Fabrication of Composite Ultrafiltration Membrane by Coating Urea Formaldehyde Resin on Filter Paper
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of the Composite Membranes
2.3. Determination of Porosity
2.4. Determination of Pure Water Flux
2.5. Determination of Rejection Ratio
2.6. Morphological Study Methods
3. Results and Discussion
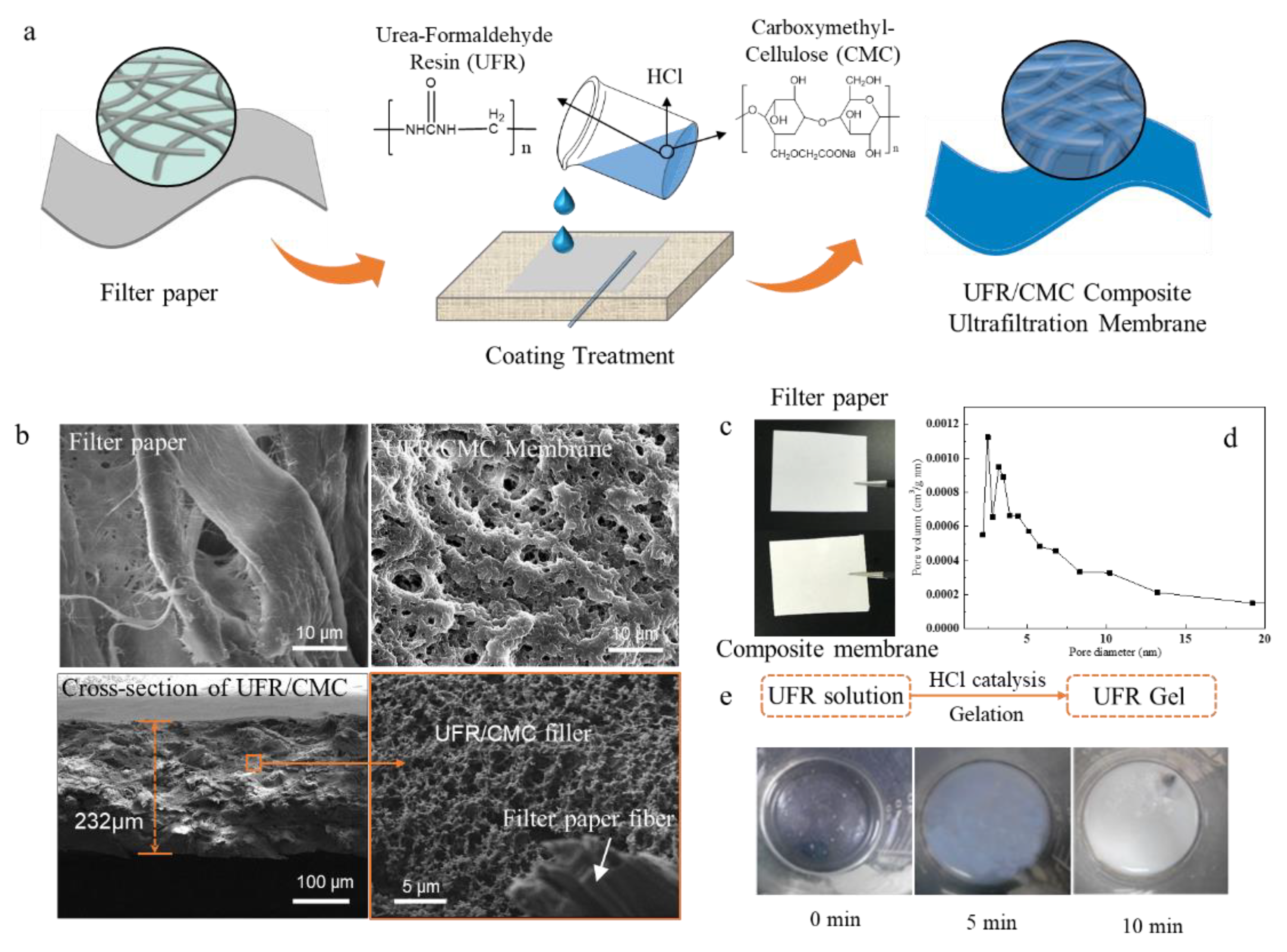
3.1. Preparation of Paper-Based UFR/CMC Composite Ultrafiltration Membrane and Its Formation Mechanism
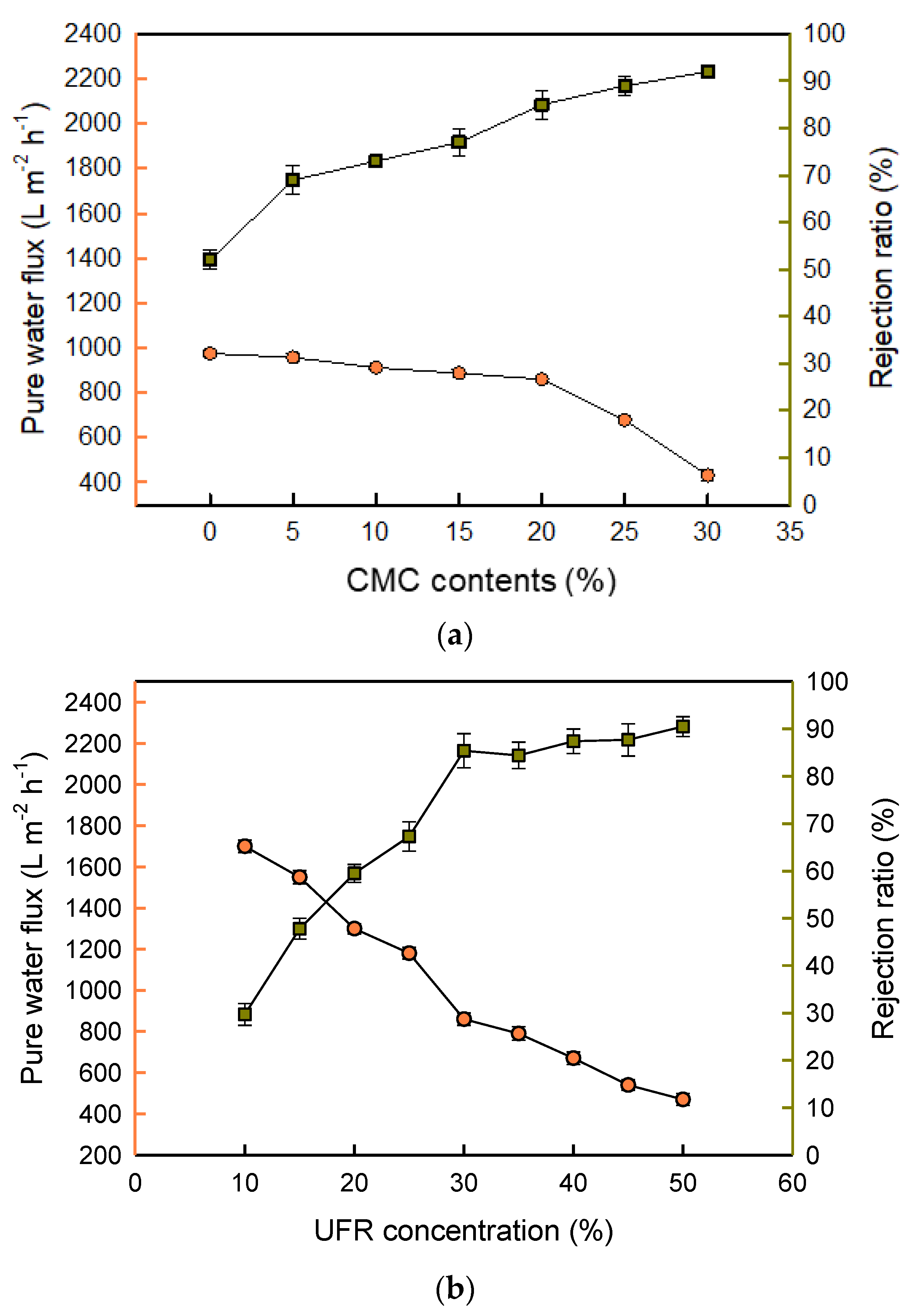
3.2. Effects of CMC Content and UFR Concentration on the Performance of the Composite Membrane
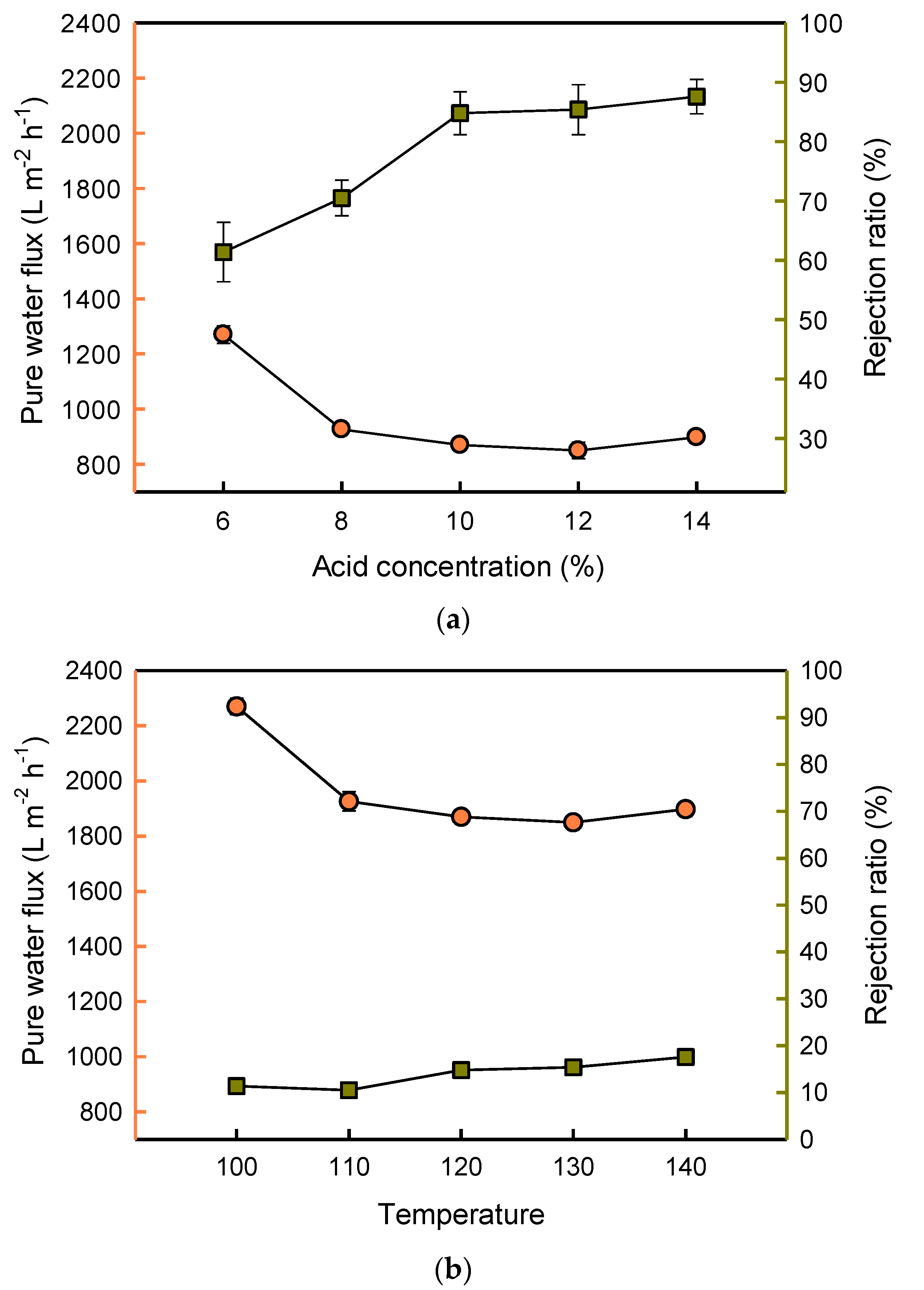
3.3. Effects of Acid on the Performance of the Composite Membrane
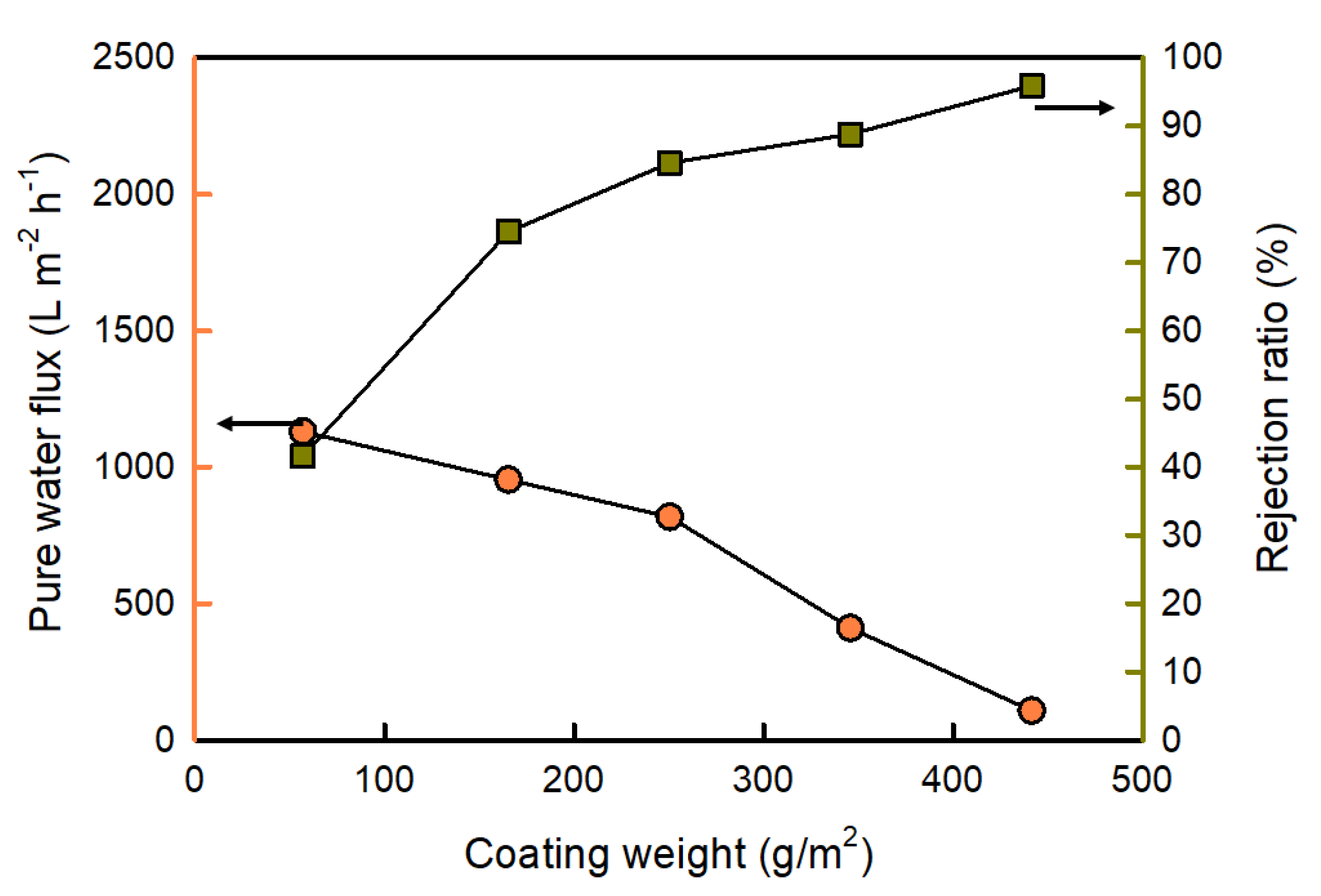
3.4. Effect of Coating Weight on the Membrane
3.5. Comparison of the Prepared Membranes with Those in the Literature
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Yong, M.; Zhang, Y.; Sun, S.; Liu, W. Properties of polyvinyl chloride (PVC) ultrafiltration membrane improved by lignin: Hydrophilicity and antifouling. J. Membr. Sci. 2019, 575, 50–59. [Google Scholar] [CrossRef]
- Huang, H.; Schwab, K.; Jacangelo, J.G. Pretreatment for Low Pressure Membranes in Water Treatment: A Review. Environ. Sci. Technol. 2009, 43, 3011–3019. [Google Scholar] [CrossRef] [PubMed]
- He, Z.; Ng, T.C.A.; Lyu, Z.; Gu, Q.; Zhang, L.; Ng, H.Y.; Wang, J. Alumina double-layered ultrafiltration membranes with enhanced water flux. Colloids Surf. A Physicochem. Eng. Asp. 2020, 587, 124324. [Google Scholar] [CrossRef]
- Shi, Q.; Su, Y.; Zhu, S.; Li, C.; Zhao, Y.; Jiang, Z. A facile method for synthesis of pegylated polyethersulfone and its application in fabrication of antifouling ultrafiltration membrane. J. Membr. Sci. 2007, 303, 204–212. [Google Scholar] [CrossRef]
