Improvement of CoCr Alloy Characteristics by Ti-Based Carbonitride Coatings Used in Orthopedic Applications
Abstract
1. Introduction
2. Materials and Methods
2.1. Coatings Preparation
2.2. Coatings Characterization
3. Results
3.1. Elemental and Phase Composition
3.2. Morphology and Roughness (Before Corrosion)
3.3. Roughness of Surface
3.4. Hardness and Elastic Modulus
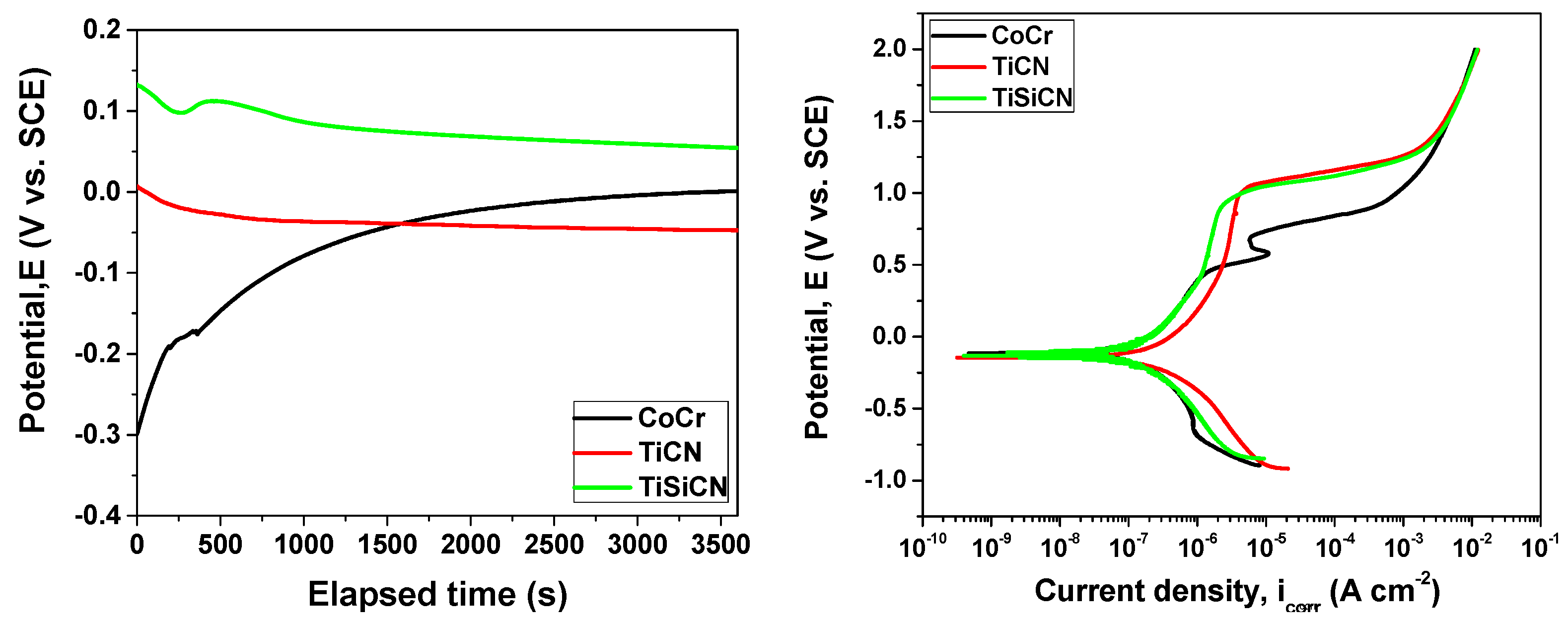
3.5. In Vitro Corrosion Investigations
3.5.1. Potentiodynamic Polarization
3.5.2. Electrochemical Impedance Spectroscopy (EIS)
3.6. Morphology and Roughness after Corrosion
4. Discussions
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Nomenclature
| Roman Symbols | |
| CPEdl | Constant phase element that models the behavior of a double layer |
| CPElayer | Constant phase element that models the behavior of a layer |
| d | Crystallite size |
| E | Elastic modulus |
| Ecorr | Corrosion potential |
| EOC | Open circuit potential |
| F | Force |
| H | Hardness |
| icorr | Corrosion current density |
| Qdl | Capacitance of the double layer |
| Qlayer | Capacitance of the layer |
| Ra | Arithmetic average of the roughness profile |
| Rct | Charge transfer resistance |
| Rel | Electrolyte resistance |
| RMS | Root mean square |
| Rp | Polarization resistance |
| Rpore | Resistance associated to the current flow through the pores |
| Rq | Root means square average of the roughness profile |
| Sk | Skewness |
| T(hkl) | Texture coefficient |
| Z | Impedance |
| Zim | Imaginary part of impedance |
| Zre | Real part of impedance |
| Greek Symbols | |
| 2θ | Angle between incident beam and reflected beam |
| α dl | Exponent equaling 1 for a capacitor characteristic to the double layer |
| α layer | Exponent equaling 1 for a capacitor characteristic to the layer |
| βa | Anodic Beta coefficient of Tafel slope |
| βc | Cathodic Beta coefficient of Tafel slope |
| ε | Strain |
| χ2 | Chi-square statistic distribution |
| Acronyms | |
| CE | Counter electrode |
| CVD | Chemical vapor deposition |
| DMEM | Dulbecco’s Modified Eagle’s Medium |
| EDS | Energy dispersive spectrometry |
| EIS | Electrochemical impedance spectroscopy |
| FBS | Fetal bovine serum |
| FCC | Face centered cubic structure |
| JCPDS | Joint committee on powder diffraction standards |
| PVD | Physical vapor deposition |
| RE | Reference electrode |
| SCE | Saturated Calomel electrode |
| SEM | Scanning electron microscopy |
| XRD | X-Ray Diffraction |
References
- Garcia, J.A.; Diaz, C.; Mandl, S.; Lutz, J.; Martinez, R.; Rodriguez, R.J. Tribological improvements of plasma immersion implanted CoCr alloys. Surf. Coat. Technol. 2010, 204, 2928–2932. [Google Scholar] [CrossRef]
