Last Call for Replacement of Antimicrobials in Animal Production: Modern Challenges, Opportunities, and Potential Solutions
Abstract
1. Introduction
2. Antimicrobials and Antibiotics: Why Did Antimicrobial Resistance Appear?
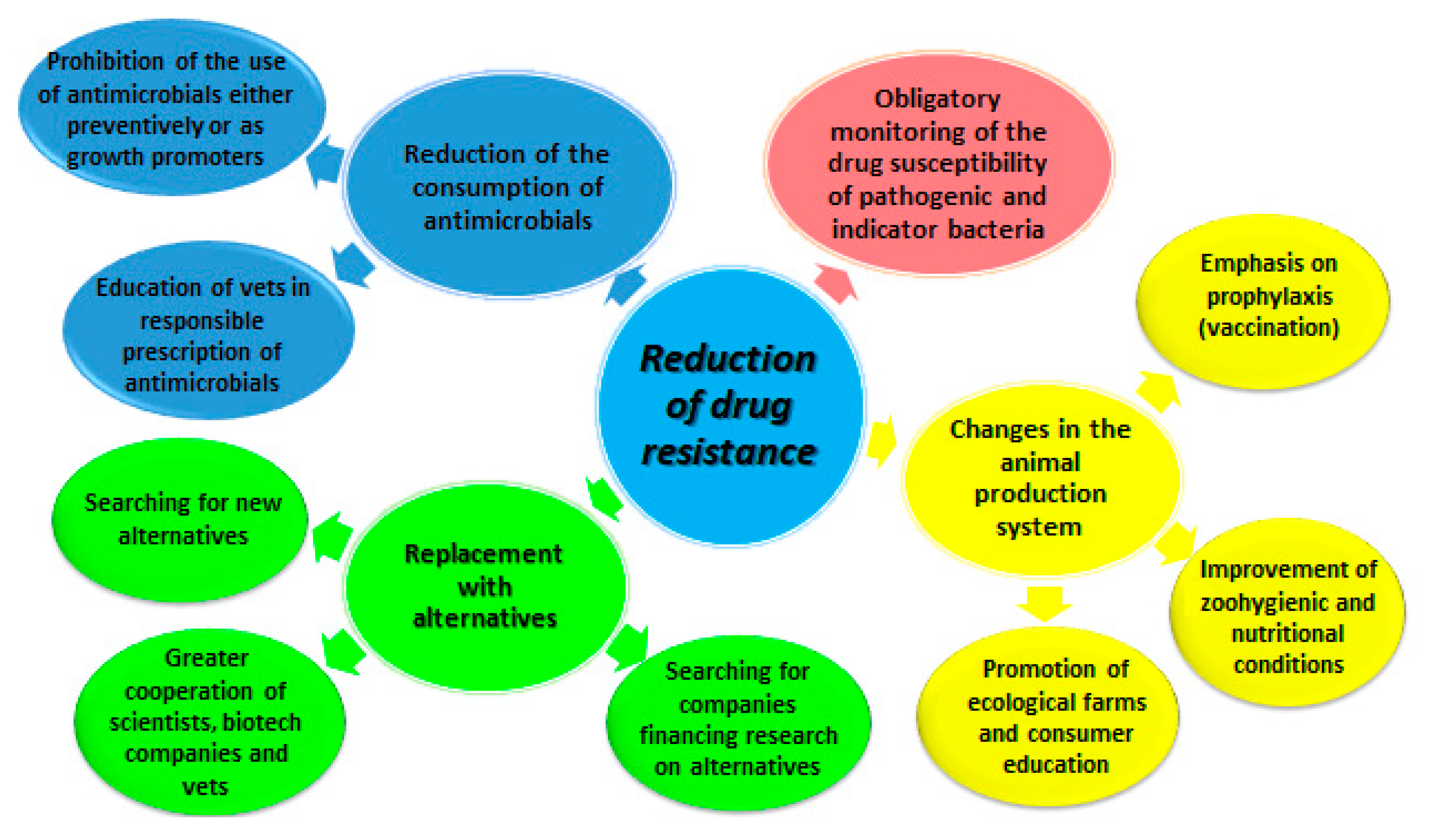
3. Yesterday and Today: Activities for Elimination of the Effects of Excessive Antibiotic Therapy
4. Factors Related to the Production and Implementation of Alternatives to Antimicrobials
5. Alternatives and Their Applicability in Animal Production
6. Bacteriophages
7. Antimicrobial Peptides
8. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Jackson, N.; Czaplewski, L.; Piddock, L.J.V. Discovery and development of new antibacterial drugs: Learning from experience? J. Antimicrob. Chemother. 2018, 73, 1452–1459. [Google Scholar] [CrossRef] [PubMed]
- Payne, D.; Gwynn, M.; Holmes, D.; Pompliano, D.L. Drugs for bad bugs: Confronting the challenges of antibacterial discovery. Nat. Rev. Drug. Discov. 2007, 6, 29–40. [Google Scholar] [CrossRef] [PubMed]
- Castanon, J.I.R. History of the use of antibiotic as growth promoters in European poultry feeds. Poultry Sci. 2007, 86, 2466–2471. [Google Scholar] [CrossRef] [PubMed]
- Kirchhelle, C. Pharming animals: A global history of antibiotics in food production (1935–2017). Palgrave Commun. 2018, 4, 96. [Google Scholar] [CrossRef]
- Raport of IACG 2019. No Time to Wait: Securing the Future from Drug-Resistant Infections. Available online: https://www.who.int/antimicrobial-resistance/interagency-coordination-group/IACG_final_summary_EN.pdf?ua=1 (accessed on 10 September 2020).
- The 2019 WHO. AWaRe Classification of Antibiotics for Evaluation and Monitoring of Use; World Health Organization: Geneva, Switzerland, 2019. [Google Scholar]
- Meek, R.W.; Vyas, H.; Piddock, L.J.V. Nonmedical uses of antibiotics: Time to restrict their use? PLoS Biol. 2015, 13, e1002266. [Google Scholar] [CrossRef]
- Raport from PEW Charitable Trust. Alternatives to Antibiotics in Animal Agriculture. 2019. Available online: https://www.pewtrusts.org/~/media/assets/2017/07/alternatives_to_antibiotics_in_animal_agriculture.pdf (accessed on 10 September 2020).
