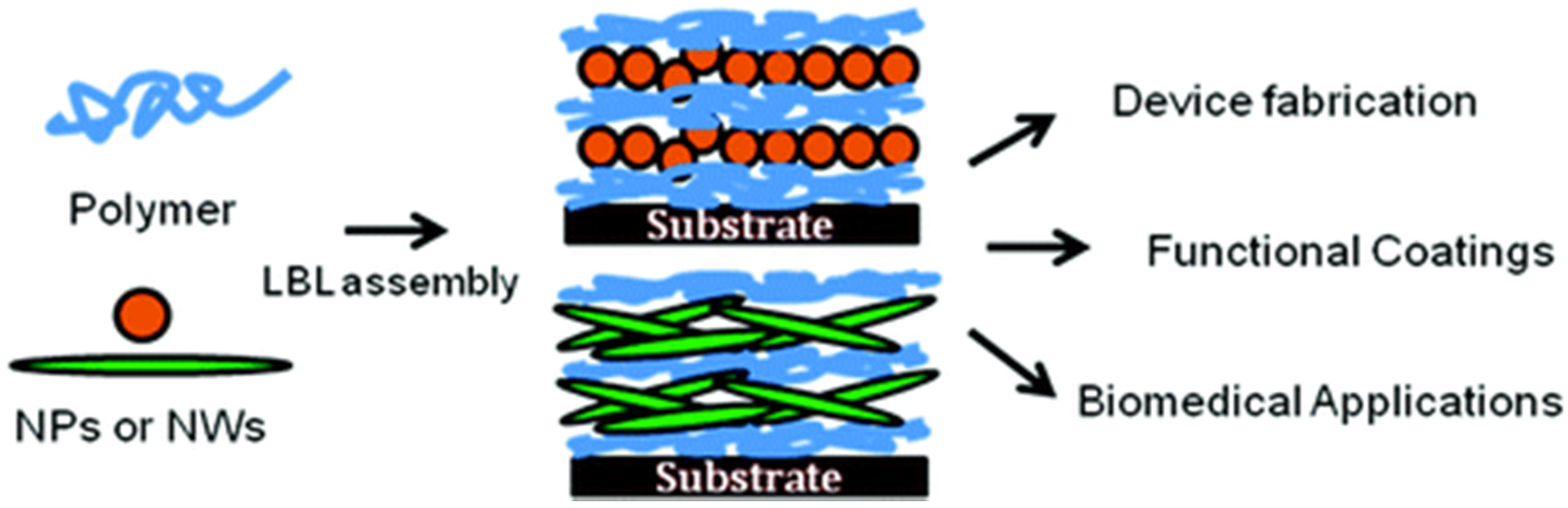
Nanoparticles in Polyelectrolyte Multilayer Layer-by-Layer (LbL) Films and Capsules—Key Enabling Components of Hybrid Coatings
Abstract
1. Introduction
2. Functionalization of Polyelectrolyte Multilayers—Organic versus Inorganic Building Blocks
2.1. Incorporation of Dyes in Layer-by-Layer (LbL) Coatings—Bringing Multifunctionality through Organic Moieties
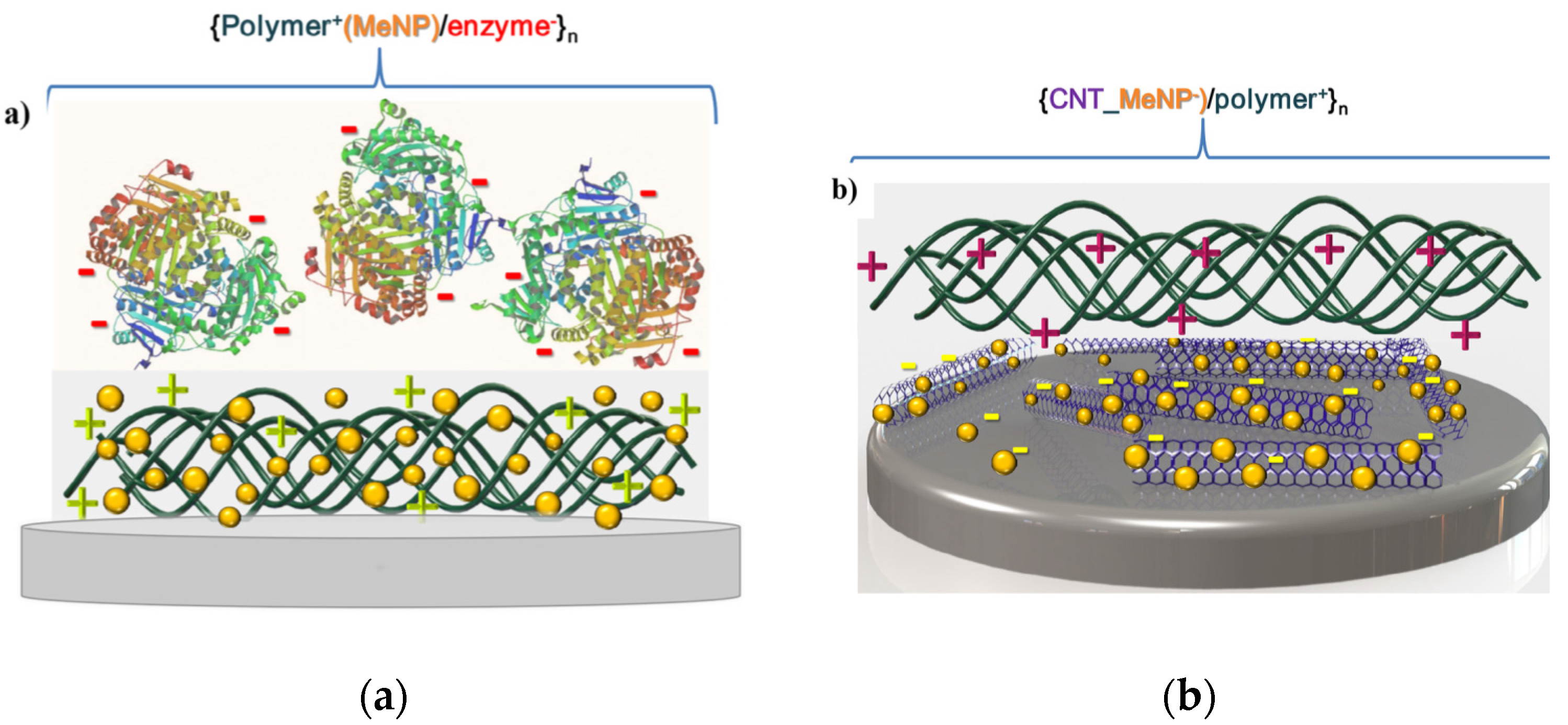
2.2. Incorporation of Nanoparticles in LbL Coatings—Hardness Enhancement and Additional Properties through Inorganic Building Blocks
3. Planar LbL Coatings and Their Functionalization by Nanoparticles
3.1. Enhancement of Mechanical Properties and Remote Acteivation of LbL Coatings
3.2. Passive and Active Activation of LbL Coatings
3.3. Assembling Janus Particles and Capsules Using LbL Coatings
3.4. Corrosion Protection of the Coatings
3.5. Development of Sensors and Biosensors Based on Layer-by-Layer Assembled Coatings
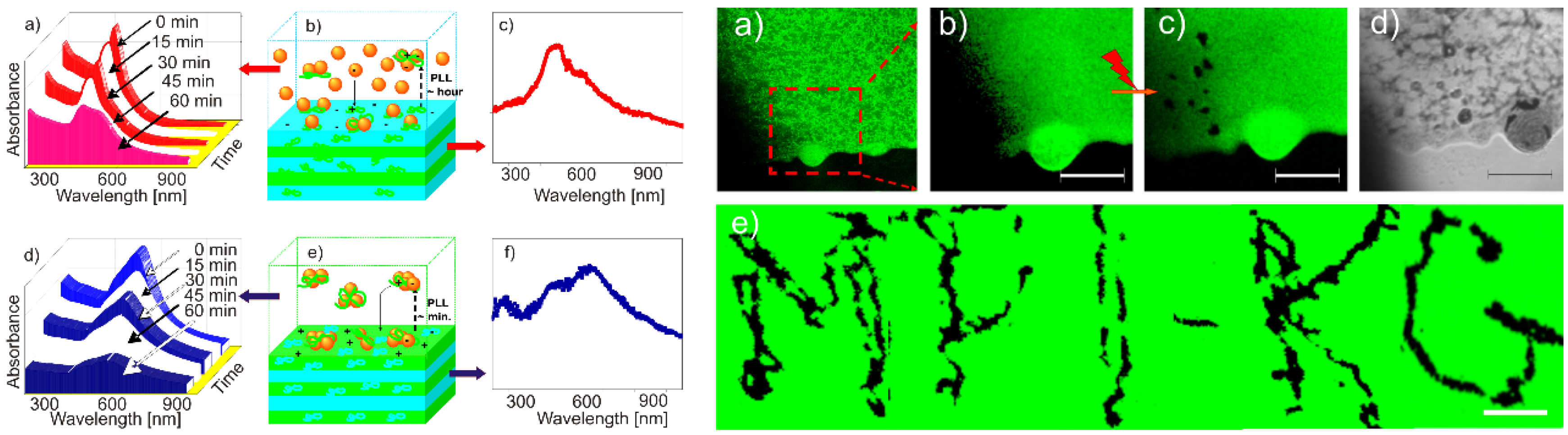
3.6. Other Nanostructured Inorganic Building Blocks in LbL Planar Coatings
4. Spherical LbL Capsules and Their Functionalization by Nanoparticles
4.1. Nanoparticle Incorporation into the Shell of LbL Capsules
4.2. Supramolecular Functionalization and Click Chemistry
4.3. Release from Microcapsules by Laser Light, Magnetic Fields and Ultrasound
4.4. Strengthening of Mechanical Properties of LbL Microcapsules by Nanoparticles
4.5. Other Nanostructured Inorganic Building Blocks in LbL Microcapsules
5. Conclusions and Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Decher, G. Fuzzy Nanoassemblies: Toward Layered Polymeric Multicomposites. Science 1997, 277, 1232–1237. [Google Scholar] [CrossRef]
- Lvov, Y.; Decher, G.; Möhwald, H. Assembly, structural characterization, and thermal behavior of layer-by-layer deposited ultrathin films of poly(vinyl sulfate) and poly(allylamine). Langmuir 1993, 9, 481–486. [Google Scholar] [CrossRef]
- Nuraje, N.; Asmatulu, R.; Cohen, R.E.; Rubner, M.F. Durable Antifog Films from Layer-by-Layer Molecularly Blended Hydrophilic Polysaccharides. Langmuir 2011, 27, 782–791. [Google Scholar] [CrossRef] [PubMed]
- Richardson, J.J.; Bjornmalm, M.; Caruso, F. Technology-driven layer-by-layer assembly of nanofilms. Science 2015, 348, aaa2491. [Google Scholar] [CrossRef] [PubMed]
- Kozlovskaya, V.; Shamaev, A.; Sukhishvili, S.A. Tuning swelling pH and permeability of hydrogel multilayer capsules. Soft Matter 2008, 4, 1499–1507. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Senger, B.; Vautier, D.; Picart, C.; Schaaf, P.; Voegel, J.-C.; Lavalle, P. Natural polyelectrolyte films based on layer-by layer deposition of collagen and hyaluronic acid. Biomaterials 2005, 26, 3353–3361. [Google Scholar] [CrossRef]
- Guin, T.; Cho, J.H.; Xiang, F.; Ellison, C.J.; Grunlan, J.C. Water-Based Melanin Multilayer Thin Films with Broadband UV Absorption. ACS Macro Lett. 2015, 4, 335–338. [Google Scholar] [CrossRef]
- Gribova, V.; Auzely-Velty, R.; Picart, C. Polyelectrolyte Multilayer Assemblies on Materials Surfaces: From Cell Adhesion to Tissue Engineering. Chem. Mater. 2012, 24, 854–869. [Google Scholar] [CrossRef]
- Kotov, N.A.; Dekany, I.; Fendler, J.H. Layer-by-Layer Self-Assembly of Polyelectrolyte-Semiconductor Nanoparticle Composite Films. J. Phys. Chem. 1995, 99, 13065–13069. [Google Scholar] [CrossRef]
- Lee, D.; Rubner, M.F.; Cohen, R.E. All-Nanoparticle Thin-Film Coatings. Nano Lett. 2006, 6, 2305–2312. [Google Scholar] [CrossRef]
- Lvov, Y.; Ariga, K.; Onda, M.; Ichinose, I.; Kunitake, T. Alternate Assembly of Ordered Multilayers of SiO2 and Other Nanoparticles and Polyions. Langmuir 1997, 13, 6195–6203. [Google Scholar] [CrossRef]
- Delcea, M.; Möhwald, H.; Skirtach, A.G. Stimuli-responsive LbL capsules and nanoshells for drug delivery. Adv. Drug Deliv. Rev. 2011, 63, 730–747. [Google Scholar] [CrossRef] [PubMed]
- Déjugnat, C.; Haložan, D.; Sukhorukov, G.B. Defined Picogram Dose Inclusion and Release of Macromolecules using Polyelectrolyte Microcapsules. Macromol. Rapid Commun. 2005, 26, 961–967. [Google Scholar] [CrossRef]
- Mauser, T.; Déjugnat, C.; Sukhorukov, G.B. Balance of Hydrophobic and Electrostatic Forces in the pH Response of Weak Polyelectrolyte Capsules. J. Phys. Chem. B 2006, 110, 20246–20253. [Google Scholar] [CrossRef]
- Saveleva, M.S.; Eftekhari, K.; Abalymov, A.A.; Douglas, T.E.L.; Volodkin, D.V.; Parakhonskiy, B.V.; Skirtach, A.G. Hierarchy of Hybrid Materials—The Place of Inorganics-in-Organics in it, Their Composition and Applications. Front. Chem. 2019, 7, 179. [Google Scholar] [CrossRef]
- Kaniewska, K.; Karbarz, M.; Katz, E. Nanocomposite hydrogel films and coatings—Features and applications. Appl. Mater. Today 2020, 20, 100776. [Google Scholar] [CrossRef]
- Rial, R.; Liu, Z.; Ruso, J.M. Soft Actuated Hybrid Hydrogel with Bioinspired Complexity to Control Mechanical Flexure Behavior for Tissue Engineering. Nanomaterials 2020, 10, 1302. [Google Scholar] [CrossRef]
- Park, M.-K.; Advincula, R.C. The Layer-by-Layer Assemblies of Polyelectrolytes and Nanomaterials as Films and Particle Coatings. In Functional Polymer Films; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2011; pp. 73–112. [Google Scholar]
- Ariga, K.; Hill, J.P.; Ji, Q. Organic-Inorganic Supramolecular Materials. In Supramolecular Soft Matter; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2011; pp. 43–55. [Google Scholar]
- Katagiri, K. Organic-inorganic nanohybrid particles for biomedical applications. In Bioceramics; Elsevier: Amsterdam, The Netherlands, 2011; pp. 113–135. [Google Scholar]
- Yan, X.; Zhu, P.; Li, J. Self-assembly and application of diphenylalanine-based nanostructures. Chem. Soc. Rev. 2010, 39, 1877–1890. [Google Scholar] [CrossRef]
- Liu, X.; Zhou, L.; Geng, W.; Sun, J. Layer-by-Layer-Assembled Multilayer Films of Polyelectrolyte-Stabilized Surfactant Micelles for the Incorporation of Noncharged Organic Dyes. Langmuir 2008, 24, 12986–12989. [Google Scholar] [CrossRef]
- Szabó, T.; Péter, Z.; Illés, E.; Janovák, L.; Talyzin, A. Stability and dye inclusion of graphene oxide/polyelectrolyte layer-by-layer self-assembled films in saline, acidic and basic aqueous solutions. Carbon N. Y. 2017, 111, 350–357. [Google Scholar] [CrossRef]
- Olszyna, M.; Debrassi, A.; Üzüm, C.; Dähne, L. Label-Free Bioanalysis Based on Low-Q Whispering Gallery Modes: Rapid Preparation of Microsensors by Means of Layer-by-Layer Technology. Adv. Funct. Mater. 2019, 29, 1805998. [Google Scholar] [CrossRef]
- Ma, W.; Zhang, Y.; Li, F.; Kou, D.; Lutkenhaus, J. Layer-by-Layer Assembly and Electrochemical Study of Alizarin Red S-Based Thin Films. Polymers 2019, 11, 165. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, U.; Singha, T.; Chianelli, R.R.; Hansda, C.; Kumar Paul, P. Organic-inorganic hybrid layer-by-layer electrostatic self-assembled film of cationic dye Methylene Blue and a clay mineral: Spectroscopic and Atomic Force microscopic investigations. J. Lumin. 2017, 187, 322–332. [Google Scholar] [CrossRef]
- Hu, X.; Qureishi, Z.; Thomas, S.W. Light-Controlled Selective Disruption, Multilevel Patterning, and Sequential Release with Polyelectrolyte Multilayer Films Incorporating Four Photocleavable Chromophores. Chem. Mater. 2017, 29, 2951–2960. [Google Scholar] [CrossRef]
- Skirtach, A.G.A.G.; Antipov, A.A.; Shchukin, D.G.; Sukhorukov, G.B. Remote Activation of Capsules Containing Ag Nanoparticles and IR Dye by Laser Light. Langmuir 2004, 20, 6988–6992. [Google Scholar] [CrossRef]
- Erokhina, S.; Benassi, L.; Bianchini, P.; Diaspro, A.; Erokhin, V.; Fontana, M.P. Light-Driven Release from Polymeric Microcapsules Functionalized with Bacteriorhodopsin. J. Am. Chem. Soc. 2009, 131, 9800–9804. [Google Scholar] [CrossRef]
