In Vitro Activity of Essential Oils Against Planktonic and Biofilm Cells of Extended-Spectrum β-Lactamase (ESBL)/Carbapenamase-Producing Gram-Negative Bacteria Involved in Human Nosocomial Infections
Abstract
1. Introduction
2. Results
2.1. Phenotypic Identification of ESBLs and Carbapenamase
2.2. Polymerase Chain Reaction and Sequencing of ESBL and Carbapenemase Genes
2.3. Antibacterial Activity Evaluation of the Essential Oils
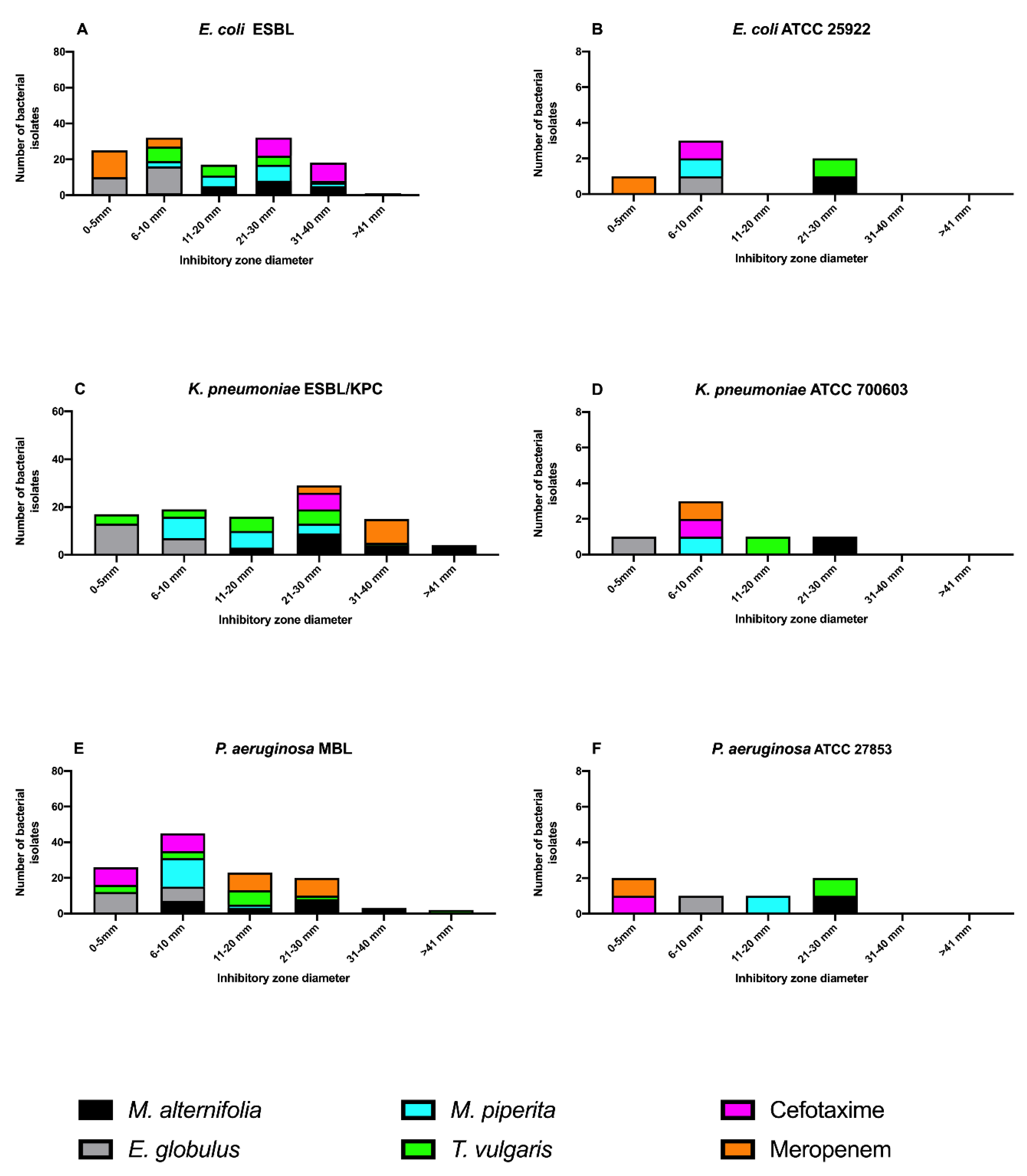
2.3.1. Agar Disk Diffusion Assay
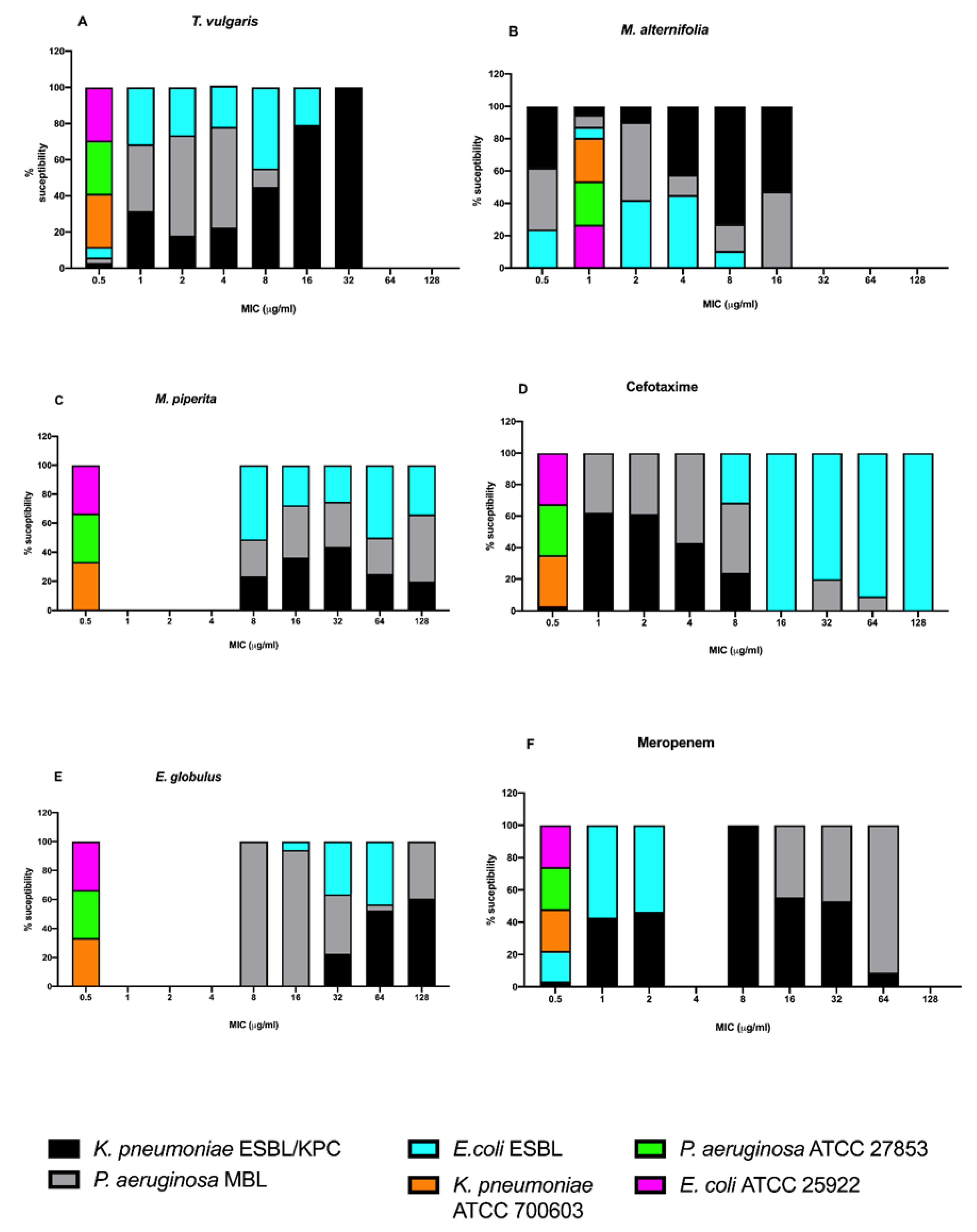
2.3.2. Minimal Inhibitory Concentration (MIC)
2.4. Biofilm Assay
3. Discussion
4. Materials and Methods
4.1. Bacterial Strains and Phenotypic Identification
4.2. Polymerase Chain Reaction and Sequencing of ESBL and Carbapenemase Genes
4.3. Antibacterial Activity Evaluation of the Essential Oils
4.3.1. Agar Disk Diffusion Assay
4.3.2. Minimum Inhibitory Concentration (MIC) Determination
4.4. Essential Oils’ Activity on Biofilm
4.5. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Zaman, S.B.; Hussain, M.A.; Nye, R.; Mehta, V.; Mamun, K.T.; Hossain, N. A Review on Antibiotic Resistance: Alarm Bells are Ringing. Cureus 2017, 9, e1403. [Google Scholar] [CrossRef] [PubMed]
- Iseppi, R.; Di Cerbo, A.; Messi, P.; Sabia, C. Antibiotic Resistance and Virulence Traits in Vancomycin-Resistant Enterococci (VRE) and Extended-Spectrum beta-Lactamase/AmpC-producing (ESBL/AmpC) Enterobacteriaceae from Humans and Pets. Antibiotics (Basel) 2020, 9. [Google Scholar] [CrossRef]
- Di Cerbo, A.; Canello, S.; Guidetti, G.; Laurino, C.; Palmieri, B. Unusual antibiotic presence in gym trained subjects with food intolerance; a case report. Nutr. Hosp. 2014, 30, 395–398. [Google Scholar] [CrossRef]
- Palmieri, B.; Di Cerbo, A.; Laurino, C. Antibiotic treatments in zootechnology and effects induced on the food chain of domestic species and, comparatively, the human specie. Nutr. Hosp. 2014, 29, 1427–1433. [Google Scholar] [CrossRef] [PubMed]
- Falagas, M.E.; Koletsi, P.K.; Bliziotis, I.A. The diversity of definitions of multidrug-resistant (MDR) and pandrug-resistant (PDR) Acinetobacter baumannii and Pseudomonas aeruginosa. J. Med. Microbiol. 2006, 55, 1619–1629. [Google Scholar] [CrossRef] [PubMed]
