Measurements of Carbon Diffusivity and Surface Transfer Coefficient by Electrical Conductivity Relaxation during Carburization: Experimental Design by Theoretical Analysis
Abstract
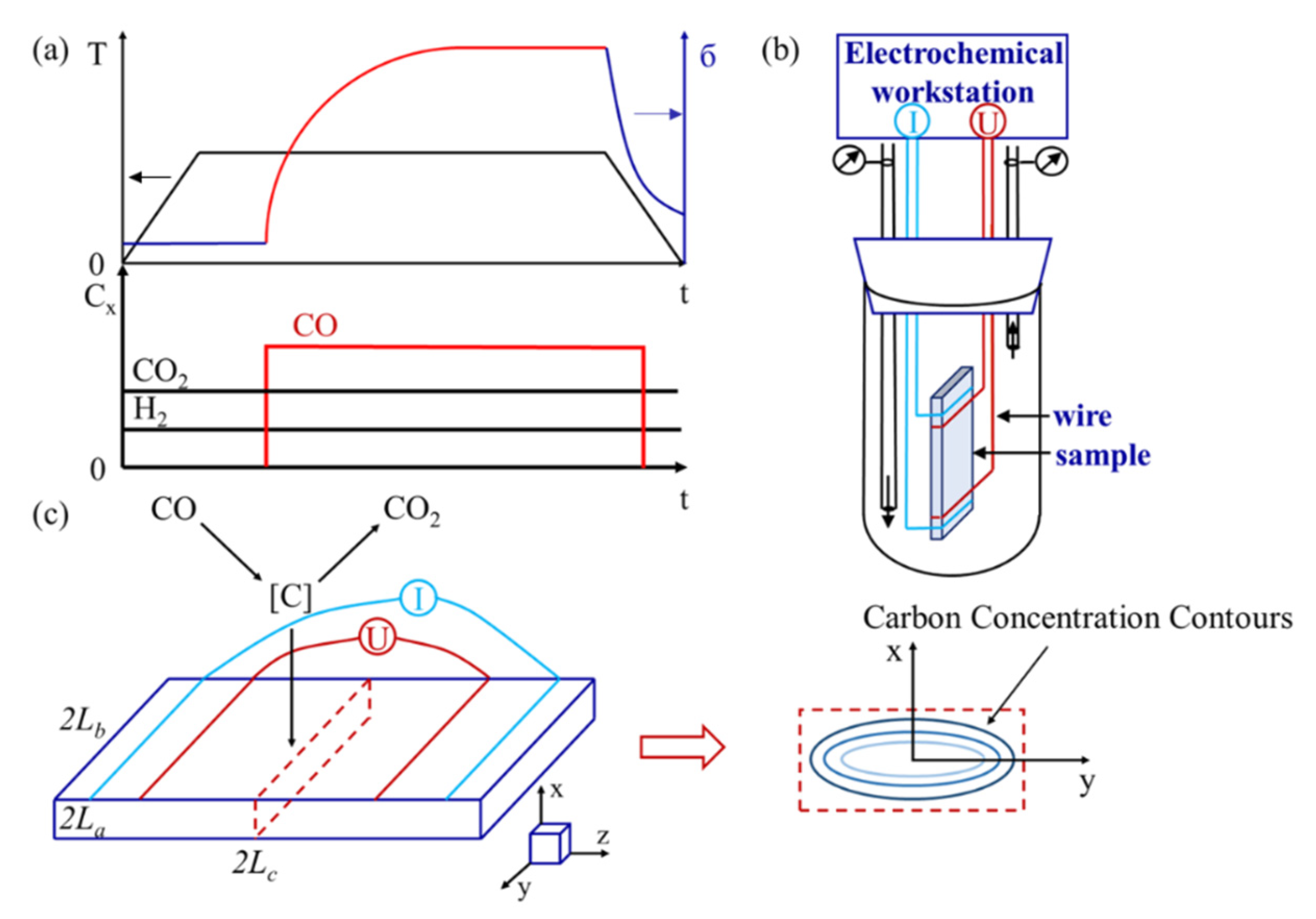
:1. Introduction
2. Modelling of Gas Carburization
3. Result and Discussion
3.1. The Influence of Width-to-Thickness Ratio
3.2. The Influence of Carbon Potential Build-Up Duration
4. Conclusions
- (1)
- The width-to-thickness ratio of sample in the experiment should be kept above 50, which can ensure the accuracy of the values of (β, D).
