MAPLE Deposition of Binary and Ternary Organic Bulk Heterojunctions Based on Zinc Phthalocyanine
Abstract
1. Introduction
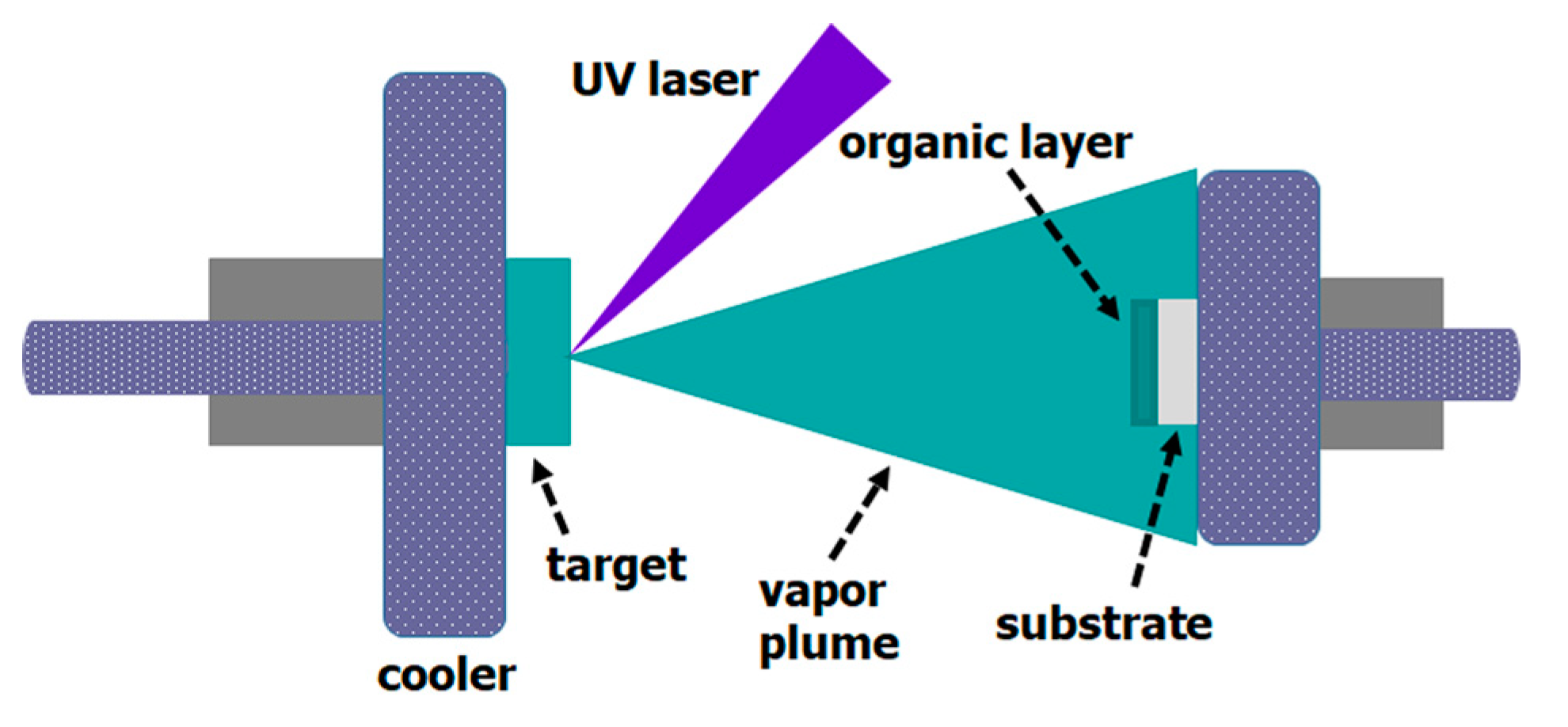
2. Experimental
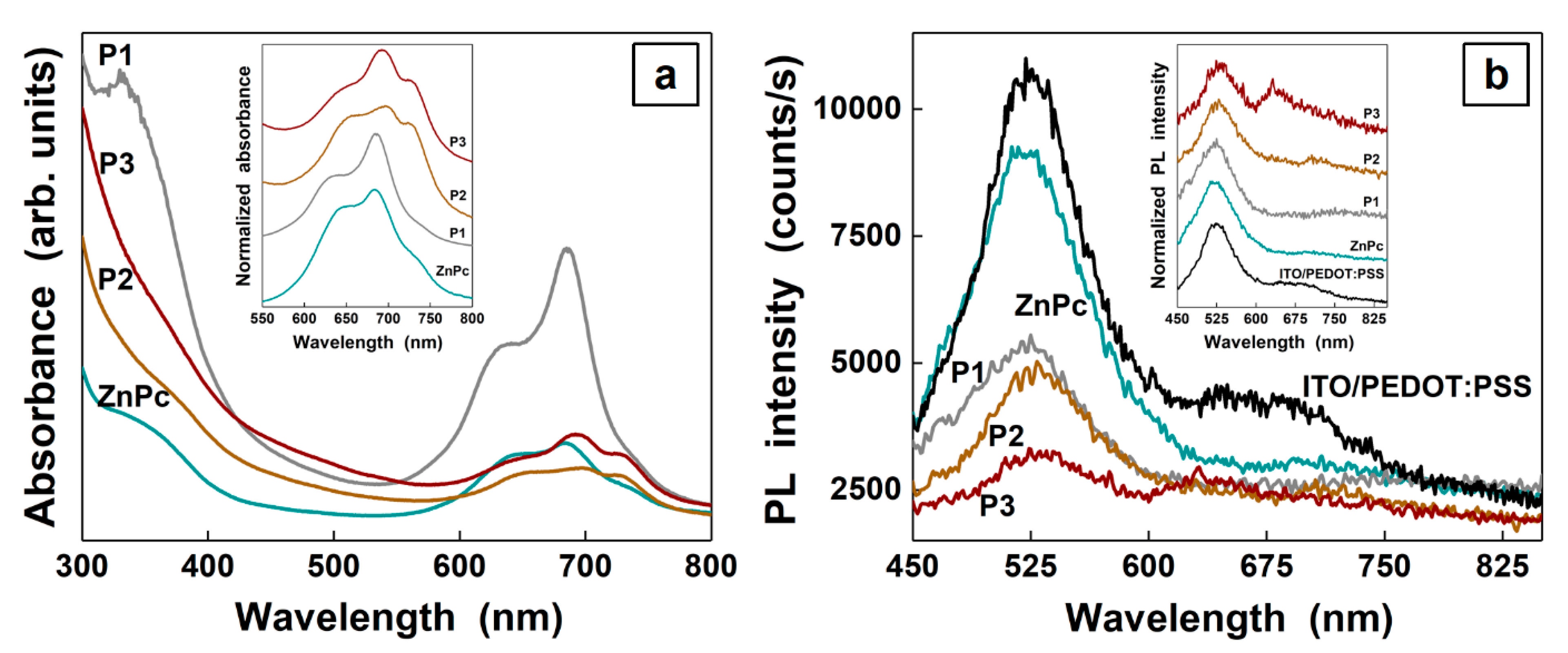
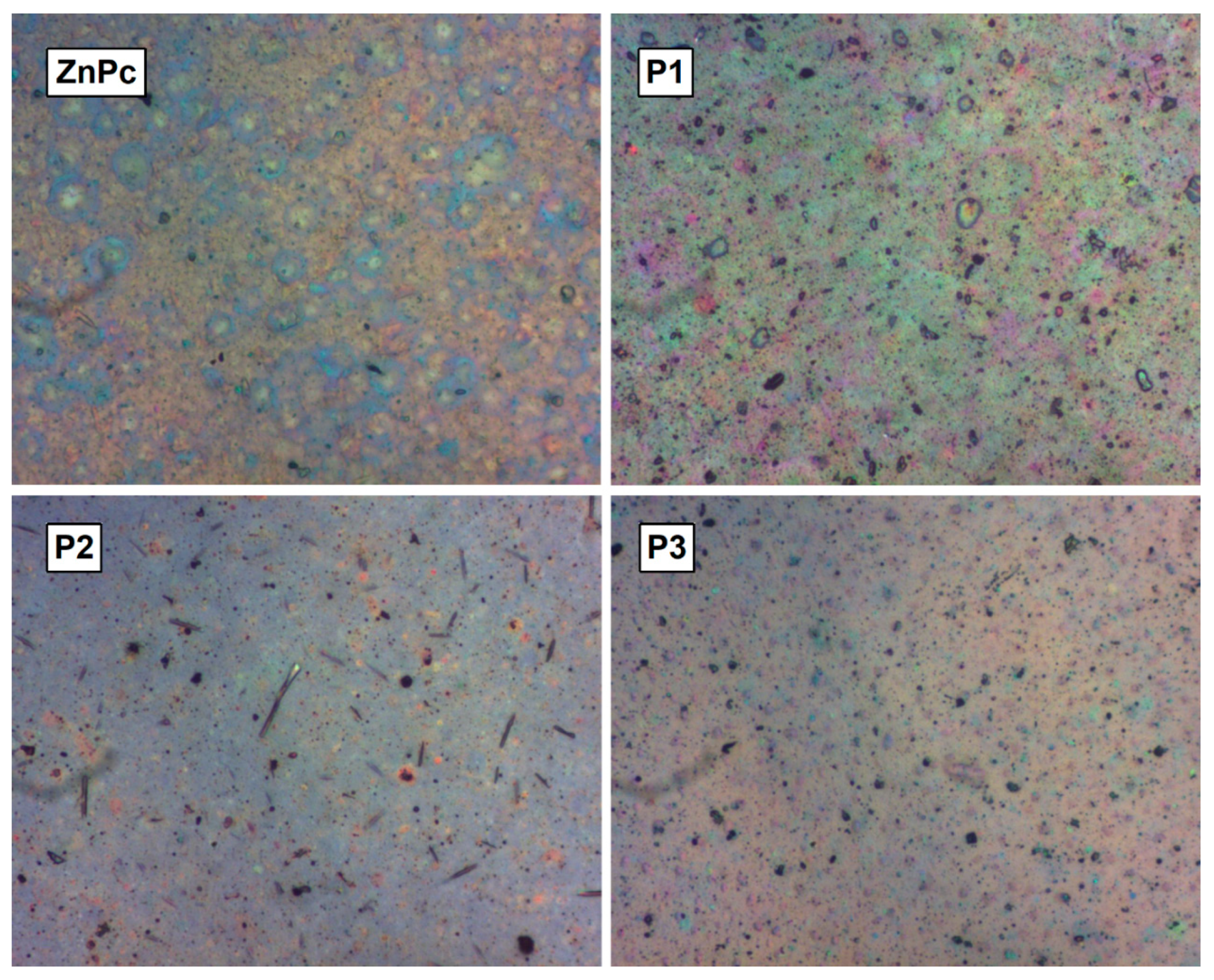
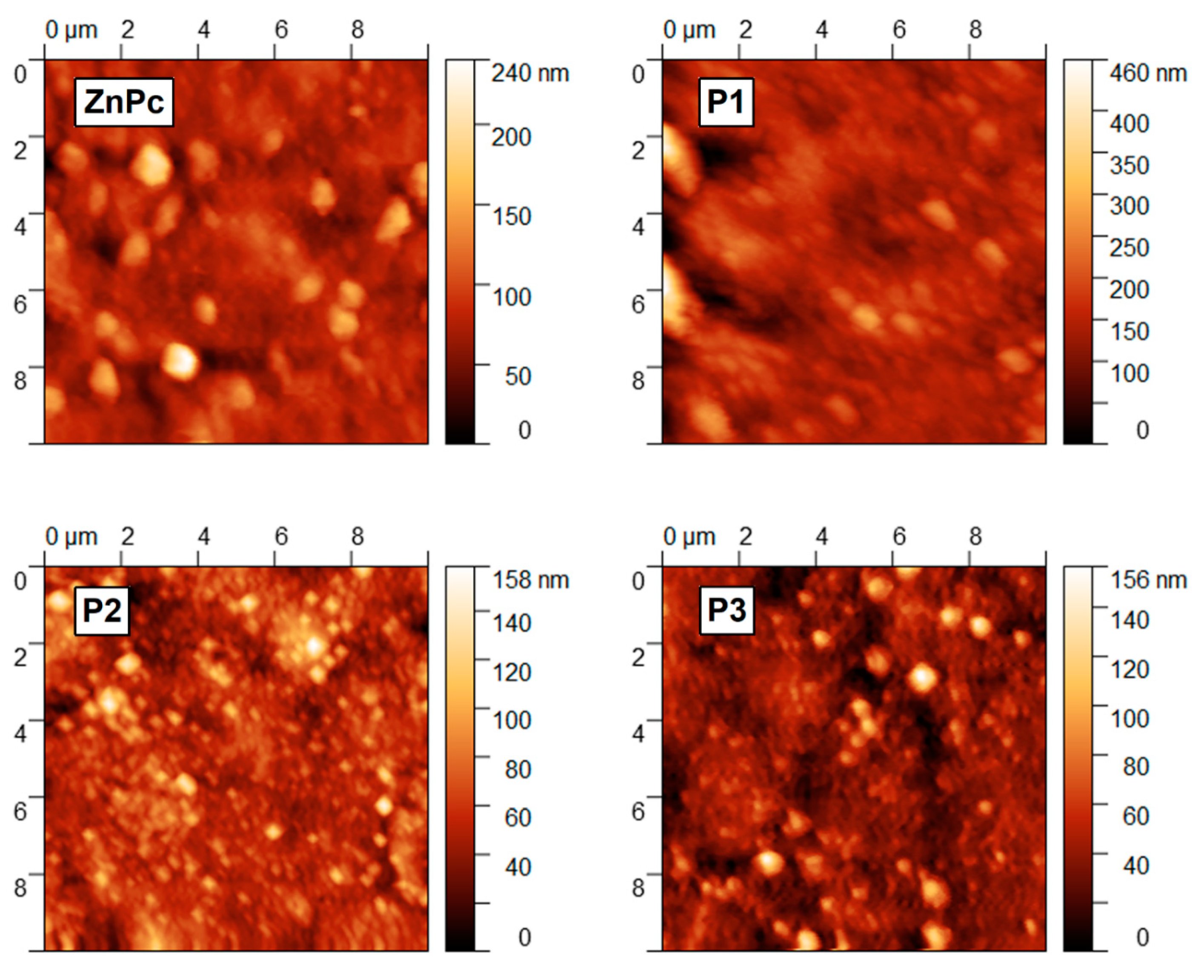
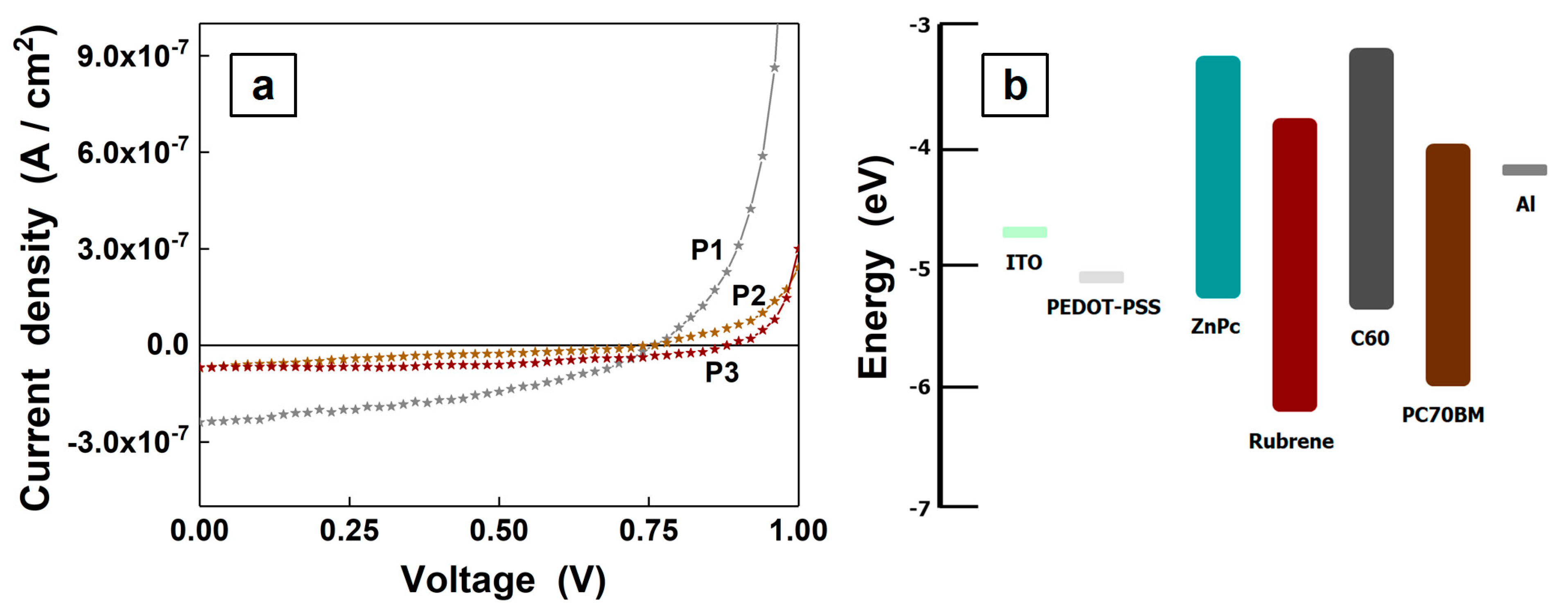
3. Results and Discussions
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Engmann, S.; Barito, A.J.; Bittle, E.G.; Giebink, N.C.; Richter, L.J.; Gundlach, D.J. Higher order effects in organic LEDs with sub-bandgap turn-on. Nat. Commun. 2019, 10, 227. [Google Scholar] [CrossRef] [PubMed]
- Jazbinsek, M.; Puc, U.; Abina, A.; Zidansek, A. Organic crystals for THz photonics. Appl. Sci. 2019, 9, 882. [Google Scholar] [CrossRef]
- Stanculescu, A.; Stanculescu, F.; Alexandru, H.V.; Socol, M. Doped aromatic derivatives wide-gap crystalline semiconductor structured layers for electronic applications. Thin Solid Films 2006, 495, 389–393. [Google Scholar] [CrossRef]
- Gusain, A.; Faria, R.M.; Miranda, P.B. Polymer solar cells—Interfacial processes related to performance issues. Front. Chem. 2019, 7, 61. [Google Scholar] [CrossRef] [PubMed]
- Chang, J.; Sonar, P.; Lin, Z.; Zhang, C.; Zhang, J.; Hao, Y.; Wu, J. Controlling aggregation and crystallization of solution processed diketopyrrolopyrrole based polymer for high performance thin film transistors by pre-metered slot die coating process. Org. Electron. 2016, 36, 113–119. [Google Scholar] [CrossRef]
