Antifungal Drug Repurposing
Abstract
1. Introduction
2. Drug Repurposing Approaches
2.1. Repurposing Approaches for the Human Therapeutic Drugs (Non-Antifungals)
2.2. Repurposing Approaches for the New Antifungal Drugs
3. Antifungal Drug Repurposing: Current Measures
3.1. In Silico/Computational Repurposing Approaches
3.2. Experimental Repurposing Approaches
3.2.1. Standard Dilution Methods: CLSI and EUCAST
3.2.2. Biofilm Analysis
3.2.3. Phenotypic Variability of Infecting Fungi: Conidia, Hyphal, Yeast, and Filamentous Growth
3.2.4. Animal Model Systems
3.3. Synergism between Repurposed Agents and Conventional Antifungals
4. Challenges
4.1. Pioglitazone: Needs for In Vivo Drug Validation
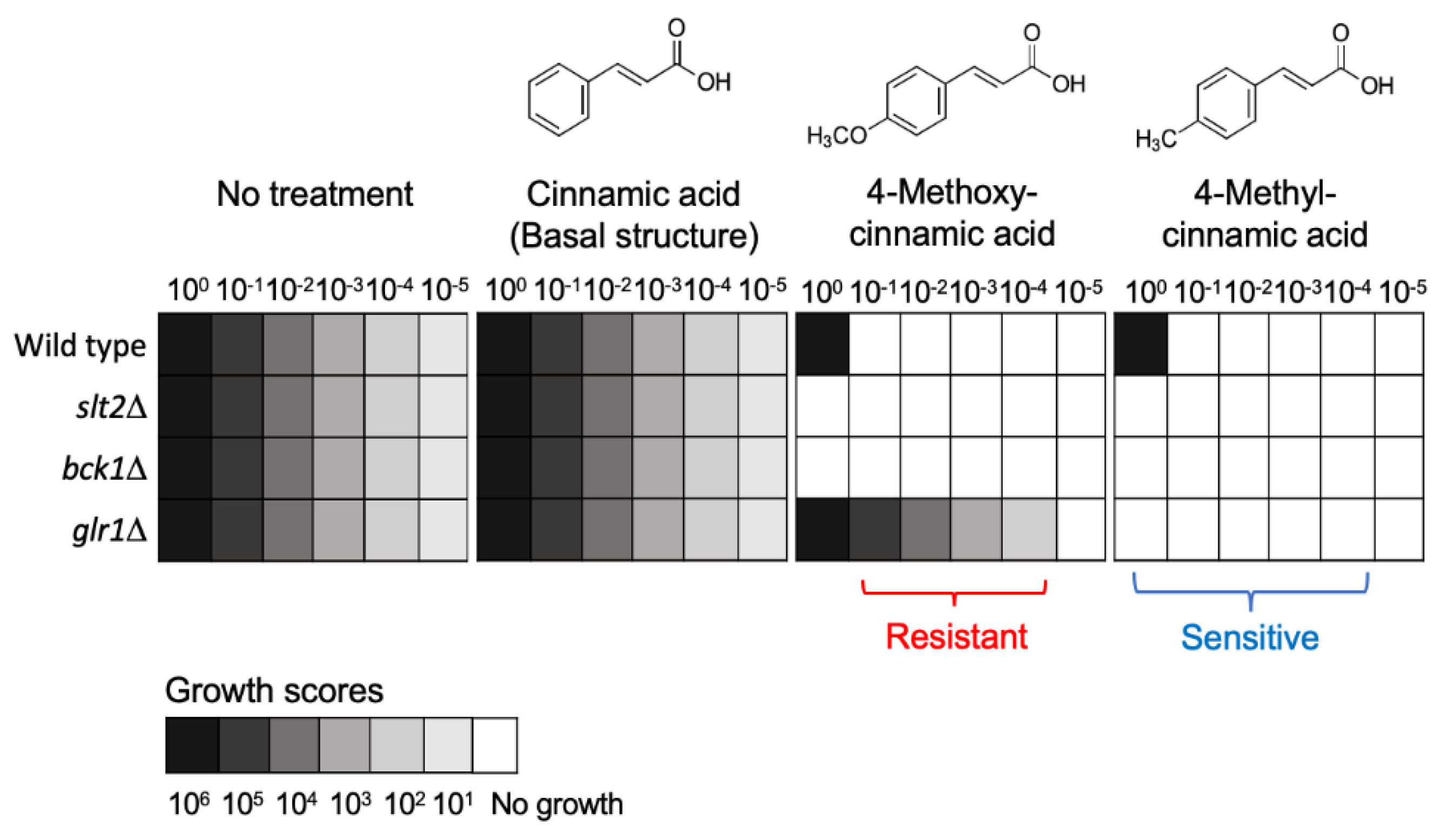
4.2. Resistance to Repurposed Drugs/Compounds: Cinnamic and Benzoic Derivatives
5. Summary
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Perfect, J.R. The antifungal pipeline: A reality check. Nat. Rev. Drug Discov. 2017, 16, 603–616. [Google Scholar] [CrossRef] [PubMed]
- Roemer, T.; Krysan, D.J. Antifungal drug development: Challenges, unmet clinical needs, and new approaches. Cold Spring Harb. Perspect. Med. 2014, 4. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, M.W.; Kontoyiannis, D.P.; Cornely, O.A.; Perfect, J.R.; Walsh, T.J. Novel agents and drug targets to meet the challenges of resistant Fungi. J. Infect. Dis. 2017, 216, S474–S483. [Google Scholar] [CrossRef] [PubMed]
- CDC. Antibiotic Resistance Threats in the United States; U.S. Department of Health and Human Services, CDC: Atlanta, GA, USA, 2019. [CrossRef]
- Bowyer, P.; Denning, D.W. Environmental fungicides and triazole resistance in Aspergillus. Pest. Manag. Sci. 2014, 70, 173–178. [Google Scholar] [CrossRef] [PubMed]
- Nett, J.E.; Andes, D.R. Antifungal agents: Spectrum of activity, pharmacology, and clinical indications. Infect. Dis. Clin. N. Am. 2016, 30, 51–83. [Google Scholar] [CrossRef] [PubMed]
- Nami, S.; Aghebati-Maleki, A.; Morovati, H.; Aghebati-Maleki, L. Current antifungal drugs and immunotherapeutic approaches as promising strategies to treatment of fungal diseases. Biomed. Pharmacother. 2019, 110, 857–868. [Google Scholar] [CrossRef] [PubMed]
- Houšť, J.; Spížek, J.; Havlíček, V. Antifungal drugs. Metabolites 2020, 10, 106. [Google Scholar] [CrossRef]
- Marena, G.D.; Ramos, M.A.D.S.; Bauab, T.M.; Chorilli, M. A critical review of analytical methods for quantification of amphotericin B in biological samples and pharmaceutical formulations. Crit. Rev. Anal. Chem. 2020. [Google Scholar] [CrossRef]
- Hamill, R.J. Amphotericin B formulations: A comparative review of efficacy and toxicity. Drugs 2013, 73, 919–934. [Google Scholar] [CrossRef]
- Tillotson, J.; Tillotson, G.S. The regulatory pathway for antifungal drugs: A US perspective. Clin. Infect. Dis. 2015, 61, S678–S683. [Google Scholar] [CrossRef]
- Cha, Y.; Erez, T.; Reynolds, I.J.; Kumar, D.; Ross, J.; Koytiger, G.; Kusko, R.; Zeskind, B.; Risso, S.; Kagan, E.; et al. Drug repurposing from the perspective of pharmaceutical companies. Br. J. Pharmacol. 2018, 175, 168–180. [Google Scholar] [CrossRef] [PubMed]
- Hernandez, J.J.; Pryszlak, M.; Smith, L.; Yanchus, C.; Kurji, N.; Shahani, V.M.; Molinski, S.V. Giving drugs a second chance: Overcoming regulatory and financial hurdles in repurposing approved drugs as cancer therapeutics. Front. Oncol. 2017, 7. [Google Scholar] [CrossRef] [PubMed]
- National Center for Biotechnology Information. PubMed. Available online: https://www.ncbi.nlm.nih.gov/pubmed (accessed on 14 August 2020).
- De Oliveira, A.A.; Neves, B.J.; Silva, L.D.C.; Soares, C.M.D.A.; Andrade, C.H.; Pereira, M. Drug repurposing for paracoccidioidomycosis through a computational chemogenomics framework. Front. Microbiol 2019, 10. [Google Scholar] [CrossRef] [PubMed]
- Khedr, M.A.; Massarotti, A.; Mohamed, M.E. Rational discovery of (+) (S) abscisic acid as a potential antifungal agent: A repurposing approach. Sci. Rep. 2018, 8, 8565. [Google Scholar] [CrossRef]
- Krajaejun, T.; Lohnoo, T.; Yingyong, W.; Rujirawat, T.; Kumsang, Y.; Jongkhajornpong, P.; Theerawatanasirikul, S.; Kittichotirat, W.; Reamtong, O.; Yolanda, H. The repurposed drug disulfiram inhibits urease and aldehyde dehydrogenase and prevents In Vitro growth of the oomycete Pythium insidiosum. Antimicrob. Agents Chemother. 2019, 63, e00609–e00619. [Google Scholar] [CrossRef]
- Rana, R.; Sharma, R.; Kumar, A. Repurposing of fluvastatin against Candida albicans CYP450 lanosterol 14 α-demethylase, a target enzyme for antifungal therapy: An In silico and In vitro study. Curr. Mol. Med. 2019, 19, 506–524. [Google Scholar] [CrossRef]
- Tavakkoli, A.; Johnston, T.P.; Sahebkar, A. Antifungal effects of statins. Pharmacol. Ther. 2020, 208, 107483. [Google Scholar] [CrossRef]
- Capoci, I.R.G.; Faria, D.R.; Sakita, K.M.; Rodrigues-Vendramini, F.A.V.; Bonfim-Mendonça, P.D.S.; Becker, T.C.A.; Kioshima, É.S.; Svidzinski, T.I.E.; Maigret, B. Repurposing approach identifies new treatment options for invasive fungal disease. Bioorg. Chem. 2019, 84, 87–97. [Google Scholar] [CrossRef]
- Gowri, M.; Jayashree, B.; Jeyakanthan, J.; Girija, E.K. Sertraline as a promising antifungal agent: Inhibition of growth and biofilm of Candida auris with special focus on the mechanism of action in vitro. J. Appl. Microbiol. 2020, 128, 426–437. [Google Scholar] [CrossRef]
- Khan, S.; Singhal, S.; Mathur, T.; Upadhyay, D.J.; Rattan, A. Antifungal potential of disulfiram. Nippon Ishinkin Gakkai Zasshi 2007, 48, 109–113. [Google Scholar] [CrossRef][Green Version]
- Spitzer, M.; Griffiths, E.; Blakely, K.M.; Wildenhain, J.; Ejim, L.; Rossi, L.; De Pascale, G.; Curak, J.; Brown, E.; Tyers, M.; et al. Cross-species discovery of syncretic drug combinations that potentiate the antifungal fluconazole. Mol. Syst. Biol. 2011, 7, 499. [Google Scholar] [CrossRef] [PubMed]
- Villanueva-Lozano, H.; González, G.M.; Espinosa-Mora, J.E.; Bodden-Mendoza, B.A.; Andrade, A.; Martínez-Reséndez, M.F.; Treviño-Rangel, R.d.J. Evaluation of the expanding spectrum of sertraline against uncommon fungal pathogens. J. Infect. Chemother. 2020, 26, 309–311. [Google Scholar] [CrossRef] [PubMed]
- Wall, G.; Chaturvedi, A.K.; Wormley, F.L.; Wiederhold, N.P.; Patterson, H.P.; Patterson, T.F.; Lopez-Ribot, J.L. Screening a repurposing library for inhibitors of multidrug-resistant Candida auris identifies ebselen as a repositionable candidate for antifungal drug development. Antimicrob. Agents Chemother. 2018, 62, e01084-18. [Google Scholar] [CrossRef] [PubMed]
- Venturini, T.P.; Chassot, F.; Loreto, É.S.; Keller, J.T.; Azevedo, M.I.; Zeni, G.; Santurio, J.M.; Alves, S.H. Antifungal activities of diphenyl diselenide and ebselen alone and in combination with antifungal agents against Fusarium spp. Med. Mycol. 2016, 54, 550–555. [Google Scholar] [CrossRef] [PubMed]
- Giurg, M.; Gołąb, A.; Suchodolski, J.; Kaleta, R.; Krasowska, A.; Piasecki, E.; Piętka-Ottlik, M. Reaction of bis[(2-chlorocarbonyl)phenyl] diselenide with phenols, aminophenols, and other amines towards diphenyl diselenides with antimicrobial and antiviral properties. Molecules 2017, 22, 974. [Google Scholar] [CrossRef] [PubMed]
- Felli Kubiça, T.; Bedin Denardi, L.; de Loreto, É.S.; Zeni, G.; Weiblen, C.; Oliveira, V.; Morais Santurio, J.; Hartz Alves, S. In vitro activity of diphenyl diselenide and ebselen alone and in combination with antifungal agents against Trichosporon. Asahii. Mycoses 2019, 62, 428–433. [Google Scholar] [CrossRef] [PubMed]
- Cong, L.; Liao, Y.; Yang, S.; Yang, R. In vitro antifungal activity of sertraline and synergistic effects in combination with antifungal drugs against planktonic forms and biofilms of clinical Trichosporon asahii isolates. PLoS ONE 2016, 11, e0167903. [Google Scholar] [CrossRef]
- De Cremer, K.; Lanckacker, E.; Cools, T.L.; Bax, M.; De Brucker, K.; Cos, P.; Cammue, B.P.A.; Thevissen, K. Artemisinins, new miconazole potentiators resulting in increased activity against Candida albicans biofilms. Antimicrob. Agents Chemother. 2015, 59, 421–426. [Google Scholar] [CrossRef]
- Gao, L.; Sun, Y.; He, C.; Zeng, T.; Li, M. Synergy between pyrvinium pamoate and azoles against Exophiala dermatitidis. Antimicrob. Agents Chemother. 2018, 62, e02361-17. [Google Scholar] [CrossRef]
- Holbrook, S.Y.L.; Garzan, A.; Dennis, E.K.; Shrestha, S.K.; Garneau-Tsodikova, S. Repurposing antipsychotic drugs into antifungal agents: Synergistic combinations of azoles and bromperidol derivatives in the treatment of various fungal infections. Eur. J. Med. Chem. 2017, 139, 12–21. [Google Scholar] [CrossRef]
- Simm, C.; May, R.C. Zinc and iron homeostasis: Target-based drug screening as new route for antifungal drug development. Front. Cell Infect. Microbiol. 2019, 9. [Google Scholar] [CrossRef] [PubMed]
- Zeng, B.; Li, J.; Wang, Y.; Chen, P.; Wang, X.; Cui, J.; Liu, L.; Hu, X.; Cao, Q.; Xiao, Y.; et al. In vitro and in vivo effects of suloctidil on growth and biofilm formation of the opportunistic fungus Candida albicans. Oncotarget 2017, 8, 69972. [Google Scholar] [CrossRef] [PubMed]
- Yousfi, H.; Ranque, S.; Rolain, J.-M.; Bittar, F. In vitro polymyxin activity against clinical multidrug-resistant fungi. Antimicrob. Resist. Infect. Control 2019, 8, 66. [Google Scholar] [CrossRef] [PubMed]
