1. Introduction
Intimate hygiene products are widely used by women as part of their daily cleansing routine; however, there is a lack of scientific literature about the impact of intimate personal hygiene product use on the vulvar area and even less information is available on the impact on vulvar microbiome stability. Proper feminine hygiene is critical for women’s intimate health, as the vulva is a woman’s first line of defense to protect the genital tract from infection [
1]. Vulvar skin is transitional in nature, changing from keratinized skin to non-keratinized mucosal epithelium, and can be differentiated from other skin sites based on many attributes, such as the level of hydration, friction, and permeability [
2]. Being covered by a thin stratum corneum with large hair follicles renders vulvar skin to be more susceptible to topical agents and irritation, leading to dermatitis and other dermatologic conditions when its barrier function is affected by factors such as moisture (urine, vaginal discharge), enzymes (stool residue), friction, and heat [
2,
3]. Symptoms of vulvovaginal disorders are common and can have a significant impact on quality of life.
The microbial inhabitants of the skin are highly dependent on the microenvironment of the biogeographic site and, based on a large-scale body-mapping study, can be grouped into dry, moist, and sebaceous sites [
4]. However, not much is known about the transitional vulvar skin, which could be considered a combination of moist and sebaceous sites. In particular, the vast majority of research to date has focused on determining the composition of bacterial communities present in the vaginal orifice of healthy and some symptomatic women. Similar to skin health, the natural microflora of the vulva plays a major role in immunity and maintaining vulvovaginal health by creating a low pH environment that is inhospitable to transient or invading organisms from taking foothold. Although what constitutes a “healthy vulvovaginal microbiome” has not been clearly defined, the concept of dysbiosis is understood as a condition attributed to a microbial imbalance or impaired microbiota where the naturally occurring dominant species are outcompeted by overgrowth of species associated with illness and disorders such as bacterial vaginosis and yeast infection.
A healthy vaginal microbiome is dominated by
Lactobacillus: Lactobacillus iners, Lactobacillus crispatus, Lactobacillus jensenii, and Lactobacillus gasseri [
5]. These organisms support a healthy vaginal environment by mechanisms such as the natural production of lactic acid and maintaining a low pH, secreting bacteriocins, competing for nutrients and receptors, and contributing to innate immunity via hormonal cycling induced by glycogen release and continual sloughing of epithelial cells [
6,
7,
8]. Healthy women can also have non-
Lactobacillus dominated vaginal and/or vulvar flora, such as
Atopobium, Corynebacterium, Anaerococcus, Peptoniphilus, Prevotella, Mobiluncus, Gardnerella, and
Sneathia [
6,
9,
10].
Supported by international guidelines [
11,
12], daily gentle cleansing of the vulva is important for feminine hygiene and overall intimate health. Routine washing of the vulva prevents the accumulation of vaginal discharge, urine, and fecal contamination to avoid offensive body odor and help maintain healthy vulvar skin for defense against infection. Gentle, proper cleansing care of the vulvar skin is essential, as harsh soaps may irritate this sensitive skin area. Studies of repeated washing of the skin with common alkaline soaps and synthetic detergents have shown that even minor differences in pH of the cleansing products affects both the skin surface pH and bacterial microflora [
13,
14,
15]. While there has been a recent increase in the availability of intimate hygiene products for cleanliness and odor control, some of these products may upset the natural pH of the vulvovaginal area, which may subsequently impact the composition of the normal vulvovaginal microbiota that is needed for protection against infection [
16]. In vitro data also suggest that some over-the-counter vaginal products may negatively affect beneficial
Lactobacillus species in the normal vaginal flora and alter the vaginal immune barrier [
16,
17,
18], which are needed for protection against infection.
Thus, ideal female intimate hygiene products that support intimate health should be specifically formulated and clinically tested for the vulvovaginal area and need to be mild and hypoallergenic, provide protection against dryness, and also maintain the natural pH and microflora. Due to the risks associated with internal washing or douching, external feminine washes are considered more appropriate and may serve as beneficial adjunct therapy in women with vaginal infections or taking antibiotics [
19].
