Examining Safety of Biocolourants from Fungal and Plant Sources-Examples from Cortinarius and Tapinella, Salix and Tanacetum spp. and Dyed Woollen Fabrics
Abstract
1. Introduction
1.1. Background
1.2. Safety and Medicinal Use Related to Fungal and Plant Colourants
1.3. Flavonoids and Carotenoids in Tanacetum Vulgare
1.4. Tannins in Salix spp.
1.5. Anthraquinones in Cortinarius Semisanguineus
1.6. Terphenyl Quinones in Tapinella Atrotomentosa
1.7. Objectives
2. Results
2.1. Colourants and Colourfastness
2.2. Toxicity
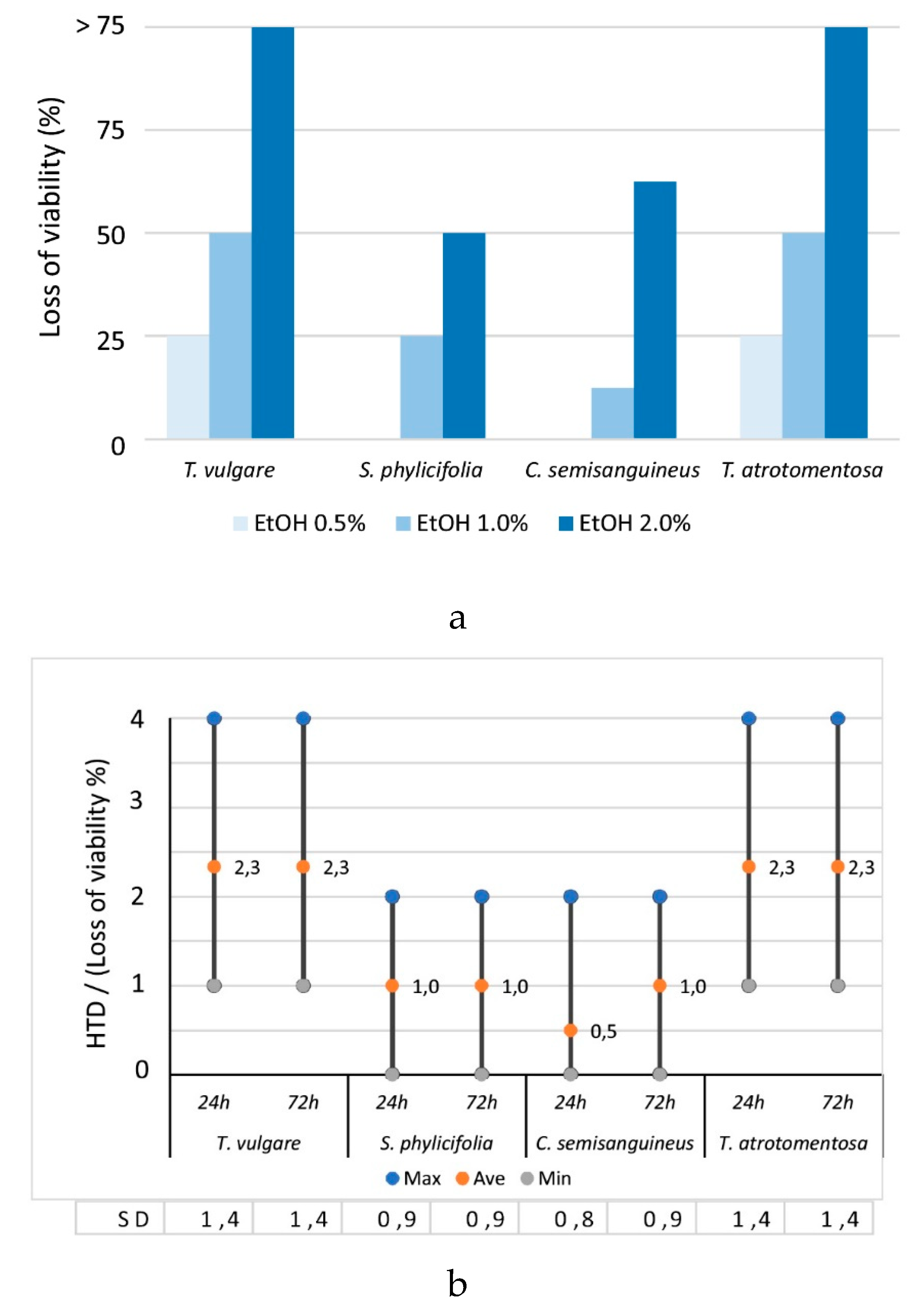
2.2.1. The Effect of EtOH Concentration on the Cell Morphology and Viability
2.2.2. The Toxicity of the Plant and Fungal Materials
2.2.3. The Toxicity of the Undyed Fabrics
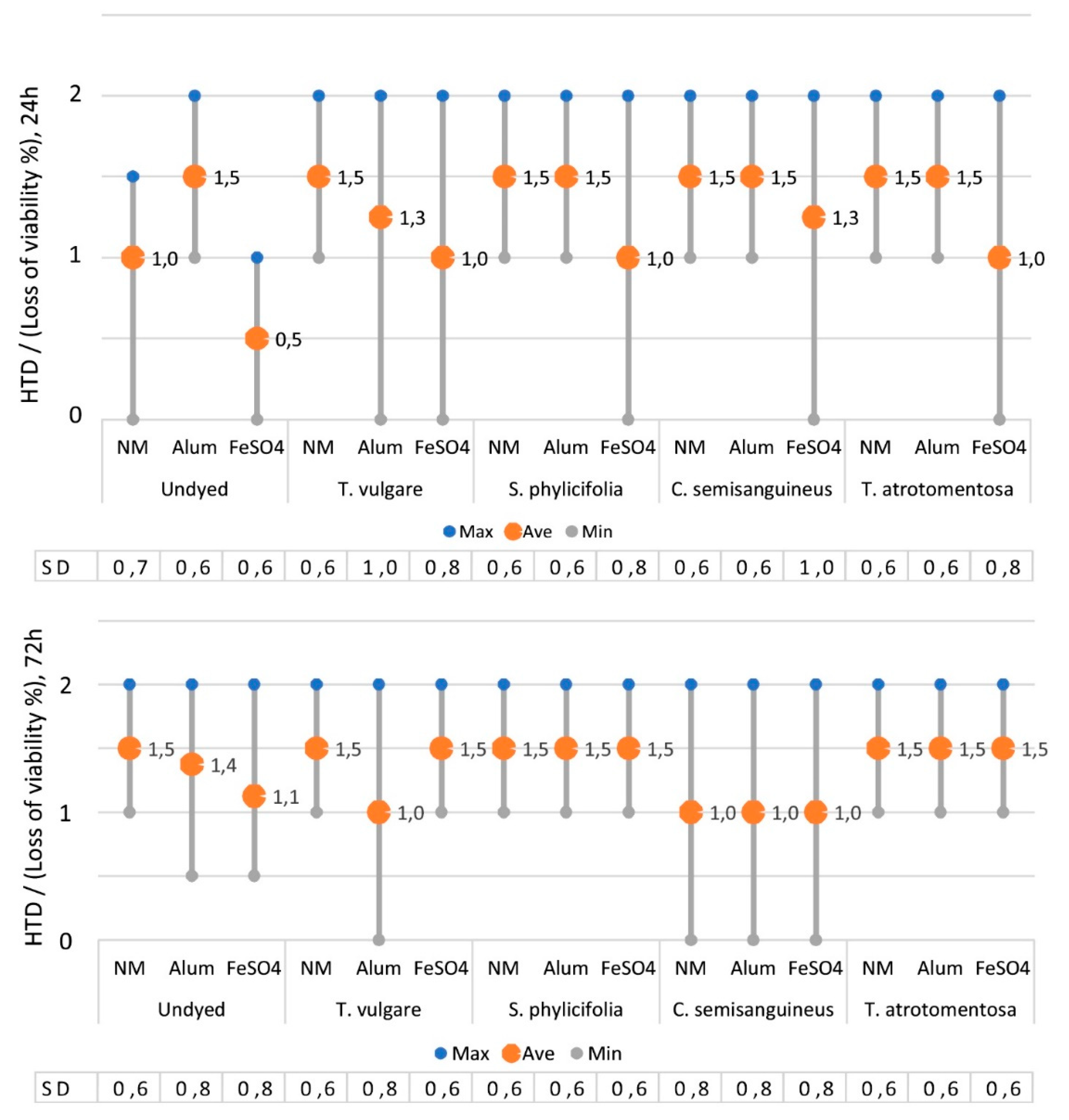
2.2.4. The Toxicity of the Dyed Fabrics
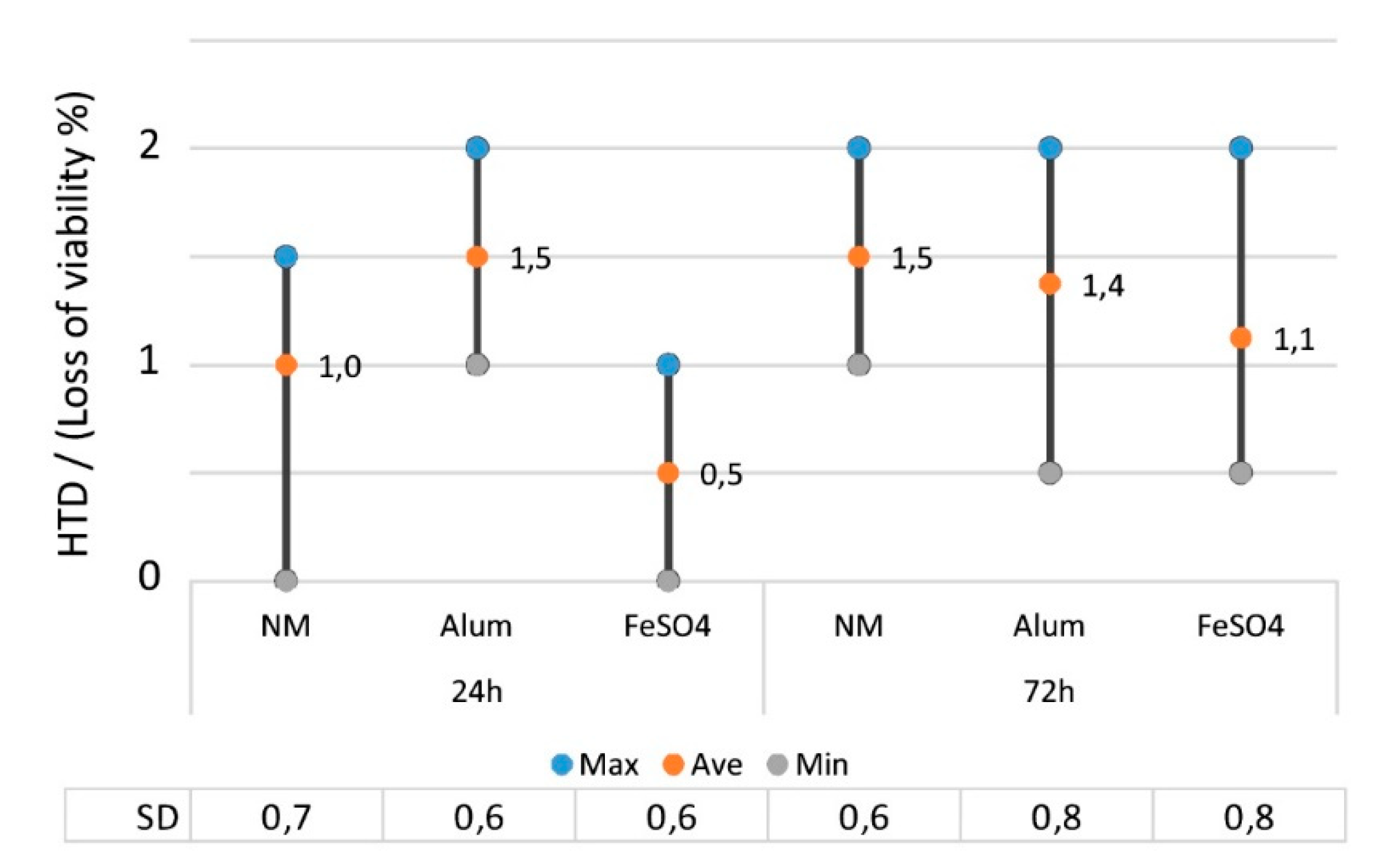
2.2.5. The Effect of Mordant on the Toxicity
3. Discussion
4. Materials and Methods
4.1. Plant and Fungal Material
4.2. Methods for Compound Identification
4.3. Dyeing of Fabric Samples and the Colour Fastness Testing
4.4. Preparation of Plant, Fungal and Fabric Extracts for Toxicity Testing
4.5. Cytotoxicity Assays
4.6. Statistical Analyses
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Holkar, C.R.; Jadhav, A.J.; Pinjari, D.V.; Mahamuni, N.M.; Pandit, A.B. A critical review on textile wastewater treatments: Possible approaches. J. Environ. Manag. 2016, 182, 351–366. [Google Scholar] [CrossRef]
- Hunger, K. (Ed.) Industrial Dyes: Chemistry, Properties, Applications; Wiley: Weinheim, Gemany, 2003. [Google Scholar]
- Leme, D.M.; Primo, F.L.; Gobo, G.G.; Da Costa, C.R.; Tedesco, A.C.; De Oliveira, D.P.J. Genotoxicity assessment of reactive and disperse textile dyes using human dermal equivalent (3D cell culture system). J. Toxicol. Environ. Health A 2015, 78, 466–480. [Google Scholar] [CrossRef]
- Oliveira, G.A.R.D.; Leme, D.M.; De Lapuente, J.; Brito, L.B.; Porredón, C.; Rodrigues, L.D.B.; Brull, N.; Serret, J.T.; Borràs, M.; Disner, G.R.; et al.; et al. A test battery for assessing the ecotoxic effects of textile dyes. Chem. Biol. Interact. 2018, 291, 171–179. [Google Scholar] [CrossRef]
- Tang, A.Y.L.; Lo, C.K.Y.; Kan, C. Textile dyes and human health: A systematic and citation network analysis review. Color. Tehnol. 2018, 134, 245–257. [Google Scholar] [CrossRef]
- Wink, M. Plant secondary metabolism diversity, function and its evolution. Nat. Prod. Commun. 2008, 3, 1205–1216. [Google Scholar] [CrossRef]
- Simmler, C.; Chen, S.-N.; Anderson, J.; Lankin, D.C.; Phansalkar, R.; Krause, E.; Dietz, B.; Bolton, J.L.; Nikolic, D.; Van Breeman, R.B.; et al. Botanical integrity: Part 2. Traditional and modern analytical approaches. Herbal. Gram. 2016, 109, 60–61. [Google Scholar]
- Macheix, J.-J.; Fleuriet, A.; Billot, J. Fruit Phenolics; CRC Press: Boca Raton, FL, USA, 1990. [Google Scholar]
- Guinot, P.; Roge, A.; Gargadennec, A.; Garcia, M.; Dupont, D.; Lecoeur, E.; Candelier, L.; Andary, C. Dyeing plants screening: An approach to combine past heritage and present development. Color. Technol. 2006, 122, 93–101. [Google Scholar] [CrossRef]
- Lavola, A.; Maukonen, M.; Julkunen-Tiitto, R. Variability in the composition of phenolic compounds in winter-dormant Salix pyrolifolia in relation to plant part and age. Phytochemistry 2018, 153, 102–110. [Google Scholar] [CrossRef]
- Caro, Y.; Venkatachalam, M.; Lebeau, J.; Fouillaud, M.; Dufossé, L. Pigments and colourant from filamentous fungi. In Fungal Metabolites; Mérillon, J.-M., Ramawat, K.G., Eds.; Springer: Cham, Switzerland, 2017; pp. 499–568. [Google Scholar]
- Schweppe, H. Handbuch der Naturfarbstoffe; Ecomed: Landsberg/Lech, Germany, 1993. [Google Scholar]
- Räisänen, R.; Primetta, A.; Niinimäki, K. Dyes from Nature; Archetype Publications: London, UK, 2016. [Google Scholar]
- Palo, R.T. Distribution of birch (Betula spp.), willow (Salix spp.) and poplar (Populus spp.) secondary metabolites and their potential role as chemical defense against herbivores. J. Chem. Ecol. 1984, 10, 499–520. [Google Scholar] [CrossRef]
- Németh, É.Z.; Héthelyi, É.; Bernáth, J. Comparison studies on Tanacetum vulgare L. chemotypes. J. Herbs Spices Med. Plants 1994, 2, 85–92. [Google Scholar] [CrossRef]
- Goudarzi, T.; Saharkhiz, M.J.; Rowshan, V. Ontogenetic variation of essential oil content and constituents in tansy (Tanacetum vulgare L.). J. Appl. Res. Med. Aromat. Plants 2015, 2, 48–53. [Google Scholar] [CrossRef]
- Zidorn, C. Seasonal variation of natural products in European trees. Phytochem. Rev. 2018, 17, 923–935. [Google Scholar] [CrossRef]
- Keskitalo, M. Exploring Biodiversity to Enhance Bioactivity in the Genus Tanacetum through Protoplast Fusion. Doctoral Dissertation, University of Helsinki, Helsinki, Finland, 1999. Available online: http://urn.fi/URN:ISBN:951-45-8965-3 (accessed on 14 October 2019).
- Hynninen, P.H.; Räisänen, R.; Elovaara, P.; Nokelainen, E. Preparative isolation of anthraquinones from the fungus Dermocybe sanguinea using enzymatic hydrolysis by the endogenous β-glucosidase. Z. Naturforsch. 2000, 55c, 600–610. [Google Scholar] [CrossRef]
- Spitaler, R.; Schlorhaufer, P.D.; Ellmerer, E.P.; Merfort, I.; Bortenschlager, S.; Stuppner, H.; Zidorn, C. Altitudinal variation of secondary metabolite profiles in flowering heads of Arnica montana cv. ARBO. Phytochemistry 2006, 67, 409–417. [Google Scholar] [CrossRef]
- Gaspar, H.; Moiteiro, C.; Turkman, A.; Coutinho, J.; Carnide, V. Influence of soil fertility on dye flavonoids production in weld (Reseda luteola L.) accessions from Portugal. J. Sep. Sci. 2009, 32, 4234–4240. [Google Scholar] [CrossRef]
- Jaakola, L.; Hohtola, A. Effect of latitude on flavonoid biosynthesis in plants. Plant Cell Environ. 2010, 33, 1239–1247. [Google Scholar] [CrossRef]
- Orians, C.M. Preserving leaves for tannin and phenolic glycoside analyses: A comparison of methods using three willow taxa. J. Chem. Ecol. 1995, 21, 1235–1243. [Google Scholar] [CrossRef]
- Brondz, I.; Ekeberg, D.; Høiland, K.; Bell, D.S.; Annino, A.R. The real nature of the indole alkaloids in Cortinarius infractus: Evaluation of artifact formation through solvent extraction method development. J. Chromatogr. A 2007, 1148, 1–7. [Google Scholar] [CrossRef]
- Saxena, S.; Raja, A.S.M. Natural dyes: Sources, chemistry, application and sustainability issues. In Roadmap to Sustainable Textiles and Clothing. Textile Science and Clothing Technology; Muthu, S.S., Ed.; Springer Science + Business Media: Singapore, 2014; pp. 37–80. [Google Scholar]