- Liu, Q.; Jiang, Y.; Jin, K.; Qin, J.; Xu, J.; Li, W.; Xiong, J.; Liu, J.; Xiao, Z.; Sun, K.; et al. 18% Efficiency organic solar cells. Sci. Bull. 2020, 65, 272–275. [Google Scholar] [CrossRef]
- Kearns, D.; Calvin, M. Photovoltaic effect and photoconductivity in laminated organic systems. J. Chem. Phys. 1958, 29, 950–951. [Google Scholar] [CrossRef]
- Tang, C.W. Two-layer organic photovoltaic cell. Appl. Phys. Lett. 1986, 48, 183–185. [Google Scholar] [CrossRef]
- Yu, G.; Gao, J.; Hummelen, J.C.; Wudl, F.; Heeger, A.J. Polymer photovoltaic cells: Enhanced efficiencies via a network of internal donor-acceptor heterojunctions. Science 1995, 270, 1789–1791. [Google Scholar] [CrossRef]
- Islam, Z.U.; Tahir, M.; Syed, W.A.; Aziz, F.; Wahab, F.; Said, S.M.; Sarker, R.M.; Md Ali, S.H.; Sabri, M.F.M. Fabrication and photovoltaic properties of organic solar cell based on zinc phthalocyanine. Energies 2020, 13, 962. [Google Scholar] [CrossRef]
- Jiang, H.; Hu, P.; Ye, J.; Ganguly, R.; Li, Y.; Long, Y.; Fichou, D.; Hu, W.; Kloc, C. Hole mobility modulation in single–crystal metal phthalocyanines by changing the metal–π/π–π interactions. Angew. Chem. Int. Ed. 2018, 57, 10112–10117. [Google Scholar] [CrossRef] [PubMed]
- Stanculescu, F.; Rasoga, O.; Catargiu, A.M.; Vacareanu, L.; Socol, M.; Breazu, C.; Preda, N.; Socol, G.; Stanculescu, A. MAPLE prepared heterostructures with arylene based polymer active layer for photovoltaic applications. Appl. Surf. Sci. 2015, 336, 240–248. [Google Scholar] [CrossRef]
