Robust Superhydrophobic Coatings for Enhanced Corrosion Resistance and Dielectric Properties
Abstract
1. Introduction
2. Experiment
2.1. Materials
2.2. Preparation of the Superhydrophobic Coating
2.3. Characterization
2.4. Robust Superhydrophobicity Test
2.5. Corrosion Resistance Test
2.6. Dielectric Performance Tests
3. Results and Discussion
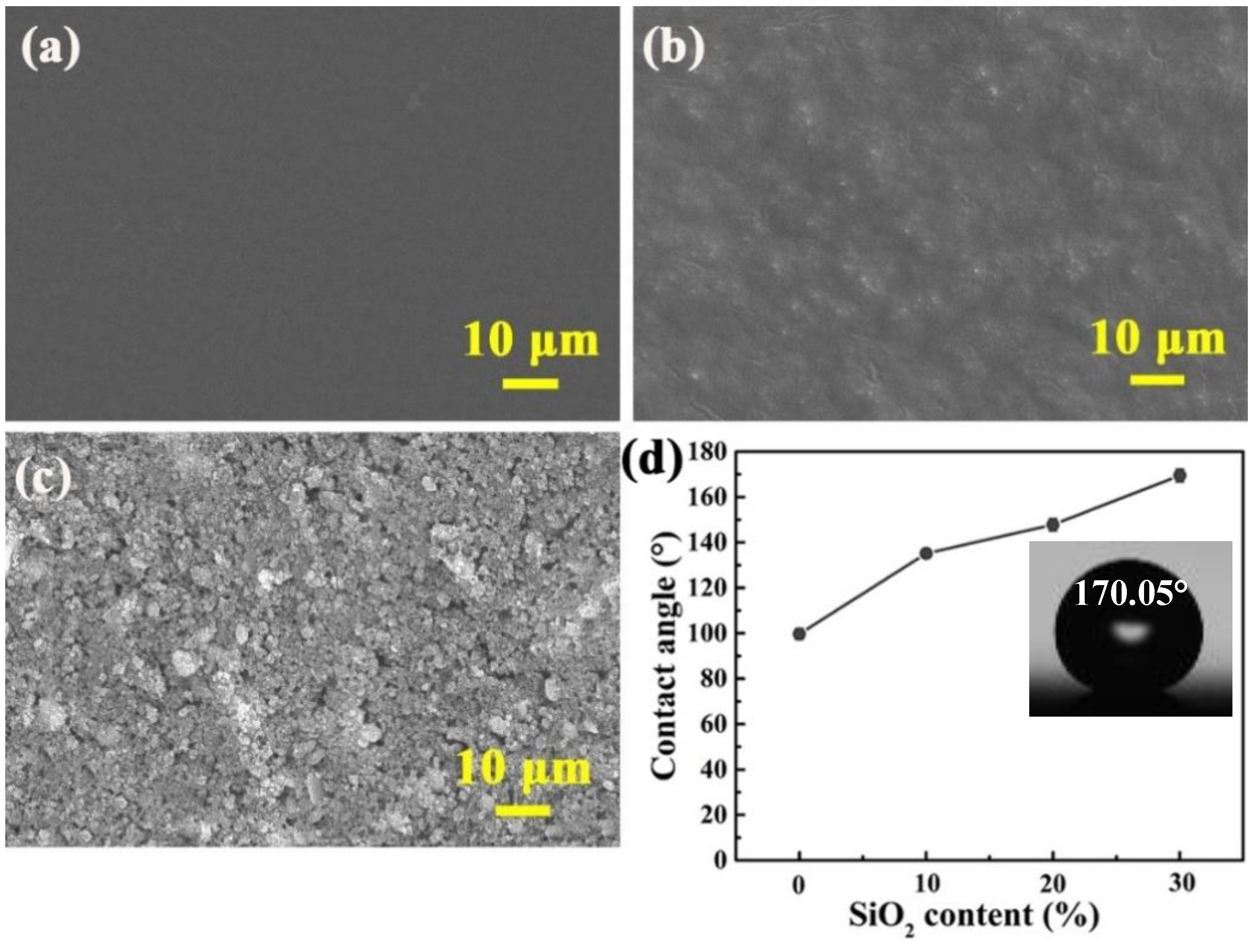
3.1. Surface Morphology and Wettability
3.2. Corrosion Resistance
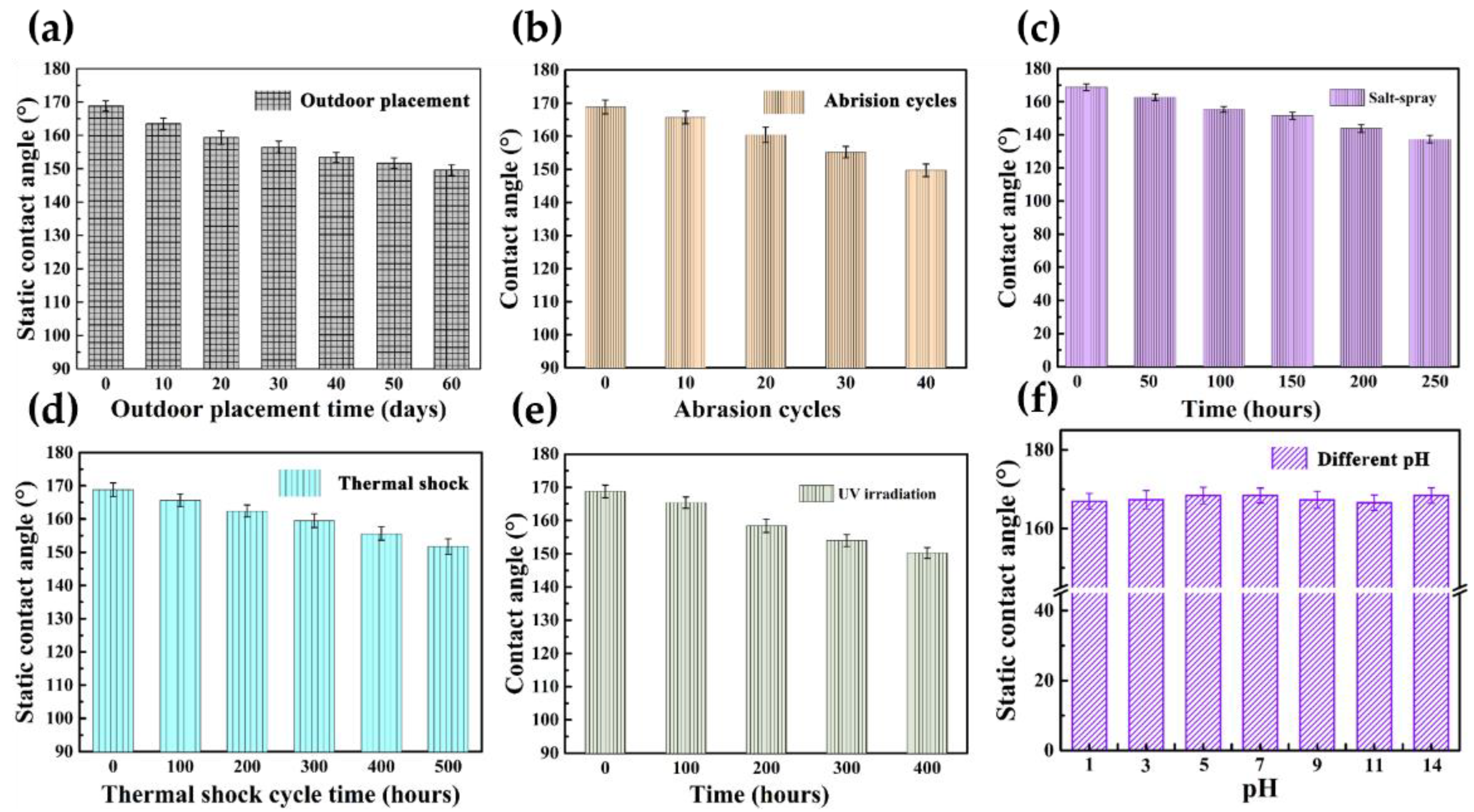
3.3. Environmental Suitability
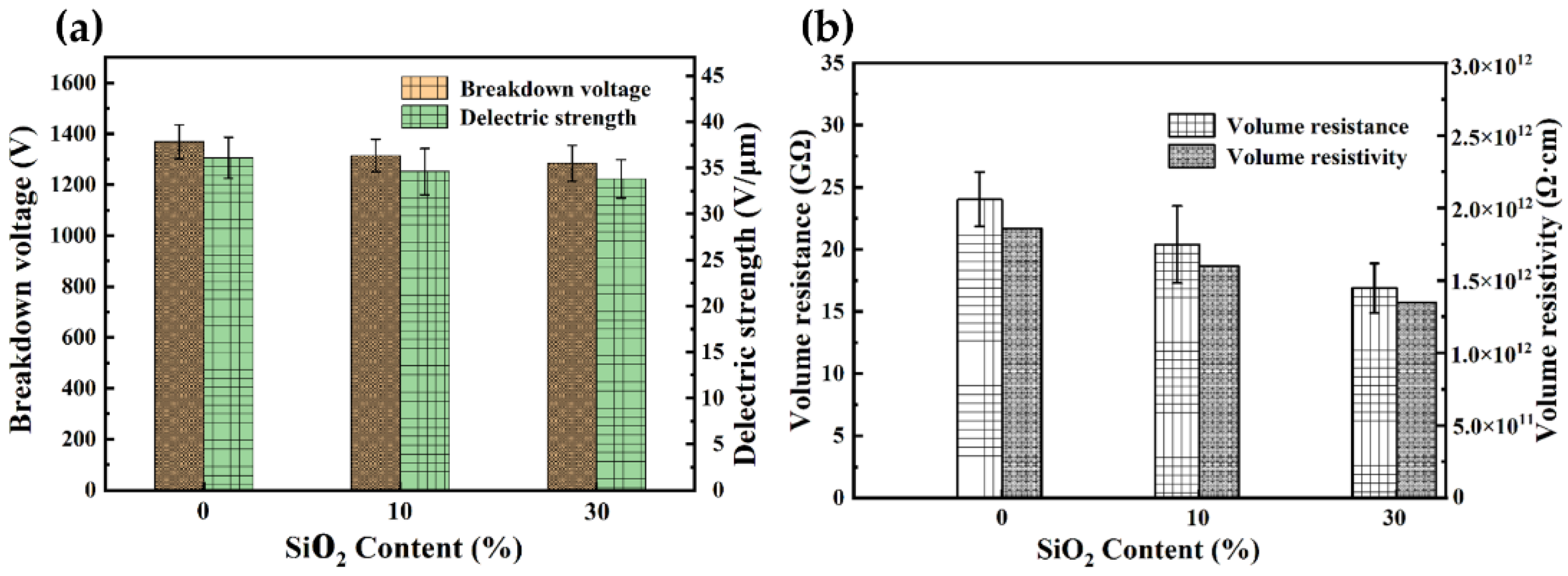
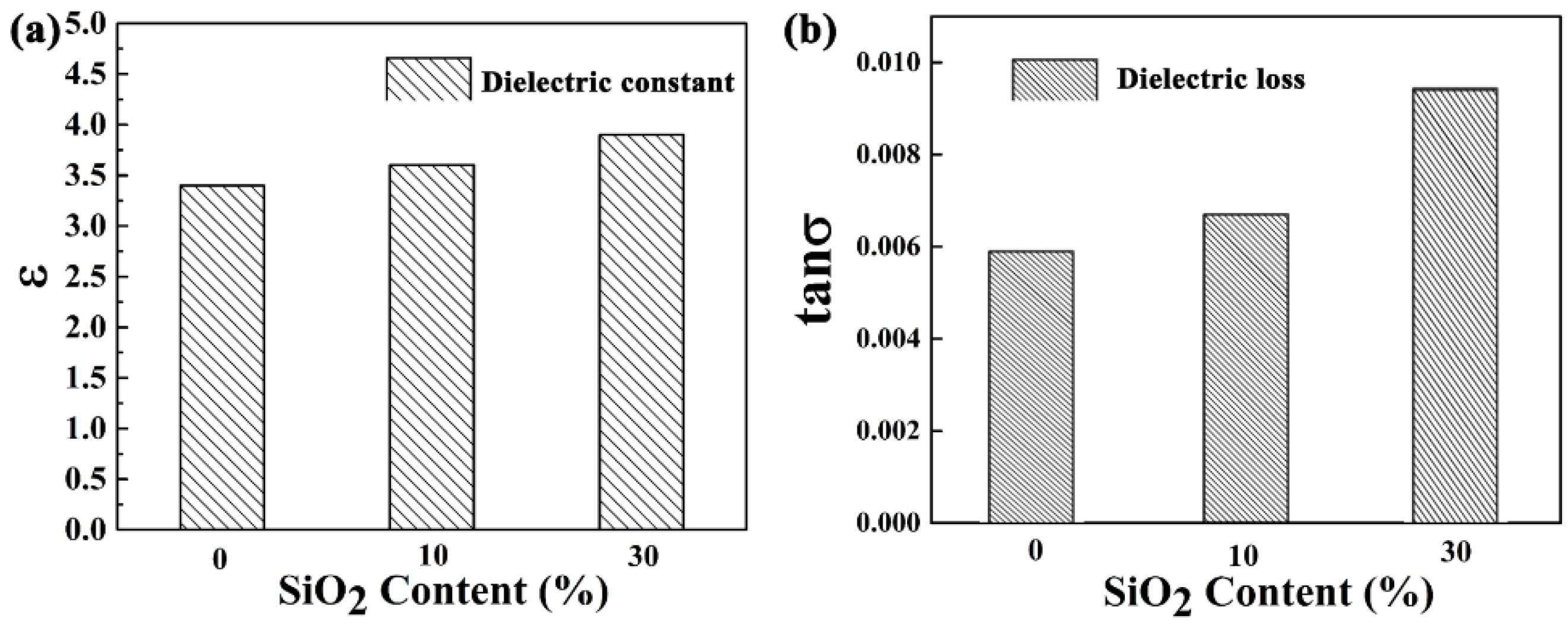
3.4. Dielectric Properties
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Yan, L.K.; Hou, Y.X. The optimization strategies and application of transmission network in power system. Electric Power Info. Commun. Technol. 2017, 107–112. [Google Scholar]
- Kaneto, K. Carrier transports in organic materials related to functional nanometric interface controlled electronic (NICE) devices. Thin Solid Film. 2001, 393, 249–258. [Google Scholar] [CrossRef]
- Gao, Y.; MacKenzie, R.C.I.; Liu, Y.; Xu, B.; van Loosdrecht, P.H.M.; Tian, W. Organic Electronics: Engineering Ultra Long Charge Carrier Lifetimes in Organic Electronic Devices at Room Temperature. Adv. Mater. Interfaces 2015, 2, 1400555. [Google Scholar] [CrossRef]
- Lin, C.H.; Huang, C.M.; Wong, T.I.; Chang, H.C.; Juang, T.Y.; Su, W.C. High-Tg and low-dielectric epoxy thermosets based on a propargyl ether-containing phosphinated benzoxazine. J. Polym. Sci. Polym. Chem. 2014, 52, 1359–1367. [Google Scholar] [CrossRef]