- (2)
- The duration of carbon potential build-up τCp should be as small as possible to ensure the accuracy of the diffusion coefficient D. If τCp is not small enough, a larger width-to-thickness ratio (more than 50) is proper with Bi = 1.
- (3)
- The ECR method shows the feasibility of measuring the values of (β, D) and even the value of τCp in practical experiments.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Thete, M.M. Runner-up Simulation of Gas Carburising: Development of Computer Program with Systematic Analyses of Process Variables Involved. Surf. Eng. 2003, 19, 217–228. [Google Scholar] [CrossRef]
- Moiseev, B.; Brunzel, Y.M.; Shvartsman, L. Kinetics of carburizing in an endothermal atmosphere. Met. Sci. Heat Treat. 1979, 21, 437–442. [Google Scholar] [CrossRef]
- Ruck, A.; Monceau, D.; Grabke, H.J. Effects of tramp elements Cu, P, Pb, Sb and Sn on the kinetics of carburization of case hardening steels. Steel Res. 1996, 67, 240–246. [Google Scholar] [CrossRef]
- Turpin, T.; Dulcy, J.; Gantois, M. Carbon diffusion and phase transformations during gas carburizing of high-alloyed stainless steels: Experimental study and theoretical modeling. Metall. Mater. Trans. A 2005, 36, 2751–2760. [Google Scholar] [CrossRef]
- Yan, M.; Liu, Z. The Mathematical Model of Surface Carbon Concentration Growth during Gas Carburization. Chin. J. Mater. Res. 1992, 6, 223–226. [Google Scholar]
- Karabelchtchikova, O.; Sisson, R.D. Carbon Diffusion in Steels: A Numerical Analysis Based on Direct Integration of the Flux. J. Phase Equilibria Diffus. 2006, 27, 598–604. [Google Scholar] [CrossRef]
- Allen, S.M.; Balluffi, R.W.; Carter, W.C. Kinetics of Materials; John Wiley & Sons: New York, NY, USA, 2005; pp. 99–114. ISBN 978-0-471-24689-3. [Google Scholar]
- Yan, M.; Liu, Z. Study on the distribution function of carbon concentration in the carburized layer of 20 steel during gas carburization in a multi-purpose furnace. Mater. Sci. Technol. 1997, 5, 3. [Google Scholar]
- Peng, Y.; Zhe, L.; Yong, J.; Bo, W.; Gong, J.; Somers, M. Experimental and numerical analysis of residual stress in carbon-stabilized expanded austenite. Scr. Mater. 2018, 157, 106–109. [Google Scholar] [CrossRef]
- Christiansen, T.L.; Somers, M. The Influence of Stress on Interstitial Diffusion—Carbon Diffusion Data in Austenite Revisited. Defect Diffus. Forum 2010, 297, 1408–1413. [Google Scholar] [CrossRef]
- Liu, Z.; Zhang, S.; Wang, S.; Feng, Y.; Peng, Y.; Gong, J.; Somers, M. Redistribution of carbon and residual stress in low-temperature gaseous carburized austenitic stainless steel during thermal and mechanical loading. Surf. Coat. Technol. 2021, 426, 127809. [Google Scholar] [CrossRef]
- Niessen, F.; Villa, M.; Danoix, F.; Hald, J.; Somers, M. In-situ analysis of redistribution of carbon and nitrogen during tempering of low interstitial martensitic stainless steel. Scr. Mater. 2018, 154, 216–219. [Google Scholar] [CrossRef]