- Magiorakos, A.P.; Srinivasan, A.; Carey, R.B.; Carmeli, Y.; Falagas, M.E.; Giske, C.G.; Harbarth, S.; Hindler, J.F.; Kahlmeter, G.; Olsson-Liljequist, B.; et al. Multidrug-resistant, extensively drug-resistant and pandrug-resistant bacteria: An international expert proposal for interim standard definitions for acquired resistance. Clin. Microbiol. Infect 2012, 18, 268–281. [Google Scholar] [CrossRef] [PubMed]
- Putman, M.; van Veen, H.W.; Konings, W.N. Molecular properties of bacterial multidrug transporters. Microbiol. Mol. Biol. Rev. 2000, 64, 672–693. [Google Scholar] [CrossRef]
- Tapia-Rodriguez, M.R.; Bernal-Mercado, A.T.; Gutierrez-Pacheco, M.M.; Vazquez-Armenta, F.J.; Hernandez-Mendoza, A.; Gonzalez-Aguilar, G.A.; Martinez-Tellez, M.A.; Nazzaro, F.; Ayala-Zavala, J.F. Virulence of Pseudomonas aeruginosa exposed to carvacrol: Alterations of the Quorum sensing at enzymatic and gene levels. J. Cell Commun. Signal 2019, 13, 531–537. [Google Scholar] [CrossRef]
- Maita, P.; Boonbumrung, K. Association between biofilm formation of Pseudomonas aeruginosa clinical isolates versus antibiotic resistance and genes involved with biofilm. J. Chem. Pharm. Res. 2014, 6, 1022–1028. [Google Scholar]
- Macia, M.D.; Rojo-Molinero, E.; Oliver, A. Antimicrobial susceptibility testing in biofilm-growing bacteria. Clin. Microbiol. Infect 2014, 20, 981–990. [Google Scholar] [CrossRef]
- Singh, S.; Singh, S.K.; Chowdhury, I.; Singh, R. Understanding the Mechanism of Bacterial Biofilms Resistance to Antimicrobial Agents. Open Microbiol. J. 2017, 11, 53–62. [Google Scholar] [CrossRef] [PubMed]
- Burt, S. Essential oils: Their antibacterial properties and potential applications in foods—A review. Int. J. Food Microbiol. 2004, 94, 223–253. [Google Scholar] [CrossRef] [PubMed]
- Silva, N.; Fernandes Júnior, A. Biological properties of medicinal plants: A review of their antimicrobial activity. J. Venom. Anim. Toxins Incl. Tropical Dis. 2010, 16, 402–413. [Google Scholar] [CrossRef]
- Pei, R.S.; Zhou, F.; Ji, B.P.; Xu, J. Evaluation of combined antibacterial effects of eugenol, cinnamaldehyde, thymol, and carvacrol against E. coli with an improved method. J. Food Sci. 2009, 74, M379–383. [Google Scholar] [CrossRef] [PubMed]