- Okazaki, Y.; Gotoh, E. Comparison of metal release from various metallic biomaterials in vitro. Biomaterials 2005, 26, 11–21. [Google Scholar] [CrossRef] [PubMed]
- Sahoo, P.; Das, S.K.; Paulo Davim, J. 1—Tribology of materials for biomedical applications. In Mechanical Behaviour of Biomaterials; Davim, J.P., Ed.; Woodhead Publishing: Cambridge, UK, 2019; pp. 1–45. [Google Scholar] [CrossRef]
- Goharian, A.; Kadir, M.R.A.; Abdullah, M.R. Trauma Plating Systems: Biomechanical, Material, Biological, and Clinical Aspects; Elsevier Science: Amsterdam, The Netherlands, 2017. [Google Scholar]
- Lamerain, M.; Bachy, M.; Delpont, M.; Kabbaj, R.; Mary, P.; Vialle, R. CoCr rods provide better frontal correction of adolescent idiopathic scoliosis treated by all-pedicle screw fixation. Eur. Spine J. 2014, 23, 1190–1196. [Google Scholar] [CrossRef] [PubMed]
- Serhan, H.; Mhatre, D.; Newton, P.; Giorgio, P.; Sturm, P. Would CoCr Rods Provide Better Correctional Forces Than Stainless Steel or Titanium for Rigid Scoliosis Curves? J. Spinal Disord. Tech. 2013, 26, E70–E74. [Google Scholar] [CrossRef] [PubMed]
- Hanawa, T. 1—Overview of metals and applications. In Metals for Biomedical Devices; Niinomi, M., Ed.; Woodhead Publishing: Cambridge, UK, 2010; pp. 3–24. [Google Scholar] [CrossRef]
- Minnath, M.A. 7—Metals and alloys for biomedical applications. In Fundamental Biomaterials: Metals; Balakrishnan, P., Sreekala, M.S., Thomas, S., Eds.; Woodhead Publishing: Cambridge, UK, 2018; pp. 167–174. [Google Scholar] [CrossRef]
- Nine, M.J.; Choudhury, D.; Hee, A.C.; Mootanah, R.; Abu Osman, N.A. Wear Debris Characterization and Corresponding Biological Response: Artificial Hip and Knee Joints. Materials 2014, 7, 980–1016. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.T.; Ge, S.R.; Cao, S.F.; Wang, S.B. Comparison of wear debris generated from ultra high molecular weight polyethylene in vivo and in artificial joint simulator. Wear 2011, 271, 647–652. [Google Scholar] [CrossRef]
- Mattei, L.; Di Puccio, F.; Piccigallo, B.; Ciulli, E. Lubrication and wear modelling of artificial hip joints: A review. Tribol. Int. 2011, 44, 532–549. [Google Scholar] [CrossRef]
- Fujiwara, N.; Kobayashi, K. Macrophages in inflammation. Curr Drug Targets Inflamm Allergy 2005, 4, 281–286. [Google Scholar] [CrossRef]
- Man, K.; Jiang, L.H.; Foster, R.; Yang, X.B.B. Immunological Responses to Total Hip Arthroplasty. J. Funct. Biomater. 2017, 8, 33. [Google Scholar] [CrossRef]