- Voget, M.; Lorenz, D.; Lieber-Tenorio, E.; Hauck, R.; Meyer, M.; Cieslicki, M. Is transmission electron microscopy (TEM) a promising approach for qualitative and quantitative investigations of polymyxin B and miconazole interactions with cellular and subcellular structures of Staphylococcus pseudintermedius, Escherichia coli, Pseudomonas aeruginosa and Malassezia pachydermatis? Vet. Microbiol. 2015, 181, 261–270. [Google Scholar] [CrossRef] [PubMed]
- Schemuth, H.; Dittmer, S.; Lackner, M.; Sedlacek, L.; Hamprecht, A.; Steinmann, E.; Buer, J.; Rath, P.-M.; Steinmann, J. In vitro activity of colistin as single agent and in combination with antifungals against filamentous fungi occurring in patients with cystic fibrosis. Mycoses 2013, 56, 297–303. [Google Scholar] [CrossRef] [PubMed]
- Mashaly, G.; Shrief, R. Candida glabrata complex from patients with healthcare-associated infections in Mansoura University Hospitals, Egypt: Distribution, antifungal susceptibility and effect of fluconazole and polymyxin B combination. Germs 2019, 9, 125–132. [Google Scholar] [CrossRef] [PubMed]
- Cui, J.; Ren, B.; Tong, Y.; Dai, H.; Zhang, L. Synergistic combinations of antifungals and anti-virulence agents to fight against Candida albicans. Virulence 2015, 6, 362–371. [Google Scholar] [CrossRef]
- Kirchhoff , L.; Olsowski, M.; Zilmans, K.; Dittmer, S.; Haase, G.; Sedlacek, L.; Steinmann, E.; Buer, J.; Rath, P.-M.; Steinmann, J. Biofilm formation of the black yeast-like fungus Exophiala dermatitidis and its susceptibility to antiinfective agents. Sci. Rep. 2017, 7, 42886. [Google Scholar] [CrossRef]
- Hsu, L.-H.; Wang, H.-F.; Sun, P.-L.; Hu, F.-R.; Chen, Y.-L. The antibiotic polymyxin B exhibits novel antifungal activity against Fusarium species. Int. J. Antimicrob. Agents 2017, 49, 740–748. [Google Scholar] [CrossRef]
- Borjihan, B.; Ogita, A.; Fujita, K.-i.; Doe, M.; Tanaka, T. The cyclic organosulfur compound zwiebelane a from onion (Allium cepa) functions as an enhancer of polymyxin b in fungal vacuole disruption. Planta Med. 2010, 76, 1864–1866. [Google Scholar] [CrossRef]
- Blooi, M.; Pasmans, F.; Rouffaer, L.; Haesebrouck, F.; Vercammen, F.; Martel, A. Successful treatment of Batrachochytrium salamandrivorans infections in salamanders requires synergy between voriconazole, polymyxin E and temperature. Sci. Rep. 2015, 5, 11788. [Google Scholar] [CrossRef] [PubMed]
- Bidaud, A.L.; Djenontin, E.; Botterel, F.; Chowdhary, A.; Dannaoui, E. Colistin interacts synergistically with echinocandins against Candida auris. Int. J. Antimicrob. Agents 2020, 55, 105901. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Yang, H.; Zhou, X.; Luo, H.; Tang, F.; Yang, J.; Alterovitz, G.; Cheng, L.; Ren, B. Lovastatin synergizes with itraconazole against planktonic cells and biofilms of Candida albicans through the regulation on ergosterol biosynthesis pathway. Appl. Microbiol. Biotechnol. 2018, 102, 5255–5264. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Fang, Y.; Jaiseng, W.; Hu, L.; Lu, Y.; Ma, Y.; Furuyashiki, T. Characterization of tamoxifen as an antifungal agent using the yeast schizosaccharomyces pombe model organism. Kobe J. Med. Sci. 2015, 61, E54–E63. [Google Scholar] [PubMed]
- Muthular, M.; Bálsamo, F.; Passero, P.; Jewtuchowicz, V.; Miozza, V.; Villalba, M.B.; Brusca, M.I.; Pérez, C. Effects of tamoxifen on periodontal disease and Candida albicans of patients with breast cancer and other pathologies. Future Microbiol. 2019, 14, 129–137. [Google Scholar] [CrossRef] [PubMed]
- Hai, T.P.; Van, A.D.; Ngan, N.T.T.; Nhat, L.T.H.; Lan, N.P.H.; Chau, N.V.V.; Thwaites, G.E.; Krysan, D.; Day, J.N. The combination of tamoxifen with amphotericin B, but not with fluconazole, has synergistic activity against the majority of clinical isolates of Cryptococcus neoformans. Mycoses 2019, 62, 818–825. [Google Scholar] [CrossRef]
- Butts, A.; Koselny, K.; Chabrier-Roselló, Y.; Semighini, C.P.; Brown, J.C.S.; Wang, X.; Annadurai, S.; DiDone, L.; Tabroff, J.; Childers, W.E.; et al. Estrogen receptor antagonists are anti-cryptococcal agents that directly bind ef hand proteins and synergize with fluconazole In Vivo. mBio 2014, 5, e00765-13. [Google Scholar] [CrossRef]
- Naeimi Eshkaleti, M.; Kordbacheh, P.; Hashemi, S.J.; Falahati, M.; Zaini, F.; Mirhendi, H.; Safara, M.; Hosseinpoor, L. In vitro activity of amphotericin B in combination with statins against clinical and environmental Rhizopus oryzae strains. Iran. J. Public Health 2019, 48, 943–948. [Google Scholar] [CrossRef]
- Nyilasi, I.; Kocsubé, S.; Krizsán, K.; Galgóczy, L.; Papp, T.; Pesti, M.; Nagy, K.; Vágvölgyi, C. Susceptibility of clinically important dermatophytes against statins and different statin-antifungal combinations. Med. Mycol. 2013, 52, 140–148. [Google Scholar] [CrossRef]
- Nyilasi, I.; Kocsubé, S.; Krizsán, K.; Galgóczy, L.; Pesti, M.; Papp, T.; Vágvölgyi, C. In vitro synergistic interactions of the effects of various statins and azoles against some clinically important fungi. FEMS Microbiol. Lett. 2010, 307, 175–184. [Google Scholar] [CrossRef]
- Qiao, J.; Kontoyiannis, D.P.; Wan, Z.; Li, R.; Liu, W. Antifungal activity of statins against Aspergillus species. Med. Mycol. 2007, 45, 589–593. [Google Scholar] [CrossRef] [PubMed]
- Roze, L.V.; Linz, J.E. Lovastatin triggers an apoptosis-like cell death process in the fungus Mucor. Racemosus. Fungal. Genet. Biol. 1998, 25, 119–133. [Google Scholar] [CrossRef] [PubMed]
- Cabral, M.E.; Figueroa, L.I.C.; Fariña, J.I. Synergistic antifungal activity of statin–azole associations as witnessed by Saccharomyces cerevisiae- and Candida utilis-bioassays and ergosterol quantification. Rev. Iberoam. Micol. 2013, 30, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Chamilos, G.; Lewis, R.E.; Kontoyiannis, D.P. Lovastatin has significant activity against zygomycetes and interacts synergistically with voriconazole. Antimicrob. Agents Chemother. 2006, 50, 96–103. [Google Scholar] [CrossRef] [PubMed]
- Galgóczy, L.; Papp, T.; Kovács, L.; Ördögh, L.; Vágvölgyi, C. In vitro activity of phenothiazines and their combinations with amphotericin B against Zygomycetes causing rhinocerebral zygomycosis. Med. Mycol. 2009, 47, 331–335. [Google Scholar] [CrossRef][Green Version]
- Lorenz, R.T.; Parks, L.W. Effects of lovastatin (mevinolin) on sterol levels and on activity of azoles in Saccharomyces cerevisiae. Antimicrob. Agents Chemother. 1990, 34, 1660–1665. [Google Scholar] [CrossRef]
- Macreadie, I.G.; Johnson, G.; Schlosser, T.; Macreadie, P.I. Growth inhibition of Candida species and Aspergillus fumigatus by statins. FEMS Microbiol. Lett. 2006, 262, 9–13. [Google Scholar] [CrossRef]
- Alem, M.A.S.; Douglas, L.J. Effects of aspirin and other nonsteroidal anti-inflammatory drugs on biofilms and planktonic cells of Candida albicans. Antimicrob. Agents Chemother. 2004, 48, 41–47. [Google Scholar] [CrossRef]
- Leeuw, N.J.; Swartt, C.W.; Ncango, D.M.; Kriell, W.M.; Pohl, C.H.; van Wyk, P.W.J.; Kock, J.L.F. Anti-inflammatory drugs selectively target sporangium development in Mucor. Can. J. Microbiol. 2009, 55, 1392–1396. [Google Scholar] [CrossRef]
- Yang, S.; Liao, Y.; Cong, L.; Lu, X.; Yang, R. In Vitro interactions between non-steroidal anti-inflammatory drugs and antifungal agents against planktonic and biofilm forms of Trichosporon asahii. PLoS ONE 2016, 11, e0157047. [Google Scholar] [CrossRef]
- Ogundeji, A.O.; Pohl, C.H.; Sebolai, O.M. Repurposing of aspirin and ibuprofen as candidate anti-Cryptococcus drugs. Antimicrob. Agents Chemother. 2016, 60, 4799–4808. [Google Scholar] [CrossRef] [PubMed]
- Rosato, A.; Catalano, A.; Carocci, A.; Carrieri, A.; Carone, A.; Caggiano, G.; Franchini, C.; Corbo, F.; Montagna, M.T. In vitro interactions between anidulafungin and nonsteroidal anti-inflammatory drugs on biofilms of Candida spp. Bioorg. Med. Chem. 2016, 24, 1002–1005. [Google Scholar] [CrossRef] [PubMed]
- Yasmin, S.; Alcazar-Fuoli, L.; Gründlinger, M.; Puempel, T.; Cairns, T.; Blatzer, M.; Lopez, J.F.; Grimalt, J.O.; Bignell, E.; Haas, H. Mevalonate governs interdependency of ergosterol and siderophore biosyntheses in the fungal pathogen Aspergillus fumigatus. Proc. Natl. Acad. Sci. USA 2012, 109, E497–E504. [Google Scholar] [CrossRef] [PubMed]
- Rossi, S.A.; de Oliveira, H.C.; Agreda-Mellon, D.; Lucio, J.; Mendes-Giannini, M.J.S.; García-Cambero, J.P.; Zaragoza, O. Identification of off-patent drugs that show synergism with amphotericin B or that present antifungal action against Cryptococcus neoformans and Candida spp. Antimicrob. Agents Chemother. 2020, 64, e01921-19. [Google Scholar] [CrossRef]
- Król, J.; Nawrot, U.; Bartoszewicz, M. Anti-candidal activity of selected analgesic drugs used alone and in combination with fluconazole, itraconazole, voriconazole, posaconazole and isavuconazole. J. Mycol. Med. 2018, 28, 327–331. [Google Scholar] [CrossRef]
- Delattin, N.; De Brucker, K.; Vandamme, K.; Meert, E.; Marchand, A.; Chaltin, P.; Cammue, B.P.A.; Thevissen, K. Repurposing as a means to increase the activity of amphotericin B and caspofungin against Candida albicans biofilms. J. Antimicrob. Chemother. 2013, 69, 1035–1044. [Google Scholar] [CrossRef]
