Magnesium Doped Hydroxyapatite-Based Coatings Obtained by Pulsed Galvanostatic Electrochemical Deposition with Adjustable Electrochemical Behavior
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of the Coatings
2.2. Characterization of Coatings
3. Results and Discussion
3.1. Morphology
3.2. Elemental Composition
3.3. Phase Composition
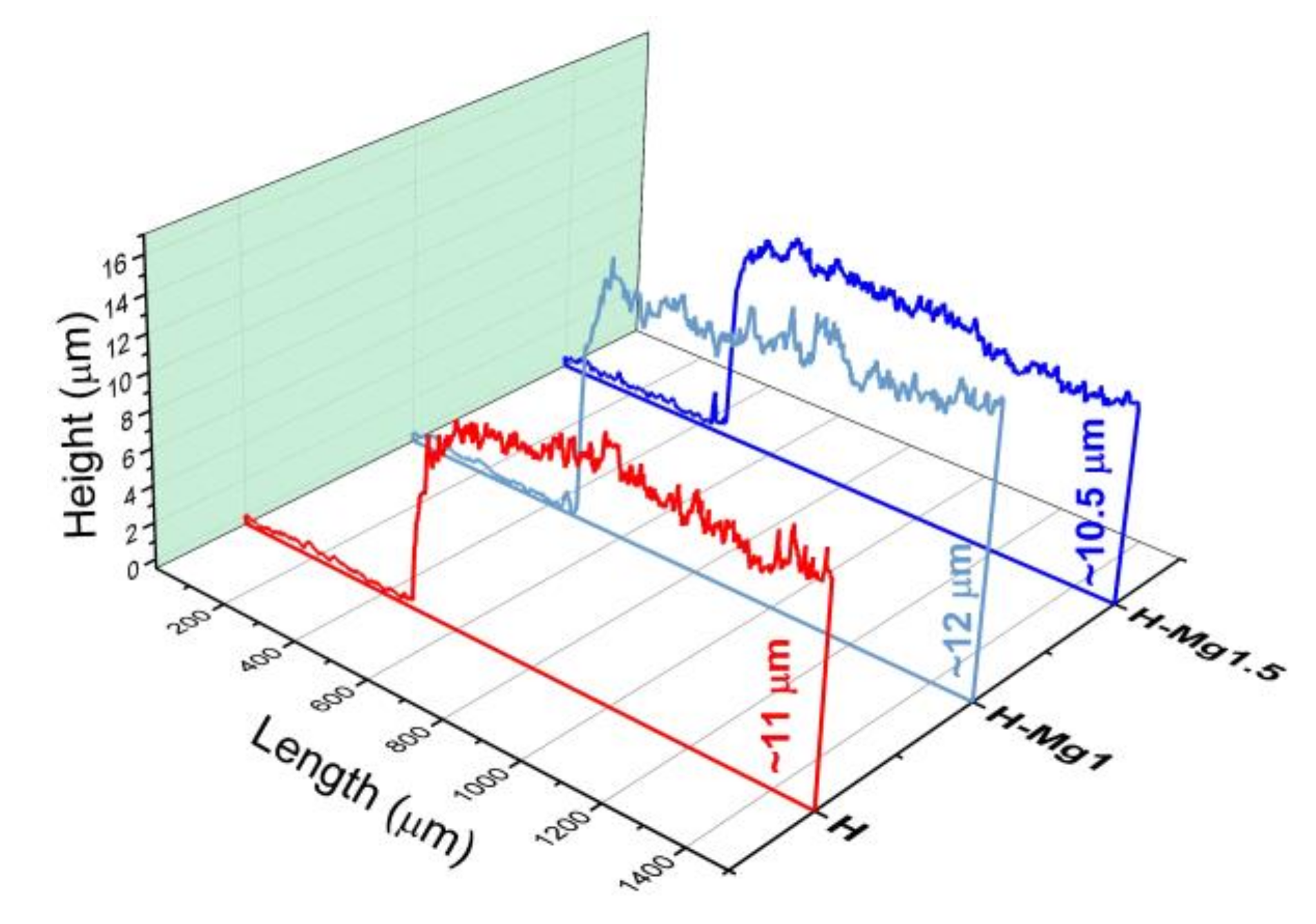
3.4. Roughness
3.5. Adhesion
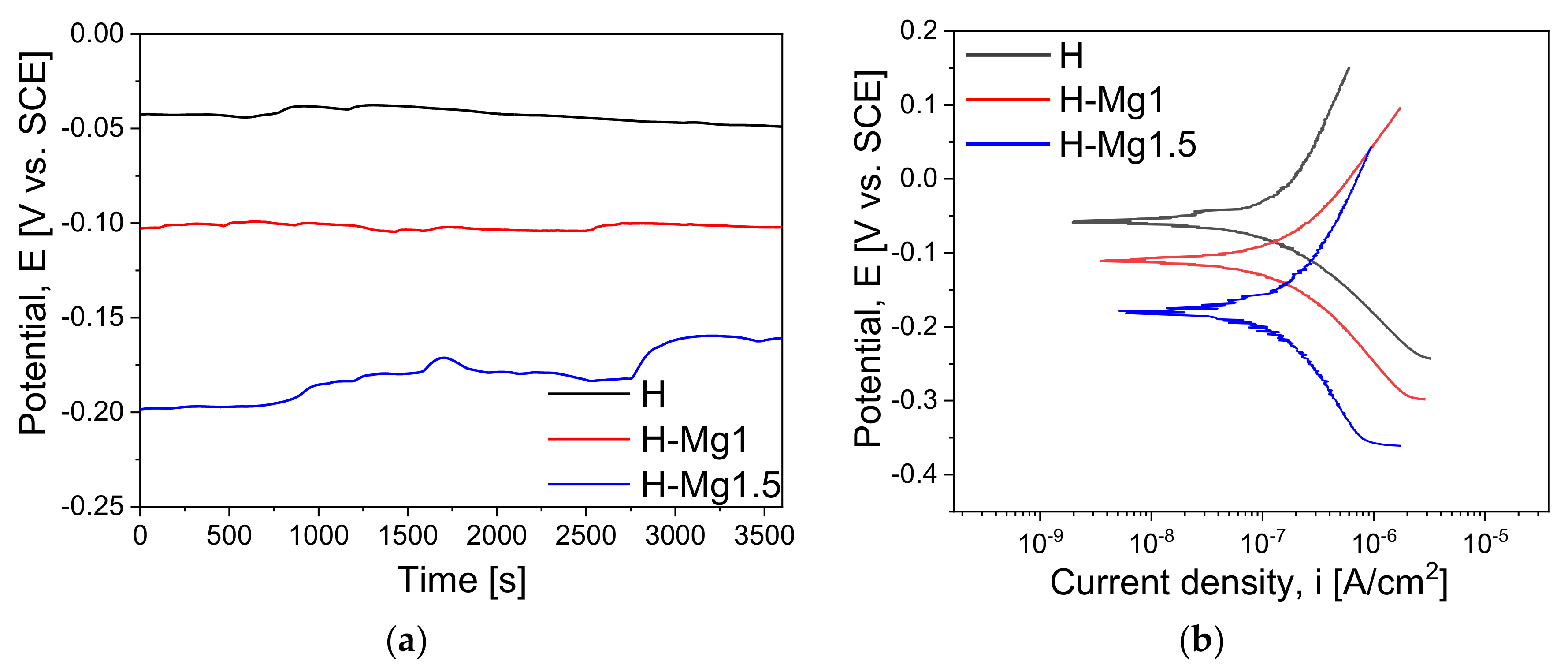
3.6. Electrochemical Behaviour
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Goodman, S.B.; Yao, Z.; Keeney, M.; Yang, F. The future of biologic coatings for orthopaedic implants. Biomaterials 2013, 34, 3174–3183. [Google Scholar] [CrossRef]
- Surmenev, R.A.; Surmeneva, M.A.; Ivanova, A.A. Significance of calcium phosphate coatings for the enhancement of new bone osteogenesis—A review. Acta Biomater. 2014, 10, 557–579. [Google Scholar] [CrossRef]
- Raphel, J.; Holodniy, M.; Goodman, S.B.; Heilshorn, S.C. Multifunctional coatings to simultaneously promote osseointegration and prevent infection of orthopaedic implants. Biomaterials 2016, 84, 301–314. [Google Scholar] [CrossRef] [PubMed]
- Canillas, M.; Pena, P.; de Aza, A.H.; Rodríguez, M.A. Calcium phosphates for biomedical applications. Boletín Soc. Española Cerámica Vidr. 2017, 56, 91–112. [Google Scholar] [CrossRef]
- Furko, M.; Havasi, V.; Kónya, Z.; Grünewald, A.; Detsch, R.; Boccaccini, A.R.; Balázsi, C. Development and characterization of multi-element doped hydroxyapatite bioceramic coatings on metallic implants for orthopedic applications. Bol. La Soc. Esp. Ceram. Y Vidr. 2018, 57, 55–65. [Google Scholar] [CrossRef]
- Damiati, L.; Eales, M.G.; Nobbs, A.H.; Su, B.; Tsimbouri, P.M.; Salmeron-Sanchez, M.; Dalby, M.J. Impact of surface topography and coating on osteogenesis and bacterial attachment on titanium implants. J. Tissue Eng. 2018, 9, 1–19. [Google Scholar] [CrossRef]
- Souza, J.C.M.; Sordi, M.B.; Kanazawa, M.; Ravindran, S.; Henriques, B.; Silva, F.S.; Aparicio, C.; Cooper, L.F. Nano-scale modification of titanium implant surfaces to enhance osseointegration. Acta Biomater. 2019, 94, 112–131. [Google Scholar] [CrossRef] [PubMed]
- Mitran, V.; Ion, R.; Miculescu, F.; Necula, M.; Mocanu, A.-C.; Stan, G.; Antoniac, I.; Cimpean, A. Osteoblast Cell Response to Naturally Derived Calcium Phosphate-Based Materials. Materials 2018, 11, 1097. [Google Scholar] [CrossRef]
- Dascalu, C.-A.; Miculescu, F.; Mocanu, A.-C.; Constantinescu, A.E.; Butte, T.M.; Pandele, A.M.; Ciocoiu, R.-C.; Voicu, S.I.; Ciocan, L.T. Novel Synthesis of Core-Shell Biomaterials from Polymeric Filaments with a Bioceramic Coating for Biomedical Applications. Coatings 2020, 10, 283. [Google Scholar] [CrossRef]