- Perez-Maceda, B.T.; Lopez-Fernandez, M.E.; Diaz, I.; Kavanaugh, A.; Billi, F.; Escudero, M.L.; Garcia-Alonso, M.C.; Lozano, R.M. Macrophage Biocompatibility of CoCr Wear Particles Produced under Polarization in Hyaluronic Acid Aqueous Solution. Materials 2018, 11, 756. [Google Scholar] [CrossRef]
- Hauer, G.; Leitner, L.; Ackerl, C.M.; Klim, S.; Vielgut, I.; Ehall, R.; Glehr, M.; Leithner, A.; Sadoghi, P. Titanium-Nitride Coating Does Not Result in a Better Clinical Outcome Compared to Conventional Cobalt-Chromium Total Knee Arthroplasty after a Long-Term Follow-Up: A Propensity Score Matching Analysis. Coatings 2020, 10, 442. [Google Scholar] [CrossRef]
- Eltit, F.; Assiri, A.; Garbuz, D.; Duncan, C.; Masri, B.; Greidanus, N.; Bell, R.; Sharma, M.; Cox, M.; Wang, R.Z. Adverse reactions to metal on polyethylene implants: Highly destructive lesions related to elevated concentration of cobalt and chromium in synovial fluid. J. Biomed. Mater. Res. Part A 2017, 105, 1876–1886. [Google Scholar] [CrossRef]
- José, A.; García, P.J.R.; Rocío, O.; Iban, Q.; Rafael, J. Rodríguez. Advanced Surface Treatments for Improving the Biocompatibility of Prosthesis and Medical Implants. In Advanced Surface Engineering Research; Chowdhury, M.A., Ed.; IntechOpen: London, UK, 2018. [Google Scholar] [CrossRef]
- Anders, A. Energetic deposition using filtered cathodic arc plasmas. Vacuum 2002, 67, 673–686. [Google Scholar] [CrossRef]
- Sanders, D.M.; Anders, A. Review of cathodic arc deposition technology at the start of the new millennium. Surf. Coat. Technol. 2000, 133, 78–90. [Google Scholar] [CrossRef]
- Setsuhara, Y. 4.12—Plasma Sources in Thin Film Deposition. In Comprehensive Materials Processing; Hashmi, S., Batalha, G.F., Van Tyne, C.J., Yilbas, B., Eds.; Elsevier: Oxford, UK, 2014; pp. 307–324. [Google Scholar] [CrossRef]
- Karlsson, L.; Hultman, L.; Johansson, M.P.; Sundgren, J.E.; Ljungcrantz, H. Growth, microstructure, and mechanical properties of arc evaporated TiCxN1-x (0 <= x <= 1). Surf. Coat. Technol. 2000, 126, 1–14. [Google Scholar] [CrossRef]
- Liskiewicz, T.; Rybiak, R.; Fouvry, S.; Wendler, B. Fretting wear of Ti(CxNy) PVD coatings under variable environmental conditions. Proc. Inst. Mech. Eng. Part J-J. Eng. Tribol. 2006, 220, 125–134. [Google Scholar] [CrossRef]
- Lotfi, M.; Ashrafi, H.; Amini, S.; Farid, A.A.; Jahanbakhsh, M. Characterization of various coatings on wear suppression in turning of Inconel 625: A three-dimensional numerical simulation. Proc. Inst. Mech. Eng. Part J-J. Eng. Tribol. 2017, 231, 734–744. [Google Scholar] [CrossRef]
- Chandrashekharaiah, T.M.; Kori, S.A. Effect of grain refinement and modification on the dry sliding wear behaviour of eutectic Al-Si alloys. Tribol. Int. 2009, 42, 59–65. [Google Scholar] [CrossRef]
