Antibiogram, Prevalence of OXA Carbapenemase Encoding Genes, and RAPD-Genotyping of Multidrug-Resistant Acinetobacter baumannii Incriminated in Hidden Community-Acquired Infections
Abstract
:1. Introduction
2. Results
2.1. Identification and Prevalence of the Hidden Community-Acquired A. baumannii
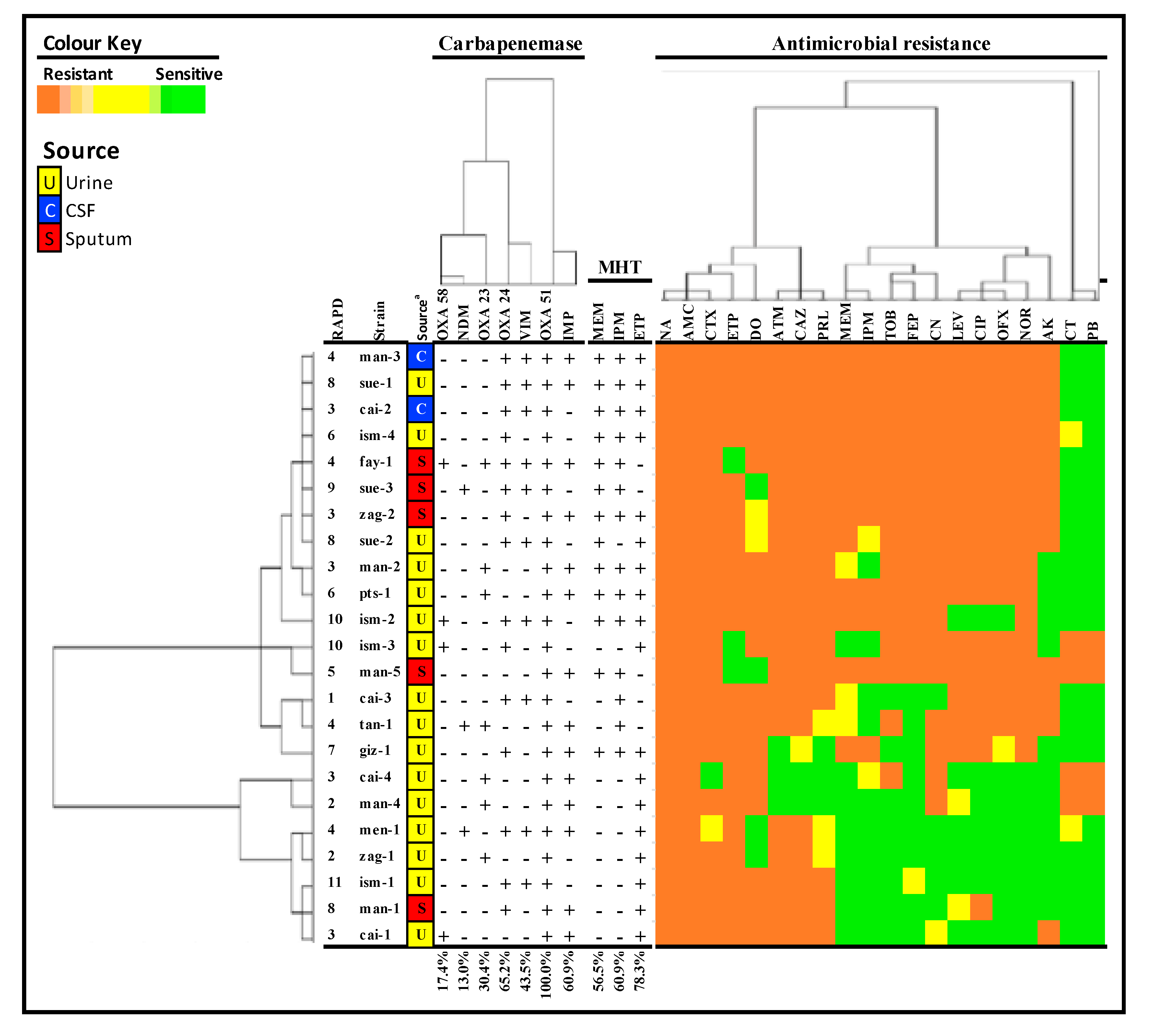
2.2. Antimicrobial Susceptibility Profiles of A. baumannii
2.3. Prevalence of Carbapenemase Producers
2.4. Prevalence of Carbapenemase Encoding Genes
2.5. Random Amplification of Polymorphic DNA (RAPD) PCR Fingerprinting
3. Discussion
4. Materials and Methods
4.1. Ethical Approval
4.2. Study Design and Bacterial Isolates
4.3. Molecular Identification of A. baumannii
4.4. Antimicrobial Susceptibility Testing of A. baumannii
4.5. Screening of Carbapenemases Production
4.6. Molecular Detection of Carbapenemase Encoding Genes
4.7. RAPD—Genotyping of A. baumannii
4.8. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- World Health Organization. WHO Publishes List of Bacteria for Which New Antibiotics Are Urgently Needed. Available online: https://www.who.int/en/news-room/detail/27-02-2017-who-publishes-list-of-bacteria-for-which-new-antibiotics-are-urgently-needed (accessed on 20 August 2019).