- Lei, Y.; Wang, Q.; Huo, J. Fabrication of durable superhydrophobic coatings with hierarchical structure on inorganic radome materials. Ceram. Int. 2014, 40, 10907–10914. [Google Scholar] [CrossRef]
- Zhang, R.F.; Zhang, S.F. Formation of micro-arc oxidation coatings on AZ91HP magnesium alloys. Corros. Sci. 2009, 51, 2820–2825. [Google Scholar] [CrossRef]
- Yu, S.; Jia, R.L.; Zhang, T.; Wang, F.H.; Hou, J.; Zhang, H.X. Effect of Different Scale Precipitates on Corrosion Behavior of Mg-10Gd-3Y-0.4Zr Alloy. Acta Metall. Sin. Engl. 2019, 32, 433–442. [Google Scholar] [CrossRef]
- Wu, Y.F.; Wang, Y.M.; Jing, Y.B.; Zhuang, J.P.; Yan, J.L.; Shao, Z.K.; Jin, M.S.; Wu, C.J.; Zhou, Y. In vivo study of microarc oxidation coated biodegradable magnesium plate to heal bone fracture defect of 3 mm width. Colloid Surf. B 2017, 158, 147–156. [Google Scholar] [CrossRef]
- Wu, Y.; Wang, Y.; Tian, S.; Jing, Y.; Zhuang, J.; Guo, L.; Jia, D.; Zhou, Y. Hydrothermal fabrication of rGO/Apatite layers on AZ31 magnesium alloy for enhanced bonding strength and corrosion resistance. Appl. Surf. Sci. 2019, 470, 430–438. [Google Scholar] [CrossRef]
- Wang, S.Q.; Wang, Y.M.; Chen, J.C.; Zou, Y.C.; Ouyang, J.H.; Jia, D.C.; Zhou, Y. Simple and scalable synthesis of super-repellent multilayer nanocomposite coating on Mg alloy with mechanochemical robustness, high-temperature endurance and electric protection. J. Magnes. Alloy. 2022, 10, 2446–2459. [Google Scholar] [CrossRef]
- Xu, W.J.; Song, J.L.; Sun, J.; Lu, Y.; Yu, Z.Y. Rapid Fabrication of Large-Area, Corrosion-Resistant Superhydrophobic Mg Alloy Surfaces. ACS Appl. Mater. Interfaces 2011, 3, 4404–4414. [Google Scholar] [CrossRef] [PubMed]
