1. Introduction
Corrosion in distribution systems pipes resulted in not only pipe material destruction, but also deterioration in drinking water quality, i.e., water infection with other wastewater or any other water. Which leads to corrosion of valves or pumps in addition to blockage in pipes as a result of solid corrosion products.Unwanted chemical and biochemical reactions that occur in the distribution systems that release iron into distributed water can accumulate, creating tubers [
1]. Corrosion measurements (tubers) consist of reactive types that modify the physical and chemical parameters of water in the distribution system not only by releasing Fe oxyhydroxides, but also by interactions, for example, with by-products of chlorinated disinfection [
2], nitrates, or with natural organic substance [
3]. Salinity (chloride) is one of the most aggressive substances in seawater. Oxygen in seawater also affects metal pipes corrosion rate. Moreover, the amount of oxygen affected with the temperature, and consequently influences the rate of corrosion [
4].
Salts in the sea water cause corrosion inside the surfaces of pipes that transport saltwater in desalination water treatment plants. Also, the existence of air, salts on the ground, moisture, and other factors lead to outside pipe corrosion in the form of small holes or rough surface.
In any case, corrosion causes a short lifetime of the pipe, hydraulic effects, aesthetic effects, including increasing pumping costs, water leaks, and the buildup of corrosion products. Pipe replacement is not possible due to the high cost, so it is necessary to isolate pipe material from water and any corrosive agents [
5].
Corrosion inhibitors are largely utilized as a part of industry, as for instance, corrosive pickling of steel and iron, overflow cleaning and preparing, generation of metal and well oil fermentation [
6,
7,
8]. Improving the acidic environment needed the progress of altered corrosion control tests among which the implementation of chemical restraints has been the most economical test for the hindrance corrosion of acid [
9,
10,
11,
12,
13,
14]. Several organic composites, such as heterocyclic assembled, acetylenic alcohol, and quaternary ammonium salts are normally utilized as inhibitors in altered industries. The selected atoms adsorbed on the surface of metal among hetero atoms which include N, S, and O due to its protection for the active centers and to form a physical barrier to lowering the transmit of erosion sample to the metal surface [
15,
16,
17,
18,
19,
20,
21]. The heterocyclic affluences containing nitrogen atoms, like 4-aminoantipyrine (pyrazole derivative) are excellent corrosion hindrance with corrosive solution because rise hindrance of corrosion and prevent the odor irritating for alloys in altered aggressive environment [
22,
23,
24,
25,
26,
27]. Therefore, the development of novel adjuster inhibitors consisting of a pyrazol ring and the study of the relations among the inhibitors chemical structure and their inhibition led to the greater significance in theoretical points and industrial application.
In this study, the hindrance effect and electrochemical habit of spiropyrazole products for Q235 steel including 1.0 M HCl are given by the TP, EIS) and EFM tests. A few quantum-chemistry tests and molecular docking have been conducted in order to record the inhibition protection to the molecular properties of the altered kind of assembled [
28,
29].
3. Results and Discussion
3.1. TP Tests
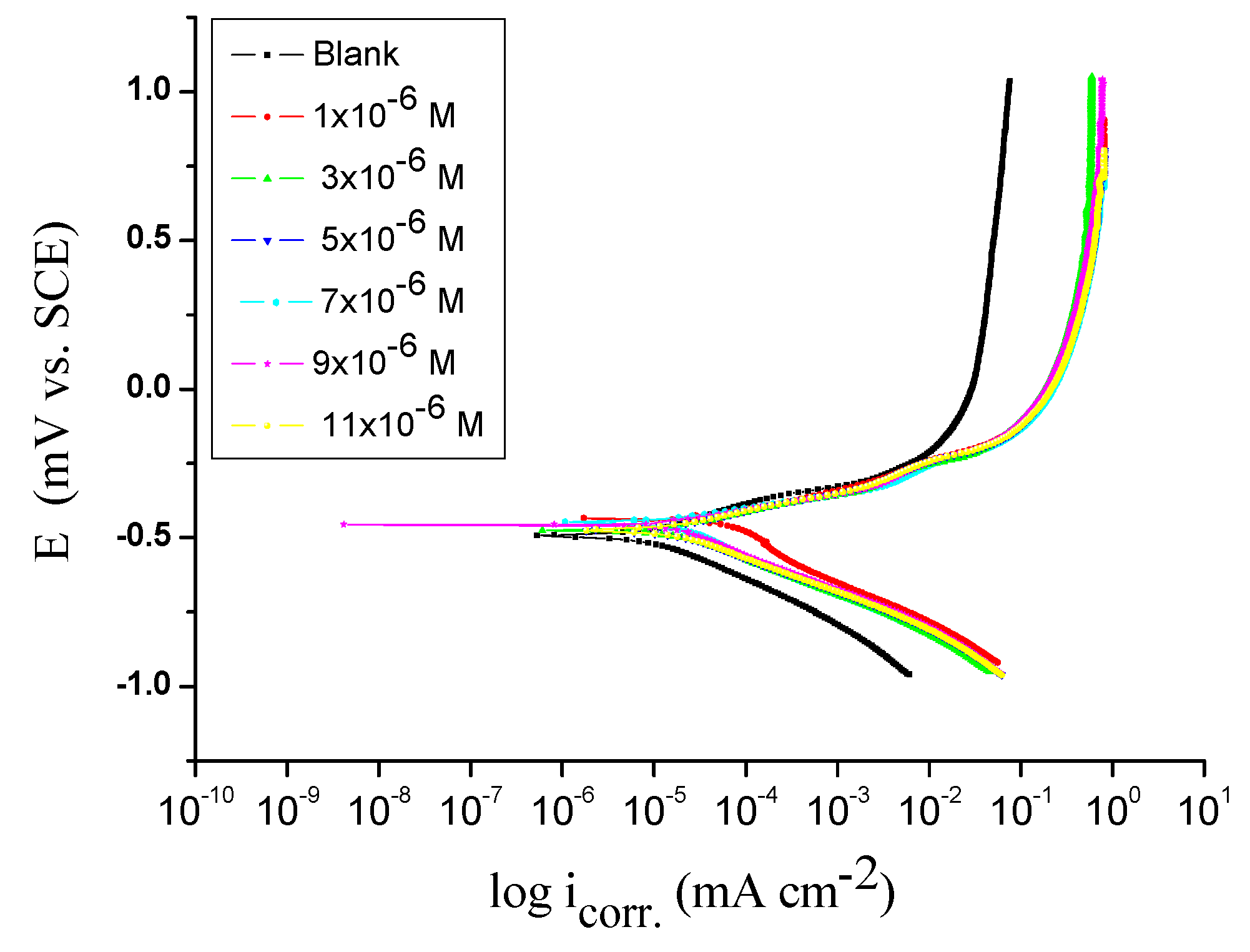
TP tests were conducted to obtain information regarding the kinetics of the anodic and cathodic reactions.