- Aelenei, P.; Miron, A.; Trifan, A.; Bujor, A.; Gille, E.; Aprotosoaie, A.C. Essential Oils and Their Components as Modulators of Antibiotic Activity against Gram-Negative Bacteria. Medicines (Basel) 2016, 3. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, S.H.; Mohamed, M.S.M.; Khalil, M.S.; Azmy, M.; Mabrouk, M.I. Combination of essential oil and ciprofloxacin to inhibit/eradicate biofilms in multidrug-resistant Klebsiella pneumoniae. J. Appl. Microbiol. 2018, 125, 84–95. [Google Scholar] [CrossRef]
- Nouzha, H.; Souhila, B.; Manel, M.; Yasmine, O.; Lamraoui, A. Screening for antibacterial activity of some essential oils and evaluation of their synergistic effec. Int. J. Biosci. 2018, 12, 292–301. [Google Scholar]
- Sakkas, H.; Gousia, P.; Economou, V.; Sakkas, V.; Petsios, S.; Papadopoulou, C. In vitro antimicrobial activity of five essential oils on multidrug resistant Gram-negative clinical isolates. J. Intercult. Ethnopharmacol. 2016, 5, 212–218. [Google Scholar] [CrossRef]
- Valdivieso-Ugarte, M.; Gomez-Llorente, C.; Plaza-Diaz, J.; Gil, A. Antimicrobial, Antioxidant, and Immunomodulatory Properties of Essential Oils: A Systematic Review. Nutrients 2019, 11. [Google Scholar] [CrossRef]
- Iseppi, R.; Brighenti, V.; Licata, M.; Lambertini, A.; Sabia, C.; Messi, P.; Pellati, F.; Benvenuti, S. Chemical Characterization and Evaluation of the Antibacterial Activity of Essential Oils from Fibre-Type Cannabis sativa L. (Hemp). Molecules 2019, 24. [Google Scholar] [CrossRef]
- Condo, C.; Anacarso, I.; Sabia, C.; Iseppi, R.; Anfelli, I.; Forti, L.; de Niederhausern, S.; Bondi, M.; Messi, P. Antimicrobial activity of spices essential oils and its effectiveness on mature biofilms of human pathogens. Nat. Prod. Res. 2020, 34, 567–574. [Google Scholar] [CrossRef] [PubMed]
- Yap, P.S.; Lim, S.H.; Hu, C.P.; Yiap, B.C. Combination of essential oils and antibiotics reduce antibiotic resistance in plasmid-conferred multidrug resistant bacteria. Phytomedicine 2013, 20, 710–713. [Google Scholar] [CrossRef] [PubMed]