- Derksen, G.C.H.; Naayer, M.; Van Beek, T.A.; Capelle, A.; Haaksman, I.K.; Van Doren, H.A.; De Groot, Æ. Chemical and enzymatic hydrolysis of anthraquinone glycosides from madder roots. Phytochem. Anal. 2003, 14, 137–144. [Google Scholar] [CrossRef]
- Jäger, I.; Hafner, C.; Welsch, C.; Schneider, K.; Iznaguen, H.; Westendorf, J. The mutagenic potential of madder root in dyeing processes in the textile industry. Mutat. Res. 2006, 605, 22–29. [Google Scholar] [CrossRef]
- Ford, L.; Rayner, C.M.; Blackburn, R.S. Degradation of lucidin: New insights into the fate of this natural pigment present in dyer’s madder (Rubia tinctorum L.) during the extraction of textile artefacts. Dyes Pigments 2018, 154, 290–295. [Google Scholar] [CrossRef]
- Yasui, Y.; Takeda, N. Identification of a mutagenic substance, in Rubia tinctorum L. (madder) root, as lucidin. Mutat. Res. 1983, 121, 185–190. [Google Scholar] [CrossRef]
- Mouri, C.; Laursen, R. Identification of anthraquinone markers for distinguishing Rubia species in madder-dyed textiles by HPLC. Microchim. Acta 2012, 179, 105–113. [Google Scholar] [CrossRef]
- Zhang, X.; Laursen, R. Application of LC-MS to the analysis of dyes in objects of historical interest. Int. J. Mass Spectrom. 2009, 284, 108–114. [Google Scholar] [CrossRef]
- Kawasaki, Y.; Goda, Y.; Yoshihira, K. The mutagenic constituents of Rubia tinctorum. Chem. Pharm. Bull. 1992, 40, 1504–1509. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.P. A review of the genetic effects of naturally occurring flavonoids, anthraquinones and related compounds. Mutat. Res. 1980, 75, 243–277. [Google Scholar] [CrossRef]
- Inoue, K.; Yoshida, M.; Takahashi, M.; Fujimoto, H.; Shibutani, M.; Hirose, M.; Nishikawa, A. Carcinogenic potential of alizarin and rubiadin, component of madder color, in a rat medium-term multi-organ bioassay. Cancer Sci. 2009, 100, 2261–2267. [Google Scholar] [CrossRef]
- Fotia, C.; Avnet, S.; Granchi, D.; Baldini, N. The natural compound alizarin as an osteotropic drug for the treatment of bone tumors. J. Orthop. Res. 2012, 30, 1486–1492. [Google Scholar] [CrossRef]
- Liktor-Busa, E.; Kovács, B.; Urbán, E.; Hohmann, J.; Ványolós, A. Investigation of Hungarian mushrooms for antibacterial activity and synergistic effects with standard antibiotics against resistant bacterial strains. Lett. Appl. Microbiol. 2016, 62, 437–443. [Google Scholar] [CrossRef]
- Devrnja, N.; Anđelkovićb, B.; Aranđelovićc, S.; Radulovićc, S.; Sokovića, M.; Krstić-Miloševića, D.; Ristićd, M.; Ćalić, D. Comparative studies on the antimicrobial and cytotoxic activities of Tanacetum vulgare L. essential oil and methanol extracts. S. Afr. J. Bot. 2017, 111, 212–221. [Google Scholar] [CrossRef]
- Béni, Z.; Dékány, M.; Kovács, B.; Csupor-Löffler, B.; Zomborszki, Z.P.; Kerekes, E.; Szekeres, A.; Urbán, E.; Hohmann, J.; Ványolós, A. Bioactivity-guided isolation of antimicrobial and antioxidant metabolites from the mushroom Tapinella atrotomentosa. Molecules 2018, 23, 1082. [Google Scholar] [CrossRef] [PubMed]
- Greim, H. Aims and mission of regulatory toxicology. In Regulatory Toxicology; Schwenk, M., Reichl, F., Eds.; Springer Reference: Berlin/Heidelberg, Germany, 2014; pp. 3–20. [Google Scholar]
- Abad, M.J.; Bermejo, P.; Villar, A. An approach to the genus Tanacetum L. (Compositae): Phytochemical and pharmacological review. Phytother. Res. 1995, 9, 79–92. [Google Scholar] [CrossRef]
- Keskitalo, M.; Pehu, E.; Simon, J.E. Variation in volatile compounds from tansy (Tanacetum vulgare L.) related to genetic and morphological differences of genotypes. Biochem. Syst. Ecol. 2001, 29, 267–285. [Google Scholar] [CrossRef]
- Rosselli, S.; Bruno, M.; Raimondo, F.M.; Spadaro, V.; Varol, M.; Koparal, A.T.; Maggio, A. Cytotoxic effect of eudesmanolides isolated from flowers of Tanacetum vulgare ssp. siculum. Molecules 2012, 17, 8186–8195. [Google Scholar] [CrossRef]
- Uehara, A.; Akiyam, S.; Iwashina, T. Foliar flavonoids from Tanacetum vulgare var. boreale and their geographical variation. Nat. Prod. Commun. 2015, 10, 403–405. [Google Scholar] [CrossRef]
- Radulović, N.S.; Genčić, M.S.; Stojanović, N.M.; Randjelović, P.J.; Stojanović-Radić, Z.Z.; Stojiljković, N.I. Toxic essential oils. Part V. Behaviour modulating and toxic properties of thujones and thujone-containing essential oils of Salvia officinalis L.; Artemisia absinthium L., Thuja occidentalis L. and Tanacetum vulgare L. Food Chem. Toxicol. 2017, 105, 355–369. [Google Scholar] [CrossRef]
- Orians, C.M. The effect of hybridization in plants on secondary chemistry: Implications for the ecology and evolution of plant-herbivory interactions. Am. J. Bot. 2000, 87, 1749–1756. [Google Scholar] [CrossRef]
- Hynninen, S. Tansy as a Dye Plant–Hue and Colour Fastness of Wool Dyed with Different Tansy Populations [in Finnish]. Master’s Thesis, University of Joensuu, Savonlinna, Finland, 2009. [Google Scholar]
- Horváth, G.; Turcsi, E.; Molnár, P.; Szabó, L.G.; Deli, J. Carotenoid content of the flower of tansy (Tanacetum vulgare L.). Planta Medica 2007, 73, 911. [Google Scholar] [CrossRef]
- Wodjylo, A.; Oszmiański, J.; Czemerys, R. Antioxidant activity and phenolic compounds in 32 selected herbs. Food Chem. 2007, 105, 940–949. [Google Scholar] [CrossRef]
- Mureşan, M.; Benedec, D.; Vlase, L.; Oprean, R.; Toiu, A.; Oniga, I. Screening of polyphenolic compounds, antioxidant and antimicrobial properties of Tanacetum vulgare from Transylvania. Studia UBB Chemia 2015, 1, 127–138. [Google Scholar]
- Chaturvedi, D. Sesquiterpene lactones: Structural diversity and their biological activities. In Opportunity, Challenge and Scope of Natural Products in Medicinal Chemistry; Tiwari, V.K., Mishra, B.B., Eds.; Research Signpost: Kerala, India, 2011; pp. 313–334. [Google Scholar]
- Lahlou, S.; Tahraoui, A.; Israili, Z.; Lyoussi, B. Diuretic activity of the aqueous extracts of Carum carvi and Tanacetum vulgare in normal rats. J. Ethnopharmacol. 2007, 110, 458–463. [Google Scholar] [CrossRef] [PubMed]
- Klemola, K. Textile toxicity. Cytotoxicity and Spermatozoa motility Inhibition Resulting from Reactive Dyes and Dyed Fabrics. Doctoral Dissertation, Department of Biosciences, University of Kuopio, Kuopio, Finland, 2008. Available online: http://urn.fi/URN:ISBN:978-951-27-1094-2 (accessed on 14 October 2019).