- Al-Janabi, A.A.H.S. Determination of antidermatophytic effects of non-steroidal anti-inflammatory drugs on Trichophyton mentagrophytes and Epidermophyton floccosum. Mycoses 2011, 54, e443–e448. [Google Scholar] [CrossRef]
- Al-Bakri, A.G.; Othman, G.; Bustanji, Y. The assessment of the antibacterial and antifungal activities of aspirin, EDTA and aspirin-EDTA combination and their effectiveness as antibiofilm agents. J. Appl. Microbiol. 2009, 107, 280–286. [Google Scholar] [CrossRef]
- Roohparvar, R.; Huser, A.; Zwiers, L.-H.; De Waard, M.A. Control of Mycosphaerella graminicola on wheat seedlings by medical drugs known to modulate the activity of ATP-binding cassette transporters. Appl. Environ. Microbiol. 2007, 73, 5011–5019. [Google Scholar] [CrossRef]
- Loreto, E.S.; Tondolo, J.S.M.; Santurio, J.M.; Alves, S.H. Screening of antibacterial drugs for antimicrobial activity against Pythium insidiosum. Med. Mycol. 2018, 57, 523–525. [Google Scholar] [CrossRef]
- Ko, H.-T.; Hsu, L.-H.; Yang, S.-Y.; Chen, Y.-L. Repurposing the thrombopoietin receptor agonist eltrombopag as an anticryptococcal agent. Med. Mycol. 2019, 58, 493–504. [Google Scholar] [CrossRef] [PubMed]
- Caldara, M.; Marmiroli, N. Tricyclic antidepressants inhibit Candida albicans growth and biofilm formation. Int. J. Antimicrob. Agents 2018, 52, 500–505. [Google Scholar] [CrossRef] [PubMed]
- Butts, A.; DiDone, L.; Koselny, K.; Baxter, B.K.; Chabrier-Rosello, Y.; Wellington, M.; Krysan, D.J. A repurposing approach identifies off-patent drugs with fungicidal cryptococcal activity, a common structural chemotype, and pharmacological properties relevant to the treatment of cryptococcosis. Eukaryot. Cell 2013, 12, 278–287. [Google Scholar] [CrossRef] [PubMed]
- Bagar, T.; Benčina, M. Antiarrhythmic drug amiodarone displays antifungal activity, induces irregular calcium response and intracellular acidification of Aspergillus niger—Amiodarone targets calcium and pH homeostasis of A. niger. Fungal. Genet. Biol. 2012, 49, 779–791. [Google Scholar] [CrossRef] [PubMed]
- Eldesouky, H.E.; Salama, E.A.; Li, X.; Hazbun, T.R.; Mayhoub, A.S.; Seleem, M.N. Repurposing approach identifies pitavastatin as a potent azole chemosensitizing agent effective against azole-resistant Candida species. Sci. Rep. 2020, 10, 7525. [Google Scholar] [CrossRef]
- Ji, C.; Liu, N.; Tu, J.; Li, Z.; Han, G.; Li, J.; Sheng, C. Drug repurposing of haloperidol: Discovery of new benzocyclane derivatives as potent antifungal agents against cryptococcosis and candidiasis. ACS Infect. Dis. 2020, 6, 768–786. [Google Scholar] [CrossRef]
- Stylianou, M.; Kulesskiy, E.; Lopes, J.P.; Granlund, M.; Wennerberg, K.; Urban, C.F. Antifungal application of nonantifungal drugs. Antimicrob. Agents Chemother. 2014, 58, 1055–1062. [Google Scholar] [CrossRef]
- Venturini, T.P.; Al-Hatmi, A.M.S.; Rossato, L.; Azevedo, M.I.; Keller, J.T.; Weiblen, C.; Santurio, J.M.; Alves, S.H. Do antibacterial and antifungal combinations have better activity against clinically relevant fusarium species? in vitro synergism. Int. J. Antimicrob. Agents 2018, 51, 784–788. [Google Scholar] [CrossRef]
- Aneke, C.I.; Rhimi, W.; Otranto, D.; Cafarchia, C. Synergistic effects of efflux pump modulators on the azole antifungal susceptibility of Microsporum canis. Mycopathologia 2020, 185, 279–288. [Google Scholar] [CrossRef]
- Iatta, R.; Puttilli, M.R.; Immediato, D.; Otranto, D.; Cafarchia, C. The role of drug efflux pumps in Malassezia pachydermatis and Malassezia furfur defence against azoles. Mycoses 2017, 60, 178–182. [Google Scholar] [CrossRef]
- Mamouei, Z.; Alqarihi, A.; Singh, S.; Xu, S.; Mansour, M.K.; Ibrahim, A.S.; Uppuluri, P. Alexidine Dihydrochloride Has Broad-Spectrum Activities against Diverse Fungal Pathogens. mSphere 2018, 3. [Google Scholar] [CrossRef] [PubMed]
- Shekhar-Guturja, T.; Gunaherath, G.M.K.B.; Wijeratne, E.M.K.; Lambert, J.-P.; Averette, A.F.; Lee, S.C.; Kim, T.; Bahn, Y.-S.; Tripodi, F.; Ammar, R.; et al. Dual action antifungal small molecule modulates multidrug efflux and TOR signaling. Nat. Chem. Biol. 2016, 12, 867–875. [Google Scholar] [CrossRef] [PubMed]
- Tong, Y.; Liu, M.; Zhang, Y.; Liu, X.; Huang, R.; Song, F.; Dai, H.; Ren, B.; Sun, N.; Pei, G.; et al. Beauvericin counteracted multi-drug resistant Candida albicans by blocking ABC transporters. Synth. Syst. Biotechnol. 2016, 1, 158–168. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Yan, K.; Zhang, Y.; Huang, R.; Bian, J.; Zheng, C.; Sun, H.; Chen, Z.; Sun, N.; An, R.; et al. High-throughput synergy screening identifies microbial metabolites as combination agents for the treatment of fungal infections. Proc. Natl. Acad. Sci. USA 2007, 104, 4606–4611. [Google Scholar] [CrossRef] [PubMed]
- Shekhar-Guturja, T.; Tebung, W.A.; Mount, H.; Liu, N.; Köhler, J.R.; Whiteway, M.; Cowen, L.E. Beauvericin potentiates azole activity via inhibition of multidrug efflux, blocks Candida albicans morphogenesis, and is effluxed via Yor1 and circuitry controlled by Zcf29. Antimicrob. Agents Chemother. 2016, 60, 7468–7480. [Google Scholar] [CrossRef]
- Mishra, N.N.; Ali, S.; Shukla, P.K. Arachidonic acid affects biofilm formation and PGE2 level in Candida albicans and non-albicans species in presence of subinhibitory concentration of fluconazole and terbinafine. Braz. J. Infect. Dis. 2014, 18, 287–293. [Google Scholar] [CrossRef]
- Fukuda, T.; Arai, M.; Yamaguchi, Y.; Masuma, R.; Tomoda, H.; Omura, S. New beauvericins, potentiators of antifungal miconazole activity, Produced by Beauveria sp. FKI-1366. I. Taxonomy, fermentation, isolation and biological properties. J. Antibiot. 2004, 57, 110–116. [Google Scholar] [CrossRef]
- Ells, R.; Kock, J.L.F.; Van Wyk, P.W.J.; Botes, P.J.; Pohl, C.H. Arachidonic acid increases antifungal susceptibility of Candida albicans and Candida dubliniensis. J. Antimicrob. Chemother. 2008, 63, 124–128. [Google Scholar] [CrossRef]
- Coelho, R.A.; Joffe, L.S.; Alves, G.M.; Figueiredo-Carvalho, M.H.G.; Brito-Santos, F.; Amaral, A.C.F.; Rodrigues, M.L.; Almeida-Paes, R. A screening of the MMV Pathogen Box® reveals new potential antifungal drugs against the etiologic agents of chromoblastomycosis. PLoS ONE 2020, 15, e0229630. [Google Scholar] [CrossRef]
- Thangamani, S.; Maland, M.; Mohammad, H.; Pascuzzi, P.E.; Avramova, L.; Koehler, C.M.; Hazbun, T.R.; Seleem, M.N. Repurposing approach identifies auranofin with broad spectrum antifungal activity that targets Mia40-Erv1 pathway. Front. Cell Infect. Microbiol. 2017, 7. [Google Scholar] [CrossRef]
- Wiederhold, N.P.; Patterson, T.F.; Srinivasan, A.; Chaturvedi, A.K.; Fothergill, A.W.; Wormley, F.L.; Ramasubramanian, A.K.; Lopez-Ribot, J.L. Repurposing auranofin as an antifungal: In vitro activity against a variety of medically important fungi. Virulence 2017, 8, 138–142. [Google Scholar] [CrossRef] [PubMed]
- Yousfi, H.; Cassagne, C.; Ranque, S.; Rolain, J.-M.; Bittar, F. Repurposing of ribavirin as an adjunct therapy against invasive Candida strains in an In Vitro study. Antimicrob. Agents Chemother. 2019, 63. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Yan, H.; Lu, M.; Wang, D.; Sun, S. Antifungal activity of ribavirin used alone or in combination with fluconazole against Candida albicans is mediated by reduced virulence. Int. J. Antimicrob. Agents 2020, 55, 105804. [Google Scholar] [CrossRef] [PubMed]
- Yousfi, H.; Ranque, S.; Cassagne, C.; Rolain, J.-M.; Bittar, F. Identification of repositionable drugs with novel antimycotic activity by screening the Prestwick Chemical Library against emerging invasive moulds. J. Glob. Antimicrob. Resist. 2020, 21, 314–317. [Google Scholar] [CrossRef]
- Xu, Y.; He, Y.; Zhou, L.; Gao, C.; Sun, S.; Wang, X.; Pang, G. Effects of contact lens solution disinfectants against filamentous fungi. Optom. Vis. Sci. 2014, 91, 1440–1445. [Google Scholar] [CrossRef]
- Venturini, T.P.; Rossato, L.; Chassot, F.; Keller, J.T.; Piasentin, F.B.; Santurio, J.M.; Alves, S.H. In vitro synergistic combinations of pentamidine, polymyxin B, tigecycline and tobramycin with antifungal agents against Fusarium spp. J. Med. Microbiol. 2016, 65, 770–774. [Google Scholar] [CrossRef]
- Santos, V.A.; Viera, P.V.; Oliveira, A.M.; Zanin, M.H.; Borsatti, M.A. Antifungal effect of electrospun nanofibers containing cetylpyridinium chloride against Candida albicans. Braz. Oral. Res. 2014, 28. [Google Scholar] [CrossRef]
- Ortiz, S.C.; Huang, M.; Hull, C.M. Spore germination as a target for antifungal therapeutics. Antimicrob. Agents Chemother. 2019, 63. [Google Scholar] [CrossRef]
- Sun, W.; Park, Y.-D.; Sugui, J.A.; Fothergill, A.; Southall, N.; Shinn, P.; McKew, J.C.; Kwon-Chung, K.J.; Zheng, W.; Williamson, P.R. Rapid identification of antifungal compounds against Exserohilum rostratum using high throughput drug repurposing screens. PLoS ONE 2013, 8, e70506, Erratum in 2013, 8, doi:10.1371/annotation/df5a5a46-cf30-4842-bf11-b6cee36c1f9b. [Google Scholar] [CrossRef]
- Schwarz, P.; Schwarz, P.V.; Felske-Zech, H.; Dannaoui, E. In vitro interactions between isavuconazole and tacrolimus, cyclosporin A or sirolimus against Mucorales. J. Antimicrob. Chemother. 2019, 74, 1921–1927. [Google Scholar] [CrossRef]