- Jana, S.; Saikia, A.; Purkait, M.K.; Mohanty, K. Chitosan based ceramic ultrafiltration membrane: Preparation, characterization and application to remove Hg(II) and As(III) using polymer enhanced ultrafiltration. Chem. Eng. J. 2011, 170, 209–219. [Google Scholar] [CrossRef]
- Sun, Z.; Chen, H.; Ren, X.; Zhang, Z.; Guo, L.; Zhang, F.; Cheng, H. Preparation and application of zinc oxide/poly(m-phenylene isophthalamide) hybrid ultrafiltration membranes. J. Appl. Polym. Sci. 2019, 136, 47583. [Google Scholar] [CrossRef]
- Chen, W.; Peng, J.; Su, Y.; Zheng, L.; Wang, L.; Jiang, Z. Separation of oil/water emulsion using Pluronic F127 modified polyethersulfone ultrafiltration membranes. Sep. Purif. Technol. 2009, 66, 591–597. [Google Scholar] [CrossRef]
- Qiu, Z.; Ji, X.; He, C. Fabrication of a loose nanofiltration candidate from Polyacrylonitrile/Graphene oxide hybrid membrane via thermally induced phase separation. J. Hazard. Mater. 2018, 360, 122–131. [Google Scholar] [CrossRef]
- He, X.; Chen, C.; Jiang, Z.; Su, Y. Computer simulation of formation of polymeric ultrafiltration membrane via immersion precipitation. J. Membr. Sci. 2011, 371, 108–116. [Google Scholar] [CrossRef]
- Altinkaya, S. Modeling of asymmetric membrane formation by dry-casting method. J. Membr. Sci. 2004, 230, 71–89. [Google Scholar] [CrossRef][Green Version]
- Dehban, A.; Kargari, A.; Zokaee Ashtiani, F. Preparation and characterization of an antifouling poly (phenyl sulfone) ultrafiltration membrane by vapor-induced phase separation technique. Sep. Purif. Technol. 2019, 212, 986–1000. [Google Scholar] [CrossRef]
- Zhang, X.; Chen, Y.; Konsowa, A.H.; Zhu, X.; Crittenden, J.C. Evaluation of an innovative polyvinyl chloride (PVC) ultrafiltration membrane for wastewater treatment. Sep. Purif. Technol. 2009, 70, 71–78. [Google Scholar] [CrossRef]
