Synthesis and Characterization of Efficient ZnO/g-C3N4 Nanocomposites Photocatalyst for Photocatalytic Degradation of Methylene Blue
Abstract
1. Introduction
2. Experimental Details
2.1. Materials
2.2. Preparation of g-CN/Zn Ophotocatalyst
2.3. Materials Characterization
2.4. Photocatalytic Study
3. Results and Discussion
3.1. Photocatalytic Activity
3.2. Reactive Species Studies
3.3. Reusability Studies
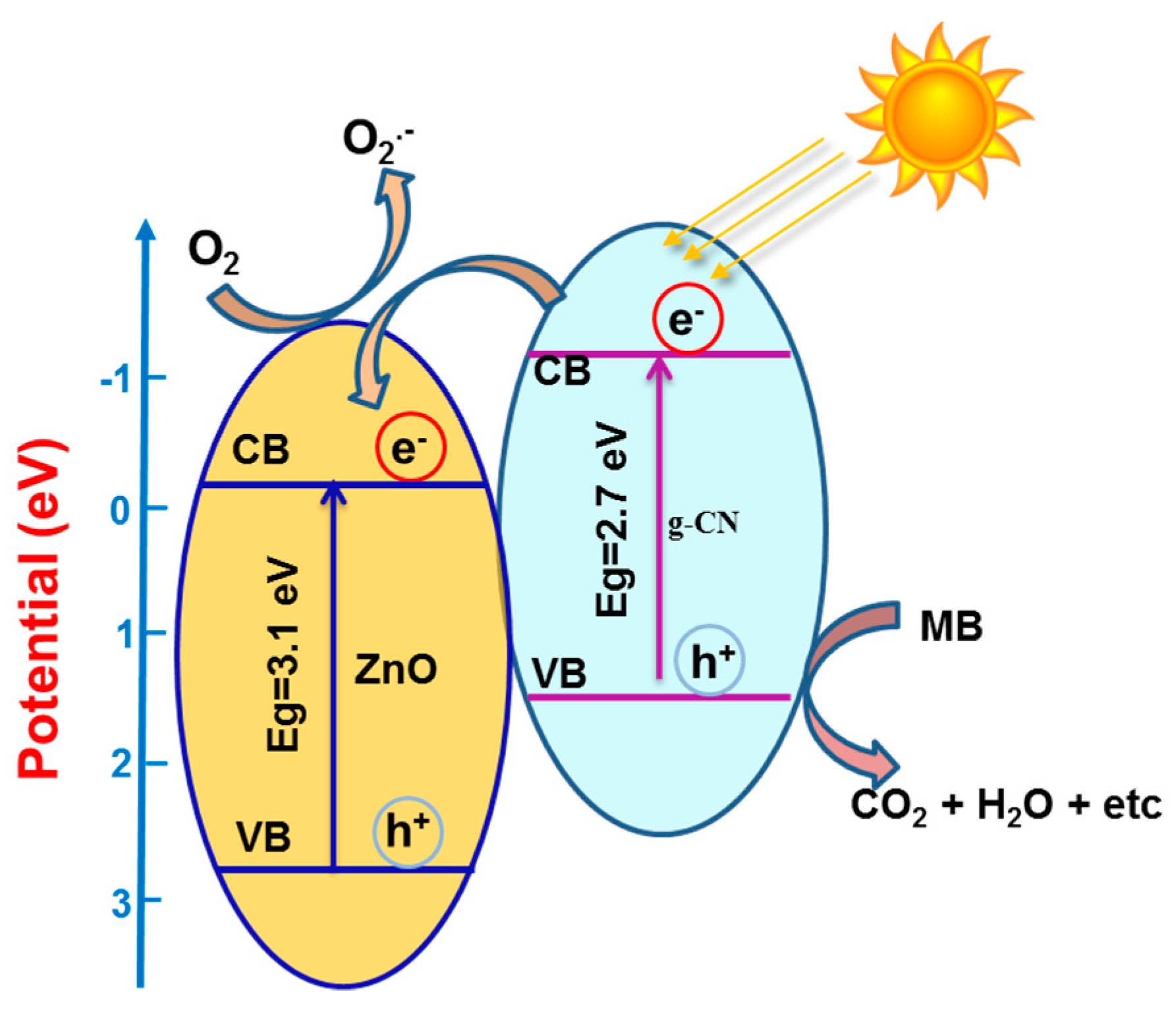
3.4. Mechanism
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gaya, U.I.; Abdullah, A.H. Heterogeneous photocatalytic degradation of organic contaminants over titanium dioxide: A review of fundamentals, progress and problems. J. Photochem. Photobiol. C Photochem. Rev. 2008, 9, 1–12. [Google Scholar] [CrossRef]
- Guo, F.; Cai, Y.; Guan, W.; Huang, H.; Liu, Y. Graphite carbon nitride/ZnIn2S4 heterojunction photocatalyst with enhanced photocatalytic performance for degradation of tetracycline under visible light irradiation. J. Phys. Chem. Solids 2017, 110, 370–378. [Google Scholar] [CrossRef]
- Bora, L.V.; Mewada, R.K. Visible/solar light active photocatalysts for organic effluent treatment: Fundamentals, mechanisms and parametric review. Renew. Sustain. Energy Rev. 2017, 76, 1393–1421. [Google Scholar] [CrossRef]
- Xu, Q.; Feng, J.; Li, L.; Xiao, Q.; Wang, J. Hollow ZnFe2O4/TiO2 composites: High-performance and recyclable visible-light photocatalyst. J. Alloys Compd. 2015, 641, 110–118. [Google Scholar] [CrossRef]
- Yu, C.; Cao, F.; Li, G.; Wei, R.; Yu, J.C.-M.; Jin, R.; Fan, Q.; Wang, C. Novel noble metal (Rh, Pd, Pt)/BiOX(Cl, Br, I) composite photocatalysts with enhanced photocatalytic performance in dye degradation. Sep. Purif. Technol. 2013, 120, 110–122. [Google Scholar] [CrossRef]
- Yang, L.; Duan, W.; Jiang, H.; Luo, S.; Luo, Y. Mesoporous TiO2@Ag3PO4 photocatalyst with high adsorbility and enhanced photocatalytic activity under visible light. Mater. Res. Bull. 2015, 70, 129–136. [Google Scholar] [CrossRef]
- Nazari, M.; Golestani-Fard, F.; Bayati, R.; Eftekhari-Yekta, B. Enhanced photocatalytic activity in anodized WO3-loaded TiO2 nanotubes. Superlattices Microstruct. 2015, 80, 91–101. [Google Scholar] [CrossRef]
