Social and Contextual Influences on Antibiotic Prescribing and Antimicrobial Stewardship: A Qualitative Study with Clinical Commissioning Group and General Practice Professionals
Abstract
1. Introduction
2. Methods
2.1. Study Participants
2.2. Data Collection
2.3. Data Analysis
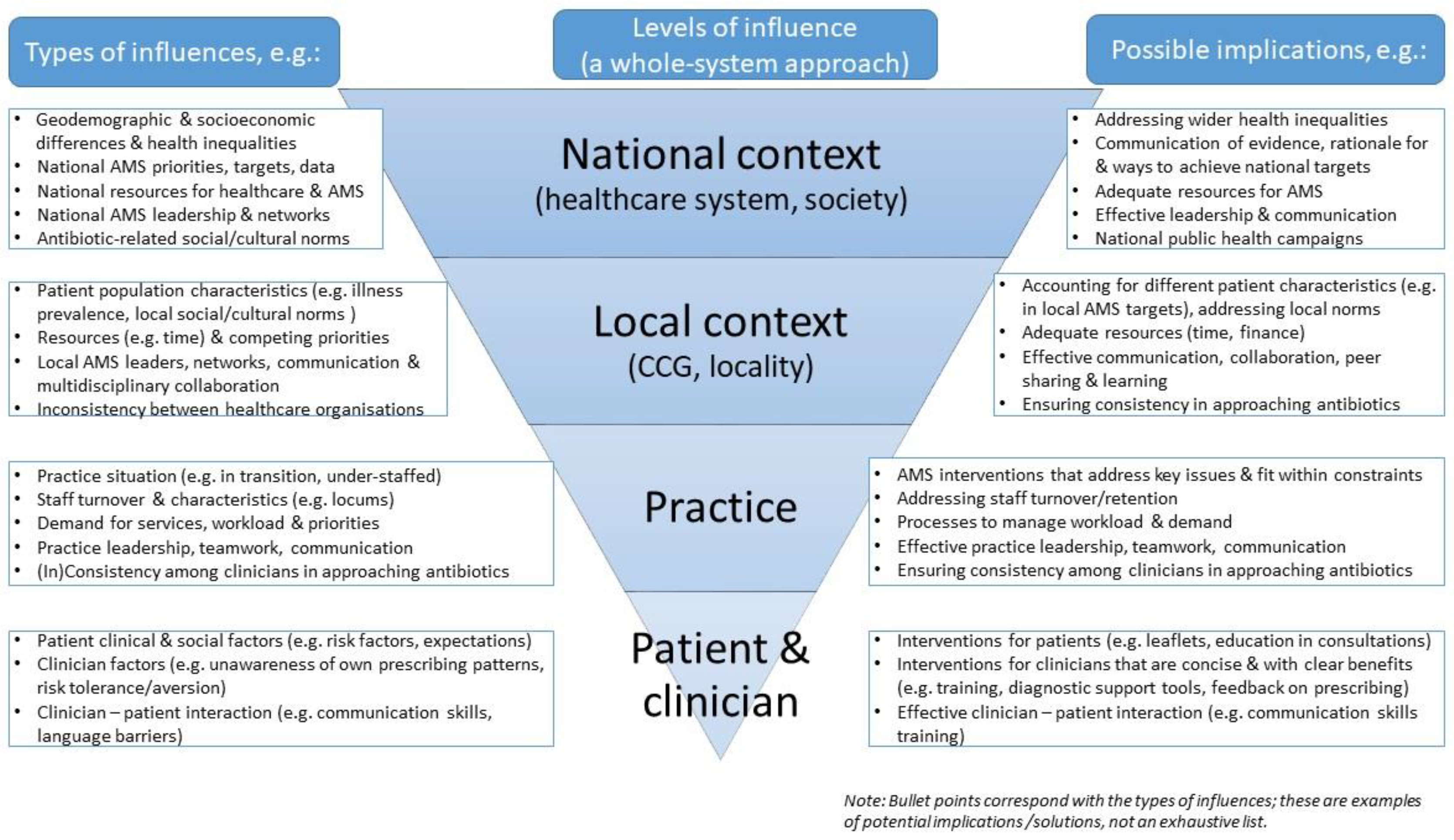
3. Results
3.1. Immediate Context: Patient, Clinician, and Practice Characteristics
The practice is in a deprived area and the patient expectation here is that they will get a prescription for an antibiotic. Not only that, but people live in overcrowded situations and there is cross-infection with wider infections etc. (…) I think there may be a clinical reason but not sufficient to explain the complete high prevalence but I feel we often get into a situation where the patient’s expectation is for a prescription for antibiotic and when we refuse or try and explain that it’s not appropriate, we get a very irate patient, and I can understand that because there is a pressure on the patient to get well and get back to work as their income levels are lower in the patch we work in. Also, children with coughs and colds, parents would expect antibiotics because their child staying at home would mean that the parent would have to take time off work and again, loss of income. And so a patient more or less expects a prescription.(GP-7, high-prescribing)
…we have a large Pakistani community and I think culturally there is this expectation that when you go to the doctors, they’ll give you a prescription and sometimes it doesn’t even matter what that prescription is, but you have to have some medication and also there’s a language barrier quite often as well… and some of our patients aren’t particularly well educated as well, so trying to give people some understanding of even basic statistics or basic kind of biology is quite difficult and trying to explain to somebody that you don’t need a prescription.(GP-10, low/medium-prescribing)
We have good response from the practices that have stable staff. Unfortunately, there are quite a lot of practices that have a lot of locums, transient staff, maybe inexperienced staff and what we find is that in those areas and often areas that have high levels of chronic disease we tend to get a high prescribing rate and it’s difficult to address that particularly when you can’t engage the staff as they’re not there long-term. (…) There’s been a lot of retirement from practices in [area] and there’s been takeovers of practices and as a consequence you’ve had lots of locum staff or short-term staff there. I think once there’s less ownership of the problem, you tend to see a deterioration in the antibiotic prescribing rates. I would argue we could map that across the city and say, we know these practices have a problem and will continue because they are struggling as general practices and as a consequence that impacts on their antimicrobial prescribing.(CCG-5, high-prescribing)
This is a bit controversial, but there’s quite a lot of locum GPs and there’s quite a lot of older GPs… some of the older GPs aren’t quite as up to date on the guidelines on what to prescribe so they tend to prescribe drugs that they’ve prescribed a lot in the past and the guys who come as locums don’t have that same kind of… it’s my permanent base so I’m very strict on what I prescribe and I stick to the guidelines, but some of the locums don’t and because they work across different places… and the guidelines may be slightly different.(GP-11, high-prescribing)
People’s thresholds as to what’s reasonable can be subjective so it depends on how open-minded you are when you go into an audit. If you do an audit feeling defensive, then it’s probably a waste of time because you’re going to find an excuse to justify things (…) practices that are practicing defensively or under a lot of pressure seem to be the ones that are much less likely to change.(CCG-9, multiple CCGs)