- Mahanty, B.; Satpati, A.K.; Kumar, S.; Leoncini, A.; Huskens, J.; Verboom, W.; Mohapatra, P.K. Development of polyvinyl chloride (PVC)-based highly efficient potentiometric sensors containing two benzene-centered tripodal diglycolamides as ionophores. Sens. Actuators B Chem. 2020, 318, 127961. [Google Scholar] [CrossRef]
- Freger, V. Nanoscale heterogeneity of polyamide membranes formed by interfacial polymerization. Langmuir 2003, 19, 4791–4797. [Google Scholar] [CrossRef]
- Paseta, L.; Luque-Alled, J.M.; Malankowska, M.; Navarro, M.; Gorgojo, P.; Coronas, J.; Téllez, C. Functionalized graphene-based polyamide thin film nanocomposite membranes for organic solvent nanofiltration. Sep. Purif. Technol. 2020, 247, 116995. [Google Scholar] [CrossRef]
- Kaur, H.; Bulasara, V.K.; Gupta, R.K. Influence of pH and temperature of dip-coating solution on the properties of cellulose acetate-ceramic composite membrane for ultrafiltration. Carbohydr. Polym. 2018, 195, 613–621. [Google Scholar] [CrossRef] [PubMed]
- Zavastin, D.; Cretescu, I.; Bezdadea, M.; Bourceanu, M.; Drăgan, M.; Lisa, G.; Mangalagiu, I.; Vasić, V.; Savić, J. Preparation, characterization and applicability of cellulose acetate–polyurethane blend membrane in separation techniques. Colloids Surf. A Physicochem. Eng. Asp. 2010, 370, 120–128. [Google Scholar] [CrossRef]
- Yan, S.; Song, W.; Lu, J.; Wang, J.; Zheng, Y.; Xiao, R. Post-fabrication modifications of thermoplastic polymeric nanofiber membranes with electroactive polymers for triboelectric nanogenerators. Nano Energy 2019, 59, 697–704. [Google Scholar] [CrossRef]
- Shao, J.; Qin, S.; Davidson, J.; Li, W.; He, Y.; Zhou, H.S. Recovery of nickel from aqueous solutions by complexation-ultrafiltration process with sodium polyacrylate and polyethylenimine. J. Hazard. Mater. 2013, 244, 472–477. [Google Scholar] [CrossRef]
- Ouni, H.; Dhahbi, M. Spectrometric study of crystal violet in presence of polyacrylic acid and polyethylenimine and its removal by polyelectrolyte enhanced ultrafiltration. Sep. Purif. Technol. 2010, 72, 340–346. [Google Scholar] [CrossRef]