- Miculescu, F.; Mocanu, A.C.; Stan, G.E.; Miculescu, M.; Maidaniuc, A.; Cîmpean, A.; Mitran, V.; Voicu, S.I.; Machedon-Pisu, T.; Ciocan, L.T. Influence of the modulated two-step synthesis of biogenic hydroxyapatite on biomimetic products’ surface. Appl. Surf. Sci. 2018, 438, 147–157. [Google Scholar] [CrossRef]
- Bhattacharjee, A.; Gupta, A.; Verma, M.; Anand, M.P.; Sengupta, P.; Saravanan, M.; Manna, I.; Balani, K. Antibacterial and magnetic response of site-specific cobalt incorporated hydroxyapatite. Ceram. Int. 2020, 46, 513–522. [Google Scholar] [CrossRef]
- Thanh, T.T.; Cotrut, C.M.; Vranceanu, M.D.; Ungureanu, E.; Tarcolea, M. Studies of microstructure and composition of modified hydroxyapatite coatings via SEM investigations. U.P.B. Sci. Bull. Ser. B 2020, 82, 145–154. [Google Scholar]
- Huang, Y.; Hao, M.; Nian, X.; Qiao, H.; Zhang, X.; Zhang, X.; Song, G.; Guo, J.; Pang, X.; Zhang, H. Strontium and copper co-substituted hydroxyapatite-based coatings with improved antibacterial activity and cytocompatibility fabricated by electrodeposition. Ceram. Int. 2016, 42, 11876–11888. [Google Scholar] [CrossRef]
- Li, T.; Ling, L.; Lin, M.; Peng, H.; Ren, H.; Lou, C.-W.; Lin, J.-H. Recent advances in multifunctional hydroxyapatite coating by electrochemical deposition. J. Mater. Sci. 2020, 55, 6352–6374. [Google Scholar] [CrossRef]
- Sharma, M.; Nagar, R.; Meena, V.K.; Singh, S. Electro-deposition of bactericidal and corrosion-resistant hydroxyapatite nanoslabs. RSC Adv. 2019, 9, 11170–11178. [Google Scholar] [CrossRef]
- Munirathinam, B.; Jaladurgam, N.R.; Magesh, J.; Narayanan, R.; Mol, J.M.C.; Neelakantan, L. Improved corrosion protection of titanium implant material by crystallographic texturing of Sr doped calcium phosphate electrodeposits. Thin Solid Films 2019, 675, 115–121. [Google Scholar] [CrossRef]
- Morejón-Alonso, L.; Mochales, C.; Nascimento, L.; Müller, W. Electrochemical deposition of Sr and Sr/Mg-co-substituted hydroxyapatite on Ti-40Nb alloy. Mater. Lett. 2019, 248, 65–68. [Google Scholar] [CrossRef]
- Huang, Y.; Song, G.; Chang, X.; Wang, Z.; Zhang, X.; Han, S.; Su, Z.; Yang, H.; Yang, D.; Zhang, X. Nanostructured Ag+-substituted fluorhydroxyapatite-TiO2 coatings for enhanced bactericidal effects and osteoinductivity of Ti for biomedical applications. Int. J. Nanomed. 2018, 13, 2665–2684. [Google Scholar] [CrossRef]