- Schwarz, P.; Dannaoui, E. In vitro interaction between isavuconazole and tacrolimus, cyclosporin A, or sirolimus against Aspergillus species. J. Fungi 2020, 6, 103. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Chan, K.L.; Cheng, L.W.; Tell, L.A.; Byrne, B.A.; Clothier, K.; Land, K.M. High efficiency drug repurposing design for new antifungal agents. Meth. Protoc. 2019, 2, 31. [Google Scholar] [CrossRef] [PubMed]
- Clark, A.; Hemmelgarn, T.; Danziger-Isakov, L.; Teusink, A. Intravenous pentamidine for Pneumocystis carinii/jiroveci pneumonia prophylaxis in pediatric transplant patients. Pediatr. Transplant. 2015, 19, 326–331. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Zhang, Z.-W.; Luo, Y.; Liang, N.; Pi, X.-X.; Fan, Y.-M. Molecular epidemiology, in vitro susceptibility and exoenzyme screening of Malassezia clinical isolates. J. Med. Microbiol. 2020, 69, 436–442. [Google Scholar] [CrossRef]
- Nixon, G.L.; McEntee, L.; Johnson, A.; Farrington, N.; Whalley, S.; Livermore, J.; Natal, C.; Washbourn, G.; Bibby, J.; Berry, N.; et al. Repurposing and reformulation of the antiparasitic agent flubendazole for treatment of cryptococcal meningoencephalitis, a neglected fungal disease. Antimicrob. Agents Chemother. 2018, 62, e01909–e01917. [Google Scholar] [CrossRef] [PubMed]
- Pandey, N.; Tripathi, M.; Gupta, M.K.; Tilak, R. Overexpression of efflux pump transporter genes and mutations in ERG11 pave the way to fluconazole resistance in Candida tropicalis: A study from a North India region. J. Glob. Antimicrob. Resist. 2020, 22, 374–378. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.-D.; Sun, W.; Salas, A.; Antia, A.; Carvajal, C.; Wang, A.; Xu, X.; Meng, Z.; Zhou, M.; Tawa, G.J.; et al. Identification of multiple cryptococcal fungicidal drug targets by combined gene dosing and drug affinity responsive target stability screening. mBio 2016, 7. [Google Scholar] [CrossRef]
- Truong, M.; Monahan, L.G.; Carter, D.A.; Charles, I.G. Repurposing drugs to fast-track therapeutic agents for the treatment of cryptococcosis. PeerJ 2018, 6, e4761. [Google Scholar] [CrossRef]
- Afeltra, J.; Vitale, R.G.; Mouton, J.W.; Verweij, P.E. Potent synergistic in vitro interaction between nonantimicrobial membrane-active compounds and itraconazole against clinical isolates of Aspergillus fumigatus resistant to itraconazole. Antimicrob. Agents Chemother. 2004, 48, 1335–1343. [Google Scholar] [CrossRef]
- Alnajjar, L.M.; Bulatova, N.R.; Darwish, R.M. Evaluation of four calcium channel blockers as fluconazole resistance inhibitors in Candida glabrata. J. Glob. Antimicrob. Resist. 2018, 14, 185–189. [Google Scholar] [CrossRef]
- Eldesouky, H.E.; Mayhoub, A.; Hazbun, T.R.; Seleem, M.N. Reversal of azole resistance in Candida albicans by sulfa antibacterial drugs. Antimicrob. Agents Chemother. 2018, 62, e00701–e00717. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Yue, L.; Gu, W.; Li, X.; Zhang, L.; Sun, S. Synergistic effect of fluconazole and calcium channel blockers against resistant Candida albicans. PLoS ONE 2016, 11, e0150859. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, A.G.; Araujo, R.; Pina-Vaz, C. Interaction of local anaesthetics with other antifungal agents against pathogenic Aspergillus. Int. J. Antimicrob. Agents 2006, 27, 339–343. [Google Scholar] [CrossRef] [PubMed]
- Brilhante, R.S.N.; Caetano, E.P.; Lima, R.A.C.; Castelo Branco, D.S.C.M.; Serpa, R.; Oliveira, J.S.; Monteiro, A.J.; Rocha, M.F.G.; Cordeiro, R.A.; Sidrim, J.J.C. In vitro antifungal activity of miltefosine and levamisole: Their impact on ergosterol biosynthesis and cell permeability of dimorphic fungi. J. Appl. Microbiol. 2015, 119, 962–969. [Google Scholar] [CrossRef]
- Brilhante, R.S.N.; Silva, M.L.Q.D.; Pereira, V.S.; de Oliveira, J.S.; Maciel, J.M.; Silva, I.N.G.D.; Garcia, L.G.S.; Guedes, G.M.D.M.; Cordeiro, R.D.A.; Pereira-Neto, W.D.A.; et al. Potassium iodide and miltefosine inhibit biofilms of Sporothrix schenckii species complex in yeast and filamentous forms. Med. Mycol. 2018, 57, 764–772. [Google Scholar] [CrossRef] [PubMed]
- Spadari, C.d.C.; Vila, T.; Rozental, S.; Ishida, K. Miltefosine has a postantifungal effect and induces apoptosis in Cryptococcus yeasts. Antimicrob. Agents Chemother. 2018, 62, e00312–e00318. [Google Scholar] [CrossRef]
- Wall, G.; Herrera, N.; Lopez-Ribot, J.L. Repositionable compounds with antifungal activity against multidrug resistant Candida auris identified in the medicines for malaria venture’s pathogen box. J. Fungi 2019, 5, 92. [Google Scholar] [CrossRef]
- Wu, Y.; Grossman, N.; Totten, M.; Memon, W.; Fitzgerald, A.; Ying, C.; Zhang, S.X. Antifungal susceptibility profiles and drug resistance mechanisms of clinical Lomentospora prolificans isolates. Antimicrob. Agents Chemother. 2020, 64, e00318–e00320. [Google Scholar] [CrossRef]
- Bulatova, N.R.; Darwish, R.M. Effect of chemosensitizers on minimum inhibitory concentrations of fluconazole in Candida albicans. Med. Prince Pract. 2008, 17, 117–121. [Google Scholar] [CrossRef]
- Harrison, T.S.; Griffin, G.E.; Levitz, S.M. Conditional lethality of the diprotic weak bases chloroquine and quinacrine against Cryptococcus neoformans. J. Infect. Dis. 2000, 182, 283–289. [Google Scholar] [CrossRef]
- Kulkarny, V.V.; Chavez-Dozal, A.; Rane, H.S.; Jahng, M.; Bernardo, S.M.; Parra, K.J.; Lee, S.A. Quinacrine inhibits Candida albicans growth and filamentation at neutral pH. Antimicrob. Agents Chemother. 2014, 58, 7501–7509. [Google Scholar] [CrossRef] [PubMed]
- Siles, S.A.; Srinivasan, A.; Pierce, C.G.; Lopez-Ribot, J.L.; Ramasubramanian, A.K. High-throughput screening of a collection of known pharmacologically active small compounds for identification of Candida albicans biofilm inhibitors. Antimicrob. Agents Chemother. 2013, 57, 3681–3687. [Google Scholar] [CrossRef] [PubMed]
- Vallières, C.; Raulo, R.; Dickinson, M.; Avery, S.V. Novel combinations of agents targeting translation that synergistically inhibit fungal pathogens. Front. Microbiol. 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- Zani, C.; Bisceglie, F.; Restivo, F.M.; Feretti, D.; Pioli, M.; Degola, F.; Montalbano, S.; Galati, S.; Pelosi, G.; Viola, G.V.C.; et al. A battery of assays as an integrated approach to evaluate fungal and mycotoxin inhibition properties and cytotoxic/genotoxic side-effects for the prioritization in the screening of thiosemicarbazone derivatives. Food Chem. Toxicol. 2017, 105, 498–505. [Google Scholar] [CrossRef]
- Sun, N.; Li, D.; Zhang, Y.; Killeen, K.; Groutas, W.; Calderone, R. Repurposing an inhibitor of ribosomal biogenesis with broad anti-fungal activity. Sci. Rep. 2017, 7, 17014. [Google Scholar] [CrossRef]
- Rogolino, D.; Gatti, A.; Carcelli, M.; Pelosi, G.; Bisceglie, F.; Restivo, F.M.; Degola, F.; Buschini, A.; Montalbano, S.; Feretti, D.; et al. Thiosemicarbazone scaffold for the design of antifungal and antiaflatoxigenic agents: Evaluation of ligands and related copper complexes. Sci. Rep. 2017, 7, 11214. [Google Scholar] [CrossRef]
- Degola, F.; Morcia, C.; Bisceglie, F.; Mussi, F.; Tumino, G.; Ghizzoni, R.; Pelosi, G.; Terzi, V.; Buschini, A.; Restivo, F.M.; et al. In vitro evaluation of the activity of thiosemicarbazone derivatives against mycotoxigenic fungi affecting cereals. Int. J. Food Microbiol. 2015, 200, 104–111. [Google Scholar] [CrossRef]
- De Sá, N.P.; Cisalpino, P.S.; Bertollo, C.M.; Santos, P.C.; Rosa, C.A.; de Souza, D.D.G.; Barbeira, P.J.S.; Alves, T.M.D.A.; Zani, C.L.; Johann, S. Thiosemicarbazone of lapachol acts on cell membrane in Paracoccidioides brasiliensis. Med. Mycol. 2018, 57, 332–339. [Google Scholar] [CrossRef]
- Schmidt, S.; Hogardt, M.; Demir, A.; Röger, F.; Lehrnbecher, T. Immunosuppressive compounds affect the fungal growth and viability of defined Aspergillus species. Pathogens 2019, 8, 273. [Google Scholar] [CrossRef]
- Noto, T.; Sawada, M.; Ando, K.; Koyama, K. Some biological properties of mycophenolic acid. J. Antibiot. 1969, 22, 165–169. [Google Scholar] [CrossRef][Green Version]
- Kim, K.; Zilbermintz, L.; Martchenko, M. Repurposing FDA approved drugs against the human fungal pathogen, Candida albicans. Ann. Clin. Microbiol. Antimicrob. 2015, 14, 32. [Google Scholar] [CrossRef] [PubMed]
- Bartoli, J.; Montalbano, S.; Spadola, G.; Rogolino, D.; Pelosi, G.; Bisceglie, F.; Restivo, F.M.; Degola, F.; Serra, O.; Buschini, A.; et al. Antiaflatoxigenic thiosemicarbazones as crop-protective agents: A cytotoxic and genotoxic study. J. Agric. Food Chem. 2019, 67, 10947–10953. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, D.; Burkard, L.; Panepinto, J.C. Inhibition of nucleotide biosynthesis potentiates the antifungal activity of amphotericin B. PLoS ONE 2014, 9, e87246. [Google Scholar] [CrossRef] [PubMed]
- Zanette, R.A.; Jesus, F.P.K.; Pilotto, M.B.; Weiblen, C.; Pötter, L.; Ferreiro, L.; Alves, S.H.; Santurio, J.M. Micafungin alone and in combination therapy with deferasirox against Pythium insidiosum. J. Mycol. Med. 2015, 25, 91–94. [Google Scholar] [CrossRef] [PubMed]
- Puri, S.; Kumar, R.; Rojas, I.G.; Salvatori, O.; Edgerton, M. Iron chelator deferasirox reduces Candida albicans invasion of oral epithelial cells and infection levels in murine oropharyngeal Candidiasis. Antimicrob. Agents Chemother. 2019, 63, e02152-18. [Google Scholar] [CrossRef] [PubMed]