I still don’t think there is the level of gravity placed on inappropriate prescribing. It’s still perceived widespread as a sort of poor judgement call. If you imagine that we were prescribing drugs for diabetes or hypertension or other long-term conditions inappropriately, it would be seen as a fairly serious deficit in care. Whereas if it’s an antibiotic, it’s seen as poor judgement or just GPs taking the easy life.(CCG-21, high-prescribing)
3.2. Wider Context: Pressures on the System and Resources
The thing that everyone recognizes is that it takes so much longer to explain why you’re not prescribing than it does to just churn out a script for an antibiotic so time is key and over-pressurized practices with insufficient staff for the workload might be predicted to prescribe more or more often inappropriately, possibly. I think we need to keep an eye on the staffing and workload levels because having time with the patient is the most effective way of exploring their actual issues and what really is worrying them….(GP-18, medium/low-prescribing)
So the issue really isn’t just simply about antimicrobials. It’s about antimicrobial prescribing within a constrained under-pressure system that in some areas is really very very delicate. Banging on and saying messages about the antibiotic problems ahead is very hard when you’ve got everyday pressures in general practices you’ve got to contend with and a waiting room full of people that you’ve got to get through. […] I think people are tired with change and tired of the relentless messages, do this, do that. So I think at the moment if it’s going to make people’s lives harder or add to workload, then you’re probably reducing the chances of that change happening.(CCG-9, multiple CCGs)
I don’t think you can have a single approach. I think if you find that somebody isn’t following what you think is sensible advice, you need to go and have a conversation with them to understand what the barriers are to implement the advice that you think is sensible. Usually those barriers will have some substance. You then have to look at what the barriers are, you have to work out whether it’s possible to move those barriers.(CCG-8, low-prescribing)
3.3. Collaborative and Whole System Approaches
Sharing experience, sharing best practices and helping clinicians who are struggling is the only way. We already have a severe workforce crisis, we can’t have a GP leaving because they can’t deal with that.(GP-15, low-prescribing)
I’d love to learn from those that have done really well and how they’ve achieved it because there is such a difference particularly with the London practices, I don’t know how they’re doing it. (…) It would be really interesting just to know how people have done it.(CCG-4, high-prescribing)
I think it’s really important personally to have a multi-pronged approach so that you are all doing the same thing, and you don’t have a GP saying ‘No, you can’t have an antibiotic’ in their surgery, and then they walk to the local casualty at 7 pm and come home with Augmentin... It’s trying to have consistent messages from all areas of the NHS which is the hardest ask.(CCG-8, low-prescribing)
I have to say, the engagement from the Chief Medical Officer (Dame Sally Davies), I think that’s been very profound and supported what you’re trying to do on the ground. When you have such high-level leadership, it makes it a lot easier because there’s so many different things on the agenda of GPs, it’s sometimes hard to fight for space but when you’ve got buy-in at that level and also the Quality Premium, it makes shifting your thing up the agenda easier.(CCG-4, high-prescribing)