- Vranceanu, D.M.; Tran, T.; Ungureanu, E.; Negoiescu, V.; Tarcolea, M.; Dinu, M.; Vladescu, A.; Zamfir, R.; Timotin, A.C.; Cotrut, C.M. Pulsed electrochemical deposition of Ag doped hydroxyapatite bioactive coatings on Ti6Al4V for medical purposes. UPB Sci. Bull. Ser. B Chem. Mater. Sci. 2018, 80, 173–184. [Google Scholar]
- Yajing, Y.; Qiongqiong, D.; Yong, H.; Han, S.; Pang, X. Magnesium substituted hydroxyapatite coating on titanium with nanotublar TiO2 intermediate layer via electrochemical deposition. Appl. Surf. Sci. 2014, 305, 77–85. [Google Scholar] [CrossRef]
- Lin, W.-C.; Chuang, C.-C.; Wang, P.-T.; Tang, C.-M. A Comparative Study on the Direct and Pulsed Current Electrodeposition of Cobalt-Substituted Hydroxyapatite for Magnetic Resonance Imaging Application. Materials 2018, 12, 116. [Google Scholar] [CrossRef] [PubMed]
- Vranceanu, D.M.; Parau, A.C.; Cotrut, C.M.; Kiss, A.E.; Constantin, L.R.; Braic, V.; Vladescu, A. In vitro evaluation of Ag doped hydroxyapatite coatings in acellular media. Ceram. Int. 2019, 45, 11050–11061. [Google Scholar] [CrossRef]
- Vlădescu, A.; Pârâu, A.; Pană, I.; Cotruț, C.M.; Constantin, L.R.; Braic, V.; Vrânceanu, D.M. In Vitro Activity Assays of Sputtered HAp Coatings with SiC Addition in Various Simulated Biological Fluids. Coatings 2019, 9, 389. [Google Scholar] [CrossRef]
- Ergun, C.; Webster, T.J.; Bizios, R.; Doremus, R.H. Hydroxylapatite with substituted magnesium, zinc, cadmium, and yttrium. I. Structure and microstructure. J. Biomed. Mater. Res. 2002, 59, 305–311. [Google Scholar] [CrossRef]
- Antoniac, I.; Miculescu, F.; Cotrut, C.; Ficai, A.; Rau, J.V.; Grosu, E.; Antoniac, A.; Tecu, C.; Cristescu, I. Controlling the Degradation Rate of Biodegradable Mg-Zn-Mn Alloys for Orthopedic Applications by Electrophoretic Deposition of Hydroxyapatite Coating. Materials 2020, 13, 263. [Google Scholar] [CrossRef]
- Antoniac, I.; Popescu, D.; Zapciu, A.; Antoniac, A.; Miculescu, F.; Moldovan, H. Magnesium Filled Polylactic Acid (PLA) Material for Filament Based 3D Printing. Materials 2019, 12, 719. [Google Scholar] [CrossRef]
- Vladescu, A.; Mihai Cotrut, C.; Ak Azem, F.; Bramowicz, M.; Pana, I.; Braic, V.; Birlik, I.; Kiss, A.; Braic, M.; Abdulgader, R.; et al. Sputtered Si and Mg doped hydroxyapatite for biomedical applications. Biomed. Mater. 2018, 13, 025011. [Google Scholar] [CrossRef]
- Rau, J.V.; Antoniac, I.; Filipescu, M.; Cotrut, C.; Fosca, M.; Nistor, L.C.; Birjega, R.; Dinescu, M. Hydroxyapatite coatings on Mg-Ca alloy prepared by Pulsed Laser Deposition: Properties and corrosion resistance in Simulated Body Fluid. Ceram. Int. 2018, 44, 16678–16687. [Google Scholar] [CrossRef]