- Dong, F.; Zhao, Z.; Xiong, T.; Ni, Z.; Zhang, W.; Sun, Y.; Ho, W.K. In situ construction of g-C3N4/g-C3N4 metal-free heterojunction for enhanced visible-light photocatalysis. ACS Appl. Mater. Interfaces 2013, 5, 11392–11401. [Google Scholar] [CrossRef]
- Xiang, Q.; Yu, J.; Jaroniec, M. Graphene-based semiconductor photocatalysts. Chem. Soc. Rev. 2012, 41, 782–796. [Google Scholar] [CrossRef]
- Tong, H.; Ouyang, S.; Bi, Y.; Umezawa, N.; Oshikiri, M.; Ye, J. Nano-photocatalytic materials: Possibilities and challenges. Adv. Mater. 2012, 24, 229–251. [Google Scholar] [CrossRef]
- Di Paola, A.; García-López, E.; Marcì, G.; Palmisano, L. A survey of photocatalytic materials for environmental remediation. J. Hazard. Mater. 2012, 211, 3–29. [Google Scholar] [CrossRef]
- Priya, A.; Arumugam, M.; Arunachalam, P.; Al-Mayouf, A.M.; Madhavan, J.; Theerthagiri, J.; Choi, M.Y. Fabrication of visible-light active BiFeWO6/ZnO nanocomposites with enhanced photocatalytic activity. Colloids Surf. A Physicochem. Eng. Asp. 2020, 586, 124294. [Google Scholar]
- Theerthagiri, J.; Salla, S.; Senthil, R.A.; Nithyadharseni, P.; Madankumar, A.; Arunachalam, P.; Maiyalagan, T.; Kim, H.-S.; Jayaraman, T.; Kim, H.S.; et al. A review on ZnO nanostructured materials: Energy, environmental and biological applications. Nanotechnology 2019, 30, 392001. [Google Scholar] [CrossRef]
- Malathi, A.; Madhavan, J.; Ashokkumar, M.; Arunachalam, P. A review on BiVO4 photocatalyst: Activity enhancement methods for solar photocatalytic applications. Appl. Catal. A Gen. 2018, 555, 47–74. [Google Scholar] [CrossRef]
- Karthikeyan, C.; Arunachalam, P.; Ramachandran, K.; Al-Mayouf, A.M.; Karuppuchamy, S. Recent advances in semiconductor metal oxides with enhanced methods for solar photocatalytic applications. J. Alloys Compd. 2020, 828, 154281. [Google Scholar] [CrossRef]
- Arunachalam, P.; Amer, M.S.; Ghanem, M.A.; Almayouf, A.M.; Zhao, D. Activation effect of silver nanoparticles on the photoelectrochemical performance of mesoporous TiO2 nanospheres photoanodes for water oxidation reaction. Int. J. Hydrogen Energy 2017, 42, 11346–11355. [Google Scholar] [CrossRef]