- EURL ECVAM DB-ALM. DB-ALM Method Summary: Protein Determination Assays (Lowry, Kenacid Blue, Coomassie Blue). 2010. Available online: https://ecvam-dbalm.jrc.ec.europa.eu/methods-and-protocols/method-summary/1555/protein-determination-assays-(lowry,-kenacid-blue,-coomassie-blue)-summary/datasheet (accessed on 14 October 2019).
- Lahlou, S.; Israili, Z.H.; Lyoussi, B. Acute and chronic toxicity of a lyophilised aqueous extract of Tanacetum vulgare leaves in rodents. J. Ethnopharmacol. 2008, 117, 221–227. [Google Scholar] [CrossRef] [PubMed]
- Falchero, L.; Brown, R.H.; Mueller-Harvey, I.; Hanley, S.; Shield, I.; Karp, A. Condensed tannins in willow (Salix spp.): A first step to evaluate novel feeds nutritionally improved animal products. Grassl. Sci. Eur. 2011, 16, 434–436. [Google Scholar]
- Tyśkiewicz, K.; Konkol, M.; Kowalski, R.; Rój, E.; Warmiński, K.; Krzyżaniak, M.; Gil, Ł.; Stolarski, M.J. Characterization of bioactive compounds in the biomass of black locust, poplar and willow. A review. Trees 2019, 33, 1235–1263. [Google Scholar] [CrossRef]
- Shara, M.; Stohs, S.J. Efficacy and safety of white willow bark (Salix alba) extracts. Phytother. Res. 2015, 29, 1112–1116. [Google Scholar] [CrossRef]
- Matyjaszczyk, E.; Schumann, R. Risk assessment of white willow (Salix alba) in food. EFSA J. 2018, 16 (Suppl. 1), e16081. [Google Scholar] [CrossRef]
- Juntheikki, M.R.; Julkunen-Tiitto, R. Inhibition of β-glucosidase and esterase by tannins from Betula, Salix and Pinus species. J. Chem. Ecol. 2000, 26, 1151–1165. [Google Scholar] [CrossRef]
- Sulima, P.; Krauze-Baranowska, M.; Przyborowski, J.A. Variation in the chemical composition and content of salicylic glycosides in the bark of Salix purpurea from natural locations and their significance for breeding. Fitoterapia 2017, 118, 118–125. [Google Scholar] [CrossRef]
- Schofield, P.; Mbugua, D.M.; Pell, A.N. Analysis of condensed tannins: A review. Anim. Feed Sci. Technol. 2001, 91, 21–40. [Google Scholar] [CrossRef]
- Jürgenliemk, G.; Petereit, F.; Nahrstedt, A. Flavan-3-ols and procyanidins from the bark of Salix purpurea L. Pharmazie 2007, 62, 231–234. [Google Scholar]
- Dou, J.; Xu, W.; Koivisto, J.J.; Mobley, J.K.; Padmakshan, D.; Kögler, M.; Xu, C.; Willför, S.; Ralph, J.; Vuorinen, T. Characteristics of hot Water Extracts from the Bark of Cultivated Willow (Salix sp.). ACS Sustain. Chem. Eng. 2018, 6, 5566–5573. [Google Scholar] [CrossRef]
- Steglich, W.; Lösel, W.; Austel, V. Pilzpigmente IV. Anthrachinon-Pigmente aus Dermocybe sanguinea (Wulf. ex Fr.) Wünsche und D. semisanguinea (Fr.). Chem. Ber. 1969, 102, 4104–4118. [Google Scholar] [CrossRef]
- Steglich, W.; Lösel, W.; Pilzpigmente, X. Anthrachinon-glucoside aus Dermocybe sanguinea (Wulf. ex Fr.) Wünsche. Chem. Ber. 1972, 105, 2928–2932. [Google Scholar] [CrossRef] [PubMed]