- Armin, A.; Hambsch, M.; Wolfer, P.; Jin, H.; Li, J.; Shi, Z.; Burn, P.L.; Meredith, P. Efficient, large area, and thick junction polymer solar cells with balanced mobilities and low defect densities. Adv. Energy Mater. 2014, 5, 1401221. [Google Scholar] [CrossRef]
- Armin, A.; Wolfer, P.; Shaw, P.E.; Hambsch, M.; Maasoumi, F.; Ullah, M.; Gann, E.; McNeill, C.R.; Li, J.; Shi, Z.; et al. Simultaneous enhancement of charge generation quantum yield and carrier transport in organic solar cells. J. Mater. Chem. C 2015, 41, 10799–10812. [Google Scholar] [CrossRef]
- Zhang, F.; Zhuo, Z.; Zhang, J.; Wang, X.; Xu, X.; Wang, Z.; Xin, Y.; Wang, J.; Wang, J.; Tang, W.; et al. Influence of PC60BM or PC70BM as electron acceptor on the performance of polymer solar cells. Sol. Energy Mater. Sol. Cells. 2012, 97, 71–77. [Google Scholar] [CrossRef]
- Pandey, A. Highly efficient spin-conversion effect leading to energy up-converted electroluminescence in singlet fission photovoltaics. Sci. Rep. 2015, 5, 7787. [Google Scholar] [CrossRef]
- Hasegawa, T.; Takeya, J. Organic field-effect transistors using single crystals. Sci. Technol. Adv. Mater. 2009, 10, 024314. [Google Scholar] [CrossRef]
- Dayneko, S.V.; Hendsbee, A.D.; Cann, J.R.; Cabanetos, C.; Welch, G.C. Ternary organic solar cells: Using molecular donor or acceptor third components to increase open circuit voltage. New J. Chem. 2019, 43, 10442–10448. [Google Scholar] [CrossRef]
- Gasparini, N.; Salleo, A.; McCulloch, I.; Baran, D. The role of the third component in ternary organic solar cells. Nat. Rev. Mater. 2019, 4, 229–242. [Google Scholar] [CrossRef]