- Magalhães, T.F.F.; Costa, M.C.; Holanda, R.A.; Ferreira, G.F.; Carvalho, V.S.D.; Freitas, G.J.C.; Ribeiro, N.Q.; Emídio, E.C.P.; Carmo, P.H.F.; de Brito, C.B.; et al. N-acetylcysteine reduces amphotericin B deoxycholate nephrotoxicity and improves the outcome of murine cryptococcosis. Med. Mycol. 2020, 58, 835–844. [Google Scholar] [CrossRef]
- Lewis, R.E.; Albert, N.D.; Kontoyiannis, D.P. Comparative pharmacodynamics of posaconazole in neutropenic murine models of invasive pulmonary aspergillosis and mucormycosis. Antimicrob. Agents Chemother. 2014, 58, 6767–6772. [Google Scholar] [CrossRef]
- Homa, M.; Galgóczy, L.; Tóth, E.; Tóth, L.; Papp, T.; Chandrasekaran, M.; Kadaikunnan, S.; Alharbi, N.S.; Vágvölgyi, C. In vitro antifungal activity of antipsychotic drugs and their combinations with conventional antifungals against Scedosporium and Pseudallescheria isolates. Med. Mycol. 2015, 53, 890–895. [Google Scholar] [CrossRef]
- You, Z.; Ran, X.; Dai, Y.; Ran, Y. Clioquinol, an alternative antimicrobial agent against common pathogenic microbe. J. Mycol. Med. 2018, 28, 492–501. [Google Scholar] [CrossRef]
- Thangamani, S.; Eldesouky, H.E.; Mohammad, H.; Pascuzzi, P.E.; Avramova, L.; Hazbun, T.R.; Seleem, M.N. Ebselen exerts antifungal activity by regulating glutathione (GSH) and reactive oxygen species (ROS) production in fungal cells. Biochim. Biophys. Acta Gen. Subj. 2017, 1861, 3002–3010. [Google Scholar] [CrossRef] [PubMed]
- Su, S.; Shi, X.; Xu, W.; Li, Y.; Chen, X.; Jia, S.; Sun, S. Antifungal activity and potential mechanism of panobinostat in combination with fluconazole against Candida albicans. Front. Microbiol. 2020, 11. [Google Scholar] [CrossRef] [PubMed]
- Leonardelli, F.; Macedo, D.; Dudiuk, C.; Theill, L.; Cabeza, M.S.; Gamarra, S.; Garcia-Effron, G. In Vitro activity of combinations of zinc chelators with amphotericin b and posaconazole against six mucorales species. Antimicrob. Agents Chemother. 2019, 63. [Google Scholar] [CrossRef]
- Jaromin, A.; Zarnowski, R.; Piętka-Ottlik, M.; Andes, D.R.; Gubernator, J. Topical delivery of ebselen encapsulated in biopolymeric nanocapsules: Drug repurposing enhanced antifungal activity. Nanomedicine 2018, 13, 1139–1155. [Google Scholar] [CrossRef]
- Ben-Gigi, G.; Polacheck, I.; Eilam, Y. In vitro synergistic activity of ketoconazole with trifluoperazine and with chlorpromazine against medically important yeasts. Chemotherapy 1988, 34, 96–100. [Google Scholar] [CrossRef]
- Butts, A.; Palmer, G.E.; Rogers, P.D. Antifungal adjuvants: Preserving and extending the antifungal arsenal. Virulence 2017, 8, 198–210. [Google Scholar] [CrossRef] [PubMed]
- Jung, E.H.; Meyers, D.J.; Bosch, J.; Casadevall, A. Novel antifungal compounds discovered in medicines for malaria venture’s malaria box. mSphere 2018, 3. [Google Scholar] [CrossRef] [PubMed]
- Montoya, M.C.; DiDone, L.; Heier, R.F.; Meyers, M.J.; Krysan, D.J. Antifungal Phenothiazines: Optimization, characterization of mechanism, and modulation of neuroreceptor activity. ACS Infect. Dis. 2018, 4, 499–507. [Google Scholar] [CrossRef] [PubMed]
- Siavoshi, F.; Tavakolian, A.; Foroumadi, A.; Hosseini, N.M.; Massarrat, S.; Pedramnia, S.; Saniee, P. Comparison of the effect of non-antifungal and antifungal agents on Candida isolates from the gastrointestinal tract. Arch. Iran. Med. 2012, 15, 27–31. [Google Scholar]
- Singh, S.; Fatima, Z.; Ahmad, K.; Hameed, S. Repurposing of respiratory drug theophylline against Candida albicans: Mechanistic insights unveil alterations in membrane properties and metabolic fitness. J. Appl. Microbiol. 2020. [Google Scholar] [CrossRef]
- Rajasekharan, S.K.; Lee, J.-H.; Lee, J. Aripiprazole repurposed as an inhibitor of biofilm formation and sterol biosynthesis in multidrug-resistant Candida albicans. Int. J. Antimicrob. Agents 2019, 54, 518–523. [Google Scholar] [CrossRef]
- Pic, E.; Burgain, A.; Sellam, A. Repurposing the anthelminthic salicylanilide oxyclozanide against susceptible and clinical resistant Candida albicans strains. Med. Mycol. 2018, 57, 387–390. [Google Scholar] [CrossRef] [PubMed]
- Nile, C.; Falleni, M.; Cirasola, D.; Alghamdi, A.; Anderson, O.F.; Delaney, C.; Ramage, G.; Ottaviano, E.; Tosi, D.; Bulfamante, G.; et al. Repurposing pilocarpine hydrochloride for treatment of Candida albicans infections. mSphere 2019, 4. [Google Scholar] [CrossRef] [PubMed]
- Montoya, M.C.; Beattie, S.; Alden, K.M.; Krysan, D.J. Derivatives of the antimalarial drug mefloquine are broad-spectrum antifungal molecules with activity against drug-resistant clinical isolates. Antimicrob. Agents Chemother. 2020, 64. [Google Scholar] [CrossRef] [PubMed]
- Fonseca, D.; Leal-Pinto, S.M.; Roa-Cordero, M.V.; Vargas, J.D.; Moreno-Moreno, E.M.; Macías, M.A.; Suescun, L.; Muñoz-Castro, Á.; Hurtado, J.J. Inhibition of C. albicans dimorphic switch by Cobalt(II) complexes with ligands derived from pyrazoles and dinitrobenzoate: Synthesis, characterization and biological activity. Int. J. Mol. Sci. 2019, 20, 3237. [Google Scholar] [CrossRef]
- Joffe, L.S.; Schneider, R.; Lopes, W.; Azevedo, R.; Staats, C.C.; Kmetzsch, L.; Schrank, A.; Del Poeta, M.; Vainstein, M.H.; Rodrigues, M.L. The anti-helminthic compound mebendazole has multiple antifungal effects against Cryptococcus neoformans. Front. Microbiol. 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Liang, S.; Zhang, Y.; Yang, D.; Wang, R. Development of anti-fungal pesticides from protein kinase inhibitor-based anticancer agents. Eur. J. Med. Chem. 2018, 148, 349–358. [Google Scholar] [CrossRef]
- Ngo, H.X.; Shrestha, S.K.; Garneau-Tsodikova, S. Identification of ebsulfur analogues with broad-spectrum antifungal activity. ChemMedChem 2016, 11, 1507–1516. [Google Scholar] [CrossRef]
- Veri, A.; Cowen, L.E. Progress and prospects for targeting Hsp90 to treat fungal infections. Parasitology 2014, 141, 1127–1137. [Google Scholar] [CrossRef]
- She, P.; Liu, Y.; Wang, Y.; Tan, F.; Luo, Z.; Wu, Y. Antibiofilm efficacy of the gold compound auranofin on dual species biofilms of Staphylococcus aureus and Candida sp. J. Appl. Microbiol. 2020, 128, 88–101. [Google Scholar] [CrossRef]
- Nobile, C.J.; Ennis, C.L.; Hartooni, N.; Johnson, A.D.; Lohse, M.B. A selective serotonin reuptake inhibitor, a proton pump inhibitor, and two calcium channel blockers inhibit Candida albicans biofilms. Microorganisms 2020, 8, 756. [Google Scholar] [CrossRef]
- Eldesouky, H.E.; Salama, E.A.; Lanman, N.A.; Hazbun, T.R.; Seleem, M.N. Potent synergistic interactions between lopinavir and azole antifungal drugs against emerging multidrug-resistant Candida auris. Antimicrob. Agents Chemother. 2020. [Google Scholar] [CrossRef] [PubMed]
- Chavez-Dozal, A.A.; Lown, L.; Jahng, M.; Walraven, C.J.; Lee, S.A. In vitro analysis of finasteride activity against Candida albicans urinary biofilm formation and filamentation. Antimicrob. Agents Chemother. 2014, 58, 5855–5862. [Google Scholar] [CrossRef] [PubMed]
- Bernardo, S.M.; Allen, C.P.; Waller, A.; Young, S.M.; Oprea, T.; Sklar, L.A.; Lee, S.A. An automated high-throughput cell-based multiplexed flow cytometry assay to identify novel compounds to target Candida albicans virulence-related proteins. PLoS ONE 2014, 9, e110354. [Google Scholar] [CrossRef] [PubMed]
- Meng, L.-N.; Liu, J.-Y.; Wang, Y.-T.; Ni, S.-S.; Xiang, M.-J. The discovery of potential phosphopantetheinyl transferase Ppt2 inhibitors against drug-resistant Candida albicans. Braz. J. Microbiol. 2020. [Google Scholar] [CrossRef]
- Mei, Y.; Jiang, T.; Zou, Y.; Wang, Y.; Zhou, J.; Li, J.; Liu, L.; Tan, J.; Wei, L.; Li, J.; et al. FDA approved drug library screening identifies robenidine as a repositionable antifungal. Front. Microbiol. 2020, 11. [Google Scholar] [CrossRef]
- Li, Z.; Fu, B.; Green, C.M.; Liu, B.; Zhang, J.; Lang, Y.; Chaturvedi, S.; Belfort, M.; Liao, G.; Li, H. Cisplatin protects mice from challenge of Cryptococcus neoformans by targeting the Prp8 intein. Emerg. Microbes Infect. 2019, 8, 895–908. [Google Scholar] [CrossRef]
- Garcia, C.; Burgain, A.; Chaillot, J.; Pic, É.; Khemiri, I.; Sellam, A. A phenotypic small-molecule screen identifies halogenated salicylanilides as inhibitors of fungal morphogenesis, biofilm formation and host cell invasion. Sci. Rep. 2018, 8, 11559. [Google Scholar] [CrossRef]
- Dehdashti, S.J.; Abbott, J.; Nguyen, D.T.; McKew, J.C.; Williamson, P.R.; Zheng, W. A high-throughput screening assay for assessing the viability of Cryptococcus neoformans under nutrient starvation conditions. Anal. Bioanl. Chem. 2013, 405, 6823–6829. [Google Scholar] [CrossRef][Green Version]
- Clinical and Laboratory Standards Institute (CLSI). Reference Method for Broth Dilution Antifungal Susceptibility Testing of Yeasts: Approved Standard, 2nd ed.; CLSI document M27-A3; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2008. [Google Scholar]
- The European Committee on Antimicrobial Susceptibility Testing. EUCAST Method for Susceptibility Testing of Yeasts (v 7.3.2 Valid from 22 April 2020). Available online: https://www.eucast.org/astoffungi/methodsinantifungalsusceptibilitytesting/susceptibility_testing_of_yeasts/ (accessed on 21 August 2020).