- Hume, M.E. Historic perspective: Prebiotics, probiotics, and other alternatives to antibiotics, Food Safety Symposium: Potential impact of reduced antibiotic use and the roles of prebiotics, probiotics, and other alternatives in antibiotic-free broiler production. Poultry Sci. 2011, 90, 2663–2669. [Google Scholar] [CrossRef]
- Cheng, G.; Hao, H.; Xie, S.; Wang, X.; Dai, M.; Huang, L.; Yuan, Z. Antibiotic alternatives:the substitution of antibiotics in animal husbandry? Front. Microbiol. 2014, 5, 217. [Google Scholar] [CrossRef]
- Greenwood, D. Chapter 1—Historical introduction. In Antibiotic and Chemotherapy, 9th ed.; Elsevier Ltd.: Amsterdam, The Netherlands, 2010; pp. 2–9. [Google Scholar] [CrossRef]
- McManus, P.S.; Stockwell, V.O. Antibiotic use for plant disease management in the United States. Plant Health Prog. 2001. [Google Scholar] [CrossRef]
- Deatherage, F.E. Use of antibiotics in the preservation of meats and other food products. Am. J. Public Health 1957, 47, 594–600. [Google Scholar] [CrossRef]
- Ventola, C. L The antibiotic resistance crisis. Part 1: Causes and threats. Pharm. Ther. 2015, 40, 277–283. [Google Scholar]
- Kirchhelle, C. Swann song British antibiotic regulation in livestock production (1953–2006). Bull. Hist. Med. 2018, 92, 317–350. [Google Scholar] [CrossRef]
- Williams Smith, H. Drug-resistant bacteria in domestic animals (presentation in section of epidemiology and preventive medicine. symposium on epidemiological risks of antibiotics, 21.02.1958). Proc. R. Soc. Med. 1958, 51, 812–813. [Google Scholar]
- Laxminarayan, R.; Van Boeckel, T.; Teillant, A. The Economic Costs of Withdrawing Antimicrobial Growth Promoters from the Livestock Sector. OECD Food Agricult. Fish. Pap. 2015, 78. [Google Scholar] [CrossRef]
- National Research Council. The Effects on Human Health of Subtherapeutic Use of Antimicrobials in Animal Feeds. Committee to Study the Human Health Effects of Subtherapeutic Antibiotic Use in Animal Feeds; NRC: Washington, DC, USA, 1980. [Google Scholar] [CrossRef]
- Manten, A. The non-medical use of antibiotics and the risk of causing microbial drug-resistance. Bull. World Health Organ. 1963, 29, 387–400. [Google Scholar]
- Marshall, B.M.; Levy, S.B. Food animals and antimicrobials: Impacts on human health. Clin. Microbiol. Rev. 2011, 24, 718–733. [Google Scholar] [CrossRef] [PubMed]
- Davies, J.; Davies, D. Origins and evolution of antibiotic resistance. Microbiol. Mol. Biol. R. 2010, 74, 417–433. [Google Scholar] [CrossRef]
- Watanabe, T. Infective heredity of multiple drug resistance in bacteria. Bacteriol. Rev. 1963, 27, 87–115. [Google Scholar] [CrossRef] [PubMed]
- Harkins, C.P.; Pichon, B.; Doumith, M.; Parkhill, J.; Westh, H.; Tomasz, A.; de Lencastre, H.; Bentley, S.D.; Kearns, A.M.; Holden, M.T.G. Methicillin-resistant Staphylococcus aureus emerged long before the introduction of methicillin into clinical practice. Genome Biol. 2017, 18, 130. [Google Scholar] [CrossRef]
- Aponte-González, J.; González-Acuña, A.; Lopez, J.; Brown, P.; Eslava-Schmalbach, J. Perceptions in the community about the use of antibiotics without a prescription: Exploring ideas behind this practice. Pharm. Pract. 2019, 17, 1394. [Google Scholar] [CrossRef]
- Kirchhelle, C. Toxic confusion: The dilemma of antibiotic regulation in West German food production (1951–1990). Endeavour 2016, 40, 114–127. [Google Scholar] [CrossRef]
- Roca, I.; Akova, M.; Baquero, F.; Carlet, J.; Cavaleri, M.; Coenen, S.; Cohen, J.H.M.; Findlay, D.; Gyssens, I.C.J.; Heure, O.E.; et al. The global threat of antimicrobial resistance: Science for intervention. New Microbes New Infect. 2015, 6, 22–29. [Google Scholar] [CrossRef] [PubMed]
- Casewell, M.; Friis, C.; Marco, E.; McMullin, P.; Phillips, J. The European ban on growth-promoting antibiotics and emerging consequences for human and animal health. J. Antimicrob. Chemoth. 2003, 52, 159–161. [Google Scholar] [CrossRef] [PubMed]
- Markestad, A.; Grave, K. Reduction of antibacterial drug use in Norwegian fish farming due to vaccination. Dev. Biol. Stand. 1997, 90, 365–369. [Google Scholar] [PubMed]
- Acar, J.; Casewell, M.; Freeman, J.; Friis, C.; Goossens, H. Avoparcin and virginiamycin as animal growth promoters: A plea for science in decision-making. Clin. Microbiol. Infec. 2000, 6, 477–482. [Google Scholar] [CrossRef] [PubMed]
- Elstrøm, P.; Astrup, E.; Hegstad, K.; Samuelsen, Ø.; Enger, H.; Kacelnik, O. The fight to keep resistance at bay, epidemiology of carbapenemase producing organisms (CPOs), vancomycin resistant enterococci (VRE) and methicillin resistant Staphylococcus aureus (MRSA) in Norway, 2006–2017. PLoS ONE 2019, 14, e0211741. [Google Scholar] [CrossRef]
- WHO (World Health Organisation). Global Priority List of Antibiotic-Resistant Bacteria to Guide Research, Discovery, and Development of New Antibiotics. 2017. Available online: https://www.who.int/medicines/publications/global-priority-list-antibiotic-resistant-bacteria/en/ (accessed on 12 August 2020).
- Millet, S. The European ban on antibiotic growth promoters in animal feed: From challenges to opportunities. Vet. J. 2011, 187, 143–144. [Google Scholar] [CrossRef]
- EMA (European Medicines Agency); EFSA (European Food Safety Authority). EMA and EFSA Joint Scientific Opinion on measures to reduce the need to use antimicrobial agents in animal husbandry in the European Union, and the resulting impacts on food safety (RONAFA). EFSA J. 2017, 15, 245. [Google Scholar] [CrossRef]
- European Medicines Agency, European Surveillance of Veterinary Antimicrobial Consumption. Sales of Veterinary Antimicrobial Agents in 31 European Countries in 2017; European Medicines Agency: Amsterdam, The Netherlands, 2019. [Google Scholar]
- European Food Safety Authority and European Centre for Disease Prevention and Control. The European Union summary report on antimicrobial resistance in zoonotic and indicator bacteria from humans, animals and food in 2017/2018. EFSA J. 2020, 18, 6007. [Google Scholar] [CrossRef]
- More, S.J. European perspectives on efforts to reduce antimicrobial usage in food animal production. Ir. Vet. J. 2020, 73, 2. [Google Scholar] [CrossRef]
- O’Neill, J. Antimicrobial Resistance: Tackling a Crisis for the Health and Wealth of Nations. 2014. Available online: https://amr-review.org/sites/default/files/AMR%20Review%20Paper%20-%20Tackling%20a%20crisis%20for%20the%20health%20and%20wealth%20of%20nations_1.pdf (accessed on 10 August 2020).
- Price, R.; O’Neill, J. Report on antimicrobial resistance: Funding for antimicrobial specialists should be improved. Eur. J. Hosp. Pharm. 2016, 23, 245–247. [Google Scholar] [CrossRef]
- Rabello, R.F.; Bonelli, R.R.; Penna, B.A.; Albuquerque, J.P.; Souza, R.M.; Cerqueira, A.M.F. Antimicrobial resistance in farm animals in Brazil: An update overview. Animals 2020, 10, 552. [Google Scholar] [CrossRef] [PubMed]
- WHO. Critically Important Antimicrobials for Human Medicine. 2019. Available online: https://www.who.int/foodsafety/publications/antimicrobials-sixth/en/ (accessed on 10 September 2020).
- European Commision. The New EU One Health Action Plan Against Antimicrobial Resistance. 2017. Available online: https://ec.europa.eu/health/antimicrobial-resistance/eu-action-on-antimicrobial-resistance_en (accessed on 12 September 2020).
