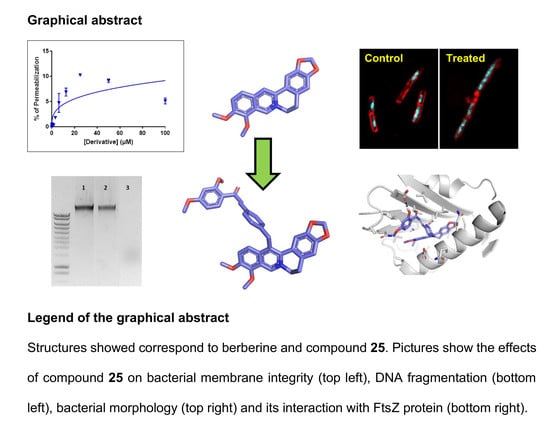
Synthesis and Evaluation of the Antibacterial Activities of 13-Substituted Berberine Derivatives
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemistry
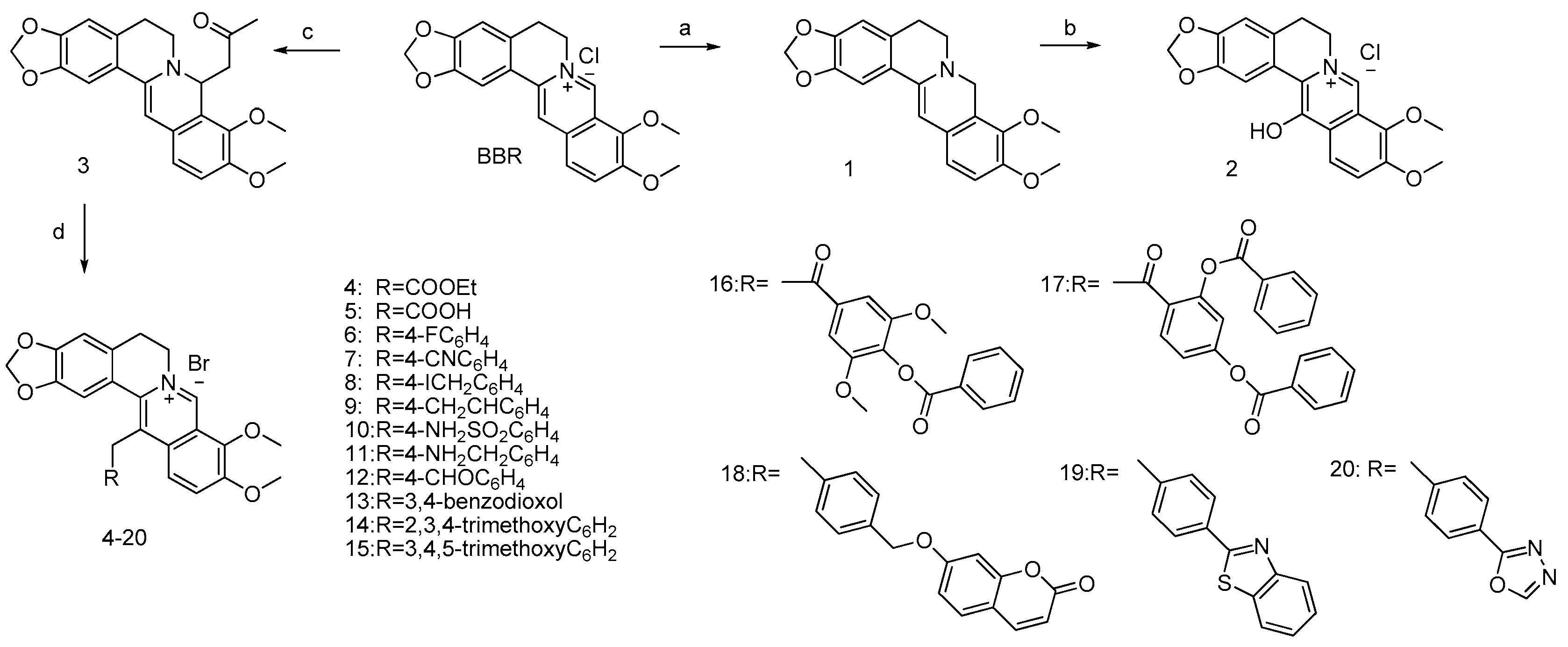
2.1.1. General Procedure for the Synthesis of Compounds 4–20
13-(Acetic Acid Ethylester) Berberine (Compound 4)
13-(Acetic Acid) Berberine (Compound 5)
13-(4-Fluorobenzyl)Berberine (Compound 6)
13-(4-Cyanobenzyl)Berberine (Compound 7)
13-(4-Iodomethylbenzyl)Berberine (Compound 8)
13-(4-Ethenylbenzyl)Berberine (Compound 9)
13-(4-Sulfamoylbenzyl)Berberine (Compound 10)
13-(4-Aminomethylbenzyl)Berberine (Compound 11)
13-(4-Formylbenzyl)Berberine (Compound 12)
13-[(2H-1,3-Benzodioxol-5-yl)methyl]Berberine (Compound 13)
13[(2,3,4-Trimethoxyphenyl)methyl]Berberine (Compound 14)
13[(3,4,5-Trimethoxyphenyl)methyl]Berberine (Compound 15)
4-[2-(Berberin-13-yl) acetyl]-2,6-dimethoxyphenyl Benzoate (Compound 16)
4-[2-(Berberin-13-yl)acetyl]-3-(benzoyloxy)phenyl Benzoate (Compound 17)
8-{4-[(Berberin-13-yl)methyl]phenoxy}-2H-chromen-2-one (Compound 18)
9-{[4-(1,3-Benzothiazol-2-yl)phenyl]methyl}berberine (Compound 19)
9-{[4-(1,3,4-Oxadiazol-2-yl)phenyl]methyl}berberine (Compound 20)
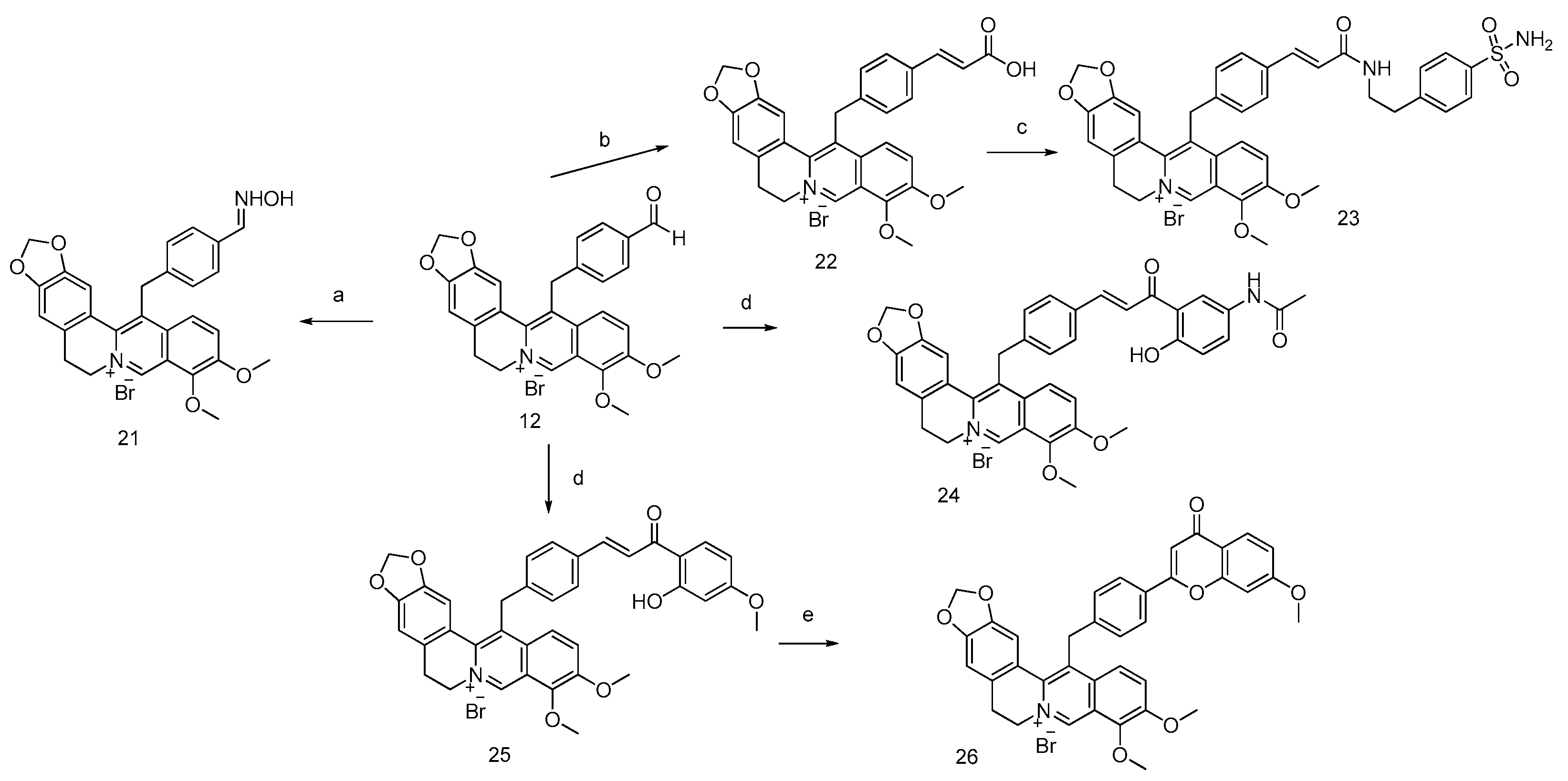
N-({4-[(Berberin-13-yl)methyl]phenyl}methylidene)hydroxylamine (Compound 21)
(2E)-3-{4-[(Berberin-13-yl)methyl]phenyl}prop-2-enoic Acid (Compound 22)
(2E)-3-{4-[(Berberin-13-yl)methyl]phenyl}-N-[2-(4-sulfamoylphenyl)ethyl]prop-2-enamide (Compound 23)
N-{3-[(2E)-3-{4-[(Berberin-13-yl)methyl]phenyl}prop-2-enoyl]-4-hydroxyphenyl}acetamide (Compound 24)
(2E)-3-{4-[(Berberin-13-yl)methyl]phenyl}-1-(2-hydroxy-4-methoxyphenyl)prop-2-en-1-one (Compound 25)
2-{4-[(Berberin-13-yl)methyl]phenyl}-7-methoxy-4H-chromen-4-one (Compound 26)
2.2. Biology
2.2.1. Microorganism Strains Used and Growth Conditions
2.2.2. Antimicrobial Activity Assay
2.2.3. Cytotoxic Assay on Human Cells
2.2.4. Bacterial Membrane Permeabilization Assay
2.2.5. DNA Fragmentation Assay
2.2.6. Docking Studies
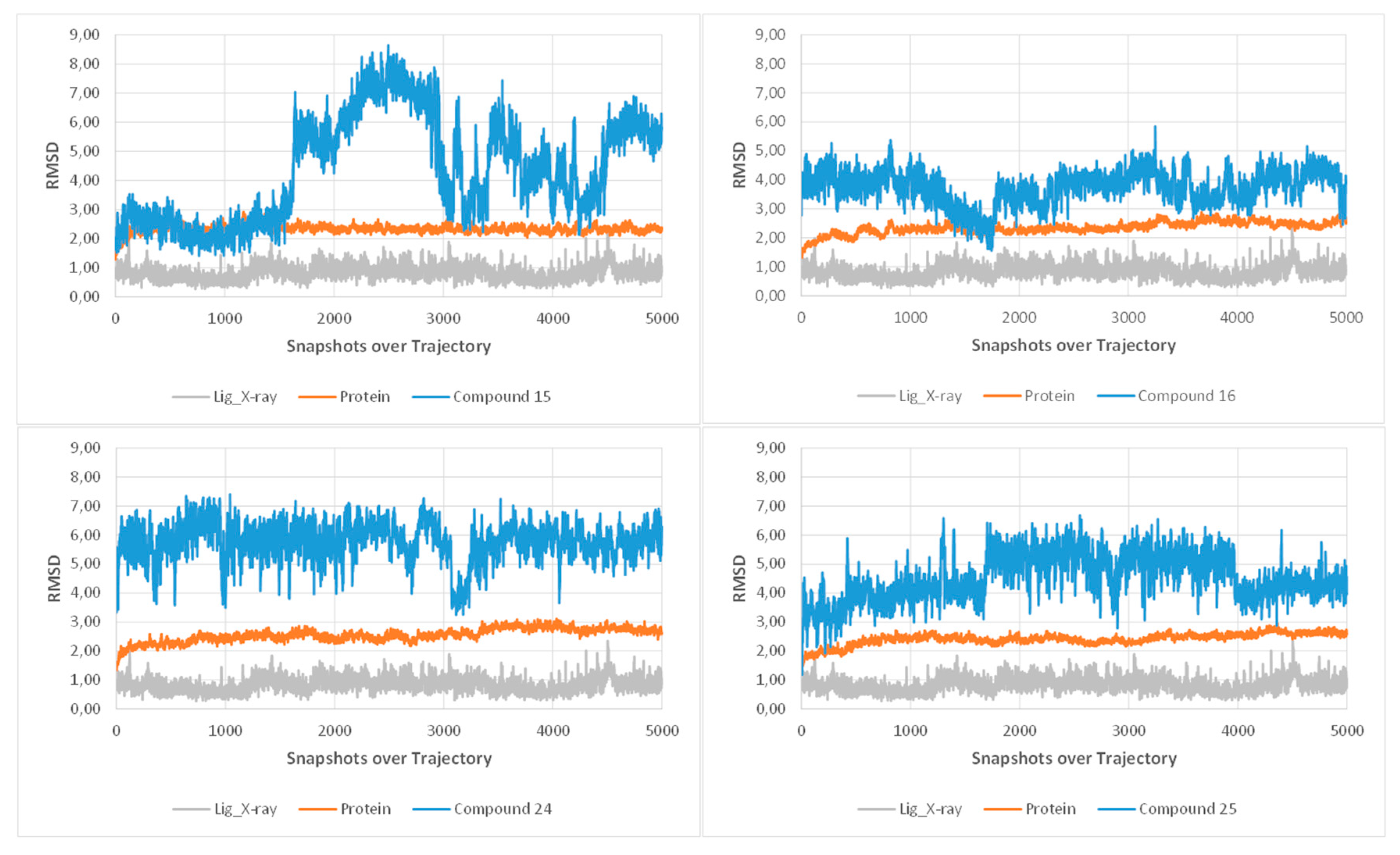
2.2.7. Molecular Dynamics Simulations
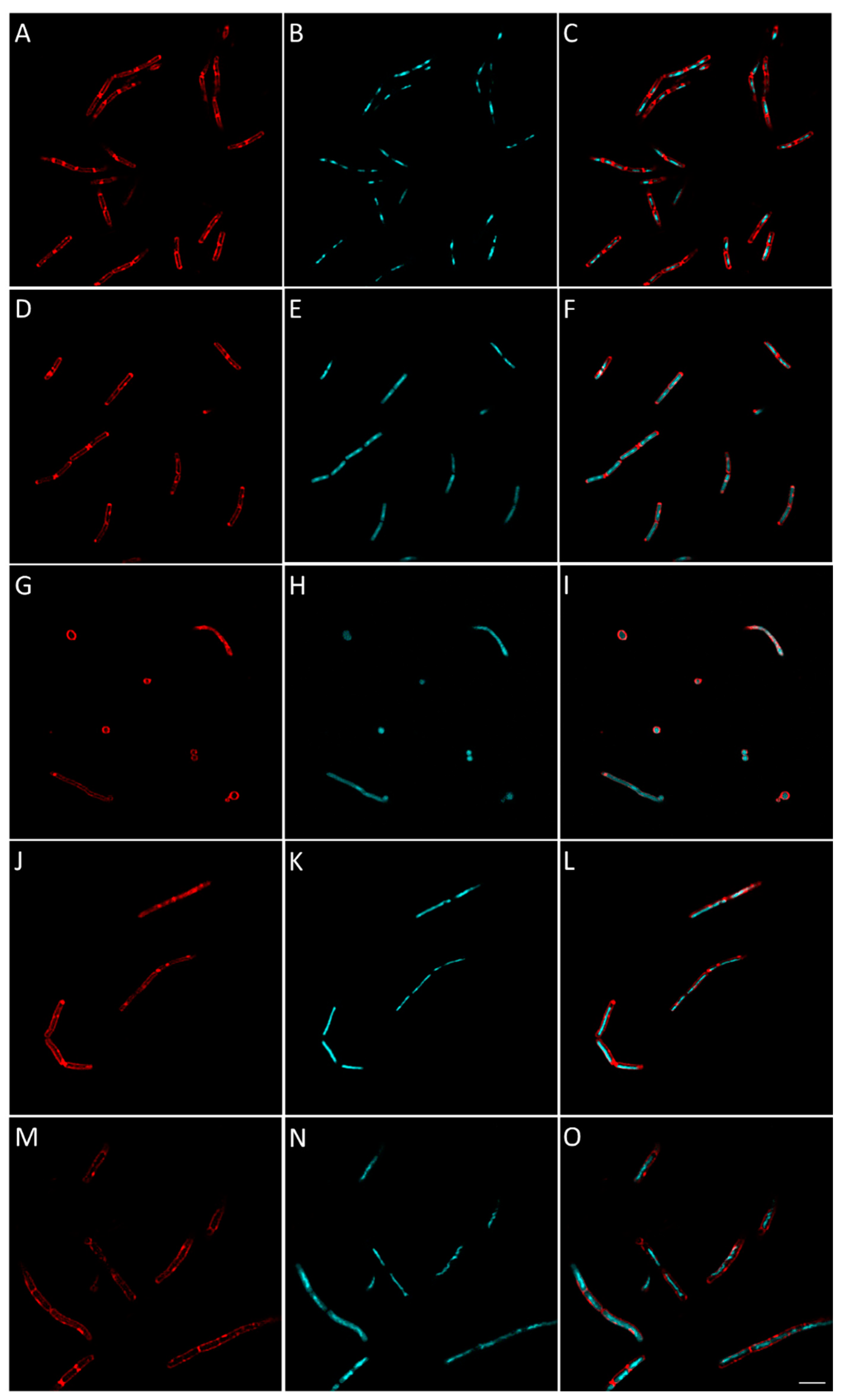