- Sakthivel, S.; Neppolian, B.; Shankar, M.V.; Arabindoo, B.; Palanichamy, M.; Murugesan, V. Solar photocatalytic degradation of azo dye: Comparison of photocatalytic efficiency of ZnO and TiO2. Sol. Energy Mater. Sol. cells 2003, 77, 65–82. [Google Scholar] [CrossRef]
- Lu, J.; Hu, H.; Yang, S.; Shanmugam, P.; Wei, W.; Selvaraj, M.; Xie, J. ZnS@carbonaceous aerogel composites fabricated in production of hydrogen and for removal of organic pollutants. J. Mater. Sci. Mater. Electron. 2018, 29, 8523–8534. [Google Scholar] [CrossRef]
- Shi, M.; Wei, W.; Jiang, Z.; Han, H.; Gao, J.; Xie, J. Biomass-derived multifunctional TiO2/carbonaceous aerogel composite as a highly efficient photocatalyst. RSC Adv. 2016, 6, 25255–25266. [Google Scholar] [CrossRef]
- Vadivel, S.; Naveen, A.N.; Theerthagiri, J.; Madhavan, J.; Priya, T.S.; Balasubramanian, N. Solvothermal synthesis of BiPO4 nanorods/MWCNT (1D-1D) composite for photocatalyst and supercapacitor applications. Ceram. Int. 2016, 42, 14196–14205. [Google Scholar] [CrossRef]
- Shanmugam, P.; Murthy, A.P.; Theerthagiri, J.; Wei, W.; Madhavan, J.; Kim, H.-S.; Maiyalagan, T.; Xie, J. Robust bifunctional catalytic activities of N-doped carbon aerogel-nickel composites for electrocatalytic hydrogen evolution and hydrogenation of nitrocompounds. Int. J. Hydrogen Energy 2019, 44, 13334–13344. [Google Scholar] [CrossRef]
- Priya, A.; Senthil, R.A.; Selvi, A.; Arunachalam, P.; Kumar, C.S.; Madhavan, J.; Boddula, R.; Pothu, R.; Al-Mayouf, A.M. A study of photocatalytic and photoelectrochemical activity of as-synthesized WO3/g-C3N4 composite photocatalysts for AO7 degradation. Mater. Sci. Energy Technol. 2020, 3, 43–50. [Google Scholar] [CrossRef]
- Thiagarajan, K.; Bavani, T.; Arunachalam, P.; Lee, S.J.; Theerthagiri, J.; Madhavan, J.; Pollet, B.G.; Choi, M.Y. Nanofiber NiMoO4/g-C3N4 Composite Electrode Materials for Redox Supercapacitor Applications. Nanomaterials 2020, 10, 392. [Google Scholar] [CrossRef]
- Senthil, R.A.; Theerthagiri, J.; Madhavan, J.; Murugan, K.; Arunachalam, P.; Arof, A.K. Enhanced performance of dye-sensitized solar cells based on organic dopant incorporated PVDF-HFP/PEO polymer blend electrolyte with g-C3N4/TiO2 photoanode. J. Solid State Chem. 2016, 242, 199–206. [Google Scholar] [CrossRef]