- Räisänen, R. Anthraquinones from the Fungus Dermocybe sanguinea as Textile Dyes. Doctoral Dissertation, University of Helsinki, Helsinki, Finland, 2002. Available online: http://urn.fi/URN:ISBN:978-952-10-5928-5 (accessed on 14 October 2019).
- Stensrud, Ø.; Orr, R.J.S.; Reier-Røberg, K.; Schumacher, T.; Høiland, K. Phylogenetic relationships in Cortinarius with focus on North European species. Karstenia 2014, 54, 57–71. [Google Scholar] [CrossRef]
- Räisänen, R. Fungal colourants in applications—Focus on Cortinarius species. Color. Tehnol. 2019, 135, 22–31. [Google Scholar] [CrossRef]
- Beattie, K.D.; Rouf, R.; Gander, L.; May, T.W.; Ratkowsky, R.; Donner, C.D.; Gill, M.; Grice, I.D.; Tiralongo, E. Antibacterial metabolites from Australian macrofungi from the genus Cortinarius. Phytochemistry 2010, 71, 948–955. [Google Scholar] [CrossRef]
- Fouillaud, M.; Venkatachalam, M.; Girard-Valenciennes, E.; Caro, Y.; Dufossé, L. Anthraquinones and 70Derivatives from Marine-Derived Fungi: Structural Diversity and Selected Biological Activities. Mar. Drugs 2016, 14, 64. [Google Scholar] [CrossRef]
- Berger, K.J.; Guss, D.A. Mycotoxins revisited: Part II. J. Emerg. Med. 2005, 28, 175–183. [Google Scholar] [CrossRef]
- Dinis-Oliveira, R.J.; Soares, M.; Rocha-Pereira, C.; Carvalho, F. Human and experimental toxicology of orellanine. Hum. Exp. Toxicol. 2016, 35, 1016–1029. [Google Scholar] [CrossRef]
- Shao, D.; Tang, S.; Healy, R.A.; Imerman, P.M.; Schrunk, D.E.; Rumbeiha, W.K. A novel orellanine containing mushroom Cortinarius armillatus. Toxicon 2016, 114, 65–74. [Google Scholar] [CrossRef]
- Danel, V.; Saviuc, P.F.; Garon, D. Main features of Cortinarius spp. poisoning: A literature review. Toxicon 2001, 39, 1053–1060. [Google Scholar] [CrossRef]
- FinBIF. Finnish Biodiversity Information Facility. Cortinarius semisanguineus and Salix phylicifolia. 2018. Available online: https://laji.fi/taxon/MX.72871 (accessed on 14 October 2019).
- Dong, X.; Fu, J.; Yin, X.; Cao, S.; Li, X.; Lin, L.; Huyiligeqi; Ni, J. Emodin: A review of its pharmacology, toxicity and pharmacokinetics. Phytother. Res. 2016, 30, 1207–1218. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Gao, J.; Wang, T.-S.; Guo, C.; Yan, Y.-J.; Mao, C.-Y.; Gu, L.-W.; Yang, Y.; Li, Z.-F.; Liu, A. NMR-based metabolomic techniques identify the toxicity of emodin in HepG2 cells. Sci. Rep. 2018, 8, 9379. [Google Scholar] [CrossRef] [PubMed]
- Von Wright, A.; Raatikainen, O.; Taipale, H.; Kärenlampi, S.; Mäki-Paakkanen, J. Directly acting geno- and cytotoxic agents from a wild mushroom Dermocybe sanguinea. Mutat. Res. Fundam. Mol. Mech. Mutagen. 1992, 269, 27–33. [Google Scholar] [CrossRef]
- Salo, P.; Niemelä, T.; Salo, U. Suomen Sieniopas; Handbook of Fungi in Finlan; LUOMUS Finnish Museum of Natural History, Botany, WSOY: Helsinki, Finland, 2006. [Google Scholar]
- Holzapfel, M.; Kilpert, C.; Steglich, W. Uber Leucomentine, farblose Vorstufen des Atromentins aus dem SamtfuBkrempling (Paxillus atrotomentosus). Liebigs Ann. Chem. 1989, 8, 797–801. [Google Scholar] [CrossRef]