- Yan, M. Mathematical Modeling and Computer Simulation of Gas Carburization and Rare Earth Carburization Processes. Ph.D. Thesis, Harbin Institute of Technology, Harbin, China, 1993. [Google Scholar]
- Hummelshoj, T.S.; Christiansen, T.; Somers, M. Determination of Concentration Dependent Diffusion Coefficients of Carbon in Expanded Austenite. Defect Diffus. Forum 2008, 273, 306–311. [Google Scholar] [CrossRef]
- Gao, W.; Long, J.M.; Kong, L.; Hodgson, P.D. Influence of the Geometry of an Immersed Steel Workpiece on Mass Transfer Coefficient in a Chemical Heat Treatment Fluidised Bed. ISIJ Int. 2004, 44, 869–877. [Google Scholar] [CrossRef]
- Bengtson, A. Quantitative depth profile analysis by glow discharge. Spectrochim. Acta Part B At. Spectrosc. 1994, 49, 411–429. [Google Scholar] [CrossRef]
- Angeli, J.; Bengtson, A.; Bogaerts, A.; Hoffmann, V.; Hodoroaba, V.D.; Steers, E. Glow discharge optical emission spectrometry: Moving towards reliable thin film analysis–A short review. J. Anal. At. Spectrom. 2003, 18, 670–679. [Google Scholar] [CrossRef]
- Shimizu, K.; Habazaki, H.; Skeldon, P.; Thompson, G.E.; Wood, G.C. Influence of argon pressure on the depth resolution during GDOES depth profiling analysis of thin films. Surf. Interface Anal. 2015, 29, 155–159. [Google Scholar] [CrossRef]
- Bellini, S.; Cilia, M.; Piccolo, E.L. Innovative sample preparation for GDOES analysis of decarburized layers in cylindrical metal specimens. Surf. Interface Anal. 2001, 31, 1100–1103. [Google Scholar] [CrossRef]
- Kilner, J.A. Measuring oxygen diffusion and oxygen surface exchange by conductivity relaxation. Solid State Ion. 2000, 136, 997–1001. [Google Scholar] [CrossRef]
- Yasuda, I.; Hishinuma, M. Electrical Conductivity and Chemical Diffusion Coefficient of Strontium-Doped Lanthanum Manganites. J. Solid State Chem. 1996, 123, 382–390. [Google Scholar] [CrossRef]
- Yeh, T.C.; Routbort, J.L.; Mason, T.O. Oxygen transport and surface exchange properties of Sr0.5Sm0.5CoO3-δ. Solid State Ion. 2013, 232, 138–143. [Google Scholar] [CrossRef]
- Chen, D.; Shao, Z. Surface Exchange and Bulk Diffusion Properties of Ba0.5Sr0.5Co0.8Fe0.2O3-δ Mixed Conductor. Int. J. Hydrogen Energy 2011, 36, 6948–6956. [Google Scholar] [CrossRef]
- Grimaud, A.; Bassat, J.M.; Mauvy, F.; Pollet, M.; Wattiaux, A.; Marrony, M.; Grenier, J.C. Oxygen reduction reaction of PrBaCo2-xFexO5+δ compounds as H+-SOFC cathodes: Correlations with physical properties. J. Mater. Chem. A 2014, 2, 3594–3604. [Google Scholar] [CrossRef]
- Lei, Z.; Liu, Y.; Zhang, Y.; Xiao, G.; Chen, F.; Xia, C. Enhancement in surface exchange coefficient and electrochemical performance of Sr2Fe1.5Mo0.5O6 electrodes by Ce0.8Sm0.2O1.9 nanoparticles. Electrochem. Commun. 2011, 13, 711–713. [Google Scholar] [CrossRef]
- Egger, A.; Bucher, E.; Sitte, W. Oxygen Exchange Kinetics of the IT-SOFC Cathode Material Nd2NiO4+δ and Comparison with La0.6Sr0.4CoO3-δ. J. Electrochem. Soc. 2015, 158, 573–579. [Google Scholar] [CrossRef]