- Mbareck, C.; Nguyen, Q.T.; Alaoui, O.T.; Barillier, D. Elaboration, characterization and application of polysulfone and polyacrylic acid blends as ultrafiltration membranes for removal of some heavy metals from water. J. Hazard. Mater. 2009, 171, 93–101. [Google Scholar] [PubMed]
- Cheng, H.; Lan, J.-F.; Wei, G.-H.; Huang, W.-H.; Cheng, J.-K. Study on antifouling performance of single-walled carbon nanotubes modified electrode and its application in determination of 5-hydroxytryptamine. Chin. J. Anal. Chem. 2013, 41, 540. [Google Scholar] [CrossRef]
- Luo, M.-L.; Zhao, J.-Q.; Tang, W.; Pu, C.-S. Hydrophilic modification of poly(ether sulfone) ultrafiltration membrane surface by self-assembly of TiO2 nanoparticles. Appl. Surf. Sci. 2005, 249, 76–84. [Google Scholar] [CrossRef]
- Yan, L.; Li, Y.S.; Xiang, C.B.; Xianda, S. Effect of nano-sized Al2O3-particle addition on PVDF ultrafiltration membrane performance. J. Membr. Sci. 2006, 276, 162–167. [Google Scholar] [CrossRef]
- Yu, H.; Zhang, X.; Zhang, Y.; Liu, J.; Zhang, H. Development of a hydrophilic PES ultrafiltration membrane containing SiO2@N-Halamine nanoparticles with both organic antifouling and antibacterial properties. Desalination 2013, 326, 69–76. [Google Scholar] [CrossRef]
- Yan, X.; Qian, X.; Chang, Y. Preparation and characterization of urea formaldehyde @ epoxy resin microcapsule on waterborne wood coatings. Coatings 2019, 9, 475. [Google Scholar] [CrossRef]
- Raquez, J.-M.; Deléglise, M.; Lacrampe, M.-F.; Krawczak, P. Thermosetting (bio)materials derived from renewable resources: A critical review. Prog. Polym. Sci. 2010, 35, 487–509. [Google Scholar] [CrossRef]
- Gourichon, B.; Deléglise, M.; Binetruy, C.; Krawczak, P. Dynamic void content prediction during radial injection in liquid composite molding. Compos. Part A Appl. Sci. Manuf. 2008, 39, 46–55. [Google Scholar] [CrossRef]