Figure 1 demonstrations the TP performance of Q235 steel electrode in corrosive solution nonexistence and attendance unlike dose of spiropyrazoles derivatives (1).
Figure 1 shows that the %IE
p rise as the spiropyrazoles dose rise, while the cathodic reaction is efficient protective, i.e., the adding of spiropyrazoles decrease the anodic liquefaction of alloyand also hindrance the cathodic reactions. Therefore, spiropyrazoles are acts as mixed kind inhibitors.
The (θ) and %IE were measured from relation (1):
where
and
are the current lack and attendance of solution inhibitor, consecutively.
It is evident from
Table 2 that the adsorbed inhibitors lessened the surface area for corrosion without effect on the mechanism of alloy corrosion in acidic solution [
34,
35]. The orders of IE% were: (1) > (2) > (3).
3.2. EIS Tests
One of the most effective tests in corrosion study is EIS. The properties of mechanical materials for surface and electrode motility can be obtained using impedance diagrams [
36,
37,
38,
39,
40].
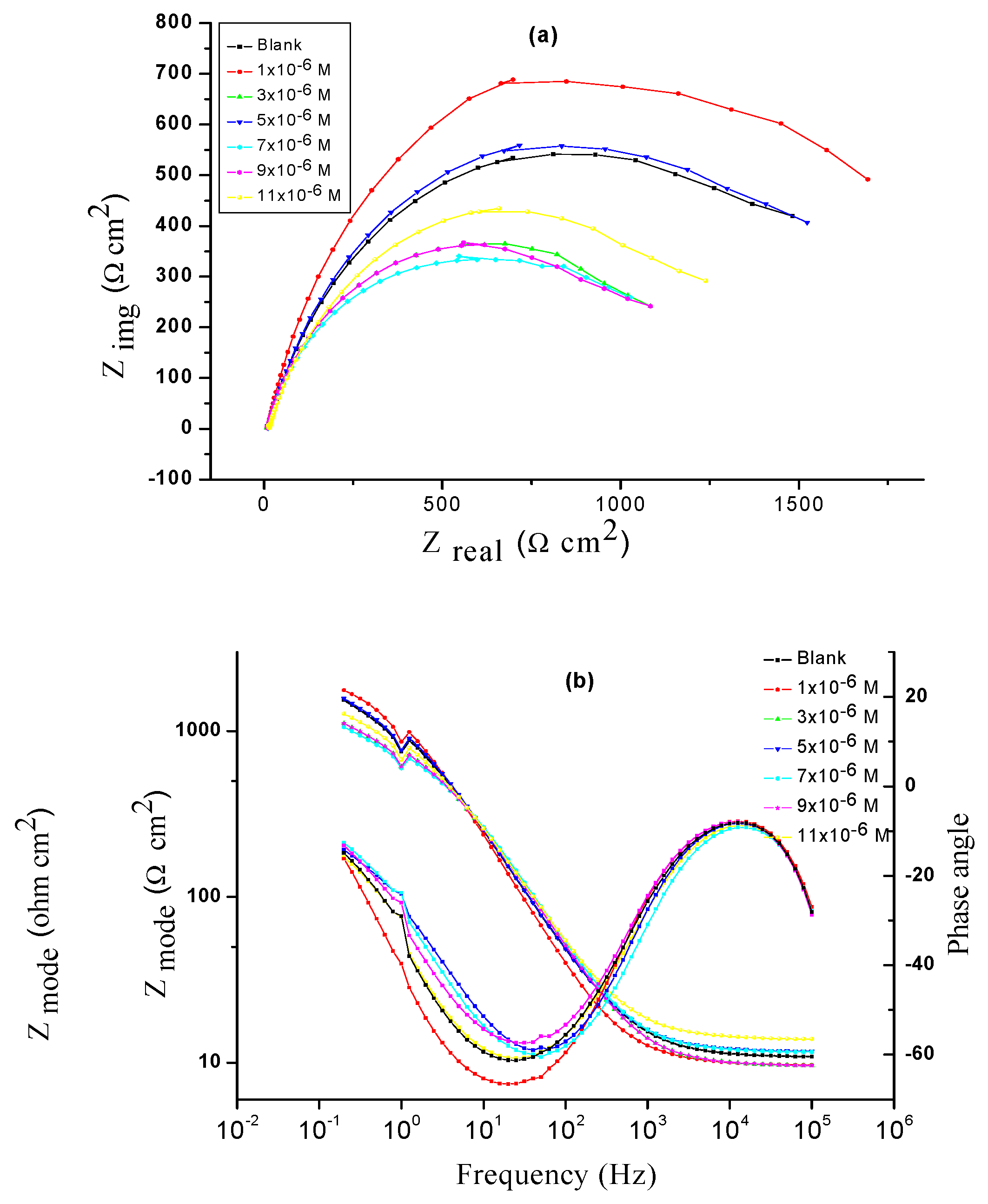
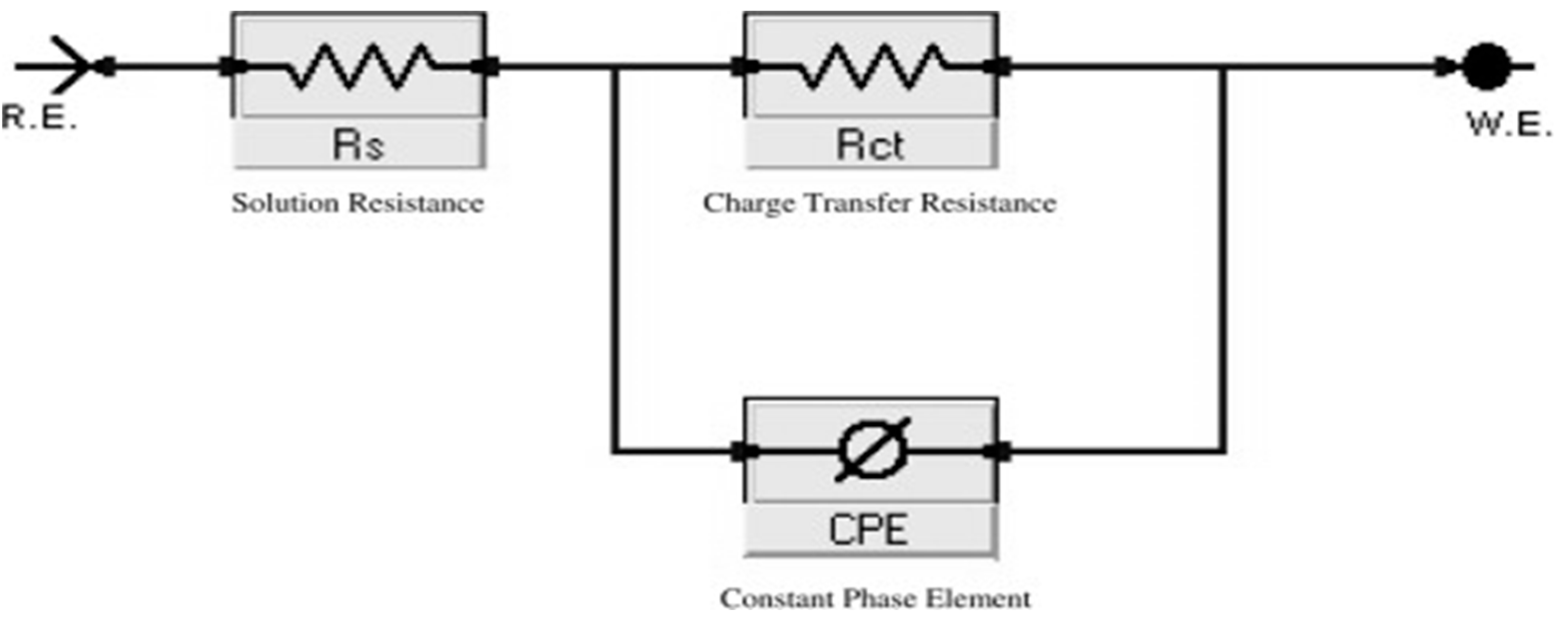
Figure 2 illustrated Nyquist (a) and Bode (b) bends given at OCP both in lack and attendance of improving dose of spiropyrazole derivatives. The values from EIS tests for a Q235 steel electrode were given utilizing the equivalent circuit demonstrated in
Figure 3.The improvement in the size of the capacitive loop with the attachment of spiropyrazole derivatives demonstrate that a barrier gradually forms on the surface of metal [
41,
42]. The higher in the size of capacitive loop
Figure 2 aimproves, at a fixed inhibitor dose, conformed the order: (1) > (2) > (3). The C
dl is measured from Equation (2):
where ω = 2π
fmax,
fmax = the maximum frequency.