- Zeng, Y.; Wang, X.-L.; Yang, Y.-J.; Chen, J.-F.; Fu, J.; Tao, X. Assembling photosensitive capsules by phthalocyanines and polyelectrolytes for photodynamic therapy. Polymer (Guildf) 2011, 52, 1766–1771. [Google Scholar] [CrossRef]
- Bédard, M.F.; Sadasivan, S.; Sukhorukov, G.B.; Skirtach, A. Assembling polyelectrolytes and porphyrins into hollow capsules with laser-responsive oxidative properties. J. Mater. Chem. 2009, 19, 2226–2233. [Google Scholar] [CrossRef]
- Li, C.; Li, Z.-Y.; Zhang, J.; Wang, K.; Gong, Y.-H.; Luo, G.-F.; Zhuo, R.-X.; Zhang, X.-Z. Porphyrin containing light-responsive capsules for controlled drug release. J. Mater. Chem. 2012, 22, 4623–4626. [Google Scholar] [CrossRef]
- Marchenko, I.V.; Borodina, T.N.; Trushina, D.B.; Nabatov, B.V.; Logachev, V.V.; Plotnikov, G.S.; Baranov, A.N.; Saletskii, A.M.; Ryabova, A.V.; Bukreeva, T.V. Incorporation of Naphthalocyanine into Shells of Polyelectrolyte Capsules and Their Disruption under Laser Radiation. Colloid J. 2018, 80, 399–406. [Google Scholar] [CrossRef]
- Srivastava, S.; Kotov, N.A. Composite Layer-by-Layer (LBL) assembly with inorganic nanoparticles and nanowires. Acc. Chem. Res. 2008, 41, 1831–1841. [Google Scholar] [CrossRef] [PubMed]
- Gittleson, F.S.; Hwang, D.; Ryu, W.-H.; Hashmi, S.M.; Hwang, J.; Goh, T.; Taylor, A.D. Ultrathin Nanotube/Nanowire Electrodes by Spin–Spray Layer-by-Layer Assembly: A Concept for Transparent Energy Storage. ACS Nano 2015, 9, 10005–10017. [Google Scholar] [CrossRef] [PubMed]
- Danglad-Flores, J.; Eftekhari, K.; Skirtach, A.G.; Riegler, H. Controlled Deposition of Nanosize and Microsize Particles by Spin-Casting. Langmuir 2019, 35, 3404–3412. [Google Scholar] [CrossRef] [PubMed]
- Hu, H.; Pauly, M.; Felix, O.; Decher, G. Spray-assisted alignment of Layer-by-Layer assembled silver nanowires: A general approach for the preparation of highly anisotropic nano-composite films. Nanoscale 2017, 9, 1307–1314. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Cheng, M.; Zhu, X.; Zhang, Y.; An, Q.; Shi, F. A facile method for the construction of stable polymer–inorganic nanoparticle composite multilayers. J. Mater. Chem. A 2013, 1, 11329–11334. [Google Scholar] [CrossRef]
- Abd El-Hady, M.M.; Sharaf, S.; Farouk, A. Highly hydrophobic and UV protective properties of cotton fabric using layer by layer self-assembly technique. Cellulose 2020, 27, 1099–1110. [Google Scholar] [CrossRef]
- Abuid, N.J.; Gattás-Asfura, K.M.; Schofield, E.A.; Stabler, C.L. Layer-by-Layer Cerium Oxide Nanoparticle Coating for Antioxidant Protection of Encapsulated Beta Cells. Adv. Healthc. Mater. 2019, 8, 1801493. [Google Scholar] [CrossRef]
- Ghostine, R.A.; Jisr, R.M.; Lehaf, A.; Schlenoff, J.B. Roughness and Salt Annealing in a Polyelectrolyte Multilayer. Langmuir 2013, 29, 11742–11750. [Google Scholar] [CrossRef]
- Buron, C.C.; Filiâtre, C.; Membrey, F.; Bainier, C.; Buisson, L.; Charraut, D.; Foissy, A. Surface morphology and thickness of a multilayer film composed of strong and weak polyelectrolytes: Effect of the number of adsorbed layers, concentration and type of salts. Thin Solid Films 2009, 517, 2611–2617. [Google Scholar] [CrossRef]
- Lavalle, P.; Gergely, C.; Cuisinier, F.J.G.; Decher, G.; Schaaf, P.; Voegel, J.C.; Picart, C. Comparison of the Structure of Polyelectrolyte Multilayer Films Exhibiting a Linear and an Exponential Growth Regime: An in Situ Atomic Force Microscopy Study. Macromolecules 2002, 35, 4458–4465. [Google Scholar] [CrossRef]
- Campbell, J.; Vikulina, A.S. Layer-By-Layer Assemblies of Biopolymers: Build-Up, Mechanical Stability and Molecular Dynamics. Polymers 2020, 12, 1949. [Google Scholar] [CrossRef] [PubMed]
- Mzyk, A.; Lackner, J.M.; Wilczek, P.; Lipińska, L.; Niemiec-Cyganek, A.; Samotus, A.; Morenc, M. Polyelectrolyte multilayer film modification for chemo-mechano-regulation of endothelial cell response. RSC Adv. 2016, 6, 8811–8828. [Google Scholar] [CrossRef]
- Schneider, A.; Francius, G.; Obeid, R.; Schwinté, P.; Hemmerlé, J.; Frisch, B.; Schaaf, P.; Voegel, J.-C.; Senger, B.; Picart, C. Polyelectrolyte Multilayers with a Tunable Young’s Modulus: Influence of Film Stiffness on Cell Adhesion. Langmuir 2006, 22, 1193–1200. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, S.; Madaboosi, N.; Uhlig, K.; Köhler, D.; Skirtach, A.; Duschl, C.; Möhwald, H.; Volodkin, D.V. Control of cell adhesion by mechanical reinforcement of soft polyelectrolyte films with nanoparticles. Langmuir 2012, 28, 7249–7257. [Google Scholar] [CrossRef] [PubMed]
- Abalymov, A.A.; Parakhonskiy, B.V.; Skirtach, A.G. Colloids-at-surfaces: Physicochemical approaches for facilitating cell adhesion on hybrid hydrogels. Colloids Surf. A Physicochem. Eng. Asp. 2020, 603, 125185. [Google Scholar] [CrossRef]
- Kovačević, D.; Pratnekar, R.; Godič Torkar, K.; Salopek, J.; Dražić, G.; Abram, A.; Bohinc, K. Influence of Polyelectrolyte Multilayer Properties on Bacterial Adhesion Capacity. Polymers 2016, 8, 345. [Google Scholar]
- Guo, S.; Kwek, M.Y.; Toh, Z.Q.; Pranantyo, D.; Kang, E.-T.; Loh, X.J.; Zhu, X.; Jańczewski, D.; Neoh, K.G. Tailoring Polyelectrolyte Architecture To Promote Cell Growth and Inhibit Bacterial Adhesion. ACS Appl. Mater. Interfaces 2018, 10, 7882–7891. [Google Scholar] [CrossRef]
- Carvalho, A.L.; Vale, A.C.; Sousa, M.P.; Barbosa, A.M.; Torrado, E.; Mano, J.F.; Alves, N.M. Antibacterial bioadhesive layer-by-layer coatings for orthopedic applications. J. Mater. Chem. B 2016, 4, 5385–5393. [Google Scholar] [CrossRef]
- Abalymov, A.A.; Van Der Meeren, L.; Saveleva, M.; Prikhozhdenko, E.; Dewettinck, K.; Parakhonskiy, B.; Skirtach, A.G. Cells-Grab-on Particles: A Novel Approach to Control Cell Focal Adhesion on Hybrid Thermally Annealed Hydrogels. ACS Biomater. Sci. Eng. 2020, 6, 3933–3944. [Google Scholar] [CrossRef]
- Abalymov, A.A.; Parakhonskiy, B.; Skirtach, A.G. Polymer- and Hybrid-Based Biomaterials for Interstitial, Connective, Vascular, Nerve, Visceral and Musculoskeletal Tissue Engineering. Polymers 2020, 12, 620. [Google Scholar] [CrossRef]
- Kolesnikova, T.A.; Kohler, D.; Skirtach, A.G.; Möhwald, H. Laser-Induced Cell Detachment, Patterning, and Regrowth on Gold Nanoparticle Functionalized Surfaces. ACS Nano 2012, 6, 9585–9595. [Google Scholar] [CrossRef] [PubMed]
- Bai, J.; Zuo, X.; Feng, X.; Sun, Y.; Ge, Q.; Wang, X.; Gao, C. Dynamic Titania Nanotube Surface Achieves UV-Triggered Charge Reversal and Enhances Cell Differentiation. ACS Appl. Mater. Interfaces 2019, 11, 36939–36948. [Google Scholar] [CrossRef] [PubMed]
- Xing, R.; Jiao, T.; Ma, K.; Ma, G.; Möhwald, H.; Yan, X. Regulating Cell Apoptosis on Layer-by-Layer Assembled Multilayers of Photosensitizer-Coupled Polypeptides and Gold Nanoparticles. Sci. Rep. 2016, 6, 26506. [Google Scholar] [CrossRef] [PubMed]
- Skirtach, A.G.; Volodkin, D.V.; Möhwald, H. Bio-interfaces-Interaction of PLL/HA Thick Films with Nanoparticles and Microcapsules. ChemPhysChem 2010, 11, 822–829. [Google Scholar] [CrossRef]
- De Gennes, P.-G. Soft Matter (Nobel Lecture). Angew. Chem. Int. Ed. Engl. 1992, 31, 842–845. [Google Scholar] [CrossRef]
- Skorb, E.V.; Möhwald, H. 25th Anniversary Article: Dynamic Interfaces for Responsive Encapsulation Systems. Adv. Mater. 2013, 25, 5029–5043. [Google Scholar] [CrossRef]
- Li, J.; Gao, W.; Dong, R.; Pei, A.; Sattayasamitsathit, S.; Wang, J. Nanomotor lithography. Nat. Commun. 2014, 5, 5026. [Google Scholar] [CrossRef]
- Guix, M.; Meyer, A.K.; Koch, B.; Schmidt, O.G. Carbonate-based Janus micromotors moving in ultra-light acidic environment generated by HeLa cells in situ. Sci. Rep. 2016, 6, 21701. [Google Scholar] [CrossRef]