- Regulation (EU) 2019/6 of the European Parliament and of the Council of 11 December 2018 on Veterinary Medicinal Products and Repealing Directive 2001/82/EC (Text with EEA Relevance). Available online: https://eur-lex.europa.eu/eli/reg/2019/6/oj (accessed on 3 August 2020).
- Kurt, T.; Wong, N.; Fowler, H.; Gay, C.; Lillehoj, H.; Plummer, P.; Scott, H.M.; Hoelzer, K. Strategic priorities for research on antibiotic alternatives in animal agriculture—Results from an expert workshop. Front. Vet. Sci. 2019, 6, 429. [Google Scholar] [CrossRef] [PubMed]
- Speksnijder, D.C.; Wagenaar, J.A. Reducing antimicrobial use in farm animals: How to support behavioral change of veterinarians and farmers. Anim. Front. 2018, 8, 2. [Google Scholar] [CrossRef] [PubMed]
- Ekakoro, J.E.; Caldwell, M.; Strand, E.B.; Okafor, C.C. Drivers, alternatives, knowledge, and perceptions towards antimicrobial use among Tennessee beef cattle producers: A qualitative study. BMC Vet. Res. 2019, 15, 16. [Google Scholar] [CrossRef] [PubMed]
- Gigante, A.; Atterbury, R.J. Veterinary use of bacteriophage therapy in intensively-reared livestock. Virol. J. 2019, 16, 155. [Google Scholar] [CrossRef] [PubMed]
- Kitching, M.; Mathur, H.; Flynn, J.; Byrne, N.; Dillon, P.; Sayers, R.; Rea, M.C.; Hill, C.; Ross, R.P. A live bio-therapeutic for mastitis, containing lactococcus lactis dpc3147 with comparable efficacy to antibiotic treatment. Front. Microbiol. 2019, 10, 2220. [Google Scholar] [CrossRef]
- Kierończyk, B.; Pruszyńska-Oszmałek, E.; Świątkiewicz, S.; Rawski, S.; Długosz, J.; Engberg, E.M.; Józefiak, D. The nisin improves broiler chicken growth performance and interacts with salinomycin in terms of gastrointestinal tract microbiota composition. J. Anim. Feed Sci. 2016, 25, 309–316. [Google Scholar] [CrossRef]
- Verstappen, K.M.; Tulinski, P.; Duim, B.; Fluit, A.C.; Carney, J.; Van Nes, A.; Wagenaar, J.A. The Effectiveness of Bacteriophages against Methicillin-Resistant Staphylococcus aureus ST398 Nasal Colonization in Pigs. PLoS ONE 2016, 11, e0160242. [Google Scholar] [CrossRef]
- Lhermie, G.; Wernli, D.; Jørgensen, P.S.; Kenkel, D.; Tauer, L.W.; Gröhn, Y.T. Global resistance to antimicrobials and their sustainable use in agriculture. Lancet Planet. Health 2019, 3, 109–110. [Google Scholar] [CrossRef]
- Bouchot, A.; Bordier, M. The OIE strategy to address threats at the interface between humans, animals and ecosystems. In Socio-Ecological Dimensions of Infectious Diseases in Southeast Asia; Morand, S., Dujardin, J.P., Lefait-Robin, R., Apiwathnasorn, C., Eds.; Springer: Singapore, 2015. [Google Scholar] [CrossRef]
- Laurent, J.W. Alternatives to Common Preventive Uses of Antibiotics for Cattle, Swine, and Chickens. NRDC Report. 2018. Available online: https://www.nrdc.org/resources/alternatives-common-preventive-uses-antibiotics-cattle-swine-and-chickens (accessed on 3 August 2020).
- Gallois, M.; Rothkotter, H.J.; Bailey, M.; Stokes, C.R.; Oswald, I.P. Natural alternatives to in-feed antibiotics in pig production: Can immunomodulators play a role? Animal 2009, 3, 1644–1661. [Google Scholar] [CrossRef]
- Seal, B.S.; Drider, D.; Oakley, B.B.; Brüssow, H.; Bikard, D.; Rich, J.O.; Miller, S.; Devillard, E.; Kwan, J.; Bertin, G.; et al. Microbial-derived products as potential new antimicrobials. Vet. Res. 2018, 49, 66. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, C.; Sarkar, P.; Issa, R.; Haldar, J. Alternatives to conventional antibiotics in the era of antimicrobial resistance. Trends Microbiol. 2019, 27, 4. [Google Scholar] [CrossRef] [PubMed]
- Wittebole, X.; De Roock, S.; Opal, S.M. A historical overview of bacteriophage therapy as an alternative to antibiotics for the treatment of bacterial pathogens. Virulence 2014, 5, 226–235. [Google Scholar] [CrossRef] [PubMed]
- Pelfrene, E.; Willebrand, E.; Cavaleiro Sanches, A.; Sebris, Z.; Cavaleri1, M. Bacteriophage therapy: A regulatory perspective. J. Antimicrob. Chemother. 2016, 71, 2071–2074. [Google Scholar] [CrossRef]
- Dąbrowska, K. Phage therapy: What factors shape phage pharmacokinetics and bioavailability? Systematic and critical review. Med. Res. Rev. 2019, 39, 2000–2025. [Google Scholar] [CrossRef]
- Mutti, M.; Corsini, L. Robust approaches for the production of active ingredient and drug product for human phage therapy. Front. Microbiol. 2019, 10, 2289. [Google Scholar] [CrossRef]
- Principi, N.; Silvestri, E.; Esposito, S. Advantages and limitations of bacteriophages for the treatment of bacterial infections. Front. Pharmacol. 2019, 10, 513. [Google Scholar] [CrossRef]
- Górski, A.; Jończyk-Matysiak, E.; Międzybrodzki, R.; Weber-Dąbrowska, B.; Łusiak-Szelachowska, M.; Bagińska, N.; Borysowski, J.; Łobocka, M.B.; Węgrzyn, A.; Węgrzyn, G. Phage therapy: Beyond antibacterial action. Front. Med. 2018, 5, 146. [Google Scholar] [CrossRef]
- Van Belleghem, J.D.; Dąbrowska, K.; Vaneechoutte, M.; Barr, J.J.; Bollyky, P.L. Interactions between bacteriophage, bacteria, and the mammalian immune system. Viruses 2019, 11, 10. [Google Scholar] [CrossRef]
- Gazev, S. Applications of Phage Therapy in Veterinary Medicine. Ph.D. Thesis, Faculty of Veterinary Medicine and Animal Science, Swedish University of Agricultural Sciences, Uppsala, Sweden, 2018. [Google Scholar]
- Zhao, Y.; Ye, M.; Zhang, X.; Sun, M.; Zhang, Z.; Chao, H.; Huang, D.; Wan, J.; Zhang, S.; Jiang, X.; et al. Comparing polyvalent bacteriophage and bacteriophage cocktails for controlling antibiotic-resistant bacteria in soil-plant system. Sci. Total. Environ. 2019, 657, 918–925. [Google Scholar] [CrossRef]
- Yu, P.; Mathieu, J.; Li, M.; Dai, Z.; Alvarez, P.J.J. Isolation of polyvalent bacteriophages by sequential multiple-host approaches. Appl. Environ. Microbiol. 2016, 82, 808–815. [Google Scholar] [CrossRef] [PubMed]
- Bogovazova, G.G.; Voroshilova, N.N.; Bondarenko, V.M. The efficacy of Klebsiella pneumoniae bacteriophage in the therapy of experimental Klebsiella infection. Zhurnal Mikrobiol. Epidemiol. Immunobiol. 1991, 4, 5–8. [Google Scholar]