- Cheesman, M.J.; Ilanko, A.; Blonk, B.; Cock, I.E. Developing New Antimicrobial Therapies: Are Synergistic Combinations of Plant Extracts/Compounds with Conventional Antibiotics the Solution? Pharmacogn. Rev. 2017, 11, 57–72. [Google Scholar] [CrossRef] [PubMed]
- Safaei-Ghomi, J.; Ahd, A.A. Antimicrobial and antifungal properties of the essential oil and methanol extracts of Eucalyptus largiflorens and Eucalyptus intertexta. Pharmacogn. Mag. 2010, 6, 172–175. [Google Scholar] [CrossRef]
- Ait Said, L.; Zahlane, K.; Ghalbane, I.; El Messoussi, S.; Romane, A.; Cavaleiro, C.; Salgueiro, L. Chemical composition and antibacterial activity of Lavandula coronopifolia essential oil against antibiotic-resistant bacteria. Nat. Prod. Res. 2015, 29, 582–585. [Google Scholar] [CrossRef]
- Predoi, D.; Iconaru, S.L.; Buton, N.; Badea, M.L.; Marutescu, L. Antimicrobial Activity of New Materials Based on Lavender and Basil Essential Oils and Hydroxyapatite. Nanomaterials (Basel) 2018, 8. [Google Scholar] [CrossRef]
- Borges, A.; Lopez-Romero, J.C.; Oliveira, D.; Giaouris, E.; Simões, M. Prevention, removal and inactivation of Escherichia coli and Staphylococcus aureus biofilms using selected monoterpenes of essential oils. J. Appl. Microbiol. 2017, 123, 104–115. [Google Scholar] [CrossRef]
- El-Shouny, W.A.; Ali, S.S.; Sun, J.; Samy, S.M.; Ali, A. Drug resistance profile and molecular characterization of extended spectrum beta-lactamase (ESbetaL)-producing Pseudomonas aeruginosa isolated from burn wound infections. Essential oils and their potential for utilization. Microb. Pathog 2018, 116, 301–312. [Google Scholar] [CrossRef]
- Vazquez-Armenta, F.J.; Hernandez-Onate, M.A.; Martinez-Tellez, M.A.; Lopez-Zavala, A.A.; Gonzalez-Aguilar, G.A.; Gutierrez-Pacheco, M.M.; Ayala-Zavala, J.F. Quercetin repressed the stress response factor (sigB) and virulence genes (prfA, actA, inlA, and inlC), lower the adhesion, and biofilm development of L. monocytogenes. Food Microbiol. 2020, 87, 103377. [Google Scholar] [CrossRef]
- Ortega-Ramirez, L.A.; Gutiérrez-Pacheco, M.M.; Vargas-Arispuro, I.; González-Aguilar, G.A.; Martínez-Téllez, M.A.; Ayala-Zavala, J.F. Inhibition of Glucosyltransferase Activity and Glucan Production as an Antibiofilm Mechanism of Lemongrass Essential Oil against Escherichia coli O157:H7. Antibiotics 2020, 9, 102. [Google Scholar] [CrossRef]