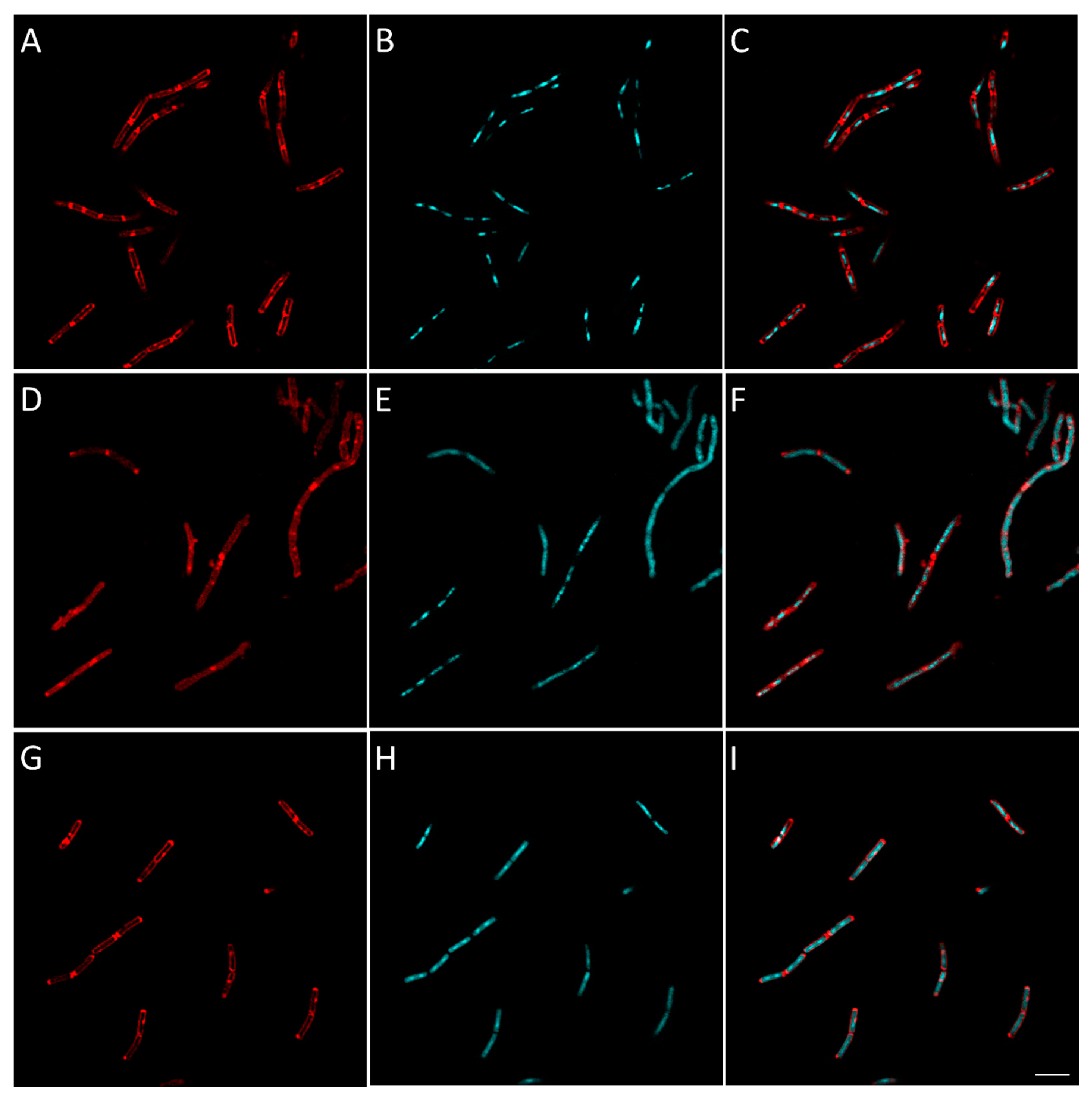
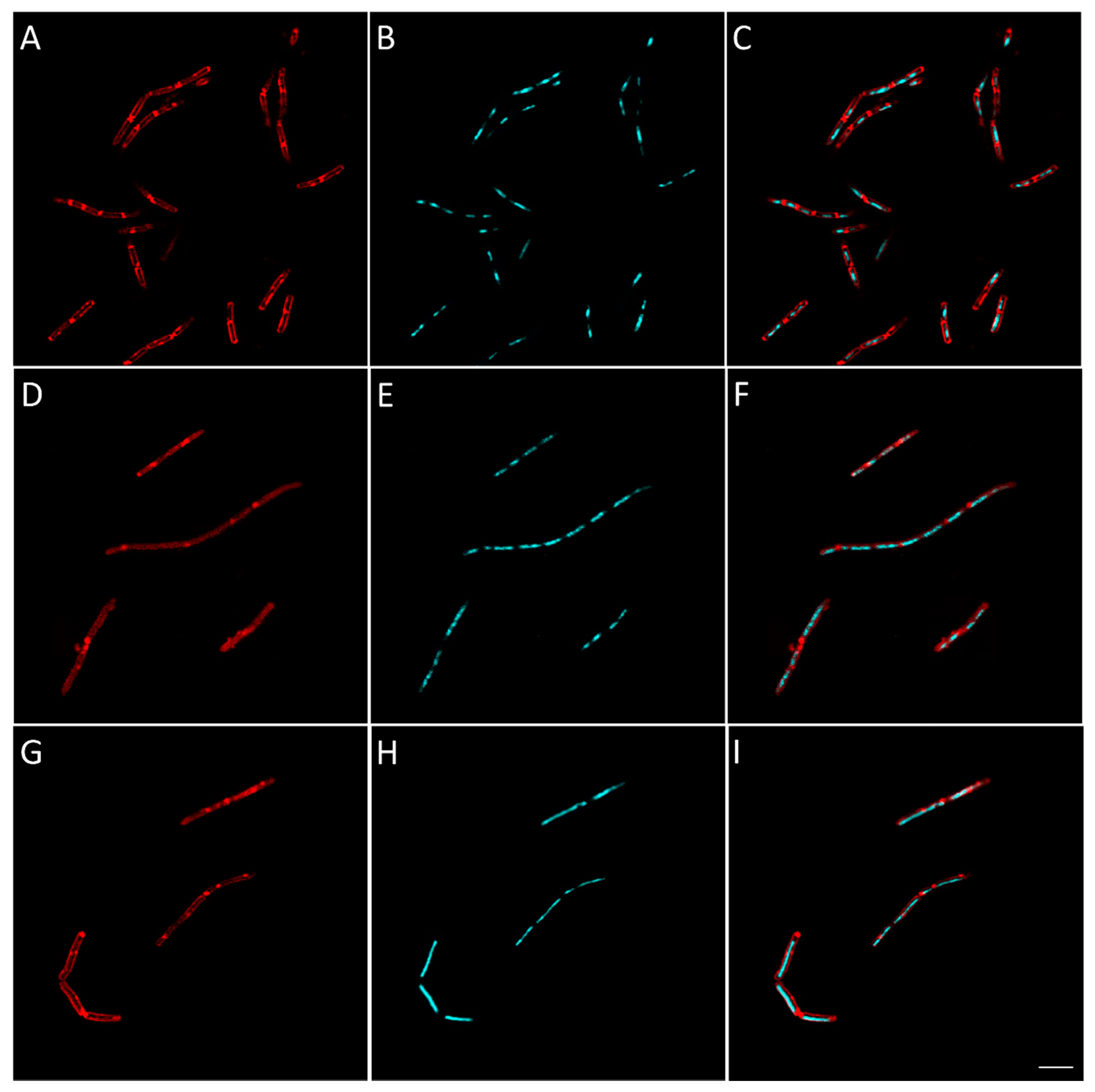
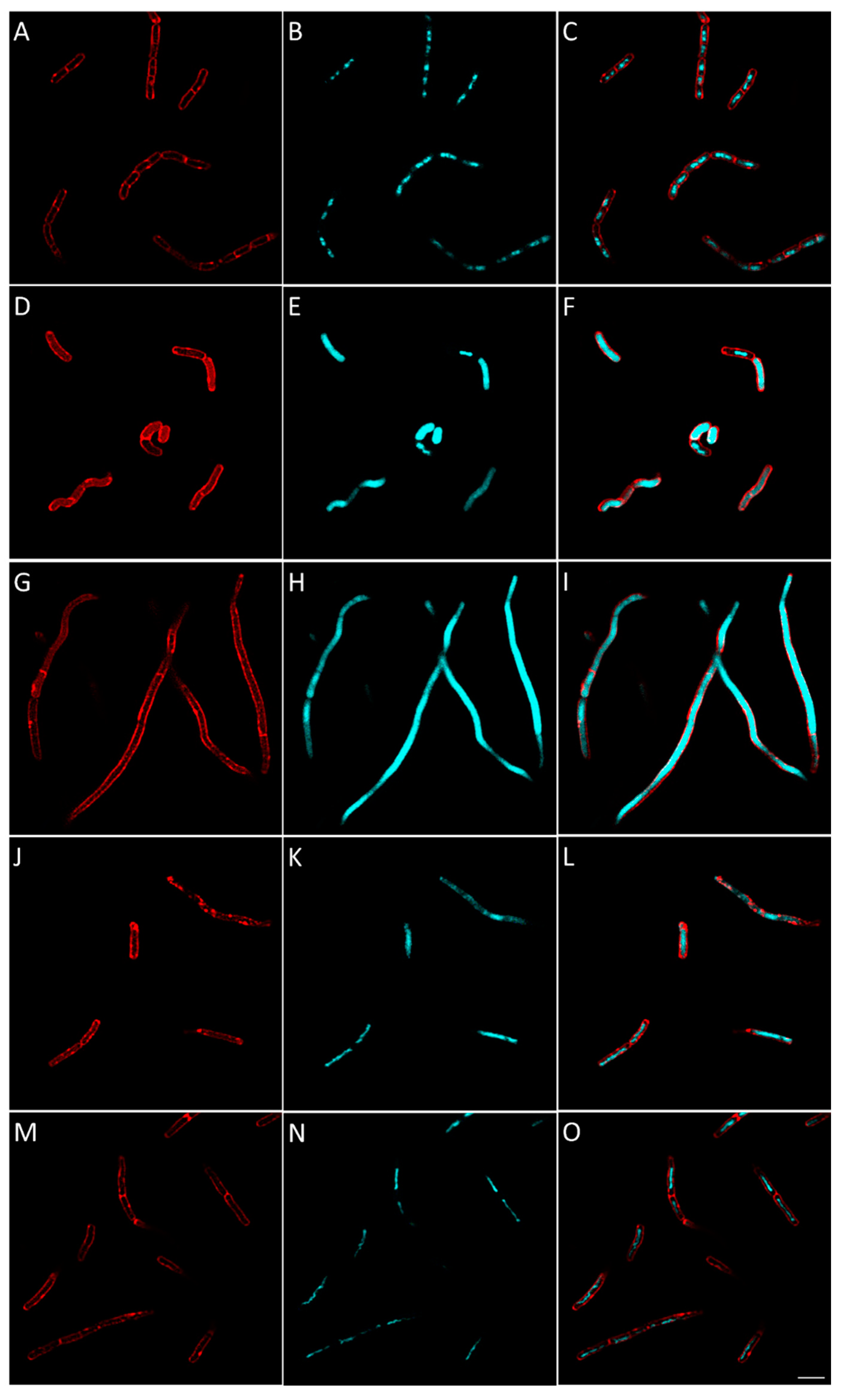
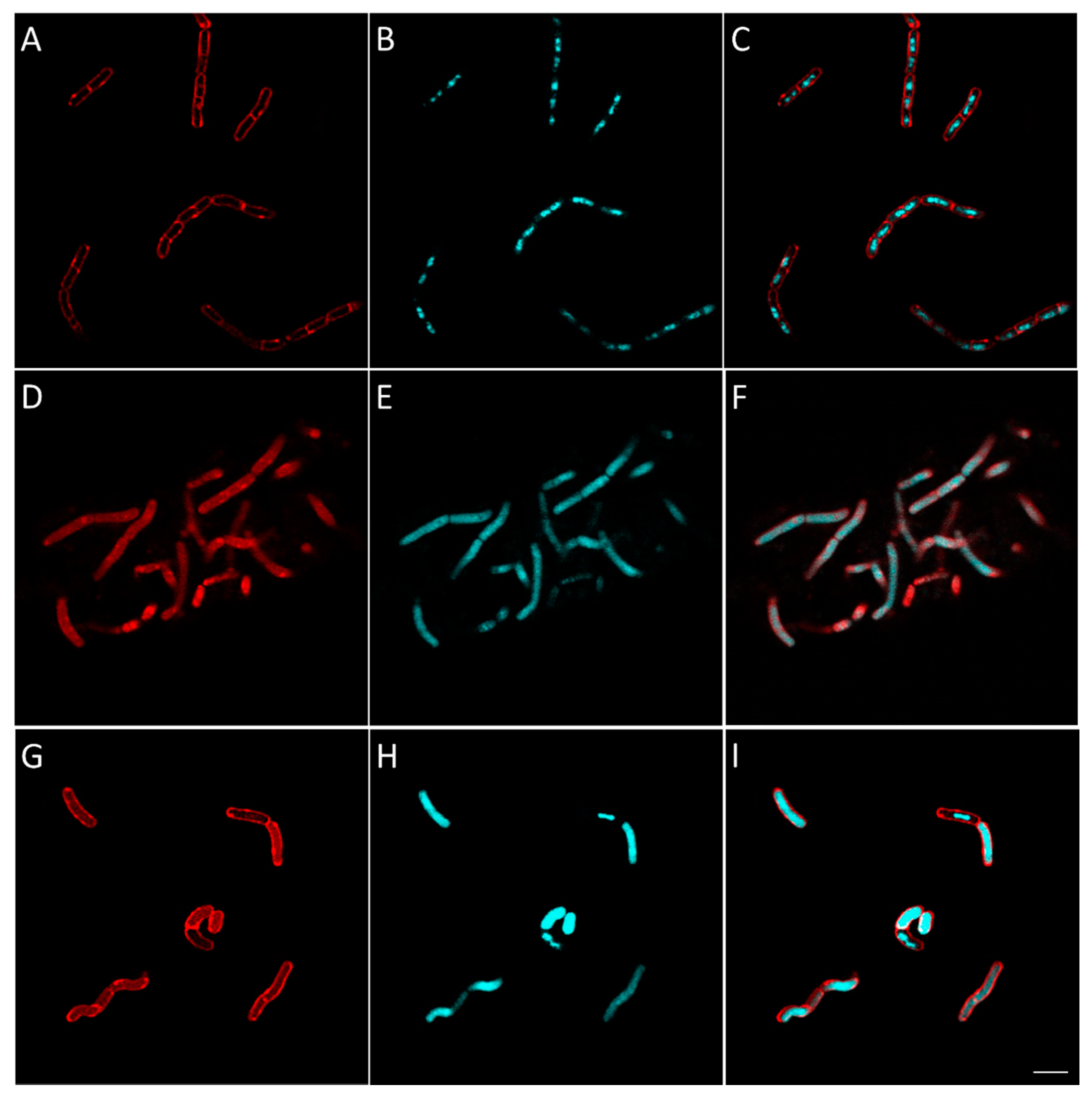
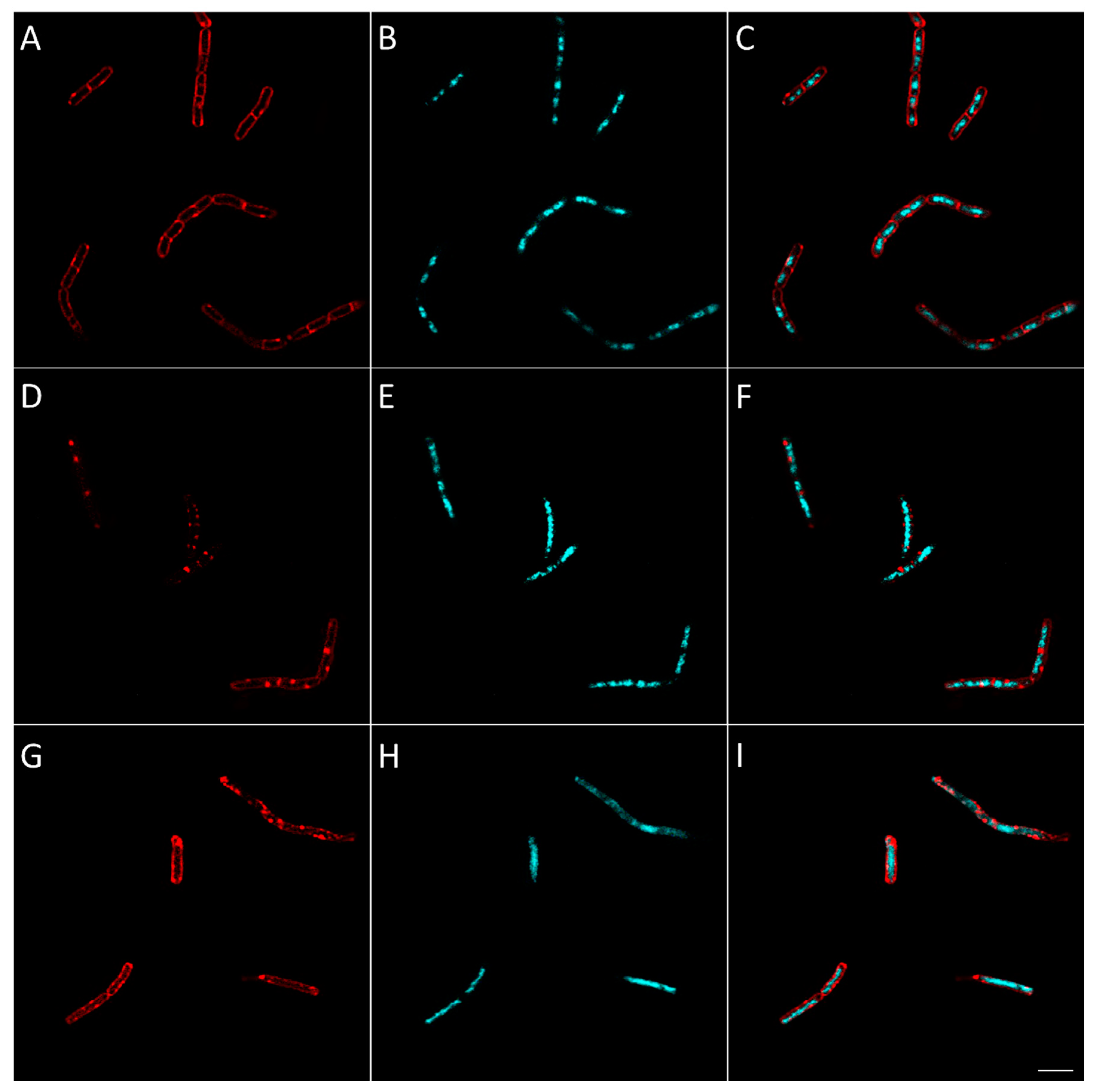
2.2.8. Evaluation of the Effect of Berberine’s Derivatives on the Synthesis of Macromolecules by Bacteria Using Fluorescent Microscopy
3. Results
3.1. Synthesis
3.2. Antibacterial Activity
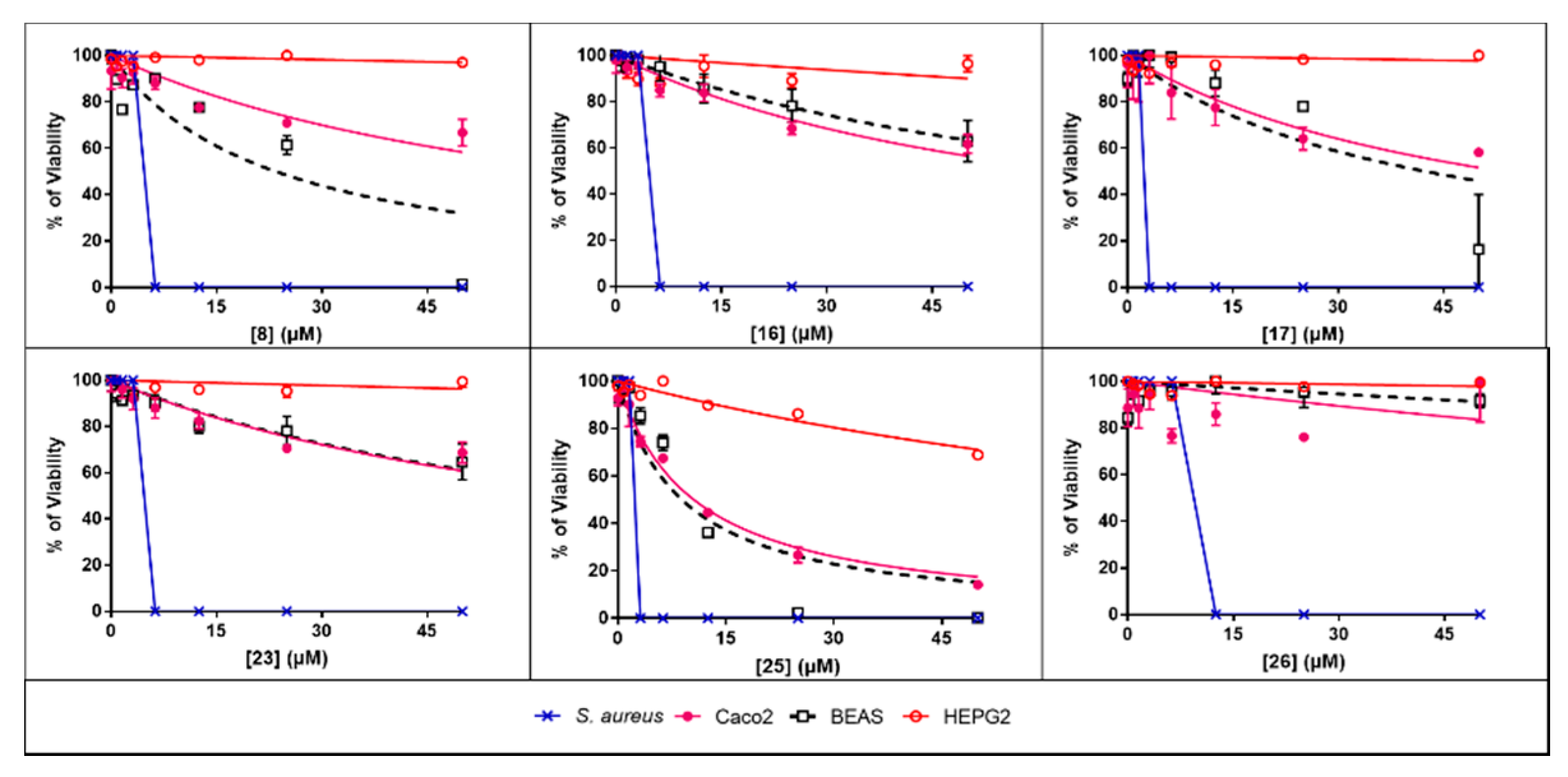
3.3. Toxicity Evaluation
3.4. Mechanistic Analysis
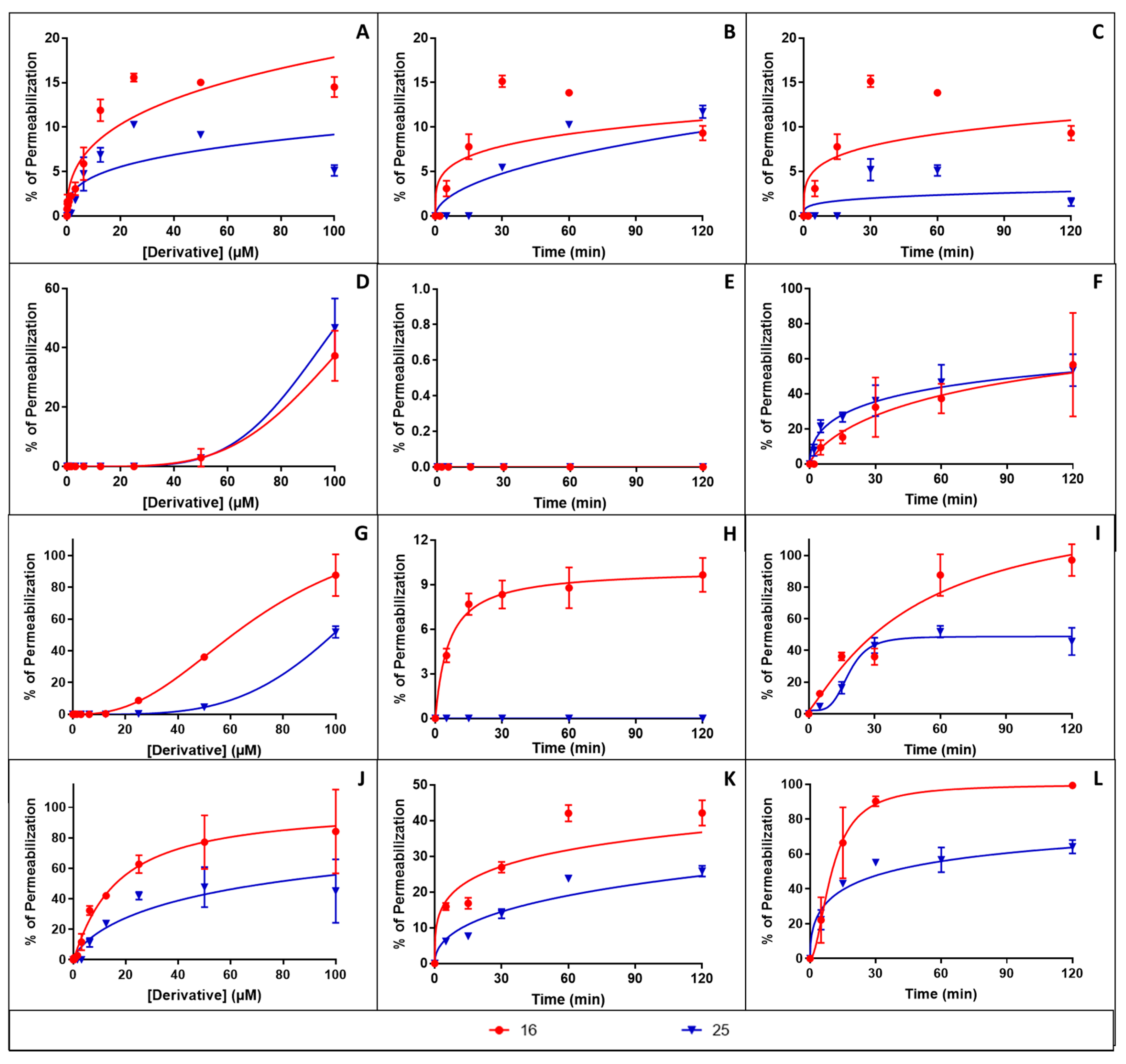
3.4.1. Membrane Permeabilisation Assay
3.4.2. DNA Fragmentation Assay
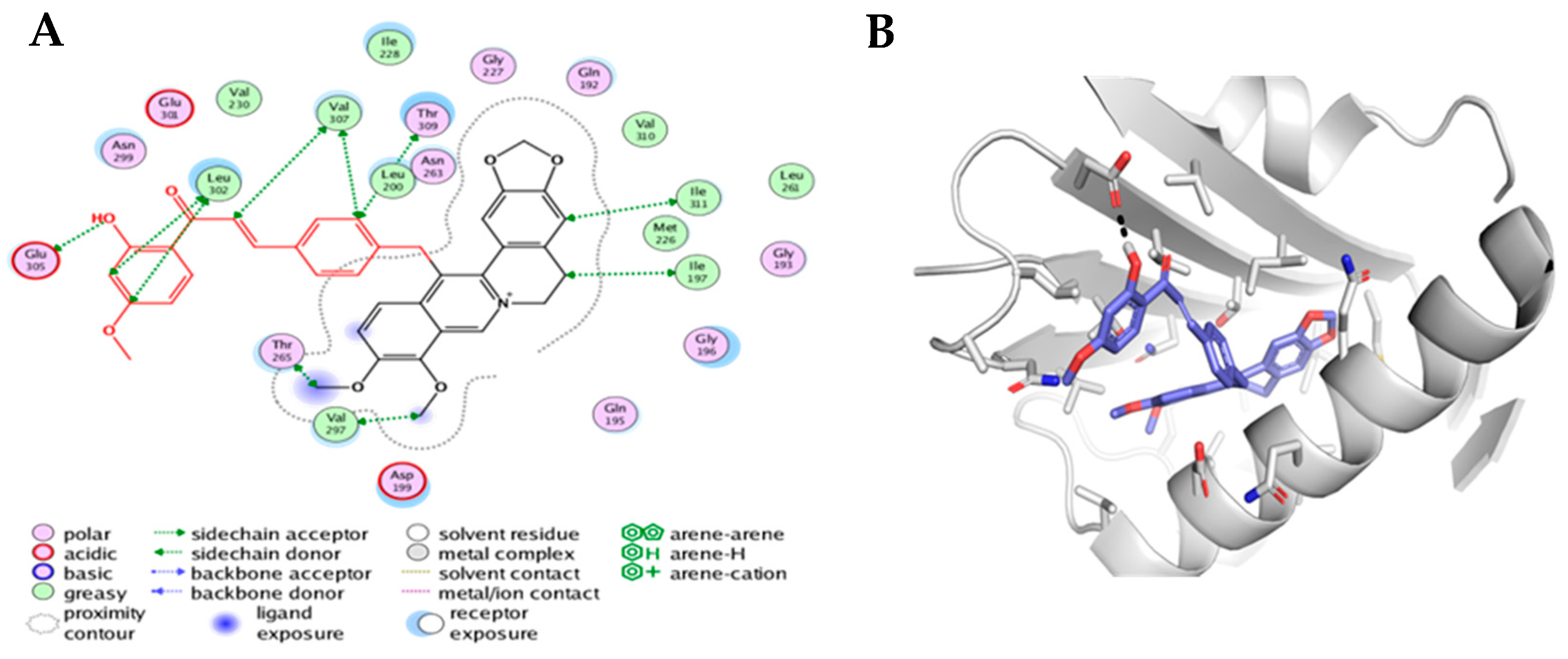
3.4.3. Molecular Docking
3.4.4. Macromolecule Synthesis Inhibition Assay
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Brown, E.D.; Wright, G.D. Antibacterial drug discovery in the resistance era. Nature 2016, 529, 336–343. [Google Scholar] [CrossRef] [PubMed]
- Arendrup, M.C.; Patterson, T.F. Multidrug-Resistant Candida: Epidemiology, Molecular Mechanisms, and Treatment. J. Infect. Dis. 2017, 216, S445–S451. [Google Scholar] [CrossRef] [PubMed]
- Derosa, G.; Maffioli, P. Alkaloids in the nature: Pharmacological applications in clinical practice of berberine and mate tea. Curr. Top. Med. Chem. 2014, 14, 200–206. [Google Scholar] [CrossRef] [PubMed]
- Xiao, D.; Liu, Z.; Zhang, S.; Zhou, M.; He, F.; Zou, M.; Peng, J.; Xie, X.; Liu, Y.; Peng, D. Berberine Derivatives with Different Pharmacological Activities via Structural Modifications. Mini Rev. Med. Chem. 2018, 18, 1424–1441. [Google Scholar] [CrossRef] [PubMed]
- Iwasa, K.; Kim, H.-S.; Wataya, Y.; Lee, D.-U. Antimalarial activity and structure-activity relationships of protoberberine alkaloids. Eur. J. Med. Chem. 1998, 33, 65–69. [Google Scholar] [CrossRef]
- Vennerstrom, J.L.; Lovelace, J.K.; Waits, V.B.; Hanson, W.L.; Klayman, D.L. Berberine derivatives as antileishmanial drugs. Antimicrob. Agents Chemother. 1990, 34, 918–921. [Google Scholar] [CrossRef]
- Wang, Y.-X.; Yang, L.; Wang, H.-Q.; Zhao, X.-Q.; Liu, T.; Li, Y.-H.; Zeng, Q.-X.; Li, Y.-H.; Song, D.-Q. Synthesis and Evolution of Berberine Derivatives as a New Class of Antiviral Agents against Enterovirus 71 through the MEK/ERK Pathway and Autophagy. Molecules 2018, 23, 2084. [Google Scholar] [CrossRef]