- Bertinetti, L.; Drouet, C.; Combes, C.; Rey, C.; Tampieri, A.; Coluccia, S.; Martra, G. Surface characteristics of nanocrystalline apatites: Effect of Mg surface enrichment on morphology, surface hydration species, and cationic environments. Langmuir 2009, 25, 5647–5654. [Google Scholar] [CrossRef]
- Landi, E.; Logroscino, G.; Proietti, L.; Tampieri, A.; Sandri, M.; Sprio, S. Biomimetic Mg-substituted hydroxyapatite: From synthesis to in vivo behaviour. J. Mater. Sci. Mater. Med. 2008, 19, 239–247. [Google Scholar] [CrossRef]
- Chen, W.; Liu, Y.; Courtney, H.S.; Bettenga, M.; Agrawal, C.M.; Bumgardner, J.D.; Ong, J.L. In vitro anti-bacterial and biological properties of magnetron co-sputtered silver-containing hydroxyapatite coating. Biomaterials 2006, 27, 5512–5517. [Google Scholar] [CrossRef]
- Zhao, S.; Jiang, Q.; Peel, S.; Wang, X.; He, F. Effects of magnesium-substituted nanohydroxyapatite coating on implant osseointegration. Clin. Oral Implant. Res. 2013, 24, 34–41. [Google Scholar] [CrossRef] [PubMed]
- Bucur, A.I.; Linul, E.; Taranu, B.-O. Hydroxyapatite coatings on Ti substrates by simultaneous precipitation and electrodeposition. Appl. Surf. Sci. 2020, 527, 146820. [Google Scholar] [CrossRef]
- Dorozhkin, S.V. Calcium orthophosphate deposits: Preparation, properties and biomedical applications. Mater. Sci. Eng. C 2015, 55, 272–326. [Google Scholar] [CrossRef]
- Eliaz, N.; Sridh, T.M. Electrocrystallization of hydroxyapatite and its dependence on solution conditions. Cryst. Growth Des. 2008, 8, 3965–3977. [Google Scholar] [CrossRef]
- Cotrut, C.M.; Vladescu, A.; Dinu, M.; Vranceanu, D.M. Influence of deposition temperature on the properties of hydroxyapatite obtained by electrochemical assisted deposition. Ceram. Int. 2017, 44, 669–677. [Google Scholar] [CrossRef]
- Vladescu, A.; Vranceanu, D.M.; Kulesza, S.; Ivanov, A.N.; Bramowicz, M.; Fedonnikov, A.S.; Braic, M.; Norkin, I.A.; Koptyug, A.; Kurtukova, M.O.; et al. Influence of the electrolyte’s pH on the properties of electrochemically deposited hydroxyapatite coating on additively manufactured Ti64 alloy. Sci. Rep. 2017, 7. [Google Scholar] [CrossRef]
- Oriň, R.; Oriň, A.; Kupková, M.; Hrubovčáková, M.; Škantárová, L.; Arlinghaus, H.F. Study of Electrochemical Deposition and Degradation of Hydroxyapatite Coated Iron Biomaterials. Int. J. Electrochem. Sci. 2015, 10, 659–670. [Google Scholar]
- ASTM International. Standard Test Methods for Rating Adhesion by Tape Test; ASTM D3359-17; ASTM International: West Conshohocken, PA, USA, 2017; pp. 1–9. [Google Scholar]
- Kokubo, T.; Takadama, H. How useful is SBF in predicting in vivo bone bioactivity? Biomaterials 2006, 27, 2907–2915. [Google Scholar] [CrossRef]