- Quave, C.L.; Horswill, A.R. Flipping the switch: Tools for detecting small molecule inhibitors of staphylococcal virulence. Front Microbiol. 2014, 5, 706. [Google Scholar] [CrossRef]
- Carson, C.F.; Hammer, K.A.; Riley, T.V. Melaleuca alternifolia (Tea Tree) oil: A review of antimicrobial and other medicinal properties. Clin. Microbiol. Rev. 2006, 19, 50–62. [Google Scholar] [CrossRef] [PubMed]
- Oliva, A.; Costantini, S.; De Angelis, M.; Garzoli, S.; Bozovic, M.; Mascellino, M.T.; Vullo, V.; Ragno, R. High Potency of Melaleuca alternifolia Essential Oil against Multi-Drug Resistant Gram-Negative Bacteria and Methicillin-Resistant Staphylococcus aureus. Molecules 2018, 23. [Google Scholar] [CrossRef] [PubMed]
- Ferrini, A.M.; Mannoni, V.; Aureli, P.; Salvatore, G.; Piccirilli, E.; Ceddia, T.; Pontieri, E.; Sessa, R.; Oliva, B. Melaleuca alternifolia essential oil possesses potent anti-staphylococcal activity extended to strains resistant to antibiotics. Int. J. Immunopathol. Pharmacol. 2006, 19, 539–544. [Google Scholar] [CrossRef] [PubMed]
- Sienkiewicz, M.; Kowalczyk, E.; Wasiela, M. Recent Patents Regarding Essential Oils and the Significance of their Con-stituents in Human Health and Treatment. Recent Patents Anti-Infective Drug Discov. 2012, 7, 133–140. [Google Scholar] [CrossRef] [PubMed]
- Chouhan, S.; Sharma, K.; Guleria, S. Antimicrobial Activity of Some Essential Oils-Present Status and Future Perspectives. Medicines (Basel) 2017, 4. [Google Scholar] [CrossRef]
- Warnke, P.H.; Lott, A.J.; Sherry, E.; Wiltfang, J.; Podschun, R. The ongoing battle against multi-resistant strains: In-vitro inhibition of hospital-acquired MRSA, VRE, Pseudomonas, ESBL E. coli and Klebsiella species in the presence of plant-derived antiseptic oils. J. Craniomaxillofac Surg. 2013, 41, 321–326. [Google Scholar] [CrossRef]
- De Billerbeck, V.G. Huiles essentielles et bactéries résistantes aux antibiotiques. Phytothérapie 2007, 5, 249–253. [Google Scholar] [CrossRef]
- El Asbahani, A.; Miladi, K.; Badri, W.; Sala, M.; Ait Addi, E.H.; Casabianca, H.; El Mousadik, A.; Hartmann, D.; Jilale, A.; Renaud, F.N.; et al. Essential oils: From extraction to encapsulation. Int. J. Pharm. 2015, 483, 220–243. [Google Scholar] [CrossRef]