- Boinovich, L.; Emelyanenko, A.M.; Modestov, A.D.; Domantovsky, A.G.; Emelyanenko, K.A. Synergistic effect of superhydrophobicity and oxidized layers on corrosion resistance of aluminum alloy surface textured by nanosecond laser treatment. ACS Appl. Mater. Interfaces 2015, 7, 150813132712001. [Google Scholar] [CrossRef] [PubMed]
- Boinovich, L.B.; Emelyanenko, A.M.; Modestov, A.D.; Domantovsky, A.G.; Shiryaev, A.A.; Emelyanenko, K.A.; Dvoretskaya, O.V.; Ganne, A.A. Corrosion behavior of superhydrophobic aluminum alloy in concentrated potassium halide solutions: When the specific anion effect is manifested. Corros. Sci. 2016, 112, 517–527. [Google Scholar] [CrossRef]
- Li, L.; Huang, T.; Lei, J.; He, J.; Qu, L.; Huang, P.; Zhou, W.; Li, N.; Pan, F. Robust biomimetic-structural superhydrophobic surface on aluminum alloy. ACS Appl. Mater. Interfaces 2015, 7, 1449–1457. [Google Scholar] [CrossRef]
- Narayanan, T.S.N.S.; Park, I.S.; Lee, M.H. Strategies to improve the corrosion resistance of microarc oxidation (MAO) coated magnesium alloys for degradable implants: Prospects and challenges. Prog. Mater. Sci. 2014, 60, 1–71. [Google Scholar] [CrossRef]
- Gu, X.N.; Li, N.; Zhou, W.R.; Zheng, Y.F.; Zhao, X.; Cai, Q.Z.; Ruan, L.Q. Corrosion resistance and surface biocompatibility of a microarc oxidation coating on a Mg-Ca alloy. Acta Biomater. 2011, 7, 1880–1889. [Google Scholar] [CrossRef]
- Wu, Y.F.; Wang, Y.M.; Zhao, D.W.; Zhang, N.; Li, H.Y.; Li, J.L.; Wang, Y.X.; Zhao, Y.; Yan, J.L.; Zhou, Y. In vivo study of microarc oxidation coated Mg alloy as a substitute for bone defect repairing: Degradation behavior, mechanical properties, and bone response. Colloid Surf. B 2019, 181, 349–359. [Google Scholar] [CrossRef]