- Besl, H.; Bresinsky, A.; Geigenmuller, G.; Herrmann, R.; Kilpert, C.; Steglich, W. Flavomentine und Spiromentine, neue Terphenylchinon-Derivate aus Paxillus atrotomentosus und P. panuoides (Boletales). Liebigs Ann. Chem. 1989, 8, 803–810. [Google Scholar] [CrossRef]
- Buchanan, M.S.; Hashimoto, T.; Takaoka, S.; Asakawa, Y. (+)-osmundalactone, ɣ-lactones and spiromentins from the fungus Paxillus atrotomentosus. Phytochemistry 1995, 40, 1251–1257. [Google Scholar] [CrossRef]
- Heikkinen, S. Sustainable Colour from Nature? The Effect of Salix phylicifolia and Rheum cultorum as Mordants to Colour and Colour Fastness of Wool Dyed with Dermocybe semisanguinea [in Finnish]. Master’s Thesis, University of Helsinki, Helsinki, Finland, 2010. [Google Scholar]
- Loukojärvi, L.-K. Effect of Detergent on Colour Change and Colour Fastness of Natural Dyed Wool in Washing [in Finnish]. Master’s Thesis, University of Helsinki, Helsinki, Finland, 2010. [Google Scholar]
- Räisänen, R.; Björk, H.; Hynninen, P.H. Two-dimensional TLC separation and mass spectrometric identification of anthraquinones isolated from the fungus Dermocybe sanguinea. Z. Naturforsch. C J. Biosci. 2000, 55c, 195–202. [Google Scholar] [CrossRef]
- Huheey, J.E. Inorganic Chemistry. Principles of Structure and Reactivity, 3rd ed.; Harper & Row: New York, NY, USA, 1983. [Google Scholar]
- Triana, J.; Eiroa, J.L.; Morales, M.; Pérez, F.J.; Brouard, I.; Marrero, M.T.; Estévez, S.; Quintana, J.; Estévez, F.; Castillo, Q.A.; et al. A chemotaxonomic study of endemic species of genus Tanacetum from the Canary Islands. Phytochemistry 2013, 92, 87–104. [Google Scholar] [CrossRef]
- Bączek, K.B.; Kosakowska, O.; Przybył, J.L.; Pióro-Jabrucka, E.; Costa, R.; Mondello, L.; Gniewosz, M.; Synowiec, A.; Węglarz, Z. Antibacterial and antioxidant activity of essential oils and extracts from costmary (Tanacetum balsamita L.) and tansy (Tanacetum vulgare L.). Ind. Crop. Prod. 2017, 102, 154–163. [Google Scholar] [CrossRef]
- Venditti, A.; Frezza, C.; Sciubba, F.; Serafini, M.; Bianco, A.; Cianfaglione, K.; Lupidi, G.; Quassinti, L.; Bramucci, M.; Maggi, F. Volatile components, polar constituents and biological activity of tansy daisy (Tanacetum macrophyllum (Waldst. et Kit.) Schlutz Bip.). Ind. Crop Prod. 2018, 118, 225–235. [Google Scholar] [CrossRef]
- Coté, H.; Boucher, M.-A.; Pichette, A.; Legault, J. Anti-inflammatory, antioxidant, antibiotic and cytotoxic activities of Tanacetum vulgare L. essential oil and its constituents. Medicines 2017, 4, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Schneider, P.; Bouhired, S.; Hoffmeister, D. Characterization of the atromentin biosynthesis genes and enzymes in the homobasidiomycete Tapinella panuoides. Fungal Genet. Biol. 2008, 45, 1487–1496. [Google Scholar] [CrossRef]