- Benameur, Q.; Gervasi, T.; Pellizzeri, V.; Pľuchtová, M.; Tali-Maama, H.; Assaous, F.; Guettou, B.; Rahal, K.; Gruľová, D.; Dugo, G.; et al. Antibacterial activity of Thymus vulgaris essential oil alone and in combination with cefotaxime against blaESBL producing multidrug resistant Enterobacteriaceae isolates. Nat. Prod. Res. 2019, 33, 2647–2654. [Google Scholar] [CrossRef]
- CLSI. Performance Standards for Antimicrobial Susceptibility Testing, 29th ed.; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2019; Volume 39. [Google Scholar]
- Perez-Perez, F.J.; Hanson, N.D. Detection of plasmid-mediated AmpC beta-lactamase genes in clinical isolates by using multiplex PCR. J. Clin. Microbiol. 2002, 40, 2153–2162. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Jeon, S.; Lee, B.; Park, M.; Lee, H.; Lee, J.; Kim, S. Rapid Detection of Extended Spectrum β-Lactamase (ESBL) for Enterobacteriaceae by use of a Multiplex PCR-based Method. Infect Chemother. 2009, 41, 181–184. [Google Scholar] [CrossRef]
- Nordmann, P.; Poirel, L.; Carrer, A.; Toleman, M.A.; Walsh, T.R. How to detect NDM-1 producers. J. Clin. Microbiol. 2011, 49, 718–721. [Google Scholar] [CrossRef] [PubMed]
- Klancnik, A.; Piskernik, S.; Jersek, B.; Mozina, S.S. Evaluation of diffusion and dilution methods to determine the antibacterial activity of plant extracts. J. Microbiol. Methods 2010, 81, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Şahin, F.; Güllüce, M.; Daferera, D.; Sökmen, A.; Sökmen, M.; Polissiou, M.; Agar, G.; Özer, H. Biological activities of the essential oils and methanol extract of Origanum vulgare ssp. vulgare in the Eastern Anatolia region of Turkey. Food Control 2004, 15, 549–557. [Google Scholar] [CrossRef]
- Stepanovic, S.; Vukovic, D.; Hola, V.; Di Bonaventura, G.; Djukic, S.; Cirkovic, I.; Ruzicka, F. Quantification of biofilm in microtiter plates: Overview of testing conditions and practical recommendations for assessment of biofilm production by staphylococci. APMIS 2007, 115, 891–899. [Google Scholar] [CrossRef]
| Strain | Positive | M. alternifolia | T. vulgaris | Meropenem | Cefotaxime |