- Wang, Y.M.; Wang, F.H.; Xu, M.J.; Zhao, B.; Guo, L.X.; Ouyang, J.H. Microstructure and corrosion behavior of coated AZ91 alloy by microarc oxidation for biomedical application. Appl. Surf. Sci. 2009, 255, 9124–9131. [Google Scholar] [CrossRef]
- Wu, Y.F.; Wang, Y.M.; Liu, H.; Liu, Y.; Guo, L.X.; Jia, D.C.; Ouyang, J.H.; Zhou, Y. The fabrication and hydrophobic property of micro-nano patterned surface on magnesium alloy using combined sparking sculpture and etching route. Appl. Surf. Sci. 2016, 389, 80–87. [Google Scholar] [CrossRef]
- Wang, H.Y.; Wei, Y.H.; Liang, M.M.; Hou, L.F.; Li, Y.G.; Guo, C.L. Fabrication of stable and corrosion-resisted super-hydrophobic film on Mg alloy. Colloid Surf. A 2016, 509, 351–358. [Google Scholar] [CrossRef]
- Saji, V.S. Recent progress in superhydrophobic and superamphiphobic coatings for magnesium and its alloys. J. Magnes. Alloy 2021, 9, 748–778. [Google Scholar] [CrossRef]
- Liang, M.M.; Wei, Y.H.; Hou, L.F.; Wang, H.Y.; Li, Y.G.; Guo, C.L. Fabrication of a super-hydrophobic surface on a magnesium alloy by a simple method. J. Alloys Compd. 2016, 656, 311–317. [Google Scholar] [CrossRef]
- Tian, X.; Verho, T.; Ras, R.H.A. Moving superhydrophobic surfaces toward real-world applications. Science 2016, 352, 142–143. [Google Scholar] [CrossRef] [PubMed]
- Yao, W.H.; Wu, L.; Huang, G.S.; Jiang, B.; Atrens, A.; Pan, F.S. Superhydrophobic coatings for corrosion protection of magnesium alloys. J. Mater. Sci. Technol. 2020, 52, 100–118. [Google Scholar] [CrossRef]