- Caricato, A.P.; Cesaria, M.; Gigli, G.; Loiudice, A.; Luches, A.; Martino, M.; Resta, V.; Rizzo, A.; Taurino, A. Poly-(3-hexylthiophene)/[6,6]-phenyl-C61-butyric-acid-methyl-ester bilayer deposition by matrix-assisted pulsed laser evaporation for organic photovoltaic applications. Appl. Phys. Lett. 2012, 100, 073306. [Google Scholar] [CrossRef]
- Socol, M.; Preda, N.; Rasoga, O.; Breazu, C.; Stavarache, I.; Stanculescu, F.; Socol, G.; Gherendi, F.; Grumezescu, V.; Stefan, N.; et al. Flexible heterostructures based on metal phthalocyanines thin films obtained by MAPLE. Appl. Surf. Sci. 2016, 374, 403–410. [Google Scholar] [CrossRef]
- Ionita, I.; Bercea, A.; Brajnicov, S.; Matei, A.; Ion, V.; Marascu, V.; Mitu, B.; Constantinescu, C. Second harmonic generation (SHG) in pentacene thin films grown by matrix assisted pulsed laser evaporation (MAPLE). Appl. Surf. Sci. 2019, 480, 212–218. [Google Scholar] [CrossRef]
- Stanculescu, A.; Rasoga, O.; Mihut, L.; Socol, M.; Stanculescu, F.; Ionita, I.; Albu, A.-M.; Socol, G. Preparation and characterization of polar aniline functionalized copolymers thin films for optical non-linear applications. Ferroelectrics 2009, 389, 159–173. [Google Scholar] [CrossRef]
- Schunemann, C.; Wynands, D.; Wilde, L.; Hein, M.P.; Pfutzner, S.; Elschner, C.; Eichhorn, K.-J.; Leo, K.; Riede, M. Phase separation analysis of bulk heterojunctions in small-molecule organic solar cells using zinc-phthalocyanine and C60. Phys. Rev. B 2012, 85, 245314. [Google Scholar] [CrossRef]