|---|---|---|---|---|---|
| E. coli 5A | 2.19 ± 0.07 | 0.61 ± 0.11 | 0.56 ± 0.03 * | 0.87 ± 0.03 | 1.92 ± 0.06 |
| E. coli 5M | 2.17 ± 0.01 | 0.76 ± 0.05 | 0.64 ± 0.08 * | 0.93 ± 0.03 | 1.88 ± 0.07 |
| E. coli 5Z | 2.04 ± 0.03 | 0.43 ± 0.10 | 0.43 ± 0.10 | 0.83 ± 0.05 | 1.88 ± 0.04 |
| E. coli 6I | 1.89 ± 0.09 | 0.68 ± 0.01 | 0.44 ± 0.09 * | 0.87 ± 0.07 | 1.86 ± 0.07 |
| E. coli 7B | 1.91 ± 0.06 | 0.25 ± 0.06 * | 0.47 ± 0.11 | 0.85 ± 0.08 | 1.84 ± 0.08 |
| E. coli 7C | 1.92 ± 0.04 | 0.34 ± 0.06 * | 0.45 ± 0.12 | 0.84 ± 0.08 | 1.83 ± 0.11 |
| E. coli 7D | 1.95 ± 0.06 | 0.45 ± 0.07 | 0.23 ± 0.09 * | 0.92 ± 0.08 | 1.89 ± 0.11 |
| E. coli 7E | 1.95 ± 0.06 | 0.45 ± 0.07 | 0.23 ± 0.09 * | 0.92 ± 0.08 | 1.89 ± 0.11 |
| E. coli ATCC 25922 | 2.11 ± 0.09 | 0.58 ± 0.08 * | 0.33 ± 0.08 | 0.55 ± 0.13 | 0.39 ± 0.12 |
| Strain | Positive | M. alternifolia | T. vulgaris | Meropenem | Cefotaxime |
|---|---|---|---|---|---|
| K. pneumoniae 1B | 1.91 ± 0,06 | 0.71 ± 0.05 | 0.27 ± 0.03 | 1.42 ± 0.04 | 1.49 ± 0.05 |
| K. pneumoniae 1C | 1.88 ± 0.10 | 0.61 ± 0.04 * | 0.86 ± 0.04 | 1.45 ± 0.05 | 1.39 ± 0.02 |
| K. pneumoniae 1D | 1.95 ± 0.15 | 0.35 ± 0.11 * | 0.31 ± 0.17 * | 1.16 ± 0.21 | 0.92 ± 0.05 |
| K. pneumoniae 1E | 1.99 ± 0.10 | 0.22 ± 0.09 * | 0.44 ± 0.08 | 0.95 ± 0.13 | 1.00 ± 0.14 |
| K. pneumoniae 1F | 2.01 ± 0.10 | 0.37 ± 0.08 * | 0.54 ± 0.08 | 0.96 ± 0.14 | 0.86 ± 0.10 |
| K. pneumoniae 1G | 2.01 ± 0.11 | 0.55 ± 0.13 * | 0.78 ± 0.03 | 1.79 ± 0.10 | 1.78 ± 0.05 |
| K. pneumoniae 1H | 2.05 ± 0.10 | 0.43 ± 0.07 | 0.21 ± 0.04 * | 1.80 ± 0.07 | 1.68 ± 0.04 |
| K-pneumoniae 1V | 2.91 ± 0.02 | 0.73 ± 0.09 | 0.45 ± 0.06 ** | 0.90 ± 0.08 | 1.04 ± 0.11 |
| K. pneumoniae ATCC 700603 | 2.04 ± 0.07 | 0.19 ± 0.01 | 0.12 ± 0.003 * | 0.17 ± 0.06 | 0.34 ± 0.12 |
| Strain | Positive | M. alternifolia | T. vulgaris | Meropenem | Cefotaxime |
|---|---|---|---|---|---|
| P. aeruginosa 3A | 1.85 ± 0.46 | 0.24 ± 0.12 | 0.35 ± 0.08 | 1.84 ± 0.07 | 1.75 ± 0.09 |
| P. aeruginosa 3B | 2.03 ± 0.07 | 0.26 ± 0.07 | 0.46 ± 0.09 | 2.03 ± 0.06 | 1.92 ± 0.07 |
| P. aeruginosa 3C | 2.15 ± 0.14 | 0.36 ± 0.08 * | 0.45 ± 0.07 | 2.14 ± 0.12 | 2.43 ± 0.15 |
