Absence of mgrB Alleviates Negative Growth Effects of Colistin Resistance in Enterobacter cloacae
Abstract
1. Introduction
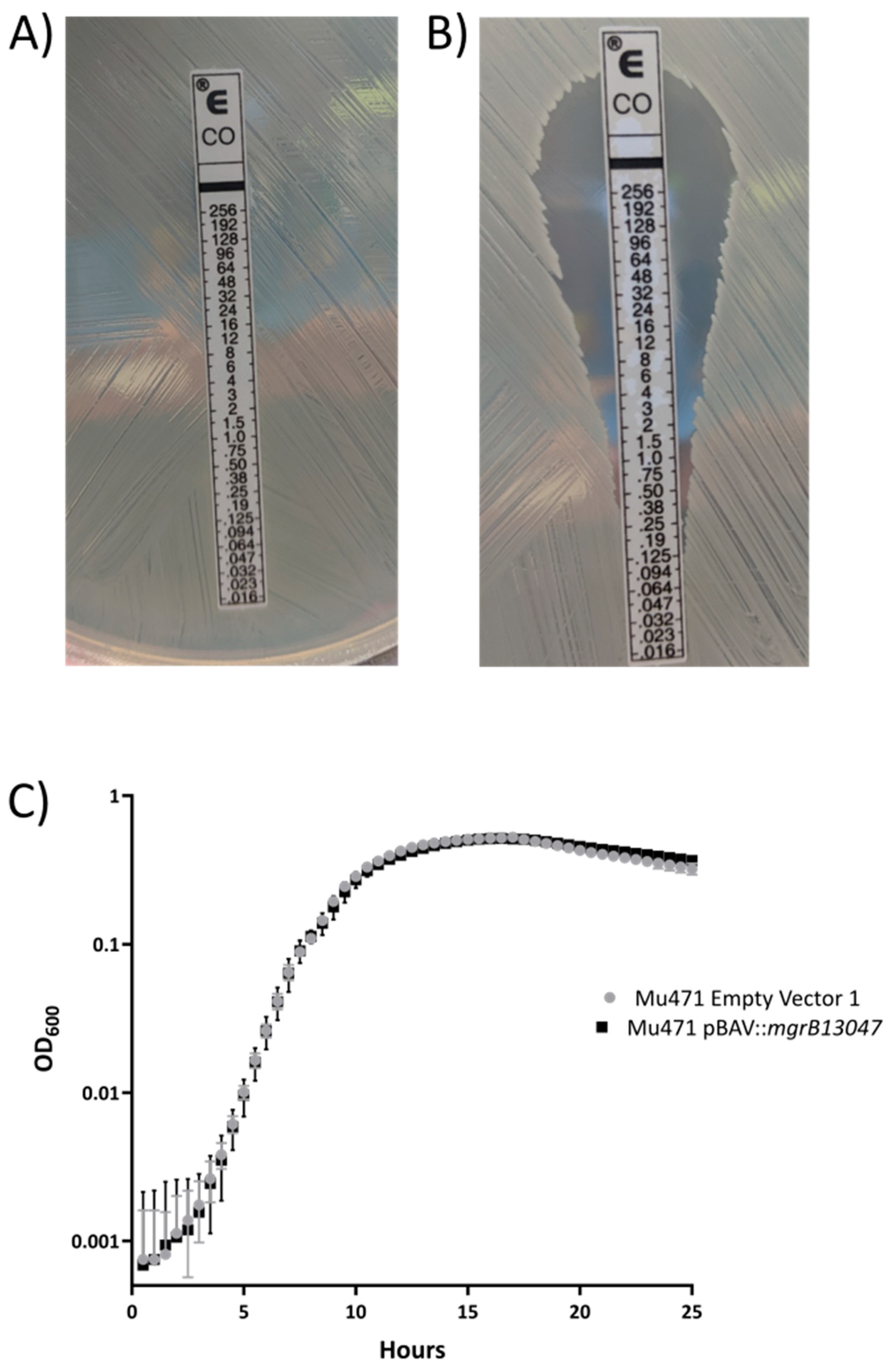
2. Results
3. Discussion
4. Materials and Methods
4.1. Bacterial Growth Conditions
4.2. Cloning
4.3. Growth Curves
4.4. Bioinformatics
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- CDC. Antibiotic Resistance Threats in the United States; Department of Health and Human Services: Atlanta, GA, USA, 2019.