- Dong, R.; Li, J.; Rozen, I.; Ezhilan, B.; Xu, T.; Christianson, C.; Gao, W.; Saintillan, D.; Ren, B.; Wang, J. Vapor-Driven Propulsion of Catalytic Micromotors. Sci. Rep. 2015, 5, 13226. [Google Scholar] [CrossRef]
- Shchukin, D.G.D.G.; Zheludkevich, M.L.; Yasakau, K.a.; Lamaka, S.; Ferreira, M.G.S.M.G.S.; Möhwald, H. Layer-by-Layer Assembled Nanocontainers for Self-Healing Corrosion Protection. Adv. Mater. 2006, 18, 1672–1678. [Google Scholar] [CrossRef]
- Andreeva, D.V.; Fix, D.; Möhwald, H.; Shchukin, D.G. Self-Healing Anticorrosion Coatings Based on pH-Sensitive Polyelectrolyte/Inhibitor Sandwichlike Nanostructures. Adv. Mater. 2008, 20, 2789–2794. [Google Scholar] [CrossRef] [PubMed]
- Tong, W.; Song, X.; Gao, C. Layer-by-layer assembly of microcapsules and their biomedical applications. Chem. Soc. Rev. 2012, 41, 6103–6124. [Google Scholar] [CrossRef] [PubMed]
- Vergaro, V.; Scarlino, F.; Bellomo, C.; Rinaldi, R.; Vergara, D.; Maffia, M.; Baldassarre, F.; Giannelli, G.; Zhang, X.; Lvov, Y.M.; et al. Drug-loaded polyelectrolyte microcapsules for sustained targeting of cancer cells. Adv. Drug Deliv. Rev. 2011, 63, 847–864. [Google Scholar] [CrossRef] [PubMed]
- Zhao, T.; Chen, L.; Wang, P.; Li, B.; Lin, R.; Abdulkareem Al-Khalaf, A.; Hozzein, W.N.; Zhang, F.; Li, X.; Zhao, D. Surface-kinetics mediated mesoporous multipods for enhanced bacterial adhesion and inhibition. Nat. Commun. 2019, 10, 4387. [Google Scholar] [CrossRef] [PubMed]
- Ling, X.Y.; Phang, I.Y.; Acikgoz, C.; Yilmaz, M.D.; Hempenius, M.A.; Vancso, G.J.; Huskens, J. Janus Particles with Controllable Patchiness and Their Chemical Functionalization and Supramolecular Assembly. Angew. Chem. Int. Ed. 2009, 48, 7677–7682. [Google Scholar] [CrossRef]
- Pawar, A.B.; Kretzschmar, I. Fabrication, Assembly, and Application of Patchy Particles. Macromol. Rapid Commun. 2010, 31, 150–168. [Google Scholar] [CrossRef]
- Yoshida, M.; Roh, K.-H.; Mandal, S.; Bhaskar, S.; Lim, D.; Nandivada, H.; Deng, X.; Lahann, J. Structurally Controlled Bio-hybrid Materials Based on Unidirectional Association of Anisotropic Microparticles with Human Endothelial Cells. Adv. Mater. 2009, 21, 4920–4925. [Google Scholar] [CrossRef]
- Chen, R.T.; Muir, B.W.; Such, G.K.; Postma, A.; McLean, K.M.; Caruso, F. Fabrication of asymmetric “Janus” particles via plasma polymerization. Chem. Commun. 2010, 46, 5121–5123. [Google Scholar] [CrossRef]
- Paunov, V.N.; Cayre, O.J. Supraparticles and “Janus” Particles Fabricated by Replication of Particle Monolayers at Liquid Surfaces Using a Gel Trapping Technique. Adv. Mater. 2004, 16, 788–791. [Google Scholar] [CrossRef]
- Li, Z.; Lee, D.; Rubner, M.F.; Cohen, R.E. Layer-by-Layer Assembled Janus Microcapsules. Macromolecules 2005, 38, 7876–7879. [Google Scholar] [CrossRef]
- Cui, J.-Q.; Kretzschmar, I. Surface-Anisotropic Polystyrene Spheres by Electroless Deposition. Langmuir 2006, 22, 8281–8284. [Google Scholar] [CrossRef] [PubMed]
- Kohler, D.; Madaboosi, N.; Delcea, M.; Schmidt, S.; De Geest, B.G.; Volodkin, D.V.; Möhwald, H.; Skirtach, A.G. Patchiness of embedded particles and film stiffness control through concentration of gold nanoparticles. Adv. Mater. 2012, 24, 1095–1100. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; Rudov, A.; Oppermann, A.; Wypysek, S.; Kather, M.; Schroeder, R.; Richtering, W.; Potemkin, I.I.; Wöll, D.; Pich, A. Synthesis of Polyampholyte Janus-like Microgels by Coacervation of Reactive Precursors in Precipitation Polymerization. Angew. Chem. Int. Ed. 2020, 59, 1248–1255. [Google Scholar] [CrossRef] [PubMed]
- Kobaku, S.P.R.; Snyder, C.S.; Karunakaran, R.G.; Kwon, G.; Wong, P.; Tuteja, A.; Mehta, G. Wettability Engendered Templated Self-Assembly (WETS) for the Fabrication of Biocompatible, Polymer–Polyelectrolyte Janus Particles. ACS Macro Lett. 2019, 8, 1491–1497. [Google Scholar] [CrossRef]
- Ji, Y.; Lin, X.; Wang, D.; Zhou, C.; Wu, Y.; He, Q. Continuously Variable Regulation of the Speed of Bubble-Propelled Janus Microcapsule Motors Based on Salt-Responsive Polyelectrolyte Brushes. Chem. Asian J. 2019, 14, 2450–2455. [Google Scholar] [CrossRef] [PubMed]
- Shao, J.; Abdelghani, M.; Shen, G.; Cao, S.; Williams, D.S.; van Hest, J.C.M. Erythrocyte Membrane Modified Janus Polymeric Motors for Thrombus Therapy. ACS Nano 2018, 12, 4877–4885. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Lin, X.; Wu, Z.; Möhwald, H.; He, Q. Self-Propelled Polymer Multilayer Janus Capsules for Effective Drug Delivery and Light-Triggered Release. ACS Appl. Mater. Interfaces 2014, 6, 10476–10481. [Google Scholar] [CrossRef]
- Wu, Y.; Si, T.; Lin, X.; He, Q. Near infrared-modulated propulsion of catalytic Janus polymer multilayer capsule motors. Chem. Commun. 2015, 51, 511–514. [Google Scholar] [CrossRef]
- Yoshizumi, Y.; Suzuki, H. Self-Propelled Metal–Polymer Hybrid Micromachines with Bending and Rotational Motions. ACS Appl. Mater. Interfaces 2017, 9, 21355–21361. [Google Scholar] [CrossRef]
- Marschelke, C.; Raguzin, I.; Matura, A.; Fery, A.; Synytska, A. Controlled and tunable design of polymer interface for immobilization of enzymes: Does curvature matter? Soft Matter 2017, 13, 1074–1084. [Google Scholar] [CrossRef]
- Lin, Z.; Wu, Z.; Lin, X.; He, Q. Catalytic Polymer Multilayer Shell Motors for Separation of Organics. Chem. Eur. J. 2016, 22, 1587–1591. [Google Scholar] [CrossRef] [PubMed]
- Hu, N.; Zhang, B.; Gai, M.; Zheng, C.; Frueh, J.; He, Q. Forecastable and Guidable Bubble-Propelled Microplate Motors for Cell Transport. Macromol. Rapid Commun. 2017, 38, 1600795. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, J.B.; O’Brien, J.S.; Suresh, H.S.; Cohen, R.E.; Rubner, M.F. Orientation-Specific Attachment of Polymeric Microtubes on Cell Surfaces. Adv. Mater. 2013, 5948–5952. [Google Scholar] [CrossRef] [PubMed]
- He, W.; Frueh, J.; Wu, Z.; He, Q. Leucocyte Membrane-Coated Janus Microcapsules for Enhanced Photothermal Cancer Treatment. Langmuir 2016, 32, 3637–3644. [Google Scholar] [CrossRef] [PubMed]
- He, W.; Frueh, J.; Wu, Z.; He, Q. How Leucocyte Cell Membrane Modified Janus Microcapsules are Phagocytosed by Cancer Cells. ACS Appl. Mater. Interfaces 2016, 8, 4407–4415. [Google Scholar] [CrossRef] [PubMed]
- He, W.; Frueh, J.; Hu, N.; Liu, L.; Gai, M.; He, Q. Guidable Thermophoretic Janus Micromotors Containing Gold Nanocolorifiers for Infrared Laser Assisted Tissue Welding. Adv. Sci. 2016, 3, 1600206. [Google Scholar] [CrossRef]
- Volodkin, D.; Skirtach, A.; Möhwald, H. Bioapplications of light-sensitive polymer films and capsules assembled using the layer-by-layer technique. Polym. Int. 2012, 61, 673–679. [Google Scholar] [CrossRef]
- Andreeva, D.V.; Skorb, E.V.; Shchukin, D.G. Layer-by-Layer Polyelectrolyte/Inhibitor Nanostructures for Metal Corrosion Protection. ACS Appl. Mater. Interfaces 2010, 2, 1954–1962. [Google Scholar] [CrossRef]
- Andreeva, D.V.; Fix, D.; Möhwald, H.; Shchukin, D.G. Buffering polyelectrolyte multilayers for active corrosion protection. J. Mater. Chem. 2008, 18, 1738–1740. [Google Scholar] [CrossRef]
- Haneder, S.; Da Como, E.; Feldmann, J.; Lupton, J.M.; Lennartz, C.; Erk, P.; Fuchs, E.; Molt, O.; Münster, I.; Schildknecht, C.; et al. Controlling the Radiative Rate of Deep-Blue Electrophosphorescent Organometallic Complexes by Singlet-Triplet Gap Engineering. Adv. Mater. 2008, 20, 3325–3330. [Google Scholar] [CrossRef]
- Grigoriev, D.O.; Köhler, K.; Skorb, E.; Shchukin, D.G.; Möhwald, H. Polyelectrolyte complexes as a “smart” depot for self-healing anticorrosion coatings. Soft Matter 2009, 5, 1426–1432. [Google Scholar] [CrossRef]
- Skorb, E.V.; Skirtach, A.G.; Sviridov, D.V.; Shchukin, D.G.; Möhwald, H. Laser-Controllable Coatings for Corrosion Protection. ACS Nano 2009, 3, 1753–1760. [Google Scholar] [CrossRef] [PubMed]