- Law, G.L.; Tisoncik-Go, J.; Korth, M.J.; Katze, M.G. Drug repurposing: A better approach for infectious disease drug discovery? Curr. Opin. Immunol. 2013, 25, 588–592. [Google Scholar] [CrossRef]
- Abadio, A.K.R.; Kioshima, E.S.; Teixeira, M.M.; Martins, N.F.; Maigret, B.; Felipe, M.S.S. Comparative genomics allowed the identification of drug targets against human fungal pathogens. BMC Genom. 2011, 12, 75. [Google Scholar] [CrossRef]
- Gowri, M.; Sofi Beaula, W.; Biswal, J.; Dhamodharan, P.; Saiharish, R.; Rohan prasad, S.; Pitani, R.; Kandaswamy, D.; Raghunathan, R.; Jeyakanthan, J.; et al. β-lactam substituted polycyclic fused pyrrolidine/pyrrolizidine derivatives eradicate C. albicans in an ex vivo human dentinal tubule model by inhibiting sterol 14-α demethylase and cAMP pathway. Biochim. Biophys. Acta 2016, 1860, 636–647. [Google Scholar] [CrossRef] [PubMed]
- OpenEye Scientific Software OMEGA; v.2.5.1; OpenEye Scientific Software: Santa Fe, NM, USA, 2019.
- OpenEye Scientific Software QUACPAC; v.1.6.3; OpenEye Scientific Software: Santa Fe, NM, USA, 2013.
- OpenEye Scientific Software OEDocking; v3.2.0; OpenEye Scientific Software: Santa Fe, NM, USA, 2017.
- Anandakrishnan, R.; Aguilar, B.; Onufriev, A.V. H++ 3.0: Automating pK prediction and the preparation of biomolecular structures for atomistic molecular modeling and simulations. Nucleic Acids Res. 2012, 40, W537–W541. [Google Scholar] [CrossRef] [PubMed]
- Biasini, M.; Bienert, S.; Waterhouse, A.; Arnold, K.; Studer, G.; Schmidt, T.; Kiefer, F.; Cassarino, T.G.; Bertoni, M.; Bordoli, L.; et al. SWISS-MODEL: Modelling protein tertiary and quaternary structure using evolutionary information. Nucleic Acids Res. 2014, 42, W252–W258. [Google Scholar] [CrossRef] [PubMed]
- Bordoli, L.; Kiefer, F.; Arnold, K.; Benkert, P.; Battey, J.; Schwede, T. Protein structure homology modeling using SWISS-MODEL workspace. Nat. Protoc. 2009, 4, 1–13. [Google Scholar] [CrossRef]
- Broad Institute. Fungal Genomics. Available online: https://www.broadinstitute.org/fungal-genome-initiative (accessed on 21 August 2020).
- Chen, V.B.; Arendall, W.B., III; Headd, J.J.; Keedy, D.A.; Immormino, R.M.; Kapral, G.J.; Murray, L.W.; Richardson, J.S.; Richardson, D.C. MolProbity: All-atom structure validation for macromolecular crystallography. Acta Crystallograph. Sect. D 2010, 66, 12–21. [Google Scholar] [CrossRef]
- De Paula e Silva, A.C.A.; Oliveira, H.C.; Silva, J.F.; Sangalli-Leite, F.; Scorzoni, L.; Fusco-Almeida, A.M.; Mendes-Giannini, M.J.S. Microplate alamarBlue assay for Paracoccidioides susceptibility testing. J. Clin. Microbiol. 2013, 51, 1250–1252. [Google Scholar] [CrossRef] [PubMed]
- Law, V.; Knox, C.; Djoumbou, Y.; Jewison, T.; Guo, A.C.; Liu, Y.; Maciejewski, A.; Arndt, D.; Wilson, M.; Neveu, V.; et al. DrugBank 4.0: Shedding new light on drug metabolism. Nucleic Acids Res. 2013, 42, D1091–D1097. [Google Scholar] [CrossRef]
- Li, Y.H.; Yu, C.Y.; Li, X.X.; Zhang, P.; Tang, J.; Yang, Q.; Fu, T.; Zhang, X.; Cui, X.; Tu, G.; et al. Therapeutic target database update 2018: Enriched resource for facilitating bench-to-clinic research of targeted therapeutics. Nucleic Acids Res. 2017, 46, D1121–D1127. [Google Scholar] [CrossRef]
- McGann, M. FRED and HYBRID docking performance on standardized datasets. J. Comput.-Aided Mol. Des. 2012, 26, 897–906. [Google Scholar] [CrossRef]
- Rodrigues, J.P.G.L.M.; Levitt, M.; Chopra, G. KoBaMIN: A knowledge-based minimization web server for protein structure refinement. Nucleic Acids Res. 2012, 40, W323–W328. [Google Scholar] [CrossRef]
- Rose, P.W.; Prlić, A.; Bi, C.; Bluhm, W.F.; Christie, C.H.; Dutta, S.; Green, R.K.; Goodsell, D.S.; Westbrook, J.D.; Woo, J.; et al. The RCSB Protein Data Bank: Views of structural biology for basic and applied research and education. Nucleic Acids Res. 2014, 43, D345–D356. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Coleman-Derr, D.; Chen, G.; Gu, Y.Q. OrthoVenn: A web server for genome wide comparison and annotation of orthologous clusters across multiple species. Nucleic Acids Res. 2015, 43, W78–W84. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Lin, Y. DEG 5.0, a database of essential genes in both prokaryotes and eukaryotes. Nucleic Acids Res. 2008, 37, D455–D458. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Ou, H.Y.; Zhang, C.T. DEG: A database of essential genes. Nucleic Acids Res. 2004, 32, D271–D272. [Google Scholar] [CrossRef] [PubMed]
- European Bioinformatics Institute. CLUSTALW. Available online: http://www.ebi.ac.uk/Tools/msa/ (accessed on 21 August 2020).
- Hess, B.; Kutzner, C.; van der Spoel, D.; Lindahl, E. GROMACS 4: Algorithms for highly efficient, load-balanced, and scalable molecular simulation. J. Chem. Theory Comput. 2008, 4, 435–447. [Google Scholar] [CrossRef]
- National Center for Biotechnology Information. BLAST. Available online: https://www.ncbi.nlm.nih.gov/BLAST/ (accessed on 21 August 2020).
- Amiri, S.; Sansom, M.S.P.; Biggin, P.C. Molecular dynamics studies of AChBP with nicotine and carbamylcholine: The role of water in the binding pocket. Protein Eng. Des. Sel. 2007, 20, 353–359. [Google Scholar] [CrossRef] [PubMed]
- Clancy, C.J.; Nguyen, M.H. Comparison of a photometric method with standardized methods of antifungal susceptibility testing of yeasts. J. Clin. Microbiol. 1997, 35, 2878–2882. [Google Scholar] [CrossRef]
- Humphrey, W.; Dalke, A.; Schulten, K. VMD: Visual molecular dynamics. J. Mol. Graph. 1996, 14, 33–38. [Google Scholar] [CrossRef]
- Molecular Operating Environment (MOE); v.2013.08; Chemical Computing Group, Inc.: Montreal, QC, Canada, 2014.
- Oostenbrink, C.; Villa, A.; Mark, A.E.; Van Gunsteren, W.F. A biomolecular force field based on the free enthalpy of hydration and solvation: The GROMOS force-field parameter sets 53A5 and 53A6. J. Comput. Chem. 2004, 25, 1656–1676. [Google Scholar] [CrossRef]
- Pronk, S.; Páll, S.; Schulz, R.; Larsson, P.; Bjelkmar, P.; Apostolov, R.; Shirts, M.R.; Smith, J.C.; Kasson, P.M.; van der Spoel, D.; et al. GROMACS 4.5: A high-throughput and highly parallel open source molecular simulation toolkit. Bioinformatics 2013, 29, 845–854. [Google Scholar] [CrossRef]
- Rarey, M.; Kramer, B.; Lengauer, T.; Klebe, G. A fast flexible docking method using an incremental construction algorithm. J. Mol. Biol. 1996, 261, 470–489. [Google Scholar] [CrossRef] [PubMed]
- Schüttelkopf, A.W.; van Aalten, D.M. PRODRG: A tool for high-throughput crystallography of protein-ligand complexes. Acta Crystallogr. D Biol. Crystallogr. 2004, 60, 1355–1363. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Dommert, F.; Holm, C. Optimizing working parameters of the smooth particle mesh Ewald algorithm in terms of accuracy and efficiency. J. Chem. Phys. 2010, 133, 034117. [Google Scholar] [CrossRef] [PubMed]
- Dallakyan, S.; Olson, A.J. Small-molecule library screening by docking with PyRx. Methods Mol. Biol. 2015, 1263, 243–250. [Google Scholar] [CrossRef]
- Morris, G.M.; Huey, R.; Lindstrom, W.; Sanner, M.F.; Belew, R.K.; Goodsell, D.S.; Olson, A.J. AutoDock4 and AutoDockTools4: Automated docking with selective receptor flexibility. J. Comput. Chem. 2009, 30, 2785–2791. [Google Scholar] [CrossRef]
- Gasteiger, J.; Rudolph, C.; Sadowski, J. Automatic generation of 3D-atomic coordinates for organic molecules. Tetrahedron. Comput. Methodol. 1990, 3, 537–547. [Google Scholar] [CrossRef]
- Jones, G.; Willett, P.; Glen, R.C.; Leach, A.R.; Taylor, R. Development and validation of a genetic algorithm for flexible docking. J. Mol. Biol. 1997, 267, 727–748. [Google Scholar] [CrossRef]
- Schäfer, T.; Kriege, N.; Humbeck, L.; Klein, K.; Koch, O.; Mutzel, P. Scaffold hunter: A comprehensive visual analytics framework for drug discovery. J. Cheminform. 2017, 9, 28. [Google Scholar] [CrossRef]
- Wallace, A.C.; Laskowski, R.A.; Thornton, J.M. LIGPLOT: A program to generate schematic diagrams of protein-ligand interactions. Protein Eng. 1995, 8, 127–134. [Google Scholar] [CrossRef]
- Meletiadis, J.; Antachopoulos, C.; Stergiopoulou, T.; Pournaras, S.; Roilides, E.; Walsh, T.J. Differential fungicidal activities of amphotericin B and voriconazole against Aspergillus species determined by microbroth methodology. Antimicrob. Agents Chemother. 2007, 51, 3329–3337. [Google Scholar] [CrossRef]
- Odds, F.C. Synergy, antagonism, and what the chequerboard puts between them. J. Antimicrob. Chemother. 2003, 52. [Google Scholar] [CrossRef] [PubMed]
- Alastruey-Izquierdo, A.; Melhem, M.S.; Bonfietti, L.X.; Rodriguez-Tudela, J.L. Susceptiblity test for fungi: Clinical and laboratorial correlations in medical mycology. Rev. Inst. Med. Trop São Paulo 2015, 57 (Suppl. 19), 57–64. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Wang, Y.; Liu, N.; Dong, G.; Sheng, C. Tackling fungal resistance by biofilm inhibitors. J. Med. Chem. 2017, 60, 2193–2211. [Google Scholar] [CrossRef] [PubMed]
- Sherry, L.; Rajendran, R.; Lappin, D.F.; Borghi, E.; Perdoni, F.; Falleni, M.; Tosi, D.; Smith, K.; Williams, C.; Jones, B.; et al. Biofilms formed by Candida albicans bloodstream isolates display phenotypic and transcriptional heterogeneity that are associated with resistance and pathogenicity. BMC Microbiol. 2014, 14, 182. [Google Scholar] [CrossRef] [PubMed]
- Desai, J.V.; Mitchell, A.P.; Andes, D.R. Fungal biofilms, drug resistance, and recurrent infection. Cold Spring Harb. Perspect. Med. 2014, 4. [Google Scholar] [CrossRef]
- Gulati, M.; Lohse, M.B.; Ennis, C.L.; Gonzalez, R.E.; Perry, A.M.; Bapat, P.; Arevalo, A.V.; Rodriguez, D.L.; Nobile, C.J. In Vitro culturing and screening of Candida albicans biofilms. Curr. Prot. Microbiol. 2018, 50, e60. [Google Scholar] [CrossRef]
- Pierce, C.G.; Srinivasan, A.; Uppuluri, P.; Ramasubramanian, A.K.; López-Ribot, J.L. Antifungal therapy with an emphasis on biofilms. Curr. Opin. Pharmacol. 2013, 13, 726–730. [Google Scholar] [CrossRef]
- Sherry, L.; Ramage, G.; Kean, R.; Borman, A.; Johnson, E.; Richardson, M.; Rautemaa-Richardson, R. Biofilm-Forming capability of highly virulent, multidrug-resistant Candida auris. Emerg. Infect. Dis. 2017, 23, 328. [Google Scholar] [CrossRef]
- Ha, K.C.; White, T.C. Effects of azole antifungal drugs on the transition from yeast cells to hyphae in susceptible and resistant isolates of the pathogenic yeast Candida albicans. Antimicrob. Agents Chemother. 1999, 43, 763–768. [Google Scholar] [CrossRef]
- Heilman, B.J.; Tadle, A.C.; Pimentel, L.R.; Mascharak, P.K. Selective damage to hyphal form through light-induced delivery of nitric oxide to Candida albicans colonies. J. Inorg. Biochem. 2013, 123, 18–22. [Google Scholar] [CrossRef]
- Higgins, J.; Pinjon, E.; Oltean, H.N.; White, T.C.; Kelly, S.L.; Martel, C.M.; Sullivan, D.J.; Coleman, D.C.; Moran, G.P. Triclosan antagonizes fluconazole activity against Candida albicans. J. Dent. Res. 2012, 91, 65–70. [Google Scholar] [CrossRef] [PubMed]
- Van de Sande, W.W.J.; Tavakol, M.; van Vianen, W.; Bakker-Woudenberg, I.A.J.M. The effects of antifungal agents to conidial and hyphal forms of Aspergillus fumigatus. Med. Mycol. 2010, 48, 48–55. [Google Scholar] [CrossRef] [PubMed]
- Mariné, M.; Bom, V.L.P.; de Castro, P.A.; Winkelstroter, L.K.; Ramalho, L.N.; Brown, N.A.; Goldman, G.H. The development of animal infection models and antifungal efficacy assays against clinical isolates of Trichosporon asahii, T. asteroides and T. inkin. Virulence 2015, 6, 476–486. [Google Scholar] [CrossRef]
- Paulussen, C.; Boulet, G.A.V.; Cos, P.; Delputte, P.; Maes, L.J.R.M. Animal models of invasive aspergillosis for drug discovery. Drug Discov. Today 2014, 19, 1380–1386. [Google Scholar] [CrossRef] [PubMed]
- National Research Council (US) Committee to Update Science, Medicine, and Animals. Regulation of animal research. In Science, Medicine, and Animals; National Academies Press: Washington, DC, USA, 2004. Available online: https://www.ncbi.nlm.nih.gov/books/NBK24650/ (accessed on 14 November 2020).
