emergence of Sulfur Quantum Dots: Unfolding Their Synthesis, Properties, and Applications. Adv. Colloid Interface Sci. 2020, 285, 102274. [Google Scholar] [CrossRef]
- Priyadarshi, R.; Riahi, Z.; Rhim, J.-W.; Han, S.; Lee, S.-G. Sulfur Quantum Dots as Fillers in Gelatin/Agar-Based Functional Food Packaging Films. ACS Appl. Nano Mater. 2021, 4, 14292–14302. [Google Scholar] [CrossRef]
- Du, T.; Cai, K.; Han, H.; Fang, L.; Liang, J.; Xiao, S. Probing the Interactions of CdTe Quantum Dots with Pseudorabies Virus. Sci. Rep. 2015, 5, 1–10. [Google Scholar] [CrossRef]
- Łoczechin, A.; Séron, K.; Barras, A.; Giovanelli, E.; Belouzard, S.; Chen, Y.T.; Metzler-Nolte, N.; Boukherroub, R.; Dubuisson, J.; Szunerits, S. Functional Carbon Quantum Dots as Medical Countermeasures to Human Coronavirus. ACS Appl. Mater. Interfaces 2019, 11, 42964–42974. [Google Scholar] [CrossRef]
- Lin, C.J.; Chang, L.; Chu, H.W.; Lin, H.J.; Chang, P.C.; Wang, R.Y.; Unnikrishnan, B.; Mao, J.Y.; Chen, S.Y.; Huang, C.C. High Amplification of the Antiviral Activity of Curcumin through Transformation into Carbon Quantum Dots. Small 2019, 15, 1902641. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.; Gu, J.; Ye, J.; Fang, B.; Wan, S.; Wang, C.; Ashraf, U.; Li, Q.; Wang, X.; Shao, L.; et al. Benzoxazine Monomer Derived Carbon Dots as a Broad-Spectrum Agent to Block Viral Infectivity. J. Colloid Interface Sci. 2019, 542, 198–206. [Google Scholar] [CrossRef] [PubMed]
- De Souza E Silva, J.M.; Hanchuk, T.D.M.; Santos, M.I.; Kobarg, J.; Bajgelman, M.C.; Cardoso, M.B. Viral Inhibition Mechanism Mediated by Surface-Modified Silica Nanoparticles. ACS Appl. Mater. Interfaces 2016, 8, 16564–16572. [Google Scholar] [CrossRef] [PubMed]
- LaBauve, A.E.; Rinker, T.E.; Noureddine, A.; Serda, R.E.; Howe, J.Y.; Sherman, M.B.; Rasley, A.; Brinker, C.J.; Sasaki, D.Y.; Negrete, O.A. Lipid-Coated Mesoporous Silica Nanoparticles for the Delivery of the ML336 Antiviral to Inhibit Encephalitic Alphavirus Infection. Sci. Rep. 2018, 8, 13990. [Google Scholar] [CrossRef] [Green Version]
- Bimbo, L.M.; Denisova, O.V.; Mäkilä, E.; Kaasalainen, M.; De Brabander, J.K.; Hirvonen, J.; Salonen, J.; Kakkola, L.; Kainov, D.; Santos, H.A. Inhibition of Influenza A Virus Infection in vitro by Saliphenylhalamide-Loaded Porous Silicon Nanoparticles. ACS Nano 2013, 7, 6884–6893. [Google Scholar] [CrossRef]
- Jyothi, K.R.; Beloor, J.; Jo, A.; Nguyen, M.N.; Choi, T.G.; Kim, J.H.; Akter, S.; Lee, S.K.; Maeng, C.H.; Baik, H.H.; et al. Liver-Targeted Cyclosporine A-Encapsulated Poly (Lactic-Co-Glycolic) Acid Nanoparticles Inhibit Hepatitis C Virus Replication. Int. J. Nanomed. 2015, 10, 903. [Google Scholar] [CrossRef] [Green Version]