- Shtansky, D.V.; Gloushankova, N.A.; Sheveiko, A.N.; Kiryukhantsev-Korneev, P.V.; Bashkova, I.A.; Mavrin, B.N.; Ignatov, S.G.; Filippovich, S.Y.; Rojas, C. Si-doped multifunctional bioactive nanostructured films. Surf. Coat. Technol. 2010, 205, 728–739. [Google Scholar] [CrossRef]
- Shi, Z.F.; Wang, Y.J.; Du, C.; Huang, N.; Wang, L.; Ning, C.Y. Silicon nitride films for the protective functional coating: Blood compatibility and biomechanical property study. J. Mech. Behav. Biomed. Mater. 2012, 16, 9–20. [Google Scholar] [CrossRef]
- Endler, I.; Hohn, M.; Schmidt, J.; Scholz, S.; Herrmann, M.; Knaut, M. Ternary and quarternary TiSiN and TiSiCN nanocomposite coatings obtained by Chemical Vapor Deposition. Surf. Coat. Technol. 2013, 215, 133–140. [Google Scholar] [CrossRef]
- Hatem, A.; Lin, J.L.; Wei, R.H.; Torres, R.D.; Laurindo, C.; de Souza, G.B.; Soares, P. Tribocorrosion behavior of low friction TiSiCN nanocomposite coatings deposited on titanium alloy for biomedical applications. Surf. Coat. Technol. 2018, 347, 1–12. [Google Scholar] [CrossRef]
- Johnson, L.J.S.; Rogstrom, L.; Johansson, M.P.; Oden, M.; Hultman, L. Microstructure evolution and age hardening in (Ti,Si)(C,N) thin films deposited by cathodic arc evaporation. Thin Solid Film. 2010, 519, 1397–1403. [Google Scholar] [CrossRef]
- Wei, R.H. Plasma enhanced magnetron sputter deposition of Ti-Si-C-N based nanocomposite coatings. Surf. Coat. Technol. 2008, 203, 538–544. [Google Scholar] [CrossRef]
- Guruvenket, S.; Li, D.; Klemberg-Sapieha, J.E.; Martinu, L.; Szpunar, J. Mechanical and tribological properties of duplex treated TiN, nc-TiN/a-SiNx and nc-TiCN/a-SiCN coatings deposited on 410 low alloy stainless steel. Surf. Coat. Technol. 2009, 203, 2905–2911. [Google Scholar] [CrossRef]
- Ma, S.L.; Ma, D.Y.; Guo, Y.; Xu, B.; Wu, G.Z.; Xu, K.W.; Chu, P.K. Synthesis and characterization of super hard, self-lubricating Ti-Si-C-N nanocomposite coatings. Acta Mater. 2007, 55, 6350–6355. [Google Scholar] [CrossRef]
- Constantin, L.R.; Parau, A.C.; Balaceanu, M.; Dinu, M.; Vladescu, A. Corrosion and tribological behaviour in a 3.5% NaCl solution of vacuum arc deposited ZrCN and Zr-Cr-Si-C-N coatings. Proc. Inst. Mech. Eng. Part J-J. Eng. Tribol. 2019, 233, 158–169. [Google Scholar] [CrossRef]
- Wang, Y.; Li, J.L.; Dang, C.Q.; Wang, Y.X.; Zhu, Y.J. Influence of bias voltage on structure and tribocorrosion properties of TiSiCN coating in artificial seawater. Mater. Charact. 2017, 127, 198–208. [Google Scholar] [CrossRef]