- Pfuetzner, S.; Meiss, J.; Petrich, A.; Riede, M.; Leo, K. Thick C60: ZnPc bulk heterojunction solar cells with improved performance by film deposition on heated substrates. Appl. Phys. Lett. 2009, 94, 253303. [Google Scholar] [CrossRef]
- Sanculescu, F.; Stanculescu, A.; Socol, M. Effect of the metallic contact on the electrical properties of organic semiconductor film. J. Optoelectron. Adv. Mater. 2007, 9, 1352–1357. [Google Scholar]
- Gaffo, L.; Cordeiro, M.R.; Freitas, A.R.; Moreira, W.C.; Girotto, E.M.; Zucolotto, V. The effects of temperature on the molecular orientation of zinc phthalocyanine films. J. Mater. Sci. 2010, 45, 1366–1370. [Google Scholar] [CrossRef]
- Pu, J.; Mo, Y.; Wan, S.; Wang, L. Fabrication of novel graphene–fullerene hybrid lubricating films based on self-assembly for MEMS applications. Chem. Commun. 2014, 50, 469–471. [Google Scholar] [CrossRef]
- Blazinic, V.; Ericsson, L.K.; Muntean, S.A.; Moons, E. Photo-degradation in air of spin-coated PC60BM and PC70BM films. Synth. Met. 2018, 241, 26–30. [Google Scholar] [CrossRef]
- Gavrilko, T.; Nechytaylo, V.; Viduta, L.; Baran, J. Optical properties and stability of bilayer rubrene-Alq3 films fabricated by vacuum deposition. Ukr. J. Phys. 2018, 63, 362. [Google Scholar] [CrossRef]
- Zanfolim, A.A.; Volpati, D.; Olivati, C.A.; Job, A.E.; Constantino, C.J.L. Structural and electric-optical properties of zinc phthalocyanine evaporated thin films: Temperature and thickness effects. J. Phys. Chem. C 2010, 114, 12290–12299. [Google Scholar] [CrossRef]