- Illescas, B.M.; Rojo, J.; Delgado, R.; Martín, N. Multivalent Glycosylated Nanostructures To Inhibit Ebola Virus Infection. J. Am. Chem. Soc. 2017, 139, 6018–6025. [Google Scholar] [CrossRef] [Green Version]
- Kandeel, M.; Al-Taher, A.; Park, B.K.; Kwon, H.J.; Al-Nazawi, M. A Pilot Study of the Antiviral Activity of Anionic and Cationic Polyamidoamine Dendrimers against the Middle East Respiratory Syndrome Coronavirus. J. Med. Virol. 2020, 92, 1665–1670. [Google Scholar] [CrossRef]
- Cagno, V.; Andreozzi, P.; D’Alicarnasso, M.; Jacob Silva, P.; Mueller, M.; Galloux, M.; Le Goffic, R.; Jones, S.T.; Vallino, M.; Hodek, J.; et al. Broad-Spectrum Non-Toxic Antiviral Nanoparticles with a Virucidal Inhibition Mechanism. Nat. Mater. 2018, 17, 195–203. [Google Scholar] [CrossRef]
- Donskyi, I.; Drüke, M.; Silberreis, K.; Lauster, D.; Ludwig, K.; Kühne, C.; Unger, W.; Böttcher, C.; Herrmann, A.; Dernedde, J.; et al. Interactions of Fullerene-Polyglycerol Sulfates at Viral and Cellular Interfaces. Small 2018, 14, 1800189. [Google Scholar] [CrossRef]
- Chen, L.; Liang, J. An Overview of Functional Nanoparticles as Novel Emerging Antiviral Therapeutic Agents. Mater. Sci. Eng. C 2020, 112, 110924. [Google Scholar] [CrossRef] [PubMed]
- Wani, A.R.; Yadav, K.; Khursheed, A.; Rather, M.A. An Updated and Comprehensive Review of the Antiviral Potential of Essential Oils and Their Chemical Constituents with Special Focus on Their Mechanism of Action against Various Influenza and Coronaviruses. Microb. Pathog. 2021, 152, 104620. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Yao, L. Antiviral Effects of Plant-Derived Essential Oils and Their Components: An Updated Review. Molecules 2020, 25, 2627. [Google Scholar] [CrossRef] [PubMed]
- Pang, J.; Wang, M.X.; Ang, I.Y.H.; Tan, S.H.X.; Lewis, R.F.; Chen, J.I.P.; Gutierrez, R.A.; Gwee, S.X.W.; Chua, P.E.Y.; Yang, Q.; et al. Potential Rapid Diagnostics, Vaccine and Therapeutics for 2019 Novel Coronavirus (2019-NCoV): A Systematic Review. J. Clin. Med. 2020, 9, 623. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Astani, A.; Reichling, J.; Schnitzler, P. Screening for Antiviral Activities of Isolated Compounds from Essential Oils. Evid. Based Complement. Altern. Med. 2011, 2011, 253643. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Salem, M.A.; Ezzat, S.M. The Use of Aromatic Plants and Their Therapeutic Potential as Antiviral Agents: A Hope for Finding Anti-COVID 19 Essential Oils. J. Essent. Oil Res. 2021, 33, 105–113. [Google Scholar] [CrossRef]