- Sundgren, J.E.; Johansson, B.O.; Hentzell, H.T.G.; Karlsson, S.E. Mechanisms of reactive sputtering of titanium nitride and titanium carbide.3. Influence of substrate bias on composition and structure. Thin Solid Film. 1983, 105, 385–393. [Google Scholar] [CrossRef]
- Saoula, N.; Henda, K.; Kesri, R. Effect Of The Plasma Deposition Parameters On The Properties Of Ti/TiN Multilayers For Hard Coatings Applications. In Proceedings of the 1st International Conference on Laser and Plasma Applications in Materials Science, Algiers, Algeria, 23–26 June 2008; p. 256. [Google Scholar]
- Constantin, L.; Braic, M.; Dinu, M.; Balaceanu, M.; Braic, V.; Farcau, C.; Vladescu, A. Effects of Zr, Nb, or Si addition on the microstructural, mechanical, and corrosion resistance of TiCN hard coatings. Mater. Corros. -Werkst. Und Korros. 2016, 67, 929–938. [Google Scholar] [CrossRef]
- Ritchie, R.O. The conflicts between strength and toughness. Nat. Mater. 2011, 10, 817–822. [Google Scholar] [CrossRef] [PubMed]
- Grieveson, P. An investigation of the Ti-C-N system. Proc. Br. Ceram. Soc. 1967, 8, 137–153. [Google Scholar]
- Terry, B.S.; Grieveson, P. Observations on the Role of Interfacial Phenomena in Materials Processing. ISIJ Int. 1993, 33, 166–175. [Google Scholar] [CrossRef]
- Levi, G.; Kaplan, W.D.; Bamberger, M. Structure refinement of titanium carbonitride (TiCN). Mater. Lett. 1998, 35, 344–350. [Google Scholar] [CrossRef]
- Nietubyc, R.; Lorkiewicz, J.; Sekutowicz, J.; Smedley, J.; Kosinska, A. Optimization of cathodic arc deposition and pulsed plasma melting techniques for growing smooth superconducting Pb photoemissive films for SRF injectors. Nucl. Instrum. Methods Phys. Res. Sect. A-Accel. Spectrometers Detect. Assoc. Equip. 2018, 891, 78–86. [Google Scholar] [CrossRef]
- Panjan, P.; Drnovšek, A.; Gselman, P.; Čekada, M.; Panjan, M. Review of Growth Defects in Thin Films Prepared by PVD Techniques. Coatings 2020, 10, 447. [Google Scholar] [CrossRef]
- ISO 2517:1974 Diacetone Alcohol for Industrial Use–List of Methods of Test. Edition No. 3, Publication Date: 1974–04. Available online: https://www.iso.org/standard/7452.html (accessed on 21 May 2020).
- Schuh, C.A. Nanoindentation studies of materials. Mater. Today 2006, 9, 32–40. [Google Scholar] [CrossRef]
- Fujisawa, N.; Lukomski, M. Nanoindentation near the edge of a viscoelastic solid with a rough surface. Mater. Des. 2019, 184. [Google Scholar] [CrossRef]
- Fischer-Cripps, A.C. Critical review of analysis and interpretation of nanoindentation test data. Surf. Coat. Technol. 2006, 200, 4153–4165. [Google Scholar] [CrossRef]