- Jafari, A.H.; Hosseini, S.M.A.; Jamalizadeh, E. Investigation of Smart Nanocapsules Containing Inhibitors for Corrosion Protection of Copper. Electrochim. Acta 2010, 55, 9004–9009. [Google Scholar] [CrossRef]
- Kopeć, M.; Szczepanowicz, K.; Mordarski, G.; Podgórna, K.; Socha, R.P.; Nowak, P.; Warszyński, P.; Hack, T. Self-healing epoxy coatings loaded with inhibitor-containing polyelectrolyte nanocapsules. Prog. Org. Coat. 2015, 84, 97–106. [Google Scholar] [CrossRef]
- Zhao, X.; Yuan, S.; Jin, Z.; Zhang, B.; Liu, N.; Chen, S.; Liu, S.; Sun, X.; Duan, J. Perfect Combination of LBL with Sol–Gel Film to Enhance the Anticorrosion Performance on Al Alloy under Simulated and Accelerated Corrosive Environment. Materials 2019, 13, 111. [Google Scholar] [CrossRef]
- Fan, F.; Zhou, C.; Wang, X.; Szpunar, J. Layer-by-Layer Assembly of a Self-Healing Anticorrosion Coating on Magnesium Alloys. ACS Appl. Mater. Interfaces 2015, 7, 27271–27278. [Google Scholar] [CrossRef]
- Skorb, E.V.; Sviridov, D.V.; Möhwald, H.; Shchukin, D.G. Light responsive protective coatings. Chem. Commun. 2009, 6041–6043. [Google Scholar] [CrossRef]
- Skorb, E.V.; Fix, D.; Andreeva, D.V.; Möhwald, H.; Shchukin, D.G. Surface-Modified Mesoporous SiO2 Containers for Corrosion Protection. Adv. Funct. Mater. 2009, 19, 2373–2379. [Google Scholar] [CrossRef]
- Skorb, E.V.; Shchukin, D.G.; Möhwald, H.; Sviridov, D.V. Photocatalytically-active and photocontrollable coatings based on titania-loaded hybrid sol–gel films. J. Mater. Chem. 2009, 19, 4931–4937. [Google Scholar] [CrossRef]
- Ulasevich, S.A.; Brezesinski, G.; Möhwald, H.; Fratzl, P.; Schacher, F.H.; Poznyak, S.K.; Andreeva, D.V.; Skorb, E.V. Light-Induced Water Splitting Causes High-Amplitude Oscillation of pH-Sensitive Layer-by-Layer Assemblies on TiO2. Angew. Chem. Int. Ed. 2016, 55, 13001–13004. [Google Scholar] [CrossRef]
- Maltanava, H.M.; Poznyak, S.K.; Andreeva, D.V.; Quevedo, M.C.; Bastos, A.C.; Tedim, J.; Ferreira, M.G.S.; Skorb, E.V. Light-Induced Proton Pumping with a Semiconductor: Vision for Photoproton Lateral Separation and Robust Manipulation. ACS Appl. Mater. Interfaces 2017, 9, 24282–24289. [Google Scholar] [CrossRef] [PubMed]
- Ryzhkov, N.V.; Skorb, E.V. A platform for light-controlled formation of free-stranding lipid membranes. J. R. Soc. Interface 2020, 17, 20190740. [Google Scholar] [CrossRef]
- Fenoy, G.E.; Maza, E.; Zelaya, E.; Marmisollé, W.A.; Azzaroni, O. Layer-by-layer assemblies of highly connected polyelectrolyte capped-Pt nanoparticles for electrocatalysis of hydrogen evolution reaction. Appl. Surf. Sci. 2017, 416, 24–32. [Google Scholar] [CrossRef]
- Skorb, E.V.; Antonouskaya, L.I.; Belyasova, N.A.; Shchukin, D.G.; Möhwald, H.; Sviridov, D.V. Antibacterial activity of thin-film photocatalysts based on metal-modified TiO2 and TiO2:In2O3 nanocomposite. Appl. Catal. B Environ. 2008, 84, 94–99. [Google Scholar] [CrossRef]
- Lanchuk, Y.; Nikitina, A.; Brezhneva, N.; Ulasevich, S.A.; Semenov, S.N.; Skorb, E.V. Photocatalytic Regulation of an Autocatalytic Wave of Spatially Propagating Enzymatic Reactions. ChemCatChem 2018, 10, 1798–1803. [Google Scholar] [CrossRef]
- Nikitina, A.A.; Ulasevich, S.A.; Kassirov, I.S.; Bryushkova, E.A.; Koshel, E.I.; Skorb, E.V. Nanostructured Layer-by-Layer Polyelectrolyte Containers to Switch Biofilm Fluorescence. Bioconjug. Chem. 2018, 29, 3793–3799. [Google Scholar] [CrossRef]
- Ryzhkov, N.V.; Yurova, V.Y.; Ulasevich, S.A.; Skorb, E.V. Photoelectrochemical photocurrent switching effect on a pristine anodized Ti/TiO2 system as a platform for chemical logic devices. RSC Adv. 2020, 10, 12355–12359. [Google Scholar] [CrossRef]
- Lanchuk, Y.V.; Ulasevich, S.A.; Fedotova, T.A.; Kolpashchikov, D.M.; Skorb, E.V. Towards sustainable diagnostics: Replacing unstable H2O2 by photoactive TiO2 in testing systems for visible and tangible diagnostics for use by blind people. RSC Adv. 2018, 8, 37735–37739. [Google Scholar] [CrossRef]
- Stekolshchikova, A.A.; Radaev, A.V.; Orlova, O.Y.; Nikolaev, K.G.; Skorb, E.V. Thin and Flexible Ion Sensors Based on Polyelectrolyte Multilayers Assembled onto the Carbon Adhesive Tape. ACS Omega 2019, 4, 15421–15427. [Google Scholar] [CrossRef]
- Nikolaev, K.G.; Kalmykov, E.V.; Shavronskaya, D.O.; Nikitina, A.A.; Stekolshchikova, A.A.; Kosareva, E.A.; Zenkin, A.A.; Pantiukhin, I.S.; Orlova, O.Y.; Skalny, A.V.; et al. ElectroSens Platform with a Polyelectrolyte-Based Carbon Fiber Sensor for Point-of-Care Analysis of Zn in Blood and Urine. ACS Omega 2020, 5, 18987–18994. [Google Scholar] [CrossRef]
- Andreeva, D.V.; Sviridov, D.V.; Masic, A.; Möhwald, H.; Skorb, E.V. Nanoengineered Metal Surface Capsules: Construction of a Metal-Protection System. Small 2012, 8, 820–825. [Google Scholar] [CrossRef] [PubMed]
- Gensel, J.; Borke, T.; Pérez, N.P.; Fery, A.; Andreeva, D.V.; Betthausen, E.; Müller, A.H.E.; Möhwald, H.; Skorb, E.V. Cavitation Engineered 3D Sponge Networks and Their Application in Active Surface Construction. Adv. Mater. 2012, 24, 985–989. [Google Scholar] [CrossRef] [PubMed]
- Rivero, P.J.; Goicoechea, J.; Hernaez, M.; Socorro, A.B.; Matias, I.R.; Arregui, F.J. Optical fiber resonance-based pH sensors using gold nanoparticles into polymeric layer-by-layer coatings. Microsyst. Technol. 2016, 22, 1821–1829. [Google Scholar] [CrossRef]
- Tokarev, I.; Tokareva, I.; Minko, S. Optical Nanosensor Platform Operating in Near-Physiological pH Range via Polymer-Brush-Mediated Plasmon Coupling. ACS Appl. Mater. Interfaces 2011, 3, 143–146. [Google Scholar] [CrossRef]
- Kozlovskaya, V.; Kharlampieva, E.; Khanal, B.P.; Manna, P.; Zubarev, E.R.; Tsukruk, V.V. Ultrathin Layer-by-Layer Hydrogels with Incorporated Gold Nanorods as pH-Sensitive Optical Materials. Chem. Mater. 2008, 20, 7474–7485. [Google Scholar] [CrossRef]
- De Oliveira, R.D.; Calaça, G.N.; Santos, C.S.; Fujiwara, S.T.; Pessôa, C.A. Preparation, characterization and electrochemistry of Layer-by-Layer films of silver nanoparticles and silsesquioxane polymer. Colloids Surf. A Physicochem. Eng. Asp. 2016, 509, 638–647. [Google Scholar] [CrossRef]
- Irigoyen, J.; Laakso, T.; Politakos, N.; Dahne, L.; Pihlajamäki, A.; Mänttäri, M.; Moya, S.E. Design and Performance Evaluation of Hybrid Nanofiltration Membranes Based on Multiwalled Carbon Nanotubes and Polyelectrolyte Multilayers for Larger Ion Rejection and Separation. Macromol. Chem. Phys. 2016, 217, 804–811. [Google Scholar] [CrossRef]
- Stanojev, J.; Bajac, B.; Cvejic, Z.; Matovic, J.; Srdic, V.V. Development of MWCNT thin film electrode transparent in the mid-IR range. Ceram. Int. 2020, 46, 11340–11345. [Google Scholar] [CrossRef]
- Barsan, M.M.; Brett, C.M.A. Recent advances in layer-by-layer strategies for biosensors incorporating metal nanoparticles. TrAC Trends Anal. Chem. 2016, 79, 286–296. [Google Scholar] [CrossRef]
- McShane, M.; Ritter, D. Microcapsules as optical biosensors. J. Mater. Chem. 2010, 20, 8189–8193. [Google Scholar] [CrossRef]
- Sukhorukov, G.B.; Rogach, A.L.; Garstka, M.; Springer, S.; Parak, W.J.; Muñoz-Javier, A.; Kreft, O.; Skirtach, A.G.; Susha, A.S.; Ramaye, Y.; et al. Multifunctionalized polymer microcapsules: Novel tools for biological and pharmacological applications. Small 2007, 3, 944–955. [Google Scholar] [CrossRef] [PubMed]
- Van der Meeren, L.; Li, J.; Parakhonskiy, B.V.; Krysko, D.V.; Skirtach, A.G. Classification of analytics, sensorics, and bioanalytics with polyelectrolyte multilayer capsules. Anal. Bioanal. Chem. 2020, 412, 5015–5029. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Liu, C.; Chen, H.; Zhang, Y.; Li, L.; Tang, N. Layer-by-Layer Deposition of Hyaluronan and Quercetin-Loaded Chitosan Nanoparticles onto Titanium for Improving Blood Compatibility. Coatings 2020, 10, 256. [Google Scholar] [CrossRef]