- Viterisi, A.; Montcada, N.F.; Kumar, C.V.; Guirado, F.G.; Martin, E.; Escuderoa, E.; Palomares, E. Unambiguous determination of molecular packing in crystalline donor domains of small molecule solution processed solar cell devices using routine X-ray diffraction techniques. J. Mater. Chem. A 2014, 2, 3536–3542. [Google Scholar] [CrossRef]
- Ahn, H.; Chu, T.-C. Annealing-induced phase transition in zinc phthalocyanine ultrathin films. Opt. Mater. Express 2016, 6, 3587. [Google Scholar] [CrossRef]
- Peng, Y.; Zhang, L.; Chengand, N.; Andrew, T. ITO-free transparent organic solar cell with distributed bragg reflector for solar harvesting windows. Energies 2017, 10, 707. [Google Scholar] [CrossRef]
- Hou, J.; Guo, X. Active layer materials for organic solar cells. In Organic Solar Cells. Green Energy and Technology, 1st ed.; Choy, W., Ed.; Springer: London, UK, 2013; pp. 17–42. [Google Scholar]
- Zhang, X.; Wu, Z.; Jiao, B.; Wang, D.; Wang, D.; Hou, X.; Huang, W. Solution-processed white organic light-emitting diodes with mixed-host structures. J. Lumin. 2012, 132, 697–701. [Google Scholar] [CrossRef]
- Zafar, Q.; Fatima, N.; Karimov, K.S.; Ahmed, M.M.; Sulaiman, K. Realizing broad-bandwidth visible wavelength photodiode based on solution-processed ZnPc/PC71BM dyad. Opt. Mater. 2017, 64, 131–136. [Google Scholar] [CrossRef]
- Elistratova, M.A.; Zakharova, I.B.; Romanov, N.M. Obtaining and investigation of C60 <A2B6> semiconductor compounds with a view to create effective solar cells. J. Phys. Conf. Ser. 2015, 661, 012030. [Google Scholar] [CrossRef]