- Wen, M.Q.; Xiong, T.; Zang, Z.G.; Wei, W.; Tang, X.S.; Dong, F. Synthesis of MoS2/gC3N4 nanocomposites with enhanced visible-light photocatalytic activity for the removal of nitric oxide (NO). Opt. Express 2016, 24, 10205–10212. [Google Scholar] [CrossRef]
- Zhang, X.; Xie, X.; Wang, H.; Zhang, J.; Pan, B.; Xie, Y. Enhanced photoresponsive ultrathin graphitic-phase C3N4 nanosheets for bioimaging. J. Am. Chem. Soc. 2013, 135, 18–21. [Google Scholar] [CrossRef]
- Huang, L.; Li, Y.; Xu, H.; Xu, Y.; Xia, J.; Wang, K.; Xu, Y.; Cheng, X. Synthesis and characterization of CeO2/gC3N4 composites with enhanced visible-light photocatatalytic activity. RSC Adv. 2013, 3, 22269–22279. [Google Scholar] [CrossRef]
- Wang, X.; Maeda, K.; Thomas, A.; Takanabe, K.; Xin, G.; Carlsson, J.M.; Domen, K.; Antonietti, M. A metal-free polymeric photocatalyst for hydrogen production from water under visible light. Nat. Mater. 2008, 8, 76–80. [Google Scholar] [CrossRef]
- Fageria, P.; Nazir, R.; Gangopadhyay, S.; Barshilia, H.C.; Pande, S. Graphitic-carbon nitride support for the synthesis of shape-dependent ZnO and their application in visible light photocatalysts. RSC Adv. 2015, 5, 80397–80409. [Google Scholar] [CrossRef]
- Dong, F.; Wu, L.W.; Sun, Y.J.; Fu, M.; Wu, Z.B.; Lee, S.C. In Situ Construction of g-C3N4/g-C3N4 Metal-Free Heterojunction for Enhanced Visible-Light Photocatalysis. J. Mater. Chem. 2011, 21, 15171–15174. [Google Scholar] [CrossRef]
- Zhang, Y.; Gong, H.; Li, G.; Zeng, H.; Zhong, L.; Liu, K.; Cao, H.; Yan, H. Synthesis of graphitic carbon nitride by heating mixture of urea and thiourea for enhanced photocatalytic H2 production from water under visible light. Int. J. Hydrogen Energy 2017, 42, 143–151. [Google Scholar] [CrossRef]
- Dong, F.; Sun, Y.; Wu, L.; Fu, M.; Wu, Z. Facile transformation of low cost thiourea into nitrogen-rich graphitic carbon nitride nanocatalyst with high visible light photocatalytic performance. Catal. Sci. Technol. 2012, 2, 1332–1335. [Google Scholar] [CrossRef]
- Lu, J.; Zhang, Q.; Wang, J.; Saito, F.; Uchida, M. Synthesis of N-Doped ZnO by grinding and subsequent heating ZnO-urea mixture. Powder Technol. 2006, 162, 33–37. [Google Scholar] [CrossRef]