- Pana, I.; Vitelaru, C.; Kiss, A.; Zoita, N.C.; Dinu, M.; Braic, M. Design, fabrication and characterization of TiO2-SiO2 multilayer with tailored color glazing for thermal solar collectors. Mater. Des. 2017, 130, 275–284. [Google Scholar] [CrossRef]
- Martinez, E.; Romero, J.; Lousa, A.; Esteve, J. Nanoindentation stress-strain curves as a method for thin-film complete mechanical characterization: Application to nanometric CrN/Cr multilayer coatings. Appl. Phys. A-Mater. Sci. Process. 2003, 77, 419–426. [Google Scholar] [CrossRef]
- Shan, L.; Wang, Y.X.; Li, J.L.; Li, H.; Wu, X.D.; Chen, J.M. Tribological behaviours of PVD TiN and TiCN coatings in artificial seawater. Surf. Coat. Technol. 2013, 226, 40–50. [Google Scholar] [CrossRef]
- Li, W.; Liu, P.; Xue, Z.H.; Ma, F.C.; Zhang, K.; Chen, X.H.; Feng, R.; Liaw, P.K. Microstructures, mechanical behavior and strengthening mechanism of TiSiCN nanocomposite films. Sci. Rep. 2017, 7. [Google Scholar] [CrossRef]
- Musil, J.; Jilek, R.; Meissner, M.; Tolg, T.; Cerstvy, R. Two-phase single layer Al-O-N nanocomposite films with enhanced resistance to cracking. Surf. Coat. Technol. 2012, 206, 4230–4234. [Google Scholar] [CrossRef]
- Musil, J.; Jirout, M. Toughness of hard nanostructured ceramic thin films. Surf. Coat. Technol. 2007, 201, 5148–5152. [Google Scholar] [CrossRef]
- Pauleau, Y.; Thiery, E. Deposition and characterization of nanostructured metal/carbon composite films. Surf. Coat. Technol. 2004, 180, 313–322. [Google Scholar] [CrossRef]
- Cordoba-Torres, P. Relationship between constant-phase element (CPE) parameters and physical properties of films with a distributed resistivity. Electrochim. Acta 2017, 225, 592–604. [Google Scholar] [CrossRef]
- Hirschorn, B.; Orazem, M.E.; Tribollet, B.; Vivier, V.; Frateur, I.; Musiani, M. Determination of effective capacitance and film thickness from constant-phase-element parameters. Electrochim. Acta 2010, 55, 6218–6227. [Google Scholar] [CrossRef]
- Carlos Valero, V.; Anna Igual, M. Electrochemical Aspects in Biomedical Alloy Characterization: Electrochemical Impedance Spectrosopy; IntechOpen: London, UK, 2011. [Google Scholar] [CrossRef]
- Dinu, M.; Hauffman, T.; Cordioli, C.; Vladescu, A.; Braic, M.; Hubin, A.; Cotrut, C.M. Protective performance of Zr and Cr based silico-oxynitrides used for dental applications by means of potentiodynamic polarization and odd random phase multisine electrochemical impedance spectroscopy. Corros. Sci. 2017, 115, 118–128. [Google Scholar] [CrossRef]
- Reid, S.A.; Eden, D.A. Assessment of Corrosion. U.S. Patent No. 6,264,824, 24 July 2001. [Google Scholar]