- Al-Harrasi, A.; Bhatia, S.; Behl, T.; Kaushik, D. Essential Oils in the Treatment of Respiratory Tract Infections. In Role of Essential Oils in the Management of COVID-19; CRC Press: Boca Raton, FL, USA, 2022; pp. 319–328. [Google Scholar] [CrossRef]
- Garzoli, S.; Mohamed, M.E.; Tawfeek, N.; Elbaramawi, S.S.; Fikry, E. Agathis Robusta Bark Essential Oil Effectiveness against COVID-19: Chemical Composition, In Silico and In Vitro Approaches. Plants 2022, 11, 663. [Google Scholar] [CrossRef]
- Badraoui, R.; Saoudi, M.; Hamadou, W.S.; Elkahoui, S.; Siddiqui, A.J.; Alam, J.M.; Jamal, A.; Adnan, M.; Suliemen, A.M.E.; Alreshidi, M.M.; et al. Antiviral Effects of Artemisinin and Its Derivatives against SARS-CoV-2 Main Protease: Computational Evidences and Interactions with ACE2 Allelic Variants. Pharmaceuticals 2022, 15, 129. [Google Scholar] [CrossRef]
- Cagno, V.; Sgorbini, B.; Sanna, C.; Cagliero, C.; Ballero, M.; Civra, A.; Donalisio, M.; Bicchi, C.; Lembo, D.; Rubiolo, P. In Vitro Anti-Herpes Simplex Virus-2 Activity of Salvia desoleana Atzei & V. Picci Essential Oil. PLoS ONE 2017, 12, e0172322. [Google Scholar] [CrossRef] [Green Version]
- Pourghanbari, G.; Nili, H.; Moattari, A.; Mohammadi, A.; Iraji, A. Antiviral Activity of the Oseltamivir and Melissa officinalis L. Essential Oil against Avian Influenza A Virus (H9N2). VirusDisease 2016, 27, 170–178. [Google Scholar] [CrossRef] [Green Version]
- Roy, S.; Priyadarshi, R.; Ezati, P.; Rhim, J.W. Curcumin and its uses in active and smart food packaging applications-A comprehensive review. Food Chem. 2021, 375, 131885. [Google Scholar] [CrossRef] [PubMed]
- Gilling, D.H.; Kitajima, M.; Torrey, J.R.; Bright, K.R. Antiviral Efficacy and Mechanisms of Action of Oregano Essential Oil and Its Primary Component Carvacrol against Murine Norovirus. J. Appl. Microbiol. 2014, 116, 1149–1163. [Google Scholar] [CrossRef] [PubMed]
- Vimalanathan, S.; Hudson, J. Anti-Influenza Virus Activity of Essential Oils and Vapors. Am. J. Essent. Oils Nat. Prod. 2014, 2, 47–53. [Google Scholar]
- Selvarani, V.; James, H. The Activity of Cedar Leaf Oil Vapor Against Respiratory Viruses: Practical Applications ARTICLE INFO ABSTRACT. J. Appl. Pharm. Sci. 2013, 3, 11. [Google Scholar] [CrossRef]
- Feriotto, G.; Marchetti, N.; Costa, V.; Beninati, S.; Tagliati, F.; Mischiati, C. Chemical Composition of Essential Oils from Thymus vulgaris, Cymbopogon citratus, and Rosmarinus officinalis, and Their Effects on the HIV-1 Tat Protein Function. Chem. Biodivers. 2018, 15, e1700436. [Google Scholar] [CrossRef]