- Wang, S.Q.; Wang, Y.M.; Zou, Y.C.; Chen, G.L.; Ouyang, J.H.; Jia, D.C.; Zhou, Y. Scalable-Manufactured Superhydrophobic Multilayer Nanocomposite Coating with Mechanochemical Robustness and High-Temperature Endurance. ACS Appl. Mater. Interfaces 2020, 12, 35502–35512. [Google Scholar] [CrossRef]
- Seongmin, K.; Jae, H.H.; Handong, C.; Dukhyun, C.; Woonbong, H. Repeatable replication method with liquid infiltration to fabricate robust, flexible, and transparent, anti-reflective superhydrophobic polymer films on a large scale. Chem. Eng. J. 2018, 350, 225–232. [Google Scholar]
- Ou, J.; Hu, W.; Xue, M.; Wang, F.; Li, W. Superhydrophobic surfaces on light alloy substrates fabricated by a versatile process and their corrosion protection. ACS Appl. Mater. Interfaces 2013, 5, 3101–3107. [Google Scholar] [CrossRef]
- Long, M.; Peng, S.; Deng, W.; Yang, X.; Deng, W. Robust and thermal-healing superhydrophobic surfaces by spin-coating of polydimethylsiloxane. J. Colloid Interface Sci. 2017, 508, 18. [Google Scholar] [CrossRef]
- Wang, S.; Wang, Y.; Cao, G.; Chen, J.; Zou, Y.; Yang, B.; Ouyang, J.; Jia, D.; Zhou, Y. Highly reliable double-layer coatings on magnesium alloy surfaces for robust superhydrophobicity, chemical durability and electrical property. Ceram. Int. 2021, 47, 35037–35047. [Google Scholar] [CrossRef]
- Lu, S.; Chen, Y.; Xu, W.; Wei, L. Controlled growth of superhydrophobic films by sol-gel method on aluminum substrate. Appl. Surf. Sci. 2010, 256, 6072–6075. [Google Scholar] [CrossRef]
- Zhang, F.Z.; Zhao, L.L.; Chen, H.; Xu, S.; Evans, D.G.; Duan, X. Corrosion Resistance of Superhydrophobic Layered Double Hydroxide Films on Aluminum. Acta Metal. Sin. 2008, 47, 2466–2469. [Google Scholar]