- Peleg, A.Y.; de Breij, A.; Adams, M.D.; Cerqueira, G.M.; Mocali, S.; Galardini, M.; Nibbering, P.H.; Earl, A.M.; Ward, D.V.; Paterson, D.L.; et al. The success of acinetobacter species; genetic, metabolic and virulence attributes. PLoS ONE 2012, 7, e46984. [Google Scholar] [CrossRef] [PubMed]
- Joly-Guillou, M.L. Clinical impact and pathogenicity of acinetobacter. Clin. Microbiol. Infect. 2005, 11, 868–873. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Porter, K.A.; Rhodes, J.; Dejsirilert, S.; Henchaichon, S.; Siludjai, D.; Thamthitiwat, S.; Prapasiri, P.; Jorakate, P.; Kaewpan, A.; Peruski, L.F.; et al. Acinetobacter bacteraemia in Thailand: Evidence for infections outside the hospital setting. Epidemiol. Infect. 2014, 142, 1317–1327. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dexter, C.; Murray, G.L.; Paulsen, I.T.; Peleg, A.Y. Community-acquired acinetobacter baumannii: Clinical characteristics, epidemiology and pathogenesis. Expert Rev. Anti-Infect. Ther. 2015, 13, 567–573. [Google Scholar] [CrossRef] [PubMed]
- De Vos, D.; Pirnay, J.-P.; Bilocq, F.; Jennes, S.; Verbeken, G.; Rose, T.; Keersebilck, E.; Bosmans, P.; Pieters, T.; Hing, M.; et al. Molecular epidemiology and clinical impact of acinetobacter calcoaceticus-baumannii complex in a Belgian burn wound center. PLoS ONE 2016, 11, e0156237. [Google Scholar] [CrossRef] [PubMed]
- Son, Y.W.; Jung, I.Y.; Ahn, M.Y.; Jeon, Y.D.; Ann, H.W.; Ahn, J.Y.; Ku, N.S.; Han, S.H.; Choi, J.Y.; Song, Y.G.; et al. A case of community-acquired pneumonia caused by multidrug-resistant acinetobacter baumannii in Korea. Infect. Chemother. 2017, 49, 297–300. [Google Scholar] [CrossRef] [Green Version]
- Peng, C.; Zong, Z.; Fan, H. Acinetobacter baumannii isolates associated with community-acquired pneumonia in West China. Clin. Microbiol. Infect. 2012, 18, E491–E493. [Google Scholar] [CrossRef] [Green Version]
- Ong, C.W.; Lye, D.C.; Khoo, K.L.; Chua, G.S.; Yeoh, S.F.; Leo, Y.S.; Tambyah, P.A.; Chua, A.C. Severe community-acquired acinetobacter baumannii pneumonia: An emerging highly lethal infectious disease in the Asia-Pacific. Respirology 2009, 14, 1200–1205. [Google Scholar] [CrossRef]
- Funke, G.; Funke-Kissling, P. Evaluation of the new VITEK 2 card for identification of clinically relevant gram-negative rods. J. Clin. Microbiol. 2004, 42, 4067–4071. [Google Scholar] [CrossRef] [Green Version]
- Kiska, D.L.; Kerr, A.; Jones, M.C.; Caracciolo, J.A.; Eskridge, B.; Jordan, M.; Miller, S.; Hughes, D.; King, N.; Gilligan, P.H. Accuracy of four commercial systems for identification of burkholderia cepacia and other gram-negative nonfermenting bacilli recovered from patients with cystic fibrosis. J. Clin. Microbiol. 1996, 34, 886–891. [Google Scholar] [CrossRef] [Green Version]
- Kim, S.; Kim, M.H.; Lee, W.I.; Kang, S.Y.; Jeon, Y. Misidentification of acinetobacter baumannii as alcaligenes faecalis by VITEK 2 system; Case report. Lab. Med. 2017, 49, e14–e17. [Google Scholar] [CrossRef] [PubMed]
- Greer, R.C.; Wangrangsimakul, T.; Amornchai, P.; Wuthiekanun, V.; Laongnualpanich, A.; Dance, D.A.B.; Limmathurotsakul, D. Misidentification of burkholderia pseudomallei as acinetobacter species in northern Thailand. Trans. R. Soc. Trop. Med. Hyg. 2019, 113, 48–51. [Google Scholar] [CrossRef]