- Bodiwala, H.S.; Sabde, S.; Mitra, D.; Bhutani, K.K.; Singh, I.P. Synthesis of 9-substituted derivatives of berberine as anti-HIV agents. Eur. J. Med. Chem. 2011, 46, 1045–1049. [Google Scholar] [CrossRef]
- Yu, P.; Li, D.; Ni, J.; Zhao, L.; Ding, G.; Wang, Z.; Xiao, W. Predictive QSAR modeling study on berberine derivatives with hypolipidemic activity. Chem. Biol. Drug Des. 2018, 91, 867–873. [Google Scholar] [CrossRef]
- Xiao, D.; He, F.; Peng, D.; Zou, M.; Peng, J.; Liu, P.; Liu, Y.; Liu, Z. Synthesis and Anticancer Activity of 9-O-Pyrazole Alkyl Substituted Berberine Derivatives. Anticancer Agents Med. Chem. 2018, 18, 1639–1648. [Google Scholar] [CrossRef]
- Vieira, S.; Castelli, S.; Falconi, M.; Takarada, J.; Fiorillo, G.; Buzzetti, F.; Lombardi, P.; Desideri, A. Role of 13-(di)phenylalkyl berberine derivatives in the modulation of the activity of human topoisomerase IB. Int. J. Biol. Macromol. 2015, 77, 68–75. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Yang, T.; Chen, H.; Xu, Y.-N.; Yu, L.-F.; Liu, T.; Tang, J.; Yi, Z.; Yang, C.-G.; Xue, W.; et al. The synthesis and antistaphylococcal activity of 9, 13-disubstituted berberine derivatives. Eur. J. Med. Chem. 2017, 127, 424–433. [Google Scholar] [CrossRef] [PubMed]
- Fan, T.-Y.; Wang, Y.-X.; Tang, S.; Hu, X.-X.; Zen, Q.-X.; Pang, J.; Yang, Y.-S.; You, X.-F.; Song, D.-Q. Synthesis and antibacterial evaluation of 13-substituted cycloberberine derivatives as a novel class of anti-MRSA agents. Eur. J. Med. Chem. 2018, 157, 877–886. [Google Scholar] [CrossRef] [PubMed]
- Park, K.D.; Lee, J.H.; Kim, S.H.; Kang, T.H.; Moon, J.S.; Kim, S.U. Synthesis of 13-(substituted benzyl) berberine and berberrubine derivatives as antifungal agents. Bioorg. Med. Chem. Lett. 2006, 16, 3913–3916. [Google Scholar] [CrossRef]
- Olleik, H.; Yahiaoui, S.; Roulier, B.; Courvoisier-Dezord, E.; Perrier, J.; Pérès, B.; Hijazi, A.; Baydoun, E.; Raymond, J.; Boumendjel, A.; et al. Aurone derivatives as promising antibacterial agents against resistant Gram-positive pathogens. Eur. J. Med. Chem. 2019, 165, 133–141. [Google Scholar] [CrossRef]