- Sydow, S.; de Cassan, D.; Hänsch, R.; Gengenbach, T.R.; Easton, C.D.; Thissen, H.; Menzel, H. Layer-by-layer deposition of chitosan nanoparticles as drug-release coatings for PCL nanofibers. Biomater. Sci. 2019, 7, 233–246. [Google Scholar] [CrossRef] [PubMed]
- Campos, P.P.; Dunne, A.; Delaney, C.; Moloney, C.; Moulton, S.E.; Benito-Lopez, F.; Ferreira, M.; Diamond, D.; Florea, L. Photoswitchable Layer-by-Layer Coatings Based on Photochromic Polynorbornenes Bearing Spiropyran Side Groups. Langmuir 2018, 34, 4210–4216. [Google Scholar] [CrossRef] [PubMed]
- Rouster, P.; Dondelinger, M.; Galleni, M.; Nysten, B.; Jonas, A.M.; Glinel, K. Layer-by-layer assembly of enzyme-loaded halloysite nanotubes for the fabrication of highly active coatings. Colloids Surf. B Biointerfaces 2019, 178, 508–514. [Google Scholar] [CrossRef]
- Rogach, A.L. (Ed.) Semiconductor Nanocrystal Quantum Dots; Springer Vienna: Vienna, Austria, 2008; ISBN 978-3-211-75235-7. [Google Scholar]
- Abd Rahman, S.; Ariffin, N.; Yusof, N.; Abdullah, J.; Mohammad, F.; Ahmad Zubir, Z.; Nik Abd Aziz, N. Thiolate-Capped CdSe/ZnS Core-Shell Quantum Dots for the Sensitive Detection of Glucose. Sensors 2017, 17, 1537. [Google Scholar] [CrossRef]
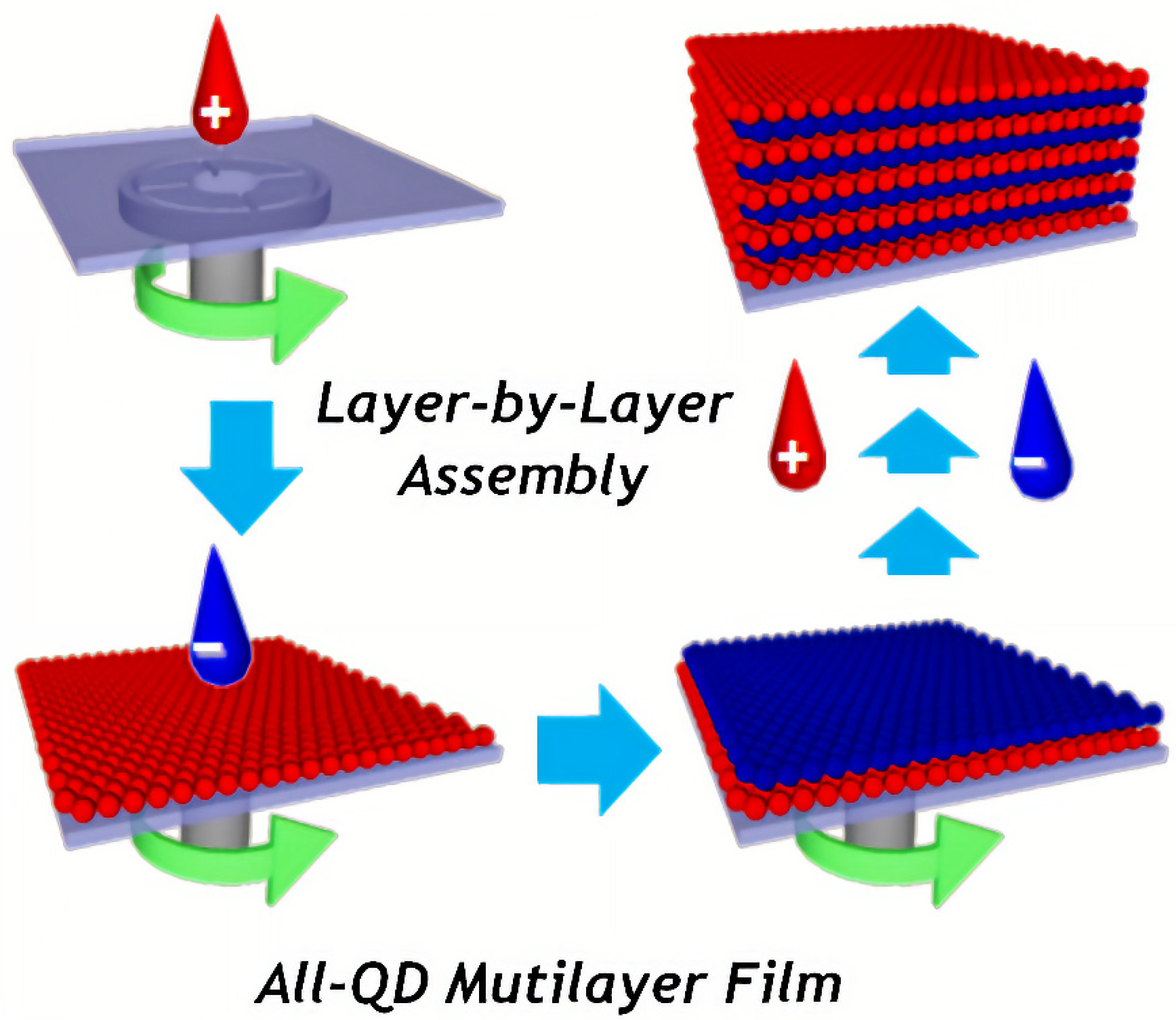
- Bae, W.K.; Kwak, J.; Lim, J.; Lee, D.; Nam, M.K.; Char, K.; Lee, C.; Lee, S. Multicolored Light-Emitting Diodes Based on All-Quantum-Dot Multilayer Films Using Layer-by-Layer Assembly Method. Nano Lett. 2010, 10, 2368–2373. [Google Scholar] [CrossRef]
- Jaffar, S.; Nam, K.T.; Khademhosseini, A.; Xing, J.; Langer, R.S.; Belcher, A.M. Layer-by-Layer Surface Modification and Patterned Electrostatic Deposition of Quantum Dots. Nano Lett. 2004, 4, 1421–1425. [Google Scholar] [CrossRef]
- Nagaraja, A.T.; Sooresh, A.; Meissner, K.E.; McShane, M.J. Processing and Characterization of Stable, pH-Sensitive Layer-by-Layer Modified Colloidal Quantum Dots. ACS Nano 2013, 7, 6194–6202. [Google Scholar] [CrossRef]
- Zimnitsky, D.; Jiang, C.; Xu, J.; Lin, Z.; Zhang, L.; Tsukruk, V.V. Photoluminescence of a Freely Suspended Monolayer of Quantum Dots Encapsulated into Layer-by-Layer Films. Langmuir 2007, 23, 10176–10183. [Google Scholar] [CrossRef] [PubMed]
- Constantine, C.A.; Gattás-Asfura, K.M.; Mello, S.V.; Crespo, G.; Rastogi, V.; Cheng, T.-C.; DeFrank, J.J.; Leblanc, R.M. Layer-by-Layer Biosensor Assembly Incorporating Functionalized Quantum Dots. Langmuir 2003, 19, 9863–9867. [Google Scholar] [CrossRef]
- Xiao, F.-X.; Miao, J.; Liu, B. Layer-by-Layer Self-Assembly of CdS Quantum Dots/Graphene Nanosheets Hybrid Films for Photoelectrochemical and Photocatalytic Applications. J. Am. Chem. Soc. 2014, 136, 1559–1569. [Google Scholar] [CrossRef] [PubMed]
- Nifontova, G.; Ramos-Gomes, F.; Baryshnikova, M.; Alves, F.; Nabiev, I.; Sukhanova, A. Cancer Cell Targeting With Functionalized Quantum Dot-Encoded Polyelectrolyte Microcapsules. Front. Chem. 2019, 7, 34. [Google Scholar] [CrossRef] [PubMed]
- Nifontova, G.; Efimov, A.; Agapova, O.; Agapov, I.; Nabiev, I.; Sukhanova, A. Bioimaging Tools Based on Polyelectrolyte Microcapsules Encoded with Fluorescent Semiconductor Nanoparticles: Design and Characterization of the Fluorescent Properties. Nanoscale Res. Lett. 2019, 14, 29. [Google Scholar] [CrossRef]
- Adamczak, M.; Hoel, H.J.; Gaudernack, G.; Barbasz, J.; Szczepanowicz, K.; Warszyński, P. Polyelectrolyte multilayer capsules with quantum dots for biomedical applications. Colloids Surf. B Biointerfaces 2012, 90, 211–216. [Google Scholar] [CrossRef]
- Rogach, A.L.; Kotov, N.A.; Koktysh, D.S.; Susha, A.S.; Caruso, F. II–VI semiconductor nanocrystals in thin films and colloidal crystals. Colloids Surf. A Physicochem. Eng. Asp. 2002, 202, 135–144. [Google Scholar] [CrossRef]
- Wang, D.; Rogach, A.L.; Caruso, F. Semiconductor Quantum Dot-Labeled Microsphere Bioconjugates Prepared by Stepwise Self-Assembly. Nano Lett. 2002, 2, 857–861. [Google Scholar] [CrossRef]
- Hashimoto, M.; Feng, J.; York, R.L.; Ellerbee, A.K.; Morrison, G.; Thomas III, S.W.; Mahadevan, L.; Whitesides, G.M. Infochemistry: Encoding Information as Optical Pulses Using Droplets in a Microfluidic Device. J. Am. Chem. Soc. 2009, 131, 12420–12429. [Google Scholar] [CrossRef]
- Ryzhkov, N.V.; Andreeva, D.V.; Skorb, E.V. Coupling pH-Regulated Multilayers with Inorganic Surfaces for Bionic Devices and Infochemistry. Langmuir 2019, 35, 8543–8556. [Google Scholar] [CrossRef]
- Han, M.; Gao, X.; Su, J.Z.; Nie, S. Quantum-dot-tagged microbeads for multiplexed optical coding of biomolecules. Nat. Biotechnol. 2001, 19, 631–635. [Google Scholar] [CrossRef] [PubMed]
- Pinchasik, B.-E.; Tauer, K.; Möhwald, H.; Skirtach, A.G. Polymer Brush Gradients by Adjusting the Functional Density Through Temperature Gradient. Adv. Mater. Interfaces 2014, 1, 1300056. [Google Scholar] [CrossRef]
- Sailer, M.; Barrett, C.J. Fabrication of Two-Dimensional Gradient Layer-by-Layer Films for Combinatorial Biosurface Studies. Macromolecules 2012, 45, 5704–5711. [Google Scholar] [CrossRef]
- Ariga, K.; Ahn, E.; Park, M.; Kim, B. Layer-by-Layer Assembly: Recent Progress from Layered Assemblies to Layered Nanoarchitectonics. Chem. Asian J. 2019, 14, 2553–2566. [Google Scholar] [CrossRef] [PubMed]
- Yu, A.; Zhang, X.; Zhang, H.; Han, D.; Knight, A.R. Preparation and electrochemical properties of gold nanoparticles containing carbon nanotubes-polyelectrolyte multilayer thin films. Electrochim. Acta 2011, 56, 9015–9019. [Google Scholar] [CrossRef]
- Vallés, C.; Zhang, X.; Cao, J.; Lin, F.; Young, R.J.; Lombardo, A.; Ferrari, A.C.; Burk, L.; Mülhaupt, R.; Kinloch, I.A. Graphene/Polyelectrolyte Layer-by-Layer Coatings for Electromagnetic Interference Shielding. ACS Appl. Nano Mater. 2019, 2, 5272–5281. [Google Scholar] [CrossRef]
- Liu, H.; Bandyopadhyay, P.; Kshetri, T.; Kim, N.H.; Ku, B.-C.; Moon, B.; Lee, J.H. Layer-by-layer assembled polyelectrolyte-decorated graphene multilayer film for hydrogen gas barrier application. Compos. Part B Eng. 2017, 114, 339–347. [Google Scholar] [CrossRef]
- Kurapati, R.; Raichur, A.M. Graphene oxide based multilayer capsules with unique permeability properties: Facile encapsulation of multiple drugs. Chem. Commun. 2012, 48, 6013–6015. [Google Scholar] [CrossRef]
- Ermakov, A.; Lim, S.H.; Gorelik, S.; Kauling, A.P.; de Oliveira, R.V.B.; Castro Neto, A.H.; Glukhovskoy, E.; Gorin, D.A.; Sukhorukov, G.B.; Kiryukhin, M.V. Polyelectrolyte-Graphene Oxide Multilayer Composites for Array of Microchambers which are Mechanically Robust and Responsive to NIR Light. Macromol. Rapid Commun. 2019, 40, 1700868. [Google Scholar] [CrossRef]
- Pan, H.; Lu, Y.; Song, L.; Zhang, X.; Hu, Y. Construction of layer-by-layer coating based on graphene oxide/β-FeOOH nanorods and its synergistic effect on improving flame retardancy of flexible polyurethane foam. Compos. Sci. Technol. 2016, 129, 116–122. [Google Scholar] [CrossRef]
- Gao, H.; Qi, X.; Chen, Y.; Sun, W. Electrochemical deoxyribonucleic acid biosensor based on the self-assembly film with nanogold decorated on ionic liquid modified carbon paste electrode. Anal. Chim. Acta 2011, 704, 133–138. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Chai, Y.; Yuan, R.; Su, H.; Han, J. A novel label-free electrochemical microRNA biosensor using Pd nanoparticles as enhancer and linker. Analyst 2013, 138, 1060–1066. [Google Scholar] [CrossRef] [PubMed]
- Erokhina, S.; Ricci, V.; Iannotta, S.; Erokhin, V. Modification of the porous glass filter with LbL technique for variable filtration applications. Colloids Surf. A Physicochem. Eng. Asp. 2020, 606, 125459. [Google Scholar] [CrossRef]