- Shrestha, M.; Khanal, B. Acinetobacter species: Phenotypic characterization and antimicrobial resistance. J. Nobel Med. Coll. 2013, 2, 43–48. [Google Scholar] [CrossRef]
- Savini, V.; Catavitello, C.; Talia, M.; Febbo, F.; Balbinot, A.; Pompilio, A.; Di Bonaventura, G.; Piccolomini, R.; D’Antonio, D. Misidentification of ampicillin-sulbactam heteroresistance in acinetobacter baumannii strains from ICU patients. J. Infect. 2009, 58, 316–317. [Google Scholar] [CrossRef] [PubMed]
- Nie, D.; Hu, Y.; Chen, Z.; Li, M.; Hou, Z.; Luo, X.; Mao, X.; Xue, X. Outer membrane protein A (OmpA) as a potential therapeutic target for Acinetobacter baumannii infection. J. Biomed. Sci. 2020, 27, 26. [Google Scholar] [CrossRef] [Green Version]
- Fitzgerald, D.M. The road to resistance. Elife 2019, 8, e52092. [Google Scholar] [CrossRef]
- Domingues, S.; Rosario, N.; Candido, A.; Neto, D.; Nielsen, K.M.; Da Silva, G.J. Competence for natural transformation is common among clinical strains of resistant acinetobacter spp. Microorganisms 2019, 7, 30. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hu, Y.; He, L.; Tao, X.; Meng, F.; Zhang, J. High DNA uptake capacity of international clone II acinetobacter baumannii detected by a novel planktonic natural transformation assay. Front. Microbiol. 2019, 10, 2165. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Livermore, D.M. The impact of carbapenemases on antimicrobial development and therapy. Curr. Opin. Investig. Drugs 2002, 3, 218–224. [Google Scholar]
- Poirel, L.; Nordmann, P. Acquired carbapenem-hydrolyzing beta-lactamases and their genetic support. Curr. Pharm. Biotechnol. 2002, 3, 117–127. [Google Scholar] [CrossRef]
- Codjoe, F.S.; Donkor, E.S. Carbapenem resistance: A review. Med. Sci. 2017, 6, 1. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tooke, C.L.; Hinchliffe, P.; Bragginton, E.C.; Colenso, C.K.; Hirvonen, V.H.A.; Takebayashi, Y.; Spencer, J. β-Lactamases and β-Lactamase inhibitors in the 21st century. J. Mol. Biol. 2019, 431, 3472–3500. [Google Scholar] [CrossRef]
- Walther-Rasmussen, J.; Hoiby, N. OXA-type carbapenemases. J. Antimicrob. Chemother. 2006, 57, 373–383. [Google Scholar] [CrossRef] [Green Version]
- Aly, M.; Tayeb, H.T.; Al Johani, S.M.; Alyamani, E.J.; Aldughaishem, F.; Alabdulkarim, I.; Balkhy, H.H. Genetic diversity of OXA-51-like genes among multidrug-resistant acinetobacter baumannii in Riyadh, Saudi Arabia. Eur. J. Clin. Microbiol. Infect. Dis. 2014, 33, 1223–1228. [Google Scholar] [CrossRef] [PubMed]
- Turton, J.F.; Woodford, N.; Glover, J.; Yarde, S.; Kaufmann, M.E.; Pitt, T.L. Identification of acinetobacter baumannii by detection of the blaOXA-51-like carbapenemase gene intrinsic to this species. J. Clin. Microbiol. 2006, 44, 2974–2976. [Google Scholar] [CrossRef] [Green Version]
- Asif, M.; Alvi, I.A.; Rehman, S.U. Insight into acinetobacter baumannii: Pathogenesis, global resistance, mechanisms of resistance, treatment options, and alternative modalities. Infect. Drug Resist. 2018, 11, 1249–1260. [Google Scholar] [CrossRef] [Green Version]
- Zarrilli, R.; Giannouli, M.; Tomasone, F.; Triassi, M.; Tsakris, A. Carbapenem resistance in acinetobacter baumannii: The molecular epidemic features of an emerging problem in health care facilities. J. Infect. Dev. Ctries. 2009, 3, 335–341. [Google Scholar] [CrossRef]