A novel gel wash formulation containing lactic acid (pH 4.2) intended for daily external use was formulated to provide gentle cleansing, freshness, and antimicrobial protection to help maintain a healthy balance of the intimate skin area for women. The gel wash contains 2% lactic acid, a key natural antimicrobial ingredient that correlates with vulvovaginal health, as lactic acid helps maintain an acidic pH in the vulvovaginal area to augment skin homeostasis and prevent the growth of harmful bacteria [
20,
21,
22]. The formulation was tested in vitro following standardized ASTM (American Society for Testing and Materials) test methodology (ASTM E2315-03 Standard Guide for Assessment of Antimicrobial Activity Using a Time-Kill Procedure) and demonstrated effective antimicrobial activity (minimum of 1 log kill) against fungi (
Candida albicans), Gram-positive (including activity against Group B
Streptococcus and
Staphylococcus aureus) and Gram-negative bacteria (including
Escherichia coli; data not shown).
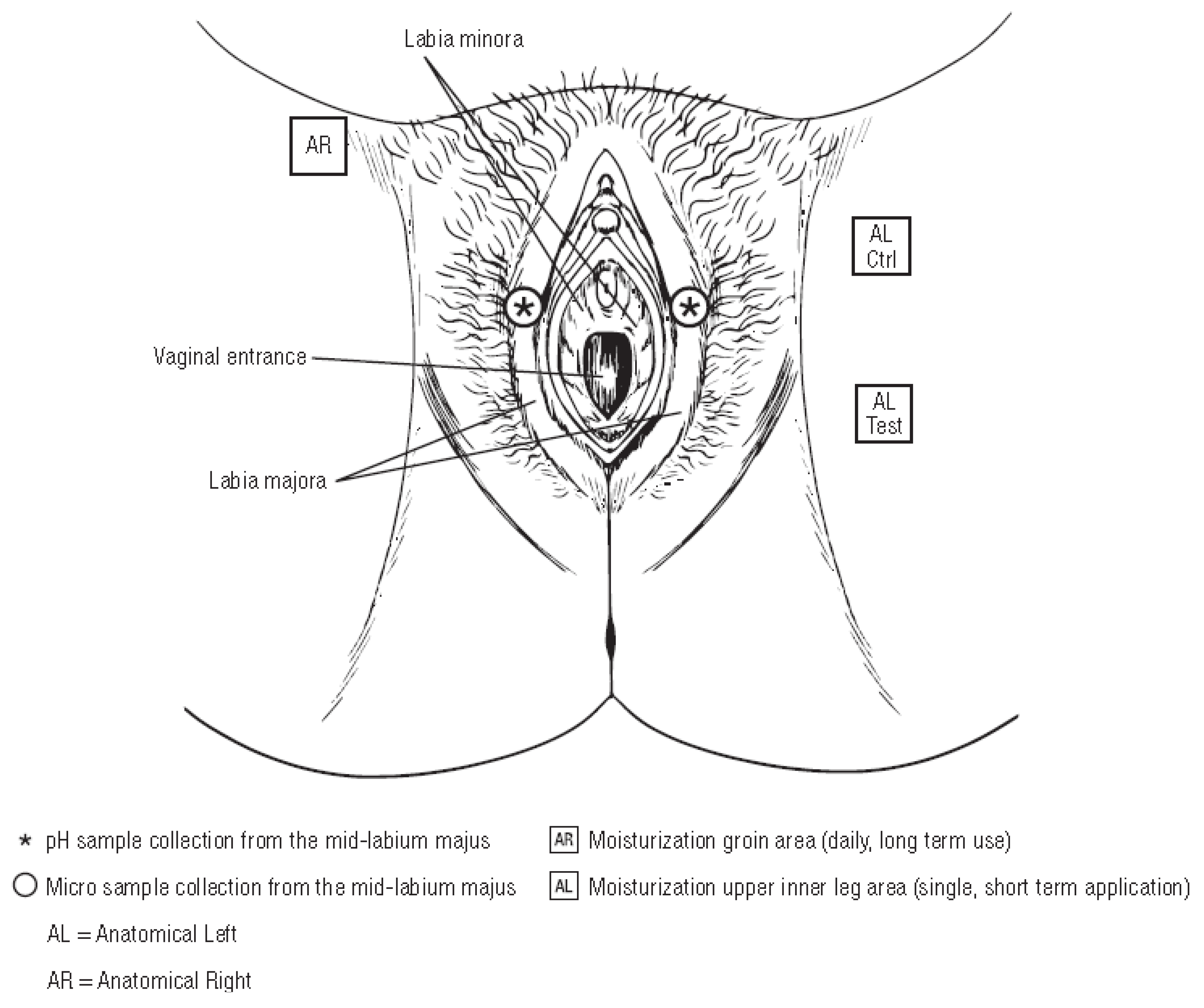
The current clinical study sought to assess, under gynecologist direction, tolerance of this new gel wash when used on the external genital area at least once daily over a 4 week period; the study also evaluated subjective tolerance, skin moisturization, and vulvar skin pH. In addition, this study assessed the impact of the gel wash on the vulvar microbiome by measuring species richness and diversity of the bacterial and fungal communities using genetic sequencing techniques. Respect for the natural pH and microflora of the skin is important for the health of the skin and vulvovaginal area. To our knowledge, this is the first time that the effect of an intimate cleansing product on vulvar skin pH and its associated microbiota has been studied this extensively in a broad age population by performing bacterial 16S rRNA gene and fungal internal transcribed spacer (ITS) microbial diversity analysis on the resident bacterial and fungal communities.
3. Discussion
Feminine hygiene is an important component of women’s overall intimate health. Because harsh soaps and surfactants may irritate the sensitive vulvar skin, intimate feminine wash products should be formulated and tested specifically for the vulvar area to ensure they are gentle enough for daily use and also respect the pH and natural microflora of the external genital area. Improper hygiene product usage and hygiene habits can lead to common vulvovaginal disorders [
1].
Under gynecologist control, this clinical study evaluated objective and subjective tolerance, pH, skin moisturization, and microbiological diversity of the vulvar area when a new lactic acid-containing gel wash was used at least once daily by women between the ages of 19 and 55 years to wash the external genital area. This new lactic acid-containing gel wash was specifically formulated to achieve a product pH of 4.2 to be compatible with the normal skin pH range and to help maintain vulvovaginal skin homeostasis and provide protection against harmful bacteria. As mentioned above, a healthy vagina is dominated by
Lactobacillus, a non-sporing, Gram-positive bacillus that produces lactic acid, resulting in a characteristic acidic environment (pH 3–4). The vaginal mucosa is also a rich source of lactic acid, which is produced during estrogen-regulated anaerobic glucose metabolism [
23]. Vaginal lactic acid is important, as it correlates with vaginal health by inhibiting the growth of bacteria known to cause vulvovaginal infections [
20] and may also play a part in local immune defense [
24]. External feminine washes, particularly those containing lactic acid and formulated to an acidic pH that enhances skin homeostasis, are considered more appropriate than internal washes or douches and may be a useful adjunct therapy for women with vaginal infections or taking antibiotics [
21].
In this clinical study, gynecologist assessment of the vulvar skin during test product use showed acceptable tolerance, as the study population showed no signs of increased dryness, redness, edema, itching, stinging, or burning, and the gynecologist concluded that the product was well tolerated by most women. Previous studies evaluated the effects of sodium lauryl sulfate (SLS, a common surfactant in body wash and feminine wash products) on vulvar skin. While irritant reactions were inconsistent (none in 1 study with low-concentration SLS [0.1–1.0%], but 50% developed irritant dermatitis in another study [SLS 2–5%]), decreases in vulvar skin stratum corneum hydration was observed in both studies [
25,
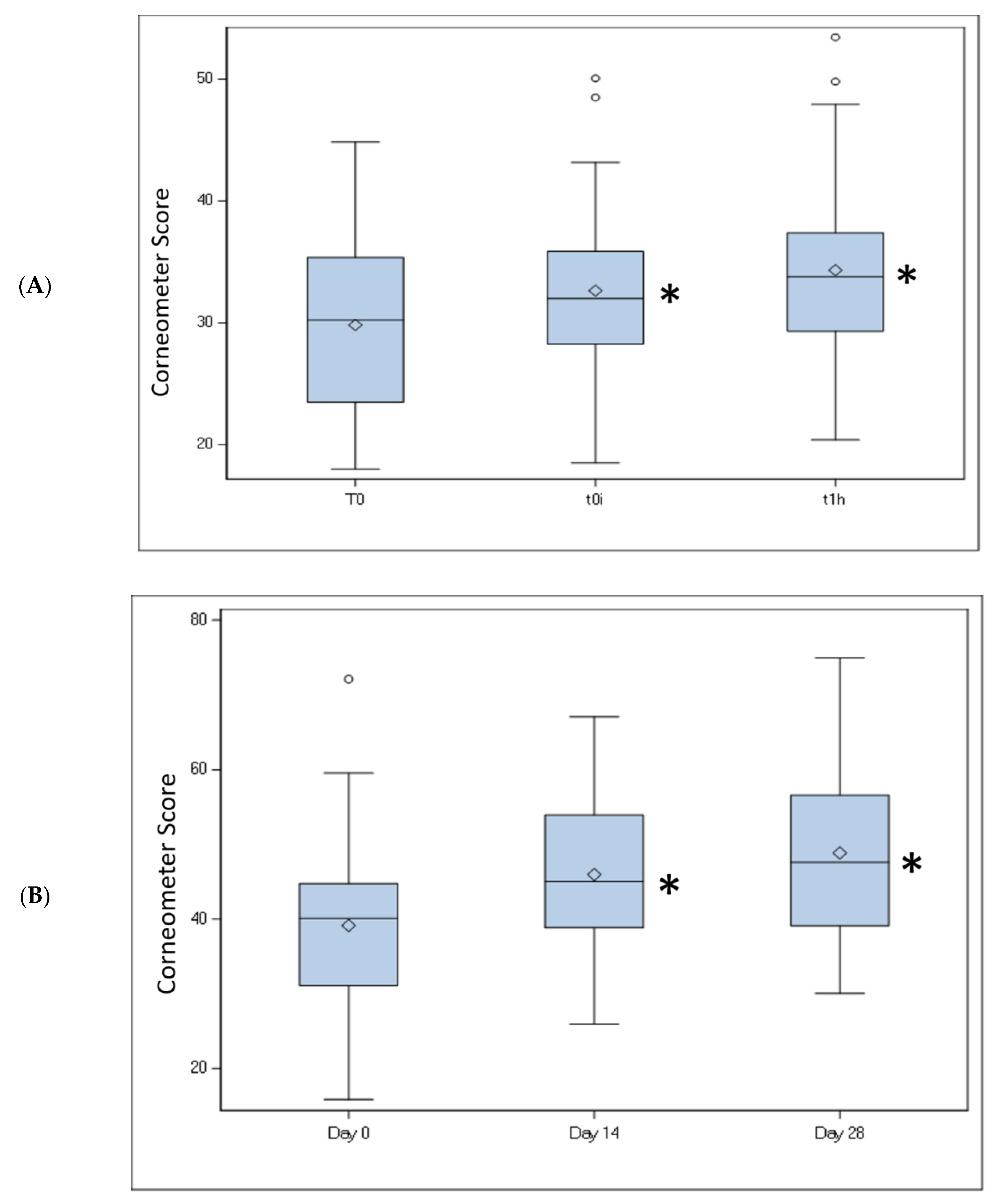
26]. Use of the gel wash in this study, however, was associated with significant increases in both short-term (single application) and longer-term (daily use for 28 days) skin moisturization, potentially reducing the possibility of skin dryness and irritation.