| P. aeruginosa 3D | 2.85 ± 0.13 | 0.22 ± 0.04 * | 0.98 ± 0.07 | 2.73 ± 0.01 | 2.84 ± 0.76 |
| P. aeruginosa 3E | 2.86 ± 0.10 | 0.56 ± 0.10 | 0.35 ± 0.08 * | 2.71 ± 0.09 | 2.84 ± 0.08 |
| P. aeruginosa 3F | 2.59 ± 0.22 | 0.55 ± 0.10 | 0.26 ± 0.08 * | 2.71 ± 0.09 | 2.85 ± 0.08 |
| P. aeruginosa 3M | 2.18 ± 0.42 | 0.50 ± 0.06 | 0.25 ± 0.07 * | 2.44 ± 0.07 | 1.86 ± 0.09 |
| P. aeruginosa 3P | 1.85 ± 0.81 | 0.54 ± 0.08 | 0.28 ± 0.04 * | 2.45 ± 0.08 | 1.74 ± 0.04 |
| P. aeruginosa ATCC 27858 | 2.28 ± 0.09 | 0.55 ± 0.06 | 0.26 ± 0.06 * | 0.67 ± 0.10 | 0.75 ± 0.04 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Iseppi, R.; Di Cerbo, A.; Aloisi, P.; Manelli, M.; Pellesi, V.; Provenzano, C.; Camellini, S.; Messi, P.; Sabia, C. In Vitro Activity of Essential Oils Against Planktonic and Biofilm Cells of Extended-Spectrum β-Lactamase (ESBL)/Carbapenamase-Producing Gram-Negative Bacteria Involved in Human Nosocomial Infections. Antibiotics 2020, 9, 272. https://doi.org/10.3390/antibiotics9050272
Iseppi R, Di Cerbo A, Aloisi P, Manelli M, Pellesi V, Provenzano C, Camellini S, Messi P, Sabia C. In Vitro Activity of Essential Oils Against Planktonic and Biofilm Cells of Extended-Spectrum β-Lactamase (ESBL)/Carbapenamase-Producing Gram-Negative Bacteria Involved in Human Nosocomial Infections. Antibiotics. 2020; 9(5):272. https://doi.org/10.3390/antibiotics9050272
Chicago/Turabian StyleIseppi, Ramona, Alessandro Di Cerbo, Piero Aloisi, Mattia Manelli, Veronica Pellesi, Cinzia Provenzano, Stefania Camellini, Patrizia Messi, and Carla Sabia. 2020. "In Vitro Activity of Essential Oils Against Planktonic and Biofilm Cells of Extended-Spectrum β-Lactamase (ESBL)/Carbapenamase-Producing Gram-Negative Bacteria Involved in Human Nosocomial Infections" Antibiotics 9, no. 5: 272. https://doi.org/10.3390/antibiotics9050272
APA StyleIseppi, R., Di Cerbo, A., Aloisi, P., Manelli, M., Pellesi, V., Provenzano, C., Camellini, S., Messi, P., & Sabia, C. (2020). In Vitro Activity of Essential Oils Against Planktonic and Biofilm Cells of Extended-Spectrum β-Lactamase (ESBL)/Carbapenamase-Producing Gram-Negative Bacteria Involved in Human Nosocomial Infections. Antibiotics, 9(5), 272. https://doi.org/10.3390/antibiotics9050272