- Qadir, M.I.; Mobeen, T.; Masood, A. Phage therapy: Progress in pharmacokinetics. Braz. J. Pharm. Sci. 2018, 54, e17093. [Google Scholar] [CrossRef]
- Colom, J.; Cano-Sarabia, M.; Otero, J.; Cortes, P.; Maspoch, D.; Llagostera, M. Liposome-encapsulated bacteriophages for enhanced oral phage therapy against Salmonella spp. Appl. Environ. Microbiol. 2015, 81, 4841–4849. [Google Scholar] [CrossRef]
- Ma, Y.-H.; Islam, G.S.; Wu, Y.; Sabour, P.M.; Chambers, J.R.; Wang, Q.; Wu, S.X.Y.; Griffiths, M.W. Temporal distribution of encapsulated bacteriophages during passage through the chick gastrointestinal tract. Poult. Sci. 2016, 95, 2911–2920. [Google Scholar] [CrossRef]
- Colom, J.; Cano-Sarabia, M.; Otero, J.; Aríñez-Soriano, J.; Cortés, P.; Maspoch, D.; Llagostera, M. Microencapsulation with alginate/CaCO3: A strategy for improved phage therapy. Sci. Rep. 2017, 7, 41441. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Pacan, J.C.; Wang, Q.; Xu, Y.; Huang, X.; Korenevsky, A.; Sabour, P.M. Microencapsulation of bacteriophage felix O1 into chitosan-alginate microspheres for oral delivery. Appl. Environ. Microbiol. 2008, 74, 4799–4805. [Google Scholar] [CrossRef] [PubMed]
- Singla, S.; Harjai, K.; Katare, O.P.; Chhibber, S. Bacteriophage-loaded nanostructured lipid carrier: Improved pharmacokinetics mediates effective resolution of Klebsiella pneumoniae-induced lobar pneumonia. J. Infect. Dis. 2015, 212, 325–334. [Google Scholar] [CrossRef] [PubMed]
- Huff, W.E.; Huff, G.R.; Rath, N.C.; Balog, J.M.; Donoghue, A.M. Bacteriophage treatment of a severe Escherichia coli respiratory infection in broiler chickens. Avian Dis. 2003, 47, 1399–1405. [Google Scholar] [CrossRef]
- Majewska, J.; Beta, W.; Lecion, D.; Hodyra-Stefaniak, K.; Kłopot, A.; Kaźmierczak, Z.; Miernikiewicz, P.; Piotrowicz, A.; Ciekot, J.; Owczarek, B.; et al. Oral application of T4 phage induces weak antibody production in the gut and in the blood. Viruses 2015, 7, 4783–4799. [Google Scholar] [CrossRef]
- Smith, H.W.; Huggins, M.B. Successful treatment of experimental Escherichia coli infections in mice using phage: Its general superiority over antibiotics. J. Gen. Microbiol. 1982, 128, 307–318. [Google Scholar] [CrossRef]
- Nilsson, A.S. Pharmacological limitations of phage therapy. Upsala J. Med. Sci. 2019, 124, 218–227. [Google Scholar] [CrossRef]
- Gutiérrez, D.; Fernández, L.; Rodríguez, A.; García, P. Role of bacteriophages in the implementation of a sustainable dairy chain. Front. Microbiol. 2019, 10, 12. [Google Scholar] [CrossRef]
- Dufour, N.; Delattre, R.; Ricard, J.-D.; Debarbieux, L. The Lysis of pathogenic Escherichia coli by bacteriophages releases less endotoxin than by β-lactams. Clin. Infect. Dis. 2017, 64, 1582–1588. [Google Scholar] [CrossRef]
- Kahn, L.H.; Bergeron, G.; Bourassa, M.W.; De Vegt, B.; Gill, J.; Gomes, F.; Malouin, F.; Opengart, K.G.; Ritter, G.D.; Singer, R.S.; et al. From farm management to bacteriophage therapy: Strategies to reduce antibiotic use in animal agriculture. Ann. N. Y. Acad. Sci. 2019, 31–39. [Google Scholar] [CrossRef]
- García, R.; Latz, S.; Romero, J.; Higuera, G.; García, K.; Bastías, R. Bacteriophage production models: An overview. Front. Microbiol. 2019, 10, 1187. [Google Scholar] [CrossRef]
- Nobrega, F.L.; Costa, A.R.; Santos, J.F.; Siliakus, M.F.; van Lent, J.W.M.; Kengen, S.W.M.; Azeredo, J.; Kluskens, L.D. Genetically manipulated phages with improved pH resistance for oral administration in veterinary medicine. Sci. Rep. 2016, 6, 39235. [Google Scholar] [CrossRef]
- Richards, P.J.; Connerton, P.L.; Connerton, I.F. Phage biocontrol of Campylobacter jejuni in chickens does not produce collateral effects on the gut microbiota. Front. Microbiol. 2019, 10, 476. [Google Scholar] [CrossRef] [PubMed]
- Nabil, N.M.; Tawakol, M.M.; Hassan, H.M. Assessing the impact of bacteriophages in the treatment of Salmonella in broiler chickens. Infect. Ecol. Epidemiol. 2018, 8, 1539056. [Google Scholar] [CrossRef]
- Rozema, E.A.; Stephens, T.P.; Bach, S.J.; Okine, E.K.; Johnson, R.P.; Stanford, K.; McAllister, T.A. Oral and Rectal Administration of Bacteriophages for Control of Escherichia coli O157:H7 in Feedlot Cattle. J. Food Prot. 2009, 72, 241–250. [Google Scholar] [CrossRef]
- Porter, J.; Anderson, J.; Carter, L.; Donjacour, E.; Paros, M. In vitro evaluation of a novel bacteriophage cocktail as a preventative for bovine coliform mastitis. J. Dairy Sci. 2016, 99, 2053–2062. [Google Scholar] [CrossRef] [PubMed]
- Sklar, I.B.; Joerger, R.D. Attempts to utilize bacteriophage to combat salmonella enterica serovar enteritidis infection in chickens. J. Food Saf. 2001, 21, 15–29. [Google Scholar] [CrossRef]
- Atterbury, R.J.; Van Bergen, M.A.P.; Ortiz, F.; Lovell, M.A.; Harris, J.A.; De Boer, A.; Wagenaar, J.A.; Allen, V.M.; Barrow, P.A. Bacteriophage Therapy To Reduce Salmonella Colonization of Broiler Chickens. Appl. Environ. Microbiol. 2007, 73, 4543–4549. [Google Scholar] [CrossRef]
- Wall, S.K.; Zhang, J.; Rostagno, M.H.; Ebner, P.D. Phage therapy to reduce preprocessing Salmonella infections in market-weight swine. Appl. Environ. Microbiol. 2010, 76, 48–53. [Google Scholar] [CrossRef]
- Wray, C.; Morris, J.A. Aspects of colibacillosis in farm animals. J. Ilyg. Camb. 1985, 95, 577–593. [Google Scholar] [CrossRef]
- Huff, W.; Huff, G.; Rath, N.; Balog, J.; Donoghue, A. Prevention of Escherichia coli infection in broiler chickens with a bacteriophage aerosol spray. Poult. Sci. 2002, 81, 1486–1491. [Google Scholar] [CrossRef]
- Jamalludeen, N.; Johnson, R.P.; Shewen, P.E.; Gyles, C.L. Evaluation of bacteriophages for prevention and treatment of diarrhea due to experimental enterotoxigenic Escherichia coli O149 infection of pigs. Vet. Microbiol. 2009, 136, 135–141. [Google Scholar] [CrossRef]