- Arvanitis, M.; Glavis-Bloom, J.; Mylonakis, E. Invertebrate models of fungal infection. Biochim. Biophys. Acta Mol. Basis Dis. 2013, 1832, 1378–1383. [Google Scholar] [CrossRef]
- De Oliveira, H.C.; Monteiro, M.C.; Rossi, S.A.; Pemán, J.; Ruiz-Gaitán, A.; Mendes-Giannini, M.J.S.; Mellado, E.; Zaragoza, O. Identification of off-patent compounds that present antifungal activity against the emerging fungal pathogen Candida auris. Front. Cell. Infect. Microbiol. 2019, 9, 83. [Google Scholar] [CrossRef]
- Gamo, F.-J.; Sanz, L.M.; Vidal, J.; de Cozar, C.; Alvarez, E.; Lavandera, J.-L.; Vanderwall, D.E.; Green, D.V.S.; Kumar, V.; Hasan, S.; et al. Thousands of chemical starting points for antimalarial lead identification. Nature 2010, 465, 305–310. [Google Scholar] [CrossRef]
- Johnson, M.D.; Perfect, J.R. Use of antifungal combination therapy: Agents, order, and timing. Curr. Fungal Infect. Rep. 2010, 4, 87–95. [Google Scholar] [CrossRef]
- Carrillo-Muñoz, A.J.; Finquelievich, J.; Tur-Tur, C.; Eraso, E.; Jauregizar, N.; Quindós, G.; Giusiano, G. Combination antifungal therapy: A strategy for the management of invasive fungal infections. Rev. Esp. Quim. 2014, 27, 141–158. [Google Scholar]
- Kim, J.H.; Haff, R.P.; Faria, N.C.G.; Martins, M.D.L.; Chan, K.L.; Campbell, B.C. Targeting the mitochondrial respiratory chain of Cryptococcus through antifungal chemosensitization: A model for control of non-fermentative pathogens. Molecules 2013, 18, 8873–8894. [Google Scholar] [CrossRef]
- Campbell, B.C.; Chan, K.L.; Kim, J.H. Chemosensitization as a means to augment commercial antifungal agents. Front. Microbiol. 2012, 3, 79. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Chan, K.L.; Cheng, L.W. Octyl gallate as an intervention catalyst to augment antifungal efficacy of caspofungin. J—Multidiscip. Sci. J. 2018, 1, 19–28. [Google Scholar] [CrossRef]
- AHFS® Patient Medication Information™. Pioglitazone. Available online: https://medlineplus.gov/druginfo/meds/a699016.html#why (accessed on 3 September 2020).
- Ribeiro, N.Q.; Santos, A.P.N.; Emídio, E.C.P.; Costa, M.C.; Freitas, G.J.C.; Carmo, P.H.F.; Silva, M.F.; de Brito, C.B.; de Souza, D.G.; Paixão, T.A.; et al. Pioglitazone as an adjuvant of amphotericin B for the treatment of cryptococcosis. Int. J. Antimicrob. Agents 2019, 54, 301–308. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H. (Western Regional Research Center, USDA-ARS, Albany, CA, USA); Haranahalli, K. (Stony Brook University, Stony Brook, NY, USA). Personal communication, 2020.
- Saccharomyces Genome Database. Available online: www.yeastgenome.org (accessed on 3 September 2020).
- Parsons, A.B.; Brost, R.L.; Ding, H.; Li, Z.; Zhang, C.; Sheikh, B.; Brown, G.W.; Kane, P.M.; Hughes, T.R.; Boone, C. Integration of chemical-genetic and genetic interaction data links bioactive compounds to cellular target pathways. Nat. Biotechnol. 2004, 22, 62–69. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Campbell, B.C.; Yu, J.; Mahoney, N.; Chan, K.L.; Molyneux, R.J.; Bhatnagar, D.; Cleveland, T.E. Examination of fungal stress response genes using Saccharomyces cerevisiae as a model system: Targeting genes affecting aflatoxin biosynthesis by Aspergillus flavus Link. Appl. Microbiol. Biotechnol. 2005, 67, 807–815. [Google Scholar] [CrossRef]
- U.S. Food and Drug Administration. Substances Added to Food. Available online: https://www.fda.gov/food/food-additives-petitions/substances-added-food-formerly-eafus (accessed on 21 July 2020).
- Fungicide Resistance Action Committee. Available online: https://www.frac.info (accessed on 21 July 2020).
- Kim, J.H.; Chan, K.L.; Cheng, L.W. Cinnamic acid analogs as intervention catalysts for overcoming antifungal tolerance. Molecules 2017, 22, 1783. [Google Scholar]
- Ma, C.-M.; Abe, T.; Komiyama, T.; Wang, W.; Hattori, M.; Daneshtalab, M. Synthesis, anti-fungal and 1,3-β-d-glucan synthase inhibitory activities of caffeic and quinic acid derivatives. Bioorg. Med. Chem. 2010, 18, 7009–7014. [Google Scholar] [CrossRef]
- Couto, N.; Wood, J.; Barber, J. The role of glutathione reductase and related enzymes on cellular redox homoeostasis network. Free Radic. Biol. Med. 2016, 95, 27–42. [Google Scholar] [CrossRef]
- Xue, T.; Nguyen, C.K.; Romans, A.; May, G.S. A mitogen-activated protein kinase that senses nitrogen regulates conidial germination and growth in Aspergillus fumigatus. Eukaryot. Cell 2004, 3, 557–560. [Google Scholar] [CrossRef]
- Reyes, G.; Romans, A.; Nguyen, C.K.; May, G.S. Novel mitogen-activated protein kinase MpkC of Aspergillus fumigatus is required for utilization of polyalcohol sugars. Eukaryot. Cell 2006, 5, 1934–1940. [Google Scholar] [CrossRef]
- Berman, J.; Krysan, D.J. Drug resistance and tolerance in fungi. Nat. Rev. Microbiol. 2020, 18, 319–331. [Google Scholar] [CrossRef] [PubMed]
- Beardsley, J.; Halliday, C.L.; Chen, S.C.-A.; Sorrell, T.C. Responding to the emergence of antifungal drug resistance: Perspectives from the bench and the bedside. Future Microbiol. 2018, 13, 1175–1191. [Google Scholar] [CrossRef] [PubMed]
- Hernando-Amado, S.; Coque, T.M.; Baquero, F.; Martínez, J.L. Defining and combating antibiotic resistance from One Health and Global Health perspectives. Nat. Microbiol. 2019, 4, 1432–1442. [Google Scholar] [CrossRef] [PubMed]
- Fisher, M.C.; Hawkins, N.J.; Sanglard, D.; Gurr, S.J. Worldwide emergence of resistance to antifungal drugs challenges human health and food security. Science 2018, 360, 739–742. [Google Scholar] [CrossRef]
- Translational Informatics Division at University of New Mexico. DrugCentral. Available online: http://drugcentral.org (accessed on 3 September 2020).
- Clinical Trials Transformation Initiative. Aggregate Content of ClinicalTrials.gov (AACT). Available online: https://aact.ctti-clinicaltrials.org/ (accessed on 3 September 2020).