- Perez, F.; Hujer, A.M.; Hujer, K.M.; Decker, B.K.; Rather, P.N.; Bonomo, R.A. Global challenge of multidrug-resistant acinetobacter baumannii. Antimicrob. Agents Chemother. 2007, 51, 3471–3484. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cillóniz, C.; Dominedò, C.; Torres, A. Multidrug resistant gram-negative bacteria in community-acquired pneumonia. Crit. Care 2019, 23, 79. [Google Scholar] [CrossRef] [Green Version]
- Robert, G.; Edwards, J.R. Overview of nosocomial infections caused by gram-negative bacilli. Clin. Infect. Dis. 2005, 41, 848–854. [Google Scholar]
- Jung, S.Y.; Lee, S.H.; Lee, S.Y.; Yang, S.; Noh, H.; Chung, E.K.; Lee, J.I. Antimicrobials for the treatment of drug-resistant acinetobacter baumannii pneumonia in critically ill patients: A systemic review and Bayesian network meta-analysis. Crit. Care 2017, 21, 319. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chang, H.C.; Wei, Y.F.; Dijkshoorn, L.; Vaneechoutte, M.; Tang, C.T.; Chang, T.C. Species-level identification of isolates of the acinetobacter calcoaceticus-acinetobacter baumannii complex by sequence analysis of the 16S-23S rRNA gene spacer region. J. Clin. Microbiol. 2005, 43, 1632–1639. [Google Scholar] [CrossRef] [Green Version]
- Lee, Y.T.; Kuo, S.C.; Chiang, M.C.; Yang, S.P.; Chen, C.P.; Chen, T.L.; Fung, C.P. Emergence of carbapenem-resistant non-baumannii species of acinetobacter harboring a blaOXA-51-like gene that is intrinsic to A. baumannii. Antimicrob. Agents Chemother. 2012, 56, 1124–1127. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maslunka, C.; Gifford, B.; Tucci, J.; Gurtler, V.; Seviour, R.J. Insertions or deletions (Indels) in the rrn 16S-23S rRNA gene internal transcribed spacer region (ITS) compromise the typing and identification of strains within the acinetobacter calcoaceticus-baumannii (Acb) complex and closely related members. PLoS ONE 2014, 9, e105390. [Google Scholar] [CrossRef]
- Benachinmardi, K.; Padmavathy, M.; Malini, J.; Naveneeth, B. Prevalence of non-fermenting gram-negative bacilli and their in vitro susceptibility pattern at a tertiary care teaching hospital. J. Sci. Soc. 2014, 41, 162. [Google Scholar] [CrossRef]
- Grewal, U.; Bakshi, R.; Walia, G.; Shah, P. Antibiotic susceptibility profiles of non-fermenting gram-negative bacilli at a tertiary care hospital in Patiala, India. Niger. Postgrad. Med. J. 2017, 24, 121. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, M.; Jena, J.; Pattnaik, D.; Mallick, B. Prevalence of nonfermentative gram-negative bacilli and their antimicrobial susceptibility profiles in a tertiary care hospital of Eastern India. Int. J. Adv. Med. 2018, 5, 366. [Google Scholar] [CrossRef] [Green Version]
- Falagas, M.E.; Karveli, E.A.; Kelesidis, I.; Kelesidis, T. Community-acquired acinetobacter infections. Eur. J. Clin. Microbiol. Infect. Dis. 2007, 26, 857–868. [Google Scholar] [CrossRef] [PubMed]
- Rebic, V.; Masic, N.; Teskeredzic, S.; Aljicevic, M.; Abduzaimovic, A.; Rebic, D. The importance of acinetobacter species in the hospital environment. Med. Arch. 2018, 72, 330. [Google Scholar] [CrossRef] [PubMed]
- Pires, S.; Peignier, A.; Seto, J.; Smyth, D.S.; Parker, D. Biological sex influences susceptibility to acinetobacter baumannii pneumonia in mice. JCI Insight 2020, 5, e132223. [Google Scholar] [CrossRef] [Green Version]