- Saveleva, M.S.; Lengert, E.V.; Gorin, D.A.; Parakhonskiy, B.V.; Skirtach, A.G. Polymeric and Lipid Membranes—From Spheres to Flat Membranes and vice versa. Membranes 2017, 7, 44. [Google Scholar] [CrossRef] [PubMed]
- Egueh, A.-N.D.; Lakard, B.; Fievet, P.; Lakard, S.; Buron, C. Charge properties of membranes modified by multilayer polyelectrolyte adsorption. J. Colloid Interface Sci. 2010, 344, 221–227. [Google Scholar] [CrossRef] [PubMed]
- Lvov, Y.; Wang, W.; Zhang, L.; Fakhrullin, R. Halloysite Clay Nanotubes for Loading and Sustained Release of Functional Compounds. Adv. Mater. 2016, 28, 1227–1250. [Google Scholar] [CrossRef] [PubMed]
- Konnova, S.A.; Sharipova, I.R.; Demina, T.A.; Osin, Y.N.; Yarullina, D.R.; Ilinskaya, O.N.; Lvov, Y.M.; Fakhrullin, R.F. Biomimetic cell-mediated three-dimensional assembly of halloysite nanotubes. Chem. Commun. 2013, 49, 4208–4210. [Google Scholar] [CrossRef]
- Dreaden, E.C.; Morton, S.W.; Shopsowitz, K.E.; Choi, J.-H.; Deng, Z.J.; Cho, N.-J.; Hammond, P.T. Bimodal Tumor-Targeting from Microenvironment Responsive Hyaluronan Layer-by-Layer (LbL) Nanoparticles. ACS Nano 2014, 8, 8374–8382. [Google Scholar] [CrossRef]
- Häuser, M.; Langer, K.; Schönhoff, M. pH-Triggered release from surface-modified poly(lactic-co-glycolic acid) nanoparticles. Beilstein J. Nanotechnol. 2015, 6, 2504–2512. [Google Scholar] [CrossRef]
- Schneider, G.; Decher, G. From Functional Core/Shell Nanoparticles Prepared via Layer-by-Layer Deposition to Empty Nanospheres. Nano Lett. 2004, 4, 1833–1839. [Google Scholar] [CrossRef]
- Yang, Q.; Li, L.; Zhao, F.; Wang, Y.; Ye, Z.; Hua, C.; Liu, Z.; Bohinc, K.; Guo, X. Spherical Polyelectrolyte Brushes as Templates to Prepare Hollow Silica Spheres Encapsulating Metal Nanoparticles. Nanomaterials 2020, 10, 799. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Bradley, S.J.; Chan, A.; Waterhouse, G.I.N.; Nann, T.; Kruger, P.E.; Telfer, S.G. Catalytically Active Bimetallic Nanoparticles Supported on Porous Carbon Capsules Derived From Metal–Organic Framework Composites. J. Am. Chem. Soc. 2016, 138, 11872–11881. [Google Scholar] [CrossRef] [PubMed]
- Zhao, W.; Wei, J.-S.; Zhang, P.; Chen, J.; Kong, J.-L.; Sun, L.-H.; Xiong, H.-M.; Möhwald, H. Self-Assembled ZnO Nanoparticle Capsules for Carrying and Delivering Isotretinoin to Cancer Cells. ACS Appl. Mater. Interfaces 2017, 9, 18474–18481. [Google Scholar] [CrossRef] [PubMed]
- De Geest, B.G.; Skirtach, A.G.; De Beer, T.R.M.; Sukhorukov, G.B.; Bracke, L.; Baeyens, W.R.G.; Demeester, J.; De Smedt, S.C. Stimuli-Responsive Multilayered Hybrid Nanoparticle/Polyelectrolyte Capsules. Macromol. Rapid Commun. 2007, 28, 88–95. [Google Scholar] [CrossRef]
- Jeyaraman, J.; Shukla, A.; Sivakumar, S. Targeted Stealth Polymer Capsules Encapsulating Ln3+-Doped LaVO4 Nanoparticles for Bioimaging Applications. ACS Biomater. Sci. Eng. 2016, 2, 1330–1340. [Google Scholar] [CrossRef]
- Wajs, E.; Nielsen, T.T.; Larsen, K.L.; Fragoso, A. Preparation of stimuli-responsive nano-sized capsules based on cyclodextrin polymers with redox or light switching properties. Nano Res. 2016, 9, 2070–2078. [Google Scholar] [CrossRef]
- Meaney, S.P.; Follink, B.; Tabor, R.F. Synthesis, Characterization, and Applications of Polymer–Silica Core–Shell Microparticle Capsules. ACS Appl. Mater. Interfaces 2018, 10, 43068–43079. [Google Scholar] [CrossRef]
- Li, F.; Feng, Y.; Wang, Z.; Yang, L.; Zhuo, L.; Tang, B. Direct electrochemistry of horseradish peroxidase immobilized on the layered calcium carbonate–gold nanoparticles inorganic hybrid composite. Biosens. Bioelectron. 2010, 25, 2244–2248. [Google Scholar] [CrossRef]
- De Geest, B.G.; Van Camp, W.; Du Prez, F.E.; De Smedt, S.C.; Demeester, J.; Hennink, W.E. Degradable multilayer films and hollow capsules via a “click” strategy. Macromol. Rapid Commun. 2008, 29, 1111–1118. [Google Scholar] [CrossRef]
- Wu, T.; Ge, Z.; Liu, S. Fabrication of Thermoresponsive Cross-Linked Poly(N-isopropylacrylamide) Nanocapsules and Silver Nanoparticle-Embedded Hybrid Capsules with Controlled Shell Thickness. Chem. Mater. 2011, 23, 2370–2380. [Google Scholar] [CrossRef]
- Such, G.K.; Quinn, J.F.; Quinn, A.; Tjipto, E.; Caruso, F. Assembly of ultrathin polymer multilayer films by click chemistry. J. Am. Chem. Soc. 2006, 128, 9318–9319. [Google Scholar] [CrossRef]
- Ochs, C.J.; Such, G.K.; Yan, Y.; van Koeverden, M.P.; Caruso, F. Biodegradable Click Capsules with Engineered Drug-Loaded Multilayers. ACS Nano 2010, 4, 1653–1663. [Google Scholar] [CrossRef] [PubMed]
- Such, G.K.; Tjipto, E.; Postma, A.; Johnston, A.P.R.; Caruso, F. Ultrathin, Responsive Polymer Click Capsules. Nano Lett. 2007, 7, 1706–1710. [Google Scholar] [CrossRef]
- Parakhonskiy, B.V.; Parak, W.J.; Volodkin, D.V.; Skirtach, A.G. Hybrids of Polymeric Capsules, Lipids, and Nanoparticles: Thermodynamics and Temperature Rise at the Nanoscale and Emerging Applications. Langmuir 2019, 35, 8574–8583. [Google Scholar] [CrossRef] [PubMed]
- Radt, B.; Smith, T.A.; Caruso, F. Optically Addressable Nanostructured Capsules. Adv. Mater. 2004, 16, 2184–2189. [Google Scholar] [CrossRef]
- Skirtach, A.G.; Muñoz Javier, A.; Kreft, O.; Köhler, K.; Piera Alberola, A.; Möhwald, H.; Parak, W.J.; Sukhorukov, G.B. Laser-Induced Release of Encapsulated Materials inside Living Cells. Angew. Chem. Int. Ed. 2006, 45, 4612–4617. [Google Scholar] [CrossRef]
- Brueckner, M.; Hollenbach-Latzko, S.; Reibetanz, U. Dual Transport of Active Substances with a Layer-by-Layer-Based Drug Delivery System to Terminate Inflammatory Processes. Macromol. Biosci. 2020, 20, 2000097. [Google Scholar] [CrossRef]
- Brkovic, N.; Zhang, L.; Peters, J.N.; Kleine-Doepke, S.; Parak, W.J.; Zhu, D. Quantitative Assessment of Endosomal Escape of Various Endocytosed Polymer-Encapsulated Molecular Cargos upon Photothermal Heating. Small 2020, 16, 2003639. [Google Scholar] [CrossRef]
- Borges, J.; Rodrigues, L.C.; Reis, R.L.; Mano, J.F. Layer-by-Layer Assembly of Light-Responsive Polymeric Multilayer Systems. Adv. Funct. Mater. 2014, 24, 5624–5648. [Google Scholar] [CrossRef]
- Zograf, G.P.; Timin, A.S.; Muslimov, A.R.; Shishkin, I.I.; Nominé, A.; Ghanbaja, J.; Ghosh, P.; Li, Q.; Zyuzin, M.V.; Makarov, S.V. All-Optical Nanoscale Heating and Thermometry with Resonant Dielectric Nanoparticles for Controllable Drug Release in Living Cells. Laser Photon. Rev. 2020, 14, 1900082. [Google Scholar] [CrossRef]
- Koryakina, I.; Kuznetsova, D.S.; Zuev, D.A.; Milichko, V.A.; Timin, A.S.; Zyuzin, M.V. Optically responsive delivery platforms: From the design considerations to biomedical applications. Nanophotonics 2020, 9, 39–74. [Google Scholar] [CrossRef]
- Lengert, E.; Parakhonskiy, B.; Khalenkow, D.; Zečić, A.; Vangheel, M.; Monje Moreno, J.M.; Braeckman, B.P.; Skirtach, A.G. Laser-induced remote release in vivo in C. elegans from novel silver nanoparticles-alginate hydrogel shells. Nanoscale 2018, 10, 17249–17256. [Google Scholar] [CrossRef] [PubMed]
- Neri, G.; Corsaro, C.; Fazio, E. Plasmon-Enhanced Controlled Drug Release from Ag-PMA Capsules. Molecules 2020, 25, 2267. [Google Scholar] [CrossRef] [PubMed]
- Timin, A.S.; Gao, H.; Voronin, D.V.; Gorin, D.A.; Sukhorukov, G.B. Inorganic/Organic Multilayer Capsule Composition for Improved Functionality and External Triggering. Adv. Mater. Interfaces 2017, 4, 1600338. [Google Scholar] [CrossRef]
- Parakhonskiy, B.V.; Bedard, M.F.; Bukreeva, T.V.; Sukhorukov, G.B.; Möhwald, H.; Skirtach, A.G. Nanoparticles on Polyelectrolytes at Low Concentration: Controlling Concentration and Size. J. Phys. Chem. C 2010, 114, 1996–2002. [Google Scholar] [CrossRef]