| Pathogens Steps | Paracoccidioides Species | Candida albicans | Candida, Aspergillus, and Trychophyton Species | Pythium insidiosum | Paracoccidioides Species | Candida auris |
|---|---|---|---|---|---|---|
| Mining and compilation of pathogen genome data | Broad Institute Fungal Genomics Database (https://www.broadinstitute.org/fungal-genome-initiative) | The Basic Local Alignment Search Tool (BLASTP), National Center for Biotechnology Information (http://www.ncbi.nlm.nih.gov/BLAST/); CLUSTALW, European Bioinformatics Institute (http://www.ebi.ac.uk/Tools/msa/) | MOE 2014.09 (PDB search module); Clustal Omega tool, UniProt server (http://www.ebi.ac.uk/Tools/msa/clustalo/) | UniProt database (https://www.uniprot.org); Protein Data Bank (https://www.rcsb.org); BLASTP (blast 2.2.28_ program) (https://www.ncbi.nlm.nih.gov) | - | - |
| Identification of orthologs in pathogen isolates | OrthoVenn server | - | - | - | - | - |
| Identification of homologs in the drug–target databases | DrugBank; Therapeutic targets database (TTD) | - | - | - | MDL Drug Data Report (MDDR) (BIOVIA and Thomson Reuters); DrugBank (https://www.drugbank.ca/); TargetMol provider (http://targetmol.com/) | - |
| Selection of essential targets in the Saccharomyces cerevisiae system | Database of Essential Genes (DEG) | - | - | - | - | - |
| Homology modeling | SWISS-MODEL server; Protein Data Bank (PDB); KoBaMIN server; HCC server; MolProbity | SWISS-MODEL server; Ramachandran plot, SAVES (http://services.mbi.ucla.edu/PROCHECK) | AMBER99 (with S. cerevisiae chorismate mutase; PDB entry code 4CSM) as a template | PubChem server (http://pubchem.ncbi.nlm.nih.gov/) (3D structure); PreADMET server (https://preadmet.bmdrc.kr/introduction/) (3D structure) | Homology modeling for Candida albicans thioredoxin reductase [174] | Cytochrome P450 51 (CYP51) modeling [175] |
| Ligand preparation and Molecular docking | OMEGA v.3.0.0.1 software by OpenEye Scientific [176]; QUACPAC v.1.7.0.2 software by OpenEye Scientific [177]; OEDocking suite v.3.2.0 by OpenEye Scientific [178]; FRED program with the ChemGauss4 score function in the OEDocking suite | Protein Preparation Wizard of Schrodinger’s suite 8.5 (Chimeric 1EA1); Docking between internal ligand (fluconazole) and 1EA1 in Schrodinger’s suite 8.5; GROMACS 5.0 for molecular dynamics simulations with GROMOS9643a1 force field; LIGPREP and MAESTRO (fluvastatin 3D structure); MATLAB version R2015b (plotting and calculations); Dundee Prodrug 2.5; GLIDE 5.0 | MOE 2014.09 (Chemical Computing Group Inc., Sherbooke St, Montreal, QC, Canada) software; Leadit 2.1.2 (BioSolveIT GmbH, Germany) software; Gromacs 4.5.5 (Molecular dynamics); PRODRG2 and GROMOS 53A6 force field (Topology); Particle Mesh Ewald (PME) method and linear constraint solver (LINCS); Xmgrace and VMD software | AutoDock Vina program; PyRx suite open-source software version 0.9.7; AutoDock Vina program; Discovery Studio Visualizer version 17.2.0 (Dassault Systemes Biovia Corp.) | Scaffold Hunter program (http://scaffoldhunter.sourceforge.net/) (selection of top ten best compounds from each database); GOLD software (docking simulation against Candida albicans thioredoxin reductase); CORINA (three-dimensional models); Visual Molecular Dynamics (VMD) program (http://www.ks.uiuc.edu/Research/vmd/); LigPlot program (https://www.ebi.ac.uk/thornton-srv/software/LIGPLOT/) | Protein preparation wizard (C. albicans CYP51); LigPrep module of the Schrodinger suite (sertraline); Schrodinger suite (LLC, New York, NY) (induced fit molecular docking analysis); Glide module (extra precision feature for sertraline) |
| Experimental validation in the target pathogen | Modified Clinical and Laboratory Standards Institute (CLSI) protocol | Microtiter bioassay | 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) microdilution assay | Agar- and broth-based assay | CLSI M27-A3 [171] | Microdilution (killing kinetics) |
| References | [15,176,177,178,179,180,181,182,183,184,185,186,187,188,189,190,191,192] | [18,193,194,195] | [16,196,197,198,199,200,201,202,203,204] | [17,205,206] | [20,171,174,207,208,209,210] | [21,175] |
| Compounds | Fungi | Effects | Ref. |
|---|---|---|---|
| Haloperidol or benzocyclane derivative | C. albicans, C. glabrata, C. neoformans | Inhibition of filamentation, melanin production, and biofilm formation | [78] |
| Aripiprazole | C. albicans | Inhibition of biofilm formation and hyphal filamentation | [152] |
| Alexidine dihydrochloride | C. albicans, C. auris, A. fumigatus | Antibiofilm activity | [83] |
| Mefloquine | C. albicans, C. neoformans, | Inhibition of the expression of virulence traits: filamentation in C. albicans and capsule formation/melanization in C. neoformans | [155] |
| Pentamidine, bifonazole, econazole, alexidine, cetylpyridinium chloride, otilonium bromide, benzethonium chloride, niclosamide, disulfiram, temsirolimus | C. neoformans | Inhibition of spore germination and yeast growth | [100] |
| Sulfonamide drugs | C. albicans | Inhibition of biofilm | [113] |
| Miltefosine | C. albicans, C. auris, C. dubliniensis, C. glabrata, C. krusei, C. parapsilosis, C. tropicalis, Sporothrix schenckii | Inhibition of both planktonic growth and biofilm formation; inhibition of Coccidioides posadasii filamentous phase and Histoplasma capsulatum filamentous/yeast phases | [116,117,119] |
| Mebendazole | C. neoformans | Antifungal activity against phagocytized C. neoformans: affected biofilms and reduced capsular dimensions | [157] |
| Quinacrine | C. albicans | Inhibition of biofilm and inhibition of planktonic growth (alkaline pH) and filamentation | [123] |
| Auranofin, pyrvinium pamoate, benzbromarone | C. albicans | Inhibition of biofilm formation | [124] |
| Finasteride | C. albicans | Inhibition of urinary biofilm formation and filamentation | [164] |
| Auranofin | C. albicans, Staphylococcus aureus | Inhibition of C. albicans and S. aureus (bacterium) mono- and dual biofilm formation | [161] |
| Panobinostat | C. albicans | Inhibition of biofilm, hyphal, and planktonic growth | [143] |
| Robenidine | A. fumigatus, C. albicans, C. neoformans, S. cerevisiae | Inhibition of yeast cell growth, filamentation, and biofilm formation | [167] |
| bis-Biguanide alexidine dihydrochloride | C. albicans | Antifungal and antibiofilm activity | [83] |
| Halogenated salicylanilide, niclosamide | C. albicans, C. auris | Antifilamentation and antibiofilm activities | [169] |
| Arachidonic acid | C. albicans, C. parapsilosis, C. glabrata, C. tropicalis | Antibiofilm activity | [88] |
| Aspirin, ibuprofen | C. albicans, Trichosporon asahii | Antibiofilm and antiplanktonic activity | [60,62] |
| Nortriptyline | C. utilis, C. krusei, C. glabrata | Antihyphal and antibiofilm activity | [74] |
| Quinine | C. albicans | Antifungal synergy with bicarbonate or hygromycin against biofilm | [125] |
| Drug/Compound Repurposed | Animal Model | Fungi | Effect | Reference |
|---|---|---|---|---|
| Raltegravir | BALB/c mice, male, 6 weeks old | Paracoccidioides species | Reduction of the fungal burden, decreased alterations in the lung structure of mice (1 mg/kg of raltegravir) | [20] |
| Thioridazine | Murine J774 phagocytes | C. neoformans | Decreased the intracellular burden of C. neoformans (2.7-fold at a concentration 16-fold below the MIC (2 μg/mL)) | [75] |
| Beauvericin | Specific pathogen-free female ICR (Crl: CD-1) mice | C. albicans or C. parapsilosis | Reduction in tissue damage and inflammatory cell infiltration in kidneys (0.5 mg/kg beauvericin and KET 1) | [85] |
| Beauvericin | BALB/c (inbred) mice, female, 7 weeks old | C. albicans | Beauvericin (4 mg/kg) and FLU 1 (0.5 mg/kg) combination exhibited a therapeutic benefit | [84] |
| Pentamidine | C57BL/6J, female, 8 to 10 weeks old | C. neoformans | Mice treated prophylactically with pentamidine (for 3 days prior to infection) resulted in a 2-fold-lower fungal burden than the control; minimized lung fungal burden in spore-mediated infections of mice | [100] |
| Deferasirox | C57BL/6 mice, female, 4 to 6 weeks old; immunosuppression model of murine oropharyngeal candidiasis | C. albicans | Preventive deferasirox treatment significantly reduced the fungal burden in tongue tissue | [137] |
| N-Acetylcysteine | C57/BL6 mice, female, 6 to 8 weeks old | C. gattii | N-Acetylcysteine + AMB 1 achieved higher survival than the control and reduced morbidity in murine-induced cryptococcosis; reduced fungal burden in lungs/brain and lower concentrations of proinflammatory cytokines in the lungs | [138] |
| Cisplatin | BALB/c mice, female, 4 to 6 weeks old | C. neoformans | Cisplatin significantly inhibited C. neoformans growth in a mouse model | [168] |
| Panobinostat | Galleria mellonella larvae | C. albicans | Panobinostat and FLU combination enhanced survival rate of G. mellonella | [143] |
| Pilocarpine hydrochloride | G. mellonella larvae | C. albicans | Pilocarpine hydrochloride protects G. mellonella larvae from C. albicans-induced mortality in a dose-dependent manner | [154] |
| Pitavastatin | Caenorhabditis elegans animal model | C. albicans | Pitavastatin–FLU combination reduced the biofilm formation of Candida species and the fungal burdens in a C. elegans infection model | [77] |
| Drug, Compound Libraries | Sources | Fungi Tested | References |
|---|---|---|---|
| MDL Drug Data Report (MDDR), DrugBank, TargetMol databases or library (L4200) | BIOVIA and Thomson Reuters https://www.drugbank.ca/, http://targetmol.com/ | C. albicans, Paracoccidioides species | [20,167] |
| Prestwick Chemical Library | Prestwick Chemical (Illkirch, France) http://www.prestwickchemical.com/ | Candida species, Cryptococcus species, Saccharomyces cerevisiae, Aspergillus fumigatus, Fusarium oxysporum, Fusarium solani, Lichtheimia species, Lomentospora prolificans, Paecilomyces variotii, Rhizopus arrhizus, Scedosporium apiospermum | [23,25,66,75,83,94,96,124,165,228] |
| Pharmakon1600 drug library | MicroSource Discovery Systems (Gaylordsville, CT, USA) http://www.msdiscovery.com/ | C. albicans, C. glabrata C. auris | [68,77,162] |
| Library of Pharmacologically Active Compounds (LOPAC1280) | Sigma-Aldrich (St. Louis, MO, USA) https://www.sigmaaldrich.com/ | Exserohilum rostratum | [101] |
| Medicines for Malaria Venture (MMV) Malaria Box | Medicines for Malaria Venture (Geneva, Switzerland) https://www.mmv.org/ [229] | C. albicans, C. gatti, C. neoformans, L. prolificans, Fonsecaea pedrosoi, Fonsecaea monophora, Fonsecaea nubica, Cladophialophora carrionii, Phialophora verrucosa, Rhinocladiela similis, Exophiala jeanselmei var. heteromorpha, Exophiala dermatitidis | [91,148] |
| Screen-Well Enzo library of 640 compounds | Enzo Life Sciences (Farmingdale, NY, USA) https://www.enzolifesciences.com/ | Aspergillus species, Candida, Cryptococcus deuterogattii, Saccharomyces | [110] |
| L1300 Selleck library, 1018 United States Food and Drug Administration (FDA)-approved Selleck library | Selleck Chemicals https://www.selleckchem.com/ | A. fumigatus, C. gattii, C. glabrata, C. neoformans, Trichophyton rubrum | [73,100] |
| Pathogen Box® chemical library | Medicines for Malaria Venture (Geneva, Switzerland) https://www.mmv.org/ | C. auris | [119] |
| United States National Institutes of Health/National Cancer Institute (NIH/NCI) compound library | Developmental Therapeutics Program of the NIH/NCI (Rockville, MD, USA) https://dtp.cancer.gov/ | A. fumigatus, Candida species, C. neoformans | [127] |
| 1547 or 1581 FDA-approved drug library | Johns Hopkins, USA Johns Hopkins Clinical Compound Library (JHCCL) version 1.0 | C. albicans, C. auris, C. krusei, C. parapsilosis, C. tropicalis | [133,163] |
| 678 Maybridge collection | Thermo Fisher Scientific (Waltham, MA, USA) https://www.thermofisher.com/us/en/home/chemicals/maybridge.html | C. albicans, C. auris | [169] |
| Repurposed Drugs/Compounds Co-Applied. | Conventional Antifungal Drugs with Synergism |
|---|---|
| N-Acetylcysteine, alexidine dihydrochloride, amiodarone, arachidonic acid, aspirin, beauvericin, bis-biguanide alexidine dihydrochloride, benzocyclane, bromperidol derivative, chenodiol, chlorcyclizine, clomiphene, cloperastine, colistin, l-cycloserine, deferasirox, drospirenon, ebselen, erythromycin, glimepiride, ibuprofen, idoxifene, lovastatin, methylene-idoxifene, miltefosine nisoldipine, nortriptyline, panobinostat, perhexiline, pitavastatin, polymyxin b, promazine, pyrvinium pamoate, quinacrine, quinine, ribavirin, riluzole, sertraline, suloctidil, tamoxifen, thioridazine, thiosemicarbazone, toremifene, trifluoperazine | Azoles (clotrimazole, fluconazole, isavuconazole, itraconazole, ketoconazole, miconazole, posaconazole, voriconazole), echinocandins (caspofungin, anidulafungin, micafungin), polyene (AMB), allylamine (terbinafine) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, J.H.; Cheng, L.W.; Chan, K.L.; Tam, C.C.; Mahoney, N.; Friedman, M.; Shilman, M.M.; Land, K.M. Antifungal Drug Repurposing. Antibiotics 2020, 9, 812. https://doi.org/10.3390/antibiotics9110812
Kim JH, Cheng LW, Chan KL, Tam CC, Mahoney N, Friedman M, Shilman MM, Land KM. Antifungal Drug Repurposing. Antibiotics. 2020; 9(11):812. https://doi.org/10.3390/antibiotics9110812
Chicago/Turabian StyleKim, Jong H., Luisa W. Cheng, Kathleen L. Chan, Christina C. Tam, Noreen Mahoney, Mendel Friedman, Mikhail Martchenko Shilman, and Kirkwood M. Land. 2020. "Antifungal Drug Repurposing" Antibiotics 9, no. 11: 812. https://doi.org/10.3390/antibiotics9110812
APA StyleKim, J. H., Cheng, L. W., Chan, K. L., Tam, C. C., Mahoney, N., Friedman, M., Shilman, M. M., & Land, K. M. (2020). Antifungal Drug Repurposing. Antibiotics, 9(11), 812. https://doi.org/10.3390/antibiotics9110812