- Goyal, S.; Ramawat, K.G.; Mérillon, J.-M. Different Shades of Fungal Metabolites: An Overview. In Fungal Metabolites; Mérillon, J.-M., Ramawat, K.G., Eds.; Springer: Cham, Switzerland, 2017; pp. 1–29. [Google Scholar]
- Mérillon, J.-M.; Ramawat, K.G. (Eds.) Fungal Metabolites; Springer: Cham, Switzerland, 2017. [Google Scholar]
- Liu, J.-K. Natural terphenyls: Developments since 1877. Chem. Rev. 2006, 106, 2209–2223. [Google Scholar] [CrossRef]
- Kopponen, P.; Asikainen, M.; Törrönen, R.; Klemola, K.; Liesivuori, J.; Kärenlampi, S. In vitro cytotoxicity of textile dyes and extracts of dyed/finished fabrics. Altern. Lab. Animals 1997, 25, 539–546. [Google Scholar]
- Benassi-Zanqueta, É.; Marques, C.F.; Valone, L.M.; Pellegrini, B.L.; Bauermeister, A.; Ferreira, I.C.P.; Lopes, N.P.; Nakamura, C.V.; Dias Filho, B.P.; Natali, M.R.; et al. Evaluation of anti-HSV-1 activity and toxicity of hydroethanolic extract of Tanacetum parthenium (L.) Sch.Bip. (Asteraceae). Phytomedicine 2019, 55, 249–254. [Google Scholar] [CrossRef]
- Pearson, J.; Liesivuori, J.; Lindström-Seppä, P. Evaluating the toxicity of fabric extracts using the hepa-1 cytotoxicity test, the HaCaT cytotoxicity test and the spermatozoa motility inhibition test. J. Text. Inst. 2009, 100, 330–337. [Google Scholar] [CrossRef]
- Pihlava, J.-M.; Hellström, J.; Kurtelius, T.; Mattila, P. Flavonoids, anthocyanins, phenolamides, benzoxazinoids, lignans and alkylresorcinols in rye (Secale cereale) and some rye products. J. Cereal Sci. 2018, 79, 183–192. [Google Scholar] [CrossRef]
- Vanden Berghe, I.; Gleba, M.; Mannering, U. Towards the identification of dyestuffs in Early Iron Age Scandinavian peat bog textiles. J. Archaeol. Sci 2019, 36, 1910–1912. [Google Scholar] [CrossRef]
- Smith, K.J. Colour-order systems, colour spaces, colour difference and colour scales. In Colour Physics for Industry, 2nd ed.; McDonald, R., Ed.; Society of Dyers and Colourists: Bradford, UK, 1997. [Google Scholar]
- Borenfreund, E.; Puerner, J.A. A simple quantitative procedure using monolayer cultures for cytotoxicity assays (HTD/NR-90). J. Tissue Cult. Methods 1984, 9, 7–9. [Google Scholar] [CrossRef]
| Chemical Compound | Structure |
|---|---|
| Alizarin (1) C14H8O4 M = 240.21 g mol−1 | 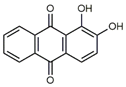 |
| Lucidin (2) C15H10O5 M = 270.24 g mol−1 | 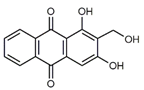 |
| Rubiadin (3) C15H10O4 M = 254.24 g mol−1 | 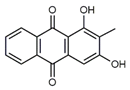 |
| Xanthopurpurin/ Purpuroxanthine (4) C14H8O4 M = 240.21 g mol−1 | 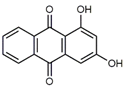 |
| Scientific Name | Tanacetum vulgare L. | Salix phylicifolia L. | Cortinarius semisanguineus (Fr.) Gillet | Tapinella atrotomentosa (Batsch.) Šutara |
|---|---|---|---|---|
| Common names | Tancy, golden buttons | Willow, tea-leaved willow | Surprise webcap | Velvet roll-rim |
| Appearance |  |  |  |  |
| CIELab coordinates | L 70.74; a* 2.54; b* 29.60 | L 56.39; a* 13.43; b* 24.98 | L 49.90; a* 21.22; b* 20.14 | L 38.64; a* 4.07; b* 18.27 |