- Anandhakumar, S.; Raichur, A.M. A facile route to synthesize silver nanoparticles in polyelectrolyte capsules. Colloids Surf. B Biointerfaces 2011, 84, 379–383. [Google Scholar] [CrossRef]
- Bédard, M.F.; De Geest, B.G.; Möhwald, H.; Sukhorukov, G.B.; Skirtach, A.G. Direction specific release from giant microgel-templated polyelectrolyte microcontainers. Soft Matter 2009, 5, 3927–3931. [Google Scholar] [CrossRef]
- Kaufman, G.; Montejo, K.A.; Michaut, A.; Majewski, P.W.; Osuji, C.O. Photoresponsive and Magnetoresponsive Graphene Oxide Microcapsules Fabricated by Droplet Microfluidics. ACS Appl. Mater. Interfaces 2017, 9, 44192–44198. [Google Scholar] [CrossRef]
- Ermakov, A.V.; Verkhovskii, R.A.; Babushkina, I.V.; Trushina, D.B.; Inozemtseva, O.A.; Lukyanets, E.A.; Ulyanov, V.J.; Gorin, D.A.; Belyakov, S.; Antipina, M.N. In Vitro Bioeffects of Polyelectrolyte Multilayer Microcapsules Post-Loaded with Water-Soluble Cationic Photosensitizer. Pharmaceutics 2020, 12, 610. [Google Scholar] [CrossRef]
- Saito, H.; Kato, N. Polyelectrolyte/carbon nanotube composite microcapsules and drug release triggered by laser irradiation. Jpn. J. Appl. Phys. 2016, 55, 03DF06. [Google Scholar] [CrossRef]
- Szczęch, M.; Szczepanowicz, K. Polymeric Core-Shell Nanoparticles Prepared by Spontaneous Emulsification Solvent Evaporation and Functionalized by the Layer-by-Layer Method. Nanomaterials 2020, 10, 496. [Google Scholar] [CrossRef] [PubMed]
- Hong, C.-S.; Park, J.H.; Lee, S.; Rhoo, K.Y.; Lee, J.T.; Paik, S.R. Fabrication of Protease-Sensitive and Light-Responsive Microcapsules Encompassed with Single Layer of Gold Nanoparticles by Using Self-Assembly Protein of α-Synuclein. ACS Appl. Mater. Interfaces 2018, 10, 26628–26640. [Google Scholar] [CrossRef] [PubMed]
- Gao, H.; Wen, D.; Tarakina, N.V.; Liang, J.; Bushby, A.J.; Sukhorukov, G.B. Bifunctional ultraviolet/ultrasound responsive composite TiO2/polyelectrolyte microcapsules. Nanoscale 2016, 8, 5170–5180. [Google Scholar] [CrossRef] [PubMed]
- Novoselova, M.V.; Voronin, D.V.; Abakumova, T.O.; Demina, P.A.; Petrov, A.V.; Petrov, V.V.; Zatsepin, T.S.; Sukhorukov, G.B.; Gorin, D.A. Focused ultrasound-mediated fluorescence of composite microcapsules loaded with magnetite nanoparticles: In vitro and in vivo study. Colloids Surf. B Biointerfaces 2019, 181, 680–687. [Google Scholar] [CrossRef] [PubMed]
- Hitchcock, J.; White, A.L.; Hondow, N.; Hughes, T.A.; Dupont, H.; Biggs, S.; Cayre, O.J. Metal-shell nanocapsules for the delivery of cancer drugs. J. Colloid Interface Sci. 2020, 567, 171–180. [Google Scholar] [CrossRef]
- Stark, K.; Hitchcock, J.P.; Fiaz, A.; White, A.L.; Baxter, E.A.; Biggs, S.; McLaughlan, J.R.; Freear, S.; Cayre, O.J. Encapsulation of Emulsion Droplets with Metal Shells for Subsequent Remote, Triggered Release. ACS Appl. Mater. Interfaces 2019, 11, 12272–12282. [Google Scholar] [CrossRef]
- White, A.L.; Langton, C.; Wille, M.-L.; Hitchcock, J.; Cayre, O.J.; Biggs, S.; Blakey, I.; Whittaker, A.K.; Rose, S.; Puttick, S. Ultrasound-triggered release from metal shell microcapsules. J. Colloid Interface Sci. 2019, 554, 444–452. [Google Scholar] [CrossRef]
- Lengert, E.; Saveleva, M.; Abalymov, A.A.; Atkin, V.; Wuytens, P.C.; Kamyshinsky, R.; Vasiliev, A.L.; Gorin, D.A.; Sukhorukov, G.B.; Skirtach, A.G.; et al. Silver Alginate Hydrogel Micro- and Nanocontainers for Theranostics: Synthesis, Encapsulation, Remote Release, and Detection. ACS Appl. Mater. Interfaces 2017, 9, 21949–21958. [Google Scholar] [CrossRef]
- Inozemtseva, O.A.; Voronin, D.V.; Petrov, A.V.; Petrov, V.V.; Lapin, S.A.; Kozlova, A.A.; Bratashov, D.N.; Zakharevich, A.M.; Gorin, D.A. Disruption of Polymer and Composite Microcapsule Shells under High-Intensity Focused Ultrasound. Colloid J. 2018, 80, 771–782. [Google Scholar] [CrossRef]
- Stavarache, C.E.; Paniwnyk, L. Controlled rupture of magnetic LbL polyelectrolyte capsules and subsequent release of contents employing high intensity focused ultrasound. J. Drug Deliv. Sci. Technol. 2018, 45, 60–69. [Google Scholar] [CrossRef]
- Kubiak, T.; Banaszak, J.; Józefczak, A.; Rozynek, Z. Direction-Specific Release from Capsules with Homogeneous or Janus Shells Using an Ultrasound Approach. ACS Appl. Mater. Interfaces 2020, 12, 15810–15822. [Google Scholar] [CrossRef] [PubMed]
- Alford, A.; Tucker, B.; Kozlovskaya, V.; Chen, J.; Gupta, N.; Caviedes, R.; Gearhart, J.; Graves, D.; Kharlampieva, E. Encapsulation and Ultrasound-Triggered Release of G-Quadruplex DNA in Multilayer Hydrogel Microcapsules. Polymers 2018, 10, 1342. [Google Scholar] [CrossRef] [PubMed]
- Gao, H.; Sapelkin, A.V.; Titirici, M.M.; Sukhorukov, G.B. In Situ Synthesis of Fluorescent Carbon Dots/Polyelectrolyte Nanocomposite Microcapsules with Reduced Permeability and Ultrasound Sensitivity. ACS Nano 2016, 10, 9608–9615. [Google Scholar] [CrossRef] [PubMed]
- Lu, Z.; Prouty, M.D.; Guo, Z.; Golub, V.O.; Kumar, C.S.S.R.; Lvov, Y.M. Magnetic switch of permeability for polyelectrolyte microcapsules embedded with Co@Au nanoparticles. Langmuir 2005, 21, 2042–2050. [Google Scholar] [CrossRef]
- Read, J.E.; Luo, D.; Chowdhury, T.T.; Flower, R.J.; Poston, R.N.; Sukhorukov, G.B.; Gould, D.J. Magnetically responsive layer-by-layer microcapsules can be retained in cells and under flow conditions to promote local drug release without triggering ROS production. Nanoscale 2020, 12, 7735–7748. [Google Scholar] [CrossRef]
- Luo, D.; Poston, R.N.; Gould, D.J.; Sukhorukov, G.B. Magnetically targetable microcapsules display subtle changes in permeability and drug release in response to a biologically compatible low frequency alternating magnetic field. Mater. Sci. Eng. C 2019, 94, 647–655. [Google Scholar] [CrossRef]
- Prabu, C.; Latha, S.; Selvamani, P.; Ahrentorp, F.; Johansson, C.; Takeda, R.; Takemura, Y.; Ota, S. Layer-by-layer assembled magnetic prednisolone microcapsules (MPC) for controlled and targeted drug release at rheumatoid arthritic joints. J. Magn. Magn. Mater. 2017, 427, 258–267. [Google Scholar] [CrossRef]
- Carregal-Romero, S.; Guardia, P.; Yu, X.; Hartmann, R.; Pellegrino, T.; Parak, W.J. Magnetically triggered release of molecular cargo from iron oxide nanoparticle loaded microcapsules. Nanoscale 2015, 7, 570–576. [Google Scholar] [CrossRef]
- Cristofolini, L.; Szczepanowicz, K.; Orsi, D.; Rimoldi, T.; Albertini, F.; Warszynski, P. Hybrid Polyelectrolyte/Fe3O4 Nanocapsules for Hyperthermia Applications. ACS Appl. Mater. Interfaces 2016, 8, 25043–25050. [Google Scholar] [CrossRef]
- Katagiri, K.; Nakamura, M.; Koumoto, K. Magnetoresponsive Smart Capsules Formed with Polyelectrolytes, Lipid Bilayers and Magnetic Nanoparticles. ACS Appl. Mater. Interfaces 2010, 2, 768–773. [Google Scholar] [CrossRef]
- Minaeva, O.V.; Brodovskaya, E.P.; Pyataev, M.A.; Gerasimov, M.V.; Zharkov, M.N.; Yurlov, I.A.; Kulikov, O.A.; Kotlyarov, A.A.; Balykova, L.A.; Kokorev, A.V.; et al. Comparative study of cytotoxicity of ferromagnetic nanoparticles and magnetitecontaining polyelectrolyte microcapsules. J. Phys. Conf. Ser. 2017, 784, 012038. [Google Scholar] [CrossRef]
- Bezbaruah, A.N.; Shanbhogue, S.S.; Simsek, S.; Khan, E. Encapsulation of iron nanoparticles in alginate biopolymer for trichloroethylene remediation. J. Nanopart. Res. 2011, 13, 6673–6681. [Google Scholar] [CrossRef]
- Gao, C.; Leporatti, S.; Moya, S.; Donath, E.; Möhwald, H. Stability and Mechanical Properties of Polyelectrolyte Capsules Obtained by Stepwise Assembly of Poly(styrenesulfonate sodium salt) and Poly(diallyldimethyl ammonium) Chloride onto Melamine Resin Particles. Langmuir 2001, 17, 3491–3495. [Google Scholar] [CrossRef]
- Fery, A.; Weinkamer, R. Mechanical properties of micro- and nanocapsules: Single-capsule measurements. Polymer (Guildf) 2007, 48, 7221–7235. [Google Scholar] [CrossRef]