- Zhang, W.; Zhang, Q.; Dong, F.; Zhao, Z. The Multiple Effects of Precursors on the Properties of Polymeric Carbon Nitride. Int. J. Photoenergy 2013, 685038. [Google Scholar] [CrossRef]
- Xiang, Q.; Yu, J.; Jaroniec, M. Preparation and Enhanced Visible-Light Photocatalytic H2-Production Activity of Graphene/C3N4 Composites. J. Phys. Chem. C 2011, 115, 7355–7363. [Google Scholar] [CrossRef]
- Hou, Y.D.; Laursen, A.B.; Zhang, J.S.; Zhang, G.G.; Zhu, Y.S.; Wang, X.C.S. Dahl an I. Chorkendorff. Angew. Chem. Int. Ed. 2013, 52, 1–6. [Google Scholar]
- Yan, H.; Yang, H. TiO2–g-C3N4 composite materials for photocatalytic H2 evolution under visible light irradiation. J. Alloys Compd. 2011, 509, L26–L29. [Google Scholar] [CrossRef]
- Yan, S.C.; Lv, S.B.; Zou, Z.; Li, Z.S. Organic–inorganic composite photocatalyst of g-C3N4 and TaON with improved visible light photocatalytic activities. Dalton Trans. 2010, 39, 1488–1491. [Google Scholar] [CrossRef]
- Sun, J.-X.; Yuan, Y.; Qiu, L.-G.; Jiang, X.; Shena, Y.; Shena, Y.; Zhu, J. Fabrication of composite photocatalyst g-C3N4–ZnO and enhancement of photocatalytic activity under visible light. Dalton Trans. 2012, 41, 6756–6763. [Google Scholar] [CrossRef]
- Wang, Y.; Bai, X.; Pan, C.; He, J.; Zhu, Y. Enhancement of photocatalytic activity of Bi2WO6 hybridized with graphite-like C3N4. J. Mater. Chem. 2012, 22, 11568–11573. [Google Scholar] [CrossRef]
- Cheng, N.; Tian, J.; Liu, Q.; Ge, C.; Qusti, A.H.; Asiri, A.M.; Al-Youbi, A.O.; Sun, X. Au-Nanoparticle-Loaded Graphitic Carbon Nitride Nanosheets: Green Photocatalytic Synthesis and Application toward the Degradation of Organic Pollutants. ACS Appl. Mater. Interfaces 2013, 5, 6815–6819. [Google Scholar] [CrossRef]
- Fu, J.; Chang, B.; Tian, Y.; Xi, F.; Dong, X. Novel C3N4–CdS composite photocatalysts with organic–inorganic heterojunctions: In situ synthesis, exceptional activity, high stability and photocatalytic mechanism. J. Mater. Chem. A 2013, 1, 3083. [Google Scholar] [CrossRef]
- Huang, L.; Xu, H.; Li, Y.; Li, H.; Cheng, X.; Xia, J.; Xu, Y.; Cai, G. Visible-light-induced WO3/gC3N4 composites with enhanced photocatalytic activity. Dalton Trans. 2013, 42, 8606–8616. [Google Scholar] [CrossRef]