- Bliznakov, E.D.; White, C.C.; Shaw, M.T. Mechanical properties of blends of HDPE and recycled urea-formaldehyde resin. J. Appl. Polym. Sci. 2000, 77, 3220–3227. [Google Scholar] [CrossRef]
- Park, B.-D.; Kang, E.C.; Park, J.Y. Effects of formaldehyde to urea mole ratio on thermal curing behavior of urea–formaldehyde resin and properties of particleboard. J. Appl. Polym. Sci. 2006, 101, 1787–1792. [Google Scholar] [CrossRef]
- Soroko, I.; Bhole, Y.; Livingston, A.G. Environmentally friendly route for the preparation of solvent resistant polyimide nanofiltration membranes. Green Chem. 2011, 13, 162–168. [Google Scholar] [CrossRef]
- Varanasi, S.; Low, Z.-X.; Batchelor, W. Cellulose nanofibre composite membranes–Biodegradable and recyclable UF membranes. Chem. Eng. J. 2015, 265, 138–146. [Google Scholar] [CrossRef]
- Qin, J.-J.; Wong, F.-S.; Li, Y.; Liu, Y.-T. A high flux ultrafiltration membrane spun from PSU/PVP (K90)/DMF/1,2-propanediol. J. Membr. Sci. 2003, 211, 139–147. [Google Scholar] [CrossRef]
- Zhao, S.; Yan, W.; Shi, M.; Wang, Z.; Wang, J.; Wang, S. Improving permeability and antifouling performance of polyethersulfone ultrafiltration membrane by incorporation of ZnO-DMF dispersion containing nano-ZnO and polyvinylpyrrolidone. J. Membr. Sci. 2015, 478, 105–116. [Google Scholar] [CrossRef]
- Yue, X.; Li, J.; Zhang, T.; Qiu, F.; Yang, D.; Xue, M. In situ one-step fabrication of durable superhydrophobic-superoleophilic cellulose/LDH membrane with hierarchical structure for efficiency oil/water separation. Chem. Eng. J. 2017, 328, 117–123. [Google Scholar] [CrossRef]
- Mohamed, M.A.; Salleh, W.N.W.; Jaafar, J.; Hir, Z.A.M.; Rosmi, M.S.; Mutalib, M.A.; Ismail, A.F.; Tanemura, M. Regenerated cellulose membrane as bio-template for in-situ growth of visible-light driven C-modified mesoporous titania. Carbohydr. Polym. 2016, 146, 166–173. [Google Scholar] [CrossRef] [PubMed]