- Burnham, J.P.; Olsen, M.A.; Kollef, M.H. Re-estimating annual deaths due to multidrug-resistant organism infections. Infect. Control Hosp. Epidemiol. 2018, 40, 112–113. [Google Scholar] [CrossRef] [PubMed]
- Logan, L.K.; Weinstein, R.A. The Epidemiology of Carbapenem-Resistant Enterobacteriaceae: The Impact and Evolution of a Global Menace. J. Infect. Dis. 2017, 215, S28–S36. [Google Scholar] [CrossRef] [PubMed]
- Soncini, F.C.; Vescovi, E.G.; Groisman, E.A. Transcriptional autoregulation of the Salmonella typhimurium phoPQ operon. J. Bacteriol. 1995, 177, 4364–4371. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Dalebroux, Z.D.; Miller, S.I. Salmonellae PhoPQ regulation of the outer membrane to resist innate immunity. Curr. Opin. Microbiol. 2014, 17, 106–113. [Google Scholar] [CrossRef]
- Groisman, E.A. The pleiotropic two-component regulatory system PhoP-PhoQ. J. Bacteriol. 2001, 183, 1835–1842. [Google Scholar] [CrossRef]
- Lippa, A.M.; Goulian, M. Feedback inhibition in the PhoQ/PhoP signaling system by a membrane peptide. PLoS Genet. 2009, 5, e1000788. [Google Scholar] [CrossRef]
- Band, V.I.; Crispell, E.K.; Napier, B.A.; Herrera, C.M.; Tharp, G.K.; Vavikolanu, K.; Pohl, J.; Read, T.D.; Bosinger, S.E.; Trent, M.S.; et al. Antibiotic failure mediated by a resistant subpopulation in Enterobacter cloacae. Nat. Microbiol. 2016, 1, 16053. [Google Scholar] [CrossRef]
- Kang, K.N.; Klein, D.R.; Kazi, M.I.; Guérin, F.; Cattoir, V.; Brodbelt, J.S.; Boll, J.M. Colistin heteroresistance in Enterobacter cloacae is regulated by PhoPQ-dependent 4-amino-4-deoxy-l-arabinose addition to lipid A. Mol. Microbiol. 2019, 111, 1604–1616. [Google Scholar] [CrossRef]
- Bhagirath, A.Y.; Li, Y.; Patidar, R.; Yerex, K.; Ma, X.; Kumar, A.; Duan, K. Two Component Regulatory Systems and Antibiotic Resistance in Gram-Negative Pathogens. Int. J. Mol. Sci. 2019, 20, 1781. [Google Scholar] [CrossRef]
- Beceiro, A.; Moreno, A.; Fernández, N.; Vallejo, J.A.; Aranda, J.; Adler, B.; Harper, M.; Boyce, J.D.; Bou, G. Biological cost of different mechanisms of colistin resistance and their impact on virulence in Acinetobacter baumannii. Antimicrob. Agents Chemother. 2014, 58, 518–526. [Google Scholar] [CrossRef]
- Nang, S.C.; Morris, F.C.; McDonald, M.J.; Han, M.L.; Wang, J.; Strugnell, R.A.; Velkov, T.; Li, J. Fitness cost of mcr-1-mediated polymyxin resistance in Klebsiella pneumoniae. J. Antimicrob. Chemother. 2018, 73, 1604–1610. [Google Scholar] [CrossRef]
- Lee, J.Y.; Park, Y.K.; Chung, E.S.; Na, I.Y.; Ko, K.S. Evolved resistance to colistin and its loss due to genetic reversion in Pseudomonas aeruginosa. Sci. Rep. 2016, 6, 25543. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-Y.; Choi, M.-J.; Choi, H.J.; Ko, K.S. Preservation of Acquired Colistin Resistance in Gram-Negative Bacteria. Antimicrob. Agents Chemother. 2015, 60, 609–612. [Google Scholar] [CrossRef] [PubMed]
- Hamel, M.; Chatzipanagiotou, S.; Hadjadj, L.; Petinaki, E.; Papagianni, S.; Charalampaki, N.; Tsiplakou, S.; Papaioannou, V.; Skarmoutsou, N.; Spiliopoulou, I.; et al. Inactivation of mgrB gene regulator and resistance to colistin is becoming endemic in carbapenem-resistant Klebsiella pneumoniae in Greece: A nationwide study from 2014 to 2017. Int. J. Antimicrob. Agents 2020, 55, 105930. [Google Scholar] [CrossRef] [PubMed]