- Fabra, M.J.; Falcó, I.; Randazzo, W.; Sánchez, G.; López-Rubio, A. Antiviral and Antioxidant Properties of Active Alginate Edible Films Containing Phenolic Extracts. Food Hydrocoll. 2018, 81, 96–103. [Google Scholar] [CrossRef]
- Bianculli, R.H.; Mase, J.D.; Schulz, M.D. Antiviral Polymers: Past Approaches and Future Possibilities. Macromolecules 2020, 53, 9158–9186. [Google Scholar] [CrossRef]
- Merigan, T.C.; Finkelstein, M.S. Interferon-Stimulating and in Vivo Antiviral Effects of Various Synthetic Anionic Polymers. Virology 1968, 35, 363–374. [Google Scholar] [CrossRef]
- Ito, M.; Baba, M.; Sato, A.; Pauwels, R.; De Clercq, E.; Shigeta, S. Inhibitory Effect of Dextran Sulfate and Heparin on the Replication of Human Immunodeficiency Virus (HIV) in Vitro. Antivir. Res. 1987, 7, 361–367. [Google Scholar] [CrossRef]
- Witvrouw, M.; De Clercq, E. Sulfated Polysaccharides Extracted from Sea Algae as Potential Antiviral Drugs. Gen. Pharmacol. Vasc. Syst. 1997, 29, 497–511. [Google Scholar] [CrossRef]
- Cermelli, C.; Cuoghi, A.; Scuri, M.; Bettua, C.; Neglia, R.G.; Ardizzoni, A.; Blasi, E.; Iannitti, T.; Palmieri, B. In vitro evaluation of antiviral and virucidal activity of a high molecular weight hyaluronic acid. Virol. J. 2011, 8, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Mallakpour, S.; Azadi, E.; Hussain, C.M. Recent Breakthroughs of Antibacterial and Antiviral Protective Polymeric Materials during COVID-19 Pandemic and after Pandemic: Coating, Packaging, and Textile Applications. Curr. Opin. Colloid Interface Sci. 2021, 55, 101480. [Google Scholar] [CrossRef] [PubMed]
- Priyadarshi, R.; Kim, H.-J.; Rhim, J.-W. Effect of Sulfur Nanoparticles on Properties of Alginate-Based Films for Active Food Packaging Applications. Food Hydrocoll. 2021, 110, 106155. [Google Scholar] [CrossRef]
- Saedi, S.; Shokri, M.; Priyadarshi, R.; Rhim, J.-W. Carrageenan-Based Antimicrobial Films Integrated with Sulfur-Coated Iron Oxide Nanoparticles (Fe3O4@SNP). ACS Appl. Polym. Mater. 2021, 3, 4913–4923. [Google Scholar] [CrossRef]
- Roy, S.; Priyadarshi, R.; Rhim, J.-W. Development of Multifunctional Pullulan/Chitosan-Based Composite Films Reinforced with ZnO Nanoparticles and Propolis for Meat Packaging Applications. Foods 2021, 10, 2789. [Google Scholar] [CrossRef]
- Basak, S.; Packirisamy, G. Nano-Based Antiviral Coatings to Combat Viral Infections. Nano-Struct. Nano-Objects 2020, 24, 100620. [Google Scholar] [CrossRef]
- Delumeau, L.V.; Asgarimoghaddam, H.; Alkie, T.; Jones, A.J.B.; Lum, S.; Mistry, K.; Aucoin, M.G.; Dewitte-Orr, S.; Musselman, K.P. Effectiveness of Antiviral Metal and Metal Oxide Thin-Film Coatings against Human Coronavirus 229E. APL Mater. 2021, 9, 111114. [Google Scholar] [CrossRef]