- Hu, M.-X.; Niu, H.-M.; Chen, X.-L.; Zhan, H.-B. Natural cellulose microfiltration membranes for oil/water nanoemulsions separation. Colloids Surf. A Physicochem. Eng. Asp. 2019, 564, 142–151. [Google Scholar] [CrossRef]
- Madaeni, S.S.; Heidary, F. Improving separation capability of regenerated cellulose ultrafiltration membrane by surface modification. Appl. Surf. Sci. 2011, 257, 4870–4876. [Google Scholar] [CrossRef]
- Rojo, E.; Peresin, M.S.; Sampson, W.W.; Hoeger, I.C.; Vartiainen, J.; Laine, J.; Rojas, O.J. Comprehensive elucidation of the effect of residual lignin on the physical, barrier, mechanical and surface properties of nanocellulose films. Green Chem. 2015, 17, 1853–1866. [Google Scholar] [CrossRef]
- Khan, A.; Wen, Y.; Huq, T.; Ni, Y. Cellulosic nanomaterials in food and nutraceutical applications: A review. J. Agric. Food Chem. 2018, 66, 8–19. [Google Scholar] [CrossRef]
- Lu, H.; Zhang, L.; Ma, J.; Alam, N.; Zhou, X.; Ni, Y. Nano-cellulose/MOF derived carbon doped CuO/Fe3O4 nanocomposite as high efficient catalyst for organic pollutant remedy. Nanomaterials 2019, 9, 277. [Google Scholar] [CrossRef]
- Lu, H.; Zhang, L.; Wang, B.; Long, Y.; Zhang, M.; Ma, J.; Khan, A.; Chowdhury, S.P.; Zhou, X.; Ni, Y. Cellulose-supported magnetic Fe3O4–MOF composites for enhanced dye removal application. Cellulose 2019, 26, 4909–4920. [Google Scholar] [CrossRef]
- Dai, L.; Long, Z.; Chen, J.; An, X.; Cheng, D.; Khan, A.; Ni, Y. Robust guar gum/cellulose nanofibrils multilayer films with good barrier properties. ACS Appl. Mater. Interfaces 2017, 9, 5477–5485. [Google Scholar] [CrossRef]
- Wang, J.; Zhou, X.; Ma, J. Preparation and characteristics of a paper-based ultrafiltration membrane. BioResources 2012, 7, 0545–0553. [Google Scholar]