- ASTM International. Standard Reference Test Method for Making Potentiostatic and Potentiodynamic Anodic Polarization Measurements; ASTM G5-94(2011)e1; ASTM International: West Conshohocken, PA, USA, 2011. [Google Scholar]
- Li, T.-T.; Ling, L.; Lin, M.-C.; Jiang, Q.; Lin, Q.; Lou, C.-W.; Lin, J.-H. Effects of ultrasonic treatment and current density on the properties of hydroxyapatite coating via electrodeposition and its in vitro biomineralization behavior. Mater. Sci. Eng. C 2019, 105, 110062. [Google Scholar] [CrossRef]
- Chakraborty, R.; Seesala, V.S.; Sengupta, S.; Dhara, S.; Saha, P.; Das, K.; Das, S. Comparison of Osteoconduction, cytocompatibility and corrosion protection performance of hydroxyapatite-calcium hydrogen phosphate composite coating synthesized in-situ through pulsed electro-deposition with varying amount of phase and crystallinity. Surf. Interfaces 2018, 10, 1–10. [Google Scholar] [CrossRef]
- Huang, Y.; Zhou, G.; Zheng, L.; Liu, H.; Niu, X.; Fan, Y. Micro-/Nano- sized hydroxyapatite directs differentiation of rat bone marrow derived mesenchymal stem cells towards an osteoblast lineage. Nanoscale 2012, 4, 2484. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Eliaz, N.; Xiang, Z.; Hsu, H.P.; Spector, M.; Hobbs, L.W. Early bone apposition in vivo on plasma-sprayed and electrochemically deposited hydroxyapatite coatings on titanium alloy. Biomaterials 2006, 27, 4192–4203. [Google Scholar] [CrossRef]
- Kanamoto, K.; Imamura, K.; Kataoka, N.; Oshitani, J.; Imanaka, H.; Nakanishi, K. Formation characteristics of calcium phosphate deposits on a metal surface by H2O2-electrolysis reaction under various conditions. Colloids Surf. A Physicochem. Eng. Asp. 2009, 350, 79–86. [Google Scholar] [CrossRef]
- Bakin, B.; Koc Delice, T.; Tiric, U.; Birlik, I.; Ak Azem, F. Bioactivity and corrosion properties of magnesium-substituted CaP coatings produced via electrochemical deposition. Surf. Coatings Technol. 2016, 301, 29–35. [Google Scholar] [CrossRef]
- Biemond, J.E.; Hannink, G.; Jurrius, A.M.G.; Verdonschot, N.; Buma, P. In Vivo Assessment of Bone Ingrowth Potential of Three-Dimensional E-Beam Produced Implant Surfaces and the Effect of Additional Treatment by Acid Etching and Hydroxyapatite Coating. J. Biomater. Appl. 2012, 26, 861–875. [Google Scholar] [CrossRef]
- Šponer, P.; Strnadová, M.; Urban, K. In vivo behaviour of low-temperature calcium-deficient hydroxyapatite: Comparison with deproteinised bovine bone. Int. Orthop. 2011, 35, 1553–1560. [Google Scholar] [CrossRef][Green Version]