- Olleik, H.; Nicoletti, C.; Lafond, M.; Courvoisier-Dezord, E.; Xue, P.; Hijazi, A.; Baydoun, E.; Perrier, J.; Maresca, M. Comparative Structure–Activity Analysis of the Antimicrobial Activity, Cytotoxicity, and Mechanism of Action of the Fungal Cyclohexadepsipeptides Enniatins and Beauvericin. Toxins 2019, 11, 514. [Google Scholar] [CrossRef]
- Chiumento, S.; Roblin, C.; Kieffer-Jaquinod, S.; Tachon, S.; Leprètre, C.; Basset, C.; Aditiyarini, D.; Olleik, H.; Nicoletti, C.; Bornet, O.; et al. Ruminococcin C, a promising antibiotic produced by a human gut symbiont. Sci. Adv. 2019, 5, eaaw9969. [Google Scholar] [CrossRef] [PubMed]
- Benkhaled, B.T.; Hadiouch, S.; Olleik, H.; Perrier, J.; Ysacco, C.; Guillaneuf, Y.; Gigmes, D.; Maresca, M.; Lefay, C. Elaboration of antimicrobial polymeric materials by dispersion of well-defined amphiphilic methacrylic SG1-based copolymers. Polym. Chem. 2018, 9, 3127–3141. [Google Scholar] [CrossRef]
- Oyama, L.B.; Girdwood, S.E.; Cookson, A.R.; Fernandez-Fuentes, N.; Privé, F.; Vallin, H.E.; Wilkinson, T.J.; Golyshin, P.N.; Golyshina, O.V.; Mikut, R.; et al. The rumen microbiome: An underexplored resource for novel antimicrobial discovery. NPJ Biofilms Microbiomes 2017, 3, 33. [Google Scholar] [CrossRef]
- Borie, C.; Mondal, S.; Arif, T.; Briand, M.; Lingua, H.; Dumur, F.; Gigmes, D.; Stocker, P.; Barbarat, B.; Robert, V.; et al. Enediynes bearing polyfluoroaryl sulfoxide as new antiproliferative agents with dual targeting of microtubules and DNA. Eur. J. Med. Chem. 2018, 148, 306–313. [Google Scholar] [CrossRef]
- Razafimanjato, H.; Benzaria, A.; Taïeb, N.; Guo, X.-J.; Vidal, N.; Di Scala, C.; Varini, K.; Maresca, M. The ribotoxin deoxynivalenol affects the viability and functions of glial cells. Glia 2011, 59, 1672–1683. [Google Scholar] [CrossRef]
- Olleik, H.; Baydoun, E.; Perrier, J.; Hijazi, A.; Raymond, J.; Manzoni, M.; Dupuis, L.; Pauleau, G.; Goudard, Y.; de La Villéon, B.; et al. Temporin-SHa and Its Analogs as Potential Candidates for the Treatment of Helicobacter pylori. Biomolecules 2019, 9, 598. [Google Scholar] [CrossRef]