- Jung, J.; Raghavendra, G.M.; Kim, D.; Seo, J. One-step synthesis of starch-silver nanoparticle solution and its application to antibacterial paper coating. Int. J. Biol. Macromol. 2018, 107, 2285–2290. [Google Scholar] [CrossRef]
- Gao, Q.; Wang, C.-Z.; Liu, S.; Hanigan, D.; Liu, S.-T.; Zhao, H.-Z. Ultrafiltration membrane microreactor (MMR) for simultaneous removal of nitrate and phosphate from water. Chem. Eng. J. 2019, 355, 238–246. [Google Scholar] [CrossRef]
- Ahmad, A.L.; Sarif, M.; Ismail, S. Development of an integrally skinned ultrafiltration membrane for wastewater treatment: Effect of different formulations of PSf/NMP/PVP on flux and rejection. Desalination 2005, 179, 257–263. [Google Scholar] [CrossRef]
- Xu, Z.; Li, X.; Teng, K.; Zhou, B.; Ma, M.; Shan, M.; Jiao, K.; Qian, X.; Fan, J. High flux and rejection of hierarchical composite membranes based on carbon nanotube network and ultrathin electrospun nanofibrous layer for dye removal. J. Membr. Sci. 2017, 535, 94–102. [Google Scholar] [CrossRef]
- Lianchao, L.; Baoguo, W.; Huimin, T.; Tianlu, C.; Jiping, X. A novel nanofiltration membrane prepared with PAMAM and TMC by in situ interfacial polymerization on PEK-C ultrafiltration membrane. J. Membr. Sci. 2006, 269, 84–93. [Google Scholar] [CrossRef]
- Zhao, Z.; Zheng, J.; Wang, M.; Zhang, H.; Han, C.C. High performance ultrafiltration membrane based on modified chitosan coating and electrospun nanofibrous PVDF scaffolds. J. Membr. Sci. 2012, 394, 209–217. [Google Scholar] [CrossRef]
- Li, M.; Li, H.; Fang, F.; Deng, X.; Ma, S. Astragaloside IV attenuates cognitive impairments induced by transient cerebral ischemia and reperfusion in mice via anti-inflammatory mechanisms. Neurosci. Lett. 2017, 639, 114–119. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Yuan, H.; Wang, C.; Zhang, S.; Zhang, L.; Liu, X.; Liu, F.; Zhu, X.; Rohani, S.; Ching, C.; et al. A novel PVDF/PFSA-g-GO ultrafiltration membrane with enhanced permeation and antifouling performances. Sep. Purif. Technol. 2020, 233, 116038. [Google Scholar] [CrossRef]