- Liu, Y.; Liu, J.; Li, S.; Liu, J.; Han, Z.; Ren, L. Biomimetic Superhydrophobic Surface of High Adhesion Fabricated with Micronano Binary Structure on Aluminum Alloy. ACS Appl. Mater. Interfaces 2013, 5, 8907–8914. [Google Scholar] [CrossRef] [PubMed]
- Xu, N.; Sarkar, D.K.; Chen, X.G.; Tong, W.P. Corrosion performance of superhydrophobic nickel stearate/nickel hydroxide thin films on aluminum alloy by a simple one-step electrodeposition process. Surf. Coat. Technol. 2016, 302, 173–184. [Google Scholar] [CrossRef]
- Kannarpady, G.K.; Khedir, K.R.; Ishihara, H.; Woo, J.; Oshin, O.D.; Trigwell, S.; Ryerson, C.; Biris, A.S. Controlled Growth of Self-Organized Hexagonal Arrays of Metallic Nanorods Using Template-Assisted Glancing Angle Deposition for Superhydrophobic Applications. ACS Appl. Mater. Interfaces 2011, 3, 2332–2340. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.Q.; Wang, Y.M.; Zou, Y.C.; Wu, Y.F.; Chen, G.L.; Ouyang, J.H.; Jia, D.C.; Zhou, Y. A self-adjusting PTFE/TiO2 hydrophobic double-layer coating for corrosion resistance and electrical insulation. Chem. Eng. J. 2020, 402, 126116. [Google Scholar] [CrossRef]
- Zhu, H.Z.; Yue, L.F.; Zhuang, C. Fabrication and characterization of self-assembled graphene oxide/silane coatings for corrosion resistance. Surf. Coating. Technol. 2016, 304, 76–84. [Google Scholar] [CrossRef]
- Wang, S.Q.; Wang, Y.M.; Zhang, H.J.; Zhou, Y.C.; Chen, G.L.; Ouyang, J.H.; Jia, D.C.; Zhou, Y. Co-growing design of super-repellent dual-layer coating for multiple heat dissipation improvement. Chem. Eng. J. 2022, 427, 131701. [Google Scholar] [CrossRef]