Maintaining the natural pH of the skin is important, as it is integral to the maintenance of the natural microflora, which is essential to protect against invading pathogenic organisms and infections [
27,
28,
29,
30]. In the current study with the new gel wash optimally formulated at pH 4.2 to be compatible with healthy skin pH, there was no significant change in vulvar skin pH for the overall population over time with daily product use. Vulvar skin pH in the overall study population ranged from 5.5 to 5.8 from baseline through product use. This is higher than the acidic range (~3.8–4.4) reported for the vagina during reproductive ages, but consistent with the literature, which reports natural skin pH in the acidic range (pH, 4.5–6.0) [
27,
28,
29], and with other studies specifically evaluating vulvar skin pH. In a study of the vulvar skin of healthy Thai women, skin pH was acidic at all sites, but was higher around the vulvar area (labia, mons pubis; pH > 5) than at the other two control sites (inner thigh, forearm; pH ≤ 5) [
31]. In a study of healthy Japanese women, skin pH was similar between sites (labia, groin, mons pubis, and inner thigh), with a mean pH of 6.0 [
32]. Previous studies showed even small changes in the pH from skin cleansing products may affect skin surface pH and microflora when tested on the forehead and forearm [
13]. The current study shows this new lactic acid-containing gel wash optimally formulated to skin pH maintains the vulva skin pH over time after repeated daily washes.
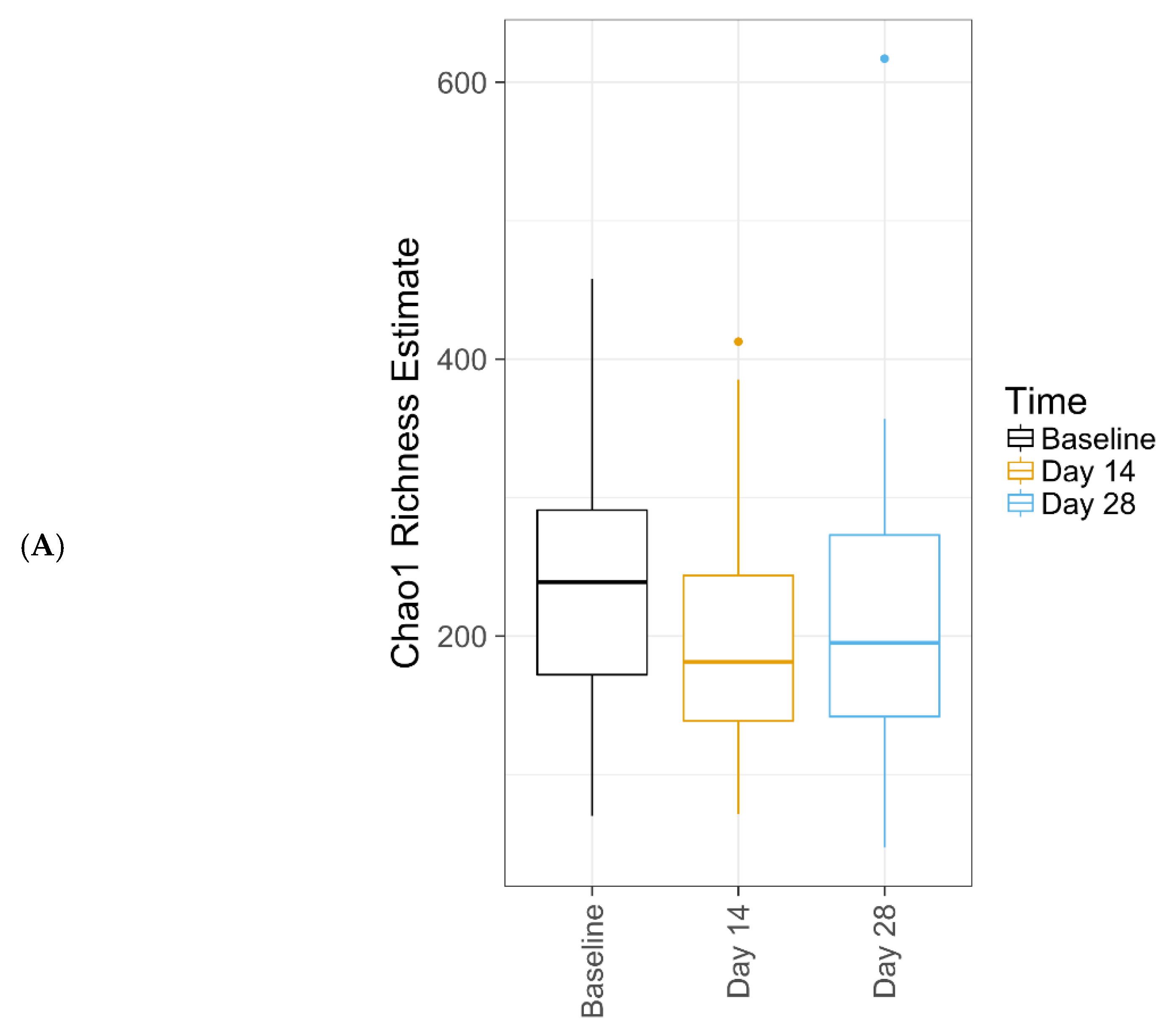
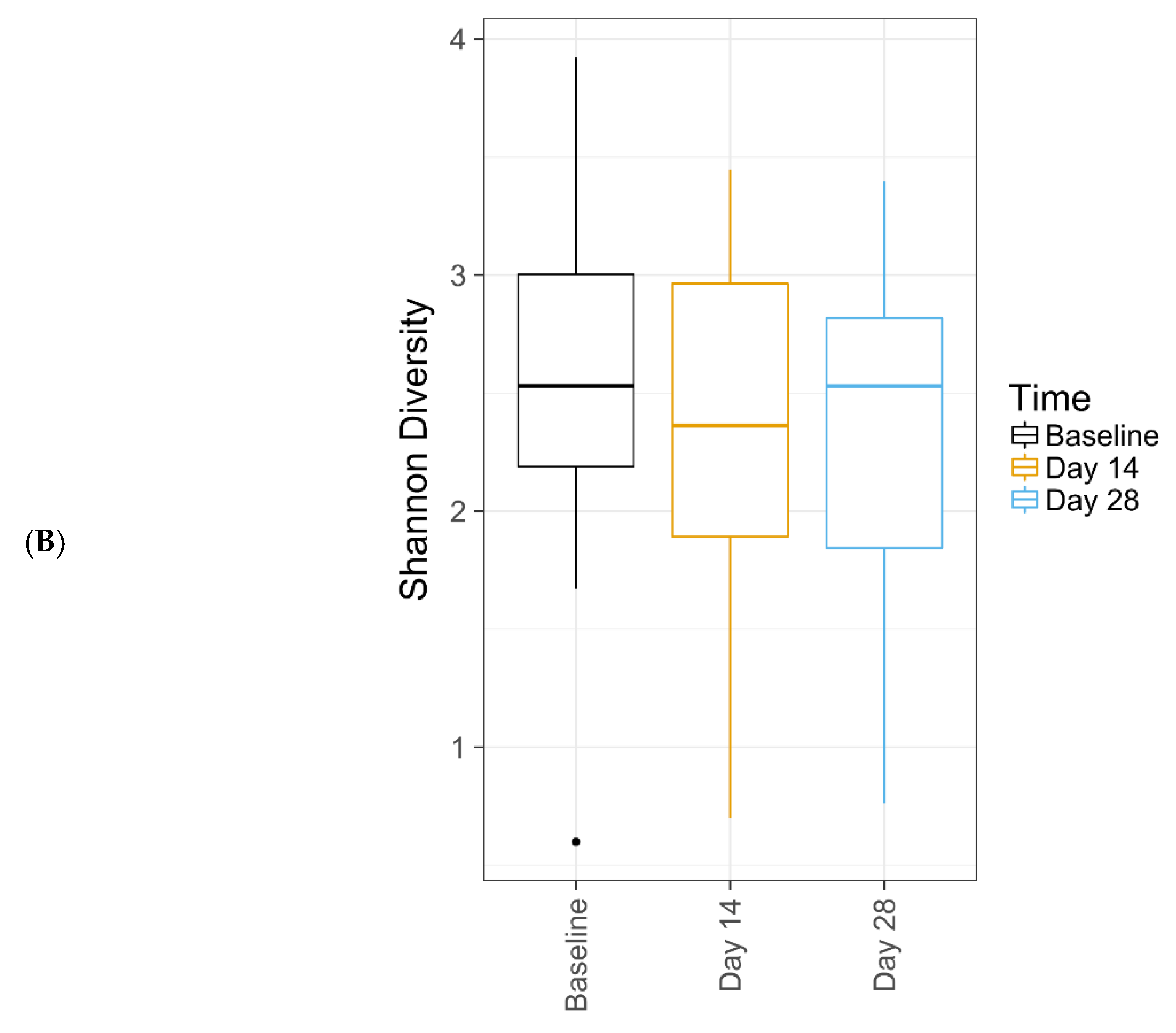
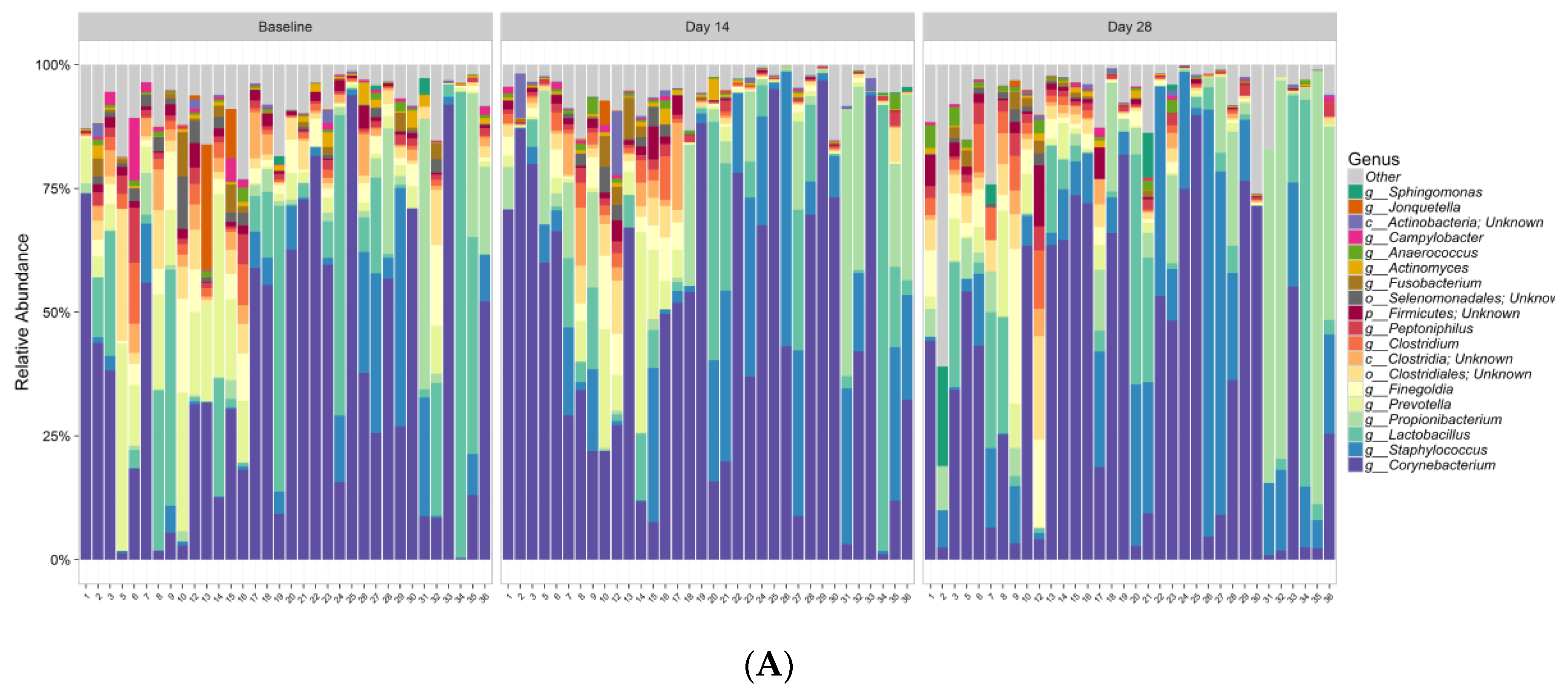
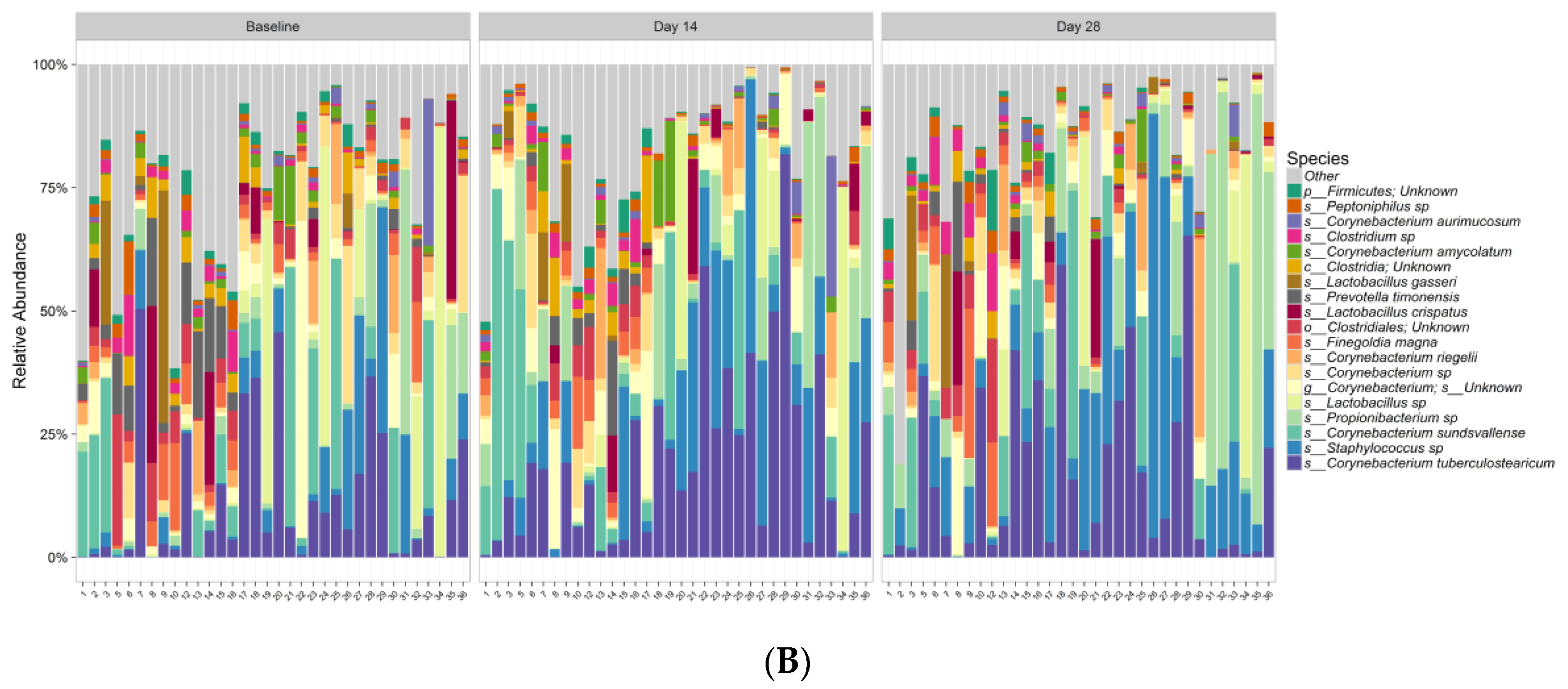
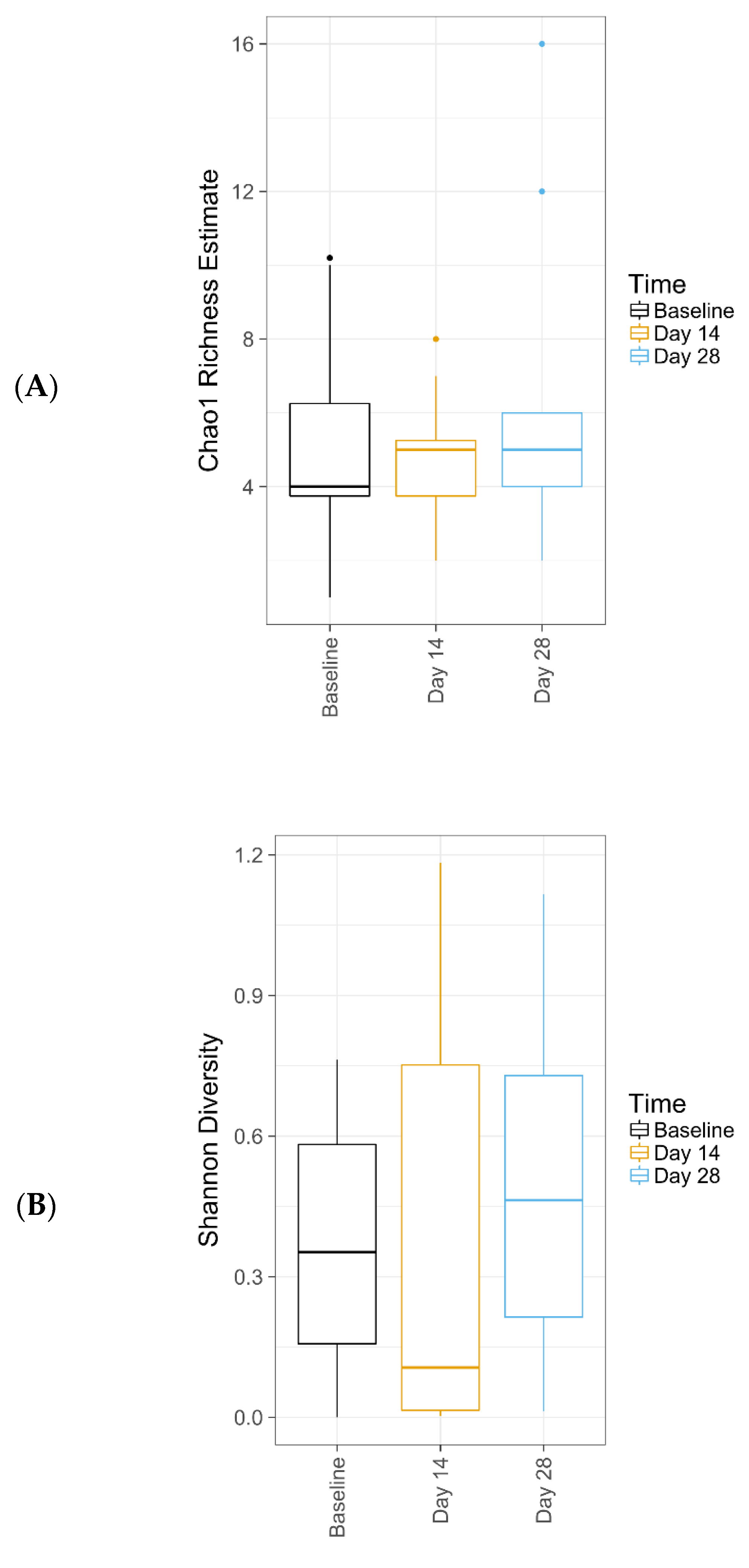
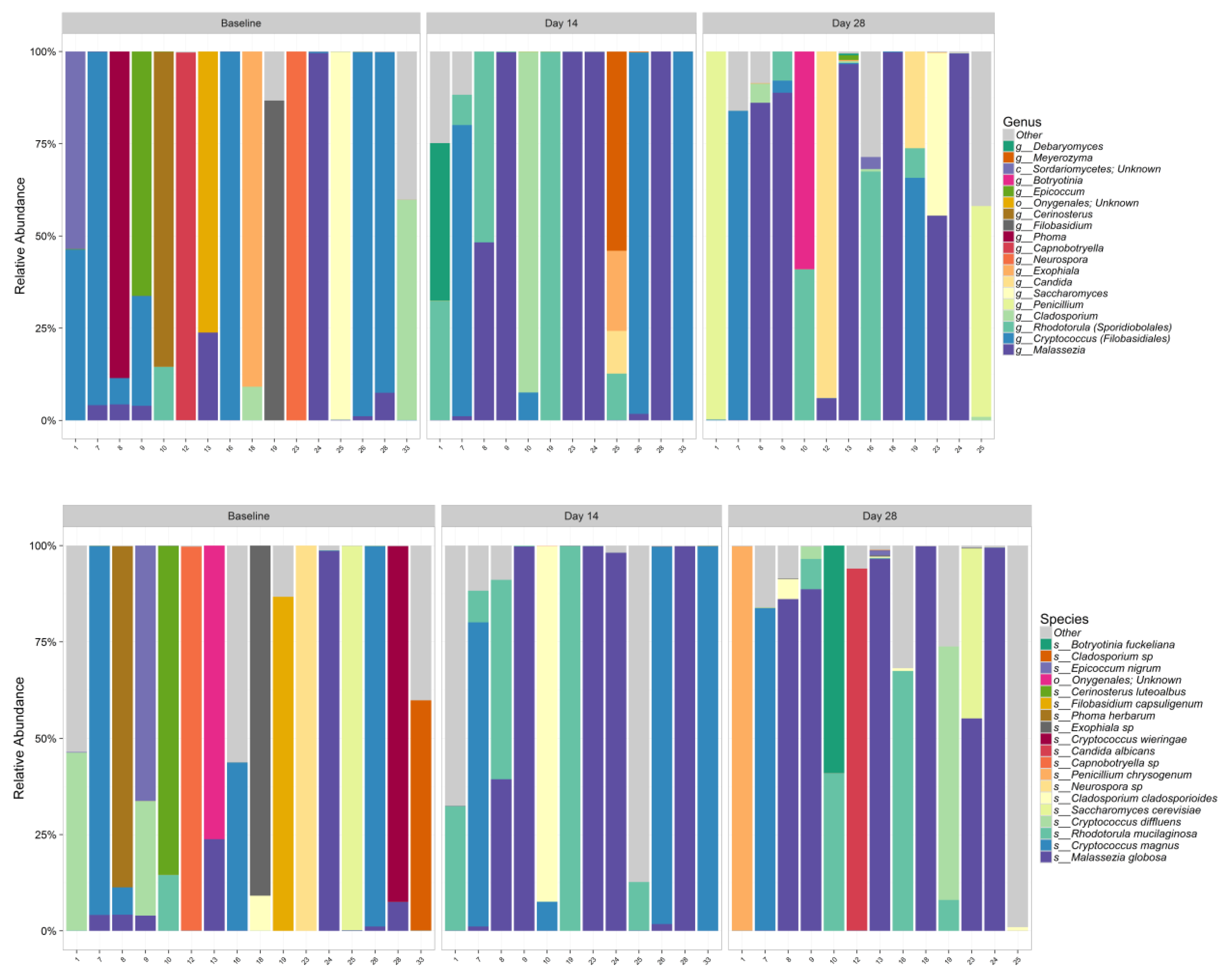
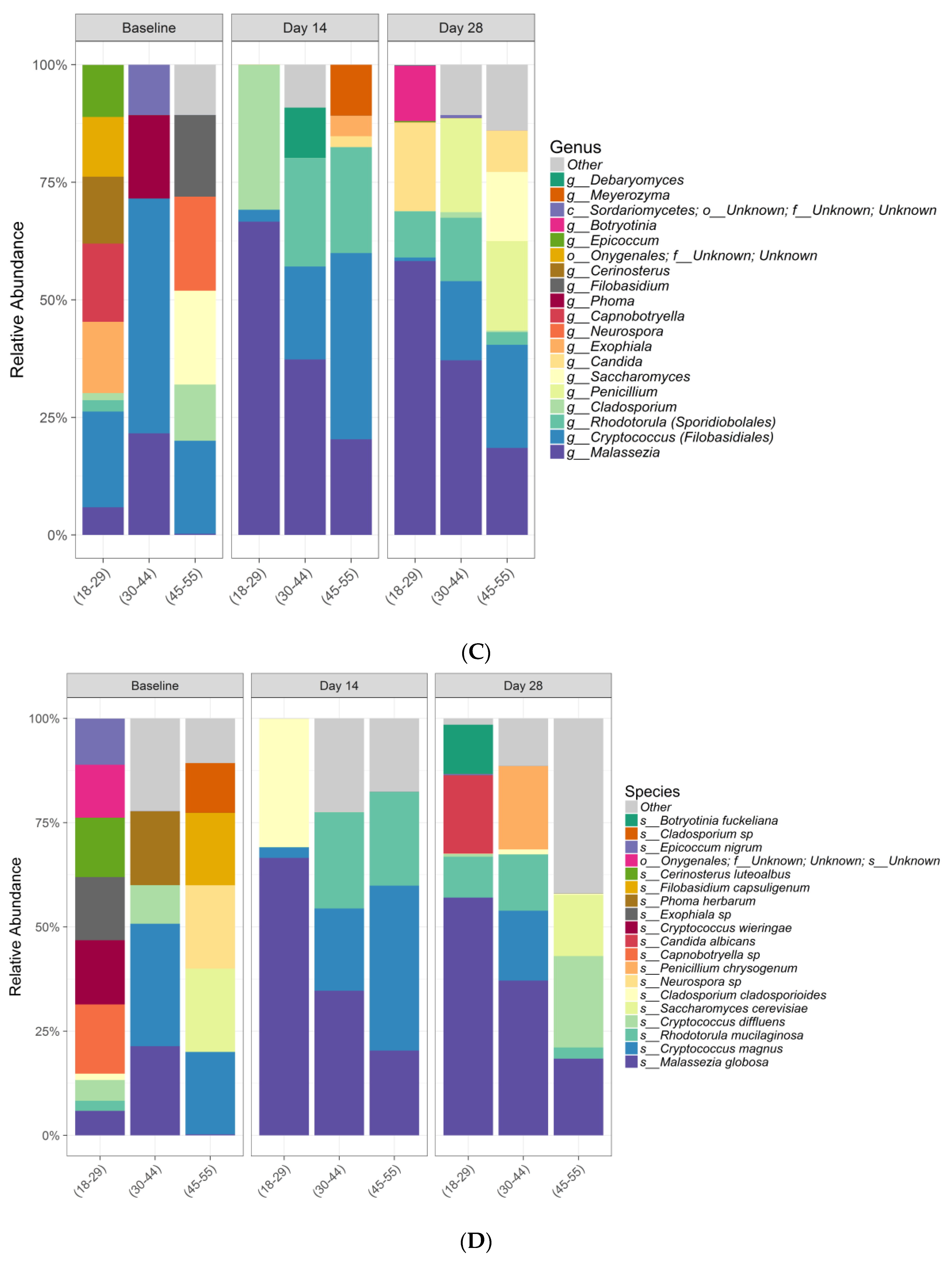
Further, this study demonstrates that this new lactic acid-containing gel wash respects the natural flora of the vulvar skin. Genetic sequencing techniques revealed that the new gel wash did not affect natural microbiome species richness or diversity. Pre- and post-test product use, the predominant bacterial genera were
Corynebacterium,
Staphylococcus,
Lactobacillus,
Actinobacteria,
Prevotella,
Clostridia, and
Propionibacterium and the predominant fungi were
Malassezia,
Cryptococcus,
Rhodoturula,
Cladosporium,
Saccharomyces, and