- Griffin, J.; Pearson, A.J.; Scarratt, N.W.; Wang, T.; Dunbar, A.D.F.; Yi, H.; Iraqi, A.; Buckley, A.R.; Lidzey, D.G. Organic photovoltaic devices with enhanced efficiency processed from non-halogenated binary solvent blends. Org. Electron. 2015, 21, 216–222. [Google Scholar] [CrossRef]
- Liang, C.; Jin-Xiang, D.; Le, K.; Min, C.; Ren-Gang, C.; Zi-Jia, Z. Optical properties of rubrene thin film prepared by thermal evaporation. Chin. Phys. B 2015, 24, 047801. [Google Scholar]
- Klyamer, D.D.; Sukhikh, A.S.; Gromilov, S.A.; Kruchinin, V.N.; Spesivtsev, E.V.; Hassan, A.K.; Basova, T.V. Influence of fluorosubstitution on the structure of zinc phthalocyanine thin films. Macroheterocycles 2018, 11, 304–311. [Google Scholar] [CrossRef]
- Lassiter, B.E.; Wei, G.; Wang, S.; Zimmerman, J.D.; Diev, V.V.; Thompson, M.E.; Forrest, S.R. Organic photovoltaics incorporating electron conducting exciton blocking layers. Appl. Phys. Lett. 2011, 98, 243307. [Google Scholar] [CrossRef]
- Singh, R.; Suranagi, S.R.; Lee, J.; Lee, H.; Kim, M.; Cho, K. Unravelling the efficiency-limiting morphological issues of the perylene diimide-based nonfullerene organic solar cells. Sci. Rep. 2018, 8, 2849. [Google Scholar] [CrossRef] [PubMed]
- Ge, W.; Li, N.K.; McCormick, R.D.; Lichtenberg, E.; Yingling, Y.G.; Stiff-Robers, A.D. Emulsion-based RIR-MAPLE deposition of conjugated polymers: Primary solvent effect and its implications on organic solar cell performance. ACS Appl. Mater. Interfaces 2016, 8, 19494–19506. [Google Scholar] [CrossRef] [PubMed]


| Sample | Composition | Ratio | Number of the Laser Pulses |
|---|---|---|---|
| P0 | ZnPc | 1 | 50 K |