| Direct dye, NM | ||||
| L 68.66; a* 3.30; b* 49.18 | L 57.09; a* 12.58; b* 26.54 | L 43.65; a* 34.49; b* 30.05 | L 33.68; a* 3.42; b* 16.50 | |
| KAl(SO4)2 | ||||
| L 35.07; a* 0.45; b* 13.64 | L 43.81; a* 5.06; b* 15.58 | L 35.62; a* 8.84; b* 10.63 | L 23.63; a* -0.24; b* 10.66 | |
| FeSO4 | ||||
| Main colourants [ref] | [12,13] | [63] | [64,66,68] | [79,80] |
Luteolin (5)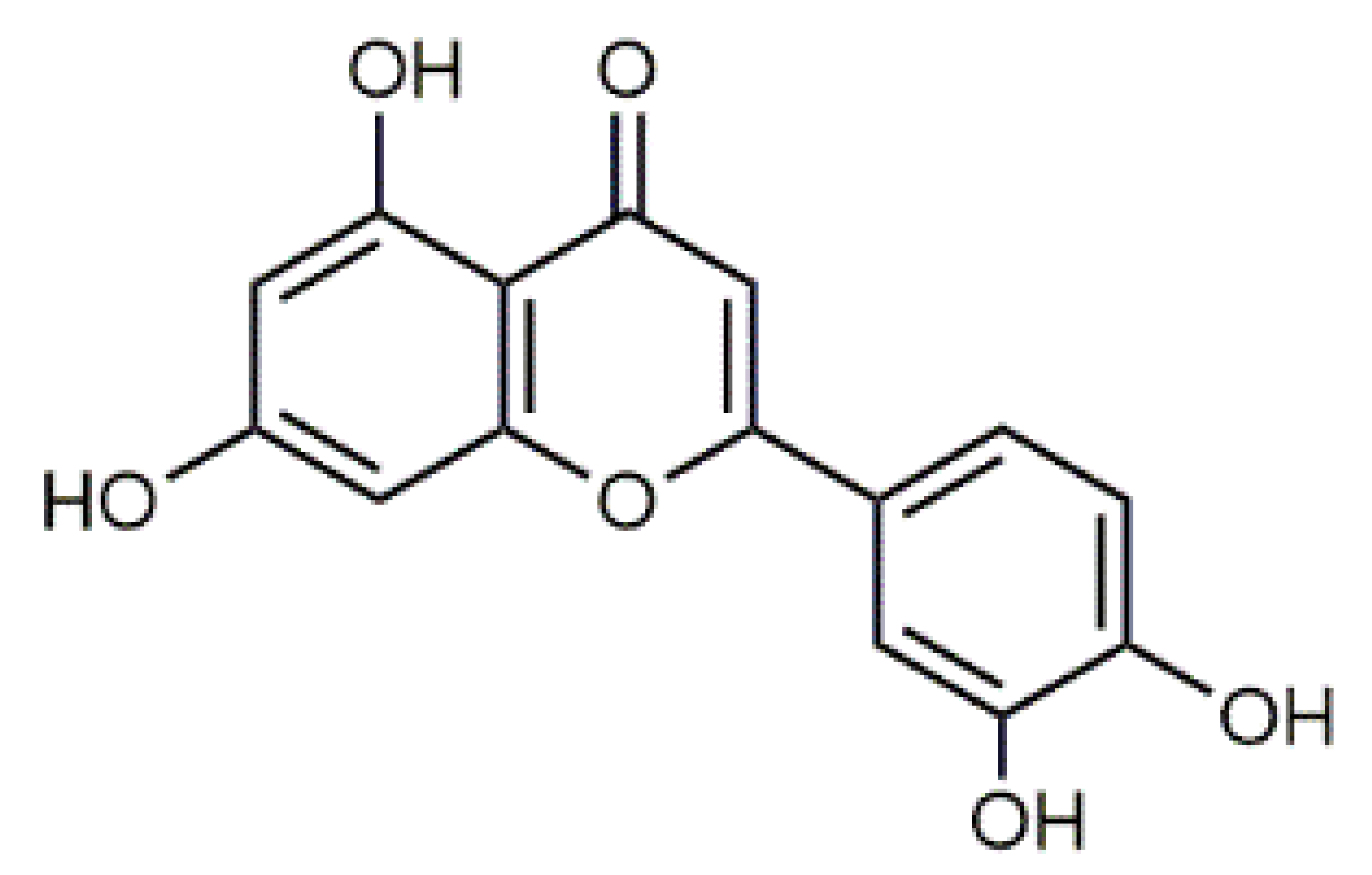 C15H10O6 M = 286.24 g·mol−1 C15H10O6 M = 286.24 g·mol−1 | Picein (9)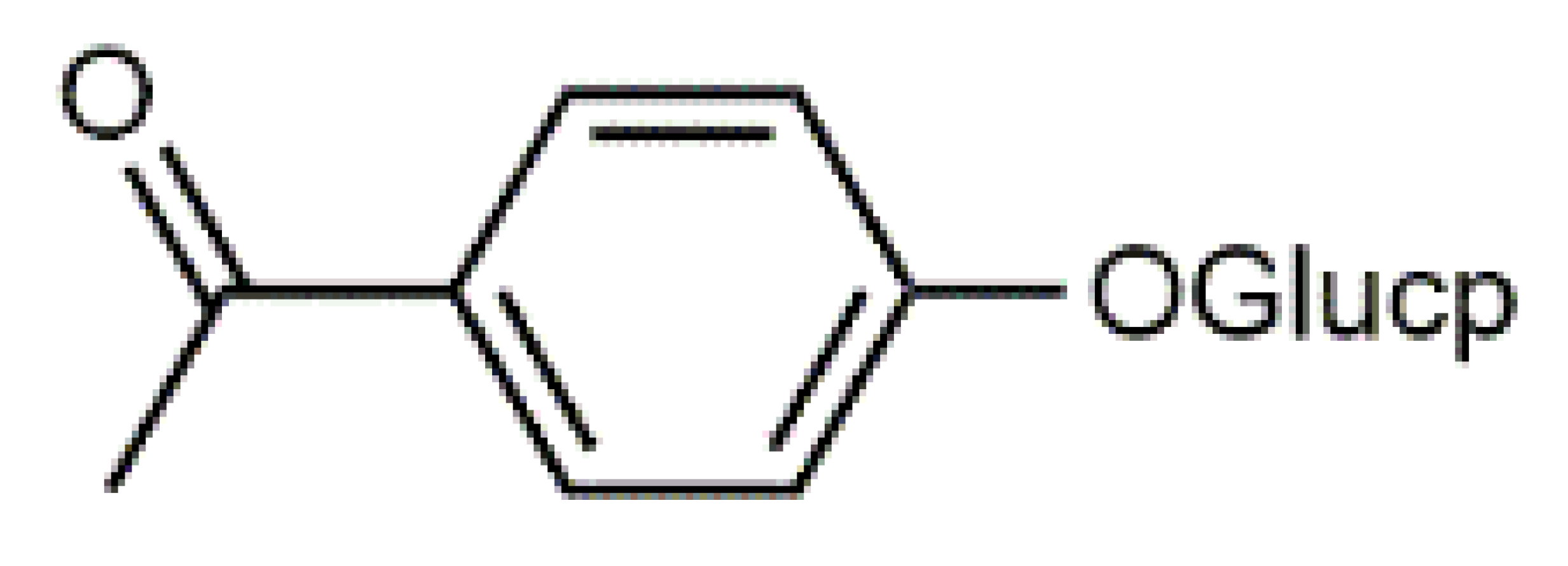 C14H18O7 M = 298.29 g·mol−1 C14H18O7 M = 298.29 g·mol−1 | Emodin (13)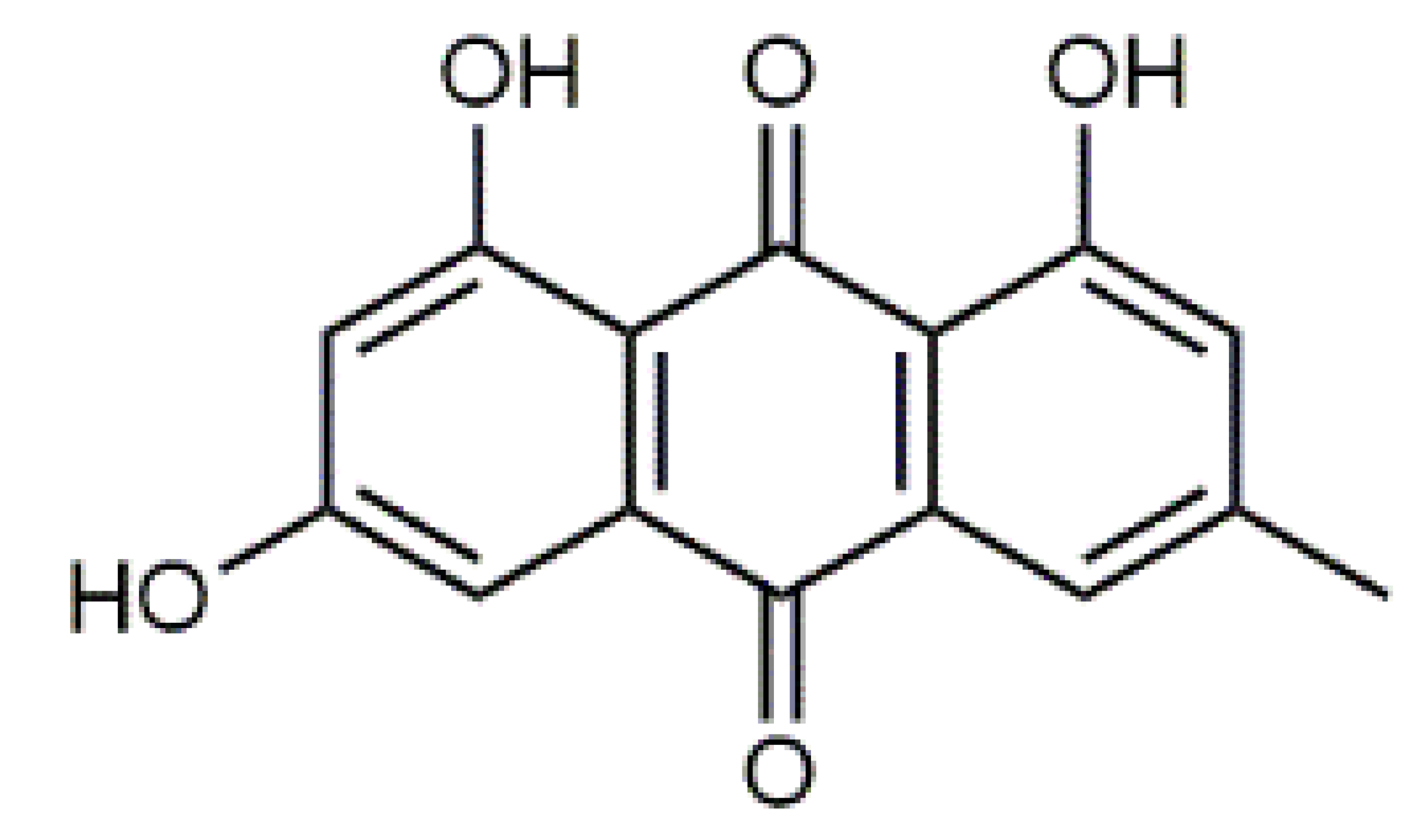 C15H10O5 M = 270.24 g·mol−1 C15H10O5 M = 270.24 g·mol−1 | Atromentin (17)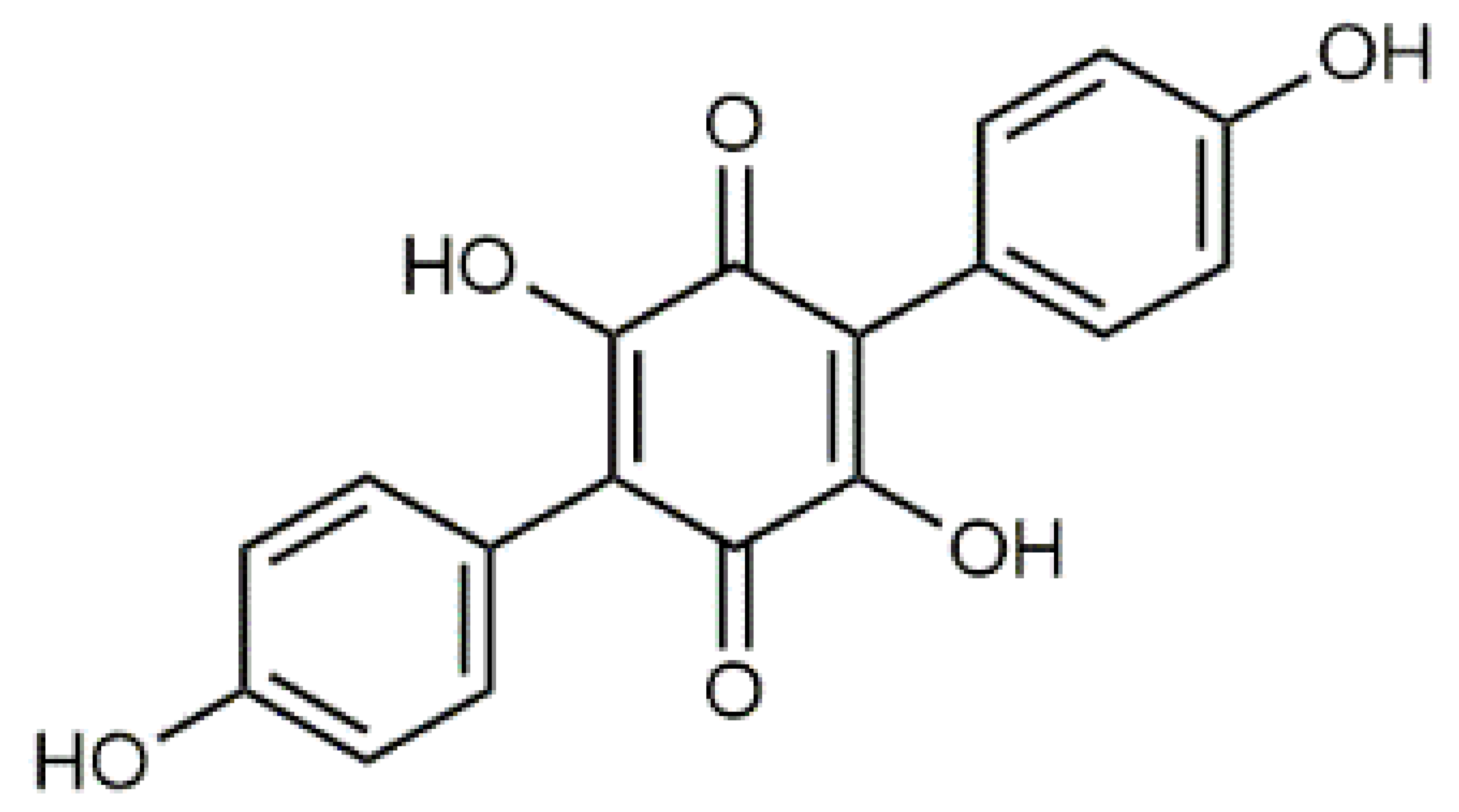 C18H12O6 M = 324.29 g·mol−1 C18H12O6 M = 324.29 g·mol−1 | |
Quercetin (6) C15H10O7 M = 302.24 g·mol−1 C15H10O7 M = 302.24 g·mol−1 | Triandrin (10)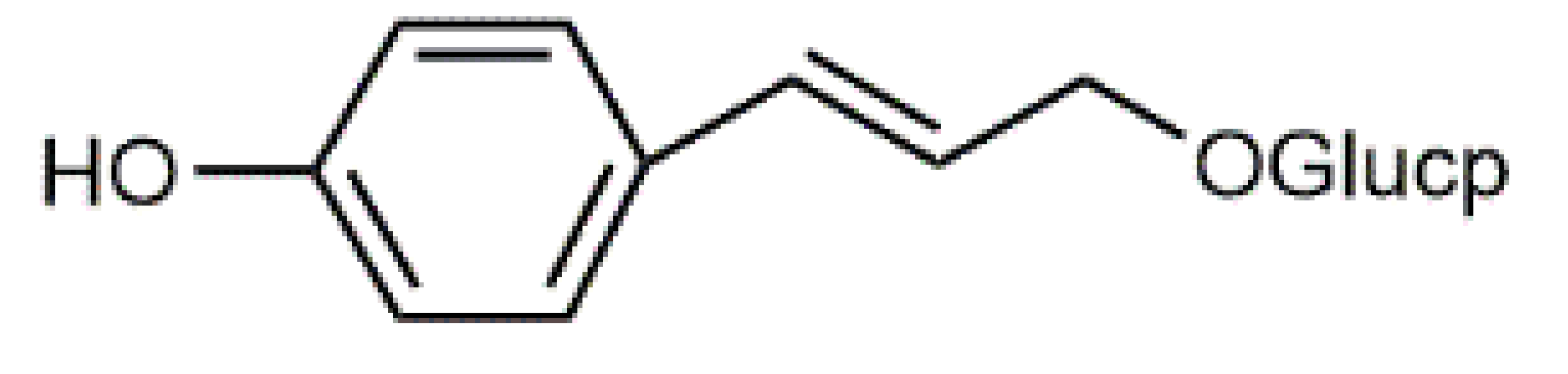 C15H20O7 M = 312.31·g mol−1 C15H20O7 M = 312.31·g mol−1 | Dermocybin (14)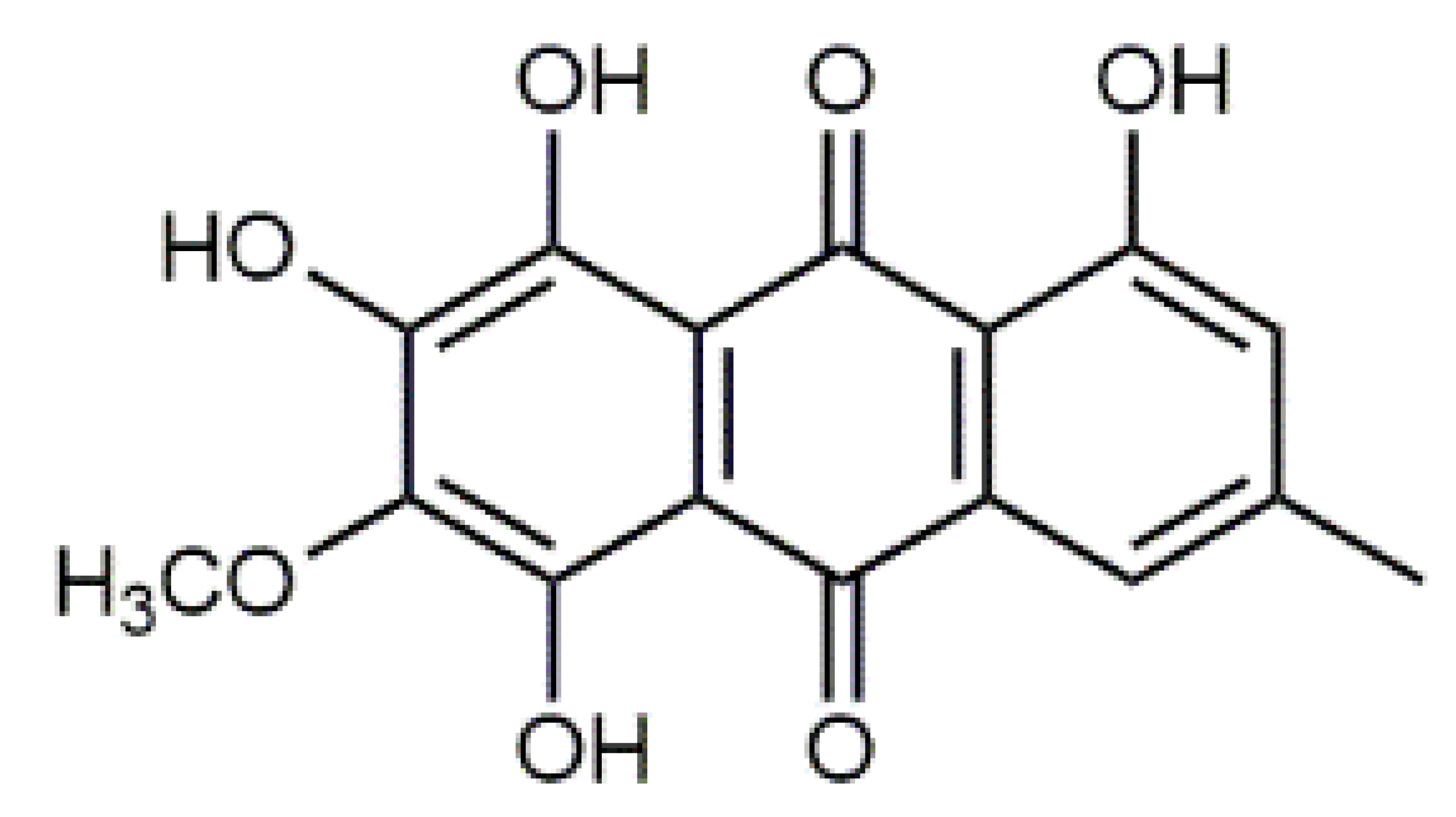 C16H12O7 M = 316.27 g·mol−1 C16H12O7 M = 316.27 g·mol−1 | Leucomentin 2 (18) C30H28O10 M = 548.54 g·mol−1 C30H28O10 M = 548.54 g·mol−1 | |
Violaxanthin (7)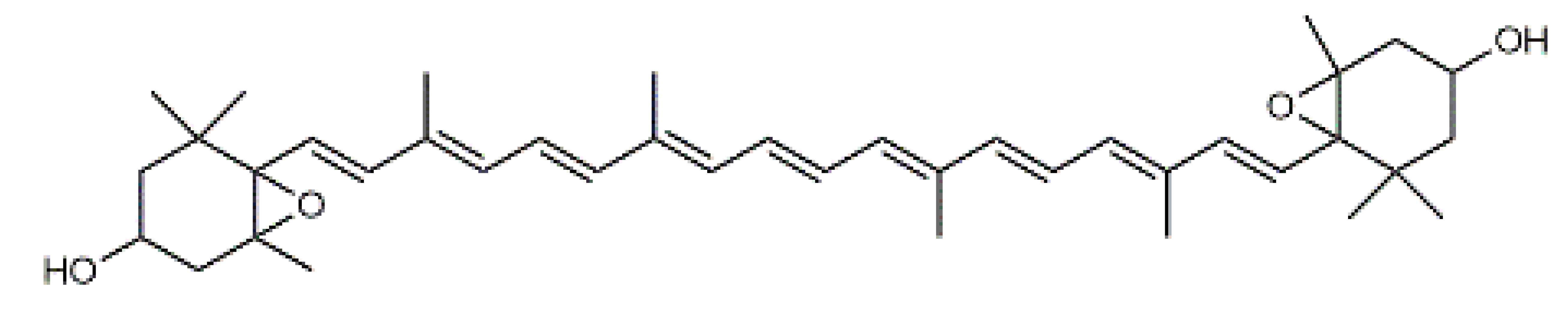 C40H56O4 M = 600.88 g·mol−1 C40H56O4 M = 600.88 g·mol−1 | (+) Catechin (11) and epicatechin differentiate in stereochemistry of C2 OH-gr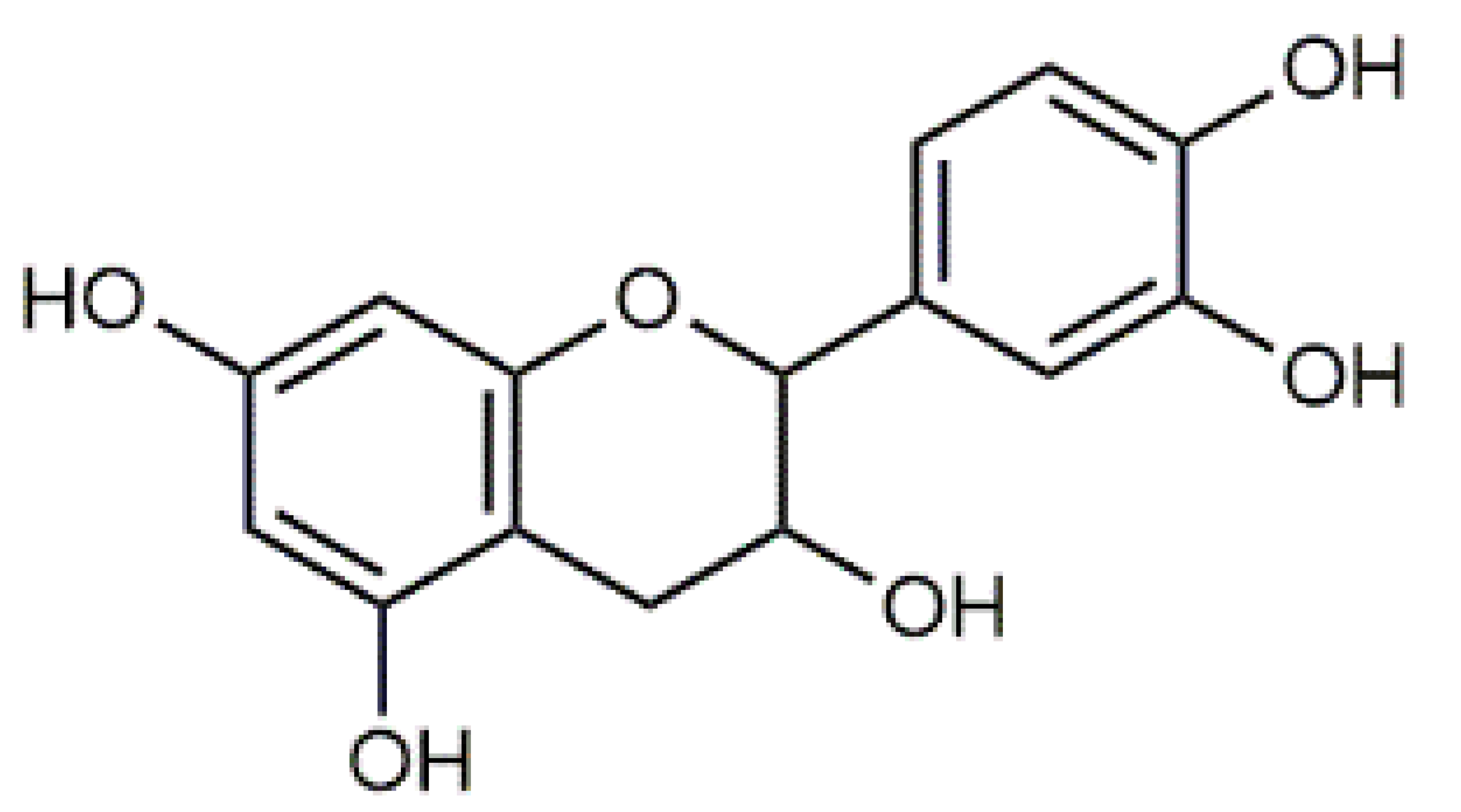 C15H14O6 M = 290.27 g·mol−1 C15H14O6 M = 290.27 g·mol−1 | Dermorubin (15)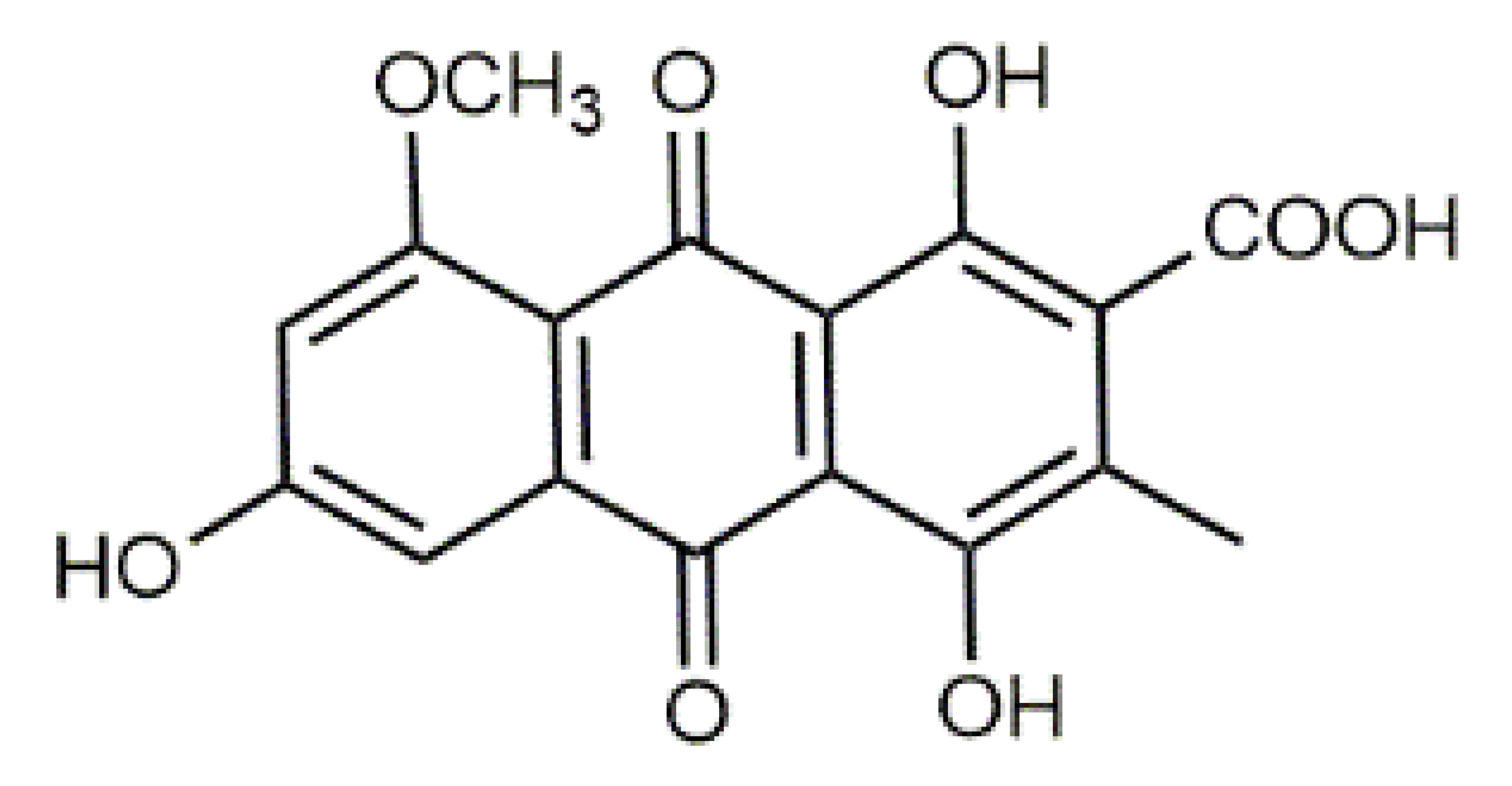 C17H12O8 M = 344.28 g·mol−1 C17H12O8 M = 344.28 g·mol−1 | Leucomentin 3 (19)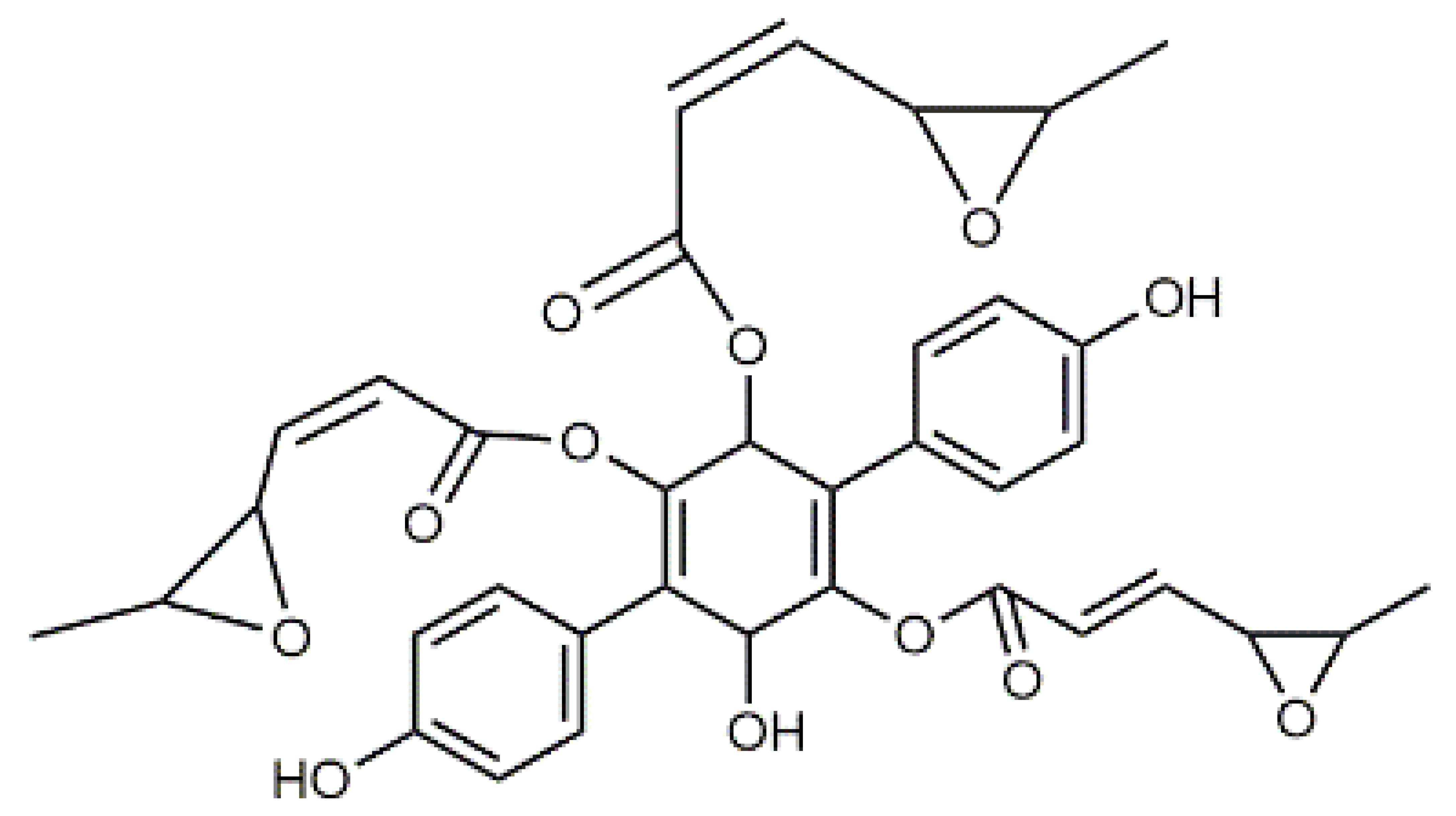 C36H34O12 M = 658.66 g·mol−1 C36H34O12 M = 658.66 g·mol−1 | |
β-Carotene (8)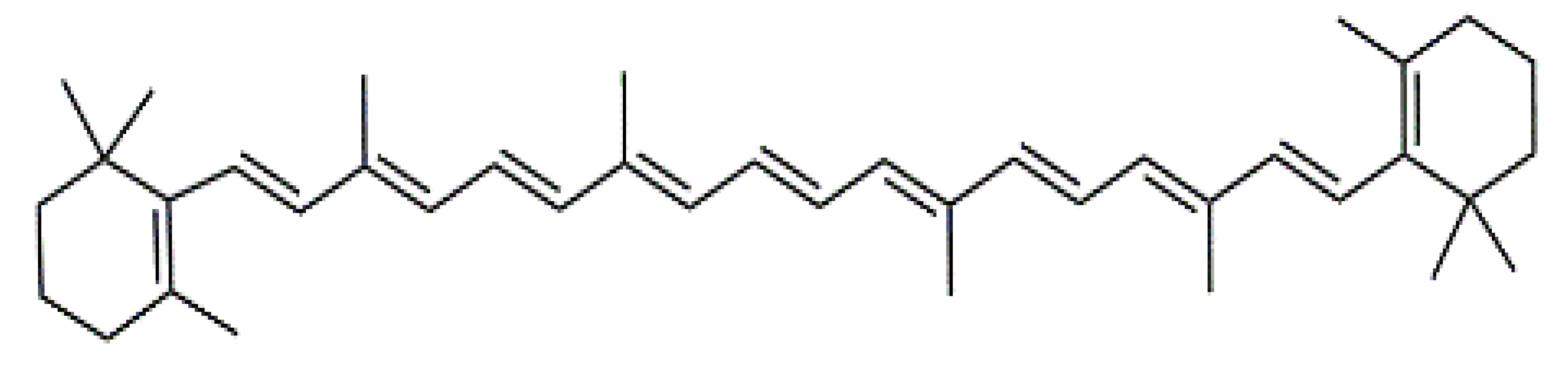 C40H56 M = 536.89 g·mol−1 C40H56 M = 536.89 g·mol−1 | Salireposide (12)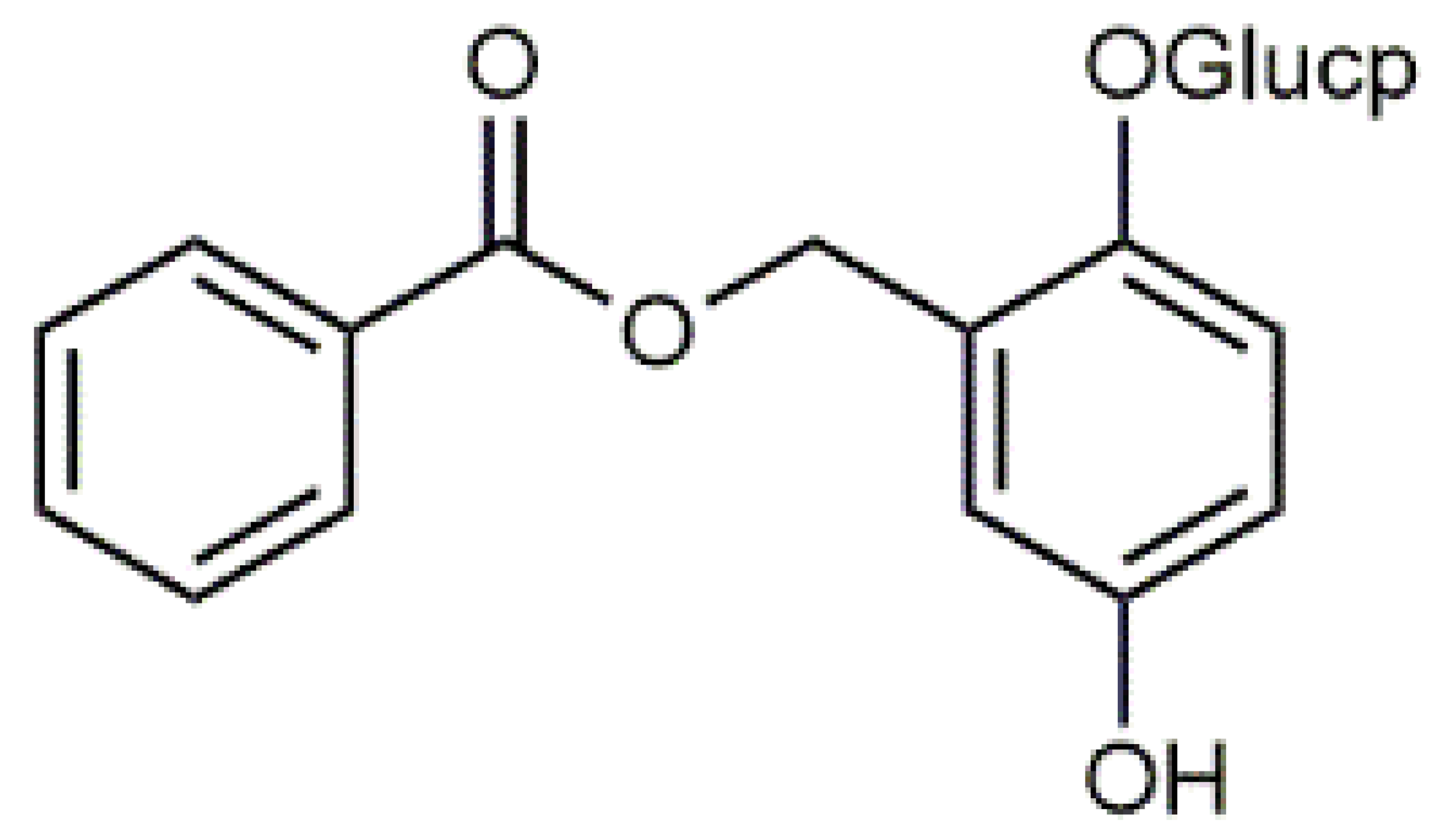 C20H22O9 M = 406.37 g·mol−1 C20H22O9 M = 406.37 g·mol−1 | Physcion (16)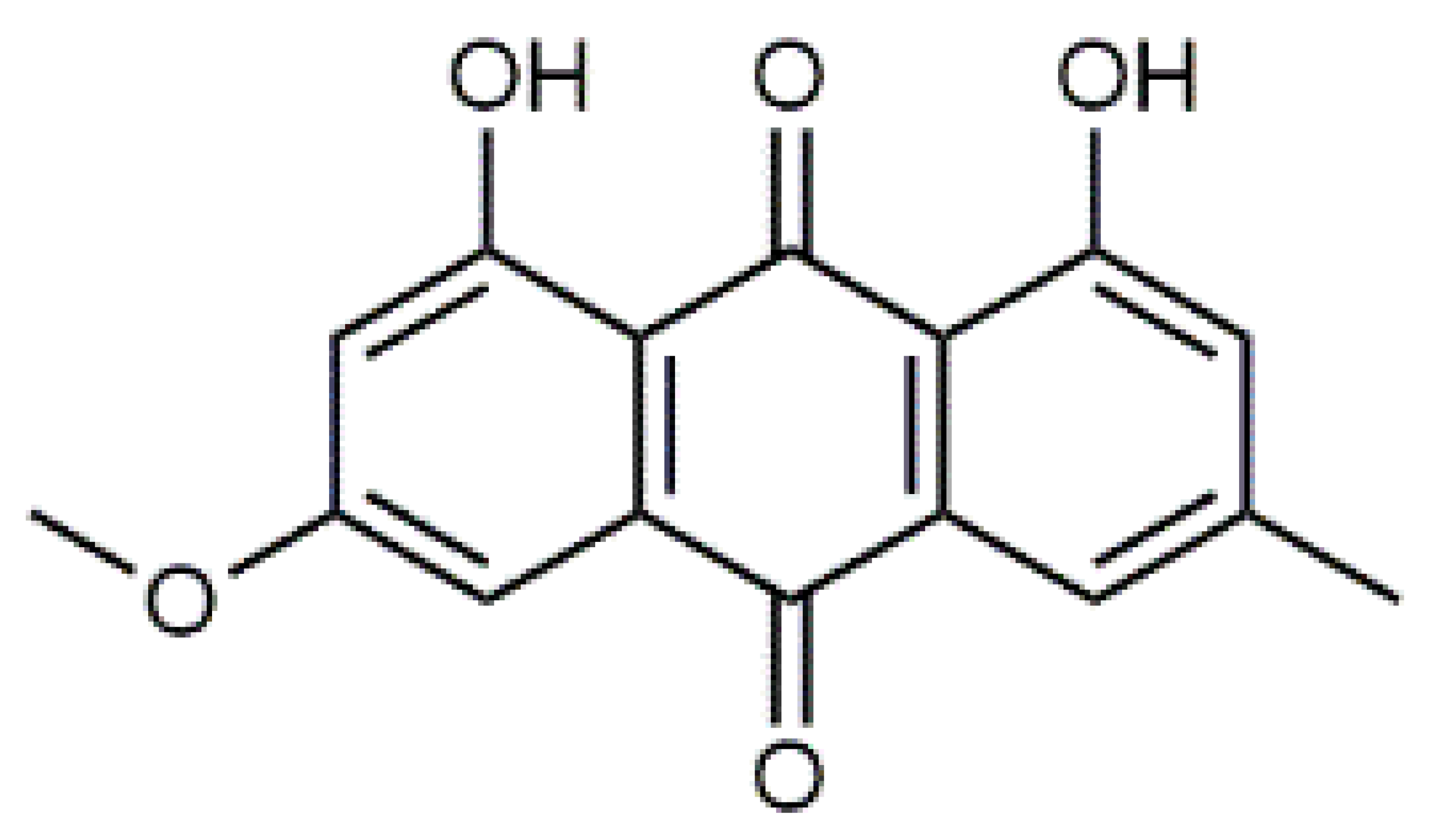 C16H12O5 M = 284.27 g·mol−1 C16H12O5 M = 284.27 g·mol−1 | Spiromentin (20)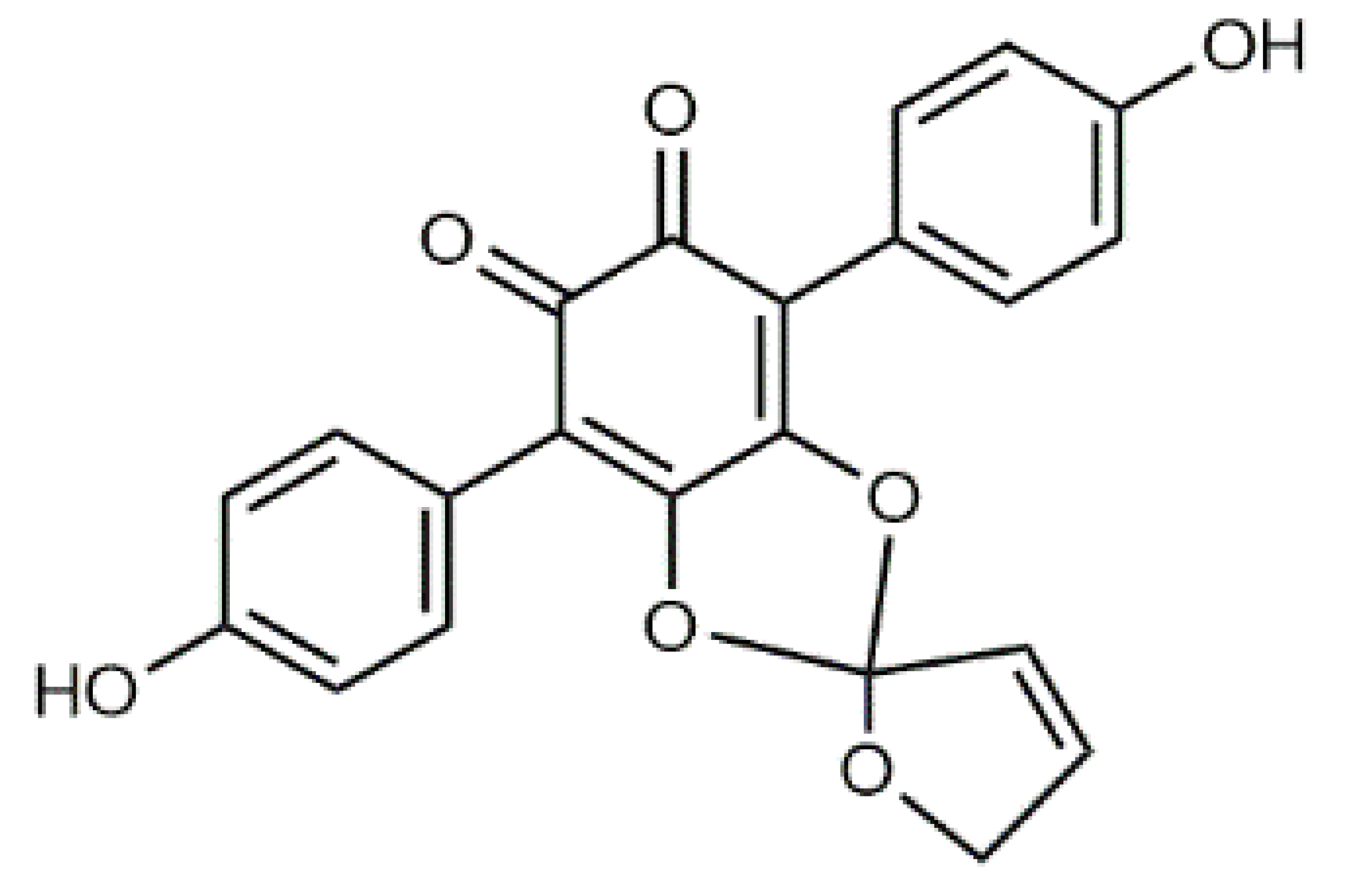 C22H14O7 M = 390.35 g·mol−1 C22H14O7 M = 390.35 g·mol−1 |
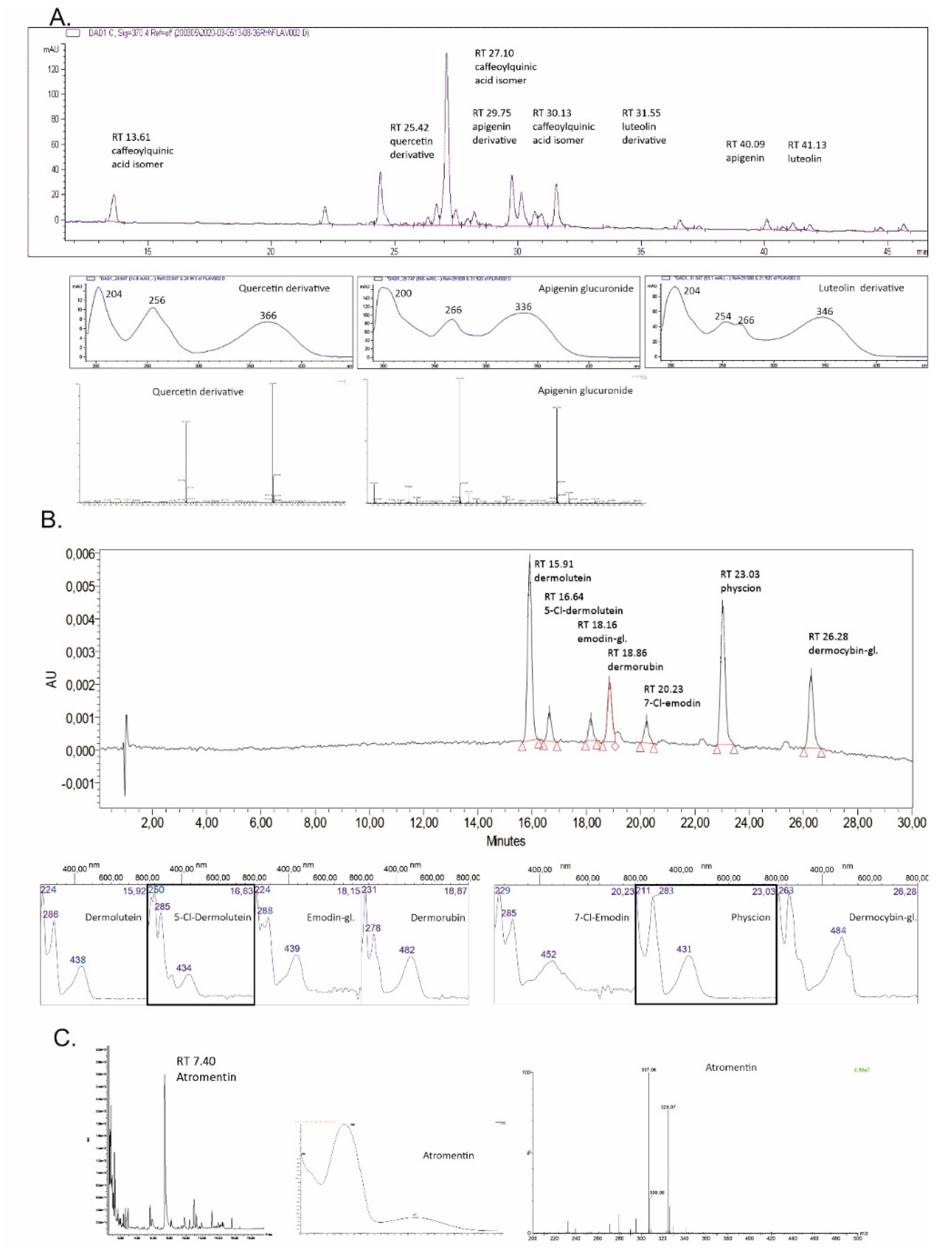
| Ret. Time | UV/Vis λmax [nm] | MS Data: m/z [M − H]/[M + H]+ and Main Fragment | Molecular Formula | Compound |
|---|---|---|---|---|
| Tanacetum vulgare, Figure 1, chromatogram (370 nm) and spectra A | ||||
| 13.61 | 218, 244, 298 sh ‡, 328 | 353.0864; 191.0535 | C16H18O9 | Caffeoylquinic acid isomer I |
| 25.42 | 204, 256, 366 | 463.0878; 301.0331 | C21H20O12 | Quercetin (6) derivative |
| 27.10 | 218, 244, 298 sh, 328 | 515.1186; 353.0859 | C25H24O12 | Caffeoylquinic acid isomer II |
| 29.75 | 200, 266, 336 | 445.0759; 269.0428 | C21H18O11 | Apigenin derivative |
| 30.13 | 218, 244, 300 sh, 328 | 515.1191; 353.0863 | C25H24O12 | Caffeoylquinic acid isomer III |
| 31.55 | 204, 254, 266, 346 | 639.3185; 285.0378 | - | Luteolin (5) derivative |