- Janež, N.; Loc-Carrillo, C. Use of phages to control Campylobacter spp. J. Microbiol. Meth. 2013, 95, 68–75. [Google Scholar] [CrossRef]
- Carrillo, C.L.; Atterbury, R.J.; El-Shibiny, A.; Connerton, P.L.; Dillon, E.; Scott, A.; Connerton, I.F. Bacteriophage therapy to reduce campylobacter jejuni colonization of broiler chickens. Appl. Environ. Microbiol. 2005, 71, 6554–6563. [Google Scholar] [CrossRef]
- Fischer, S.; Kittler, S.; Klein, G.; Glünder, G. Impact of a single phage and a phage cocktail application in broilers on reduction of Campylobacter jejuni and development of resistance. PLoS ONE 2013, 8, e78543. [Google Scholar] [CrossRef]
- Mahmood, K.; Rahman, S.U.; Hussain, I.; Abbas, R.Z.; Khaliq, T.; Arif, J.; Mahmood, F. Non-antibiotic strategies for the control of necrotic enteritis in poultry. Worlds Poult. Sci. J. 2014, 70, 865–879. [Google Scholar] [CrossRef]
- Żbikowska, K.; Michalczuk, M.; Dolka, B. The Use of bacteriophages in the poultry industry. Animals 2020, 10, 872. [Google Scholar] [CrossRef]
- Titze, I.; Krömker, V. Antimicrobial activity of a phage mixture and a lactic acid bacterium against Staphylococcus aureus from bovine mastitis. Vet. Sci. 2020, 7, 31. [Google Scholar] [CrossRef]
- Ngassam-Tchambaa, C.; Dupreza, J.N.; Fergestadb, M.; De Visscherc, A.; L’Abee-Lundb, T.; De Vliegherc, S.; Wastesonb, Y.; Touzaind, F.; Blanchardd, Y.; Lavignee, R.; et al. In vitro and in vivo assessment of phage therapy against Staphylococcus aureus causing bovine mastitis. J. Glob. Antimicrob. Resist. 2020, 22, 762–770. [Google Scholar] [CrossRef]
- Ribeiro, H.G.; Correia, R.; Moreira, T.; Vilas Boas, D.; Azeredo, J.; Oliveira, A. Bacteriophage biodistribution and infectivity from honeybee to bee larvae using a T7 phage model. Sci. Rep. 2019, 9, 620. [Google Scholar] [CrossRef]
- REGULATION (EC) No 470/2009 of the European Parliament and of the Council of 6 May 2009. Available online: https://eur-lex.europa.eu/legal-content/pl/TXT/?uri=CELEX:32009R0470 (accessed on 20 July 2020).
- Moye, Z.D.; Woolston, J.; Sulakvelidze, A. Bacteriophage applications for food production and processing. Viruses 2018, 10, 205. [Google Scholar] [CrossRef]
- Bigwood, T.; Hudson, J.A.; Billington, C.; Carey-Smith, G.V.; Heinemann, J.A. Phage inactivation of foodborne pathogens on cooked and raw meat. Food Microbiol. 2008, 25, 400–406. [Google Scholar] [CrossRef]
- Magnone, J.P.; Marek, P.J.; Sulakvelidze, A.; Senecal, A.G. Additive approach for inactivation of Escherichia coli O157:H7, Salmonella, and Shigella spp. on contaminated fresh fruits and vegetables using bacteriophage cocktail and produce wash. J. Food Prot. 2013, 76, 1336–1341. [Google Scholar] [CrossRef]
- Soni, K.A.; Nannapaneni, R. Bacteriophage significantly reduces Listeria monocytogenes on raw salmon fillet tissue. J. Food Prot. 2010, 73, 32–38. [Google Scholar] [CrossRef]
- Grant, A.; Parveen, S.; Schwarz, J.; Hashem, F.; Vimini, B. Reduction of Salmonella in ground chicken using a bacteriophage. Poult. Sci. 2017, 96, 2845–2852. [Google Scholar] [CrossRef]
- Brives, C.; Pourraz, J. Phage therapy as a potential solution in the fight against AMR: Obstacles and possible futures. Palgrave Commun. 2020, 6, 100. [Google Scholar] [CrossRef]
- Jozala, A.F.; de Lencastre Novaes, L.C.; Pessoa, A. Nisin. In Concepts, Compounds and the Alternatives of Antibacterials; InTech Concepts: London, UK, 2015; pp. 103–119. [Google Scholar] [CrossRef]
- Hassan, M.; Kjos, M.; Nes, I.F.; Diep, D.B.; Lotfipour, F. Natural antimicrobial peptides from bacteria: Characteristics and potential applications to fight against antibiotic resistance. J. Appl. Microbiol. 2012, 113, 723–736. [Google Scholar] [CrossRef]
- Józefiak, A.; Engberg, R.M. Insect proteins as a potential source of antimicrobial peptides in livestock production. A review. J. Anim. Feed Sci. 2017, 26, 87–99. [Google Scholar] [CrossRef]
- Lazzaro, B.; Zasloff, M.; Rolff, J. Antimicrobial peptides: Application informed by evolution. Science 2020, 368, 5480. [Google Scholar] [CrossRef]
- Rabel, D.; Charlet, M.; Ehret-Sabatier, L.; Cavicchioli, L.; Cudic, M.; Otvos, L.; Bulet, P. Primary structure and in vitro antibacterial properties of the Drosophila melanogaster attacin C Prodomain. J. Biol. Chem. 2004, 279, 14853–14859. [Google Scholar] [CrossRef]
- Brogden, K.A.; Ackermann, M.; McCray, P.B.; Tack, B.F. Antimicrobial peptides in animals and their role in host defences. Int. J. Antimicrob. Agents 2003, 22, 465–478. [Google Scholar] [CrossRef]
- Laukova, A.; Guba, P.; Nemcova, R.; Marekova, M. Inhibition of Salmonella enterica serovar Dusseldorf by enterocin A in gnotobiotic Japanese quails. Vet. Med. 2004, 49, 47–51. [Google Scholar] [CrossRef]
- Waghu, F.H.; Barai, R.S.; Gurung, P.; Idicula-Thomas, S. CAMPR3: A database on sequences, structures and signatures of antimicrobial peptides. Nucleic Acids Res. 2016, 44, D1094–D1097. [Google Scholar] [CrossRef]
- Yu, L.; van der Linden, D.S.; Sugiarto, H.; Anderson, R.C. Antimicrobial peptides isolated from the blood of farm animals. Anim. Prod. Sci. 2010, 50, 660–669. [Google Scholar] [CrossRef]
- Sun, Y.; Veseli, I.A.; Vaillancourt, K.; Frenette, M.; Grenier, D.; Pombert, J.-F. The bacteriocin from the prophylactic candidate Streptococcus suis 90-1330 is widely distributed across S. suis isolates and appears encoded in an integrative and conjugative element. PLoS ONE 2019, 14, e0216002. [Google Scholar] [CrossRef]
- Wang, S.; Zeng, X.; Yang, Q.; Qiao, S. Antimicrobial peptides as potential alternatives to antibiotics in food animal industry. Int. J. Mol. Sci. 2016, 17, 603. [Google Scholar] [CrossRef] [PubMed]
- Brogden, K.A. Ovine pulmonary surfactant induces killing of Pasteurella haemolytica, Escherichia coli, and Klebsiella pneumoniae by normal serum. Infect. Immun. 1992, 60, 5182–5189. [Google Scholar] [CrossRef] [PubMed]