- Harding, C.M.; Hennon, S.W.; Feldman, M.F. Uncovering the mechanisms of acinetobacter baumannii virulence. Nat. Rev. Microbiol. 2018, 16, 91–102. [Google Scholar] [CrossRef] [PubMed]
- Moustafa, N.T.; El-gendy, A.O.; Saafan, A.E.; Tawakkol, W.M. Phenotypic detection of efflux mechanism in panaminoglycoside resistant acinetobacter baumannii from Egyptian clinical isolates. Jundishapur J. Microbiol. 2018, 11. [Google Scholar] [CrossRef] [Green Version]
- Abdalhamid, B.; Hassan, H.; Itbaileh, A.; Shorman, M. Characterization of carbapenem-resistant acinetobacter baumannii clinical isolates in a tertiary care hospital in Saudi Arabia. New Microbiol. 2014, 37, 65–73. [Google Scholar] [PubMed]
- Al-Agamy, M.H.; Khalaf, N.G.; Tawfick, M.M.; Shibl, A.M.; El Kholy, A. Molecular characterization of carbapenem-insensitive acinetobacter baumannii in Egypt. Int. J. Infect. Dis. 2014, 22, 49–54. [Google Scholar] [CrossRef] [Green Version]
- Rynga, D.; Shariff, M.; Deb, M. Phenotypic and molecular characterization of clinical isolates of acinetobacter baumannii isolated from Delhi, India. Ann. Clin. Microbiol. Antimicrob. 2015, 14, 40. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bakour, S.; Touati, A.; Sahli, F.; Ameur, A.A.; Haouchine, D.; Rolain, J.M. Antibiotic resistance determinants of multidrug-resistant acinetobacter baumannii clinical isolates in Algeria. Diagn. Microbiol. Infect. Dis. 2013, 76, 529–531. [Google Scholar] [CrossRef]
- Amudhan, S.M.; Sekar, U.; Arunagiri, K.; Sekar, B. OXA beta-lactamase-mediated carbapenem resistance in acinetobacter baumannii. Indian J. Med. Microbiol. 2011, 29, 269–274. [Google Scholar]
- Al-Zahrani, I.A. Routine detection of carbapenem-resistant gram-negative bacilli in clinical laboratories. A review of current challenge. Saudi Med. J. 2018, 39, 861–872. [Google Scholar] [CrossRef]
- June, C.M.; Vallier, B.C.; Bonomo, R.A.; Leonard, D.A.; Powers, R.A. Structural origins of oxacillinase specificity in class D beta-lactamases. Antimicrob. Agents Chemother. 2014, 58, 333–341. [Google Scholar] [CrossRef] [Green Version]
- Lutgring, J.D.; Limbago, B.M. The problem of carbapenemase-producing-carbapenem-resistant-enterobacteriaceae detection. J. Clin. Microbiol. 2016, 54, 529–534. [Google Scholar] [CrossRef] [Green Version]
- Wang, J.T.; McDonald, L.C.; Chang, S.C.; Ho, M. Community-acquired acinetobacter baumannii bacteremia in adult patients in Taiwan. J. Clin. Microbiol. 2002, 40, 1526–1529. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ying, C.; Li, Y.; Wang, Y.; Zheng, B.; Yang, C. Investigation of the molecular epidemiology of acinetobacter baumannii isolated from patients and environmental contamination. J. Antibiot. 2015, 68, 562–567. [Google Scholar] [CrossRef] [PubMed]
- El-Sayed-Ahmed, M.A.E.-G.; Amin, M.A.; Tawakol, W.M.; Loucif, L.; Bakour, S.; Rolain, J.-M. High prevalence of bla(NDM-1) carbapenemase-encoding gene and 16S rRNA armA methyltransferase gene among acinetobacter baumannii clinical isolates in Egypt. Antimicrob. Agents Chemother. 2015, 59, 3602–3605. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alkasaby, N.M.; El Sayed Zaki, M. Molecular study of acinetobacter baumannii isolates for metallo-β-lactamases and extended-spectrum-β-lactamases genes in intensive care unit, Mansoura University Hospital, Egypt. Int. J. Microbiol. 2017, 2017, 3925868. [Google Scholar] [CrossRef] [Green Version]