After EIS exam the figure of the Nyquist bends, the corrosion procedure was measured principally charged-transfer [
43,
44,
45,
46]. From
Table 3 for the EIS data, we distinguished that the results of Rct improve with increasing the dose of spiropyrazoles and this result in improving in %IE. Data of Cdl are also minor to the maximum spiropyrazole inhibitor range [
47,
48]. The main merits of EIS are to monitor the corrosion performance of the metal with constant time. The %IEEIS was gotten from the EIS data from Equation (3) [
49]:
where
Roct and
Rct are the resistance values existence and lack of spiropyrazole, consecutively.
3.3. The Method of EFM
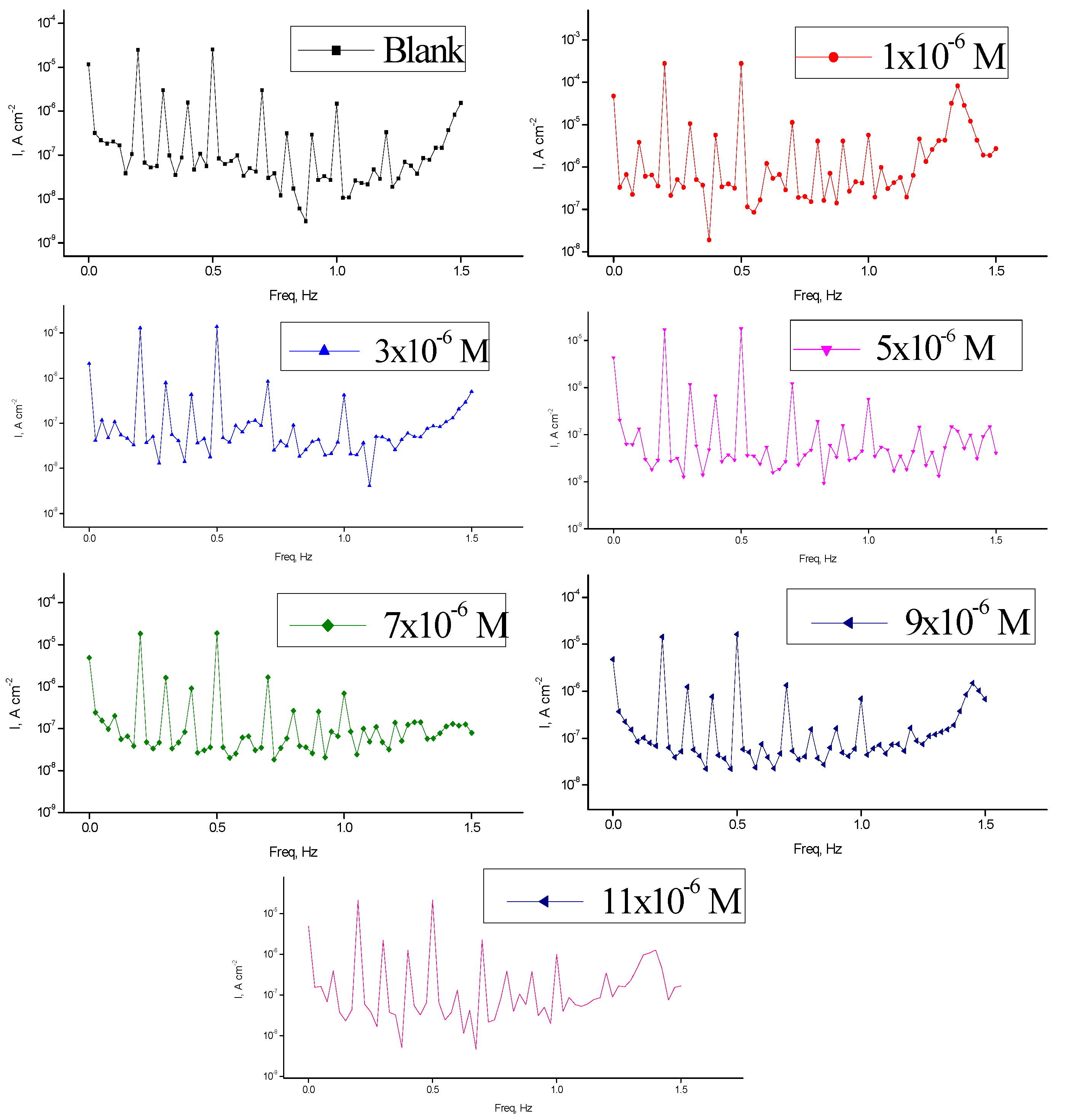
The advantages of EFM test gotten it a perfect for online monitoring of corrosion [
50]. The data of EFM in corrosive environments existance and lack of unlike dose of spiropyrazoles was obtain in
Figure 4. The results of EFM-tests were applied two unlike models: diffusion complete control of the cathodic reaction was quantified by and the “activation” model [
51]. The (
icorr), (CF-2 and CF-3), and (βc and βa) were quantified by the higher peaks. The preferable data of CF-2 and CF-3 in
Table 4 are parallel to their theoretical numbers of 2.0 and 3.0, individually result in excellent quality of the measured data.
The %IE
EFM raising by improvement the inhibitor dose and was calculated from Equation (4):
where
iocorr and
icorr are current attendance and lack of spiropyrazoles, consecutively.