We did a workshop where we got a clinician from another area. It was a doctor who had published about the work that he’d done in reducing antibiotic prescribing in his surgery and the approach that they’d taken and we got him to come and talk to all of our practices at a joint locality meeting because we felt that me standing there going, “you really ought to say no to antibiotics a bit more” probably wasn’t very helpful but a clinician who had done it, one of their peers was probably more helpful and that went down really well.(CCG-2, medium-prescribing)
…if a practice can be united in their approach to managing antimicrobial prescribing, then that seems to reflect a kind of lower overall antibiotic prescribing rate and that’s true of a lot of prescribing indicators. If you’ve got people working as a practice and not as a set of individuals, that’s really really helpful.(CCG-9, multiple CCGs)
It’s something we’ve always been passionate about. Historically we’ve had a few GPs in particular, two or three of us who strongly believe in antibiotic stewardship. (…) I think the most important strategy is creating milieu in the practice to reduce unnecessary prescribing. If you ask me what I believe personally, you need to be well led. You need to have an environment in the practice that you’re all buying into that as a principle. And I think if you do that, you’ll be more successful than if you try to do it unilaterally, on your own sort of thing. You need to try and get buy-in.(GP-6, low-prescribing)
3.4. Relativity of Appropriate Prescribing
‘Why have they chosen 10%’? And why this and why that and, ‘They just plucked these figures out of the air’, so it did create some negative feedback.(CCG-6, high-prescribing)
We’ve put in some arbitrary red, amber, green ratings on the dashboard for our antibiotics, and if people are green then they’re doing well, they’re performing to target, they’re doing what we want them to do, they wouldn’t have a great deal of additional scrutiny. If somebody is red and not moving at all, then that would be more cause for concern.(CCG-8, low-prescribing)
For us it would be helpful if everybody else would stand still and give us a chance because we are reducing our antibiotics but equally so is everybody else so whilst we are lower than we were, we are still higher than other areas….(CCG-2, medium-prescribing)
Being the lowest percentage in the UK is still high when you compare it to other countries in the world.(GP-6, low-prescribing)
This particular colleague who prescribes a lot of antibiotics used to prescribe a lot of Co-amoxiclav. Because the CCG are concentrating on that… he’s recently moved over to prescribing Azithromycin… He knows that this isn’t one that the CCG are picking up on and paying attention, so it’s a way of keeping his head down. He wants to prescribe an effective antibiotic which minimizes the risk of the patient getting worse and needing to be admitted to hospital and that’s his current way of doing it.(GP-4, high-prescribing)
Generally, now, as prescribers and the general population seem to understand the responsibility that they have so generally I feel as though I’m pushing against an open door in supporting certainly the clinicians in trying to achieve appropriate prescribing in their practices. I think maybe that wouldn’t have been the case 10 or 15 years ago, but I think the perception has changed both in the public and other healthcare professionals.(CCG-4, high-prescribing)
4. Discussion
4.1. Comparison with the Literature
4.2. Implications
4.3. Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Public Health England. English Surveillance Programme for Antimicrobial Utilisation and Resistance (ESPAUR) Report 2018–2019; Public Health England: London, UK, 2019. Available online: https://webarchive.nationalarchives.gov.uk/20191003132022/https://www.gov.uk/government/publications/english-surveillance-programme-antimicrobial-utilisation-and-resistance-espaur-report (accessed on 30 November 2020).