- Al-Hassan, L.; Zafer, M.M.; El-Mahallawy, H. Multiple sequence types responsible for healthcare-associated acinetobacter baumannii dissemination in a single centre in Egypt. BMC Infect. Dis. 2019, 19, 829. [Google Scholar] [CrossRef] [Green Version]
- Asadian, M.; Azimi, L.; Alinejad, F.; Ostadi, Y.; Lari, A. Molecular characterization of acinetobacter baumannii isolated from ventilator-associated pneumonia and burn wound colonization by random amplified polymorphic DNA polymerase chain reaction and the relationship between antibiotic susceptibility and biofilm production. Adv. Biomed. Res. 2019, 8, 58. [Google Scholar]
- Koeleman, J.G.; Stoof, J.; Biesmans, D.J.; Savelkoul, P.H.; Vandenbroucke-Grauls, C.M. Comparison of amplified ribosomal DNA restriction analysis, random amplified polymorphic DNA analysis, and amplified fragment length polymorphism fingerprinting for identification of acinetobacter genomic species and typing of acinetobacter baumannii. J. Clin. Microbiol. 1998, 36, 2522–2529. [Google Scholar] [CrossRef] [Green Version]
- Mac Faddin, J.F. Biochemical Tests for Identification of Medical Bacteria, 3rd ed.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2000. [Google Scholar]
- Evans, B.A.; Hamouda, A.; Towner, K.J.; Amyes, S.G. OXA-51-like beta-lactamases and their association with particular epidemic lineages of acinetobacter baumannii. Clin. Microbiol. Infect. 2008, 14, 268–275. [Google Scholar] [CrossRef]
- CLSI. Performance Standards for Antimicrobial Susceptibility Testing; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2017. [Google Scholar]
- Antony, B. Comparative evaluation of four phenotypic tests for detection of metallo-β-lactamase and carbapenemase production in acinetobacter baumannii. J. Clin. Microbiol. 2014, 8, DC05. [Google Scholar] [CrossRef]
- Poirel, L.; Walsh, T.R.; Cuvillier, V.; Nordmann, P. Multiplex PCR for detection of acquired carbapenemase genes. Diagn. Microbiol. Infect. Dis. 2011, 70, 119–123. [Google Scholar] [CrossRef]
- Jeon, B.C.; Jeong, S.H.; Bae, I.K.; Kwon, S.B.; Lee, K.; Young, D.; Lee, J.H.; Song, J.S.; Lee, S.H. Investigation of a nosocomial outbreak of imipenem-resistant acinetobacter baumannii producing the OXA-23 beta-lactamase in Korea. J. Clin. Microbiol. 2005, 43, 2241–2245. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- ÇİÇek, A.Ç.; Düzgün, A.Ö.; Saral, A.; Kayman, T.; Çİzmecİ, Z.; Balcı, P.Ö.; Dal, T.; Fırat, M.; Tosun, İ.; Alıtntop, Y.A.; et al. Detection of class 1 integron in acinetobacter baumannii isolates collected from nine hospitals in Turkey. Asian Pac. J. Trop. Biomed. 2013, 3, 743–747. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Chen, Y.; Tang, Y.; Zhang, K. Combination of ARDRA and RAPD genotyping techniques in identification of acinetobacter spp. genomic species. Front. Biol. China 2008, 3, 397–401. [Google Scholar] [CrossRef]
- Heras, J.; Dominguez, C.; Mata, E.; Pascual, V.; Lozano, C.; Torres, C.; Zarazaga, M. GelJ—a tool for analyzing DNA fingerprint gel images. BMC Bioinform. 2015, 16, 270. [Google Scholar] [CrossRef] [PubMed] [Green Version]


| Sample Origin | Gender | Urine | Sputum | Pus | Blood | Wound | CSF * | Total | Collecting Lab. Identification % | |
|---|---|---|---|---|---|---|---|---|---|---|