- Thanh, D.T.M.; Nam, P.T.; Phuong, N.T.; Que, L.X.; Van Anh, N.; Hoang, T.; Lam, T.D. Controlling the electrodeposition, morphology and structure of hydroxyapatite coating on 316L stainless steel. Mater. Sci. Eng. C 2013, 33, 2037–2045. [Google Scholar] [CrossRef]
- Li, T.-T.; Ling, L.; Lin, M.-C.; Jiang, Q.; Lin, Q.; Lin, J.-H.; Lou, C.-W. Properties and Mechanism of Hydroxyapatite Coating Prepared by Electrodeposition on a Braid for Biodegradable Bone Scaffolds. Nanomaterials 2019, 9, 679. [Google Scholar] [CrossRef]
- Dong, X.-L.; Zhou, H.-L.; Wu, T.; Wang, Q. Behavior Regulation of Adsorbed Proteins via Hydroxyapatite Surface Texture Control. J. Phys. Chem. B 2008, 112, 4751–4759. [Google Scholar] [CrossRef]
- Liu, X.; He, D.; Zhou, Z.; Wang, G.; Wang, Z.; Wu, X.; Tan, Z. Characteristics of (002) Oriented Hydroxyapatite Coatings Deposited by Atmospheric Plasma Spraying. Coatings 2018, 8, 258. [Google Scholar] [CrossRef]
- Kim, H.; Camata, R.P.; Lee, S.; Rohrer, G.S.; Rollett, A.D.; Hennessy, K.M.; Bellis, S.L.; Vohra, Y.K. Calcium Phosphate Bioceramics with Tailored Crystallographic Texture for Controlling Cell Adhesion. MRS Proc. 2006, 925. [Google Scholar] [CrossRef]
- Zou, Z.; Liu, X.; Chen, L.; Lin, K.; Chang, J. Dental enamel-like hydroxyapatite transformed directly from monetite. J. Mater. Chem. 2012, 22, 22637. [Google Scholar] [CrossRef]
- Mišković-Stanković, V.B. Electrophoretic Deposition of Ceramic Coatings on Metal Surfaces. In Electrodeposition and Surface Finishing: Fundamentals and Applications; Stojan, S.D., Ed.; Springer: New York, NY, USA, 2014; pp. 133–216. ISBN 978-1-4939-4830-7. [Google Scholar]
- Tamimi, F.; Le Nihouannen, D.; Eimar, H.; Sheikh, Z.; Komarova, S.; Barralet, J. The effect of autoclaving on the physical and biological properties of dicalcium phosphate dihydrate bioceramics: Brushite vs. monetite. Acta Biomater. 2012, 8, 3161–3169. [Google Scholar] [CrossRef] [PubMed]
- Batra, U.; Kapoor, S.; Sharma, S. Influence of Magnesium Ion Substitution on Structural and Thermal Behavior of Nanodimensional Hydroxyapatite. J. Mater. Eng. Perform. 2013, 22, 1798–1806. [Google Scholar] [CrossRef]
- Gopi, D.; Karthika, A.; Nithiya, S.; Kavitha, L. In vitro biological performance of minerals substituted hydroxyapatite coating by pulsed electrodeposition method. Mater. Chem. Phys. 2014, 144, 75–85. [Google Scholar] [CrossRef]
- Rusu, V.M.; Ng, C.H.; Wilke, M.; Tiersch, B.; Fratzl, P.; Peter, M.G. Size-controlled hydroxyapatite nanoparticles as self-organized organic-inorganic composite materials. Biomaterials 2005, 26, 5414–5426. [Google Scholar] [CrossRef]