- Tonkin-Crine, S.K.; Tan, P.S.; van Hecke, O.; Wang, K.; Roberts, N.W.; McCullough, A.; Hansen, M.P.; Butler, C.C.; Del Mar, C.B. Clinician-targeted interventions to influence antibiotic prescribing behaviour for acute respiratory infections in primary care: An overview of systematic reviews. Cochrane Database Syst. Rev. 2017, 9, CD012252. [Google Scholar] [CrossRef]
- McDonagh, M.S.; Peterson, K.; Winthrop, K.; Cantor, A.; Lazur, B.H.; Buckley, D.I. Interventions to reduce inappropriate prescribing of antibiotics for acute respiratory tract infections: Summary and update of a systematic review. J. Int. Med. Res. 2018, 46, 3337–3357. [Google Scholar] [CrossRef] [PubMed]
- NHS England. NHS England: Quality Premium. Available online: https://www.england.nhs.uk/ccg-out-tool/qual-prem/ (accessed on 2 July 2019).
- Balinskaite, V.; Johnson, A.P.; Holmes, A.; Aylin, P. The Impact of a National Antimicrobial Stewardship Program on Antibiotic Prescribing in Primary Care: An Interrupted Time Series Analysis. Clin. Infect. Dis. 2019, 69, 227–232. [Google Scholar] [CrossRef] [PubMed]
- Bou-Antoun, S.; Costelloe, C.; Honeyford, K.; Mazidi, M.; Hayhoe, B.W.J.; Holmes, A.; Johnson, A.P.; Aylin, P. Age-related decline in antibiotic prescribing for uncomplicated respiratory tract infections in primary care in England following the introduction of a national financial incentive (the Quality Premium) for health commissioners to reduce use of antibiotics in the community: An interrupted time series analysis. J. Antimicrob. Chemother. 2018, 73, 2883–2892. [Google Scholar] [CrossRef] [PubMed]
- Curtis, H.J.; Walker, A.J.; Mahtani, K.R.; Goldacre, B. Time trends and geographical variation in prescribing of antibiotics in England 1998–2017. J. Antimicrob. Chemother. 2019, 74, 242–250. [Google Scholar] [CrossRef] [PubMed]
- Palin, V.; Mölter, A.; Belmonte, M.; Ashcroft, D.M.; White, A.; Welfare, W.; Van Staa, T. Antibiotic prescribing for common infections in UK general practice: Variability and drivers. J. Antimicrob. Chemother. 2019, 74, 2440–2450. [Google Scholar] [CrossRef] [PubMed]
- Pouwels, K.B.; Dolk, F.C.K.; Smith, D.R.M.; Smieszek, T.; Robotham, J.V. Explaining variation in antibiotic prescribing between general practices in the UK. J. Antimicrob. Chemother. 2018, 73, ii27–ii35. [Google Scholar] [CrossRef]
- Hope, E.C.; Crump, R.E.; Hollingsworth, T.D.; Smieszek, T.; Robotham, J.V.; Pouwels, K.B. Identifying English Practices that Are High Antibiotic Prescribers Accounting for Comorbidities and Other Legitimate Medical Reasons for Variation. EClinicalMedicine 2018, 6, 36–41. [Google Scholar] [CrossRef]
- Dolk, F.C.K.; Pouwels, K.B.; Smith, D.R.M.; Robotham, J.V.; Smieszek, T. Antibiotics in primary care in England: Which antibiotics are prescribed and for which conditions? J. Antimicrob. Chemother. 2018, 73, ii2–ii10. [Google Scholar] [CrossRef]
- Wang, K.Y.; Seed, P.; Schofield, P.; Ibrahim, S.; Ashworth, M. Which practices are high antibiotic prescribers? A cross-sectional analysis. Br. J. Gen. Pract. 2009, 59, e315–e320. [Google Scholar] [CrossRef]