- She, S.; Li, Q.; Shan, B.; Tong, W.; Gao, C. Fabrication of Red-Blood-Cell-Like Polyelectrolyte Microcapsules and Their Deformation and Recovery Behavior Through a Microcapillary. Adv. Mater. 2013, 25, 5814–5818. [Google Scholar] [CrossRef]
- Kolesnikova, T.A.; Skirtach, A.G.; Möhwald, H. Red blood cells and polyelectrolyte multilayer capsules: Natural carriers versus polymer-based drug delivery vehicles. Expert Opin. Drug Deliv. 2013, 10, 47–58. [Google Scholar] [CrossRef]
- Anselmo, A.C.; Mitragotri, S. Impact of particle elasticity on particle-based drug delivery systems. Adv. Drug Deliv. Rev. 2017, 108, 51–67. [Google Scholar] [CrossRef]
- Dubreuil, F.; Elsner, N.; Fery, A. Elastic properties of polyelectrolyte capsules studied by atomic-force microscopy and RICM. Eur. Phys. J. E Soft Matter 2003, 12, 215–221. [Google Scholar] [CrossRef]
- Pan, H.M.; Seuss, M.; Neubauer, M.P.; Trau, D.W.; Fery, A. Tuning the Mechanical Properties of Hydrogel Core–Shell Particles by Inwards Interweaving Self-Assembly. ACS Appl. Mater. Interfaces 2016, 8, 1493–1500. [Google Scholar] [CrossRef]
- Frueh, J.; Rühm, A.; He, Q.; Möhwald, H.; Krastev, R.; Köhler, R. Elastic to Plastic Deformation in Uniaxially Stressed Polylelectrolyte Multilayer Films. Langmuir 2018, 34, 11933–11942. [Google Scholar] [CrossRef]
- Hegemann, J.; Knoche, S.; Egger, S.; Kott, M.; Demand, S.; Unverfehrt, A.; Rehage, H.; Kierfeld, J. Pendant capsule elastometry. J. Colloid Interface Sci. 2018, 513, 549–565. [Google Scholar] [CrossRef] [PubMed]
- Köhler, K.; Möhwald, H.; Sukhorukov, G.B. Thermal behavior of polyelectrolyte multilayer microcapsules: 2. Insight into molecular mechanisms for the PDADMAC/PSS system. J. Phys. Chem. B 2006, 110, 24002–24010. [Google Scholar] [CrossRef] [PubMed]
- Cramer, A.D.; Dong, W.-F.; Benbow, N.L.; Webber, J.L.; Krasowska, M.; Beattie, D.A.; Ferri, J.K. The influence of polyanion molecular weight on polyelectrolyte multilayers at surfaces: Elasticity and susceptibility to saloplasticity of strongly dissociated synthetic polymers at fluid–fluid interfaces. Phys. Chem. Chem. Phys. 2017, 19, 23781–23789. [Google Scholar] [CrossRef] [PubMed]
- Giménez, R.E.; Serrano, M.P.; Álvarez, R.M.S.; Martino, D.M.; Borsarelli, C.D. Fabrication and Characterization of Hollow Microcapsules from Polyelectrolytes Bearing Thymine Pendant Groups for Ultraviolet-B (UVB)-Induced Crosslinking. ChemPlusChem 2019, 84, 504–511. [Google Scholar] [CrossRef] [PubMed]
- Hastings, D.E.; Stöver, H.D.H. Crosslinked Hydrogel Capsules for Cell Encapsulation Formed Using Amino/Betaine Dual-Functional Semibatch Copolymers. ACS Appl. Polym. Mater. 2019, 1, 2055–2067. [Google Scholar] [CrossRef]
- Wu, Q.-X.; Xu, X.; Wang, Z.-L.; Yao, S.-J.; Tong, W.-Y.; Chen, Y. Effect of the cross-linking agent on performances of NaCS-CS/WSC microcapsules. Colloids Surf. B Biointerfaces 2016, 147, 416–421. [Google Scholar] [CrossRef]
- Gupta, N.; Kozlovskaya, V.; Dolmat, M.; Kharlampieva, E. Shape Recovery of Spherical Hydrogen-Bonded Multilayer Capsules after Osmotically Induced Deformation. Langmuir 2019, 35, 10910–10919. [Google Scholar] [CrossRef]
- Mueller, R.; Köhler, K.; Weinkamer, R.; Sukhorukov, G.B.; Fery, A. Melting of PDADMAC/PSS capsules investigated with AFM force spectroscopy. Macromolecules 2005, 38, 9766–9771. [Google Scholar] [CrossRef]
- Van der Meeren, L.; Li, J.; Konrad, M.; Skirtach, A.G.; Volodkin, D.; Parakhonskiy, B.V. Temperature Window for Encapsulation of an Enzyme into Thermally Shrunk, CaCO3 Templated Polyelectrolyte Multilayer Capsules. Macromol. Biosci. 2020, 20, 2000081. [Google Scholar] [CrossRef]
- Mettu, S.; Zhou, M.; Tardy, B.L.; Ashokkumar, M.; Dagastine, R.R. Temperature dependent mechanical properties of air, oil and water filled microcapsules studied by atomic force microscopy. Polymer (Guildf) 2016, 102, 333–341. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, F.; Valenzuela, L.D.; Sammalkorpi, M.; Lutkenhaus, J.L. Effect of Water on the Thermal Transition Observed in Poly(allylamine hydrochloride)–Poly(acrylic acid) Complexes. Macromolecules 2016, 49, 7563–7570. [Google Scholar] [CrossRef]
- Bédard, M.F.; Munoz-Javier, A.; Mueller, R.; del Pino, P.; Fery, A.; Parak, W.J.; Skirtach, A.G.; Sukhorukov, G.B. On the mechanical stability of polymeric microcontainers functionalized with nanoparticles. Soft Matter 2009, 5, 148–155. [Google Scholar] [CrossRef]
- Yashchenok, A.M.; Bratashov, D.N.; Gorin, D.a.; Lomova, M.V.; Pavlov, A.M.; Sapelkin, A.V.; Shim, B.S.; Khomutov, G.B.; Kotov, N.A.; Sukhorukov, G.B.; et al. Carbon Nanotubes on Polymeric Microcapsules: Free-Standing Structures and Point-Wise Laser Openings. Adv. Funct. Mater. 2010, 20, 3136–3142. [Google Scholar] [CrossRef]
- Del Mercato, L.L.; Guerra, F.; Lazzari, G.; Nobile, C.; Bucci, C.; Rinaldi, R. Biocompatible multilayer capsules engineered with a graphene oxide derivative: Synthesis, characterization and cellular uptake. Nanoscale 2016, 8, 7501–7512. [Google Scholar] [CrossRef] [PubMed]
- Kaufman, G.; Mukhopadhyay, S.; Rokhlenko, Y.; Nejati, S.; Boltyanskiy, R.; Choo, Y.; Loewenberg, M.; Osuji, C.O. Highly stiff yet elastic microcapsules incorporating cellulose nanofibrils. Soft Matter 2017, 13, 2733–2737. [Google Scholar] [CrossRef] [PubMed]
- Sciortino, F.; Thivolle, M.; Kahn, M.L.; Gaillard, C.; Chevance, S.; Gauffre, F. Structure and elasticity of composite nanoparticle/polymer nanoshells (hybridosomes). Soft Matter 2017, 13, 4393–4400. [Google Scholar] [CrossRef] [PubMed]
- Chojnacka-Górka, K.; Rozpędzik, A.; Zapotoczny, S. Robust polyelectrolyte microcapsules reinforced with carbon nanotubes. RSC Adv. 2016, 6, 114639–114643. [Google Scholar] [CrossRef]
- Ermakov, A.V.; Prikhozhdenko, E.S.; Demina, P.A.; Gorbachev, I.A.; Vostrikova, A.M.; Sapelkin, A.V.; Goryacheva, I.Y.; Sukhorukov, G.B. Composite multilayer films based on polyelectrolytes and in situ-formed carbon nanostructures with enhanced photoluminescence and conductivity properties. J. Appl. Polym. Sci. 2019, 136, 47718. [Google Scholar] [CrossRef]
- Zhao, S.; Caruso, F.; Dähne, L.; Decher, G.; De Geest, B.G.; Fan, J.; Feliu, N.; Gogotsi, Y.; Hammond, P.T.; Hersam, M.C.; et al. The Future of Layer-by-Layer Assembly: A Tribute to ACS Nano Associate Editor Helmuth Möhwald. ACS Nano 2019, 13, 6151–6169. [Google Scholar] [CrossRef]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lengert, E.V.; Koltsov, S.I.; Li, J.; Ermakov, A.V.; Parakhonskiy, B.V.; Skorb, E.V.; Skirtach, A.G. Nanoparticles in Polyelectrolyte Multilayer Layer-by-Layer (LbL) Films and Capsules—Key Enabling Components of Hybrid Coatings. Coatings 2020, 10, 1131. https://doi.org/10.3390/coatings10111131
Lengert EV, Koltsov SI, Li J, Ermakov AV, Parakhonskiy BV, Skorb EV, Skirtach AG. Nanoparticles in Polyelectrolyte Multilayer Layer-by-Layer (LbL) Films and Capsules—Key Enabling Components of Hybrid Coatings. Coatings. 2020; 10(11):1131. https://doi.org/10.3390/coatings10111131
Chicago/Turabian StyleLengert, Ekaterina V., Semyon I. Koltsov, Jie Li, Alexey V. Ermakov, Bogdan V. Parakhonskiy, Ekaterina V. Skorb, and Andre G. Skirtach. 2020. "Nanoparticles in Polyelectrolyte Multilayer Layer-by-Layer (LbL) Films and Capsules—Key Enabling Components of Hybrid Coatings" Coatings 10, no. 11: 1131. https://doi.org/10.3390/coatings10111131
APA StyleLengert, E. V., Koltsov, S. I., Li, J., Ermakov, A. V., Parakhonskiy, B. V., Skorb, E. V., & Skirtach, A. G. (2020). Nanoparticles in Polyelectrolyte Multilayer Layer-by-Layer (LbL) Films and Capsules—Key Enabling Components of Hybrid Coatings. Coatings, 10(11), 1131. https://doi.org/10.3390/coatings10111131