- Gazit, E.; Boman, A.; Boman, H.G.; Shai, Y. Interaction of the mammalian antibacterial peptide cecropin P1 with phospholipid vesicles. Biochemistry 1995, 34, 11479–11488. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-Y.; Boman, A.; Chuanxin, S.; Andersson, M.; Jörnvall, H.; Mutt, V.; Boman, H.G. Antibacterial peptides from pig intestine: Isolation of a mammalian cecropin. Proc. Natl. Acad. Sci. USA 1989, 86, 9159–9162. [Google Scholar] [CrossRef]
- Coorens, M.; Scheenstra, M.R.; Veldhuizen, E.J.A.; Haagsman, H.P. Interspecies cathelicidin comparison reveals divergence in antimicrobial activity, TLR modulation, chemokine induction and regulation of phagocytosis. Sci. Rep. 2017, 7, 40874. [Google Scholar] [CrossRef]
- Steinstraesser, L.; Tack, B.F.; Waring, A.J.; Hong, T.; Boo, L.M.; Fan, M.-H.; Remick, D.; Su, G.L.; Lehrer, R.I.; Wang, S.C. Activity of Novispirin G10 against Pseudomonas aeruginosa in vitro and in infected burns. Antimicrob. Agents Ch. 2002, 46, 1837–1844. [Google Scholar] [CrossRef]
- Yari, L.; Karkhane, A.A.; Bahrami, A.; Yakhchali, B. Bactenecin and its three improved derivatives for enhancement of antibacterial activity against Escherichia coli. Jundishapur J. Microbiol. 2019, 12, e94769. [Google Scholar] [CrossRef]
- Diarraa, M.S.; Petitclerca, D.; Deschenesa, E.; Lessardc, N.; Grondinb, G.; Talbot, B.G.; Lacassea, P. Lactoferrin against Staphylococcus aureus mastitis lactoferrin alone or in combination with Penicillin G on bovine polymorphonuclear function and mammary epithelial cells colonisation by Staphylococcus aureus. Vet. Immunol. Immunop. 2003, 95, 33–42. [Google Scholar] [CrossRef]
- Kawai, K.; Nagahata, H.; Lee, N.; Anri, A.; Shimazaki, K. Effect of infusing lactoferrin hydrolysate into bovine mammary glands with subclinical mastitis. Vet. Res. Commun. 2003, 27, 539–548. [Google Scholar] [CrossRef]
- Kawai, K.; Shimazaki, K.; Higuchi, H.; Nagahata, H. Antibacterial activity of bovine lactoferrin hydrolysate against mastitis pathogens and its effect on superoxide production of bovine neutrophils. Zoonoses Public Health 2007, 54, 160–164. [Google Scholar] [CrossRef]
- Zhang, J.X.; Zhang, S.F.; Wang, T.D.; Guo, X.J.; Hu, R.L. Mammary gland expression of antibacterial peptide genes to inhibit bacterial pathogens causing mastitis. J. Dairy Sci. 2007, 90, 5218–5225. [Google Scholar] [CrossRef]
- Wang, Y.-Z.; Shan, T.-Z.; Xu, Z.-R.; Feng, J.; Wang, Z.-Q. Effects of the lactoferrin (LF) on the growth performance, intestinal microflora and morphology of weanling pigs. Anim. Feed Sci. Technol. 2007, 135, 263–272. [Google Scholar] [CrossRef]
- Xiong, X.; Yang, H.S.; Li, L.; Wang, Y.F.; Huang, R.L.; Li, F.N.; Wang, S.P.; Qiu, W. Effects of antimicrobial peptides in nursery diets on growth performance of pigs reared on five different farms. Livest. Sci. 2014, 167, 206–210. [Google Scholar] [CrossRef]
- Lagha, A.B.; Haas, B.; Gottschalk, M.; Grenier, D. Antimicrobial potential of bacteriocins in poultry and swine production. Vet. Res. 2017, 48, 22. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.-C.; Lin, C.-H.; Sung, C.T.; Fang, J.-Y. Antibacterial activities of bacteriocins: Application in foods and pharmaceuticals. Front. Microbiol. 2014, 5, 241. [Google Scholar] [CrossRef]
- Chikindas, M.L.; Weeks, R.; Drider, D.; Chistyakov, V.A.; Dicks, L.M.T. Functions and emerging applications of bacteriocins. Curr. Opin. Biotechnol. 2018, 49, 23–28. [Google Scholar] [CrossRef]
- Józefiak, D.; Sip, A. Bacteriocins in poultry nutrition—A review. Ann. Anim. Sci. 2013, 13, 449–462. [Google Scholar] [CrossRef][Green Version]
- Gharsallaoui, A.; Oulahal, N.; Joly, C. Nisin as a food preservative: Part 1: Physicochemical properties, antimicrobial activity and main uses. Crit. Rev. Food Sci. Nutr. 2016, 56, 1262–1274. [Google Scholar] [CrossRef]
- Galvez, A.; Abriouel, H.; Lopez, R.L.; Ben Omar, N. Bacteriocin-based strategies for food biopreservation. Int. J. Food Microbiol. 2007, 120, 51–70. [Google Scholar] [CrossRef]
- Cao, L.T.; Wu, J.Q.; Xie, F.; Hu, S.H.; Mo, Y. Efficacy of nisin in treatment of clinical mastitis in lactating dairy cows. J. Dairy Sci. 2007, 90, 3980–3985. [Google Scholar] [CrossRef]
- Kaczorek, E.; Małaczewska, J.; Wójcik, R.; Rękawek, W.; Siwicki, A.K. Phenotypic and genotypic antimicrobial susceptibility pattern of Streptococcus spp. isolated from cases of clinical mastitis in dairy cattle in Poland. J. Dairy Sci. 2017, 100, 6442–6453. [Google Scholar] [CrossRef]
- Shin, J.M.; Gwak, J.W.; Kamarajan, P.; Fenno, J.C.; Rickard, A.H.; Kapila, Y.L. Biomedical applications of nisin. J. Appl. Microbiol. 2016, 120, 1449–1465. [Google Scholar] [CrossRef] [PubMed]
- Maurício, E.; Rosado, C.; Duarte, M.P.; Verissimo, J.; Bom, S.; Vasconcelos, L. Efficiency of nisin as preservative in cosmetics and topical products. Cosmetics 2017, 4, 41. [Google Scholar] [CrossRef]
- Timbermont, L.; De Smet, L.; Van Nieuwerburgh, F.; Parreira, V.R.; Van Driessche, G.; Haesebrouck, F.; Ducatelle, R.; Prescott, J.; Deforce, D.; Devreese, B.; et al. Perfrin, a novel bacteriocin associated with netB positive Clostridium perfringens strains from broilers with necrotic enteritis. Veter. Res. 2014, 45, 40. [Google Scholar] [CrossRef]
- Wang, G.; Song, Q.; Huang, S.; Wang, Y.; Cai, S.; Yu, H.; Ding, X.; Zeng, X.; Zhang, J. Effect of antimicrobial peptide Microcin J25 on growth performance, immune regulation, and intestinal microbiota in broiler chickens challenged with Escherichia coli and Salmonella. Animals 2020, 10, 345. [Google Scholar] [CrossRef]
- Svetoch, E.A.; Stern, N.J.; Eruslanov, B.V.; Kovalev, Y.N.; Volodina, L.I.; Perelygin, V.V.; Mitsevich, E.V.; Mitsevich, I.P.; Pokhilenko, V.D.; Borzenkov, V.N.; et al. Isolation of Bacillus circulans and Paenibacillus polymyxa strains inhibitory to Campylobacter jejuni and characterization of associated bacteriocins. J. Food Prot. 2005, 68, 11–17. [Google Scholar] [CrossRef]