- Toloei, A.a.S.V.a.N.D. The Relationship Between Surface Roughness and Corrosion. Asme Int. Mech. Eng. Congr. Expo. Proc. (IMECE) 2013, 2. [Google Scholar] [CrossRef]
- Musil, J. Hard and superhard nanocomposite coatings. Surf. Coat. Technol. 2000, 125, 322–330. [Google Scholar] [CrossRef]
- Roy, M.E.; Whiteside, L.A.; Xu, J.; Katerberg, B.J. Diamond-like carbon coatings enhance the hardness and resilience of bearing surfaces for use in joint arthroplasty. Acta Biomater. 2010, 6, 1619–1624. [Google Scholar] [CrossRef] [PubMed]
- Leyland, A.; Matthews, A. On the significance of the H/E ratio in wear control: A nanocomposite coating approach to optimised tribological behaviour. Wear 2000, 246, 1–11. [Google Scholar] [CrossRef]
- Ievlev, V.M.; Kostyuchenko, A.V.; Darinskii, B.M.; Barinov, S.M. Hardness and microplasticity of nanocrystalline and amorphous calcium phosphate coatings. Phys. Solid State 2014, 56, 321–329. [Google Scholar] [CrossRef]
- Surmeneva, M.A.; Tyurin, A.I.; Teresov, A.D.; Koval, N.N.; Pirozhkova, T.S.; Shuvarin, I.A.; Surmenev, R.A. Combined effect of pulse electron beam treatment and thin hydroxyapatite film on mechanical features of biodegradable AZ31 magnesium alloy. In Proceedings of the 3rd International Youth Conference on Interdisciplinary Problems of Nanotechnology, Biomedicine and Nanotoxicology (Nanobiotech), Tambov, Russia, 21–22 May 2015. [Google Scholar]
- Franceschini, D.F.; Achete, C.A.; Freire, F.L. Internal-Stress Reduction by Nitrogen Incorporation in Hard Amorphous-Carbon Thin-Films. Appl. Phys. Lett. 1992, 60, 3229–3231. [Google Scholar] [CrossRef]
- Sun, C.Q. Size dependence of nanostructures: Impact of bond order deficiency. Prog. Solid State Chem. 2007, 35, 1–159. [Google Scholar] [CrossRef]
- Sun, C.Q.; Shi, Y.; Li, C.M.; Li, S.; Yeung, T.C.A. Size-induced undercooling and overheating in phase transitions in bare and embedded clusters. Phys. Rev. B 2006, 73. [Google Scholar] [CrossRef]
- Liu, C.; Bi, Q.; Leyland, A.; Matthews, A. An electrochemical impedance spectroscopy study of the corrosion behaviour of PVD coated steels in 0.5 N NaCl aqueous solution: Part II. EIS interpretation of corrosion behaviour. Corros. Sci. 2003, 45, 1257–1273. [Google Scholar] [CrossRef]
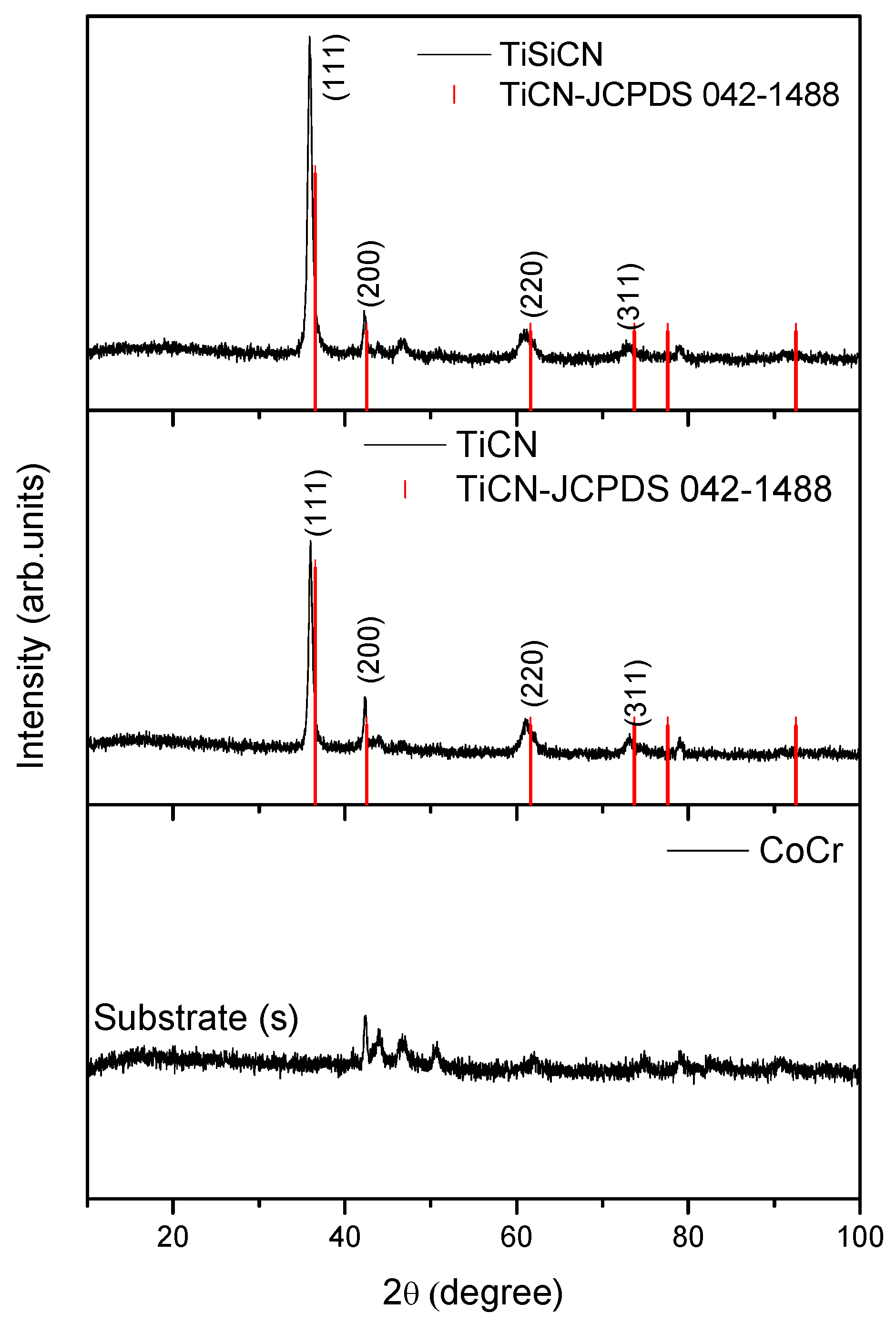




| Deposition Parameters | TiCN | TiSiCN |
|---|---|---|
| Base pressure | 2 × 10−3 Pa | |
| Working pressure | 2 × 10−2 Pa | 6 × 10−2 Pa |
| CH4 mass flow rate | 80 sccm | |
| N2 mass flow | 120 sccm | |
| Arc current on each cathode | 90 A | |
| Substrate bias voltage | −150 V | |
| Deposition duration | 40 min | |