- Kuriakose, S.; Choudhary, V.; Satpati, B.; Mohapatra, S. Facile synthesis of Ag–ZnO hybrid nanospindles for highly efficient photocatalytic degradation of methyl orange. Phys. Chem. Chem. Phys. 2014, 16, 17560–17568. [Google Scholar] [CrossRef]
- Kadam, A.; Kim, T.G.; Shin, D.-S.; Garadkar, K.M.; Park, J. Morphological evolution of Cu doped ZnO for enhancement of photocatalytic activity. J. Alloys Compd. 2017, 710, 102–113. [Google Scholar] [CrossRef]
- Barzgari, Z.; Ghazizadeh, A.; Askari, S.Z. Preparation of Mn-doped ZnO nanostructured for photocatalytic degradation of Orange G under solar light. Res. Chem. Intermed. 2015, 42, 4303–4315. [Google Scholar] [CrossRef]
- Kumar, R.; Umar, A.; Rana, D.S.; Sharma, P.; Chauhan, M.; Chauhan, G.S. Fe-doped ZnO nanoellipsoids for enhanced photocatalytic and highly sensitive and selective picric acid sensor. Mater. Res. Bull. 2018, 102, 282–288. [Google Scholar] [CrossRef]
- Kumar, S.; Singh, V.; Tanwar, A. Structural, morphological, optical and photocatalytic properties of Ag-doped ZnO nanoparticles. J. Mater. Sci. Mater. Electron. 2015, 27, 2166–2173. [Google Scholar] [CrossRef]
- Ji, Z.; Luo, Z.; Li, J.; Li, P. Enhanced Photocatalytic Activity of ZnO Toward the Degradation of Methylene Blue Dye: Effects of Fe3+ and Sn4+ Doping. Phys. Status Solidi 2019, 216, 1800947. [Google Scholar] [CrossRef]
- Raghavan, N.; Thangavel, S.; Venugopal, G. Enhanced photocatalytic degradation of methylene blue by reduced graphene-oxide/titanium dioxide/zinc oxide ternary nanocomposites. Mater. Sci. Semicond. Process. 2015, 30, 321–329. [Google Scholar] [CrossRef]
- Tien, H.N.; Luan, V.H.; Hoa, L.T.; Khoa, N.T.; Hahn, S.H.; Chung, J.S.; Shin, E.W.; Hur, S.H. One-pot synthesis of a reduced graphene oxide–zinc oxide sphere composite and its use as a visible light photocatalyst. Chem. Eng. J. 2013, 229, 126–133. [Google Scholar] [CrossRef]
- Liu, L.; Ma, D.; Zheng, H.; Li, X.; Cheng, M.; Bao, X. Synthesis and characterization of microporous carbon nitride. Microporous Mesoporous Mater. 2008, 110, 216–222. [Google Scholar] [CrossRef]