| Strain | n (%) | |||||||||
| Pseudomonas aeruginosa | Male Female Total | 36 | 18 | 6 | 10 | 21 | 3 | 94 (47) | 100% | |
| 21 | 13 | 4 | 8 | 14 | 2 | 62 (31) | ||||
| 57 | 31 | 10 | 18 | 35 | 5 | 156 (78) | ||||
| Acinetobacter baumannii | Male Female Total | 0 | 1 | - | - | - | 1 | 2 (1) | unidentified | |
| 16 | 4 | - | - | - | 1 | 21 (10.5) | ||||
| 16 | 5 | - | - | - | 2 | 23 (11.5) | ||||
| Acinetobacter lwoffii | Male Female Total | 2 | 3 | - | 1 | - | - | 6 (3) | unidentified | |
| 1 | 0 | - | 0 | - | - | 1 (0.5) | ||||
| 3 | 3 | - | 1 | - | - | 7 (3.5) | ||||
| Acinetobacter nosocomialis | Male Female Total | - | - | 1 | 1 | - | - | 2 (1) | unidentified | |
| - | - | 0 | 0 | - | - | 0 | ||||
| - | - | 1 | 1 | - | - | 2 (1) | ||||
| Acinetobacter calcoaceticus | Male Female Total | 0 | 1 | 1 | - | 2 | - | 4 (2) | unidentified | |
| 1 | 0 | 0 | - | 1 | - | 2 (1) | ||||
| 1 | 1 | 1 | - | 3 | - | 6 (3) | ||||
| Burkholderia cepacia | Male Female Total | - | 0 | - | - | 1 | - | 1 (0.5) | unidentified | |
| - | 1 | - | - | 1 | - | 2 (1) | ||||
| - | 1 | - | - | 2 | - | 3 (1.5) | ||||
| Stenotrophomonas maltophilia | Male Female Total | - | - | - | 0 | - | 1 | 1 (0.5) | unidentified | |
| - | - | - | 2 | - | 0 | 2 (1) | ||||
| - | - | - | 2 | - | 1 | 3 (1.5) | ||||
| Total | Male Female Total | 38 | 23 | 8 | 12 | 24 | 5 | 110 (55) | ||
| 39 | 18 | 4 | 10 | 16 | 3 | 90 (45) | ||||
| 77 | 41 | 12 | 22 | 40 | 8 | 200 | ||||
| Variables | Sample Origin | Sign | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Urine | Sputum | CSF | Total | |||||||
| n | % | n | % | n | % | n | ||||
| Gender | Male | 0 | 0.0 | 1 | 50 | 1 | 50 | 2 | 0.009 * | |
| Female | 16 | 76.2 | 4 | 19.0 | 1 | 4.8 | 21 | |||
| Total | 16 | 69.6 | 5 | 21.7 | 2 | 8.7 | 23 | |||
| Age | Male (mean ± SD) | 0.0 | 18.0 ± 19.02 | 52.0 ± 19.21 | 35.0 ± 13.58 | 0.049 * | ||||
| Female (mean ± SD) | 41.5 ± 4.80 | 44.0 ± 9.60 | 5.0 ± 19.21 | 30.17 ± 7.34 | ||||||
| Total (mean ± SD) | 41.5 ± 16.23 | 38.8 ± 28.40 | 28.5 ± 33.20 | 39.8 ± 19.90 | 0.695 ns | |||||
| Age groups | 0–15 years | 0 | 0.0 | 1 | 50.0 | 1 | 50.0 | 2 | 0.333 ns | |
| 16–30 years | 7 | 87.5 | 1 | 12.5 | 0 | 0.0 | 8 | |||
| 31–55 years | 4 | 66.7 | 1 | 16.7 | 1 | 16.7 | 6 | |||
| >56 years | 5 | 71.4 | 2 | 28.6 | 0 | 0.0 | 7 | |||
| Total | 16 | 69.6 | 5 | 21.7 | 2 | 8.7 | 23 | |||
| Antimicrobial | Resistance (n) % | Sign. | ||
|---|---|---|---|---|
| S | R | I | ||
| Tobramycin | (8) 34.8% | (15) 65.2% | 0 | 0.144 ns |
| Gentamycin | (5) 21.7% | (17) 73.9% | (1) 4.3% | <0.001 * |
| Amikacin | (11) 47.8% | (12) 52.2% | 0 | 0.835 ns |
| Aztreonam | (3) 13% | (20) 87% | 0 | <0.001 * |
| Norfloxacin | (7) 30.4% | (16) 69.6% | 0 | 0.061 ns |
| Levofloxacin | (6) 26.1% | (15) 65.2% | (2) 8.7% | 0.003 * |
| Ofloxacin | (8) 34.8% | (14) 60.9% | (1) 4.3% | 0.0004 * |
| Ciprofloxacin | (7) 30.4% | (16) 69.6% | 0 | 0.061 ns |
| Nalidixic acid | 0 | (23) 100% | 0 | <0.001 * |
| Piperacillin/tazobactam | (3) 13% | (19) 82.6% | (3) 13% | <0.001 * |
| Oxacillin | 0 | (23) 100% | 0 | <0.001 * |
| Amoxicillin/clavulanic acid | 0 | (23) 100% | 0 | <0.001 * |
| Ceftazidime | (2) 8.7% | (20) 87% | (1) 4.3% | <0.001 * |
| Cefotaxime | (1) 4.3% | (21) 91.3% | (1) 4.3% | <0.001 * |
| Cefepime | (9) 39.1% | (13) 56.5% | (1) 4.3% | 0.008 * |