- Fabra, M.J.; Castro-Mayorga, J.L.; Randazzo, W.; Lagarón, J.M.; López-Rubio, A.; Aznar, R.; Sánchez, G. Efficacy of Cinnamaldehyde Against Enteric Viruses and Its Activity After Incorporation Into Biodegradable Multilayer Systems of Interest in Food Packaging. Food Environ. Virol. 2016, 8, 125–132. [Google Scholar] [CrossRef] [Green Version]
- Ordon, M.; Zdanowicz, M.; Nawrotek, P.; Stachurska, X.; Mizielińska, M. Polyethylene Films Containing Plant Extracts in the Polymer Matrix as Antibacterial and Antiviral Materials. Int. J. Mol. Sci. 2021, 22, 13438. [Google Scholar] [CrossRef]
- Ordon, M.; Nawrotek, P.; Stachurska, X.; Mizielińska, M. Polyethylene Films Coated with Antibacterial and Antiviral Layers Based on CO2 Extracts of Raspberry Seeds, of Pomegranate Seeds, and of Rosemary. Coatings 2021, 11, 1179. [Google Scholar] [CrossRef]
- Mizielińska, M.; Nawrotek, P.; Stachurska, X.; Ordon, M.; Bartkowiak, A. Packaging Covered with Antiviral and Antibacterial Coatings Based on ZnO Nanoparticles Supplemented with Geraniol and Carvacrol. Int. J. Mol. Sci. 2021, 22, 1717. [Google Scholar] [CrossRef] [PubMed]
- Castro-Mayorga, J.L.; Randazzo, W.; Fabra, M.J.; Lagaron, J.M.; Aznar, R.; Sánchez, G. Antiviral Properties of Silver Nanoparticles against Norovirus Surrogates and Their Efficacy in Coated Polyhydroxyalkanoates Systems. LWT—Food Sci. Technol. 2017, 79, 503–510. [Google Scholar] [CrossRef] [Green Version]
- Ghosh, T.; Mondal, K.; Giri, B.S.; Katiyar, V. Silk Nanodisc Based Edible Chitosan Nanocomposite Coating for Fresh Produces: A Candidate with Superior Thermal, Hydrophobic, Optical, Mechanical, and Food Properties. Food Chem. 2021, 360, 130048. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, T.; Das, D.; Katiyar, V. Edible Food Packaging: Targeted Biomaterials and Synthesis Strategies. In Nanotechnology in Edible Food Packaging; Springer: Singapore, 2021; pp. 25–63. [Google Scholar]
- Ghosh, T.; Katiyar, V. Edible Food Packaging: An Introduction BT—Nanotechnology in Edible Food Packaging: Food Preservation Practices for a Sustainable Future; Katiyar, V., Ghosh, T., Eds.; Springer: Singapore, 2021; pp. 1–23. ISBN 978-981-33-6169-0. [Google Scholar]
- Falcó, I.; Flores-Meraz, P.L.; Randazzo, W.; Sánchez, G.; López-Rubio, A.; Fabra, M.J. Antiviral Activity of Alginate-Oleic Acid Based Coatings Incorporating Green Tea Extract on Strawberries and Raspberries. Food Hydrocoll. 2019, 87, 611–618. [Google Scholar] [CrossRef] [Green Version]