- Gopal, C.B.; Haile, S.M. An electrical conductivity relaxation study of oxygen transport in samarium doped ceria. J. Mater. Chem. A 2014, 2, 2405–2417. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Yan, F.; Zhang, Y.; Xu, Y.; Yan, M. Impedance spectrum model for in situ characterization of the kinetic equation parameters of vacuum thermal expansion. J. Mater. Heat Treat. 2020, 41, 129–134. [Google Scholar] [CrossRef]
- Magar, H.S.; Hassan, R.Y.; Mulchandani, A. Electrochemical impedance spectroscopy (EIS): Principles, construction, and biosensing applications. Sensors 2021, 21, 6578. [Google Scholar] [CrossRef]
- Crank, B.J. The Mathematics of Diffusion, 2nd ed.; Oxford University Press: New York, NY, USA, 1975; pp. 60–61. ISBN 978-01-9853-411-2. [Google Scholar]
- Ciucci, F. Electrical conductivity relaxation measurements: Statistical investigations using sensitivity analysis, optimal experimental design and ECRTOOLS. Solid State Ion. 2013, 239, 28–40. [Google Scholar] [CrossRef]
- Boukamp, B.A.; den Otter, M.W.; Bouwmeester, H.J.M. Transport processes in mixed conducting oxides: Combining time domain experiments and frequency domain analysis. J. Solid State Electrochem. 2004, 8, 592–598. [Google Scholar] [CrossRef]
- Otter, M.D.; Bouwmeester, H.; Boukamp, B.A.; Verweij, H. Reactor Flush Time Correction in Relaxation Experiments. J. Electrochem. Soc. 2001, 148, J1. [Google Scholar] [CrossRef]









| Bi | τCp (s) | β (cm/s) | D (cm2/s) | La (cm) | Lb (cm) | Lb/La |
|---|---|---|---|---|---|---|
| 0.01 | 0 | 1 × 10−6 | 1 × 10−8 | 1 × 10−4 | 1 × 10−4~1 × 10−2 | 1, 5, 10, 50, 100 |
| 1 | 1 × 10−2 | 1 × 10−2~1 | ||||
| 100 | 1 | 1~100 |
| Bi | β (cm/s) | D (cm2/s) | La (cm) | Lb (cm) | Lb/La | τCp (s) |
|---|---|---|---|---|---|---|
| 1 | 1 × 10−4 | 1 × 10−6 | 1 × 10−2 | 5 × 10−1 | 50 | 0 |
| 10 | ||||||
| 50 | ||||||
| 100 | ||||||
| 500 | ||||||
| 1000 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ma, W.; Sheng, J.; Wang, Y.; Yan, M.; Wu, Y.; Qin, S.; Zhou, X.; Zhang, Y. Measurements of Carbon Diffusivity and Surface Transfer Coefficient by Electrical Conductivity Relaxation during Carburization: Experimental Design by Theoretical Analysis. Coatings 2022, 12, 1886. https://doi.org/10.3390/coatings12121886
Ma W, Sheng J, Wang Y, Yan M, Wu Y, Qin S, Zhou X, Zhang Y. Measurements of Carbon Diffusivity and Surface Transfer Coefficient by Electrical Conductivity Relaxation during Carburization: Experimental Design by Theoretical Analysis. Coatings. 2022; 12(12):1886. https://doi.org/10.3390/coatings12121886
Chicago/Turabian StyleMa, Wenbo, Jianjun Sheng, Yiheng Wang, Mufu Yan, Yujian Wu, Shaohua Qin, Xiaoliang Zhou, and Yanxiang Zhang. 2022. "Measurements of Carbon Diffusivity and Surface Transfer Coefficient by Electrical Conductivity Relaxation during Carburization: Experimental Design by Theoretical Analysis" Coatings 12, no. 12: 1886. https://doi.org/10.3390/coatings12121886