- Di Pasquale, E.; Salmi-Smail, C.; Brunel, J.-M.; Sanchez, P.; Fantini, J.; Maresca, M. Biophysical studies of the interaction of squalamine and other cationic amphiphilic molecules with bacterial and eukaryotic membranes: Importance of the distribution coefficient in membrane selectivity. Chem. Phys. Lipids 2010, 163, 131–140. [Google Scholar] [CrossRef]
- Tan, C.M.; Therien, A.G.; Lu, J.; Lee, S.H.; Caron, A.; Gill, C.J.; Lebeau-Jacob, C.; Benton-Perdomo, L.; Monteiro, J.M.; Pereira, P.M.; et al. Restoring methicillin-resistant Staphylococcus aureus susceptibility to β-lactam antibiotics. Sci. Transl. Med. 2012, 4, 126ra35. [Google Scholar] [CrossRef]
- Korb, O.; Stützle, T.; Exner, T.E. Empirical scoring functions for advanced protein-ligand docking with PLANTS. J. Chem. Inf. Model. 2009, 49, 84–96. [Google Scholar] [CrossRef]
- Giordano, A.; del Gaudio, F.; Johansson, C.; Riccio, R.; Oppermann, U.; Di Micco, S. Virtual Fragment Screening Identification of a Quinoline-5,8-dicarboxylic Acid Derivative as a Selective JMJD3 Inhibitor. Chemmedchem 2018, 13, 1160–1164. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Cheng, X.; Swails, J.M.; Yeom, M.S.; Eastman, P.K.; Lemkul, J.A.; Wei, S.; Buckner, J.; Jeong, J.C.; Qi, Y.; et al. CHARMM-GUI Input Generator for NAMD, GROMACS, AMBER, OpenMM, and CHARMM/OpenMM Simulations Using the CHARMM36 Additive Force Field. J. Chem. Theory Comput. 2016, 12, 405–413. [Google Scholar] [CrossRef] [PubMed]
- Jo, S.; Kim, T.; Iyer, V.G.; Im, W. CHARMM-GUI: A web-based graphical user interface for CHARMM. J. Comput. Chem. 2008, 29, 1859–1865. [Google Scholar] [CrossRef]
- Chu, M.H.; Xiao, R.; Yin, Y.; Wang, X.; Chu, Z.; Zhang, M.; Ding, R.; Wang, Y. Berberine: A Medicinal Compound for the Treatment of Bacterial Infections. Clin. Microbiol. 2014. [Google Scholar] [CrossRef]
- Sun, N.; Chan, F.-Y.; Lu, Y.-J.; Neves, M.A.C.; Lui, H.-K.; Wang, Y.; Chow, K.-Y.; Chan, K.-F.; Yan, S.-C.; Leung, Y.-C.; et al. Rational Design of Berberine-Based FtsZ Inhibitors with Broad-Spectrum Antibacterial Activity. PLoS ONE 2014, 9, e97514. [Google Scholar] [CrossRef] [PubMed]