Penicillium. As previously discussed, the skin microbiome varies by skin site and physiology, with specific species associated with moist, dry, and sebaceous microenvironments [
4], and the vulvar skin, while not extensively studied, likely represents a combination of organisms known to inhabit moist (
Staphylococcus and
Corynebacterium spp.), sebaceous (
Propionibacterium and
Malassezia spp.), and dry (
Actinobacteria spp.) areas. The predominant organisms found in our clinical study are similar to those found previously with culture-based techniques using the same detergent scrub method for sampling the vulvar skin (lipophilic and non-lipophilic diphtheroids [including
Corynebacterium], coagulase negative
Staphylococcus, micrococci,
Lactobacilli,
Streptococci, gram-negative rods, and yeasts) [
33]. The percent incidence reported for yeast was 39% for the vulva, consistent with our finding, where 16 out of 34 subjects have shown fungal amplification. Results using non-culture based methods showed a distinctive microbiota for each individual woman tested. Contributing factors that could affect the composition of vulvar communities may include different habits and practices (e.g., frequency of bathing, kinds of clothing worn); microbiota of the labia majora included species found on the skin, including
Staphylococcus and
Corynebacterium [
9]. In a study of healthy Japanese women, the most predominant species found on the pubic area skin (labia and groin, mons pubis, and inner thigh) were
Lactobacillus spp. and
Staphylococcus epidermidis, followed by
S. aureus [
32].
Propionibacterium acnes was detected in almost all women at all sites, but was less abundant than
S. aureus, while
Prevotella spp. was detected in the labia and groin but not in other sites based on reverse transcription PCR [
32].
Particularly noteworthy, despite the antimicrobial action demonstrated in vitro, the gel wash had no significant impact on commensal species richness or diversity of the vulvar skin microbiome; in other words, the wash helped to maintain the natural flora of the external genital area. Grice and Segre [
4] discuss that maintaining the delicate balance between the skin, in this case the vulvar skin, and the diverse collection of microorganisms that regularly inhabit the area, is important and can be impacted by endogenous (e.g., genetic variations) and exogenous (e.g., environmental) factors. More specifically, soaps and other hygiene products with high surfactant and high pH have the potential to negatively affect the composition of the skin microbiome.