- Liu, M.L.; Luo, Y.F.; Jia, D.M. A Robust and Versatile Continuous Super-Repellent Polymeric Film for Easy Repair and Underwater Display. ACS Appl. Mater. Interfaces 2020, 12, 6677–6687. [Google Scholar] [CrossRef]
- Peng, C.Y.; Chen, Z.Y.; Tiwari, M.K. All-organic superhydrophobic coatings with mechanochemical robustness and liquid impalement resistance. Nat. Mater. 2018, 17, 355–360. [Google Scholar] [CrossRef]
- Li, W.; Kang, Z. Fabrication of corrosion resistant superhydrophobic surface with self-cleaning property on magnesium alloy and its mechanical stability. Surf. Coatings Technol. 2014, 253, 205–213. [Google Scholar] [CrossRef]
- Shi, L.; Hu, J.; Lin, X.D.; Fang, L.; Wu, F.; Xie, J.; Meng, F.M. A robust superhydrophobic PPS-PTFE/SiO2 composite coating on AZ31 Mg alloy with excellent wear and corrosion resistance properties. J. Alloys Compd. 2017, 721, 157–163. [Google Scholar] [CrossRef]
- Liang, Y.; Ju, J.; Deng, N. Super-hydrophobic self-cleaning bead-like SiO2@ PTFE nanofiber membranes for waterproof-breathable applications. Appl. Surf. Sci. 2018, 442, 54–64. [Google Scholar] [CrossRef]
- Zang, D.M.; Zhu, R.W.; Zhang, W.; Yu, X.Q.; Lin, L.; Guo, X.L.; Liu, M.J.; Jiang, L. Corrosion-Resistant Superhydrophobic Coatings on Mg Alloy Surfaces Inspired by Lotus Seedpod. Adv. Funct. Mater. 2017, 27, 1605446. [Google Scholar] [CrossRef]
- Xie, J.; Hu, J.; Lin, X. Robust and anti-corrosive PDMS/SiO2 superhydrophobic coatings fabricated on magnesium alloys with different-sized SiO2 nanoparticles. Appl. Surf. Sci. 2018, 457, 870–880. [Google Scholar] [CrossRef]
- Li, H.Y.; Liu, G.; Liu, B.; Chen, W.; Chen, S.T. Dielectric properties of polyimide Al2O3 hybrids synthesized by in-situ polymerization. Mater. Lett. 2007, 61, 1507–1511. [Google Scholar] [CrossRef]