- Landi, E.; Tampieri, A.; Celotti, G.; Sprio, S. Densification behaviour and mechanisms of synthetic hydroxyapatites. J. Eur. Ceram. Soc. 2000, 20, 2377–2387. [Google Scholar] [CrossRef]
- Poralan, G.M.; Gambe, J.E.; Alcantara, E.M.; Vequizo, R.M. X-ray diffraction and infrared spectroscopy analyses on the crystallinity of engineered biological hydroxyapatite for medical application. IOP Conf. Ser. Mater. Sci. Eng. 2015, 79, 012028. [Google Scholar] [CrossRef]
- Leach, R. Surface Topography Characterisation. In Fundamental Principles of Engineering Nanometrology; Elsevier: Amsterdam, The Netherlands, 2014; pp. 241–294. [Google Scholar]
- Mayer, I.; Schlam, R.; Featherstone, J.D. Magnesium-containing carbonate apatites. J. Inorg. Biochem. 1997, 66, 1–6. [Google Scholar] [CrossRef]
- Parcharoen, Y.; Kajitvichyanukul, P.; Sirivisoot, S.; Termsuksawad, P. Hydroxyapatite electrodeposition on anodized titanium nanotubes for orthopedic applications. Appl. Surf. Sci. 2014, 311, 54–61. [Google Scholar] [CrossRef]
- Zhang, Z.; Dunn, M.F.; Xiao, T.D.; Tomsia, A.P.; Saiz, E. Nanostructured Hydroxyapatite Coatings for Improved Adhesion and Corrosion Resistance for Medical Implants. MRS Proc. 2001, 703. [Google Scholar] [CrossRef]
- Baboian, R.; Scully, J.R.; Dean, S.W.J. Corrosion Tests and Standards; Fontana, M.G., Staehle, R.W., Eds.; Springer US Plenum Press: New York, NY, USA, 2005; Volume 6, ISBN 978-1-4684-8988-0. [Google Scholar]
- Cai, Y.; Zhang, S.; Zeng, X.; Wang, Y.; Qian, M.; Weng, W. Improvement of bioactivity with magnesium and fluorine ions incorporated hydroxyapatite coatings via sol–gel deposition on Ti6Al4V alloys. Thin Solid Films 2009, 517, 5347–5351. [Google Scholar] [CrossRef]
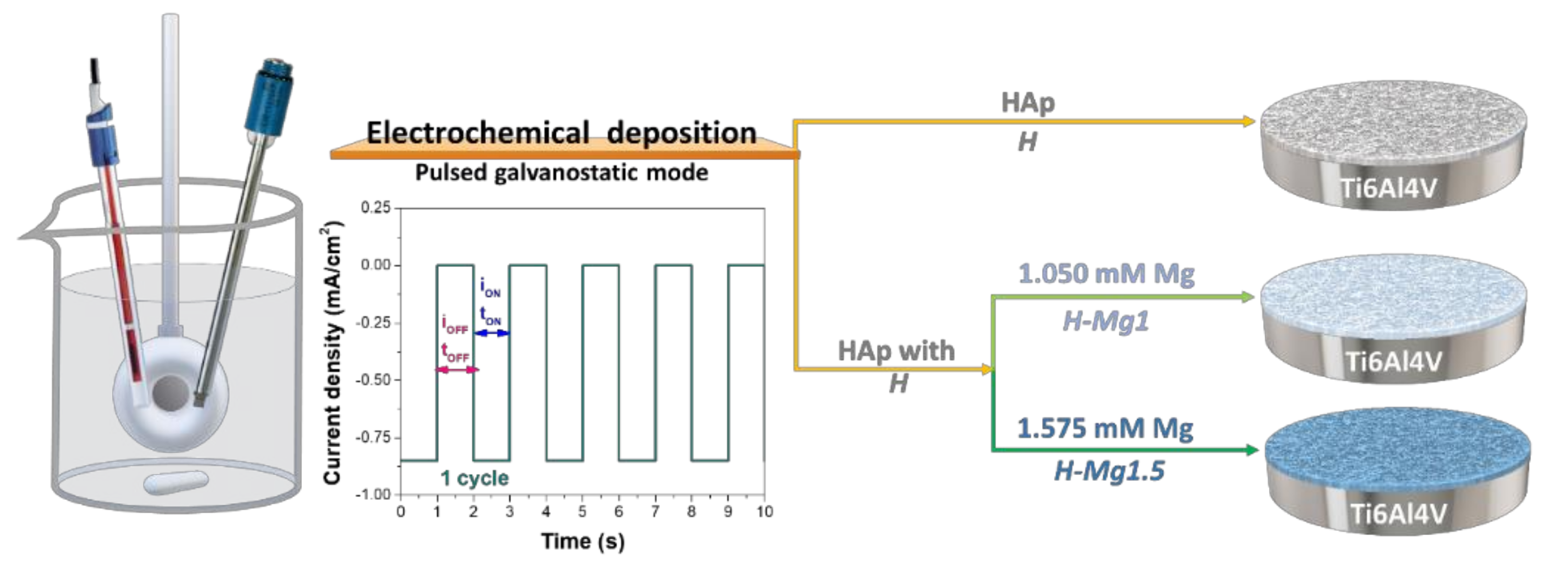


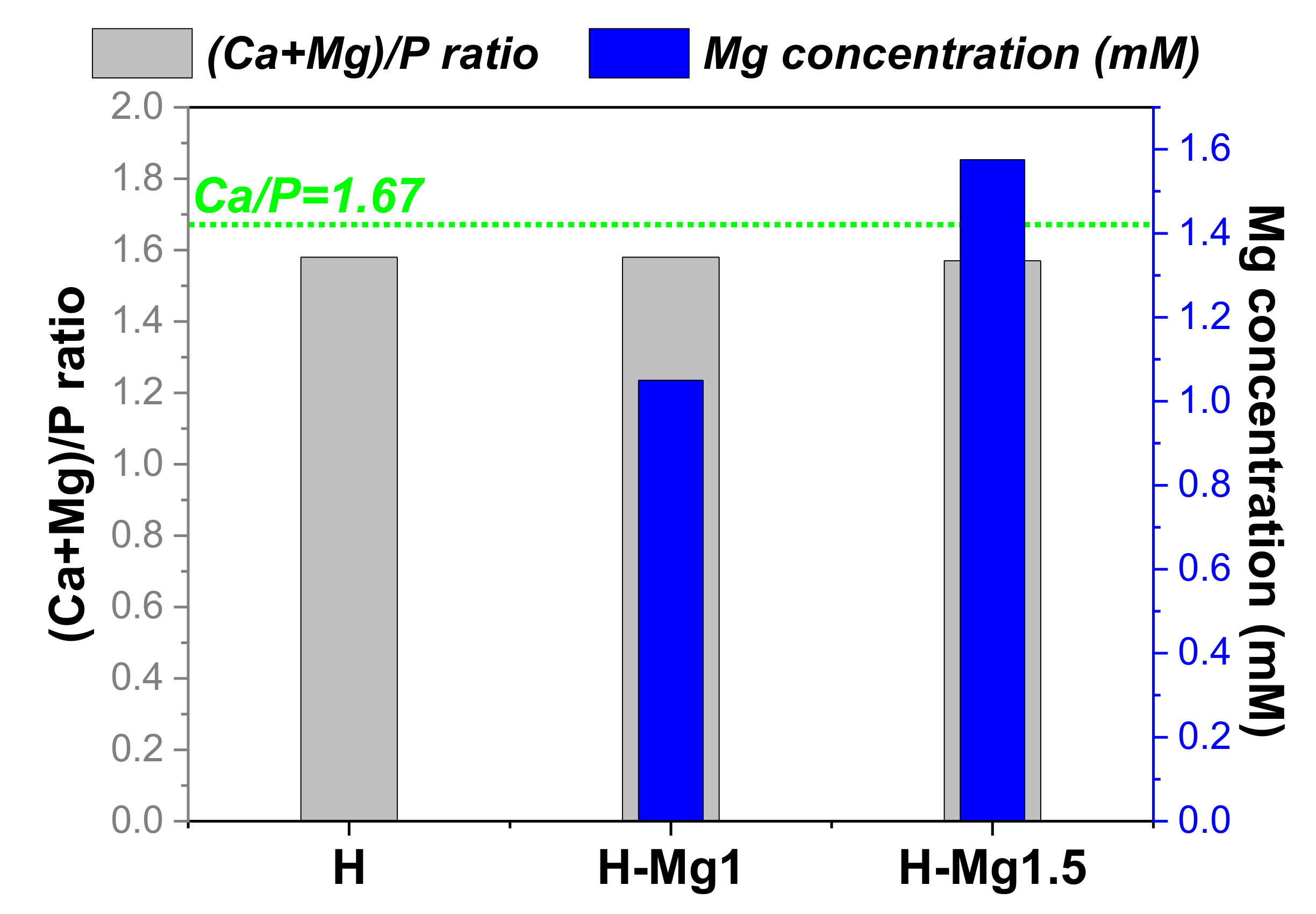
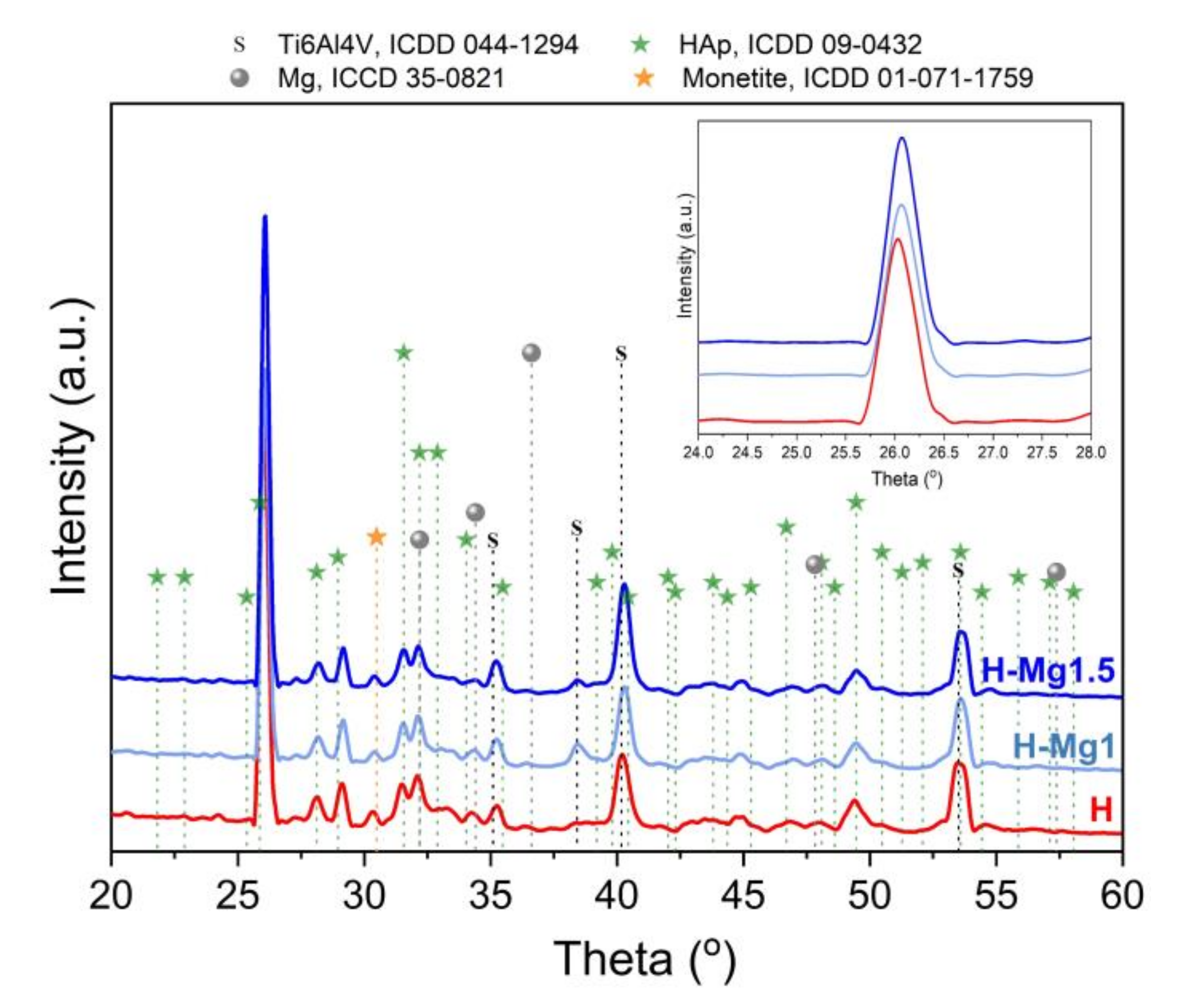
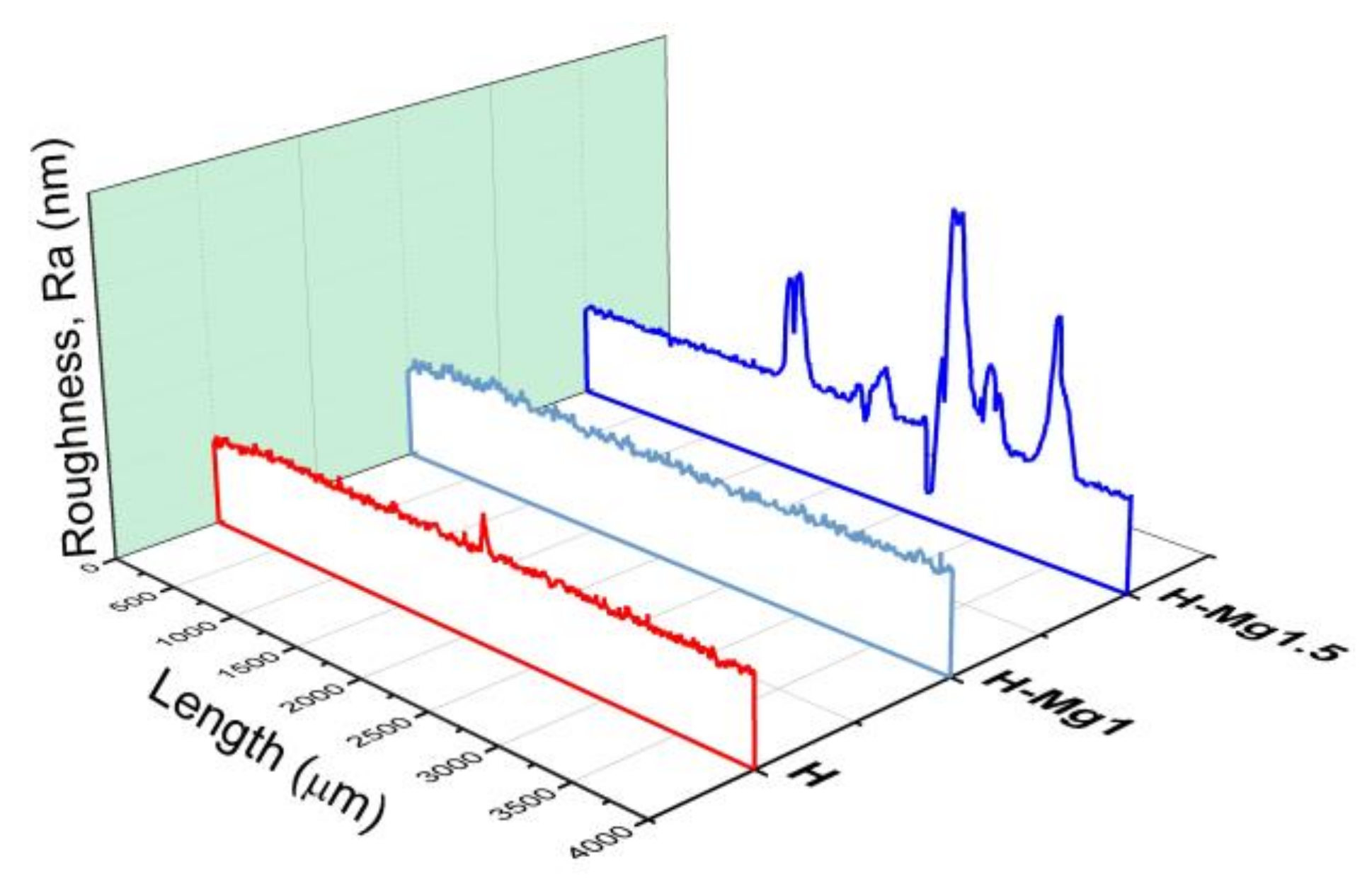

| Coating Type | Sample Codification | Chemical Composition (mM) | Molar Ratio (Ca + Mg)/P | ||
|---|---|---|---|---|---|
| Ca(NO3)2·4H2O | NH4H2PO4 | Mg(NO3)2·6H2O | |||
| HAp | H | 10.500 | 6.300 | 0 | 1.67 |
| HAp with Mg | H-Mg1 | 9.450 | 1.050 | ||
| H-Mg1.5 | 8.925 | 1.575 | |||
| H-Mg2 | 8.400 | 2.100 | |||
| Chemical Formula | Reagent Grade | Purity | Manufacturer |
|---|---|---|---|
| Ca(NO3)2·4H2O | ACS reagent | ≥99.00% | Sigma Aldrich, Munich, Germany |
| NH4H2PO | Trace metal basis | 99.99% | |
| Mg(NO3)2·6H2O | ACS reagent | 99.00% | |
| NaOH | ACS reagent | ≥98.00% |
| No. | Chemical Formula | Amount | Reagent Grade | Purity | Manufacturer |
|---|---|---|---|---|---|