- Testerman, T.L.; Morris, J. Beyond the stomach: An updated view of Helicobacter pylori pathogenesis, diagnosis, and treatment. World J. Gastroenterol. WJG 2014, 20, 12781–12808. [Google Scholar] [CrossRef]
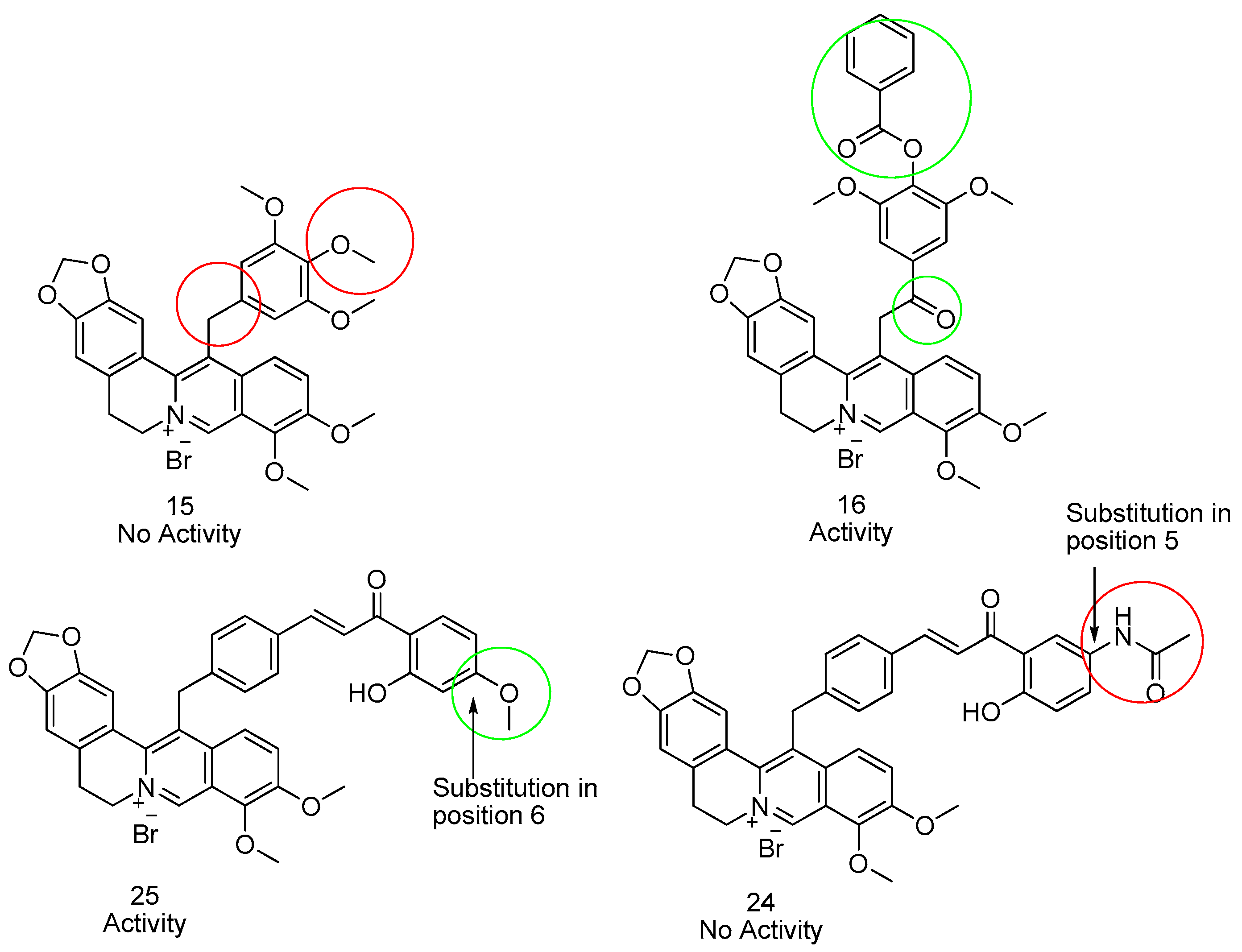
| Compound | S. aureus | B. subtilis | E. coli | P. aeruginosa | M. smegmatis | C. albicans |
|---|---|---|---|---|---|---|
| Amphotericin B | - | - | - | - | - | 1.1 |
| Berberine | >100 | >100 | >100 | >100 | 100 | 100 |
| Gemifloxacin | 0.06 | 0.02 | 0.06 | 0.25 | 2 | - |
| 1 | 100 | >100 | >100 | >100 | >100 | 100 |
| 2 | 12.5–25 | 50 | >100 | >100 | 25 | >100 |
| 3 | 100 | >100 | >100 | >100 | 100 | 100 |
| 4 | >100 | >100 | >100 | >100 | 100 | >100 |
| 5 | 100 | 100 | >100 | >100 | 50 | >100 |
| 6 | 12.5–25 | 50 | >100 | >100 | 25 | >100 |
| 7 | 100 | >100 | >100 | >100 | 100 | 100 |
| 8 | 6.25 | 6.25 | >100 | >100 | 25 | 50 |
| 9 | 3.125 | 12.5 | >100 | >100 | 6.25 | 50 |
| 10 | >100 | >100 | >100 | >100 | >100 | >100 |
| 11 | 25 | 25 | >100 | >100 | 50 | 100 |
| 12 | 100 | >100 | >100 | >100 | 100 | >100 |
| 13 | 6.25–12.5 | 25 | >100 | >100 | 12.5 | 50 |
| 14 | 25 | 50 | >100 | >100 | 25 | >100 |
| 15 | 100 | >100 | >100 | >100 | 50 | >100 |
| 16 | 6.25–12.5 | 25 | >100 | >100 | 50 | >100 |
| 17 | 6.25–12.5 | 12.5 | >100 | >100 | 100 | 25 |
| 18 | 3.125–6.25 | 12.5 | >100 | >100 | 12.5 | 12.5 |
| 19 | 1.5–3.12 | 1.5 | >100 | >100 | 12.5 | 6.25 |
| 20 | 100 | >100 | >100 | >100 | 100 | 100 |
| 21 | 50 | 100 | >100 | >100 | 50 | >100 |
| 22 | >100 | >100 | >100 | >100 | >100 | >100 |
| 23 | 6.25 | 25 | >100 | >100 | 25 | 100 |
| 24 | 50–100 | 100 | >100 | >100 | 25 | >100 |
| 25 | 3.125 | 6.25 | >100 | >100 | 25 | 12.5 |
| 26 | 12.5 | 50 | >100 | >100 | 50 | 100 |
| Compound | Caco2 | BEAS | HEPG2 |
|---|---|---|---|
| Berberine | 21.56 ± 2.7 | 40.45 ± 5.4 | 526.1 ± 151.9 |
| 2 | 34.18 ± 3.8 | 64.61 ± 4.5 | 392.2 ± 114.5 |
| 6 | 36.91 ± 4.3 | 56.25 ± 5.0 | 376.4 ± 161.4 |
| 8 | 69.90 ± 8.1 | 23.38 ± 4.5 | 1627 ± 855.1 |
| 9 | 24.77 ± 3.6 | 12.79 ± 1.6 | 61.45 ± 10.0 |
| 11 | 90.15 ± 14.3 | 150.80 ± 23.7 | 3648 ± 5425 |