- Beril Melbiah, J.S.; Kaleekkal, N.J.; Nithya Rabekkal, D.; Rana, D.; Nagendran, A.; Mohan, D. Improved permeation, separation and antifouling performance of customized polyacrylonitrile ultrafiltration membranes. Chem. Eng. Res. Des. 2020, 159, 157–169. [Google Scholar] [CrossRef]
- Liu, Q.; Li, L.; Pan, Z.; Dong, Q.; Xu, N.; Wang, T. Inorganic nanoparticles incorporated in polyacrylonitrile-based mixed matrix membranes for hydrophilic, ultrafast, and fouling-resistant ultrafiltration. J. Appl. Polym. Sci. 2019, 136, 47902. [Google Scholar] [CrossRef]
- Gul, S.; Rehan, Z.A.; Khan, S.A.; Akhtar, K.; Khan, M.A.; Khan, M.I.; Rashid, M.I.; Asiri, A.M.; Khan, S.B. Antibacterial PES-CA-Ag2O nanocomposite supported Cu nanoparticles membrane toward ultrafiltration, BSA rejection and reduction of nitrophenol. J. Mol. Liq. 2017, 230, 616–624. [Google Scholar] [CrossRef]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lu, H.; Zhang, L.; Shi, Y.; Ma, J.; Zhou, X. Fabrication of Composite Ultrafiltration Membrane by Coating Urea Formaldehyde Resin on Filter Paper. Coatings 2020, 10, 482. https://doi.org/10.3390/coatings10050482
Lu H, Zhang L, Shi Y, Ma J, Zhou X. Fabrication of Composite Ultrafiltration Membrane by Coating Urea Formaldehyde Resin on Filter Paper. Coatings. 2020; 10(5):482. https://doi.org/10.3390/coatings10050482
Chicago/Turabian StyleLu, Hailong, Lili Zhang, Yong Shi, Jinxia Ma, and Xiaofan Zhou. 2020. "Fabrication of Composite Ultrafiltration Membrane by Coating Urea Formaldehyde Resin on Filter Paper" Coatings 10, no. 5: 482. https://doi.org/10.3390/coatings10050482
APA StyleLu, H., Zhang, L., Shi, Y., Ma, J., & Zhou, X. (2020). Fabrication of Composite Ultrafiltration Membrane by Coating Urea Formaldehyde Resin on Filter Paper. Coatings, 10(5), 482. https://doi.org/10.3390/coatings10050482