- Sharif, N.; Falcó, I.; Martínez-Abad, A.; Sánchez, G.; López-Rubio, A.; Fabra, M.J. On the Use of Persian Gum for the Development of Antiviral Edible Coatings against Murine Norovirus of Interest in Blueberries. Polymers 2021, 13, 224. [Google Scholar] [CrossRef] [PubMed]

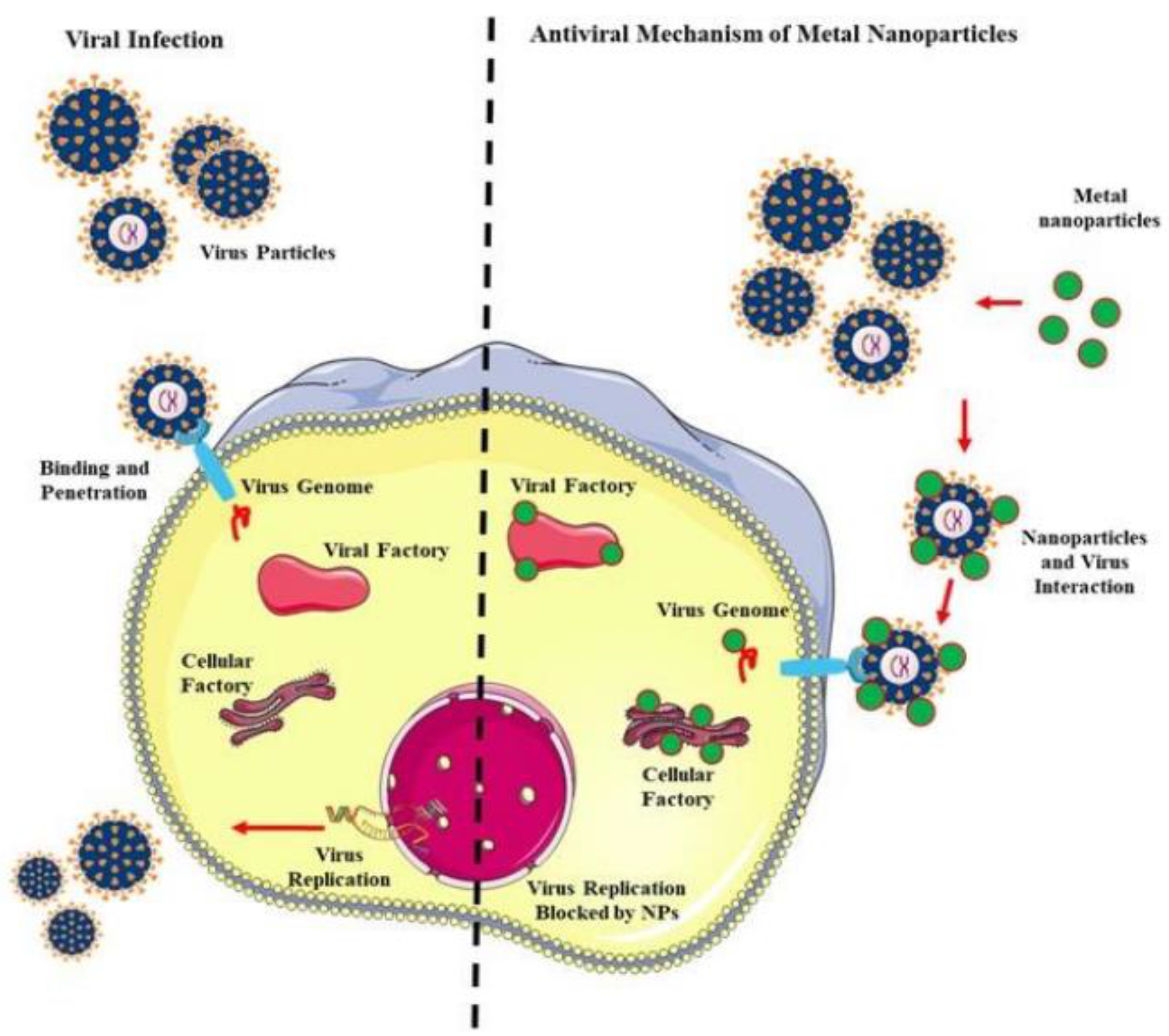

| Nanomaterial | Virus | Mechanism |
|---|---|---|
| Graphene oxide | Respiratory syncytial virus | Directly inactivates the virus and inhibits attachment |
| Nanogel | PRRSV | Shields attachment and penetration |
| Silver nanoparticle | Herpesvirus | Affects viral attachment |
| Graphene oxide | Herpesvirus | Attachment inhibition |
| Gold nanoparticles | Herpesvirus | Prevent viral attachment and penetration |
| Nanocarbon | Herpesvirus | Inhibits virus entry at the early stage |
| Silicon nanoparticles | Influenza A | Reduce the amount of progeny virus |
| Ag2S nanoclusters | Coronavirus | Block viral RNA synthesis and budding |
| Gd2O3:Tb3+/Er3+ nanoparticles | Zika virus | Antigen microcarriers for Zk2 peptide of ZIKV |
| Copper oxide nanoparticles | Herpes simplex virus type 1 | Oxidation of viral proteins and degradation of the viral genome |
| NiO nanostructures | Cucumber mosaic virus | Increase the expression of the pod, pr1, and pal1 genes |
| Zirconia nanoparticles | H5N1 influenza virus | Promote the expression of cytokines |
| Zinc oxide nanoparticles | H1N1 influenza virus | Inhibit virus only after viral entry into host cells |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Priyadarshi, R.; Purohit, S.D.; Roy, S.; Ghosh, T.; Rhim, J.-W.; Han, S.S. Antiviral Biodegradable Food Packaging and Edible Coating Materials in the COVID-19 Era: A Mini-Review. Coatings 2022, 12, 577. https://doi.org/10.3390/coatings12050577
Priyadarshi R, Purohit SD, Roy S, Ghosh T, Rhim J-W, Han SS. Antiviral Biodegradable Food Packaging and Edible Coating Materials in the COVID-19 Era: A Mini-Review. Coatings. 2022; 12(5):577. https://doi.org/10.3390/coatings12050577
Chicago/Turabian StylePriyadarshi, Ruchir, Shiv Dutt Purohit, Swarup Roy, Tabli Ghosh, Jong-Whan Rhim, and Sung Soo Han. 2022. "Antiviral Biodegradable Food Packaging and Edible Coating Materials in the COVID-19 Era: A Mini-Review" Coatings 12, no. 5: 577. https://doi.org/10.3390/coatings12050577
APA StylePriyadarshi, R., Purohit, S. D., Roy, S., Ghosh, T., Rhim, J.-W., & Han, S. S. (2022). Antiviral Biodegradable Food Packaging and Edible Coating Materials in the COVID-19 Era: A Mini-Review. Coatings, 12(5), 577. https://doi.org/10.3390/coatings12050577