- Yang, D.; Huang, S.; Ruan, M.N.; Li, S.X.; Yang, J.W.; Wu, Y.B.; Guo, W.L.; Zhang, L.Q. Mussel Inspired Modification for Aluminum Oxide/Silicone Elastomer Composites with Largely Improved Thermal Conductivity and Low Dielectric Constant. Ind. Eng. Chem. Res. 2018, 57, 3255–3262. [Google Scholar] [CrossRef]
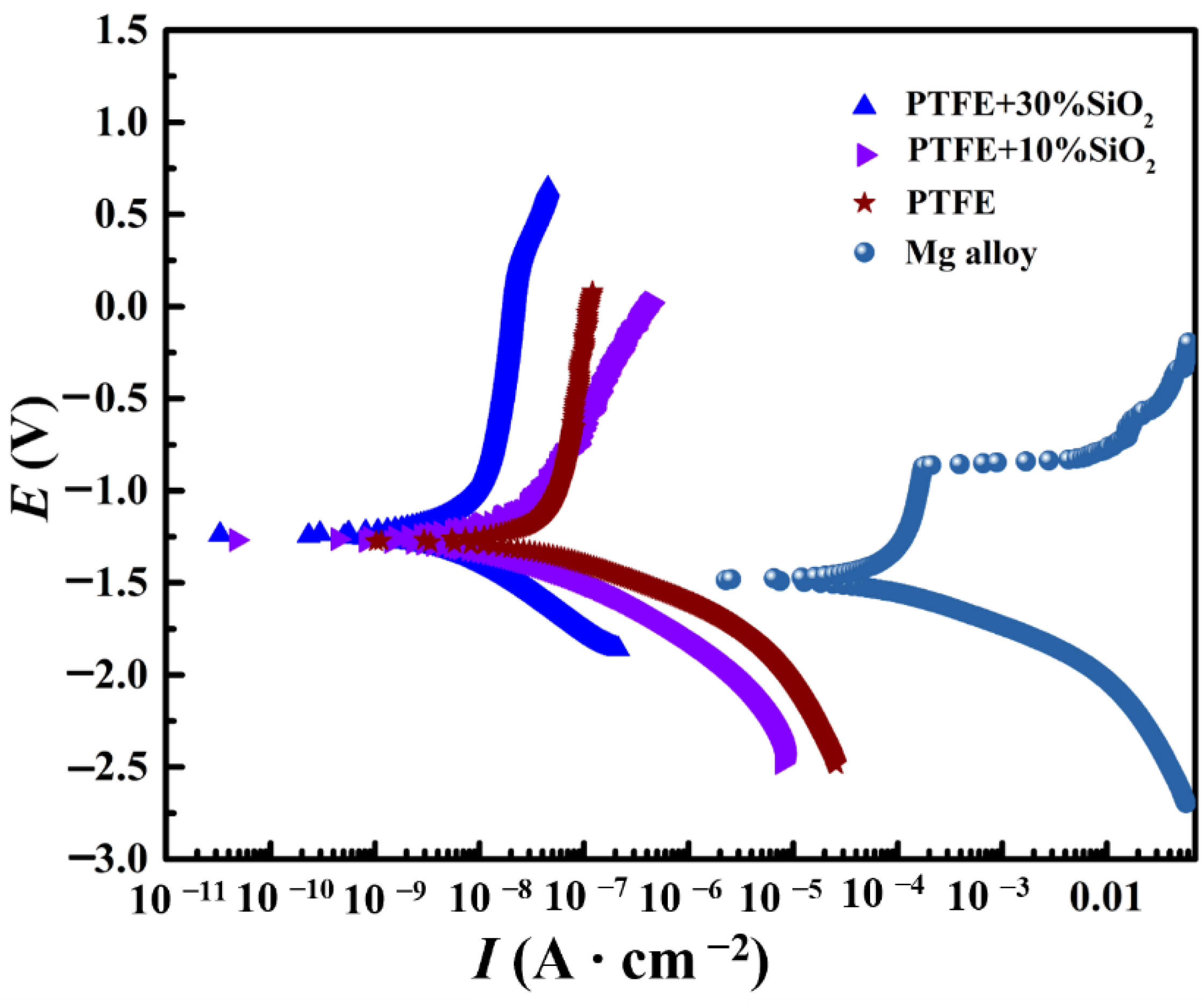


| Sample | Ecorr (V) | Icorr (A∙cm−2) | Rp (Ω∙cm−2) | η |
|---|---|---|---|---|
| AZ31 Mg | −1.483 | 3.47 × 10−5 | 1098.52 | - |
| PTFE | −1.293 | 7.89 × 10−8 | 5.84 × 105 | 97.69% |
| PTFE + 10%SiO2 | −1.257 | 7.66 × 10−8 | 5.99 × 105 | 97.79% |
| PTFE + 30%SiO2 | −1.229 | 3.39 × 10−8 | 1.20 × 106 | 99.02% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shao, W.; Kan, Q.; Bai, X.; Wang, C. Robust Superhydrophobic Coatings for Enhanced Corrosion Resistance and Dielectric Properties. Coatings 2022, 12, 1655. https://doi.org/10.3390/coatings12111655
Shao W, Kan Q, Bai X, Wang C. Robust Superhydrophobic Coatings for Enhanced Corrosion Resistance and Dielectric Properties. Coatings. 2022; 12(11):1655. https://doi.org/10.3390/coatings12111655
Chicago/Turabian StyleShao, Wentao, Qi Kan, Xinxin Bai, and Chengqian Wang. 2022. "Robust Superhydrophobic Coatings for Enhanced Corrosion Resistance and Dielectric Properties" Coatings 12, no. 11: 1655. https://doi.org/10.3390/coatings12111655
APA StyleShao, W., Kan, Q., Bai, X., & Wang, C. (2022). Robust Superhydrophobic Coatings for Enhanced Corrosion Resistance and Dielectric Properties. Coatings, 12(11), 1655. https://doi.org/10.3390/coatings12111655