- Mölter, A.; Belmonte, M.; Palin, V.; Mistry, C.; Sperrin, M.; White, A.; Welfare, W.; Van Staa, T. Antibiotic prescribing patterns in general medical practices in England: Does area matter? Health Place 2018, 53, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Smith, R. Antimicrobial resistance is a social problem requiring a social solution. BMJ 2015, 350, h2682. [Google Scholar] [CrossRef] [PubMed]
- Charani, E.; Holmes, A. Antibiotic Stewardship—Twenty Years in the Making. Antibiotics 2019, 8, 7. [Google Scholar] [CrossRef] [PubMed]
- Germeni, E.; Frost, J.; Garside, R.; Rogers, M.; Valderas, J.M.; Britten, N. Antibiotic prescribing for acute respiratory tract infections in primary care: An updated and expanded meta-ethnography. Br. J. Gen. Pract. 2018, 68, e633–e645. [Google Scholar] [CrossRef] [PubMed]
- Van Der Zande, M.M.; Dembinsky, M.; Aresi, G.; Van Staa, T.P. General practitioners’ accounts of negotiating antibiotic prescribing decisions with patients: A qualitative study on what influences antibiotic prescribing in low, medium and high prescribing practices. BMC Fam. Pract. 2019, 20, 172. [Google Scholar] [CrossRef] [PubMed]
- Borek, A.J.; Anthierens, S.; Allison, R.; McNulty, C.A.M.; Lecky, D.M.; Costelloe, C.; Holmes, A.; Butler, C.C.; Walker, A.S.; Tonkin-Crine, S. How did a Quality Premium financial incentive influence antibiotic prescribing in primary care? Views of Clinical Commissioning Group and general practice professionals. J. Antimicrob. Chemother. 2020, 75, 2681–2688. [Google Scholar] [CrossRef]
- Improving the Uptake and Sustainability of Effective Interventions to Promote Prudent Antibiotic Use in Primary Care (STEP-UP) Study. Available online: https://www.expmedndm.ox.ac.uk/stepup/home (accessed on 14 August 2019).
- Braun, V.; Clarke, V. Using thematic analysis in psychology. Qual. Res. Psychol. 2006, 3, 77–101. [Google Scholar] [CrossRef]
- Thomson, K.; Berry, R.; Robinson, T.; Brown, H.; Bambra, C.; Todd, A. An examination of trends in antibiotic prescribing in primary care and the association with area-level deprivation in England. BMC Public Health 2020, 20, 1148. [Google Scholar] [CrossRef]
- Covvey, J.R.; Johnson, B.F.; Elliott, V.; Malcolm, W.; Mullen, A.B. An association between socioeconomic deprivation and primary care antibiotic prescribing in Scotland. J. Antimicrob. Chemother. 2014, 69, 835–841. [Google Scholar] [CrossRef]
- Adekanmbi, V.; Jones, H.; Farewell, D.; Francis, N.A. Antibiotic use and deprivation: An analysis of Welsh primary care antibiotic prescribing data by socioeconomic status. J. Antimicrob. Chemother. 2020, 75, 2363–2371. [Google Scholar] [CrossRef]
- Frazer, J.S.; Frazer, G.R. GP prescribing in Northern Ireland by deprivation index: Retrospective analysis. Fam. Med. Community Health 2020, 8, e000376. [Google Scholar] [CrossRef] [PubMed]
- Borek, A.J.; Campbell, A.; Dent, E.; Butler, C.C.; Holmes, A.; Moore, M.; Walker, A.S.; Mcleod, M.; Tonkin-Crine, S. Implementing interventions to reduce antibiotic use: Views from high-prescribing practices. Under review.