| #1 | NaCl | 8.035 g/L | ACS reagent | >99.99% | Sigma Aldrich, Munich, Germany |
| #2 | NaHCO3 | 0.355 g/L | Bio Reagent | ≥99.50% | |
| #3 | KCl | 0.225 g/L | Purris p.a. | ≥99.50% | |
| #4 | K2HPO4·3H2O | 0.231 g/L | Reagent Plus | ≥99.00% | |
| #5 | MgCl2·6H2O | 0.311 g/L | Purum p.a. | ≥98.00% | |
| #6 | HCl | 39 mL | ACS reagent | ≥37.00% | |
| #7 | CaCl2 | 0.292 g/L | ACS reagent | ≥99.00% | |
| #8 | Na2SO4 | 0.072 g/L | ACS reagent | ≥99.00% | |
| #9 | (CH2OH)3CNH2 | 6.118 g/L | Standard & Buffer | ≥99.90% |
| Coating | Ra (nm) | Rq (nm) | Skew | Thickness (µm) |
|---|---|---|---|---|
| H | 439.7 (±33.9) | 587.9 (±66.7) | 0.84 (±0.1) | 11.1 (±0.9) |
| H-Mg1 | 558.2 (±37.9) | 710.9 (±47.0) | 0.38 (±0.1) | 12.2 (±0.7) |
| H-Mg1.5 | 4396.9 (±895.4) | 6609.1 (±1071.4) | 2.44 (±0.25) | 10.5 (±0.7) |
| Sample | Eoc (mV) | Ecorr (mV) | icorr (nA/cm2) | Βc (mV) | βa (mV) | Rp (kΩ·cm2) |
|---|---|---|---|---|---|---|
| H | −48.9 (±5.2) | −56.9 (±6.3) | 22.45 (±3.82) | 34.23 (±5.51) | 35.98 (±6.15) | 339.70 (±10.42) |
| H-Mg1 | −102.2 (±7.8) | −110.3 (±9.1) | 53.70 (±4.56) | 63.22 (±9.47) | 60.67 (±8.77) | 250.67 (±11.13) |
| H-Mg1.5 | −160.7 (±9.4) | −180.5 (±10.7) | 90.43 (±6.75) | 100.33 (±12.34) | 105.39 (±11.92) | 247.11 (±12.58) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vranceanu, D.M.; Ionescu, I.C.; Ungureanu, E.; Cojocaru, M.O.; Vladescu, A.; Cotrut, C.M. Magnesium Doped Hydroxyapatite-Based Coatings Obtained by Pulsed Galvanostatic Electrochemical Deposition with Adjustable Electrochemical Behavior. Coatings 2020, 10, 727. https://doi.org/10.3390/coatings10080727
Vranceanu DM, Ionescu IC, Ungureanu E, Cojocaru MO, Vladescu A, Cotrut CM. Magnesium Doped Hydroxyapatite-Based Coatings Obtained by Pulsed Galvanostatic Electrochemical Deposition with Adjustable Electrochemical Behavior. Coatings. 2020; 10(8):727. https://doi.org/10.3390/coatings10080727
Chicago/Turabian StyleVranceanu, Diana Maria, Ionut Cornel Ionescu, Elena Ungureanu, Mihai Ovidiu Cojocaru, Alina Vladescu, and Cosmin Mihai Cotrut. 2020. "Magnesium Doped Hydroxyapatite-Based Coatings Obtained by Pulsed Galvanostatic Electrochemical Deposition with Adjustable Electrochemical Behavior" Coatings 10, no. 8: 727. https://doi.org/10.3390/coatings10080727
APA StyleVranceanu, D. M., Ionescu, I. C., Ungureanu, E., Cojocaru, M. O., Vladescu, A., & Cotrut, C. M. (2020). Magnesium Doped Hydroxyapatite-Based Coatings Obtained by Pulsed Galvanostatic Electrochemical Deposition with Adjustable Electrochemical Behavior. Coatings, 10(8), 727. https://doi.org/10.3390/coatings10080727





.jpg)