The order of %IEEFM: (1) > (2) > (3).
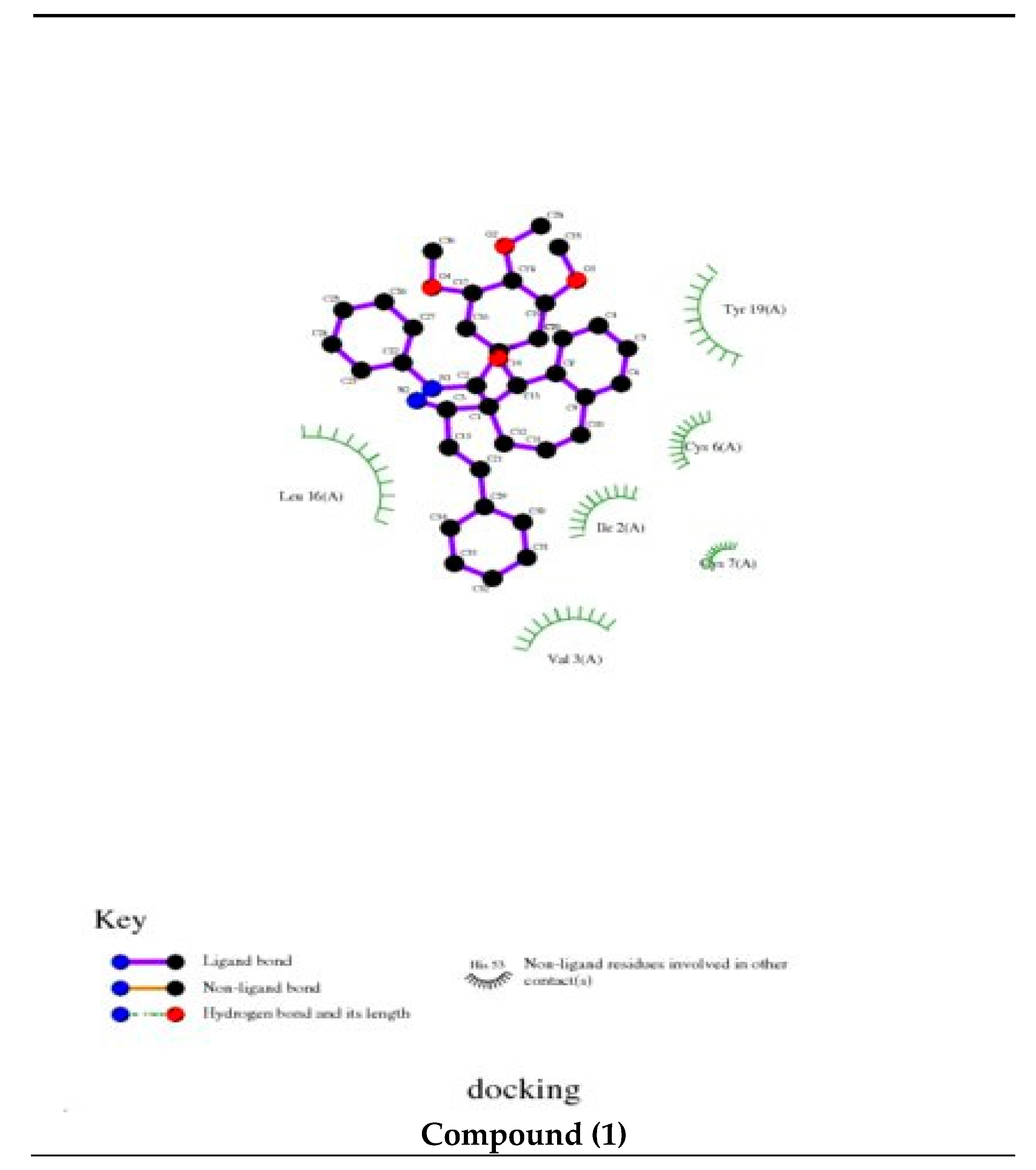
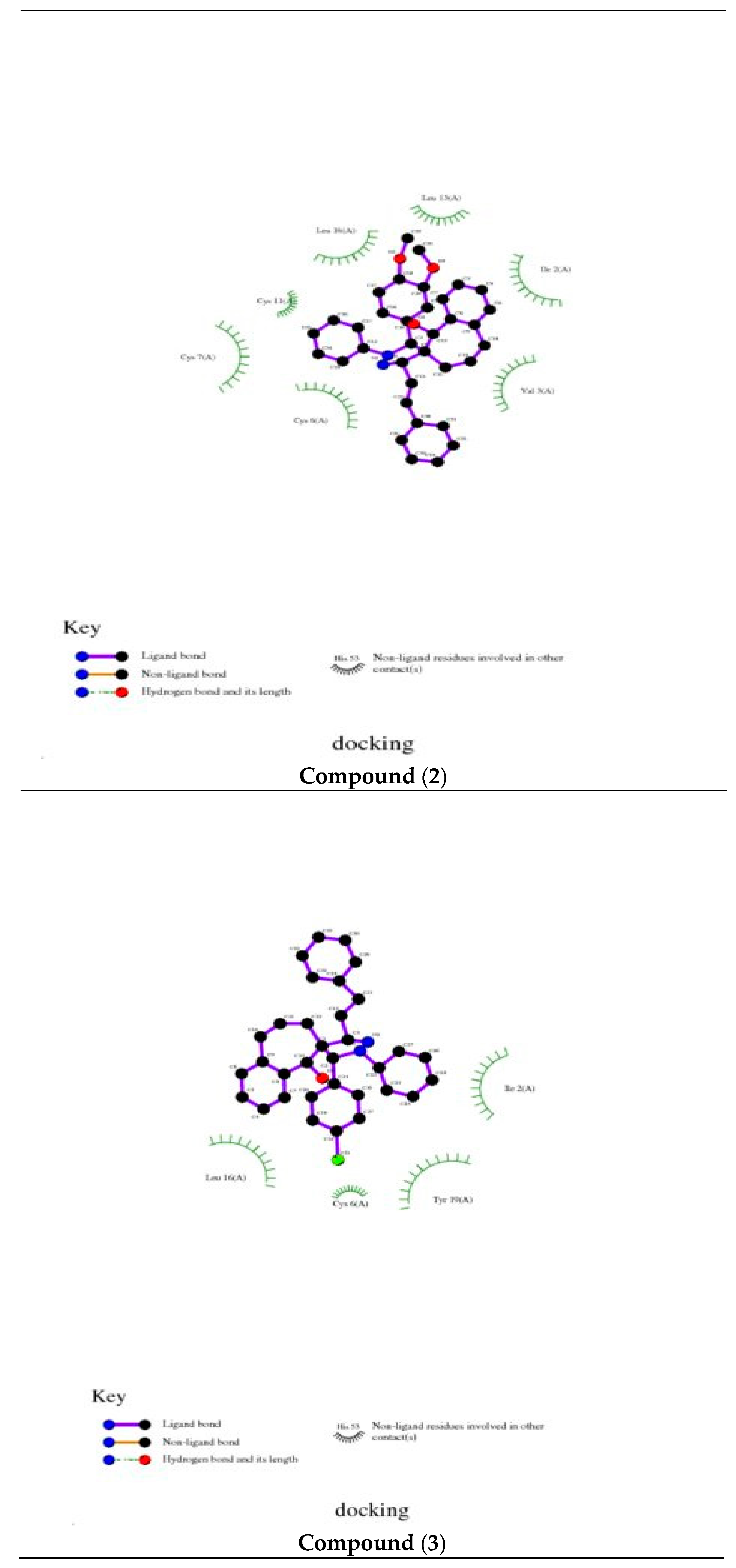
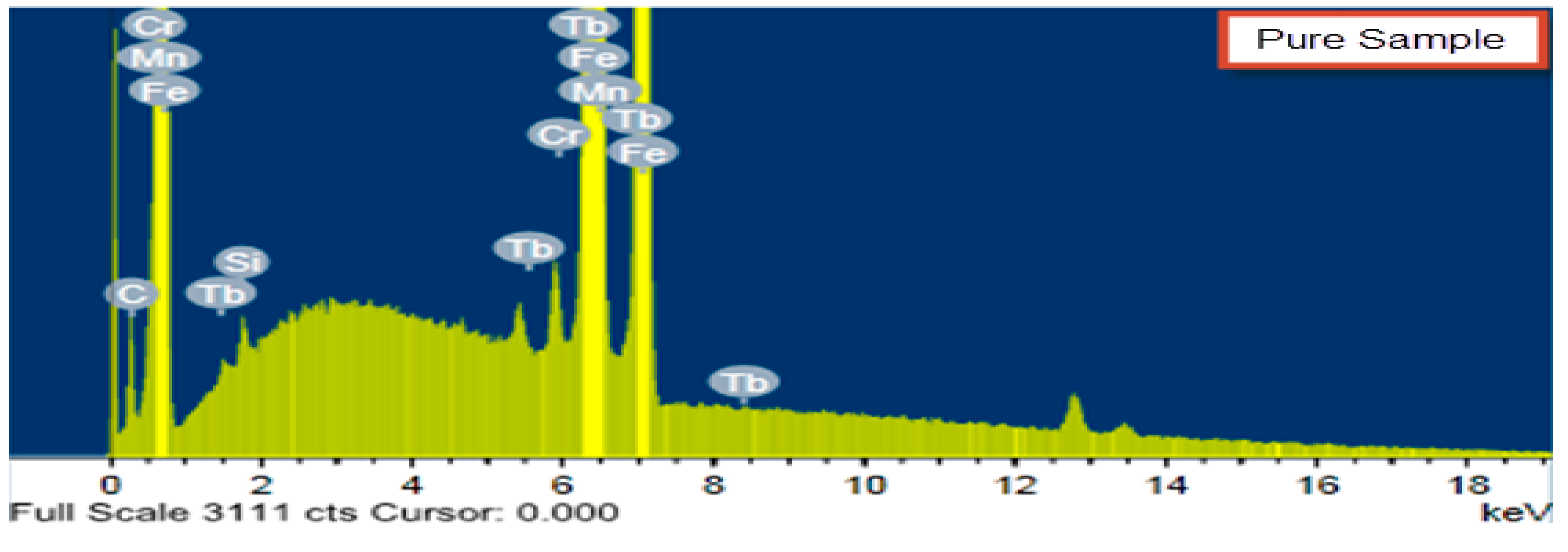
3.4. Molecular Docking
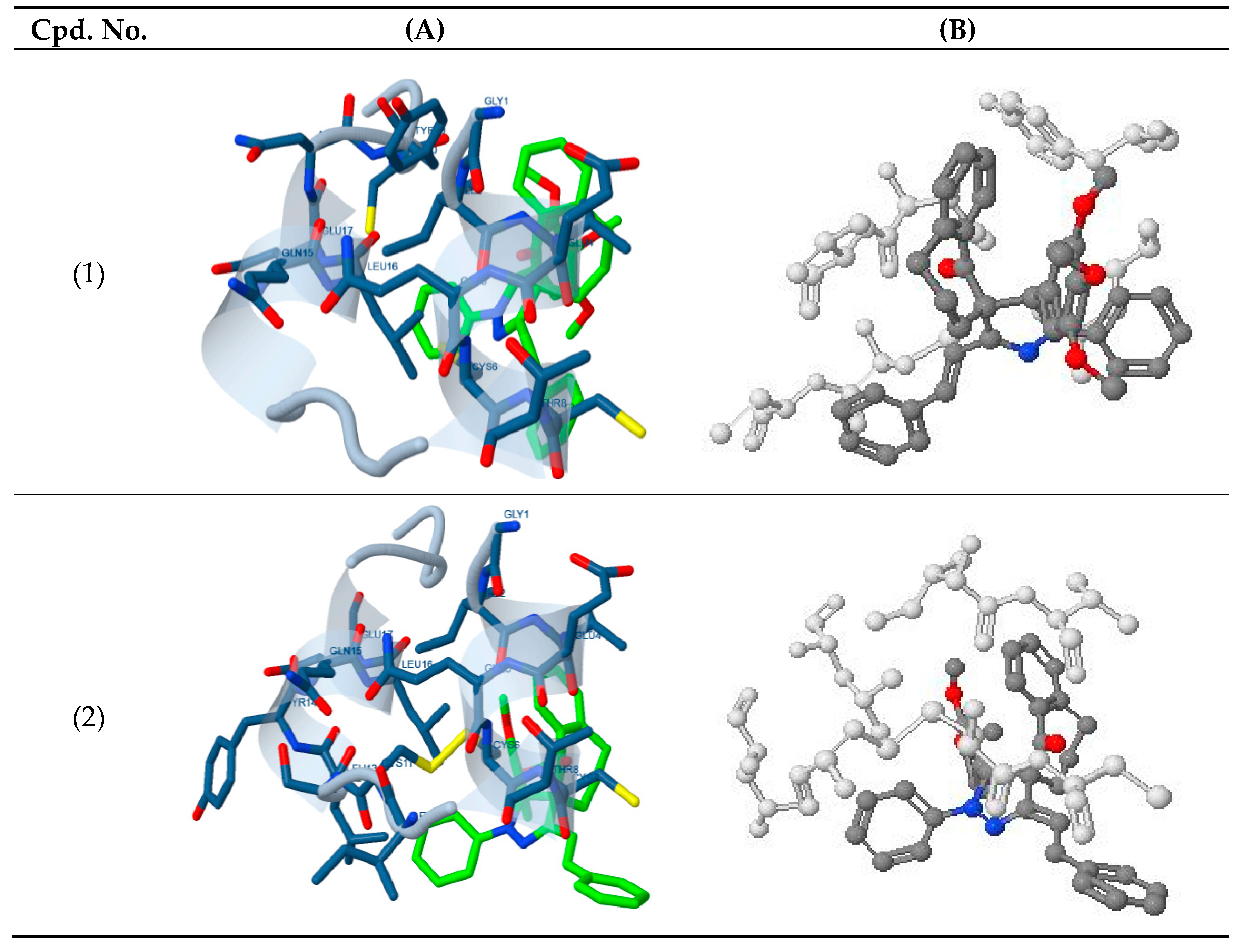
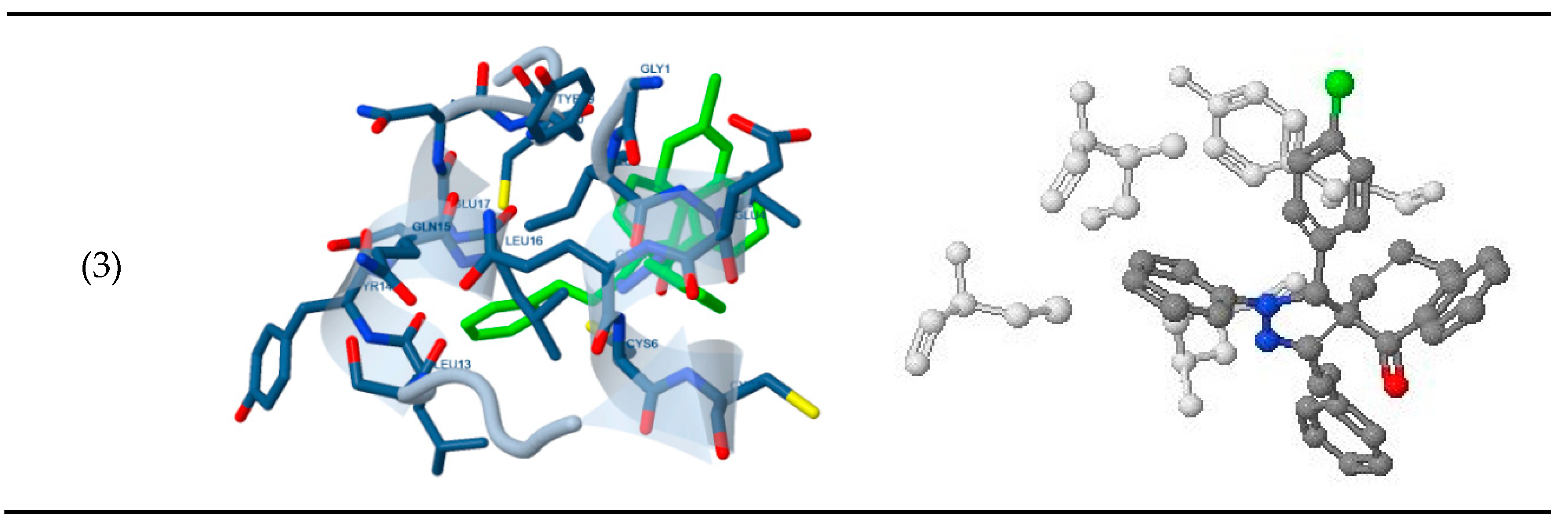
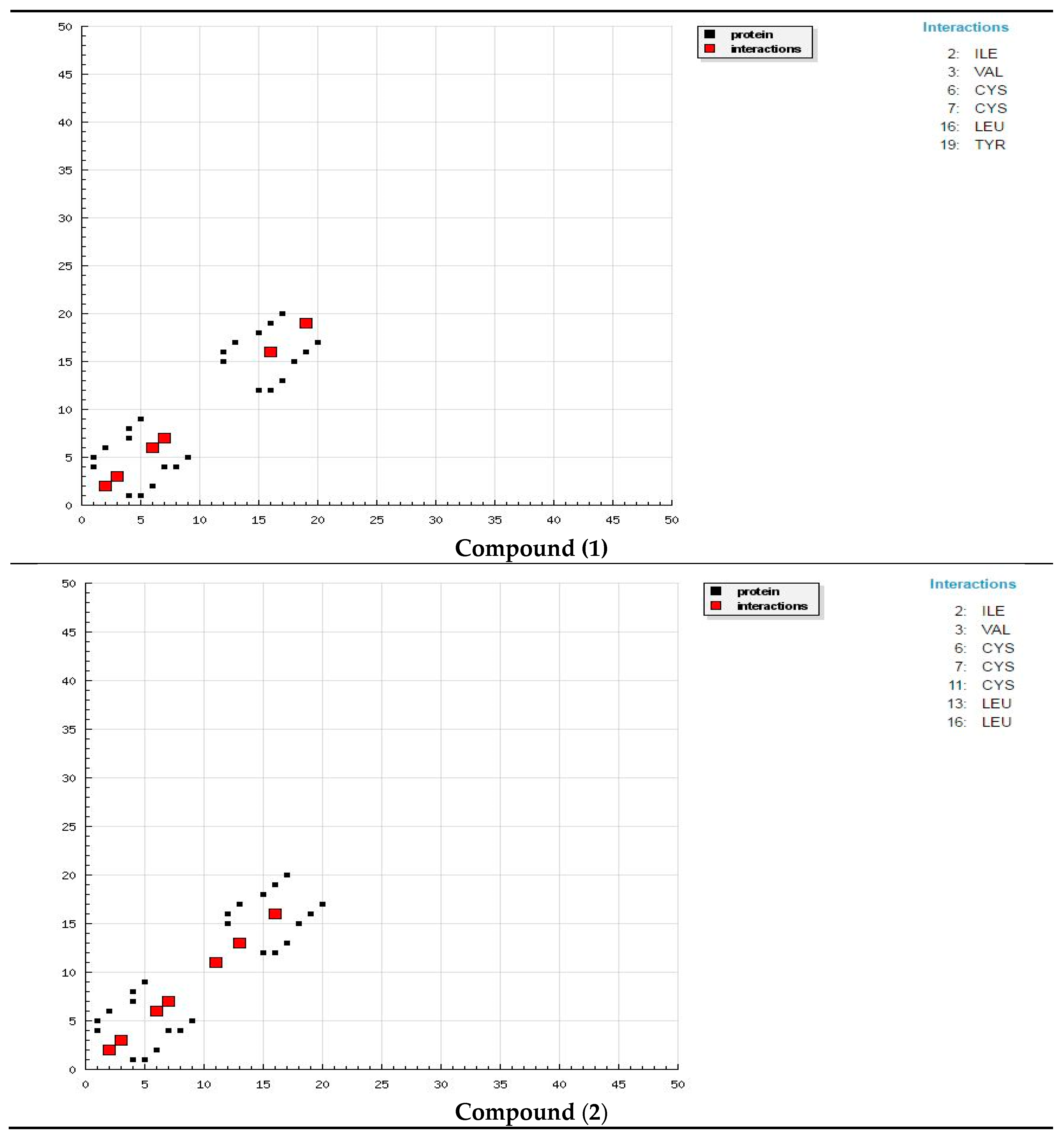
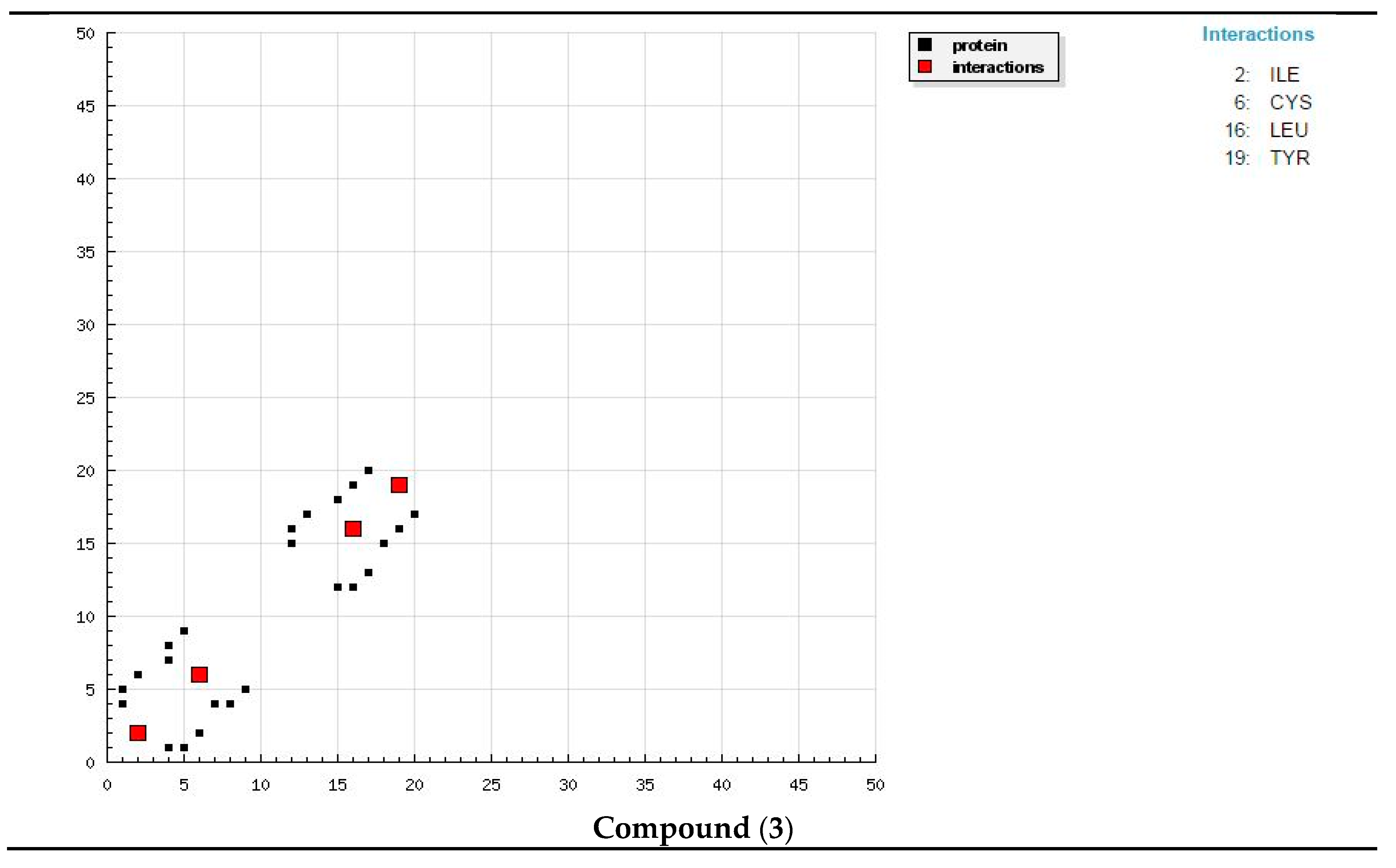
The docking study presented a favorable contact among spiropyrazoles derivatives and the receptor of 3tt8-hormone of crystal structure analysis of Cu human insulin derivative. The energy calculated is recorded in
Table 5 and
Figure 5. According to the outcome data in this study, HB diagrams specified that the spiropyrazoles derivatives bind to the proteins via hydrogen bond and disintegrated interactions energies in kcal/mol existed among the spiropyrazoles derivatives with 3tt8 receptor as exposed in
Figure 6. Also, based on this value, it can propose that interaction among the 3tt8 receptor and the spiropyrazoles is possible [
52]. Further, 2D plot bends of docking with spiropyrazole products are displayed in
Figure 7.
3.5. SEM Tests
The SEM test gotten from coins of Q235 steel existence and lack of 11 × 10
−6 M spiropyrazoles products after dipping for three days obtain in
Figure 8. The surfaces suffer from damaged corrosion attack in the blank. Due to the stress out when the composite appending in the solution, the morphology of the tests free surfaces was smoother. We observed a film creation which distributed in a random way on the whole surface of Q235 steel. This may be understood as being due to the spiropyrazole products adsorbed of the on Q235 steel which block the active center on alloy. This causes less contact among alloys and the aggressive enlivenments, and sequentially gives best protection effect [
53,
54].