| Meropenem | (8) 34.8% | (12) 52.2% | (3) 13% | 0.070 ns |
| Imipenem | (10) 39.1% | (11) 47.8% | (2) 8.7% | <0.001 * |
| Ertapenem | (3) 13% | (20) 87% | 0 | <0.001 * |
| Doripenem | (5) 21.7% | (17) 73.9% | (1) 4.3% | <0.001 * |
| Colistin | (17) 73.9% | (4) 17.4% | (2) 8.7% | <0.001 * |
| Polymyxin B | (19) 82.6% | (4) 17.4% | 0 | 0.002 * |
| Oligo | Sequence (5′→3′) | Amplicon Size (bp) | Annealing Tmp (°C) | Reference |
|---|---|---|---|---|
| (16-23S ITS) P-1512F | GTCGTAACAAGGTAGCCGTA | 607 | 60 | [33] |
| (16-23S ITS) P-6R | GGGTTC/TCCCCA/GTTCRGAAAT | |||
| blaOXA-69F (F) | CTAATAATTGATCTACTCAAG | 975 | 48 | [60] |
| blaOXA-69R (R) | CCAGTGGATGGATGGATAGATTATC | |||
| blaOXA-51F (F) | TAATGCTTTGATCGGCCTTG | 353 | 52 | [63] |
| blaOXA-51R (R) | TGGATTGCACTTCATCTTGG | |||
| blaOXA-23F (F) | GATGTGTCATAGTATTCGTCGT | 1058 | [64] | |
| blaOXA-23R (R) | TCACAACAACTAAAAGCACTGT | |||
| blaOXA-24F (F) | GGTTAGTTGGCCCCCTAAAA | 246 | [63] | |
| blaOXA-24R (R) | AGTTGAGCGAAAAGGGGATT | |||
| blaOXA-58F (F) | AAGTATTGGGGCTTGTGCTG | 599 | [63] | |
| blaOXA-58R (R) | CCCCTCTGCGCTCTACATAC | |||
| (blaIMP) IMP-F (blaIMP) IMP-R | GGAATAGAGTGGCTTAAYTCTC GGTTTAAYAAAACAACCACC | 232 | 52 | [63] |
| (blaVIM) VIM-F (blaVIM) VIM-R | GATGGTGTTTGGTCGCATA CGAATGCGCAGCACCAG | 390 | ||
| (blaNDM) NDM-F (blaNDM) NDM-R | GGTTTGGCGATCTGGTTTTC CGGAATGGCTCATCACGATC | 621 | ||
| (blaKPC) KPC-F (blaKPC) KPC-R | CGTCTAGTTCTGCTGTCTTG CTTGTCATCCTTGTTAGGCG | 798 | ||
| (blaGES) GES-F (blaGES) GES-R | ATGCGCTTCATTCACGCAC CTATTTGTCCGTGCTCAGGA | 863 | 55 | [65] |
| OPM-01 (P1) | GTTGGTGGCT | Variable | 40 | Eurofins Kit |
| OPM-07 (P2) | CCGTGACTCA | |||
| OPM-09 (P3) | GTCTTGCGGA | |||
| OPQ-06 (P4) | GAGCGCCTTG | |||
| OPQ-08 (P5) | CTCCAGCGGA | |||
| OPM-03 (P6) | GGGGGATGAG | |||
| OPQ-02 (P7) | TCTGTCGGTC | |||
| OPM-05 (P8) | GGGAACGTGT | |||
| OPQ-04 (P9) | AGTGCGCTGA | |||
| OPQ-10 (P10) | TGTGCCCGAA |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
El-Kazzaz, W.; Metwally, L.; Yahia, R.; Al-Harbi, N.; El-Taher, A.; Hetta, H.F. Antibiogram, Prevalence of OXA Carbapenemase Encoding Genes, and RAPD-Genotyping of Multidrug-Resistant Acinetobacter baumannii Incriminated in Hidden Community-Acquired Infections. Antibiotics 2020, 9, 603. https://doi.org/10.3390/antibiotics9090603
El-Kazzaz W, Metwally L, Yahia R, Al-Harbi N, El-Taher A, Hetta HF. Antibiogram, Prevalence of OXA Carbapenemase Encoding Genes, and RAPD-Genotyping of Multidrug-Resistant Acinetobacter baumannii Incriminated in Hidden Community-Acquired Infections. Antibiotics. 2020; 9(9):603. https://doi.org/10.3390/antibiotics9090603
Chicago/Turabian StyleEl-Kazzaz, Waleed, Lobna Metwally, Reham Yahia, Najwa Al-Harbi, Ayat El-Taher, and Helal F. Hetta. 2020. "Antibiogram, Prevalence of OXA Carbapenemase Encoding Genes, and RAPD-Genotyping of Multidrug-Resistant Acinetobacter baumannii Incriminated in Hidden Community-Acquired Infections" Antibiotics 9, no. 9: 603. https://doi.org/10.3390/antibiotics9090603
APA StyleEl-Kazzaz, W., Metwally, L., Yahia, R., Al-Harbi, N., El-Taher, A., & Hetta, H. F. (2020). Antibiogram, Prevalence of OXA Carbapenemase Encoding Genes, and RAPD-Genotyping of Multidrug-Resistant Acinetobacter baumannii Incriminated in Hidden Community-Acquired Infections. Antibiotics, 9(9), 603. https://doi.org/10.3390/antibiotics9090603