- Cannatelli, A.; Giani, T.; D’Andrea, M.M.; Di Pilato, V.; Arena, F.; Conte, V.; Tryfinopoulou, K.; Vatopoulos, A.; Rossolini, G.M. MgrB inactivation is a common mechanism of colistin resistance in KPC-producing Klebsiella pneumoniae of clinical origin. Antimicrob. Agents Chemother. 2014, 58, 5696–5703. [Google Scholar] [CrossRef] [PubMed]
- Formosa, C.; Herold, M.; Vidaillac, C.; Duval, R.E.; Dague, E. Unravelling of a mechanism of resistance to colistin in Klebsiella pneumoniae using atomic force microscopy. J. Antimicrob. Chemother. 2015, 70, 2261–2270. [Google Scholar] [CrossRef] [PubMed]
- Mhaya, A.; Bégu, D.; Tounsi, S.; Arpin, C. MgrB Inactivation Is Responsible for Acquired Resistance to Colistin in Enterobacter hormaechei subsp. steigerwaltii. Antimicrob. Agents Chemother. 2020, 64. [Google Scholar] [CrossRef]
- Nawfal Dagher, T.; Al-Bayssari, C.; Chabou, S.; Baron, S.; Hadjadj, L.; Diene, S.M.; Azar, E.; Rolain, J.M. Intestinal carriage of colistin-resistant Enterobacteriaceae at Saint Georges Hospital in Lebanon. J. Glob. Antimicrob. Resist. 2020, 21, 386–390. [Google Scholar] [CrossRef]
- Magill, S.S.; Edwards, J.R.; Bamberg, W.; Beldavs, Z.G.; Dumyati, G.; Kainer, M.A.; Lynfield, R.; Maloney, M.; McAllister-Hollod, L.; Nadle, J.; et al. Multistate point-prevalence survey of health care-associated infections. N. Engl. J. Med. 2014, 370, 1198–1208. [Google Scholar] [CrossRef]
- Ren, Y.; Ren, Y.; Zhou, Z.; Guo, X.; Li, Y.; Feng, L.; Wang, L. Complete genome sequence of Enterobacter cloacae subsp. cloacae type strain ATCC 13047. J. Bacteriol. 2010, 192, 2463–2464. [Google Scholar] [CrossRef]
- Bryksin, A.V.; Matsumura, I. Rational design of a plasmid origin that replicates efficiently in both gram-positive and gram-negative bacteria. PLoS ONE 2010, 5, e13244. [Google Scholar] [CrossRef] [PubMed]
- Poirel, L.; Jayol, A.; Bontron, S.; Villegas, M.V.; Ozdamar, M.; Türkoglu, S.; Nordmann, P. The mgrB gene as a key target for acquired resistance to colistin in Klebsiella pneumoniae. J. Antimicrob. Chemother. 2015, 70, 75–80. [Google Scholar] [CrossRef] [PubMed]
- Cannatelli, A.; Santos-Lopez, A.; Giani, T.; Gonzalez-Zorn, B.; Rossolini, G.M. Polymyxin resistance caused by mgrB inactivation is not associated with significant biological cost in Klebsiella pneumoniae. Antimicrob. Agents Chemother. 2015, 59, 2898–2900. [Google Scholar] [CrossRef]
- Zhang, H.; Zhao, D.; Shi, Q.; Quan, J.; Li, X.; Yu, Y. mcr-1 Gene Has No Effect on Colistin Resistance When It Coexists with Inactivated mgrB Gene in Klebsiella pneumoniae. Microb. Drug Resist. 2018, 24, 1117–1120. [Google Scholar] [CrossRef] [PubMed]
- Salazar, M.E.; Podgornaia, A.I.; Laub, M.T. The small membrane protein MgrB regulates PhoQ bifunctionality to control PhoP target gene expression dynamics. Mol. Microbiol. 2016, 102, 430–445. [Google Scholar] [CrossRef]
- EUCAST. Recommendations for MIC Determination of Colistin (Polymyxin E) as Recommended by the Joint CLSI-EUCAST Polymyxin Breakpoints Working Group. Available online: https://www.eucast.org/fileadmin/src/media/PDFs/EUCAST_files/General_documents/Recommendations_for_MIC_determination_of_colistin_March_2016.pdf (accessed on 24 September 2020).