| 13 | 31.43 ± 3.8 | 16.90 ± 1.5 | 197.9 ± 35.0 |
| 14 | 86.14 ± 9.2 | 73.62 ± 8.7 | 2217 ± 2876 |
| 16 | 64.80 ± 4.9 | 86.22 ± 7.6 | 450.9 ± 171 |
| 17 | 53.40 ± 8.4 | 42.41 ± 9.8 | 2138 ± 2243 |
| 18 | 18.26 ± 1.3 | 9.59 ± 1.2 | 60.75 ± 11.6 |
| 19 | 11.95 ± 1.3 | 4.92 ± 0.6 | 31.17 ± 7.1 |
| 23 | 77.52 ± 7.2 | 79.16 ± 7.7 | 1288 ± 711.1 |
| 25 | 10.54 ± 0.7 | 8.87 ± 1.5 | 123.1 ± 9.7 |
| 26 | 253 ± 128.4 | 519 ± 237.2 | 2259 ± 1797 |
| Compound | ||||||
|---|---|---|---|---|---|---|
| 8 | 16 | 17 | 23 | 25 | 26 | |
| Caco2 | 11.18 | 10.36 | 8.54 | 12.40 | 3.37 | 20.24 |
| BEAS | 3.74 | 13.79 | 6.78 | 12.66 | 2.83 | 41.52 |
| HEPG2 | 260.30 | 72.14 | 342.00 | 206.00 | 39.39 | 180.70 |
| Compound | 8 | 16 | 17 | 23 | 25 | 26 | |
|---|---|---|---|---|---|---|---|
| Gram-positive | A. gandavensis | 3.12 | 100 | 12.5 | 50 | 1.5 | 12.5 |
| B. cereus | 12.5 | 12.5 | 25 | 12.5 | 6.25 | 50 | |
| B. subtilis | 6.25 | 25 | 12.5 | 25 | 6.25 | 50 | |
| B. subtilis (Nisin-resistant) | 12.5 | 12.5 | 12.5–25 | 25 | 6.25 | 50 | |
| C. difficile | 100 | 100 | 100 | 50 | 12.5 | >100 | |
| C. perfringens | 100 | 25 | 50 | 50 | 25 | >100 | |
| E. faecalis (Vancomycin-resistant) | 50 | 50 | 25 | 100 | 12.5 | >100 | |
| L. lactis | 3.12–6.25 | 6.25–12.5 | 3.12–6.25 | 6.25 | 1.5–3.12 | 6.25–12.5 | |
| S. aureus | 6.25 | 6.25 | 6.25 | 6.25 | 3.125 | 12.5 | |
| S.aureus (Methicillin-resistant) | 12.5 | 12.5 | 25 | 12.5 | 6.25 | 25 | |
| S. pyogenes | 12.5 | 6.25 | 25 | 6.25 | 6.25 | 25 | |
| Gram-negative | A. baumannii | >100 | >100 | >100 | >100 | >100 | >100 |
| C. farmer | >100 | >100 | >100 | >100 | >100 | >100 | |
| C. rodentium | >100 | >100 | >100 | >100 | >100 | >100 | |
| E. coli | >100 | >100 | >100 | >100 | >100 | >100 | |
| H. pylori | 6.25 | 6.25 | 6.25–12.5 | 6.25 | 3.125 | 50 | |
| K. pneumonia | >100 | >100 | >100 | >100 | >100 | >100 | |
| P. aeruginosa | >100 | >100 | >100 | >100 | >100 | >100 | |
| P. aeruginosa (Quinolone-resistant) | >100 | >100 | >100 | >100 | >100 | >100 | |
| S. enterica | >100 | >100 | >100 | >100 | >100 | >100 | |
| S. flexneri | 50 | >100 | >100 | >100 | >100 | >100 | |
| V. diabolicus | >100 | >100 | >100 | >100 | >100 | >100 | |