Ecological body site niche is a greater determinant of microbiota composition than the individual genetic variation among healthy volunteers. In the current study, a similar microbiota was observed among healthy volunteers for this same site. In a systematic, multi-site metagenomic study of the skin from 15 healthy adults from 18 defined anatomical skin sites, Oh and colleagues [
34] concluded that both biogeography and individuality influence skin microbial composition and function.
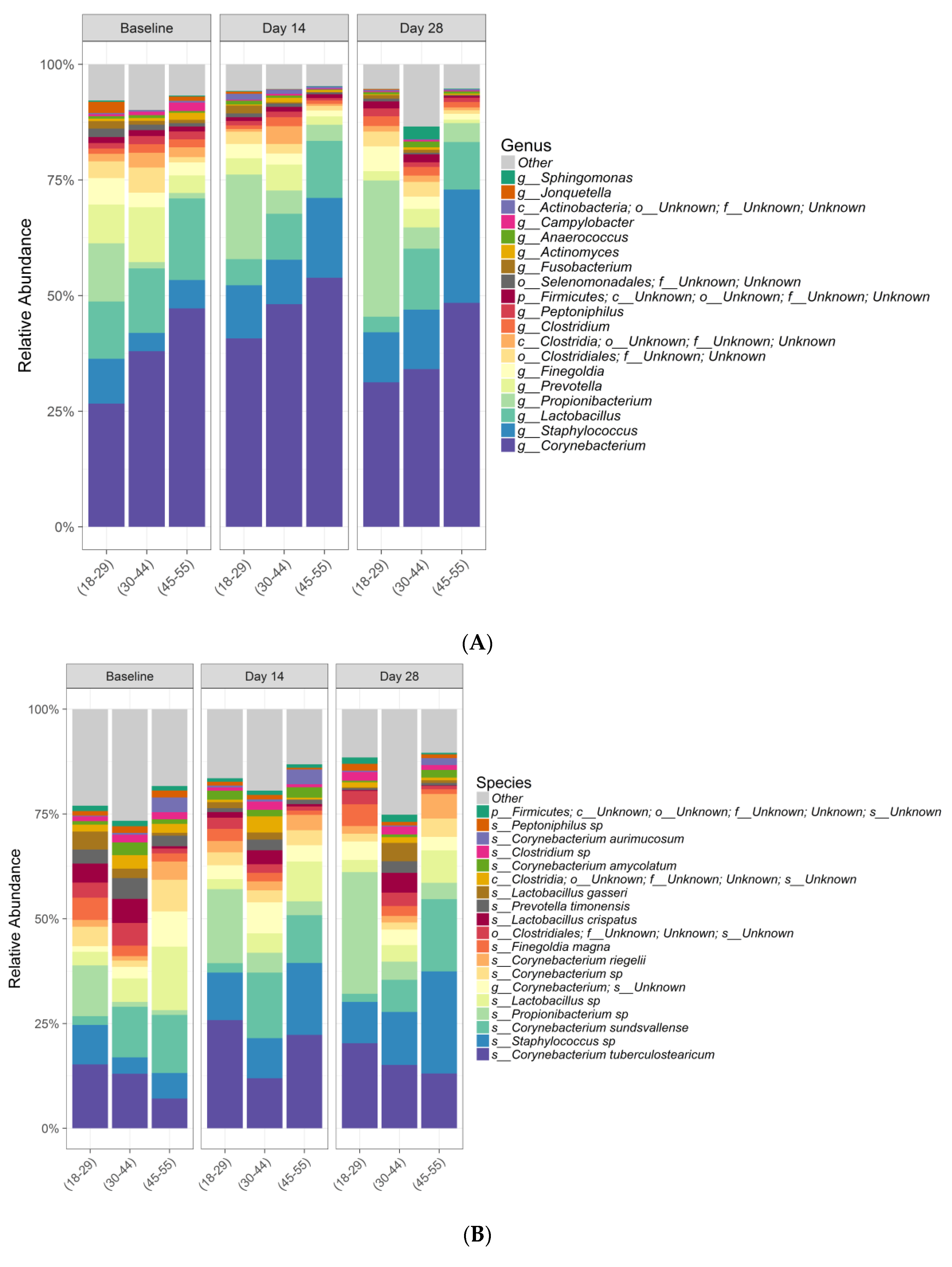
Interestingly, differences in beta diversity metrics among age groups overall were observed, with substantial interindividual variation in relative abundances of bacterial and fungal species within age groups. At baseline, considerable differences were observed in relative abundance plots at both genera and species level, especially in the fungal community. This could suggest that age is an important factor to influence microbiome composition. However, future studies with more subject numbers in each age group is warranted to confirm this interesting observation.