- Ogunbanwo, S.; Sanni, A.; Onilude, A. Influence of bacteriocin in the control of Escherichia coli infection of broiler chickens in Nigeria. World J. Microbiol. Biotechnol. 2004, 20, 51–56. [Google Scholar] [CrossRef]
- Vieco-Saiz, N.; Belguesmia, Y.; Raspoet, R.; Auclair, E.; Gancel, F.; Kempf, I.; Drider, D. Benefits and inputs from lactic acid bacteria and their bacteriocins as alternatives to antibiotic growth promoters during food-animal production. Front. Microbiol. 2019, 10, 57. [Google Scholar] [CrossRef]
- Vaillancourt, K.; LeBel, G.; Frenette, M.; Gottschalk, M.; Grenier, D. Suicin 3908, a new lantibiotic produced by a strain of Streptococcus suis serotype 2 isolated from a healthy carrier pig. PLoS ONE 2015, 10, e0117245. [Google Scholar] [CrossRef]
- Mazurek-Popczyk, J.; Pisarska, J.; Bok, E.; Baldy-Chudzik, K. Antibacterial activity of bacteriocinogenic commensal Escherichia coli against zoonotic strains resistant and sensitive to antibiotics. Antibiotics 2020, 9, 411. [Google Scholar] [CrossRef] [PubMed]
- Lasagno, M.; de los Angeles Navarro, M.; Moliva, M.; Reinoso, E. Screening of bacteriocin associated genes of Streptococcus uberis strains. Heliyon. 2019, 5, e02393. [Google Scholar] [CrossRef] [PubMed]
- Godoy-Santos, F.; Pinto, M.S.; Barbosa, A.A.T.; Brito, M.A.V.P.; Mantovani, H.C. Efficacy of a ruminal bacteriocin against pure and mixed cultures of bovine mastitis pathogens. Ind. J. Microbiol. 2019, 59, 304–312. [Google Scholar] [CrossRef] [PubMed]
- Carson, D.A.; Barkema, H.W.; Naushad, S.; De Buck, J. Bacteriocins of non-aureus staphylococci isolated from bovine milk. Appl. Environ. Microbiol. 2017, 83, e01015-17. [Google Scholar] [CrossRef] [PubMed]
- Heo, S.; Kim, M.G.; Kwon, M.; Lee, H.S.; Kim, G.-B. Inhibition of Clostridium perfringens using bacteriophages and bacteriocin producing strains. Korean J. Food Sci. 2018, 38, 88–98. [Google Scholar] [CrossRef]
- LeBel, G.; Piché, F.; Frenette, M.; Gottschalk, M.; Grenier, D. Antimicrobial activity of nisin against the swine pathogen Streptococcus suis and its synergistic interaction with antibiotics. Peptides 2013, 50, 19–23. [Google Scholar] [CrossRef]
- Al Atya, A.K.; Abriouel, H.; Kempf, I.; Jouy, E.; Auclair, E.; Vachee, A.; Drider, D. Effects of colistin and bacteriocins combinations on the in vitro growth of Escherichia coli strains from swine origin. Prob. Antimicrob. Prot. 2016, 8, 183–190. [Google Scholar] [CrossRef]
- Lin, L.; Nonejuie, P.; Munguia, J.; Hollands, A.; Olson, J.; Dam, Q.; Kumaraswamy, M.; Rivera, H.; Corriden, R.; Rohde, M.; et al. Azithromycin synergizes with cationic antimicrobial peptides to exert bactericidal and therapeutic activity against highly multidrug-resistant gram-negative bacterial pathogens. EBioMed. 2015, 2, 690–698. [Google Scholar] [CrossRef]
- Luther, A.; Urfer, M.; Zahn, M.; Müller, M.; Wang, S.-Y.; Mondal, M.; Vitale, A.; Hartmann, J.-B.; Sharpe, T.; Monte, F.L.; et al. Chimeric peptidomimetic antibiotics against Gram-negative bacteria. Nat. Cell Biol. 2019, 576, 452–458. [Google Scholar] [CrossRef]
- Soren, O.; Brinch, K.S.; Patel, D.; Liu, Y.; Liu, A.; Coates, A.; Hu, Y. Antimicrobial peptide novicidin synergizes with rifampin, ceftriaxone, and ceftazidime against antibiotic-resistant enterobacteriaceae in vitro. Antimicrob. Agents Chemother. 2015, 59, 6233–6240. [Google Scholar] [CrossRef]
- Chae, J.-H.; Kurokawa, K.; So, Y.-I.; Hwang, H.O.; Kim, M.-S.; Park, J.-W.; Jo, Y.-H.; Lee, Y.S.; Lee, B.L. Purification and characterization of tenecin 4, a new anti-Gram-negative bacterial peptide, from the beetle Tenebrio molitor. Dev. Comp. Immunol. 2012, 36, 540–546. [Google Scholar] [CrossRef]
- Fantner, G.E.; Barbero, R.J.; Gray, D.S.; Belcher, A.M. Kinetics of antimicrobial peptide activity measured on individual bacterial cells using high-speed atomic force microscopy. Nat. Nanotechnol. 2010, 5, 280–285. [Google Scholar] [CrossRef]
- Holmes, A.H.; Moore, L.S.P.; Sundsfjord, A.; Steinbakk, M.; Regmi, S.; Karkey, A.; Guerin, P.J.; Piddock, L.J.V. Understanding the mechanisms and drivers of antimicrobial resistance. Lancet 2016, 9, 176–187. [Google Scholar] [CrossRef]
- Gullberg, E.; Cao, S.; Berg, O.G.; Ilbäck, C.; Sandegren, L.; Hughes, D.; Andersson, D.I. Selection of resistant bacteria at very low antibiotic concentrations. PLoS Pathog. 2011, 7, e1002158. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Hu, Y.; Yang, Y.; Lu, Z.; Wang, Y. Antimicrobial resistance in livestock: Antimicrobial peptides provide a new solution for a growing challenge. Anim. Front. 2018, 8, 21–29. [Google Scholar] [CrossRef]
- Topman, S.; Tamir-Ariel, D.; Bochnic-Tamir, H.; Bauer, T.S.; Shafir, S.; Burdman, S.; Hayouka, Z. Random peptide mixtures as new crop protection agents. Microb. Biotechnol. 2018, 11, 1027–1036. [Google Scholar] [CrossRef]
- Unckless, R.L.; Howick, V.M.; Lazzaro, B.P. Convergent balancing selection on an antimicrobial peptide in Drosophila. Curr. Biol. 2016, 26, 257–262. [Google Scholar] [CrossRef]
- Chou, S.; Wang, J.; Shang, L.; Akhtar, M.U.; Wang, Z.; Shi, B.; Feng, X.; Shan, A. Short, symmetric-helical peptides have narrow-spectrum activity with low resistance potential and high selectivity. Biomater. Sci. 2019. [Google Scholar] [CrossRef]
- Kamata, K.; Tokuda, Y.; Gu, Y.; Ohmagari, N.; Yanagihara, K. Public knowledge and perception about antimicrobials and antimicrobial resistance in Japan: A national questionnaire survey in 2017. PLoS ONE 2018, 13, e0207017. [Google Scholar] [CrossRef]
- Tsuzuki, S.; Fujitsuka, N.; Horiuchi, K.; Ijichi, S.; Gu, Y.; Fujitomo, Y.; Takahashi, R.; Ohmagari, N. Factors associated with sufficient knowledge of antibiotics and antimicrobial resistance in the Japanese general population. Sci. Rep. 2020, 10, 3502. [Google Scholar] [CrossRef]
- European Commission. Products Covered by EU Organics Rules. Available online: https://ec.europa.eu/info/food-farming-fisheries/farming/organic-farming/organic-production-and-products_en (accessed on 15 August 2020).