| Coating | Elemental Composition (at.%) | (C+N)/ Σ(Me+Si) | T(hkl) | d (nm) | ε | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Ti | Si | C | N | (111) | (200) | (220) | (311) | ||||
| TiCN | 52.1 | - | 17.5 | 30.4 | 0.9 | 0.59 | 0.09 | 0.24 | 0.07 | 16.4 | 0.053 |
| TiSiCN | 48.4 | 3.4 | 20.1 | 28.1 | 0.9 | 0.73 | 0.06 | 0.17 | 0.05 | 14.6 | 0.012 |
| Sample | CoCr | TiCN | TiSiCN |
|---|---|---|---|
| Ra (nm) | 46.9 ± 5.9 | 534.6 ± 111.4 | 499.7 ± 28.9 |
| Rq (nm) | 59.5 ± 7.0 | 746.8 ± 181.9 | 650.4 ± 30.7 |
| Sk | 0.2 ± 0.4 | 2.1 ± 0.3 | 1.6 ± 0.1 |
| Substrate/ Coating | Eoc (mV) | Rp (kΩ cm2) | Ecorr (mV) | βa (mV dec−1) | βc (mV dec−1) | icorr (nA cm−2) |
|---|---|---|---|---|---|---|
| CoCr | 1 | 455 | −36 | 516 | 447 | 198.2 |
| TiCN | −48 | 216 | −103 | 517 | 288 | 233.0 |
| TiSiCN | 54 | 425 | −14 | 271 | 269 | 49.6 |
| Substrate/ Coating | Rel (Ω·cm2) | Qlayer (μFs(α−1) cm−2) | α Layer | Rpore (kΩ·cm2) | Qdl (μFs(α−1) cm−2) | α dl | Rct (kΩ·cm2) | χ2 |
|---|---|---|---|---|---|---|---|---|
| CoCr | 127 | 1.94 | 0.96 | 88 | 3.66 | 0.91 | 243,830 | 13 × 10−3 |
| TiCN | 113 | 16.19 | 0.87 | 216 | 2.19 | 0.99 | 112,000 | 3 × 10−3 |
| TiSiCN | 101 | 12.38 | 0.87 | 316 | 1.21 | 0.96 | 262,750 | 5 × 10−3 |
| Sample | CoCr | TiCN | TiSiCN |
|---|---|---|---|
| Ra (nm) | 1342.9 ± 192.4 | 15,584.3 ± 7462.8 | 6297.9 ± 1598.3 |
| Rq (nm) | 1758.2 ± 237.2 | 19,896.6 ± 6410.9 | 7480.5 ± 1567.1 |
| Sk | −0.1 ± 0.2 | −1.2 ± 0.8 | −0.7 ± 0.4 |
| Kurtosis | 2.4 ± 0.3 | 1.8 ± 0.4 | 0.7 ± 0.3 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dinu, M.; Pana, I.; Scripca, P.; Sandu, I.G.; Vitelaru, C.; Vladescu, A. Improvement of CoCr Alloy Characteristics by Ti-Based Carbonitride Coatings Used in Orthopedic Applications. Coatings 2020, 10, 495. https://doi.org/10.3390/coatings10050495
Dinu M, Pana I, Scripca P, Sandu IG, Vitelaru C, Vladescu A. Improvement of CoCr Alloy Characteristics by Ti-Based Carbonitride Coatings Used in Orthopedic Applications. Coatings. 2020; 10(5):495. https://doi.org/10.3390/coatings10050495
Chicago/Turabian StyleDinu, Mihaela, Iulian Pana, Petronela Scripca, Ioan Gabriel Sandu, Catalin Vitelaru, and Alina Vladescu. 2020. "Improvement of CoCr Alloy Characteristics by Ti-Based Carbonitride Coatings Used in Orthopedic Applications" Coatings 10, no. 5: 495. https://doi.org/10.3390/coatings10050495
APA StyleDinu, M., Pana, I., Scripca, P., Sandu, I. G., Vitelaru, C., & Vladescu, A. (2020). Improvement of CoCr Alloy Characteristics by Ti-Based Carbonitride Coatings Used in Orthopedic Applications. Coatings, 10(5), 495. https://doi.org/10.3390/coatings10050495





.jpg)