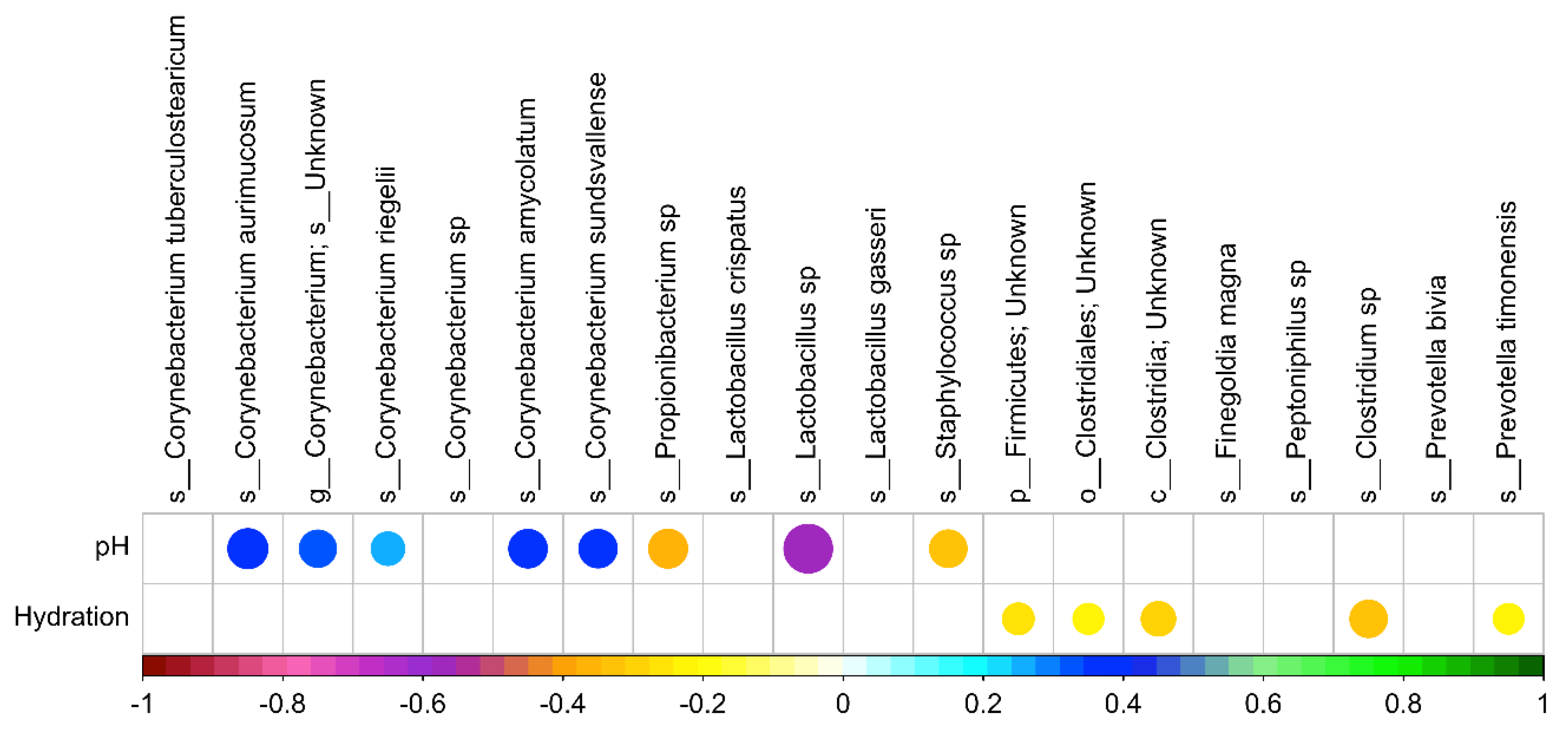
Correlation analysis was performed between skin pH and moisture levels with the top bacterial taxa, as scientific investigations have shown that applying specific probiotic strains to skin, such as
Staphylococcus epidermidis, can significantly improve skin moisture retention by increasing the lipid content and suppressing the water evaporation from the skin [
35]. Our data suggest that several coryneforms are less abundant in lower pH conditions whereas
Propionibacterium,
Staphylococcus, and
Lactobacillus species thrive better in a lower pH environment. Predominantly the
Clostridia and
Clostridium species showed a decrease with increase in moisturization.
A healthy vulvovaginal environment is particularly important for a pregnant woman to protect the baby from potential infection risk. Protection against Group B
Streptococcus is particularly important for pregnant women as it often colonizes the vagina through the gastrointestinal tract and increases the risk of preterm delivery, neonatal meningitis, and even fetal death. The association of harmful vaginal bacteria, i.e., bacterial vaginosis, with preterm labor and preterm birth is also supported by the literature [
1]. Birth mode (cesarean vs. vaginal birth) can have long-term effects on microbial diversity [
36], but data suggest that the skin microbiome of a cesarean-born infant can be at least partially restored through vaginal microbial transfer to what would be expected following vaginal birth [
37]. In contrast, Chu and colleagues showed that while the infant microbiome matures and changes over the first 6 weeks of life, the variations observed are primarily due to body site and not mode of delivery [
38]. Despite inconclusive data, pregnant women should be cautious in choosing hygiene products because they might impact the early-life microbiome, which is critical for development of infant immune function [
39].
Results from the current study should be considered in relation to the study limitations. First, there were no previous vulvar microbiome data to power the study, although data from another internal study assessing the effects of a cosmetic product on the skin pH were used to determine an appropriate subject sample size. The sample size selected for this study meets the requirements of CLT in order to sufficiently sample the population. Second, bacterial 16S rRNA gene and fungal ITS sequencing is limited to reference sequences that are present in the database and low sequence homology within those regions to allow for differentiation between taxonomic groups versus shotgun metagenomics or metatranscriptomics that can also elucidate function. Finally, this was a purely Caucasian population and was geographically limited to the United Kingdom. An additional study using shotgun metagenomics and/or metatranscriptomics including different subjects of different ethnicities is warranted to obtain complete information. Further study specific to postmenopausal women may also be of interest, as vulvovaginal dryness and infections can become increasing concerns for this subgroup of women.