- Li, X.; Zhang, J.; Shen, L.; Ma, Y.; Lei, W.; Cui, Q.; Zou, G. Preparation and characterization of graphitic carbon nitride through pyrolysis of melamine. Appl. Phys. A 2008, 94, 387–392. [Google Scholar] [CrossRef]
- Le, S.; Jiang, T.; Li, Y.; Zhao, Q.; Li, Y.; Fang, W.; Gong, M. Highly efficient visible-light-driven mesoporous graphitic carbon nitride/ZnO nanocomposite photocatalysts. Appl. Catal. B Environ. 2017, 200, 601–610. [Google Scholar] [CrossRef]
- Isai, K.A.; Shrivastava, V.S. Photocatalytic degradation of methylene blue using ZnO and 2%Fe–ZnO semiconductor nanomaterials synthesized by sol–gel method: A comparative study. SN Appl. Sci. 2019, 1, 1247. [Google Scholar] [CrossRef]
- Mao, Y.; Wu, M.; Li, G.; Dai, P.; Yu, X.; Bai, Z.; Chen, P. Photocatalytic degradation of methylene blue over boron-doped g-C3N4 together with nitrogen-vacancies under visible light irradiation. React. Kinet. Mech. Catal. 2018, 125, 1179–1190. [Google Scholar] [CrossRef]
- Adeleke, J.; Theivasanthi, T.; Thiruppathi, M.; Swaminathan, M.; Akomolafe, T.; Alabi, A. Photocatalytic degradation of methylene blue by ZnO/NiFe2O4 nanoparticles. Appl. Surf. Sci. 2018, 455, 195–200. [Google Scholar] [CrossRef]
- Li, Y.; Wang, K.; Wu, J.; Gu, L.; Lu, Z.; Wang, X.; Cao, X. Synthesis of highly permeable Fe 2 O 3/ZnO hollow spheres for printable photocatalysis. RSC Adv. 2015, 5, 88277–88286. [Google Scholar] [CrossRef]
- Zhu, H.Y.; Jiang, R.; Fu, Y.Q.; Li, R.R.; Yao, J.; Jiang, S.T. Novel multifunctional NiFe2O4/ZnO hybrids for dye removal by adsorption, photocatalysis and magnetic separation. Appl. Surf. Sci. 2016, 369, 1–10. [Google Scholar] [CrossRef]
- Chen, L.; Jiang, D.; He, T.; Wu, Z.; Chen, M. In-situ ion exchange synthesis of hierarchical AgI/BiOI microsphere photocatalyst with enhanced photocatalytic properties. CrystEngComm 2013, 15, 7556. [Google Scholar] [CrossRef]
- Malathi, A.; Arunachalam, P.; Madhavan, J.; Almayouf, A.M.; Ghanem, M.A. Rod-on-flake α-FeOOH/BiOI nanocomposite: Facile synthesis, characterization and enhanced photocatalytic performance. Colloids Surf. A Physicochem. Eng. Asp. 2018, 537, 435–445. [Google Scholar] [CrossRef]
- Fu, H.; Xu, T.; Zhu, S.; Zhu, Y. Photocorrosion Inhibition and Enhancement of Photocatalytic Activity for ZnO via Hybridization with C60. Environ. Sci. Technol. 2008, 42, 8064–8069. [Google Scholar] [CrossRef]