| V. alginolyticus | 6.25 | 3.12 | 1.5–3.12 | 6.25 | 3.12 | 50 |
| Condition | Known/Possible Target | Observed Morphology |
|---|---|---|
| Control | - |
|
| Amoxicillin | Cell wall biosynthesis |
|
| Gemifloxacin | DNA synthesis |
|
| Rifampicin | RNA biosynthesis |
|
| Tetracycline | Protein biosynthesis |
|
| 16 | Cell wall biosynthesis |
|
| 25 | RNA biosynthesis |
|
| Condition | Known/Possible Target | Observed Morphology |
|---|---|---|
| Control | - |
|
| Amoxicillin | Cell wall biosynthesis |
|
| Gemifloxacin | DNA synthesis |
|
| Rifampicin | RNA biosynthesis |
|
| Tetracycline | Protein biosynthesis |
|
| 16 | Cell wall biosynthesis |
|
| 25 | RNA biosynthesis |
|
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Olleik, H.; Yacoub, T.; Hoffer, L.; Gnansounou, S.M.; Benhaiem-Henry, K.; Nicoletti, C.; Mekhalfi, M.; Pique, V.; Perrier, J.; Hijazi, A.; et al. Synthesis and Evaluation of the Antibacterial Activities of 13-Substituted Berberine Derivatives. Antibiotics 2020, 9, 381. https://doi.org/10.3390/antibiotics9070381
Olleik H, Yacoub T, Hoffer L, Gnansounou SM, Benhaiem-Henry K, Nicoletti C, Mekhalfi M, Pique V, Perrier J, Hijazi A, et al. Synthesis and Evaluation of the Antibacterial Activities of 13-Substituted Berberine Derivatives. Antibiotics. 2020; 9(7):381. https://doi.org/10.3390/antibiotics9070381
Chicago/Turabian StyleOlleik, Hamza, Taher Yacoub, Laurent Hoffer, Senankpon Martial Gnansounou, Kehna Benhaiem-Henry, Cendrine Nicoletti, Malika Mekhalfi, Valérie Pique, Josette Perrier, Akram Hijazi, and et al. 2020. "Synthesis and Evaluation of the Antibacterial Activities of 13-Substituted Berberine Derivatives" Antibiotics 9, no. 7: 381. https://doi.org/10.3390/antibiotics9070381
APA StyleOlleik, H., Yacoub, T., Hoffer, L., Gnansounou, S. M., Benhaiem-Henry, K., Nicoletti, C., Mekhalfi, M., Pique, V., Perrier, J., Hijazi, A., Baydoun, E., Raymond, J., Piccerelle, P., Maresca, M., & Robin, M. (2020). Synthesis and Evaluation of the Antibacterial Activities of 13-Substituted Berberine Derivatives. Antibiotics, 9(7), 381. https://doi.org/10.3390/antibiotics9070381