- Sundrum, A. Organic livestock farming. Livest. Prod. Sci. 2001, 67, 207–215. [Google Scholar] [CrossRef]
| Report/Project | Published by |
|---|---|
| A report on Swedish Antibiotic Sales and Resistance in Human Medicine (Swedres) and Swedish Veterinary Antibiotic Resistance Monitoring (Svarm) | Public Health Agency of Sweden and National Veterinary Institute Annual Report https://www.sva.se/en/our-topics/antibiotics/svarm-resistance-monitoring/swedres-svarm-reports/ |
| Danish Integrated Antimicrobial Resistance Monitoring and Research Programme (DANMAP): Use of antimicrobial agents and occurrence of antimicrobial resistance in bacteria from food animals, food, and humans in Denmark | National Food Institute Statens Serum Institut (Denmark) Danish Ministry of Food, Agriculture and Fisheries and the Danish Ministry of Health in 1995. Annual Report https://www.danmap.org/ |
| Swiss Antibiotic Resistance Report | Federal Office of Public Health FOPH (Switzerland) Annual Report https://www.bundespublikationen.admin.ch/cshop_mimes_bbl/ |
| Annual Report | Scientific Board of ONERBA (the National Observatory of the Epidemiology of Bacterial Resistance to Antibiotics) France Annual Report http://onerba.org/onerba-in-english/ |
| FINRES–Vet Finnish Veterinary Antimicrobial Resistance Monitoring and Consumption of Antimicrobial Agents | Finnish Food Authority (Finland) Annual Report https://www.ruokavirasto.fi/en/farmers/animal-husbandry/animal-medication/monitoring-of-antibiotic-resistance/finres-vet-reports/ |
| UK Veterinary Antibiotic Resistance and Sales Surveillance Report | Veterinary Medicines Directorate (VMD; United Kingdom) Annual Report https://www.gov.uk/government/publications/veterinary-antimicrobial-resistance-and-sales-surveillance-2018 |
| The European Union summary report on antimicrobial resistance in zoonotic and indicator bacteria from humans, animals, and food | European Food Safety Authority and European Centre for Disease Prevention and Control Based on Data Submitted by EU Member Countries Annual Report https://www.efsa.europa.eu/en/efsajournal/pub/6007 |
| Sales of veterinary antimicrobial agents in 31 European countries in (given year) under the European Surveillance of Veterinary Antimicrobial Consumption project | the European Medicines Agency (EMA) Annual Report https://www.ema.europa.eu/en/veterinary-regulatory/overview/antimicrobial-resistance/european-surveillance-veterinary-antimicrobial-consumption-esvac |
| National Antimicrobial Resistance Monitoring System (NARMS) Annual Integrated Report | Food and Drug Administration (FDA) Annual Report https://www.fsis.usda.gov/wps/portal/fsis/topics/data-collection-and-reports/microbiology/antimicrobial-resistance/narms |
| OIE Annual Report on Antimicrobial Agents Intended for Use in Animals | World Health Organization for Animal Health Annual Report https://www.oie.int/scientific-expertise/veterinary-products/antimicrobials/ |
| Global Action Plan On Antimicrobial Resistance | World Health Organization In 2015 http://www.emro.who.int/health-topics/drug-resistance/global-action-plan.html |
| The OIE Strategy on Antimicrobial Resistance and the Prudent Use of Antimicrobials | World Health Organization for Animal Health In 2016 https://www.oie.int/fileadmin/Home/eng/Media_Center/docs/pdf/PortailAMR/EN_OIE-AMRstrategy.pdf |
| EMA and EFSA Joint Scientific Opinion on measures to reduce the need to use antimicrobial agents in animal husbandry in the European Union, and the resulting impacts on food safety (RONAFA opinion) | EMA (European Medicines Agency) and EFSA (European Food Safety Authority) in 2017 https://www.efsa.europa.eu/en/efsajournal/pub/4666 |
| Supporting Antimicrobial Stewardship In Veterinary Settings Goals For Fiscal Years 2019–2023 | FDA (U.S. Food and Drug Administration) CENTER FOR VETERINARY MEDICINE In 2018 https://health.gov/healthypeople/tools-action/browse-evidence-based-resources/supporting-antimicrobial-stewardship-veterinary-settings-goals-fiscal-years-2019-2023-fda-center-veterinary-medicine |
| The 2019 WHO AWaRe classification of antibiotics for evaluation and monitoring of use | World Health Organization In 2019 https://www.who.int/medicines/news/2019/WHO_releases2019AWaRe_classification_antibiotics/en/ |
| No Time To Wait: Securing The Future From Drug-Resistant Infections | World Health Organization Interagency Coordination Group (IACG) on Antimicrobial Resistance In 2019 https://www.who.int/antimicrobial-resistance/interagency-coordination-group/final-report/en/ |
| Antibiotic Resistance Threats In The United States | Centers for Disease Control and Prevention In 2019 https://www.cdc.gov/drugresistance/pdf/threats-report/2019-ar-threats-report-508.pdf |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nowakiewicz, A.; Zięba, P.; Gnat, S.; Matuszewski, Ł. Last Call for Replacement of Antimicrobials in Animal Production: Modern Challenges, Opportunities, and Potential Solutions. Antibiotics 2020, 9, 883. https://doi.org/10.3390/antibiotics9120883
Nowakiewicz A, Zięba P, Gnat S, Matuszewski Ł. Last Call for Replacement of Antimicrobials in Animal Production: Modern Challenges, Opportunities, and Potential Solutions. Antibiotics. 2020; 9(12):883. https://doi.org/10.3390/antibiotics9120883
Chicago/Turabian StyleNowakiewicz, Aneta, Przemysław Zięba, Sebastian Gnat, and Łukasz Matuszewski. 2020. "Last Call for Replacement of Antimicrobials in Animal Production: Modern Challenges, Opportunities, and Potential Solutions" Antibiotics 9, no. 12: 883. https://doi.org/10.3390/antibiotics9120883
APA StyleNowakiewicz, A., Zięba, P., Gnat, S., & Matuszewski, Ł. (2020). Last Call for Replacement of Antimicrobials in Animal Production: Modern Challenges, Opportunities, and Potential Solutions. Antibiotics, 9(12), 883. https://doi.org/10.3390/antibiotics9120883