- Coenen, S.; Francis, N.; Kelly, M.; Hood, K.; Nuttall, J.; Little, P.; Verheij, T.J.M.; Melbye, H.; Goossens, H.; Butler, C.C.; et al. Are Patient Views about Antibiotics Related to Clinician Perceptions, Management and Outcome? A Multi-Country Study in Outpatients with Acute Cough. PLoS ONE 2013, 8, e76691. [Google Scholar] [CrossRef] [PubMed]
- Stivers, T.; Mangione-Smith, R.; Elliott, M.N.; McDonald, L.; Heritage, J. Why do physicians think parents expect antibiotics? What parents report vs what physicians believe. J. Fam. Pract. 2003, 52, 140–147. [Google Scholar] [PubMed]
- Stuart, B.; Brotherwood, H.; Van’t Hoff, C.; Brown, A.; Van Den Bruel, A.; Hay, A.D.; Moore, M.; Little, P. Exploring the appropriateness of antibiotic prescribing for common respiratory tract infections in UK primary care. J. Antimicrob. Chemother. 2020, 75, 236–242. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Mölter, A.; White, A.; Welfare, W.; Palin, V.; Belmonte, M.; Ashcroft, D.M.; Sperrin, M.; van Staa, T.P. Relationship between prescribing of antibiotics and other medicines in primary care: A cross-sectional study. Br. J. Gen. Pract. 2019, 69, e42–e51. [Google Scholar] [CrossRef]
- Tarrant, C.; Krockow, E.M.; Nakkawita, W.M.I.D.; Bolscher, M.; Colman, A.M.; Chattoe-Brown, E.; Perera, N.; Mehtar, S.; Jenkins, D.R. Moral and Contextual Dimensions of “Inappropriate” Antibiotic Prescribing in Secondary Care: A Three-Country Interview Study. Front. Sociol. 2020, 5, 7. [Google Scholar] [CrossRef]
- Allison, R.; Lecky, D.M.; Beech, E.; Ashiru-Oredope, D.; Costelloe, C.; Owens, R.; McNulty, C.A.M. What Resources Do NHS Commissioning Organisations Use to Support Antimicrobial Stewardship in Primary Care in England? Antibiotics 2020, 9, 158. [Google Scholar] [CrossRef]
- Allison, R.; Lecky, D.M.; Beech, E.; Costelloe, C.; Ashiru-Oredope, D.; Owens, R.; McNulty, C.A.M. What antimicrobial stewardship strategies do NHS commissioning organizations implement in primary care in England? JAC Antimicrob. Resist. 2020, 2, dlaa020. [Google Scholar] [CrossRef]
- Allison, R.; Lecky, D.; Beech, E.; Ashiru-Oredope, D.; Costelloe, C.; Owens, R.; McNulty, C. Local implementation of national guidance on management of common infections in primary care in England. Pharm. J. 2020, 15, 44. [Google Scholar] [CrossRef]
- Høye, S.; Brænd, A.M.; Spehar, I. Quality improvement and antimicrobial stewardship in general practice—The role of the municipality chief medical officer. A qualitative study. Scand. J. Prim. Health Care 2020, 1–8. [Google Scholar] [CrossRef]
- Spehar, I.; Sjøvik, H.; Karevold, K.I.; Rosvold, E.O.; Frich, J.C. General practitioners’ views on leadership roles and challenges in primary health care: A qualitative study. Scand. J. Prim. Health Care 2017, 35, 105–110. [Google Scholar] [CrossRef] [PubMed]
- Hallsworth, M.; Chadborn, T.; Sallis, A.; Sanders, M.; Berry, D.; Greaves, F.; Clements, L.; Davies, S.C. Provision of social norm feedback to high prescribers of antibiotics in general practice: A pragmatic national randomised controlled trial. Lancet 2016, 387, 1743–1752. [Google Scholar] [CrossRef]
- Festinger, L. A theory of social comparison processes. Hum. Relat. 1954, 7, 117–140. [Google Scholar] [CrossRef]
- Gulliford, M.; Juszczyk, D.; Yardley, L. What is “normal” antibiotic prescribing? BMJ Br. Med J. 2019, 364, l1144. [Google Scholar] [CrossRef] [PubMed]