| 40.09 | 206, 266, 336 | 269.0427 | C15H10O5 | Apigenin |
| 41.13 | 204, 254, 266, 346 | 285.0388 | C15H10O6 | Luteolin (5) |
| Cortinarius semisanguineus, Figure 1, chromatogram (440 nm) and spectra B | ||||
| 15.91 | 224, 286, 438 | 327; 283 | C17H12O7 | Dermolutein |
| 16.64 | 250, 285, 434 | - | C17H11ClO7 | 5-Cl-Demolutein |
| 18.16 | 224, 288, 439 | 684 | C21H20O10 | Emodin-1-glucoside (13) * |
| 18.86 | 231, 278, 482 | 343; 299 | C17H12O8 | Dermorubin (15) |
| 20.23 | 229, 285, 452 | 303; 239 | C15H9ClO5 | 7-Cl-Emodin |
| 23.03 | 211, 283, 431 | 283; 240 | C16H12O5 | Physcion (16) |
| 26.28 | 263, 484 | 770 | C22H22O12 | Dermocybin-1-glucoside (14) # |
| Tapinella atrotomentosa, Figure 1, chromatogram (370 nm) and spectra C | ||||
| 07.40 | 263, 371 | 325.07; 307.06 | C18H12O6 | Atromentin (17) |
| Dyeing | LF | Washing Fastness | ||
|---|---|---|---|---|
| Cc | s, WO | s, CO | ||
| Tanacetum | ||||
| NM, direct dye | 6 | 1 | 5 | 4/5 |
| Alum mordant | 7 | 3/4 | 5 | 4 |
| FeSO4 mordant | 7 | 2/3 | 5 | 4 |
| Salix | ||||
| NM, direct dye | 5 | 1 | 5 | 5 |
| Alum mordant | 6 | 2/3 | 5 | 5 |
| FeSO4 mordant | 6 | 2 | 5 | 5 |
| Cortinarius | ||||
| NM, direct dye | 3 | 3/4 | 4/5 | 4/5 |
| Alum mordant | 4 | 4 | 4/5 | 4/5 |
| FeSO4 mordant | 5 | 4 | 4/5 | 4/5 |
| Tapinella | ||||
| NM, direct dye | 2 | 3 | 5 | 4/5 |
| Alum mordant | 4 | 3/4 | 5 | 4/5 |
| FeSO4 mordant | 5 | 3/4 | 5 | 5 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Räisänen, R.; Primetta, A.; Nikunen, S.; Honkalampi, U.; Nygren, H.; Pihlava, J.-M.; Vanden Berghe, I.; von Wright, A. Examining Safety of Biocolourants from Fungal and Plant Sources-Examples from Cortinarius and Tapinella, Salix and Tanacetum spp. and Dyed Woollen Fabrics. Antibiotics 2020, 9, 266. https://doi.org/10.3390/antibiotics9050266
Räisänen R, Primetta A, Nikunen S, Honkalampi U, Nygren H, Pihlava J-M, Vanden Berghe I, von Wright A. Examining Safety of Biocolourants from Fungal and Plant Sources-Examples from Cortinarius and Tapinella, Salix and Tanacetum spp. and Dyed Woollen Fabrics. Antibiotics. 2020; 9(5):266. https://doi.org/10.3390/antibiotics9050266
Chicago/Turabian StyleRäisänen, Riikka, Anja Primetta, Sari Nikunen, Ulla Honkalampi, Heli Nygren, Juha-Matti Pihlava, Ina Vanden Berghe, and Atte von Wright. 2020. "Examining Safety of Biocolourants from Fungal and Plant Sources-Examples from Cortinarius and Tapinella, Salix and Tanacetum spp. and Dyed Woollen Fabrics" Antibiotics 9, no. 5: 266. https://doi.org/10.3390/antibiotics9050266
APA StyleRäisänen, R., Primetta, A., Nikunen, S., Honkalampi, U., Nygren, H., Pihlava, J.-M., Vanden Berghe, I., & von Wright, A. (2020). Examining Safety of Biocolourants from Fungal and Plant Sources-Examples from Cortinarius and Tapinella, Salix and Tanacetum spp. and Dyed Woollen Fabrics. Antibiotics, 9(5), 266. https://doi.org/10.3390/antibiotics9050266