| P1 | ZnPc:C60 | 1:1 | 100 K |
| P2 | ZnPc:PC70BM | 1:1 | 100 K |
| P3 | ZnPc:rubrene:PC70BM | 1:1:1 | 100 K |
| Sample | VOC [V] | JSC [A] | Pmax [W] |
|---|---|---|---|
| P1 | 0.76 | 2.4 × 10−7 | 5.6 × 10−8 |
| P2 | 0.74 | 6.9 × 10−8 | 1.2 × 10−8 |
| P3 | 0.88 | 6.9 × 10−8 | 3.1 × 10−8 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Socol, M.; Preda, N.; Petre, G.; Costas, A.; Rasoga, O.; Popescu-Pelin, G.; Mihailescu, A.; Stanculescu, A.; Socol, G. MAPLE Deposition of Binary and Ternary Organic Bulk Heterojunctions Based on Zinc Phthalocyanine. Coatings 2020, 10, 956. https://doi.org/10.3390/coatings10100956
Socol M, Preda N, Petre G, Costas A, Rasoga O, Popescu-Pelin G, Mihailescu A, Stanculescu A, Socol G. MAPLE Deposition of Binary and Ternary Organic Bulk Heterojunctions Based on Zinc Phthalocyanine. Coatings. 2020; 10(10):956. https://doi.org/10.3390/coatings10100956
Chicago/Turabian StyleSocol, Marcela, Nicoleta Preda, Gabriela Petre, Andreea Costas, Oana Rasoga, Gianina Popescu-Pelin, Andreea Mihailescu, Anca Stanculescu, and Gabriel Socol. 2020. "MAPLE Deposition of Binary and Ternary Organic Bulk Heterojunctions Based on Zinc Phthalocyanine" Coatings 10, no. 10: 956. https://doi.org/10.3390/coatings10100956
APA StyleSocol, M., Preda, N., Petre, G., Costas, A., Rasoga, O., Popescu-Pelin, G., Mihailescu, A., Stanculescu, A., & Socol, G. (2020). MAPLE Deposition of Binary and Ternary Organic Bulk Heterojunctions Based on Zinc Phthalocyanine. Coatings, 10(10), 956. https://doi.org/10.3390/coatings10100956