- Anyanwu, P.E.; Pouwels, K.; Walker, A.; Moore, M.; Majeed, A.; Hayhoe, B.W.J.; Tonkin-Crine, S.; Borek, A.; Hopkins, S.; Mcleod, M.; et al. Investigating the mechanism of impact and differential effect of the Quality Premium scheme on antibiotic prescribing in England: A longitudinal study. BJGP Open 2020, 4. [Google Scholar] [CrossRef] [PubMed]
| Characteristics | CCG Participants (n = 22, 33 CCGs) | General Practice Participants (n = 19) |
|---|---|---|
| Sex | 17 females, 5 males | 9 females, 10 males |
| Age (years) | 35–60 (mean 48) | 36–68 (mean 49) |
| Role | Leadership role, 11 Team member/prescribing advisor-type role, 11 | General practitioners, 14 Nurse prescribers, 3 Practice managers, 2 |
| Years in current organization | 1–20 (mean 6) | 1–35 (mean 13) General practitioners, 5–35, Nurse prescribers, 1–5, Practice managers, 7 and 10 |
| Years in current role/since qualified | 1–19 (mean 4) | 2–45 (mean 21) General practitioners, 8–45 Nurse prescribers, 2 and 11 |
| Size of CCG/general practice | 9–97 practices (mean 40) Small (<25 practices), 10 CCGs Medium (25–75 practices), 20 CCGs Large (>76 practices), 3 practices | 2–24 prescribers (mean 9) Small (2–5 prescribers), 7 practices Medium (6–15 prescribers), 11 practices Large (>20 prescribers), 1 practice |
| Deprivation 1 | High (1–3 decile), 6 CCGs Medium (4–7 decile), 12 CCGs Low (8–10 decile), 15 CCGs | High (1–3 decile), 8 practices Medium (4–7 decile), 7 practices Low (8–10 decile): 4 practices |
| Antibiotic prescribing rates 2 (items/STAR-PU) | High (quintiles 4–5), 13 CCGs Medium (quintile 3), 9 CCGs Low (quintiles 1–2), 11 CCGs | High (>0.27), 4 practices Medium (0.25–0.27), 7 practices Low (<0.25), 8 practices |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Borek, A.J.; Anthierens, S.; Allison, R.; Mcnulty, C.A.M.; Anyanwu, P.E.; Costelloe, C.; Walker, A.S.; Butler, C.C.; Tonkin-Crine, S.; on behalf of the STEP-UP Study Team. Social and Contextual Influences on Antibiotic Prescribing and Antimicrobial Stewardship: A Qualitative Study with Clinical Commissioning Group and General Practice Professionals. Antibiotics 2020, 9, 859. https://doi.org/10.3390/antibiotics9120859
Borek AJ, Anthierens S, Allison R, Mcnulty CAM, Anyanwu PE, Costelloe C, Walker AS, Butler CC, Tonkin-Crine S, on behalf of the STEP-UP Study Team. Social and Contextual Influences on Antibiotic Prescribing and Antimicrobial Stewardship: A Qualitative Study with Clinical Commissioning Group and General Practice Professionals. Antibiotics. 2020; 9(12):859. https://doi.org/10.3390/antibiotics9120859
Chicago/Turabian StyleBorek, Aleksandra J., Sibyl Anthierens, Rosalie Allison, Cliodna A. M. Mcnulty, Philip E. Anyanwu, Ceire Costelloe, Ann Sarah Walker, Christopher C. Butler, Sarah Tonkin-Crine, and on behalf of the STEP-UP Study Team. 2020. "Social and Contextual Influences on Antibiotic Prescribing and Antimicrobial Stewardship: A Qualitative Study with Clinical Commissioning Group and General Practice Professionals" Antibiotics 9, no. 12: 859. https://doi.org/10.3390/antibiotics9120859
APA StyleBorek, A. J., Anthierens, S., Allison, R., Mcnulty, C. A. M., Anyanwu, P. E., Costelloe, C., Walker, A. S., Butler, C. C., Tonkin-Crine, S., & on behalf of the STEP-UP Study Team. (2020). Social and Contextual Influences on Antibiotic Prescribing and Antimicrobial Stewardship: A Qualitative Study with Clinical Commissioning Group and General Practice Professionals. Antibiotics, 9(12), 859. https://doi.org/10.3390/antibiotics9120859