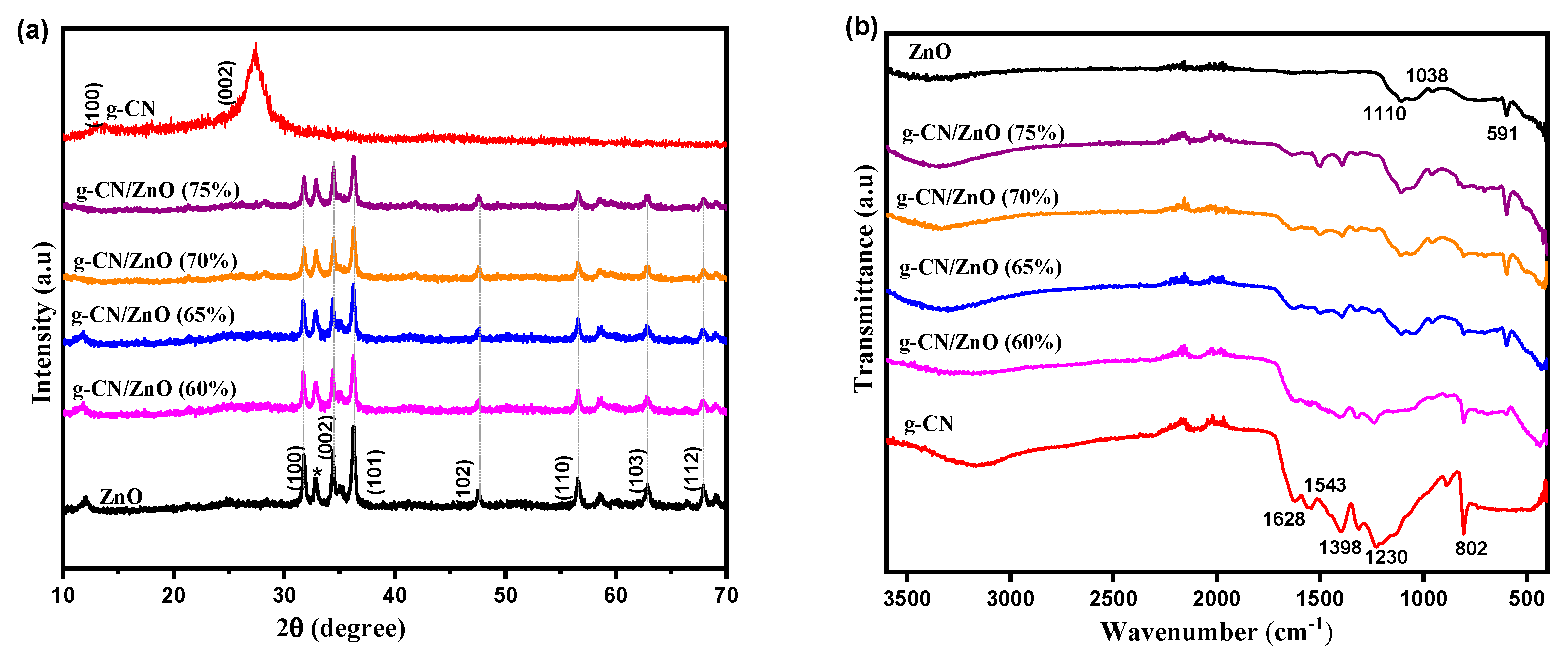

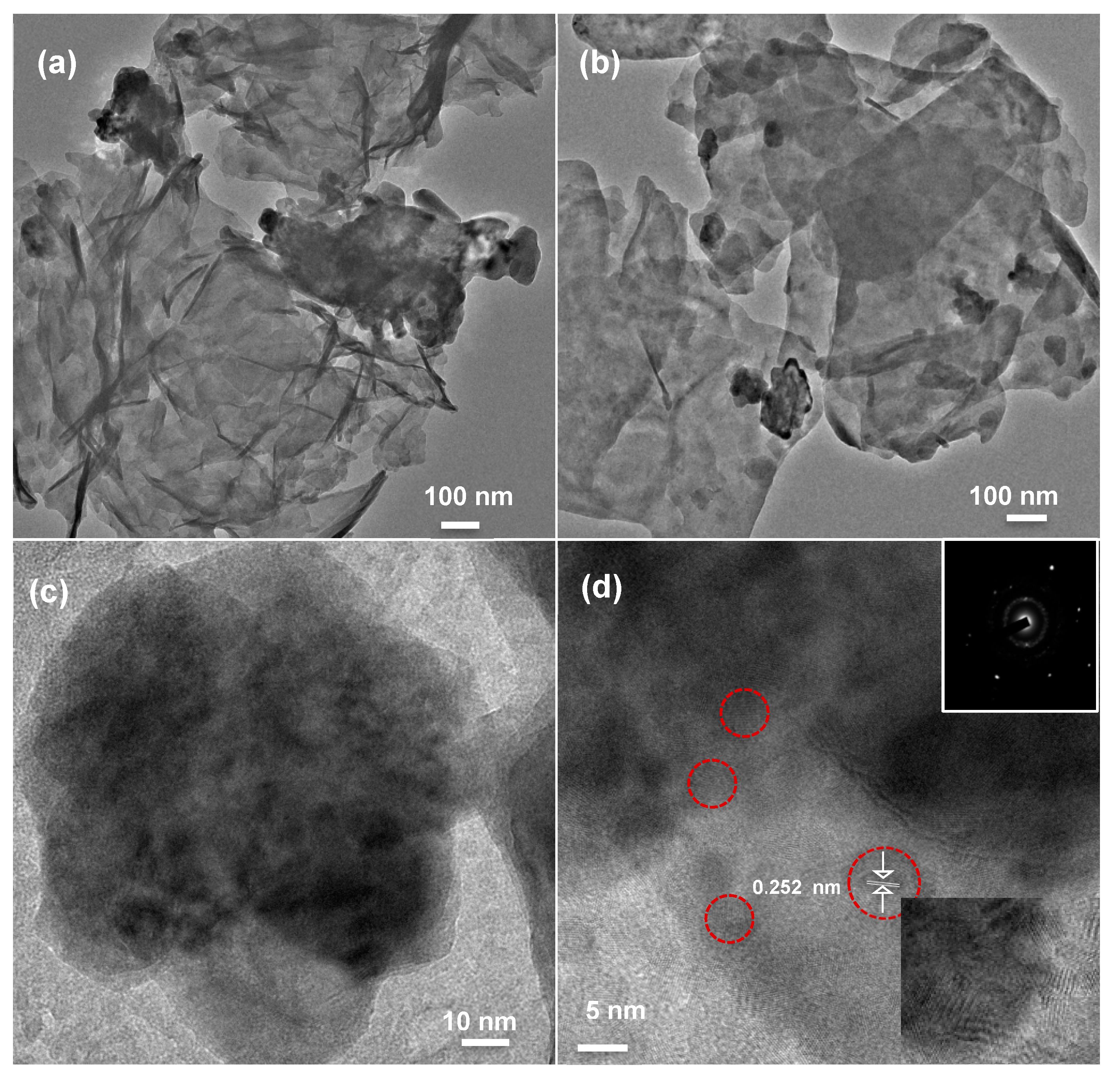
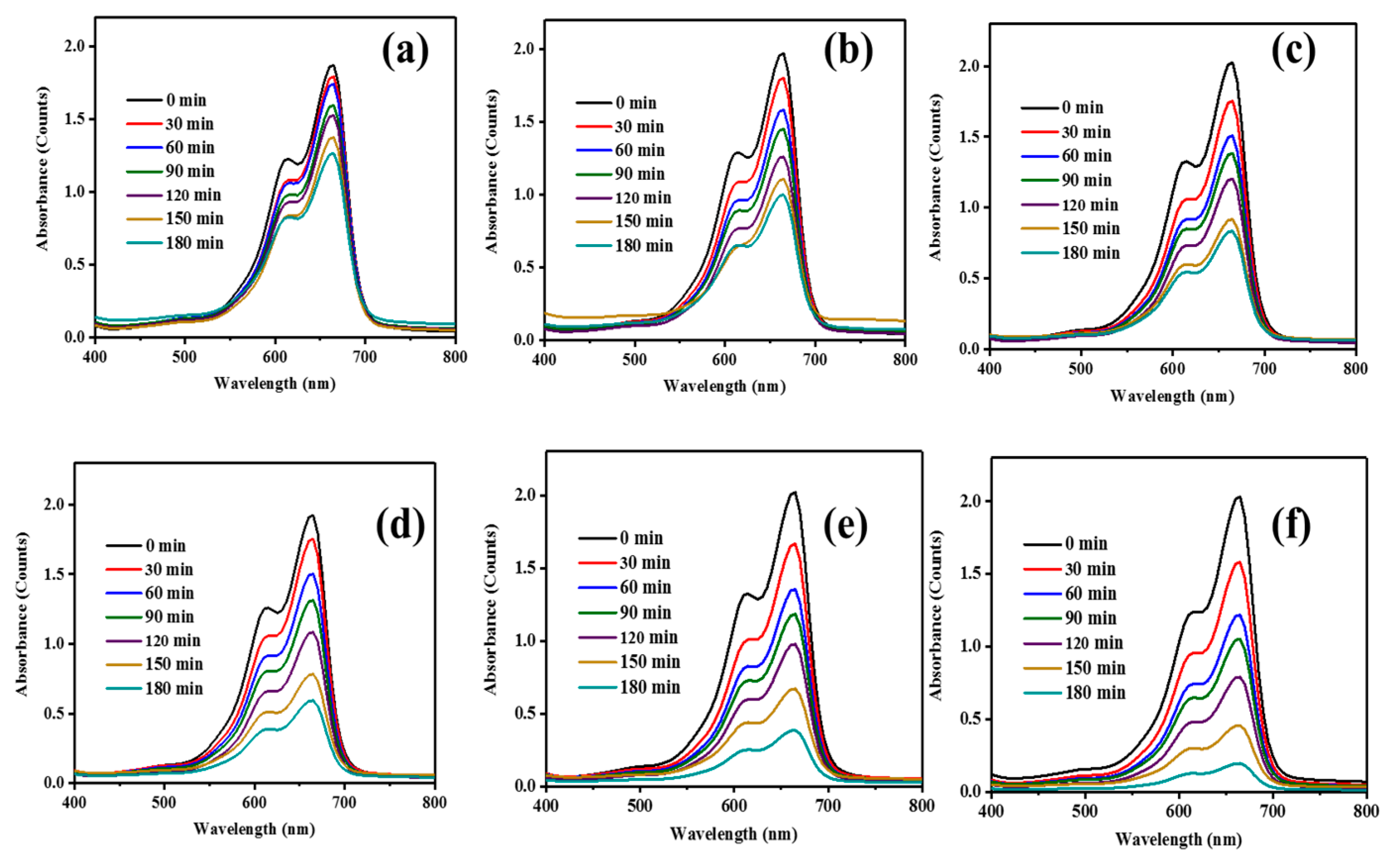
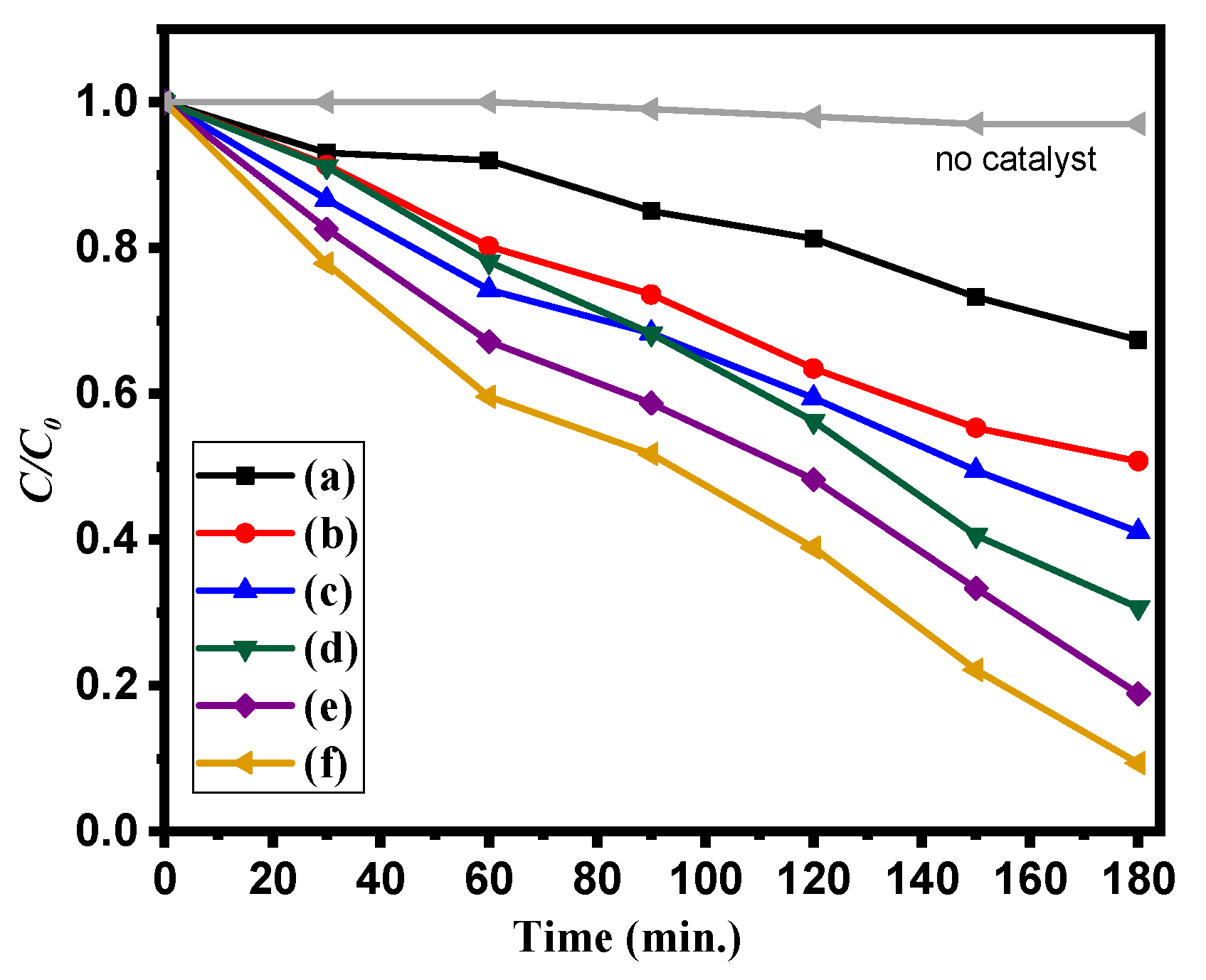
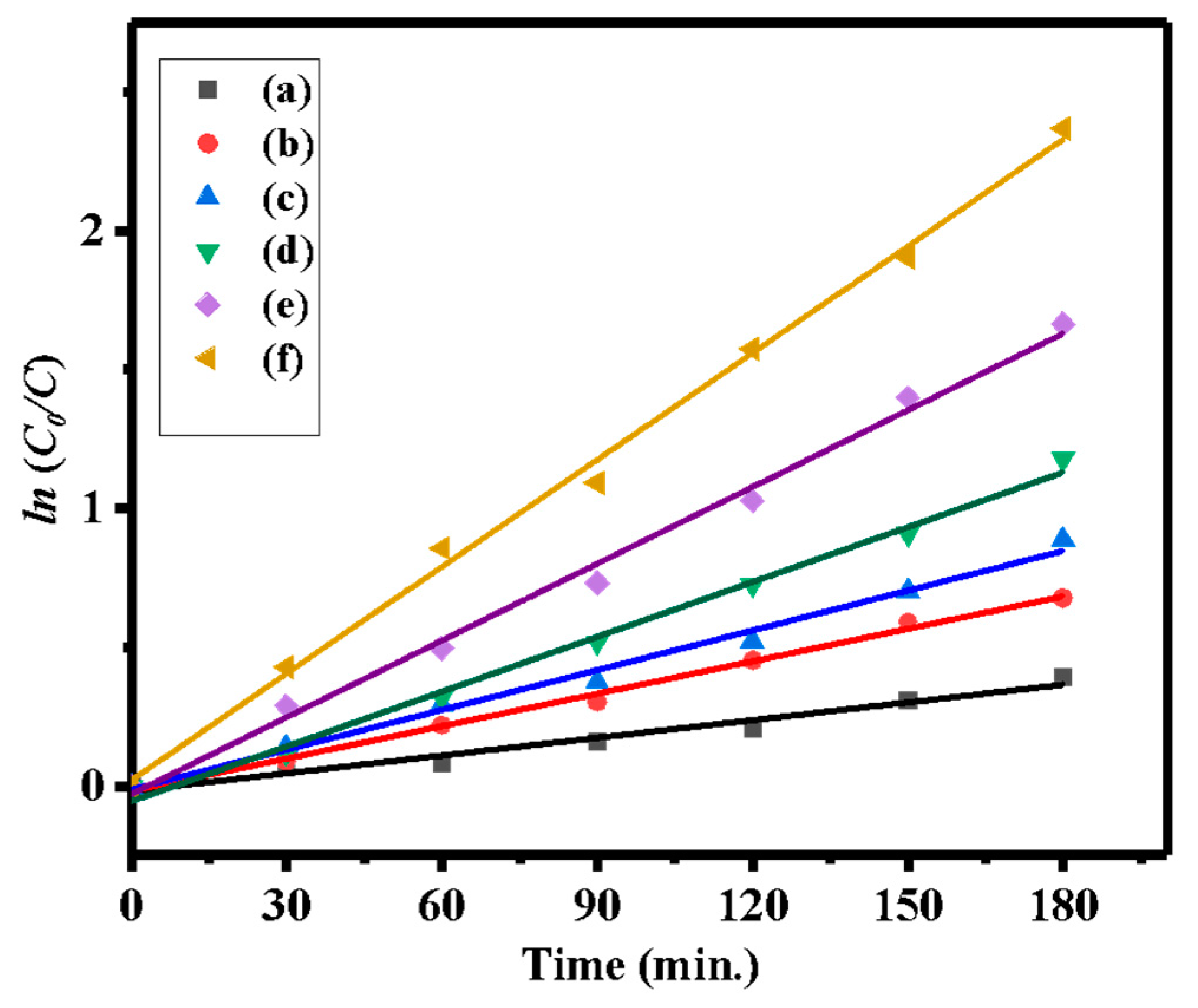
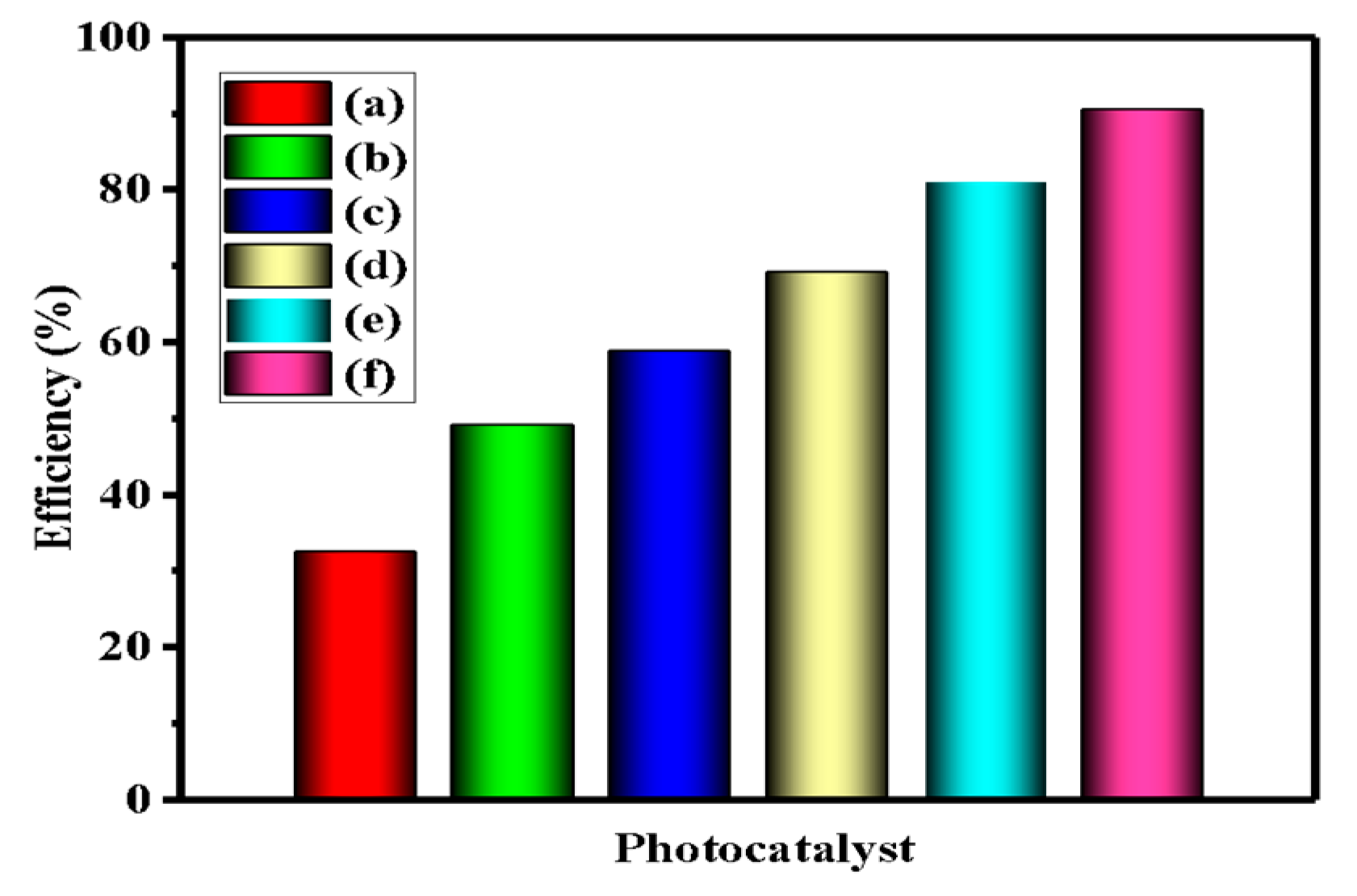
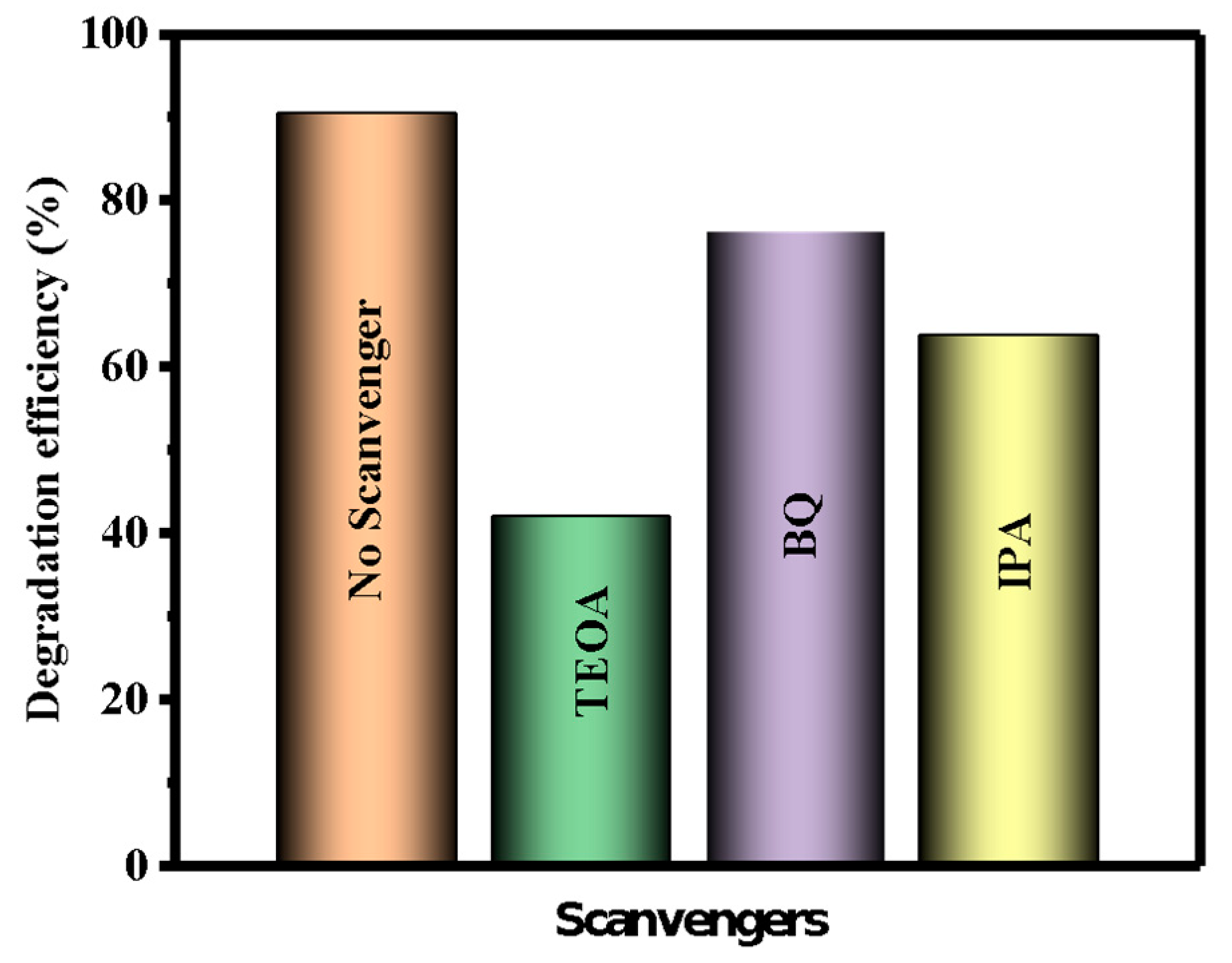
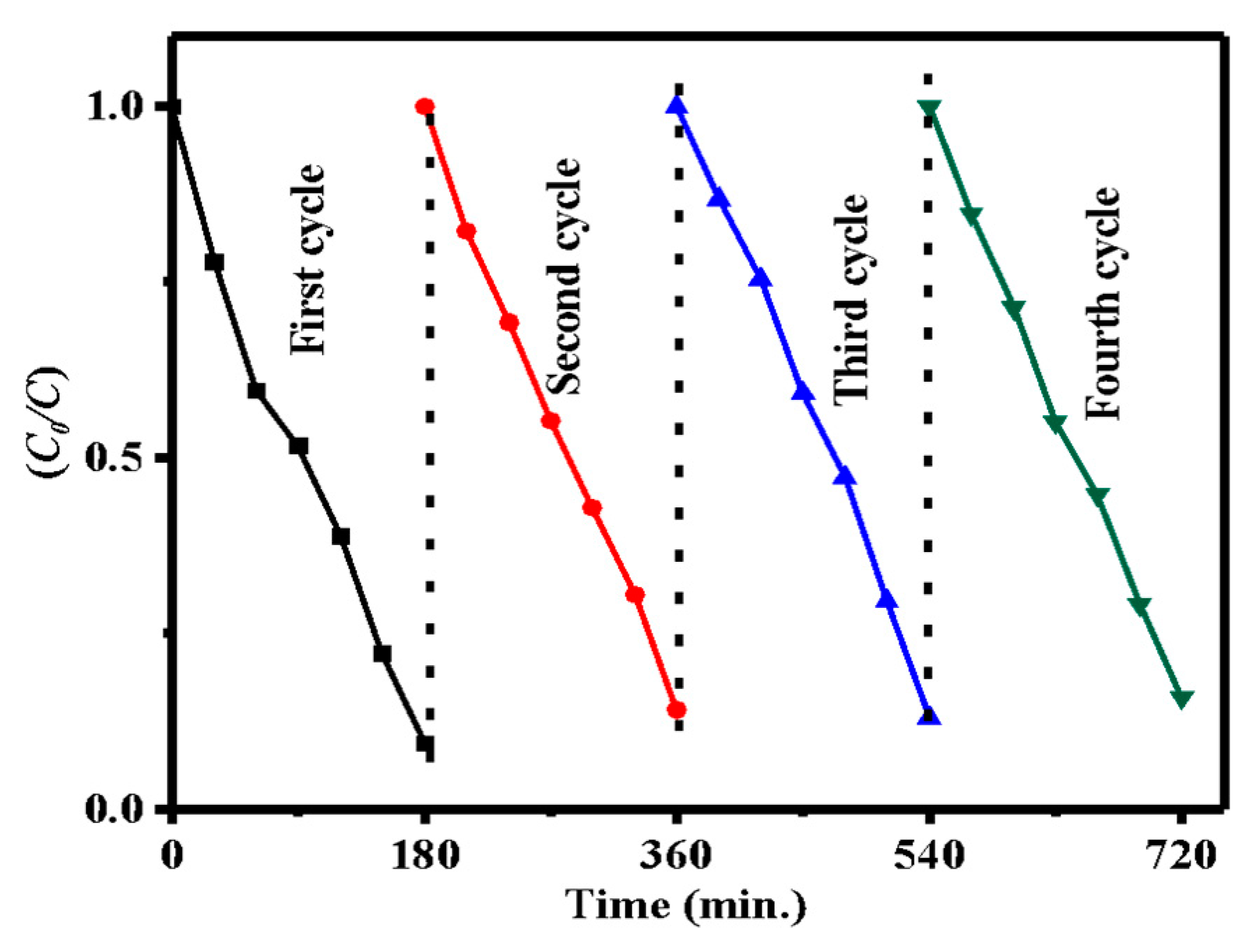
| Sample | Bare ZnO | Bare g-CN | g-CN/ZnO (60%) | g-CN/ZnO (65%) | g-CN/ZnO (70%) | g-CN/ZnO (75%) |
|---|---|---|---|---|---|---|
| Intercept | −0.0155 ± 0.018 | −0.0159 ± 0.012 | −0.010 ± 0.023 | −0.0532 ± 0.026 | −0.027 ± 0.03 | 0.0217 ± 0.038 |
| Rate Constant | 0.0021 ± 0.00017 | 0.00389 ± 0.000119 | 0.00477 ± 0.00021 | 0.00658 ± 0.000242 | 0.00922 ± 0.0003295 | 0.01283 ± 0.0003542 |
| R-Square | 0.96877 | 0.99535 | 0.99045 | 0.99328 | 0.99365 | 0.9962 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ngullie, R.C.; Alaswad, S.O.; Bhuvaneswari, K.; Shanmugam, P.; Pazhanivel, T.; Arunachalam, P. Synthesis and Characterization of Efficient ZnO/g-C3N4 Nanocomposites Photocatalyst for Photocatalytic Degradation of Methylene Blue. Coatings 2020, 10, 500. https://doi.org/10.3390/coatings10050500
Ngullie RC, Alaswad SO, Bhuvaneswari K, Shanmugam P, Pazhanivel T, Arunachalam P. Synthesis and Characterization of Efficient ZnO/g-C3N4 Nanocomposites Photocatalyst for Photocatalytic Degradation of Methylene Blue. Coatings. 2020; 10(5):500. https://doi.org/10.3390/coatings10050500
Chicago/Turabian StyleNgullie, Renathung C., Saleh O. Alaswad, Kandasamy Bhuvaneswari, Paramasivam Shanmugam, Thangavelu Pazhanivel, and Prabhakarn Arunachalam. 2020. "Synthesis and Characterization of Efficient ZnO/g-C3N4 Nanocomposites Photocatalyst for Photocatalytic Degradation of Methylene Blue" Coatings 10, no. 5: 500. https://doi.org/10.3390/coatings10050500
APA StyleNgullie, R. C., Alaswad, S. O., Bhuvaneswari, K., Shanmugam, P., Pazhanivel, T., & Arunachalam, P. (2020). Synthesis and Characterization of Efficient ZnO/g-C3N4 Nanocomposites Photocatalyst for Photocatalytic Degradation of Methylene Blue. Coatings, 10(5), 500. https://doi.org/10.3390/coatings10050500