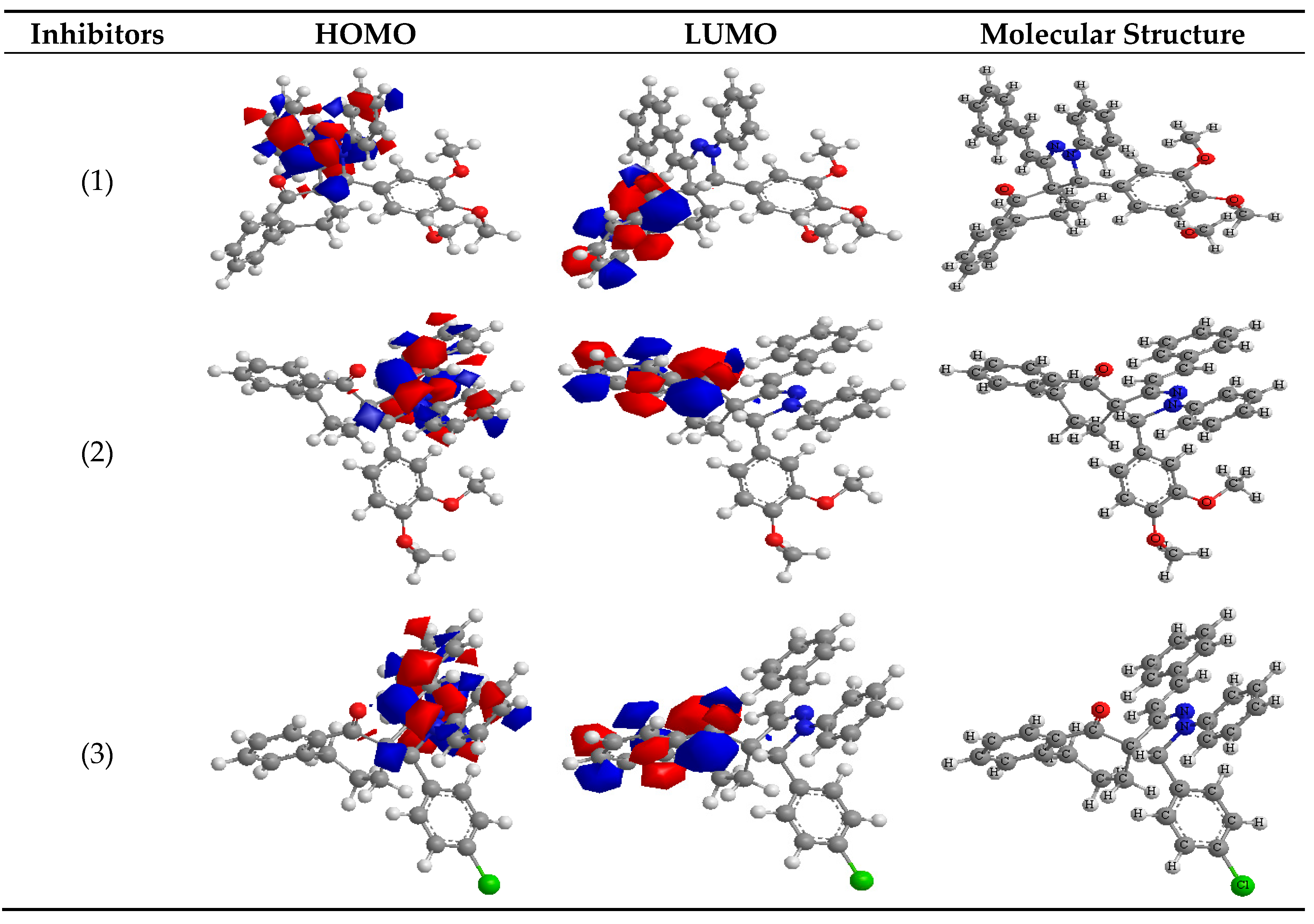
3.6. EDS Test
The EDS tests were applied to measure the elements obtain on the surface of Q235 steel and after 3 days of coated in the lack and attendance of corrosive solution.
Figure 9 gives the EDS data from the composition of Q235 steel only without the acid and presence spiropyrazoles. The EDS show that only oxygen and iron were detected, and the film passive was obtained with only Fe
2O
3.
The spectra give added lines, lead to the existence of C (C atoms of spiropyrazoles products). These data provide that the O and C atoms enclosed surface. The elemental detected is listed in
Table 6.
3.7. Quantum Chemical Calculations
The Mulliken charges and molecular orbital bends of spiropyrazole products given in
Figure 10. Theoretical tests were obtained for only the forms of neutral, in order to gotten further insight into the experimental results. Data of quantum chemical chief to ∆
E and
EHOMO and
ELUMO are measured and listed in
Table 7. The improved or lesser negative
EHOMO is inhibitor related, the higher the trend of offering electrons to unoccupied d orbital of Q235 steel, and the progress of the corrosion hindrance. The lesser
ELUMO, the greater the acceptance of plain electrons from surface of Q235 steel [
55,
56]. ∆
E assumed by the tests in case of spiropyrazole (1) is less than (3) (
Table 7) given spiropyrazole (1) molecule will absorb more highest on alloy surface than others, due to electron easy transfer between HOMO and LUMO occurred among its adsorption on the surface of Q235 steel and the maximum of hindrance productivity [
57]. It can be seen that all tests of quantum checking these results from experimental.
3.8. Mechanism of Protection
From the results of electrochemical tests, the IE% relies on metal nature, dose, surface conditions, and the kind of spiropyrazole derivatives adsorption on Q235-steel.
The outcome data of corrosion data attendance of these inhibitors:
With an increase in the dose of the inhibitor, the corrosion rate becomes lower
The exchange in Tafel lines to extreme regions of potential.
The %IE relies on excharge density and their equipment of adsorption centers in the molecule.
Metals such as iron, which are highly attractive to aromatic rings, were gotten to adsorb benzene rings in a flat direction. The order of breakdown of the %IE of the spiropyrazoles in the corrosion solution was in the following order: (1) > (2) > (3).
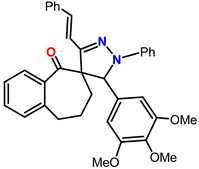
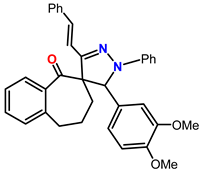
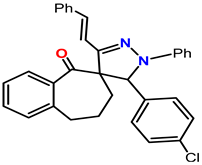
Spiropyrazoles (1) demonstrations best hindrance power because: (i) it has greater molecular size (558.25) that may enable best surface coated and bigger molecular area and (ii) its adsorption among 6 active sites (2-N and 4-O atoms). Spiropyrazoles (2) comes after (1) in %IE because it has fewer molecular size (528.24) and minus active site (1-O and 2-N atoms). Spiropyrazoles (3) is the smallest one in %IE, this is due to it having a minor molecular size (502.18), the appending of p-Cl group is electron withdrawing group with (σ Cl = +0.23), and its order of protection relies on the magnitude of their withdrawing character.
Concentration of the inhibitor is an important factor in adsorption. As illustrated in
Figure 11, at the adsorption density less than monolayer (
Figure 11a), most of the nucleus sites are still likely to be exposed to hydrochloric acid, as the inhibitor absorbs them less. When the adsorption intensity reaches monolayer adsorption (
Figure 11b), some nucleus sites begin to cover with the barrier particles. At the maximum absorption density (
Figure 11c), the inhibitor particles cover the entire surface, including the sites of the nucleus, and then complete inhibition occurs.
3.9. Conclusions
All the spiropyrazole products are potentially brilliant corrosion inhibitors for Q235 steel. The structures of these spiropyrazole inhibtors as well as the presence of certain substituents play a vital role on their effectiveness anticorrosive agents.
The results of EIS display enhancement in the charge transfer resistance and a decline in double layer capacitances. When adding an inhibitor and thus an increase in% IE due to an increase in the electrical double layer the thickness.
The outcome values from electrochemical tests were in good agreement. The % IE of these spiropyrazoles is: (1)>(2)>(3).
Molecular docking and binding energy calculations of spiropyrazolederivatives (1)–(3) with the receptor of 3tt8-hormone of crystal structure analysis of Cu human insulin derivative indicated that the spiropyrazoles are %IE of receptor of 3tt8-hormone.
The morphology of protected and no protected Q235 steel was tested by SEM and EDX.
Quantum calculation results demonstrated that the heteroatoms of N and O are the active sites ofthe spiropyrazole derivatives.