- Datsenko, K.A.; Wanner, B.L. One-step inactivation of chromosomal genes in Escherichia coli K-12 using PCR products. Proc. Natl. Acad. Sci. USA 2000, 97, 6640–6645. [Google Scholar] [CrossRef]
- Baba, T.; Ara, T.; Hasegawa, M.; Takai, Y.; Okumura, Y.; Baba, M.; Datsenko, K.A.; Tomita, M.; Wanner, B.L.; Mori, H. Construction of Escherichia coli K-12 in-frame, single-gene knockout mutants: The Keio collection. Mol. Syst. Biol 2006, 2, 2006.0008. [Google Scholar] [CrossRef]
- Chin, C.Y.; Tipton, K.A.; Farokhyfar, M.; Burd, E.M.; Weiss, D.S.; Rather, P.N. A high-frequency phenotypic switch links bacterial virulence and environmental survival in Acinetobacter baumannii. Nat. Microbiol. 2018, 3, 563–569. [Google Scholar] [CrossRef]
- Kurtz, S.; Phillippy, A.; Delcher, A.L.; Smoot, M.; Shumway, M.; Antonescu, C.; Salzberg, S.L. Versatile and open software for comparing large genomes. Genome Biol. 2004, 5, R12. [Google Scholar] [CrossRef]

| Strain Name | Species | Strain Information |
|---|---|---|
| Mu471 | Enterobacter cloacae | Collected by MuGSI from the urine of a female patient from a GA hospital in 2013 |
| ATCC13047 | Enterobacter cloacae | Purchased from ATCC |
| ATCC13047ΔmgrB | Enterobacter cloacae | Clean mgrB deletion generated as described below |
| Plasmid Name | Information |
|---|---|
| pBAV EV | Derived from pBAVIK-t5-GFP |
| pBAV mgrB | Complementation plasmid containing mgrB generated as described below |
| Primer Name | Sequence |
|---|---|
| pBAV mgrB F | ccacaattatgatagaatttgacgtcgaattccattgcctctttatctttgttgtcatgc |
| pBAV mgrB R | gcggcggcatcgatcgggccctgaggcctgcagttcaccacctcaataaaaacacgc |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wozniak, J.E.; Chande, A.T.; Burd, E.M.; Band, V.I.; Satola, S.W.; Farley, M.M.; Jacob, J.T.; Jordan, I.K.; Weiss, D.S. Absence of mgrB Alleviates Negative Growth Effects of Colistin Resistance in Enterobacter cloacae. Antibiotics 2020, 9, 825. https://doi.org/10.3390/antibiotics9110825
Wozniak JE, Chande AT, Burd EM, Band VI, Satola SW, Farley MM, Jacob JT, Jordan IK, Weiss DS. Absence of mgrB Alleviates Negative Growth Effects of Colistin Resistance in Enterobacter cloacae. Antibiotics. 2020; 9(11):825. https://doi.org/10.3390/antibiotics9110825
Chicago/Turabian StyleWozniak, Jessie E., Aroon T. Chande, Eileen M. Burd, Victor I. Band, Sarah W. Satola, Monica M. Farley, Jesse T. Jacob, I. King Jordan, and David S. Weiss. 2020. "Absence of mgrB Alleviates Negative Growth Effects of Colistin Resistance in Enterobacter cloacae" Antibiotics 9, no. 11: 825. https://doi.org/10.3390/antibiotics9110825
APA StyleWozniak, J. E., Chande, A. T., Burd, E. M., Band, V. I., Satola, S. W., Farley, M. M., Jacob, J. T., Jordan, I. K., & Weiss, D. S. (2020). Absence of mgrB Alleviates Negative Growth Effects of Colistin Resistance in Enterobacter cloacae. Antibiotics, 9(11), 825. https://doi.org/10.3390/antibiotics9110825





